Hazop Analysis of a Bioprocess for Polyhydroxyalkanoate (PHA) Production from Organic Waste: Part B
Abstract
1. Introduction
2. Materials and Methods
2.1. HAZOP Analysis
- -
- To verify the project safety;
- -
- To check operating and safety procedures;
- -
- To increase the safety of an existing system;
- -
- To verify that safety equipment is working in the best possible way.
- (1)
- Definition of the purpose and objectives of the study;
- (2)
- Team selection;
- (3)
- Study preparation;
- (4)
- Carrying out the analysis;
- (5)
- Recording the results.
- Operating procedures development;
- Verification of design values, process parameters and possible modifications;
- Request for additional alarms;
- Request for unforeseen alarms or blocks;
- Useful information for assessing and managing the risk associated with the accidental identified events.
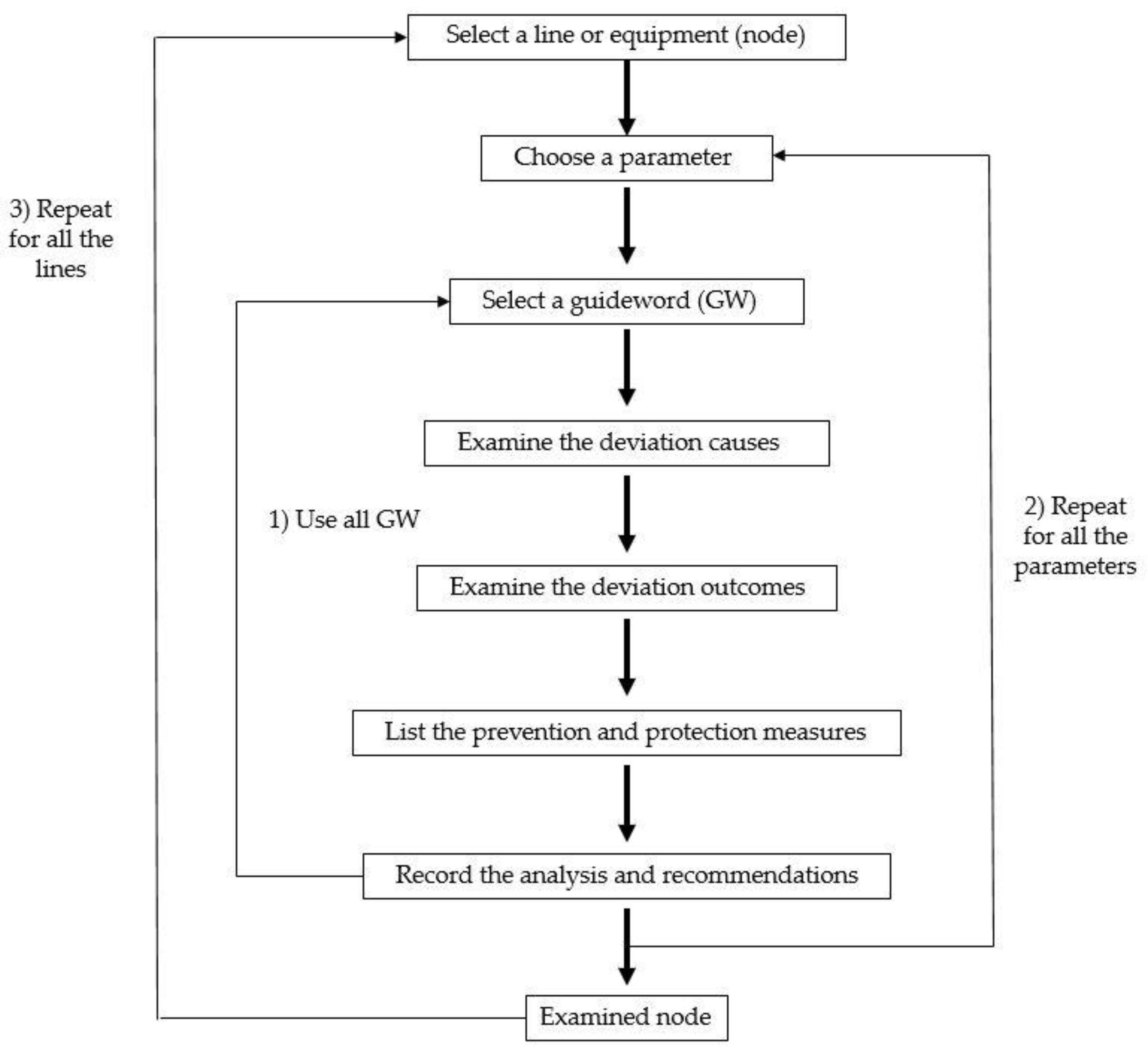
2.2. HAZOP Methodology
- The examination must be systematic;
- The analysis must be carried out with a degree of formality (e.g., forms), so that the reasons for each decision made during the analysis can be clearly identified by different people at different times;
- All working on the implementation of the project must be involved in the analysis.
- (1)
- A checklist;
- (2)
- A tailored HAZOP method (BioHazOp).
2.2.1. The Checklist
- (1)
- Process specification section (engineering process and bioprocess: substances hazard classification, biohazard, flammability, explosivity and relevant parameters);
- (2)
- General section (management outline): operating procedures, plant layout, emergency response/on-going programs and process hazard analysis.
2.2.2. BioHazOp
2.3. Piping and Instrumentation Diagram (P&ID)
3. Case-Study: Pilot Plant for the PHA Production
- (1)
- Acidogenic fermentation of the organic feedstock for volatile fatty acids (VFA) production;
- (2)
- PHA-storing microorganisms’ selection from MMC;
- (3)
- PHA accumulation maximization.
- (1)
- biomass withdrawal;
- (2)
- regeneration;
- (3)
- feeding;
- (4)
- reaction.
4. Results
Relevant Deviations Matrices and BioHazOp Worksheets
5. Discussion
- A periodic check of their side covers. In case of leakages, the screws must be re-tightened or gaskets must be replaced;
- A hydraulic damper check (lubricant level). In particular, it is recommended to use lubricant, which has a kinematic viscosity included between 30 and 50 mm²/s (ISO grade);
- Check of hydraulic circuit connections (all the components have to be perfectly tightened).
- (1)
- Technical measures (e.g., aeration of SBR performed by air bubbles under pressure, instead of surface aerators);
- (2)
- Organizational measures (e.g., only qualified employees are allowed to do specified works; signage/warnings and/or administrative controls);
- (3)
- Personal measures, such as personal protective equipment (PPE).
- Using air diffusers, such as an aeration system, instead of systems involving the mechanical agitation of fermentation liquid;
- Isolating workers from equipment with high bioaerosol emissions and using glass panes or plastic curtains;
- Forced ventilation system for activities located within indoor places;
- Organizing the work tasks to prevent workers from spending long periods in areas where the bioaerosol concentrations are higher;
- Providing adequate welfare facilities and first-aid equipment;
- Ensuring the use of PPE, such as gloves, goggles, a face shield, water-resistant suit or respiratory protective equipment (RPE), depending on the worker task;
- Making effective arrangements for monitoring the staff health.
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| RELEVANT DEVIATIONS MATRIX | ||||||||||
| Parameters | ||||||||||
| SBR | ||||||||||
| Guide words | Flow (VFA-rich stream) | Temperature | Mixing | CL (cycle length) | HRT | HRT/CL | DO (dissolved oxygen) | Organic Loading Rate (OLR) | Water flow | Withdrawal |
| more | X | X | X | |||||||
| no/less | X | X | X | X | ||||||
| reverse | X | X | ||||||||
| lower | X | X | X | |||||||
| higher | X | X | X | |||||||
| no | X | |||||||||
| longer | X | X | ||||||||
| shorter | X | X | ||||||||
| Node 2: SBR | |||||||
| Deviation | Engineering process | Biotechnology process | Existing counter measures | BioHazard | Proposed counter measures | ||
| causes | consequences | causes | consequences | ||||
| Working volume | |||||||
| MORE | -failure of IE2 -failure of E7 -failure of IE3 and E6 -failure of IE4 and E11 -failure of PLC control system | -increased VFA concentration -HRT increase due to increased SBR working Volume 1 -uncontrolled release of substrate from the tank | -total or partial inhibition of microorganisms that may increase the organic load 2 -decrease of the rate of PHA synthesis -selection of a slower and less efficient microbial community in terms of PHA production | -visual inspection -periodic maintenance of IE2 and E7 -IE2 and E7 setting check -maintenance of the level gauge installed on the tank 3 | -install a flow meter on the E7 outlet section | ||
| NO/LESS | -failure of IE2 -failure of E7 -failure of IE3 and E6 -failure of IE4 and E11 -failure of PLC control system | -decreased VFA concentration -decrease or absence of substrate in the reactor | -lowering of the organic load -microbial cells decay 4 | -visual inspection -periodic maintenance of IE2 and E7 -IE2 and E7 setting check | |||
| REVERSE | -failure of IE2 -failure of E7 -failure of non-return valve on the E7 outlet section | -absence of substrate in the reactor 5 | -visual inspection -periodic maintenance of IE2 and E7 -IE2 and E7 setting check -periodic maintenance of non-return valves | ||||
| Temperature (immersion heater) | |||||||
| LOWER (<25°C) | -heater failure | -slowing down of biomass kinetics -increased substrate degradation times -increased feast phase with selection damage -decreased PHA yield | |||||
| HIGHER (>28°C) | -heater failure | -uncontrolled and different composition of the mixture due to the possible growth of other species of microorganisms 6 -decrease in PHA yield for T>30°C | |||||
| Mixing | |||||||
| NO | -blower failure | -biomass sedimentation -little or no substrate degradation -emptying of the biomass from the reactor by the washout (pump E11) | -periodic cleaning of the plates | -blower redundancy ( installation of a second blower) -electric blower motor provided with preferential power supply (generator or uninterruptible power supply) | |||
| HRT (1–2 days) 7 | |||||||
| LONGER (>2 days) | -failure of PLD software -failure of IE2 -failure of E7 | -ineffective selection of micro-organisms | -visual inspection -periodic maintenance of IE2 and E7 -IE2 and E7 setting check | -install flow transmitters | |||
| SHORTER (<1 days) | -failure of PLD software -failure of IE2 -failure of E7 | -inactivation of micro-organisms (washout) | -visual inspection -periodic maintenance of IE2 and E7 -IE2 and E7 setting check | -install flow transmitters | |||
| HRT/CL 8 | |||||||
| LOWER | -failure of PLD software -failure of IE2 -failure of E7 | -See SHORTER HRT | -visual inspection -periodic maintenance of IE2 and E7 -IE2 and E7 setting check | -install flow transmitters | |||
| HIGHER | -failure of PLD software -failure of IE2 -failure of E7 | -See LONGER HRT | -visual inspection -periodic maintenance of IE2 and E7 -IE2 and E7 setting check | -install flow transmitters | |||
| DO (dissolved oxygen) 9 | |||||||
| NO/LESS | -blower failure -plates clogging | -biomass decay | -blower redundancy (installation of a second blower) -electric blower motor provided with preferential power supply (generator or uninterruptible power supply) -oxygen sensor redundancy | ||||
| Organic Loading Rate (OLR) 10 | |||||||
| LOWER (<4) | -failure of IE2 -failure of E7 -failure of PLC software | -feeding of less substrate -progressive lowering of working volume -HRT decrease | -biomass decay -washout | -visual inspection -periodic maintenance of IE2 and E7 -IE2 and E7 setting check -periodic monitoring of the VFA concentration in the fermented stream -addition of water to the SBR by E6 and IE3 | -online analysis of the VFA concentration 11 in the storage tank and automatic intervention of feeding system (IE2 and E7) and water addition system (IE3 and E6) to ensure a constant organic load | ||
| HIGHER (>4) | -failure of IE2 -failure of E7 -failure of PLC software | -substrate feeding increase -excessive VFA concentration -excessive increase of working volume and HRT raising | -selection inhibition -excessive duration of the feast phase | -visual inspection -periodic maintenance of IE2 and E7 -IE2 and E7 setting check -periodic monitoring of the VFA concentration in the fermented stream -addition of water to the SBR by E6 and IE3 | -online analysis of the VFA concentration12 in the storage tank and automatic intervention of the feeding system (IE2 and E7) and water addition system (IE3 and E6) to ensure a constant organic load | ||
| Water flow | |||||||
| MORE | -failure of IE3 -failure of E6 -failure of PLC software | -biomass dilution -excessive increase of working volume and HRT raising | -visual inspection -periodic maintenance of IE3 and E6 -IE3 and E6 setting check | ||||
| NO/LESS | -failure of IE3 -failure of E6 -failure of PLC software | -biomass concentration -progressive lowering of working volume and HRT decrease | -visual inspection -periodic maintenance of IE3 and E6 -IE3 and E6 setting check | ||||
| REVERSE | -failure of IE3 -failure of E6 | -visual inspection-periodic maintenance of IE3 and E6-IE3 and E6 setting check | Install a non-return valve on E6 outlet section | ||||
| Withdrawal | |||||||
| MORE | -failure of IE4 -failure of E11 -failure of PLC software | -variation in the SBR working volume -see Flow (VFA-rich stream) | |||||
| NO/LESS | -failure of IE4 -failure of E11 -failure of PLC software | -variation in the SBR working volume -see Flow (VFA-rich stream) | |||||
References
- Available online: https://www.marketsandmarkets.com/PressReleases/pha.asp (accessed on 30 January 2023).
- Khatami, K.; Perez-Zabaleta, I.M.; Owusu-Agyeman, I.; Cetecioglu, Z. Waste to bioplastics: How close are we to sustainable polyhydroxyalkanoates production? Waste Manag. 2021, 119, 374–388. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chen, Q.; Wen, Z.; Zhao, L.; Lee, D.J.; Yang, L.; Wang, Y. Insights into Feast-Famine polyhydroxyalkanoate (PHA)-producer selection: Microbial community succession, relationships with system function and underlying driving forces. Water Res. 2018, 131, 167–176. [Google Scholar] [CrossRef]
- Marang, L.; van Loosdrecht, M.C.M.; Kleerebezem, R. Enrichment of PHA-producing bacteria under continuous substrate supply. New Biotechnol. 2018, 41, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Oehmen, A.; Freitas, E.B.; Carvalho, G.; Reis, M.A.M. The link of feast-phase dissolved oxygen (DO) with substrate competition and microbial selection in PHA production. Water Res. 2017, 112, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Casson Moreno, V.; Giacomini, E.; Cozzani, V. Identification of major accident hazards in industrial biological processes. Chem. Eng. Trans. 2016, 48, 679–684. [Google Scholar]
- Casson Moreno, V.; Cozzani, V. Integrated hazard identification within the risk management of industrial biological process-es. Saf. Sci. 2018, 103, 340–351. [Google Scholar] [CrossRef]
- Valentino, F.; Moretto, G.; Lorini, L.; Bolzonella, D.; Pavan, P.; Majone, M. Pilot-Scale Polyhydroxyalkanoate Production from Combined Treatment of Organic Fraction of Municipal Solid Waste and Sewage Sludge. Ind. Eng. Chem. Res. 2019, 58, 12149–12158. [Google Scholar] [CrossRef]
- Moretto, G.; Lorini, L.; Pavan, P.; Crognale, S.; Tonanzi, B.; Rossetti, S.; Majone, M.; Valentino, F. Biopolymers from Urban Organic Waste: Influence of the Solid Retention Time to Cycle Length Ratio in the Enrichment of a Mixed Microbial Culture (MMC). ACS Sustain. Chem. Eng. 2020, 8, 14531–14539. [Google Scholar] [CrossRef]
- Dunjó, J.; Fthenakis, V.; Vílchez, J.A.; Arnaldos, J. Hazard and operability (HAZOP) analysis. A literature review. J. Hazard. Mater. 2010, 173, 19–32. [Google Scholar] [CrossRef]
- Center for Chemical Process Safety (CCPS). Guidelines for Process Safety in Bioprocess Manufacturing Facilities; Wiley: New York, NY, USA, 2010; pp. 1–248. [Google Scholar]
- Casson Moreno, V.; Guglielmi, D.; Cozzani, V. Identification of critical safety barriers in biogas facilities. Reliab. Eng. Syst. Saf. 2018, 169, 81–94. [Google Scholar] [CrossRef]
- Paltrinieri, N.; Tugnoli, A.; Buston, J.; Wardman, M.; Cozzani, V. Dynamic procedure for atypical scenarios identification (DyPASI): A new systematic HAZID tool. J. Loss Prev. Process. Ind. 2013, 26, 683–695. [Google Scholar] [CrossRef]
- Directive 2000/54/EC of the European Parliament and of the Council of 18 September 2000 on the Protection of Workers from Risks Related to Exposure to Biological Agents at Work (Seventh Individual Directive within the Meaning of Article 16(1) of Di-rective 89/391/EEC. Official Journal of the European Communities L 262/21 of 17.10.2000. Available online: https://osha.europa.eu/en/legislation/directives/exposure-to-biological-agents/77 (accessed on 1 November 2022).
- 15. Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classi-fication, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006. Official Journal of European Union of 01.03.2022. Available online: https://osha.europa.eu/en/legislation/directives/regulation-ec-no-1272-2008-classification-labelling-and-packaging-of-substances-and-mixtures (accessed on 1 November 2022).
- Nguemna Tayou, L.; Lauri, R.; Incocciati, E.; Pietrangeli, B.; Majone, M.; Micolucci, F.; Gottardo, M.; Valentino, F. Acidogenic fermentation of food waste and sewage sludge mixture: Effect of operating parameters on pro-cess performance and safety aspects. Process Saf. Environ. Prot. 2022, 163, 158–166. [Google Scholar] [CrossRef]
- Villano, M.; Valentino, F.; Barbetta, A.; Martino, L.; Scandola, M.; Majone, M. Polyhydroxyalkanoates production with mixed microbial cultures: From culture selection to polymer recovery in a high-rate continuous process. New Biotechnol. 2014, 31, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Valentino, F.; Morgan-Sagastume, F.; Campanari, S.; Villano, M.; Werker, A.; Majone, M. Carbon recovery from wastewater through bioconversion into biodegradable polymers. New Biotechnol. 2017, 37, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Crognale, S.; Tonanzi, B.; Valentino, F.; Majone, M.; Rossetti, S. Microbiome dynamics and phaC synthase genes selected in a pilot plant producing polyhydroxyalkanoate from the organic fraction of urban waste. Sci. Total Environ. 2019, 689, 765–773. [Google Scholar] [CrossRef]
- Commission Directive (EU) 2019/1833 of 24 October 2019 Amending Annexes I, III, V and VI to Directive 2000/54/EC of the European Parliament and of the Council as Regards Purely Technical Adjustments. Official Journal of European Union of 31.10.2019. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32019L1833 (accessed on 1 November 2022).
- Commission Directive (EU) 2020/739 of 3 June 2020 amending Annex III to Directive 2000/54/EC of the European Parliament and of the Council as Regards the Inclusion of SARS-CoV-2 in the List of Biological Agents Known to Infect Humans and Amending Commission Directive (EU) 2019/1833. Official Journal of European Union of 04.06.2020. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32020L0739 (accessed on 1 November 2022).
- Fernández, R.; Dinsdale, R.M.; Guwy, A.J.; Premier, G.C. Critical analysis of methods for the measurement of volatile fatty acids. Crit. Rev. Environ. Sci. Technol. 2016, 46, 209–234. [Google Scholar] [CrossRef]
- Nativ, P.; Gräber, Y.; Aviezer, Y.; Lahav, O. A Simple and Accurate Approach for Determining the VFA Concentration in Anaerobic Digestion Liquors, Relying on Two Titration Points and an External Inorganic Carbon Analysis. Chem. Eng. 2021, 5, 15. [Google Scholar] [CrossRef]
- Thorne, P.S.; Duchaine, C.; Douwes, J.; Eduard, W.; Gorny, R.; Jacobs, R.; Reponen, T.; Schierl, R.; Szponar, B. Working Group Report 4: Exposure assessment for biological agents. Am. J. Ind. Med. 2004, 46, 419–422. [Google Scholar] [CrossRef]
- European Agency for Safety and Health at Work (EU-OSHA). Expert Forecast on Emerging Biological Risks Related to Occupational Safety and Health. European risk observatory report; European Agency for Safety and Health at Work (EU-OSHA): Bilbao, Spain, 2007; pp. 1–145. ISBN 1830-5946. [Google Scholar]
- Walsera, S.M.; Gerstnera, D.G.; Brennera, B.; Büngerb, J.; Eikmannc, T.; Janssena, B.; Kolba, S.; Kolkd, A.; Nowake, D.; Raulfb, M.; et al. Evaluation of exposure–response relationships for health effects of microbial bioaerosols—A systematic review. Int. J. Hyg. Environ. Health 2015, 218, 577–589. [Google Scholar] [CrossRef]
- Bruijn, F.J. (Ed.) Stress and Environmental Regulation of Gene Expression and Adaptation in Bacteria. I&II; Wiley-Blackwell: Hoboken, NJ, USA, 2016; pp. 1–1442. [Google Scholar]
- Braga, R.M.; Nóbrega Dourado, M.; Araújo, W.L. Microbial interactions: Ecology in a molecular perspective. Braz. J. Microbiol. 2016, 47S, 86–98. [Google Scholar] [CrossRef]
- Rolain, J.M.; Canton, R.; Cornaglia, G. Emergence of antibiotic resistance: Need for a new paradigm. Clin. Microbiol. Infect. 2012, 18, 615–616. [Google Scholar] [CrossRef]
- Pietrangeli, B.; Lauri, R. Biogas production plants. A methodological approach for occupational health and safety im-provement. In Advances in Biofuel and Bioenergy; Nageswara-Rao, M., Ed.; IntechOpen: London, UK, 2018; pp. 183–200. ISBN 978-1-78923-286-8. [Google Scholar]
- Occupational Health and Safety Assessment Serie (OHSAS). 18001:2007 Occupational Health and Safety MANAGEMENT systems—Requirements; OHSAS Project Group: London, UK, 2007; ISBN 978 0 580 50802 8. [Google Scholar]
- UNI ISO 45001:2018; Sistemi di Gestione per la Salute e Sicurezza sul Lavoro—Requisiti e Guida Per l’uso. ISO: Geneva, Switzerland, 2018.
- Schlosser, O.; Huyard, A.; Catalan, V. Exposure to dust, endotoxin and airborne microorganisms in wastewater sludge thermal drying units. Water Sci. Technol. 2011, 64, 1073–1080. [Google Scholar] [CrossRef]
- Douwes, J.; Thorne, P.; Pearce, N.; Heederik, D. Bioaerosol health effects and exposure assessment: Progress and prospects. Ann. Occup. Hyg. 2003, 47, 187–200. [Google Scholar] [PubMed]
- Stagg, S.; Bowry, A.; Kelsey, A.; Crook, B. Bioaerosols from waste composting and the potential for workers’ exposure. In Health and Safety Executive; Research Report N. 786; HSE Books: Norwich, UK, 2010; pp. 1–107. [Google Scholar]
- Douwes, J.; Mannetje, A.; Heederik, D. Work-related symptoms in sewage treatment workers. Ann. Agric. Environ. Med. 2001, 8, 39–45. [Google Scholar]
- Smit, L.A.M.; Spaan, S.; Heederik, D. Endotoxin exposure and symptoms in wastewater treatment workers. Am. J. Ind. Med. 2005, 48, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Rylander, R. Health effects among workers in sewage treatment plants. Occup. Environ. Med. 1999, 56, 354–357. [Google Scholar] [CrossRef] [PubMed]


| Process Unit | ||||||
|---|---|---|---|---|---|---|
| process parameter | ||||||
| guide word 1 | ||||||
| guide word 2 | ||||||
| guide word n | ||||||
| Process Unit | |||||||
|---|---|---|---|---|---|---|---|
| Engineering process | Biotechnology process | Existing counter measures | Biohazard | Proposed counter measures | |||
| causes | consequences | causes | consequences | ||||
| process parameter | |||||||
| deviation 1 | |||||||
| deviation 2 | |||||||
| deviation n | |||||||
| HRT | Window Time (d) | Feedstock | T (°C) | pH |
|---|---|---|---|---|
| 1 | 0.5 | VFA-rich stream | 25–28 | uncontrolled |
| Run | HRT (d) | SRT (d) | OLR (g COD/L d) | CL (d) | SRT/CL (d/d) | Feeding Frequency (d−1) | Operation Lengt (d) | Load per Cycle (g COD/L) |
|---|---|---|---|---|---|---|---|---|
| Ae3 | 1 | 1 | 4 | 0.5 | 2 | 2 | 45 | 20 |
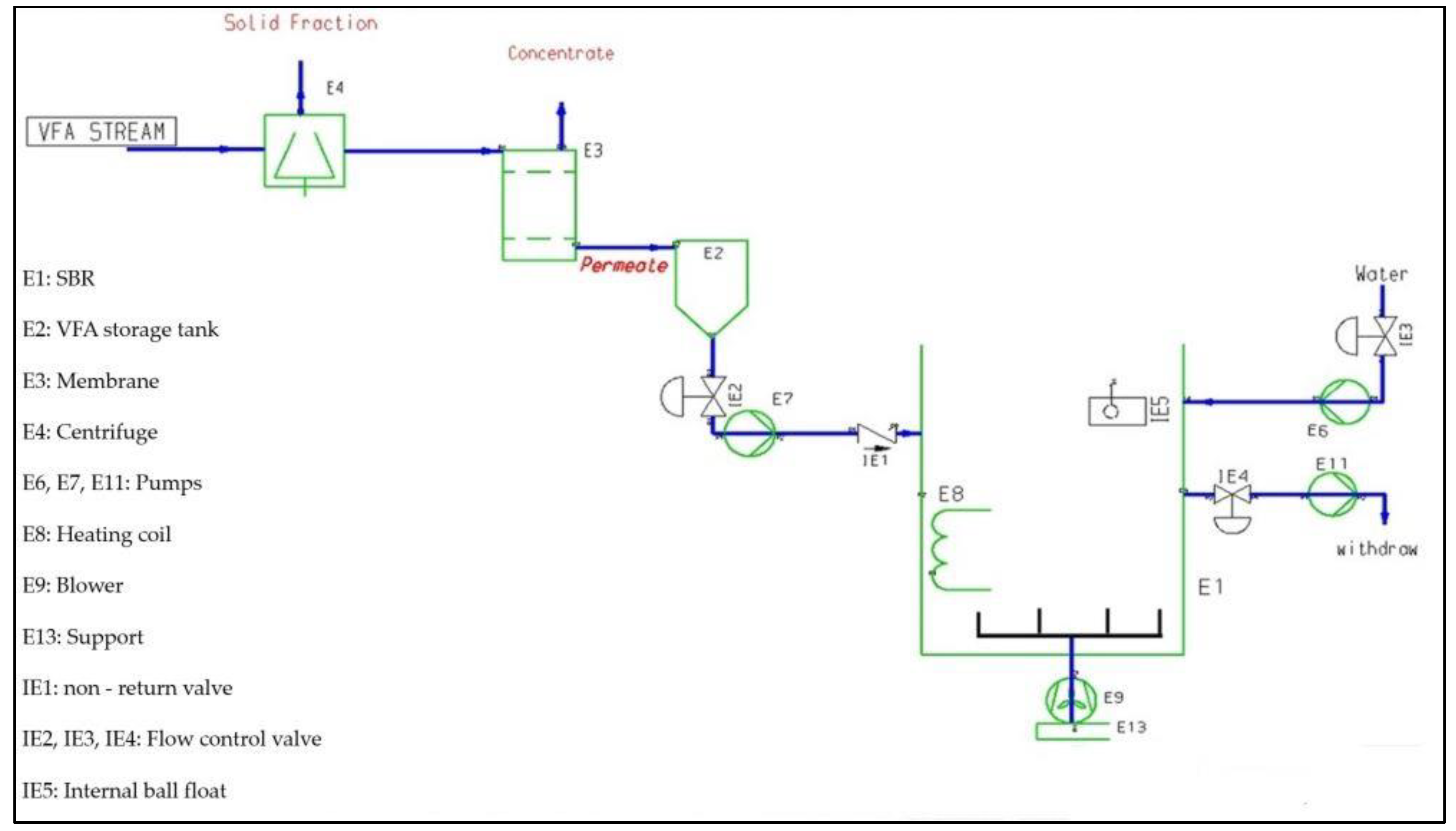
| The activated sludge from the full-scale plant is used as inoculum for the SBR (aerobic PHA microorganisms selection reactor), and the biomass was selected using an aerobic dynamic feeding strategy (feast–famine regime). Activated sludge is automatically fed to the SBR, a 100 L working-volume reactor. The dissolved oxygen (DO) concentration is maintained at a maximum of 8.0 mg O2/L with linear membrane blowers, which also allow the complete stirring of the mixed liquor. The DO concentration, oxidation reduction potential (ORP), pH and temperature are constantly monitored in real time by immersion probes and online signals are acquired through a programmable logic controller (PLC). The SBR temperature is regulated by an immersion heater and maintained between 25 °C and 28 °C. SBR operating cycles were automated and controlled by the PLC. The mixture inside the SBR contains:
The process equipment includes drains (sampling points). The process does not work in or near the flammability range. Further detailed information has been acquired with respect to the following: |
| -BIOHAZARD: it is connected with PHA producing microorganisms enriched in the SBR. The microbial consortium, enriched in the SBR growth conditions, includes many microbial genera. The microbial characterization has identified about 200 bacterial genera as reported in Crognale et al. [18], some of which may contain species assigned to the risk group 2 (low pathogenicity), according to the European classification (Annex III of Directive 2000/54/EC [13] as amended by Directives 2010/1833 [19] and 2020/739 [20]). The exposure routes to the biological agents present in the SBR include:
|
-TOXICITY and ECOTOXICITY: the toxic agents in main quantity are CH₃COOH (acetic acid, CAS No. 64-19-7) and CH3CH2CH2COOH (butyric acid, CAS No. 107-92-6) having potential health effects both acute and chronic. The substances entry routes are inhalation, skin contact, and eye contact. Specific toxic effects are associated with each substance. For example, the main toxic effects of acetic acid are:
|
| -FLAMMABILITY and EXPLOSIVITY: the SBR does not contain flammable gaseous mixtures. |
| Operating procedure |
| There is no specific written procedure to maintain the on-going integrity of the process equipment. Nevertheless, good operating practices have been applied to plants operations, such as cleaning of coaxial centrifuge and ultrafiltration membranes, and taking samples for laboratory analysis. |
| Plant layout |
| There are buffer zones between the plant and the external public (population). The pilot plant operators are potentially exposed to hazards from the wastewater treatment plant within which the PHA production process takes place. The OFMSW transfer to digester is carried out under containment conditions and therefore it has no impact on the environment and operators. The workplace layout includes the location of control rooms, laboratories and offices, drainage areas and sampling points. |
| Emergency/ongoing program |
| A number of steps in the production process are managed by programmable logic controller (PLC). There is not any system, which ensures the plant is currently kept and periodically tested. |
| Management—Process Hazard Analysis (PHA) |
No risk analysis techniques other than BioHazOp analysis (FTA, FMEA, What if) have been applied to the plant.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lauri, R.; Incocciati, E.; Pietrangeli, B.; Tayou, L.N.; Valentino, F.; Gottardo, M.; Majone, M. Hazop Analysis of a Bioprocess for Polyhydroxyalkanoate (PHA) Production from Organic Waste: Part B. Fermentation 2023, 9, 154. https://doi.org/10.3390/fermentation9020154
Lauri R, Incocciati E, Pietrangeli B, Tayou LN, Valentino F, Gottardo M, Majone M. Hazop Analysis of a Bioprocess for Polyhydroxyalkanoate (PHA) Production from Organic Waste: Part B. Fermentation. 2023; 9(2):154. https://doi.org/10.3390/fermentation9020154
Chicago/Turabian StyleLauri, Roberto, Emma Incocciati, Biancamaria Pietrangeli, Lionel Nguemna Tayou, Francesco Valentino, Marco Gottardo, and Mauro Majone. 2023. "Hazop Analysis of a Bioprocess for Polyhydroxyalkanoate (PHA) Production from Organic Waste: Part B" Fermentation 9, no. 2: 154. https://doi.org/10.3390/fermentation9020154
APA StyleLauri, R., Incocciati, E., Pietrangeli, B., Tayou, L. N., Valentino, F., Gottardo, M., & Majone, M. (2023). Hazop Analysis of a Bioprocess for Polyhydroxyalkanoate (PHA) Production from Organic Waste: Part B. Fermentation, 9(2), 154. https://doi.org/10.3390/fermentation9020154








