Variations in Essential Oil Composition and Chemotype Patterns of Wild Thyme (Thymus) Species in the Natural Habitats of Hungary
Abstract
1. Introduction
- Subsect. Isolepides: Thymus glabrescens Willd.—common thyme; Thymus pannonicus All.—Pannonian/Large/Hungarian thyme.
- Subsect. Alternanthes: Thymus pulegioides L.-broad-leaved/mountain thyme.
- Subsect. Pseudomarginati: Thymus praecox Opiz—creeping thyme.
2. Materials and Methods
3. Results
3.1. Frequency of Occurrence of Thymus Species
- The occurrence of native Thymus populations was various, depending on their ecological tolerance, habitat preferences, and social behavior types. Among 74 populations of 5 species surveyed in the Hungarian Mountain Range, T. pannonicus (TPA) was found with the highest frequency (38 populations, 51.35%), followed by T. glabrescens (TGL) (17 populations, 22.98%) (Table 1 and Table 2). Both species were explored at new sites in the last few years, which also verifies their broad ecological tolerance and generalist character (Table 1 and Table 2).
- Populations of T. pannonicus were found on diverse parent rocks, mainly on limestone (e.g., Buda Hills, Tapolca Basin), dolomite (e.g., Bakony Hills, Vértes Hills), and loess (e.g., Pilis Hills, Balaton Uplands), while they were less frequent on silicate rocks such as andesite (Visegrád Hills) or rhyolite tuff (e.g., Bükk Hills), but they were also found on the rare marble substrate in the Cserehát (Table 1).
- T. glabrescens samples were also collected from sites with limestone base rocks (e.g., Buda Hills), dolomite (e.g., Keszthely Hills), or sandy loess (Pilis Hills); however, this species survived in rather extreme conditions, provided by thin bare soil layers developed on basalt, granite, or sandstone rocks (Table 1).
- T. praecox (TPR) and T. pulegioides (TPU) have special ecological preferences: the existence of TPR (specialist, eight populations, 10.81%) populations is connected to soils on calciferous base rocks (dolomite, limestone), while TPU (nine populations, 12.16%) prefers humid areas of mountain and lowland meadows (Table 1 and Table 2).
- Where the habitat conditions were favorable, the coexistence of 1–2 thyme species was also observed in the combination of TPA/TGL/TPR (dry conditions, calciferous rocks, and grassland communities) or of TPA/TPU (humid conditions and meadows). In our studies, the most important plant communities involving wild thymes were grasslands on sand, loess, silicate stones, dolomite, and limestones (Table 1).
3.2. Essential Oil Levels in Thymus Populations
3.2.1. Essential Oil Production of Thymus Species in Different Habitats
3.2.2. The Role of the Genetic Factor in the Essential Oil Accumulating Ability
3.2.3. The Role of the Environmental Factors on the Essential Oil Accumulating Ability
3.3. Essential Oil Chemotypes in Native Thymus populations
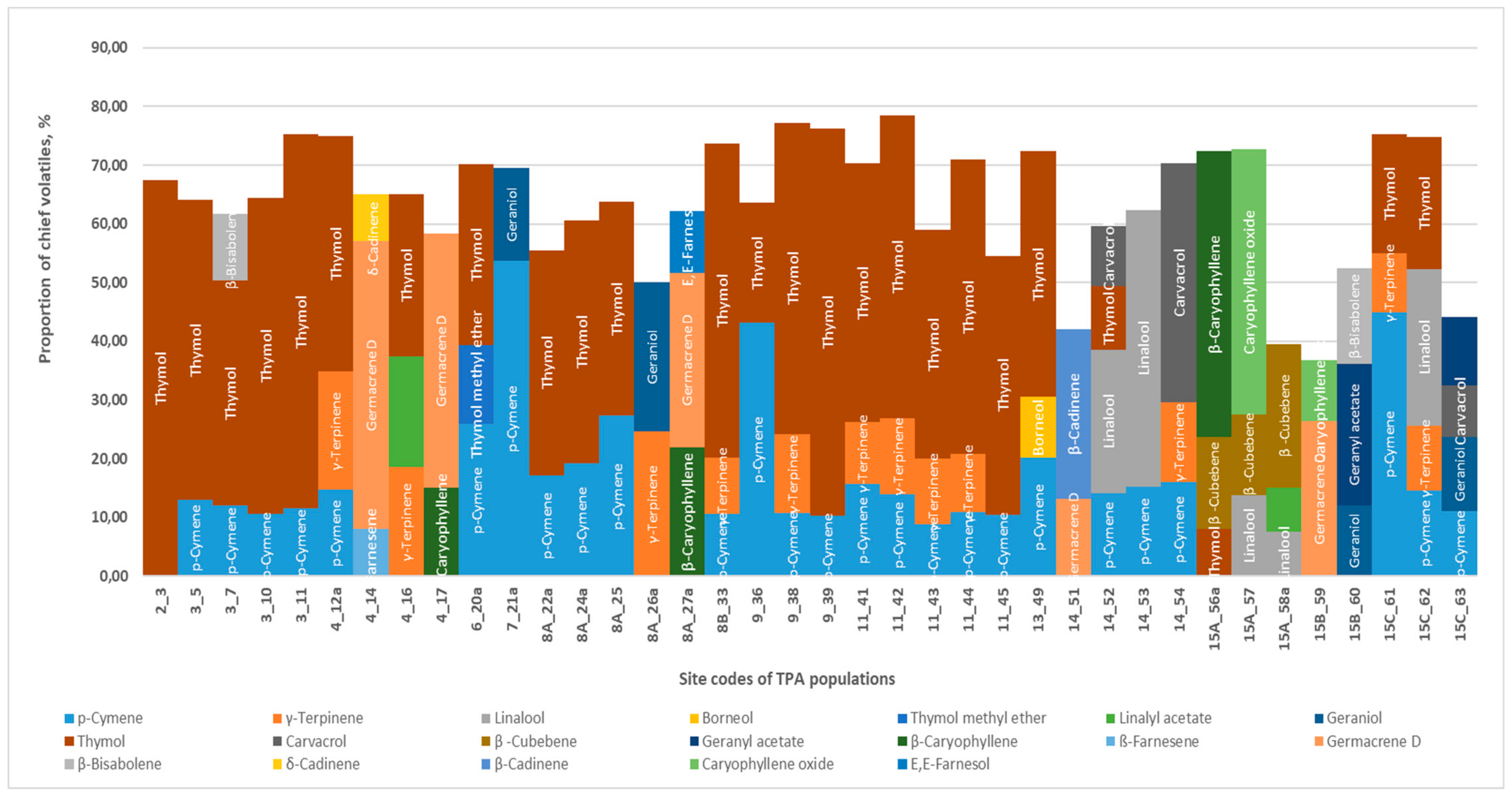
3.3.1. Thymus pannonicus
- Thymol chemotype (+ γ-terpinene + p-cymene) (18 sites)—limestone, loess, sand, andesite, and dolomite.
- Thymol + γ-terpinene+linalyl acetate (Bakony Hills, Öskü)—dolomite.
- Thymol + p-cymene +isoborneol (Mátra Hills, Sirok)—rhyolite tuff.
- Carvacrol + γ-terpinene + p-cymene (Bükk Hills, Mónosbél)—mudstone.
- Geraniol + geranyl acetate + p-cymene + carvacrol (Cserehát Hills, Sajógalgóc)—mudstone.
- Geraniol + p-cymene (Gerecse Hills, Tardosbánya)—limestone.
- Geraniol + γ-terpinene (Buda Hills, Homok Hill)—dolomite.
- Linalool + p-cymene + thymol + carvacrol (Bükk Hills, Noszvaj)—mudstone.
- 9.
- Thymol +p-cymene + β-bisabolene (Balaton Uplands, Tapolca)—limestone.
- 10.
- Thymol + β-caryophyllene + β-cubebene (Zemplén Hills, Regéc hayfield)—rhyolite.
- 11.
- Geraniol + geranyl acetate + β-bisabolene (Aggtelek Hills, Jósvafő)—limestone.
- 12.
- Linalool + linalyl acetate + β-cubebene (Zemplén Hills, Vágáshuta)—andesite.
- 13.
- Linalool + caryophyllene oxide + β-cubebene (Zemplén Hills, Bózsva)—rhyolite.
- 14.
- Germacrene D + β-caryophyllene (Bakony Hills, Litér)—dolomite.
- 15.
- Germacrene D + β-caryophyllene + farnesol (Buda Hills, Nagyszénás)—dolomite.
- 16.
- Germacrene D + caryophyllene oxide (Aggtelek Hills, Aggtelek)—limestone.
- 17.
- Germacrene D + β-farnesene + δ-cadinene (Bakony Hills, Szőc)—limestone
- 18.
- Germacrene D + β-cadinene (Bükk Hills, Cserépváralja)—rhyolite tuff.
3.3.2. Thymus glabrescens
- Thymol + γ-terpinene (Pilis Hills, Pilisszántó)—dolomite.
- Thymol + p-cymene (Bakony Hills, Csesznek)—limestone.
- Geraniol + p-cymene + linalyl acetate (Gerecse: Tardosbánya)—limestone.
- 4.
- Thymol + geraniol + germacrene D (Medves Hills, Salgó Hill)—basalt.
- 5.
- Thymol + germacrene D (Buda Hills: Kálvária Hill; Mátra Hills: Pásztó, Köves Cliff).
- 6.
- Thymol + germacrene D + β-caryophyllene (Balaton Uplands, Várvölgy; Szentbékkálla)—sand; sandstone.
- 7.
- Cis-β-Ocymene + germacrene D + β-caryophyllene (Bükk Hills, Szarvaskő)—mudstone.
- 8.
- Germacrene D + β-caryophyllene (Bakony Hills, Várpalota; Buda Hills, Érd)—dolomite, limestone.
- 9.
- Germacrene D + β-caryophyllene +bicyclogermacrene (Buda Hills, Nagykovácsi Dog Hill)—limestone.
- 10.
- Germacrene D+ β-caryophyllene + β-cadinene (Pilis Hills, Pilisszentiván, Fehér Hill)—dolomite.
- 11.
- Germacrene D + nerolidol + β -cadinene (Pilis Hills, Dorog, Strázsa Hill)—Ca-sand.
- 12.
- Germacrene D + τ-cadinol (Vértes Hills, Csákberény)—dolomite.
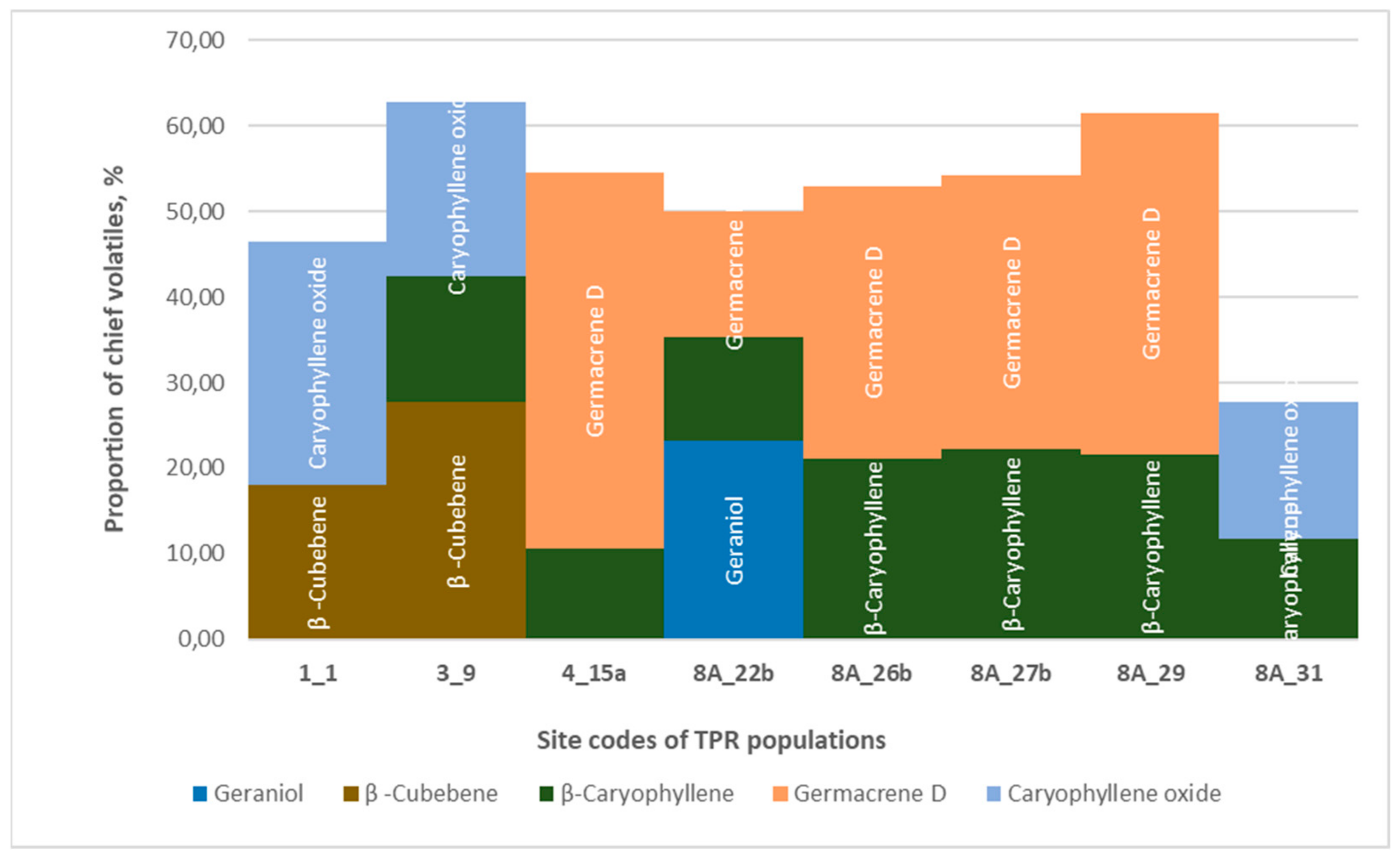
3.3.3. Thymus praecox
- Germacrene D + β-caryophyllene (Buda Hills: Sas Hill, Nagyszénás Hill, Homok Hill; Bakony Hills: Várpalota): dolomite.
- β-caryophyllene + caryophyllene oxide (Buda Hills: Tétény Hill, Diósd): Sarmathian limestone.
- β-cubebene + caryophyllene oxide (Mecsek Hills: Pécs, Kis-Tubes Hill)—limestone.
- β-cubebene + caryophyllene oxide + β-caryophyllene Balaton Uplands, Tamás Hill, Balatonfüred): dolomite.
- 5.
- Geraniol + germacrene D + β-caryophyllene (Buda Hills, Odvas Hill, Budaörs): dolomite.
3.3.4. Thymus pulegioides
- Carvacrol + thymol metylether + γ-terpinene (Balaton Uplands, Zalaszántó)—silt.
- 2.
- Geranial + linalyl acetate + neral + linalool (Mátra Hills, Mátrakeresztes)—andesite.
- 3.
- p-cymene + spathulenol +geraniol (Börzsöny Hills, Szent Mihály Hill, Zebegény)—andesite.
- 4.
- β-caryophyllene + thymol + germacrene D (Balaton Uplands, Szentbékkálla)—sandstone.
- 5.
- Germacrene D + β-caryophyllene + γ-muurulene (Visegrád Hills, Vadálló Cliffs, Dömös)—andesite.
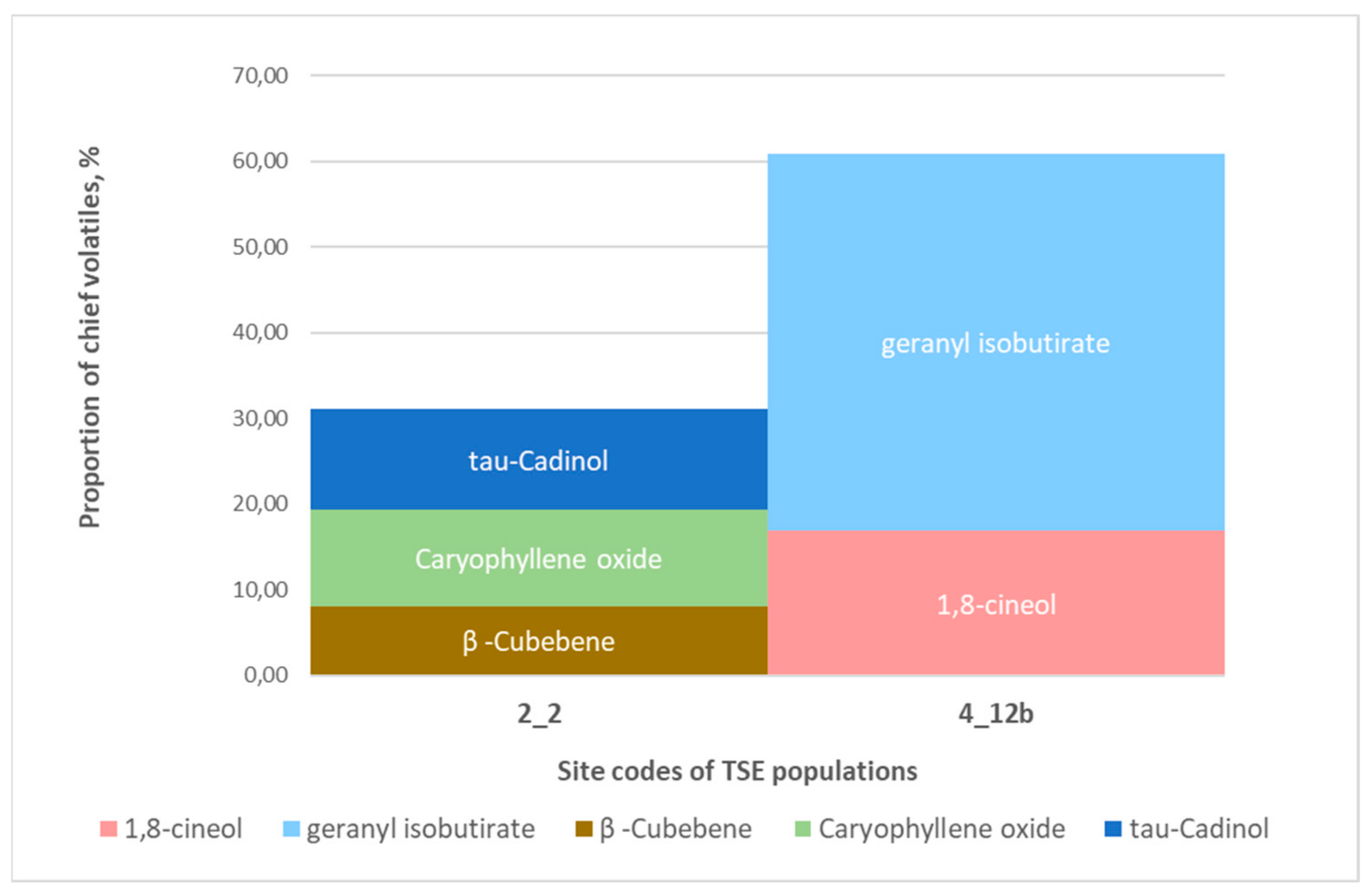
3.3.5. Thymus serpyllum
- Geraniol + geranyl isobutyrate (Bakony Hills, Fenyőfő)—basic sand.
- 2.
- τ-cadinol + caryophyllene oxide + β-cubebene (Somogy Hills, Nagybajom)—acidic sand.
Frequency of Chief Volatiles Detected in Thymus chemotypes
Classification of Thymus chemotypes by Major Terpene Compounds
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stahl-Biskup, E. Essential oil chemistry of the genus Thymus—A global view. In Thyme. The Genus Thymus, 1st ed.; Stahl-Biskup, E., Sáez, F., Eds.; Taylor & Francis: London, UK; New York, NY, USA, 2002; pp. 75–143. [Google Scholar]
- Trindade, H.; Costa, M.M.; Lima, S.B.; Pedro, L.G.; Figueiredo, A.C.; Barroso, J.G. Genetic diversity and chemical polymorphism of Thymus caespititius from Pico, Saõ Jorge and Terceira islands (Azores). Biochem. Syst. Ecol. 2008, 36, 790–797. [Google Scholar] [CrossRef]
- Mills, S.; Hutchins, R. ESCOP Monographs: Serpylli herba—Wild Thyme, 3rd ed.; Online series; Europaean Scientific Cooperative on Phytotherapy (ESCOP): Exeter, UK, 2014; p. 14. [Google Scholar]
- Kulišić, T.; Dragović-Uzelac, V.; Miloš, M. Antioxidant activity of Aqueous Tea Infusions Prepared from Oregano, Thyme and Wild Thyme. Food Tech. Biotech. 2006, 44, 485–492. [Google Scholar]
- Maksimović, Z.; Milenković, M.; Vučićević, D.; Ristić, M. Chemical composition and anti-microbial activity of Thymus pannonicus, All. (Lamiaceae) essential oil. Cent. Eur.J. Biol. 2008, 3, 149–154. [Google Scholar]
- Jovanović, A.; Balanč, B.; Petrović, P.; Djordjevic, V. Pharmacological potential of Thymus serpyllum L. (wild thyme) extracts and essential oil: A review. J. Engin. Process. Manag. 2022, 13, 32–41. [Google Scholar] [CrossRef]
- European Pharmacopoeia 9.0. Serpylli herba, 9th ed.; Council of Europe: Strasbourg, France, 2016; pp. 1559–1560. [Google Scholar]
- Boros, B.; Jakabová, S.; Dörnyei, Á.; Horváth, G.; Pluhár, Z.; Kilár, F.; Felinger, A. Determination of Polyphenolic Compounds by Liquid Chromatography-Mass Spectrometry in Thymus Species. J. Chromatogr. A 2010, 1217, 7972–7980. [Google Scholar] [CrossRef]
- Morales, R. The history, botany and taxonomy of the genus Thymus. In Thyme. The genus Thymus, 1st ed.; Stahl-Biskup, E., Sáez, F., Eds.; Taylor & Francis: London, UK; New York, NY, USA, 2002; pp. 1–42. [Google Scholar]
- Jalas, J. Thymus. In Flora Europaea; Tutin, T.G., Heywood, V.H., Burges, N.A., Valentine, D.H., Walters, S.M., Webb, D.A., Eds.; Cambridge University Press: Cambridge, UK, 1972; Volume 3, pp. 172–182. [Google Scholar]
- Karuza-Stojakovic, L.; Paolovic, S.; Zivanovic, P.; Todorovic, B. Composition and yield of essential oils of various species of the genus. Thymus Arch. Farm. 1989, 39, 105–111. [Google Scholar]
- Kustrak, D.; Martinis, Z.; Kuftinec, J.; Blazevic, N. Composition of the essential oils of some Thymus and Thymbra species. Flav. Fragr. J. 1990, 5, 227–231. [Google Scholar] [CrossRef]
- Mechtler, C.; Schneider, A.; Langer, R.; Jurenits, J. Individuelle Variabilitat der Zusammensetzung des Atherischen Öles von Quendel-Arten. Sci. Pharm. 1994, 62, 117. [Google Scholar]
- Baser, K.; Kirimer, N.; Ermin, N.; Özek, T. Composition of essential oils from three varieties of Thymus praecox Opiz growing in Turkey. J. Ess. Oil Res. 1996, 8, 319–321. [Google Scholar] [CrossRef]
- Bischof-Deichnik, C.; Stahl-Biskup, E.; Holthuijzen, J. Multivariate statistical analysis of the essential oil data from T. praecox ssp. polytrichus of the Tyrolean Alps. Flav. Fragr. J. 2000, 15, 1–6. [Google Scholar] [CrossRef]
- Loziene, J.; Vaiciuniené, J.; Venskutonis, R. Chemical composition of the essential oil of creeping thyme (Thymus serpyllum s. l.) growing wild in Lithuania. Planta Med. 1998, 64, 772–773. [Google Scholar] [CrossRef] [PubMed]
- Mártonfi, P.; Grejtovsky, A.; Repcák, M. Soil chemistry of Thymus species stands in Carpathians and Pannonia. Thaiszia J. Bot. 1996, 6, 39–48. [Google Scholar]
- Tohidi, B.; Rahimmalek, M.; Arzani, A. Essential oil composition, total phenolic, flavonoid contents, and antioxidant activity of Thymus species collected from different regions of Iran. Food Chem. 2017, 220, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Pluhár, Z.; Héthelyi, É.; Kutta, G.; Kamondy, L. Evaluation of environmental factors influencing essential oil quality of Thymus pannonicus All., and Thymus praecox Opiz. J. Herbs Spices Med. Plants 2007, 13, 23–43. [Google Scholar] [CrossRef]
- Pluhár, Z.; Sárosi, S.; Novák, I.; Kutta, G. Essential oil polymorphism of Hungarian common thyme (Thymus glabrescens Willd.) populations. Nat. Prod. Commun. 2008, 3, 1151–1154. [Google Scholar] [CrossRef]
- Pluhár, Z.; Sárosi, S.; Pintér, A.; Simkó, H. Essential oil polymorphism of wild growing Hungarian thyme (Thymus pannonicus All.) populations in the Carpathian Basin. Nat. Prod. Commun. 2010, 5, 1681–1686. [Google Scholar] [PubMed]
- Trendafilova, A.; Todorova, M.; Ivanova, V.; Zhelev, P.; Aneva, I. Essential Oil Composition of Ten Species from Sect. Serpyllum of Genus Thymus Growing in Bulgaria. Diversity 2023, 15, 759. [Google Scholar] [CrossRef]
- Kryvtsova, M.; Hrytsyna, M.; Salamon, I.; Skybitska, M.; Novykevuch, O. Chemotypes of Species of the Genus Thymus L. in Carpathians Region of Ukraine—Their Essential Oil Qualitative and Quantitative Characteristics and Antimicrobial Activity. Horticulturae 2022, 8, 1218. [Google Scholar] [CrossRef]
- Simon, T. (Ed.) Thymus. In A Magyar Edényes Flóra Határozója; Nemzeti Tankönyvkiadó: Budapest, Hungary, 2001; pp. 377–379. [Google Scholar]
- Van den Dool, H.; Kratz, P. A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of essential oil components by gas chromatography/mass spectrometry. J. Am. Soc. Mass Spectrom. 2007, 16, 1902–1903. [Google Scholar]
- Borhidi, A. Social behaviour types, the naturalness and relative ecological indicator values of the higher plants in the Hungarian Flora. Acta Bot. Hung. 1995, 39, 97–181. [Google Scholar]
- Pluhár, Z.; Szabó, D.; Sárosi, S. Thyme oil- Essential oil properties of Thymus vulgaris L. Plant Sci. Today 2016, 3, 312–326. [Google Scholar] [CrossRef]
- Vernet, P.; Gouyon, P.H.; Valdeyron, G. Genetic control of the oil content in Thymus vulgaris L.: A case of polymorphism in a biosynthetic chain. Genetics 1986, 69, 227–231. [Google Scholar] [CrossRef]
- Thompson, J.D. Population structure and the spatial dynamics of genetic polymorphism in thyme. In Thyme. The genus Thymus, 1st ed.; Stahl-Biskup, E., Sáez, F., Eds.; Taylor & Francis: London, UK; New York, NY, USA, 2002; pp. 44–50. [Google Scholar]
- Sur, S.V.; Tulyupa, F.H.; Tolok, A.Y.; Peresypkina, T.N. Composition of essential oils from the aboveground part of the thyme. Khim.-Farmatsev. Zhurn. 1988, 22, 1361–1366. [Google Scholar]
- Stahl-Biskup, E. The chemical composition of Thymus oils: A review of the literature 1960–1989. J. Ess. Oil Res. 1991, 3, 61–82. [Google Scholar] [CrossRef]
- Maggi, F.; Caprioli, G.; Papa, F.; Sagratinia, G.; Vittoria, S.; Kolarcik, V.; Mártonfi, P. Intra-population chemical polymorphism in Thymus pannonicus All. growing in Slovakia. Nat. Prod. Res. 2014, 28, 1557–1566. [Google Scholar] [CrossRef]
- Sostaric, I.; Arsenijevic, J.; Acic, S.; Dajic Stevanovic, Z. Essential oil polymorphism of Thymus pannonicus All. (Lamiaceae) in Serbia. J. Essent. Oil Bear. Plants 2012, 15, 237–243. [Google Scholar] [CrossRef]
- Simkó, H.; Sárosi, S.; Ladányi, M.; Marton, B.; Radácsi, P.; Csontos, P.; Gosztola, B.; Kun, R.; Pluhár, Z. Studies on occurence, essential oil data and habitat conditions of Hungarian Thymus pannonicus and Thymus glabrescens populations. Med. Arom. Plants 2013, 2, 119–125. [Google Scholar] [CrossRef]
- Arsenijević, J.; Drobac, M.; Šoštarić, I.; Jevđović, R.; Živković, J.; Ražić, S.; Moravčević, Đ.; Maksimović, Z. Comparison of essential oils and hydromethanol extracts of cultivated and wild growing Thymus pannonicus All. Ind. Crops. Prod. 2019, 130, 162–169. [Google Scholar] [CrossRef]
- Pavel, M.; Ristic, M.; Stevic, T. Essential oils of Thymus pulegioides and Thymus glabrescens from Romania: Chemical composition and antimicrobial activity. J. Serb. Chem. Soc. 2010, 75, 27–34. [Google Scholar] [CrossRef]
- Dajić-Stevanović, Z.; Šoštarić, I.; Marin, P.D.; Stojanović, D.; Ristić, M. Population variability in Thymus glarescens Willd from Serbia: Morphology, anatomy and essential oil composition. Arc. Biol. Sci. Belgr. 2008, 60, 475–483. [Google Scholar] [CrossRef]
- Loziené, K.; Venskutonis, P.R. Influence of environmental and genetic factors on the stability of essential oil composition of Thymus pulegioides. Biochem. Syst. Ecol. 2005, 33, 517–525. [Google Scholar] [CrossRef]

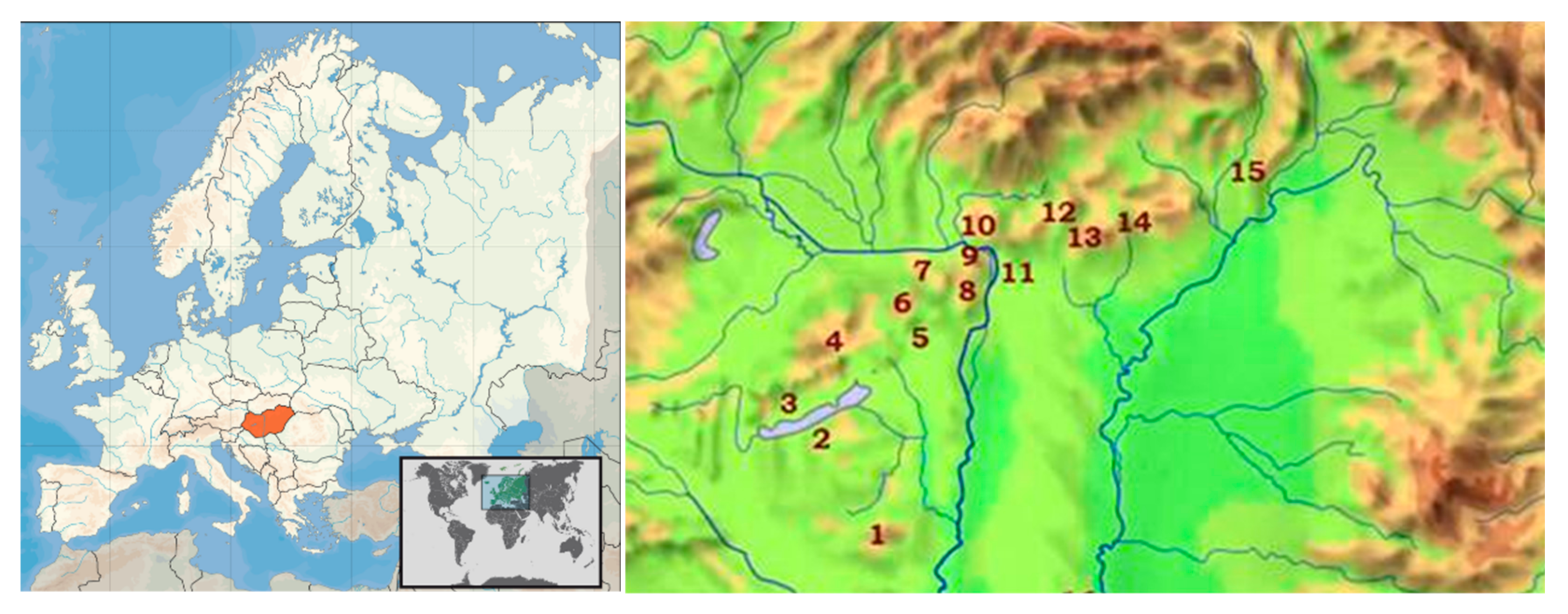
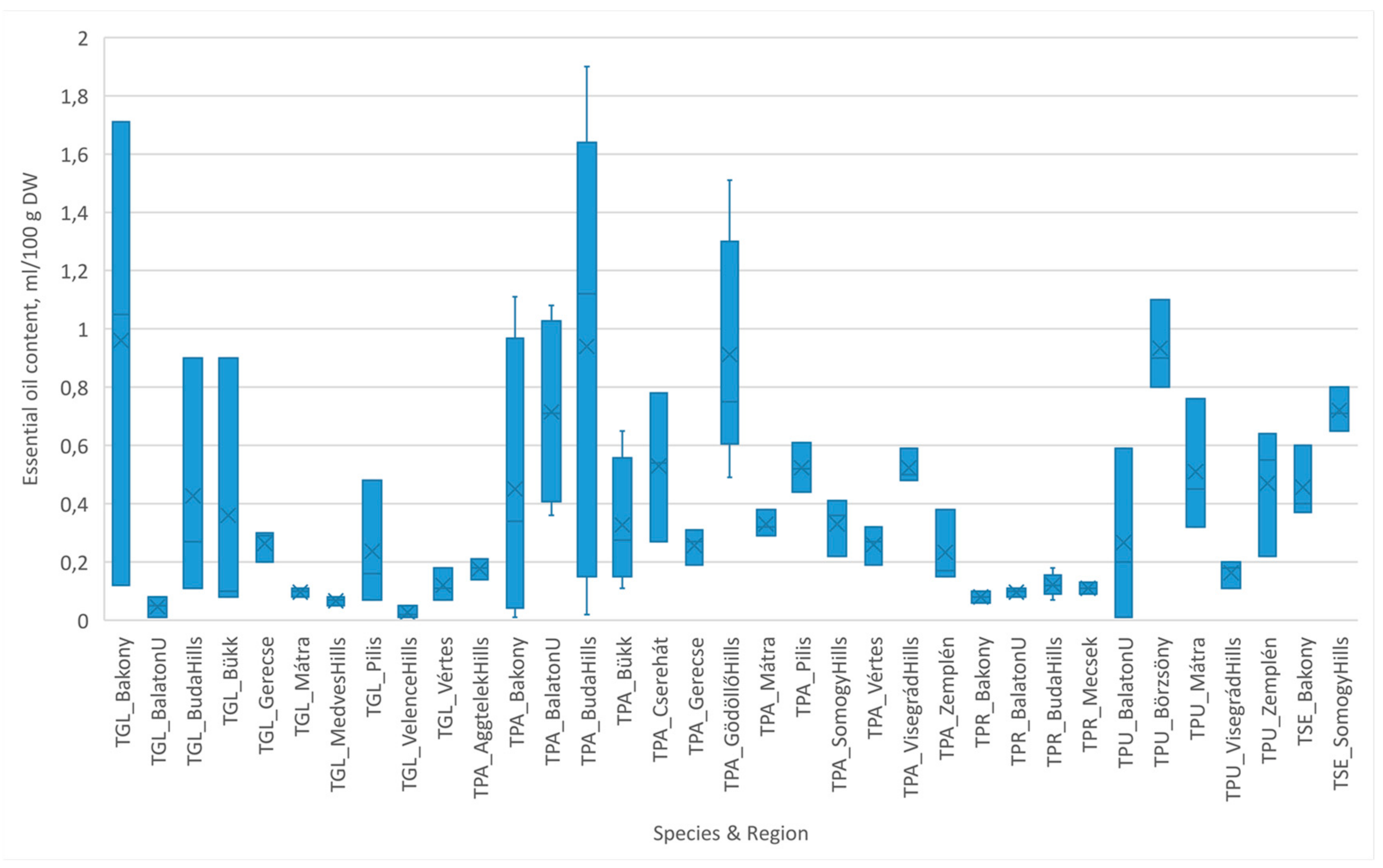
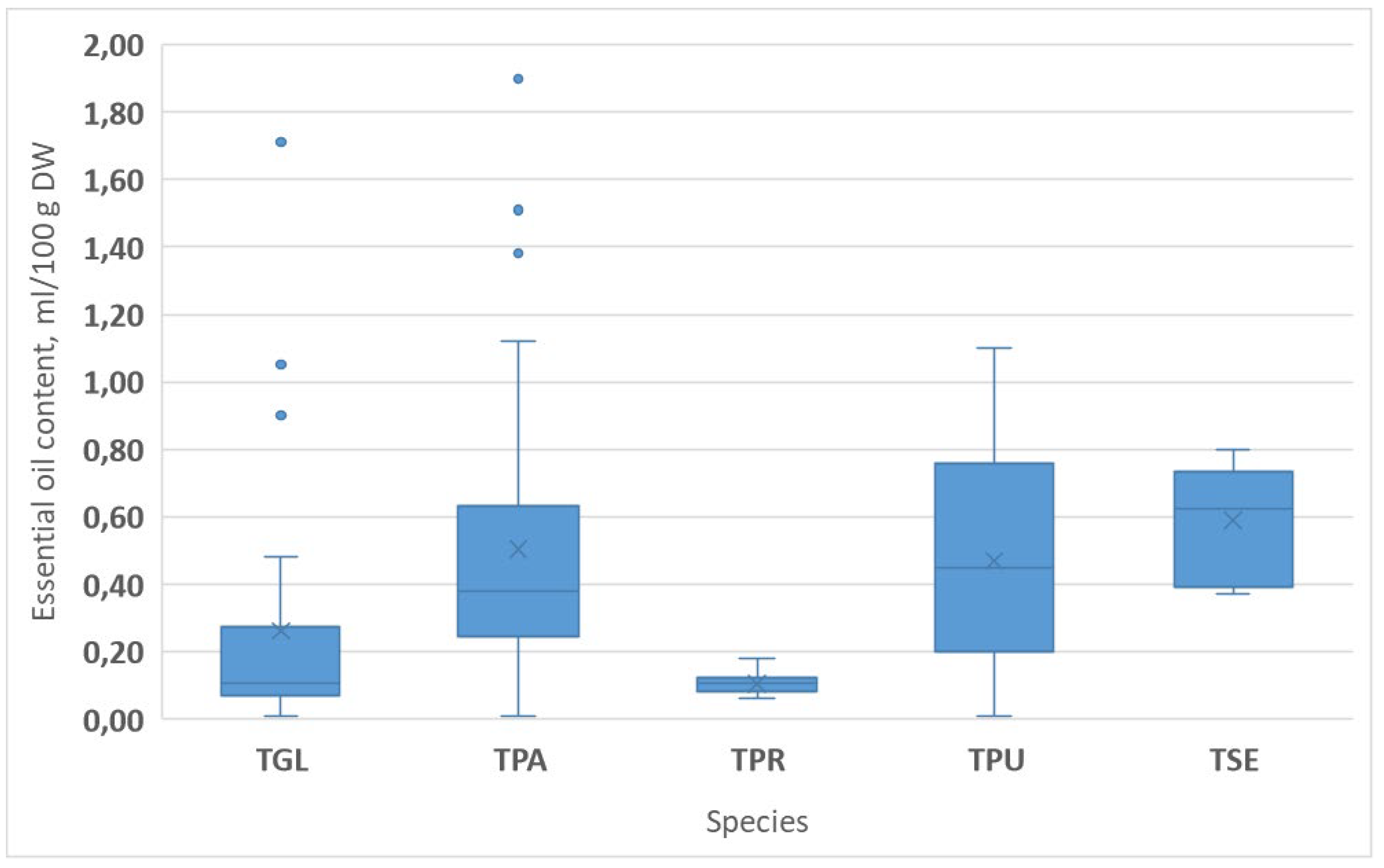

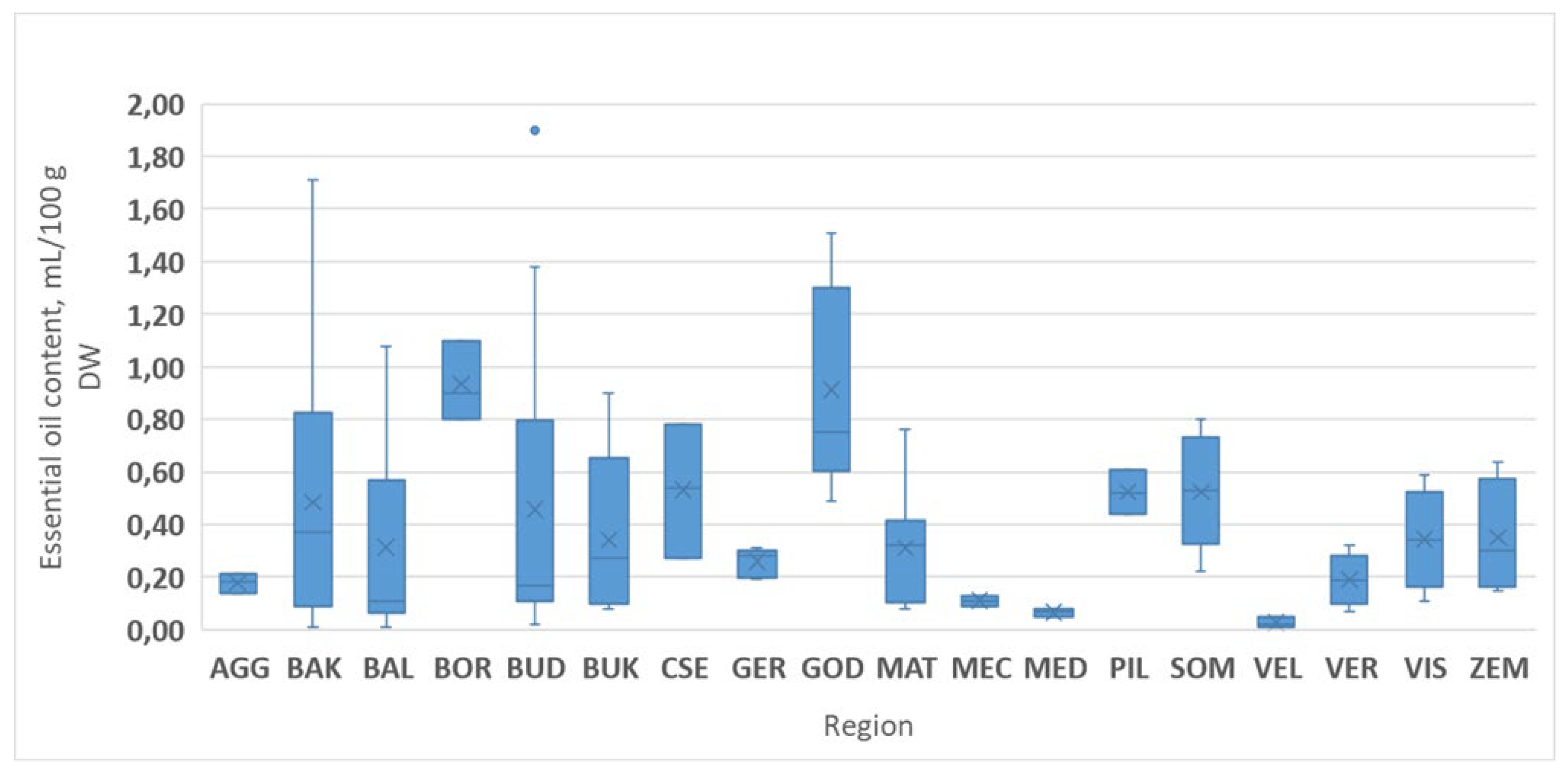



| Region | Location of Model Area | Site Code Region_Location ** | Species * Found | Base Rock Type | Soil Type |
|---|---|---|---|---|---|
| 1 Mecsek Hills | 1. Kis-Tubes Hill | 1_1 | TPR | gray limestone | black rendzina |
| 2 Somogy Hills | 2. Nagybajom, pasture | 2_2 | TSE | acidic sand | humified sand |
| 3. Köröshegy, loess hill | 2_3 | TPA | sandy loess | humified sand | |
| 3 Balaton Uplands | 4. Várvölgy, Keszthely Hills | 3_4 | TGL | calciferous sand | humified sand |
| 5. Balatongyörök, Keszthely Hills | 3_5 | TPA | dolomite | bare soil | |
| 6. Zalaszántó, Pap meadows | 3_6 | TPU | brookside silt | meadow soil | |
| 7. Tapolca Basin, Tapolca hillside | 3_7 | TPA | Dachstein limestone | black rendzina | |
| 8. Szentbékkálla, Rock Hill | 3_8a | TPU | Pannonian sandstone | bare soil | |
| 3_8b | TGL | ||||
| 9. Balatonfüred, Tamás Hill | 3_9 | TPR | dolomite | black rendzina | |
| 10. Balatonfüred, Koloska Valley | 3_10 | TPA | loess | humus carbonate soil | |
| 11. Balatonalmádi, Megye Hill | 3_11 | TPA | loess | bare soil | |
| 4 Bakony Hills | 12. Fenyőfő, Pasture Lane | 4_12a | TPA | calciferous sand | humified sand |
| 4_12b | TSE | ||||
| 13. Csesznek, Castle Hill | 4_13 | TGL | Dachstein limestone | black rendzina | |
| 14. Szőc, Pasture | 4_14 | TPA | Dachstein limestone | bare soil | |
| 15. Várpalota, Great Meadows | 4_15a | TPR | dolomite | bare soil | |
| 4_15b | TGL | ||||
| 16. Öskü, Péti Hill | 4_16 | TPA | dolomite | bare soil | |
| 17. Litér, Mogyorós Hill | 4_17 | TPA | dolomite | bare soil | |
| 5 Velence Hills | 18. Pákozd, Moveable Rocks | 5_18 | TGL | granite | bare soil |
| 6 Vértes Hills | 19. Várgesztes, Som Hill | 6_19 | TGL | Dachstein limestone | black rendzina |
| 20. Csákberény, pasture | 6_20a | TPA | dolomite | black rendzina | |
| 6_20b | TGL | ||||
| 7 Gerecse Hills | 21. Tardosbánya, rock mine plateau | 7_21a | TPA | Dachstein limestone | black rendzina |
| 7_21b | TGL | ||||
| 8A Buda Hills | 22. Budaörs, Csíki Hills, Odvas Hill | 8A_22a | TPA | dolomite | black rendzina |
| 8A_22b | TPR | ||||
| 23. Budapest, Kálvária Hill | 8A_23 | TGL | Dachstein limestone | brown forest soil | |
| 24. Budapest, Újlaki Hill | 8A_24a | TPA | Dachstein limestone | black rendzina | |
| 8A_24b | TPR | ||||
| 25. Budapest, Vörös-Kővár Hill | 8A_25 | TPA | sandstone of Hárs Hill | black rendzina | |
| 26. Budapest, Homok Hill | 8A_26a | TPA | dolomite | black rendzina | |
| 8A_26b | TPR | ||||
| 27. Nagykovácsi, Nagy-Szénás Hill | 8A_27a | TPA | dolomite, loess | black rendzina | |
| 8A_27b | TPR | ||||
| 28. Nagykovácsi, Dog Hill | 8A_28 | TGL | Dachstein limestone | black rendzina | |
| 29. Budapest, Sas Hill | 8A_29 | TPR | dolomite | black rendzina | |
| 30. Érd, Tétény Hills, | 8A_30 | TGL | Sarmathian limestone | black rendzina | |
| 31. Diósd, Tétény Hills | 8A_31 | TPR | Sarmathian limestone | black rendzina | |
| 8B Pilis Hills | 32. Dorog, Strázsa Hill | 8B_32 | TGL | calciferous sand | humified sand |
| 33. Dorog, Park | 8B_33 | TPA | calciferous sand | bare soil | |
| 34. Pilisszentiván, Fehér Hill | 8B_34 | TGL | dolomite | black rendzina | |
| 35. Pilisszántó, Pilis Hill | 8B_35 | TGL | dolomite | bare soil | |
| 9 Visegrádi Hills | 36. Szentendre, Dobos Hill | 9_36 | TPA | dolomite | black rendzina |
| 37. Dömös, Vadálló Cliffs | 9_37 | TPU | andesite | bare soil | |
| 38. Visegrád1, Nagy-Villám | 9_38 | TPA | andesite | black rendzin | |
| 39. Visegrád2, Mogyoró Hill | 9_39 | TPA | andesite | bare soil | |
| 10 Börzsöny Hills | 40. Szent Mihály Hill | 10_40 | TPU | andesite | erubase |
| 11 Gödöllő Hills | 41. Veresegyház | 11_41 | TPA | calciferous sand | brown forest soil |
| 42. Szada | 11_42 | TPA | calciferous sand | brown forest soil | |
| 43. Zsófialiget | 11_43 | TPA | railside soil | bare soil | |
| 44. Ceglédbercel, loess valley | 11_44 | TPA | loess | humified sand | |
| 45. Ceglédbercel, public park | 11_45 | TPA | calciferous sand | humified sand | |
| 12 Medves Hills | 46. Salgótarján, Salgó Hill | 12_46 | TGL | basalt | erubase |
| 13 Mátra Hills | 47. Pásztó: Köves Cliff | 13_47 | TGL | andesite | bare soil |
| 48. Mátrakeresztes: Great Meadows | 13_48a113_48a2 | TPU-L TPU-T | andesite | meadow soil | |
| 49. Sirok, Castle Hill | 13_49 | TPA | rhyolite tuff | bare soil | |
| 14 Bükk Hills | 50. Szarvaskő Hilltop | 14_50 | TGL | mudstone | bare soil |
| 51. Cserépváralja, rhyolit tuff cones | 14_51 | TPA | rhyolite tuff | bare soil | |
| 52. Noszvaj | 14_52 | TPA | mudstone | bare soil | |
| 53. Bogács | 14_53 | TPA | mudstone | black rendzina | |
| 54. Mónosbél, Szappanos Hill | 14_54 | TPA | mudstone | brown rendzina | |
| 15A Zemplén Hills | 55. Regéc, meadow | 15A_55 | TPU | rhyolite | bare soil |
| 56. Regéc, hayland | 15A_56a | TPA | andesite | bare soil | |
| 15A_56b | TPU | ||||
| 57. Bózsva, Volcanic Cliff | 15A_57 | TPA | rhyolite | bare soil | |
| 58. Vágáshuta, pasture | 15A_58a | TPA | andesite | bare soil | |
| 15A_58b | TPU | ||||
| 15B Aggtelek Hills | 59. Aggtelek, pasture | 15B_59 | TPA | Dachstein limestone | bare soil |
| 60. Jósvafő, Red Lake, meadows | 15B_60 | TPA | Dachstein limestone | black rendzina | |
| 15C Cserehát Hills | 61. Szendrőlád, Szendrő Hills | 15C_61 | TPA | Dachstein limestone | brown forest soil |
| 62. Rakaca, Szendrő Hills | 15C_62 | TPA | marble | bare soil | |
| 63. Sajógalgóc, Putnok Hills | 15C_63 | TPA | mudstone | bare soil |
| Species Name | Abbreviation | Occurrence of Populations | Social Behavior Type | |||
|---|---|---|---|---|---|---|
| Overall | New | |||||
| No | % | No | % | |||
| Thymus pannonicus All. | TPA | 38 | 51.35 | 12 | 16.21 | generalist |
| T. glabrescens Willd. | TGL | 17 | 22.98 | 5 | 6.76 | generalist |
| T. praecox Opiz | TPR | 8 | 10.81 | - | - | specialist |
| T. pulegioides L. | TPU | 9 | 12.16 | - | - | generalist |
| T. serpyllum L | TSE | 2 | 2.70 | - | - | natural competitor |
| Total | 74 | 100 | 17 | 22.87 | ||
| Site Code, Region_Location ** | Species * Found | Mean EO Content, mL/100 g DW | Relative Percentages (%) of Chief Essential Oil Compounds of Chemotypes | Terpene Class | |||
|---|---|---|---|---|---|---|---|
| 1st | 2nd | 3rd | 4th | M/MS/S ** | |||
| 1_1 | TPR | 0.11 | Caryophyllene oxide (28.50) | β-Cubebene (18.00) | S | ||
| 2_2 | TSE | 0.65 | τ-Cadinol (11.80) | Caryophyllene oxide (11.20) | β-Cubebene (8.10) | S | |
| 2_3 | TPA | 0.36 | Thymol (67.50) | M | |||
| 3_4 | TGL | 0.05 | Thymol (32.88) | β-Caryophyllene (16.50) | Germacrene D (17.62) | MS | |
| 3_5 | TPA | 1.08 | Thymol (51.10) | p-Cymene (13.00) | M | ||
| 3_6 | TPU | 0.59 | Carvacrol (32.20) | Thymol methyl ether (12.10) | γ-Terpinene (9.30) | M | |
| 3_7 | TPA | 0.36 | Thymol (38.25) | p-Cymene (12.07) | β-Bisabolene (11.43) | MS | |
| 3_8a | TPU | 0.01 | β-Caryophyllene (24.50) | Thymol (17.40) | Germacrene D (16.80) | MS | |
| 3_8b | TGL | 0.01 | Thymol (32.90) | β -Caryophyllene (16.50) | Germacrene D (13.50) | MS | |
| 3_9 | TPR | 0.08 | β-Cubebene (27.80) | Caryophyllene oxide (20.29) | β-Caryophyllene (14.70) | S | |
| 3_10 | TPA | 0.87 | Thymol (53.89) | p-Cymene (10.56) | M | ||
| 3_11 | TPA | 0.55 | Thymol (63.70) | p-Cymene (11.50) | M | ||
| 4_12a | TPA | 1.10 | Thymol (40.00) | γ-Terpinene (20.20) | p-Cymene (14.70) | M | |
| 4_12b | TSE | 0.37 | Geranyl isobutyrate (44.00) | 1,8-Cineol (16.90) | M | ||
| 4_13 | TGL | 1.71 | Thymol (34.0) | p-Cymene (22.9) | M | ||
| 4_14 | TPA | 0.01 | Germacrene D (49.00) | β-Farnesene (8.00) | δ-Cadinene (8.00) | S | |
| 4_15a | TPR | 0.06 | Germacrene D (43.90) | β-Caryophyllene (10.60) | S | ||
| 4_15b | TGL | 0.12 | Germacrene D (55.40) | β-Caryophyllene (14.80) | S | ||
| 4_16 | TPA | 0.54 | Thymol (27.70) | Linalyl acetate (18.80) | γ-Terpinene (18.60) | M | |
| 4_17 | TPA | 0.14 | Germacrene D (43.40) | β-Caryophyllene (15.00) | S | ||
| 5_18 | TGL | 0.02 | nd | nd | nd | nd | |
| 6_19 | TGL | 0.18 | Geraniol (49.00) | Germacrene D (13.60) | MS | ||
| 6_20a | TPA | 0.32 | Thymol (30.17) | p-Cymene (26.00) | Thymol methylether (13.38) | γ-Terpinene (9.55) | M |
| 6_20b | TGL | 0.07 | τ-Cadinol (43.20) | Germacrene D (15.55) | cis-γ-Cadinene (10.41) | S | |
| 7_21a | TPA | 0.27 | p-Cymene (53.70 | Geraniol (15.80) | M | ||
| 7_21b | TGL | 0.30 | p-Cymene (45.00) | Geraniol (13.60) | Linalyl acetate (9.9) | M | |
| 8A_22a | TPA | 0.28 | Thymol (38.30) | p-Cymene (17.20) | M | ||
| 8A_22b | TPR | 0.11 | Geraniol (23.20) | Germacrene D (14.70) | β-Caryophyllene (12.20) | MS | |
| 8A_23 | TGL | 0.27 | Germacrene D (43.75) | Thymol (25.03) | MS | ||
| 8A_24a | TPA | 1.10 | Thymol (41.30) | p-Cymene (19.20) | M | ||
| 8A_24b | TPR | 0.01 | Geraniol (18.20) | Germacrene D (16.60) | MS | ||
| 8A_25 | TPA | 0.22 | Thymol (36.50) | p-Cymene (27.30) | M | ||
| 8A_26a | TPA | 1.37 | Geraniol (25.30) | γ-Terpinene (24.70) | M | ||
| 8A_26b | TPR | 0.07 | Germacrene D (31.70) | β-Caryophyllene (21.20) | S | ||
| 8A_27a | TPA | 1.90 | Germacrene D (29.7) | β-Caryophyllene (22.00) | Farnesol (10.40) | S | |
| 8A_27b | TPR | 0.13 | Germacrene D (31.90) | β-Caryophyllene (22.30) | S | ||
| 8A_28 | TGL | 0.90 | Germacrene D (44.73) | β-Caryophyllene (13.88) | Bicyclogermacrene (10.45) | S | |
| 8A_29 | TPR | 0.12 | Germacrene D (39.90) | β-Caryophyllene (21.60) | S | ||
| 8A_30 | TGL | 0.11 | Germacrene D (56.40) | β-Caryophyllene (25.07) | S | ||
| 8A_31 | TPR | 0.18 | Caryophyllene oxide (16.00) | β-Caryophyllene (11.70) | S | ||
| 8B_32 | TGL | 0.16 | Germacrene D (17.80) | Nerolidol (12.99) | β-Cadinene (12.86) | β-Bisabolene (9.19) | S |
| 8B_33 | TPA | 0.52 | Thymol (53.58) | p-Cymene (10.52) | γ-Terpinene (9.63) | M | |
| 8B_34 | TGL | 0.07 | β-Caryophyllene (29.77) | Germacrene D (23.82) | β-Cadinene (11.90) | S | |
| 8B_35 | TGL | 0.48 | Thymol (69.28) | γ-Terpinene (18.28) | M | ||
| 9_36 | TPA | 0.48 | p-Cymene (45.13) | Thymol (20.48) | M | ||
| 9_37 | TPU | 0.11 | Germacrene D (21.70) | β-Caryophyllene (13.80) | γ-Muurulene (10.30) | S | |
| 9_38 | TPA | 0.59 | Thymol (52.92) | γ-Terpinene (13.52) | p-Cymene (10.72) | M | |
| 9_39 | TPA | 0.50 | Thymol (66.00) | p-Cymene (10.27) | M | ||
| 10_40 | TPU | 0.80 | p-cymene (18.70) | Spathulenol (16.10) | Geraniol (14.00) | MS | |
| 11_41 | TPA | 0.72 | Thymol (32.00–56.00) | p-Cymene (9.80–21.50) | γ-Terpinene (5.90–15.40) | M | |
| 11_42 | TPA | 1.15 | Thymol (43.00–60.00) | p-Cymene (6.90–21.10) | γ-Terpinene (7.30–18.50) | M | |
| 11_43 | TPA | 0.83 | Thymol (38.90) | p-Cymene (8.90) | γ-Terpinene (11.10) | M | |
| 11_44 | TPA | 1.09 | Thymol (48.00–53.00) | p-Cymene (5.90–15.80) | γ-Terpinene (7.10–11.00) | M | |
| 11_45 | TPA | 0.49 | Thymol (32–56) | p-Cymene (2.40–6.30) | γ-Terpinene (6.70–7.80) | M | |
| 12_46 | TGL | 0.05 | Thymol (14.40) | Germacrene D (12.10) | Geraniol (10.80) | MS | |
| 13_47 | TGL | 0.08 | Thymol (29.30) | Germacrene D (14.20) | MS | ||
| 13_48a1 | TPU-L | 0.32 | Geranial (22.20) | Linalyl acetate (19.8) | Neral (14.30) | Linalool (14.20) | M |
| 13_48a2 | TPU-T | 0.76 | Thymol (56.20) | γ-Terpinene (10.40) | Thymol methylether (9.90) | M | |
| 13_49 | TPA | 0.32 | Thymol (41.9) | p-Cymene (20.2) | Borneol (10.30) | M | |
| 14_50 | TGL | 0.10 | Germacrene D (9.40) | β-Caryophyllene (6.90) | cis-Ocymene (6.00) | MS | |
| 14_51 | TPA | 0.27 | β-Cadinene (28.82) | Germacrene D (13.18) | S | ||
| 14_52 | TPA | 0.11 | Linalool (24.44) | p-Cymene (14.14) | Thymol (10.78) | Carvacrol (10.31) | M |
| 14_53 | TPA | 0.28 | Linalool (47.12) | p-Cymene (15.18) | M | ||
| 14_54 | TPA | 0.65 | Carvacrol (40.71) | p-Cymene (15.97) | γ-Terpinene (13.67) | M | |
| 15A_55 | TPU | 0.22 | β-Caryophyllene (53.2) | β-Cubebene (19.20) | S | ||
| 15A_56a | TPA | 0.15 | β-Caryophyllene (48.70) | β-Cubebene (19.90) | Thymol (8.00) | MS | |
| 15A_56b | TPU | 0.64 | Linalool (38.1) | Geraniol (23.90) | Linalyl acetate (14.40) | M | |
| 15A_57 | TPA | 0.17 | Caryophyllene oxide (45.10) | β-Cubebene (15.70) | Linalool (13.80) | MS | |
| 15A_58a | TPA | 0.38 | β-Cubebene (24.50) | Linalool (7.59) | Linalyl acetate (7.41) | MS | |
| 15A_58b | TPU | 0.55 | Geraniol (27.50) | Linalyl acetate (20.20) | Thymol methyl ether (13.50) | M | |
| 15B_59 | TPA | 0.14 | Germacrene D (26.35) | Caryophyllene oxide (10.43) | S | ||
| 15B_60 | TPA | 0.21 | Geranyl acetate (24.08) | β-bisabolene (16.27) | Geraniol (12.00) | MS | |
| 15C_61 | TPA | 0.78 | p-Cymene (44.90) | Thymol (20.22) | γ-Terpinene (10.12) | M | |
| 15C_62 | TPA | 0.54 | Linalool (26.63) | Thymol (22.25) | p-Cymene (14.56) | γ-Terpinene (11.05) | M |
| 15C_63 | TPA | 0.27 | Geraniol (12.67) | Geranyl acetate (11.58) | p-Cymene (11.08) | Carvacrol (8.80) | M |
| No. | Compound | RT | LRI | Terpene Class ** | Frequency of Chief Compounds by Species * (Number of Occurrence) | Overall Freqency of Compounds in EO Samples | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TPA | TGL | TPR | TPU | TSE | Total No. of Occur. | Total Share, % | Mean % | |||||
| 1 | p-Cymene | 8.09 | 1026 | MH | 26 | 2 | 1 | 29 | 42.00 | 19.39 | ||
| 2 | 1,8-Cineol | 8.38 | 1034 | MO | 1 | 1 | 1.35 | 16.90 | ||||
| 3 | cis-β-Ocymene | 8.50 | 1036 | MH | 1 | 1 | 1.35 | 16.00 | ||||
| 4 | γ-Terpinene | 9.20 | 1056 | MH | 12 | 1 | 2 | 15 | 20.27 | 13.61 | ||
| 5 | Linalool | 10.76 | 1097 | MO | 5 | 2 | 7 | 9.46 | 24.55 | |||
| 6 | Isoborneol | 13.43 | 1162 | MO | 1 | 1 | 1.35 | 10.30 | ||||
| 7 | Thymol methyl ether | 16.20 | 1228 | MO | 1 | 3 | 4 | 5.41 | 12.23 | |||
| 8 | Neral | 16.58 | 1249 | MO | 1 | 1 | 1.35 | 14.30 | ||||
| 9 | Linalyl acetate | 17.11 | 1250 | MO | 2 | 1 | 2 | 5 | 6.76 | 14.14 | ||
| 10 | Geraniol | 17.20 | 1252 | MO | 4 | 3 | 1 | 4 | 12 | 16.22 | 20.44 | |
| 11 | Geranial | 17.86 | 1268 | MO | 1 | 1.35 | 22.20 | |||||
| 12 | Thymol | 18.81 | 1290 | MO | 25 | 7 | 1 | 34 | 45.95 | 38.98 | ||
| 13 | Carvacrol | 19.20 | 1300 | MO | 3 | 1 | 4 | 5.41 | 23.00 | |||
| 14 | Geranyl acetate | 22.43 | 1388 | MO | 2 | 2 | 2.7 | 17.83 | ||||
| 15 | β -Cubebene | 22.47 | 1389 | SH | 3 | 2 | 1 | 1 | 7 | 9.46 | 18.16 | |
| 16 | β-Caryophyllene | 23.68 | 1420 | SH | 3 | 7 | 7 | 3 | 1 | 21 | 28.38 | 21.25 |
| 17 | β-Farnesene | 25.27 | 1459 | SH | 1 | 1 | 1.35 | 8.00 | ||||
| 18 | γ-Muurulene | 25.99 | 1477 | SH | 1 | 1 | 1,35 | 10.30 | ||||
| 19 | Germacrene D | 26.18 | 1482 | SH | 5 | 13 | 5 | 2 | 25 | 33.79 | 27.95 | |
| 20 | Bicyclogermacrene | 26.81 | 1497 | SH | 1 | 1 | 1.35 | 10.45 | ||||
| 21 | β-Bisabolene | 27.23 | 1508 | SH | 2 | 1 | 3 | 4.05 | 12.30 | |||
| 22 | cis-γ-Cadinene | 27.49 | 1515 | SH | 1 | 1 | 1.35 | 10.41 | ||||
| 23 | δ-Cadinene | 27.80 | 1524 | SH | 1 | 1 | 1.35 | 8.00 | ||||
| 24 | Geranyl isobutyrate | 29.33 | 1566 | MO | 1 | 1 | 1.35 | 44.00 | ||||
| 25 | Nerolidol | 29.51 | 1570 | SO | 1 | 1 | 1.35 | 12.99 | ||||
| 26 | β-Cadinene | 29.87 | 1580 | SH | 1 | 2 | 3 | 4.05 | 17.86 | |||
| 27 | Spathulenol | 29.98 | 1584 | SO | 1 | 1 | 1.35 | 16.10 | ||||
| 28 | Caryophyllene oxide | 30.20 | 1590 | SO | 2 | 3 | 1 | 6 | 8.11 | 21.92 | ||
| 29 | τ-Cadinol | 32.26 | 1644 | SH | 1 | 1 | 2 | 2.70 | 43.20 | |||
| 30 | E,E-Farnesol | 35.33 | 1728 | SO | 1 | 1 | 1.35 | 10.40 | ||||
| Non-oxygenated monoterpenes (MH) | 3 | 38 | 4 | 0 | 3 | 0 | 45 | |||||
| Oxygenated monoterpenes (MO) | 12 | 41 | 11 | 1 | 14 | 2 | 73 | |||||
| Total monoterpenes (M) | 15 | 79 | 15 | 1 | 17 | 2 | 118 | |||||
| Non-oxygenated sesquiterpenes (SH) | 11 | 16 | 26 | 14 | 7 | 3 | 66 | |||||
| Oxygenated sesquiterpenes (SO) | 4 | 2 | 1 | 3 | 1 | 1 | 9 | |||||
| Total sesquiterpenes (S) | 15 | 18 | 27 | 17 | 8 | 4 | 75 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pluhár, Z.; Kun, R.; Cservenka, J.; Neumayer, É.; Tavaszi-Sárosi, S.; Radácsi, P.; Gosztola, B. Variations in Essential Oil Composition and Chemotype Patterns of Wild Thyme (Thymus) Species in the Natural Habitats of Hungary. Horticulturae 2024, 10, 150. https://doi.org/10.3390/horticulturae10020150
Pluhár Z, Kun R, Cservenka J, Neumayer É, Tavaszi-Sárosi S, Radácsi P, Gosztola B. Variations in Essential Oil Composition and Chemotype Patterns of Wild Thyme (Thymus) Species in the Natural Habitats of Hungary. Horticulturae. 2024; 10(2):150. https://doi.org/10.3390/horticulturae10020150
Chicago/Turabian StylePluhár, Zsuzsanna, Róbert Kun, Judit Cservenka, Éva Neumayer, Szilvia Tavaszi-Sárosi, Péter Radácsi, and Beáta Gosztola. 2024. "Variations in Essential Oil Composition and Chemotype Patterns of Wild Thyme (Thymus) Species in the Natural Habitats of Hungary" Horticulturae 10, no. 2: 150. https://doi.org/10.3390/horticulturae10020150
APA StylePluhár, Z., Kun, R., Cservenka, J., Neumayer, É., Tavaszi-Sárosi, S., Radácsi, P., & Gosztola, B. (2024). Variations in Essential Oil Composition and Chemotype Patterns of Wild Thyme (Thymus) Species in the Natural Habitats of Hungary. Horticulturae, 10(2), 150. https://doi.org/10.3390/horticulturae10020150







