The Effect of Biotic Stress in Plant Species Induced by ‘Candidatus Phytoplasma solani’—An Artificial Neural Network Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Phylogenetic Position of the Tested Serbian ‘Ca. Phytoplasma solani’ Strains
2.3. Biochemical Analyses of Peony, Mint, Dill, and Carrot Infected by ‘Ca. Phytoplasma solani’
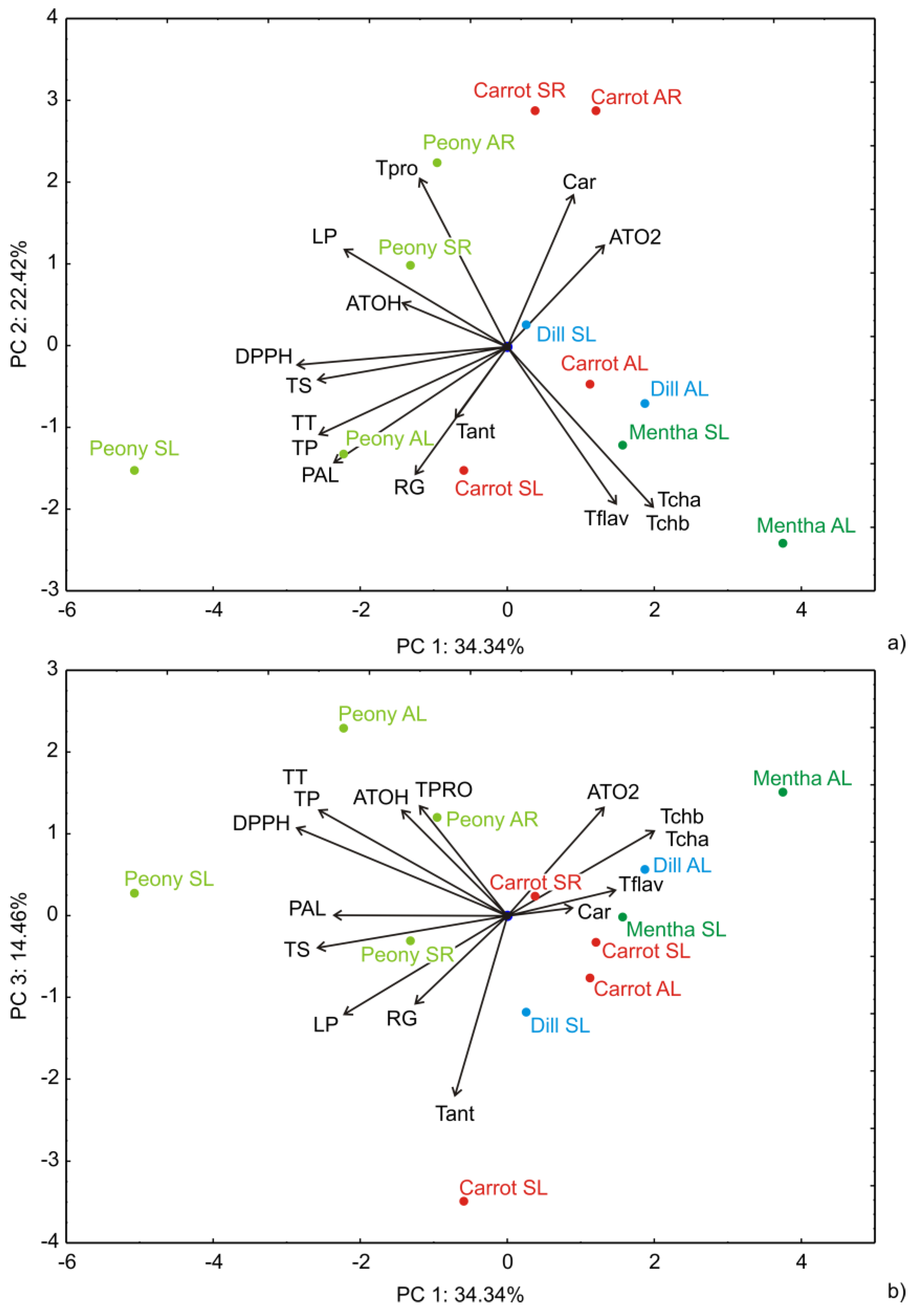
2.4. Principal Component Analysis (PCA)
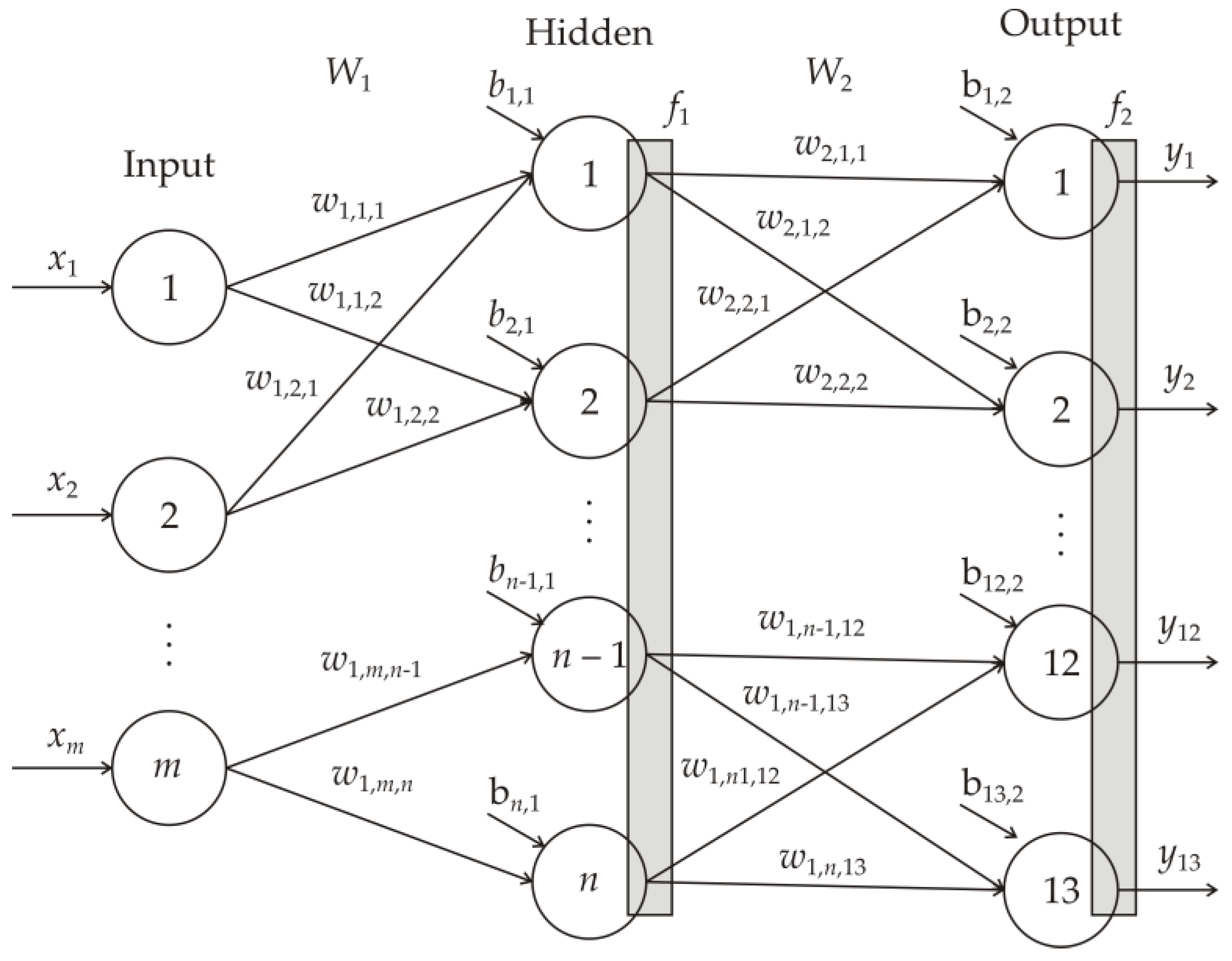
2.5. Artificial Neural Network (ANN) Modeling
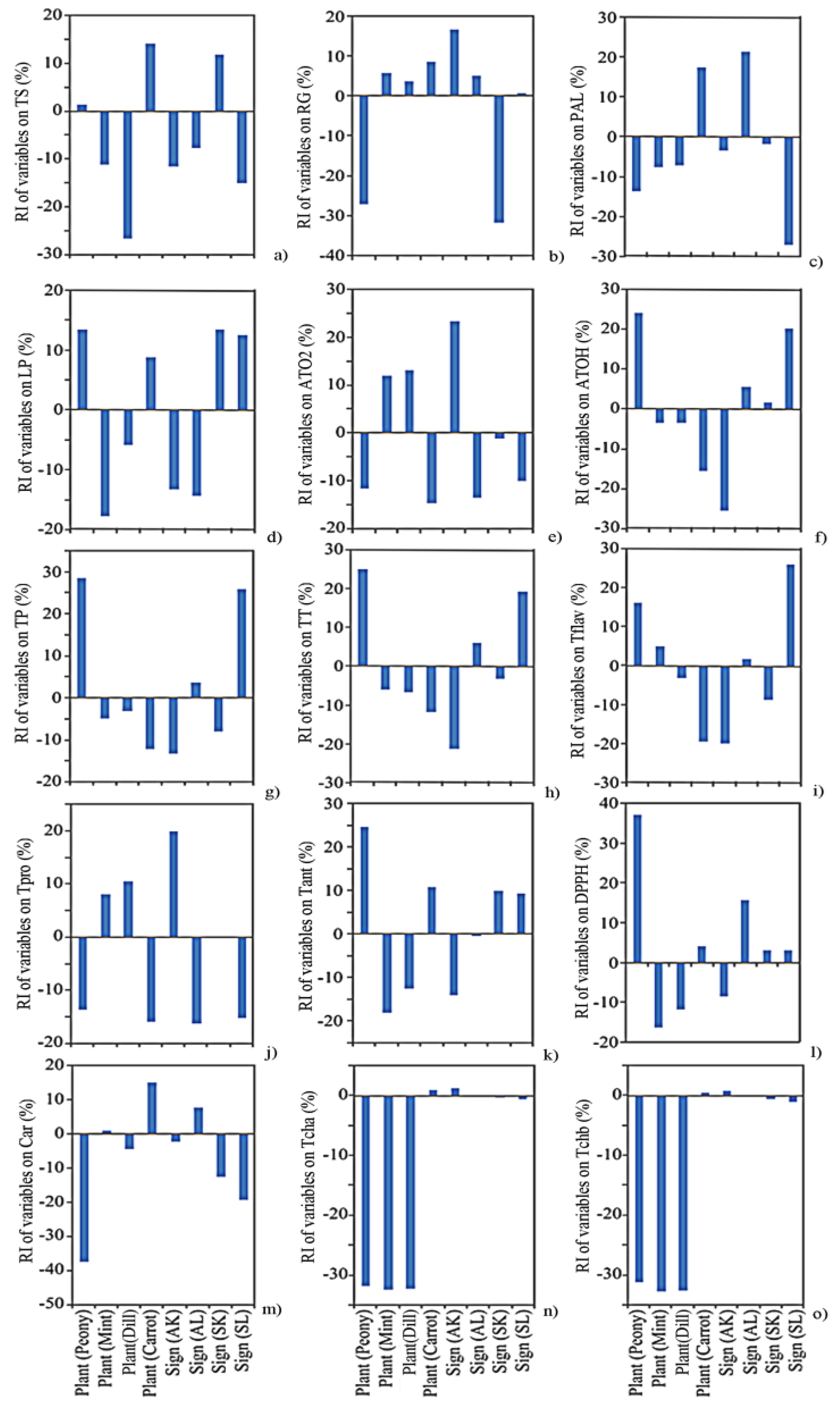
2.6. Global Sensitivity Analysis
3. Results
3.1. Phylogenetic Position of ‘Ca. Phytoplasma solani’ Strains
3.2. Biochemical Analyses of Peony, Mint, Dill, and Carrot Infected by ‘Ca. Phytoplasma solani’
3.3. Principal Component Analysis (PCA)
3.4. Neurons in the ANN Hidden Layer
3.5. Sensitivity Analysis
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ghosh, S.; Dey, G. Biotic and abiotic stress tolerance through CRISPR-Cas mediated genome editing. J. Plant Biochem. Biotechnol. 2022, 31, 227–238. [Google Scholar] [CrossRef]
- Prasad, A.; Senthil-Kumar, M.; Prasad, M. Complex molecular mechanisms determine fitness of plants to biotic and abiotic stresses. J. Plant Biochem. Biotechnol. 2021, 30, 633–635. [Google Scholar] [CrossRef]
- Ben Rejeb, I.; Pastor, V.; Mauch-Mani, B. Plant responses to simultaneous biotic and abiotic stress: Molecular mechanisms. Plants 2014, 3, 458–475. [Google Scholar] [CrossRef] [PubMed]
- Ayvacı, H.; Güldür, M.E.; Dikilitas, M. Physiological and Biochemical Changes in Lucerne (Medicago sativa) Plants Infected with ‘Candidatus Phytoplasma australasia’-Related Strain (16SrII-D Subgroup). Plant Pathol. J. 2022, 38, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Kiprovski, B.; Đalović, I.; Adamović, D.; Mitrović, P.; Marjanović-Jeromela, A.; Malenčić, Đ.; Popović, T. Biochemical changes in Oenothera biennis plants infected by ‘Candidatus Phytoplasma solani’. J. Plant Pathol. 2018, 100, 209–214. [Google Scholar] [CrossRef]
- Kumari, S.; Nagendran, K.; Rai, A.B.; Singh, B.; Rao, G.P.; Bertaccini, A. Global status of phytoplasma diseases in vegetable crops. Front. Microbiol. 2019, 10, 1349. [Google Scholar] [CrossRef] [PubMed]
- Mitrovic, P.; Djalovic, I.; Kiprovski, B.; Veljović Jovanović, S.; Trkulja, V.; Jelušić, A.; Popović, T. Oxidative stress and antioxidative activity in leaves and roots of carrot plants induced by Candidatus Phytoplasma solani. Plants 2021, 10, 337. [Google Scholar] [CrossRef]
- Popović, T.; Mitrović, P.; Kosovac, A. Molecular characterization of Candidatus Phytoplasma solani in celery: Case study in Futog. Field Veg. Crop. Res. 2021, 58, 66–71. [Google Scholar] [CrossRef]
- Mitrović, M.; Marinković, S.; Cvrković, T.; Jović, J.; Krstić, O.; Jakovljević, M. Framework for risk assessment of ‘Candidatus Phytoplasma solani’associated diseases outbreaks in agroecosystems in Serbia. J. Plant Pathol. 2022, 104, 537–552. [Google Scholar] [CrossRef]
- Zafari, S.; Niknam, V.; Musetti, R.; Noorbakhsh, S.N. Effect of phytoplasma infection on metabolite content and antioxidant enzyme activity in lime (Citrus aurantifolia). Acta Physiol. Plant. 2012, 34, 561–568. [Google Scholar] [CrossRef]
- Hameed, S.; Akhtar, K.P.; Hameed, A.; Gulzar, T.; Kiran, S.; Yousaf, S.; Abbas, G.; Asghar, M.J.; Sarwar, N. Biochemical changes in the leaves of mungbean (Vigna radiata) plants infected by phytoplasma. Turk. J. Biochem. 2017, 42, 591–599. [Google Scholar] [CrossRef]
- Junqueira, A.; Bedendo, I.; Pascholati, S. Biochemical changes in corn plants infected by the maize bushy stunt phytoplasma. Physiol. Mol. Plant Pathol. 2004, 65, 181–185. [Google Scholar] [CrossRef]
- Trkulja, V.; Adamović, D.; Đalović, I.; Mitrović, P.; Kovačić-Jošić, D.; Lukač, Z.; Komić, J. First report of stolbur phytoplasma associated with Anethum graveolens in Serbia. Plant Dis. 2016, 100, 516. [Google Scholar] [CrossRef]
- Adamovic, D.; Djalovic, I.; Mitrovic, P.; Kojic, S.; Pivic, R.; Josic, D. First Report on Natural Infection of Paeonia tenuifolia by ‘Candidatus Phytoplasma solani’in Serbia. Plant Dis. 2014, 98, 565. [Google Scholar] [CrossRef]
- Mitrović, P.; Trkulja, V.; Adamović, D.; Đalović, I.; Milovac, Ž.; Kovačić-Jošić, D.; Mihić Salapura, J. First report of stolbur phytoplasma on Mentha× piperita in Serbia. Plant Dis. 2016, 100, 853. [Google Scholar] [CrossRef]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef]
- Albalasmeh, A.A.; Berhe, A.A.; Ghezzehei, T.A. A new method for rapid determination of carbohydrate and total carbon concentrations using UV spectrophotometry. Carbohydr. Polym. 2013, 97, 253–261. [Google Scholar] [CrossRef]
- Rahman, I.; Kode, A.; Biswas, S.K. Assay for quantitative determination of glutathione and glutathione disulfide levels using enzymatic recycling method. Nat. Protoc. 2006, 1, 3159–3165. [Google Scholar] [CrossRef]
- Hodges, D.M.; DeLong, J.M.; Forney, C.F.; Prange, R.K. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 1999, 207, 604–611. [Google Scholar] [CrossRef]
- Gerasimova, N.G.; Pridvorova, S.M.; Ozeretskovskaya, O.L. Role of L–phenylalanine ammonia Lyase in the induced resistance and susceptibility of sotato plants. Appl. Biochem. Microbiol. 2005, 41, 103–105. [Google Scholar] [CrossRef]
- Von Wettstein, D. Chlorophyll-letale und der submikroskopische Formwechsel der plastiden. Exp. Cell Res. 1957, 12, 427–506. [Google Scholar] [CrossRef] [PubMed]
- Makkar, H.P.S. Quantification of Tannins in Tree and Shrub Foliage: A Laboratory Manual; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2003. [Google Scholar]
- Pękal, A.; Pyrzynska, K. Evaluation of aluminium complexation reaction for flavonoid content assay. Food Anal. Methods 2014, 7, 1776–1782. [Google Scholar] [CrossRef]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.K. Assay guided comparison for enzymatic and nonenzymatic antioxidant activities with special reference to medicinal plants. In Antioxidant Enzyme, 3rd ed.; El-Missiry, M.A., Ed.; IntechOpen: Rijeka, Croatia, 2012; Volume 14, pp. 382–400. [Google Scholar]
- Ahmed, D.; Saman, Z.; Hira, B. In vitro analysis of antioxidant activities of Oxalis corniculata Linn. fractions in various solvents. Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 158–165. [Google Scholar] [CrossRef][Green Version]
- Sánchez-Moreno, C. Methods used to evaluate the free radical scavenging activity in foods and biological systems. Int. J. Food Sci. Technol. 2002, 8, 121–137. [Google Scholar] [CrossRef]
- Brlek, T.; Pezo, L.; Voća, N.; Krička, T.; Vukmirović, Đ.; Čolović, R.; Bodroža-Solarov, M. Chemometric approach for assessing the quality of olive cake pellets. Fuel Process. Technol. 2013, 116, 250–256. [Google Scholar] [CrossRef]
- Abdi, H.; Williams, L.J. Principal component analysis. Wiley Interdiscip. Rev. Comput. Stat. 2010, 2, 433–459. [Google Scholar] [CrossRef]
- Hu, X.; Weng, Q. Estimating impervious surfaces from medium spatial resolution imagery using the self-organizing map and multi-layer perceptron neural networks. Remote Sens. Environ. 2009, 113, 2089–2102. [Google Scholar] [CrossRef]
- Karlović, S.; Bosiljkov, T.; Brnčić, M.; Jezek, D.; Tripalo, B.; Dujmić, F.; Dzineva, I.; Skupnjak, A. Comparison of artificial neural network and mathematical models for drying of apple slices pretreated with high intensity ultrasound. Bulg. J. Agric. Sci. 2013, 19, 1372–1377. [Google Scholar]
- Chattopadhyaya, P.B.; Rangarajana, R. Application of ANN in sketching spatial nonli-nearity of unconfined aquifer in agricultural basin. Agric. Water Manag. 2014, 133, 81–91. [Google Scholar] [CrossRef]
- Yoon, Y.; Swales, G., Jr.; Margavio, T.M. A Comparison of Discriminant Analysis versus Artificial Neural Networks. J. Oper. Res. Soc. 1993, 44, 51–60. [Google Scholar] [CrossRef]
- Quaglino, F.; Zhao, Y.; Casati, P.; Bulgari, D.; Bianco, P.A.; Wei, W.; Davis, R.E. ‘Candidatus Phytoplasma solani’, a novel taxon associated with stolbur-and bois noir-related diseases of plants. Int. J. Syst. Evol. Microbiol. 2013, 63, 2879–2894. [Google Scholar] [CrossRef] [PubMed]
- Mitrović, M.; Jakovljević, M.; Jović, J.; Krstić, O.; Kosovac, A.; Trivellone, V.; Jermini, M.; Toševski, I.; Cvrković, T. ‘Candidatus phytoplasma solani’genotypes associated with potato stolbur in Serbia and the role of Hyalesthes obsoletus and Reptalus panzeri (hemiptera, cixiidae) as natural vectors. Eur. J. Plant Pathol. 2016, 144, 619–630. [Google Scholar] [CrossRef]
- Martinović, M.; Bjegović, P. O nekim bolestima i štetočinama utvrđenim u NR Srbiji u 1949 godini. Zaštita Bilja 1950, 2, 59–68. (In Serbian) [Google Scholar]
- Duduk, B.; Botti, S.; Ivanović, M.; Krstić, B.; Dukić, N.; Bertaccini, A. Identification of phytoplasmas associated with grapevine yellows in Serbia. J. Phytopathol. 2004, 152, 575–579. [Google Scholar] [CrossRef]
- Duduk, B.; Bertaccini, A. Corn with symptoms of reddening: New host of stolbur phytoplasma. Plant Dis. 2006, 90, 1313–1319. [Google Scholar] [CrossRef] [PubMed]
- Jović, J.; Ember, I.; Mitrović, M.; Cvrković, T.; Krstić, O.; Krnjajić, S.; Acs, Z.; Kolber, M.; Toševski, I. Molecular detection of potato stolbur phyto-plasma in Serbia. Bull. Insectology 2011, 64, S83–S84. [Google Scholar]
- Mitrović, J.; Pavlović, S.; Duduk, B. Survey and Multigene Characterization of Stolbur Phytoplasmas on Various Plant Species in Serbia. Phytopathol. Mediterr. 2013, 52, 8. [Google Scholar] [CrossRef]
- Martini, M.; Quaglino, F.; Bertaccini, A. Multilocus Genetic Characterization of Phytoplasmas. In Phytoplasmas: Plant Pathogenic Bacteria-III: Genomics, Host Pathogen Interactions and Diagnosis; Springer: Singapore, 2019; pp. 161–200. [Google Scholar]
- Böhm, J.W.; Duckeck, D.; Duduk, B.; Schneider, B.; Kube, M. Genome Comparison of ‘Candidatus Phytoplasma rubi’with Genomes of Other 16SrV Phytoplasmas Highlights Special Group Features. Appl. Microbiol. 2023, 3, 1083–1100. [Google Scholar] [CrossRef]
- Contaldo, N.; Stepanović, J.; Pacini, F.; Bertaccini, A.; Duduk, B. Molecular Variability and Host Distribution of ‘Candidatus Phytoplasma Solani’ Strains from Different Geographic Origins. Microorganisms 2021, 9, 2530. [Google Scholar] [CrossRef]

 and D. carota
and D. carota  and 23 comparative Phytoplasma spp. strains belonging to different 16Sr Groups. Acholeplasma laidlawii strain NCTC10116 from the GenBank database served as an outgroup.
and 23 comparative Phytoplasma spp. strains belonging to different 16Sr Groups. Acholeplasma laidlawii strain NCTC10116 from the GenBank database served as an outgroup.
 and D. carota
and D. carota  and 23 comparative Phytoplasma spp. strains belonging to different 16Sr Groups. Acholeplasma laidlawii strain NCTC10116 from the GenBank database served as an outgroup.
and 23 comparative Phytoplasma spp. strains belonging to different 16Sr Groups. Acholeplasma laidlawii strain NCTC10116 from the GenBank database served as an outgroup.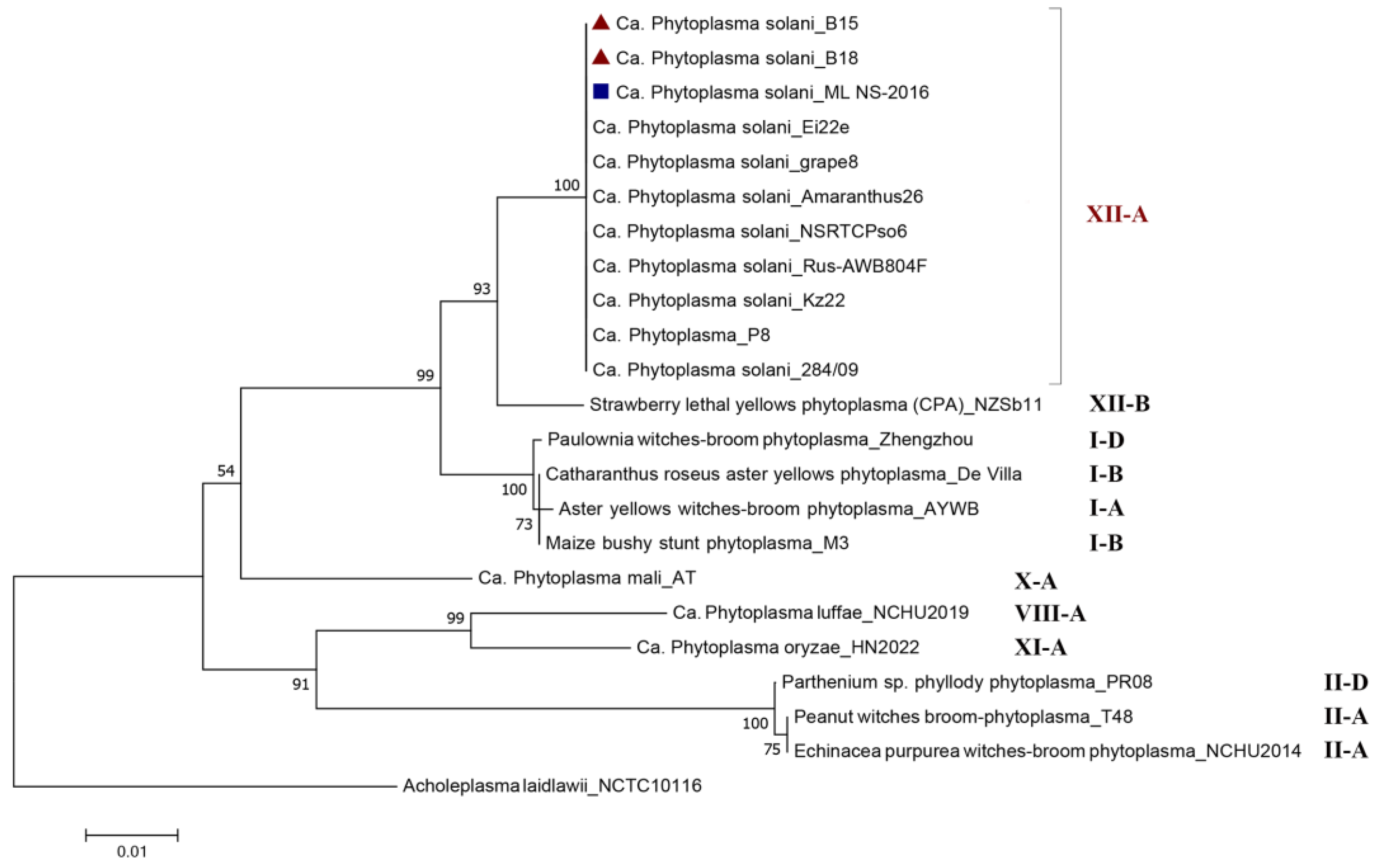
 and Mentha × piperita
and Mentha × piperita  and 13 comparative Phytoplasma spp. strains according to sequences of the plsC gene. Acholeplasma laidlawii strain DSM23060 from the GenBank database served as an outgroup.
and 13 comparative Phytoplasma spp. strains according to sequences of the plsC gene. Acholeplasma laidlawii strain DSM23060 from the GenBank database served as an outgroup.
 and Mentha × piperita
and Mentha × piperita  and 13 comparative Phytoplasma spp. strains according to sequences of the plsC gene. Acholeplasma laidlawii strain DSM23060 from the GenBank database served as an outgroup.
and 13 comparative Phytoplasma spp. strains according to sequences of the plsC gene. Acholeplasma laidlawii strain DSM23060 from the GenBank database served as an outgroup.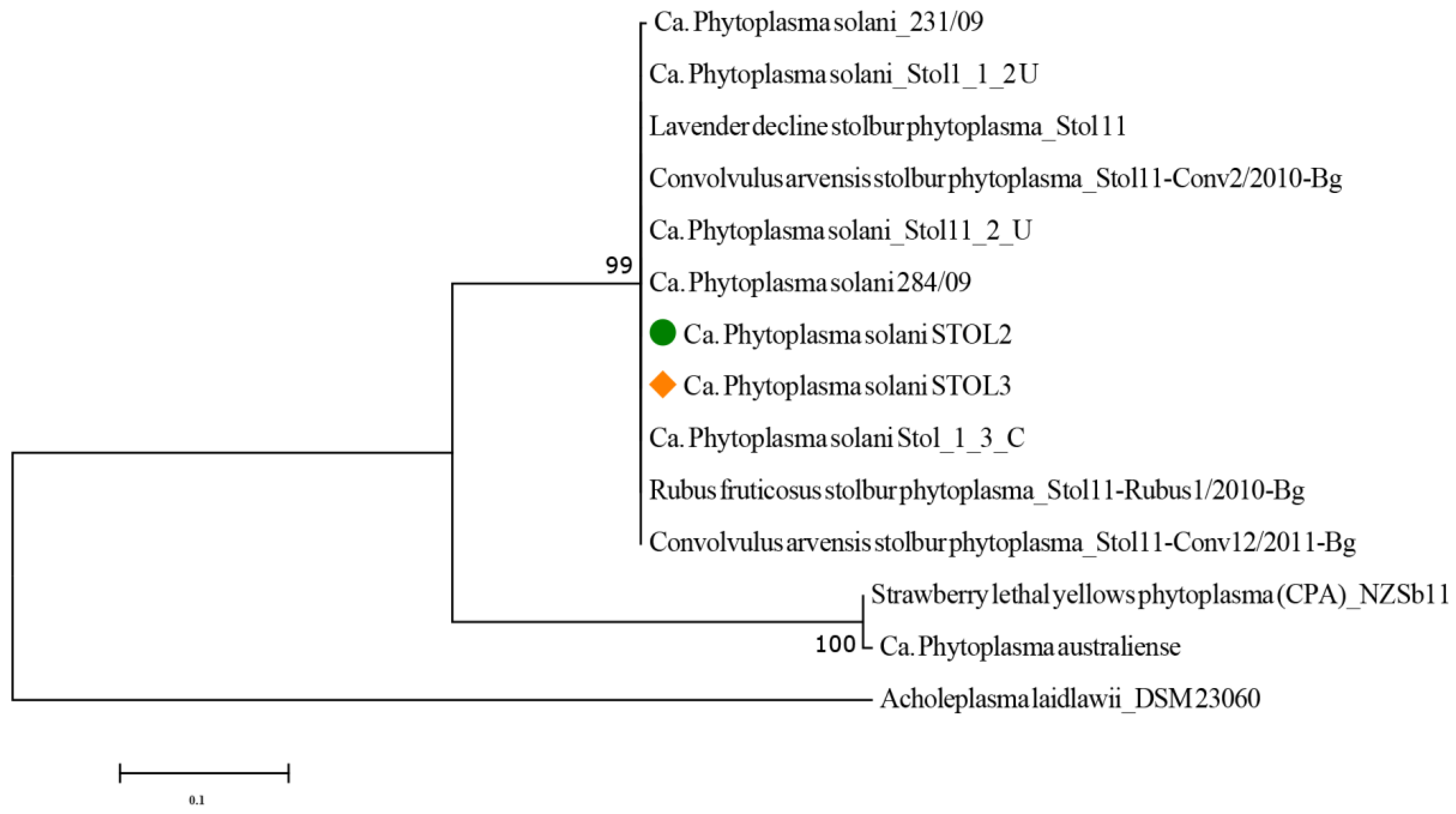
| Species | 16Sr Group Classification | Strain | Isolation Source | Country | Acc. No. |
|---|---|---|---|---|---|
| ‘Ca. Phytoplasma solani’ | XII-A | B15 a | P. tenuifolia | Serbia | KC960487 |
| B18 a | P. tenuifolia | Serbia | KF614623 | ||
| ML_NS-2016 a | D. carota | Serbia | MF503627 | ||
| Ei22 | Euscelis incisus | Serbia | MN047263 | ||
| grape8 | Vitis vinifera | Iran | MK392488 | ||
| Amaranthus26 | Amaranthus sp. | - | MN007088 | ||
| NSRTCPso6 | Prunus domestica | Jordan | MH085229 | ||
| Rus-AWB804F | Medicago sativa | Russia | KY587525 | ||
| Kz22 | Citrus sp. | Iran | MG563790 | ||
| P8 | Solanum tuberosum | Germany | PP261349 | ||
| 284/09 | Nicotiana tabacum | - | NC_022588 | ||
| Strawberry lethal yellows phytoplasma (CPA) | XII-B variant | NZSb11 | - | Australia and New Zealand | CP002548 |
| Aster yellows witches’-broom phytoplasma | I-A | AYWB | Lactuca sativa | USA | CP000061 |
| ‘Catharanthus roseus’ aster yellows phytoplasma | I-B | De Villa | Catharanthus roseus | South Africa | CP035949 |
| Maize bushy stunt phytoplasma | I-B | M3 | Dalbulus maidis | Brazil | CP015149 |
| Paulownia witches’-broom phytoplasma | I-D | Zhengzhou | Paulownia | China | CP066882 |
| Peanut witches’-broom phytoplasma | II-A | T48 | Areca catechu | China | OR239773 |
| ‘Echinacea purpurea’ witches’-broom phytoplasma | II-A | NCHU2014 | Catharanthus roseus | Taiwan | CP040925 |
| ‘Parthenium sp.’ phyllody phytoplasma | II-D | PR08 | Parthenium hysterophorus | India | CP097207 |
| ‘Ca. Phytoplasma ziziphi’ | V-B | Jwb-nky | Ziziphus jujuba | China | CP025121 |
| ‘Ca. Phytoplasma luffae’ | VIII-A | NCHU2019 | Luffa aegyptiaca | Taiwan | CP054393 |
| ‘Ca. Phytoplasma mali’ | X-A | AT | - | - | CU469464 |
| ‘Ca. Phytoplasma oryzae’ | XI-A | HN2022 | - | China | CP116038 |
| Acholeplasma laidlawii b | - | NCTC10116 | - | - | LS483439 |
| Species | Strain | Isolation Source | Country | Acc. No. |
|---|---|---|---|---|
| ‘Ca. Phytoplasma solani’ | STOL2 a | A. graveolens | Serbia | KT281866 |
| STOL3 a | Mentha × piperita | Serbia | KT281865 | |
| 284/09 | Nicotiana tabacum | - | FO393427 | |
| 231/09 | - | - | FO393428 | |
| Stol11_3_C | Convolvulus arvensis | - | JQ977746 | |
| Stol11_1_C | Convolvulus arvensis | - | JQ977744 | |
| Stol11_2_U | Urtica dioica | - | JQ977745 | |
| ‘Rubus fruticosus’ stolbur phytoplasma | Stol11-Rubus1/2010-Bg | Rubus fruticosus | Bulgaria | JN561701 |
| ‘Convolvulus arvensis’ stolbur phytoplasma | Stol11-Conv2/2010-Bg | Convolvulus arvensis | Bulgaria | JN561700 |
| ‘Convolvulus arvensis’ stolbur phytoplasma | Stol11-Conv12/2011-Bg | Convolvulus arvensis | Bulgaria | JN561699 |
| Lavender decline stolbur phytoplasma | Stol 11 | - | - | AF447596 |
| Strawberry lethal yellows phytoplasma (CPA) | NZSb11 | - | - | CP002548 |
| ’Ca. Phytoplasma australiense’ | - | - | - | AM422018 |
| Acholeplasma laidlawii b | DSM 23060 | NZ_QRDS01000001 |
| Carrot a (Daucus carota) | Mint (Mentha × piperita) | Dill (Anethum graveolens) | Peony (Paeonia tenuifolia) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| A | S | A | S | A | S | A | S | ||
| Total sugars (% fw) | ± Se | 5.79 ± 0.01 | 5.15 ± 0.01 | 3.45 ± 0.08 | 6.62 ± 0.01 | 3.44 ± 0.02 | 5.15 ± 0.01 | 4.80 ± 0.01 | 11.98 ± 0.01 |
| t-test A/S | * | * | * | * | |||||
| Reduced glutathione (μmol GSH/g fw) | ± Se | 2.75 ± 0.01 | 3.10 ± 0.01 | 3.28 ± 0.01 | 3.93 ± 0.01 | 4.21 ± 0.01 | 5.07 ± 0.02 | 4.55 ± 0.01 | 5.01 ± 0.01 |
| t-test A/S | * | * | * | * | |||||
| Phenylalanine ammonia-lyase (U/g fw) | ± Se | 258.82 ± 0.29 | 70.94 ± 0.03 | 164.33 ± 0.09 | 300.91 ± 0.06 | 247.63 ± 0.19 | 310.20 ± 0.42 | 413.51 ± 0.28 | 1450.21 ± 0.41 |
| t-test A/S | nd | * | * | * | |||||
| Lipid peroxidation (nmol MDA/g fw) | ± Se | 871.57 ± 0.26 | 1064.43 ± 3.80 | 590.77 ± 1.04 | 641.43 ± 0.30 | 597.37 ± 0.86 | 1476.27 ± 0.64 | 722.83 ± 0.12 | 1636.60 ± 1.70 |
| A/S | * | * | * | * | |||||
| Total polyphenols (mg/g dw) | ± Se | 4.03 ± 0.02 | 1.42 ± 0.01 | 4.10 ± 0.01 | 7.45 ± 0.06 | 5.17 ± 0.02 | 6.27 ± 0.01 | 94.02 ± 0.16 | 86.56 ± 0.23 |
| t-test A/S | * | * | * | * | |||||
| Total tannins (mg/g dw) | ± Se | 3.33 ± 0.04 | 1.18 ± 0.03 | 2.66 ± 0.02 | 5.83 ± 0.03 | 1.94 ± 0.01 | 2.34 ± 0.01 | 82.18 ± 0.02 | 77.35 ± 0.08 |
| t-test A/S | * | * | * | * | |||||
| Total flavonoids (mg/g dw) | ± Se | 0.043 ± 0.0 | 0.000 ± 0.0 | 0.263 ± 0.002 | 0.275 ± 0.002 | 0.032 ± 0.001 | 0.022 ± 0.001 | 0.050 ± 0.000 | 0.051 ± 0.001 |
| t-test A/S | nd | * | * | * | |||||
| Total proanthocyanidins (mg/g dw) | ± Se | 0.40 ± 0.0 | 6.02 ± 0.01 | 0.87 ± 0.01 | 0.79 ± 0.01 | 0.86 ± 0.01 | 2.20 ± 0.01 | 4.75 ± 0.01 | 3.61 ± 0.01 |
| t-test A/S | * | * | * | * | |||||
| Total anthocyanidins (mg/g dw) | ± Se | 0.003 ± 0.0 | 0.000 ± 0.0 | 0.003 ± 0.00 | 0.000 ± 0.00 | 0.009 ± 0.01 | 0.012 ± 0.00 | 0.003 ± 0.00 | 0.137 ± 0.00 |
| t-test A/S | nd | * | nd | * | |||||
| Total chlorophyll a (mg/g dw) | ± Se | 0.78 ± 0.01 | 0.54 ± 0.01 | 1.86 ± 0.01 | 0.46 ± 0.00 | 0.93 ± 0.01 | 0.27 ± 0.00 | 0.61 ± 0.01 | 0.12 ± 0.01 |
| t-test A/S | * | * | * | * | |||||
| Total chlorophyll b (mg/g dw) | ± Se | 0.18 ± 0.0 | 0.15 ± 0.0 | 0.77 ± 0.00 | 0.21 ± 0.01 | 0.42 ± 0.01 | 0.14 ± 0.00 | 0.24 ± 0.00 | 0.05 ± 0.00 |
| t-test A/S | * | * | * | * | |||||
| Carotenoids (mg/g dw) | ± Se | 0.36 ± 0.01 | 0.17 ± 0.0 | 0.39 ± 0.00 | 0.12 ± 0.00 | 0.35 ± 0.01 | 0.08 ± 0.00 | 0.29 ± 0.00 | 0.12 ± 0.00 |
| t-test A/S | * | * | * | * | |||||
| NBT test (% neutralized radicals) | ± Se | 20.33 ± 0.28 | 82.57 ± 0.23 | 93.53 ± 0.29 | 88.37 ± 0.09 | 82.10 ± 0.06 | 88.40 ± 0.06 | 44.30 ± 0.35 | 52.60 ± 0.23 |
| t-test A/S | * | * | * | nd | |||||
| •OH test (% neutralized radicals) | ± Se | 10.81 ± 2.42 | 0.69 ± 0.04 | 16.40 ± 0.15 | 15.17 ± 1.94 | 35.37 ± 1.28 | 4.70 ± 0.12 | 36.30 ± 0.46 | 30.90 ± 0.06 |
| t-test A/S | * | nd | * | * | |||||
| DPPH test (% neutralized radicals) | ± Se | 19.95 ± 0.02 | 10.90 ± 0.01 | 14.30 ± 0.01 | 15.43 ± 0.12 | 13.74 ± 0.08 | 22.80 ± 0.20 | 92.91 ± 0.06 | 90.87 ± 0.02 |
| t-test A/S | * | * | * | * | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Djalovic, I.; Mitrovic, P.; Trivan, G.; Jelušić, A.; Pezo, L.; Janić Hajnal, E.; Popović Milovanović, T. The Effect of Biotic Stress in Plant Species Induced by ‘Candidatus Phytoplasma solani’—An Artificial Neural Network Approach. Horticulturae 2024, 10, 426. https://doi.org/10.3390/horticulturae10050426
Djalovic I, Mitrovic P, Trivan G, Jelušić A, Pezo L, Janić Hajnal E, Popović Milovanović T. The Effect of Biotic Stress in Plant Species Induced by ‘Candidatus Phytoplasma solani’—An Artificial Neural Network Approach. Horticulturae. 2024; 10(5):426. https://doi.org/10.3390/horticulturae10050426
Chicago/Turabian StyleDjalovic, Ivica, Petar Mitrovic, Goran Trivan, Aleksandra Jelušić, Lato Pezo, Elizabet Janić Hajnal, and Tatjana Popović Milovanović. 2024. "The Effect of Biotic Stress in Plant Species Induced by ‘Candidatus Phytoplasma solani’—An Artificial Neural Network Approach" Horticulturae 10, no. 5: 426. https://doi.org/10.3390/horticulturae10050426
APA StyleDjalovic, I., Mitrovic, P., Trivan, G., Jelušić, A., Pezo, L., Janić Hajnal, E., & Popović Milovanović, T. (2024). The Effect of Biotic Stress in Plant Species Induced by ‘Candidatus Phytoplasma solani’—An Artificial Neural Network Approach. Horticulturae, 10(5), 426. https://doi.org/10.3390/horticulturae10050426






