Kombucha Healthy Drink—Recent Advances in Production, Chemical Composition and Health Benefits
Abstract
1. Introduction
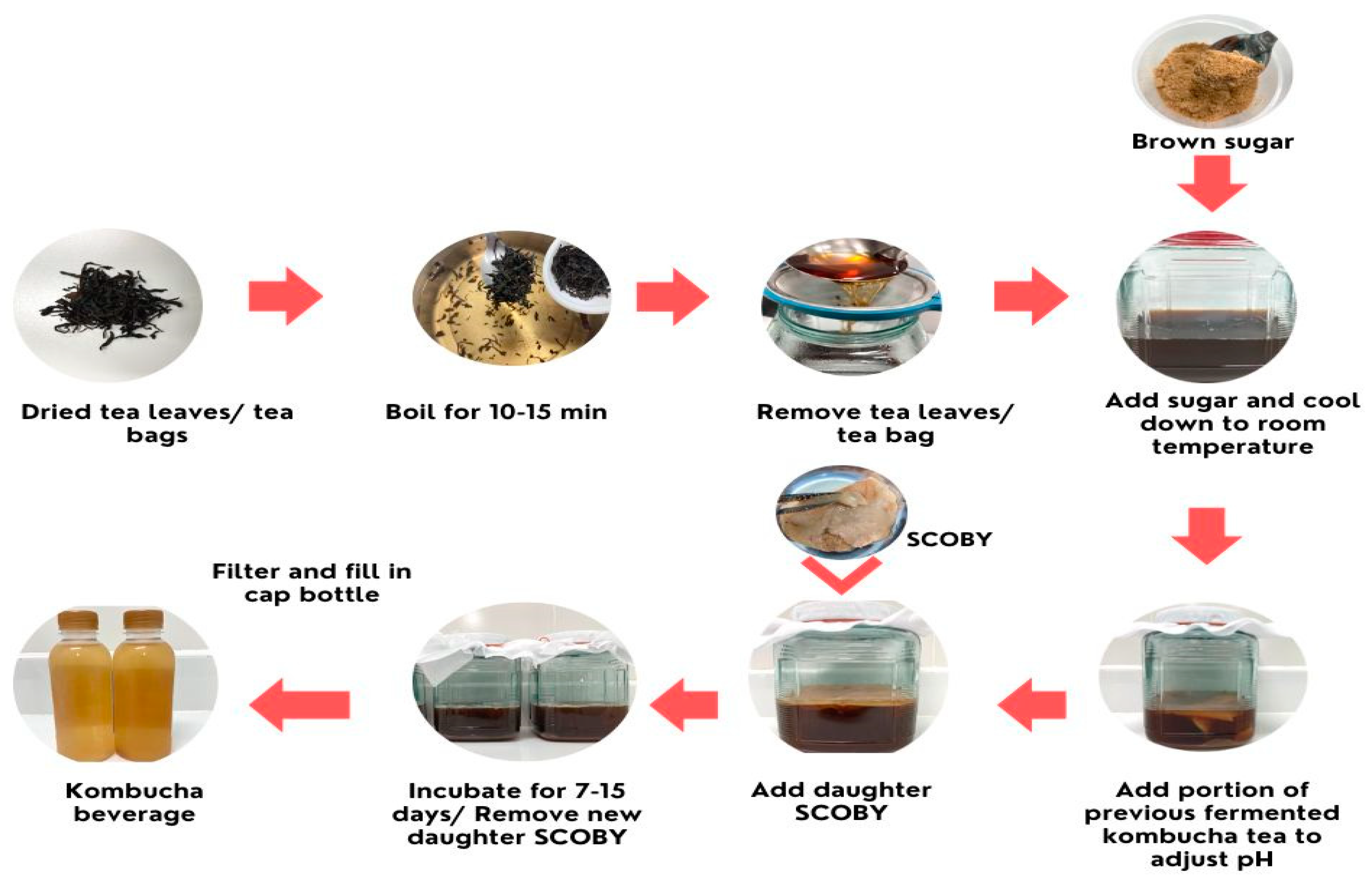
2. Kombucha Production
3. Microorganisms Involved in Kombucha Fermentation
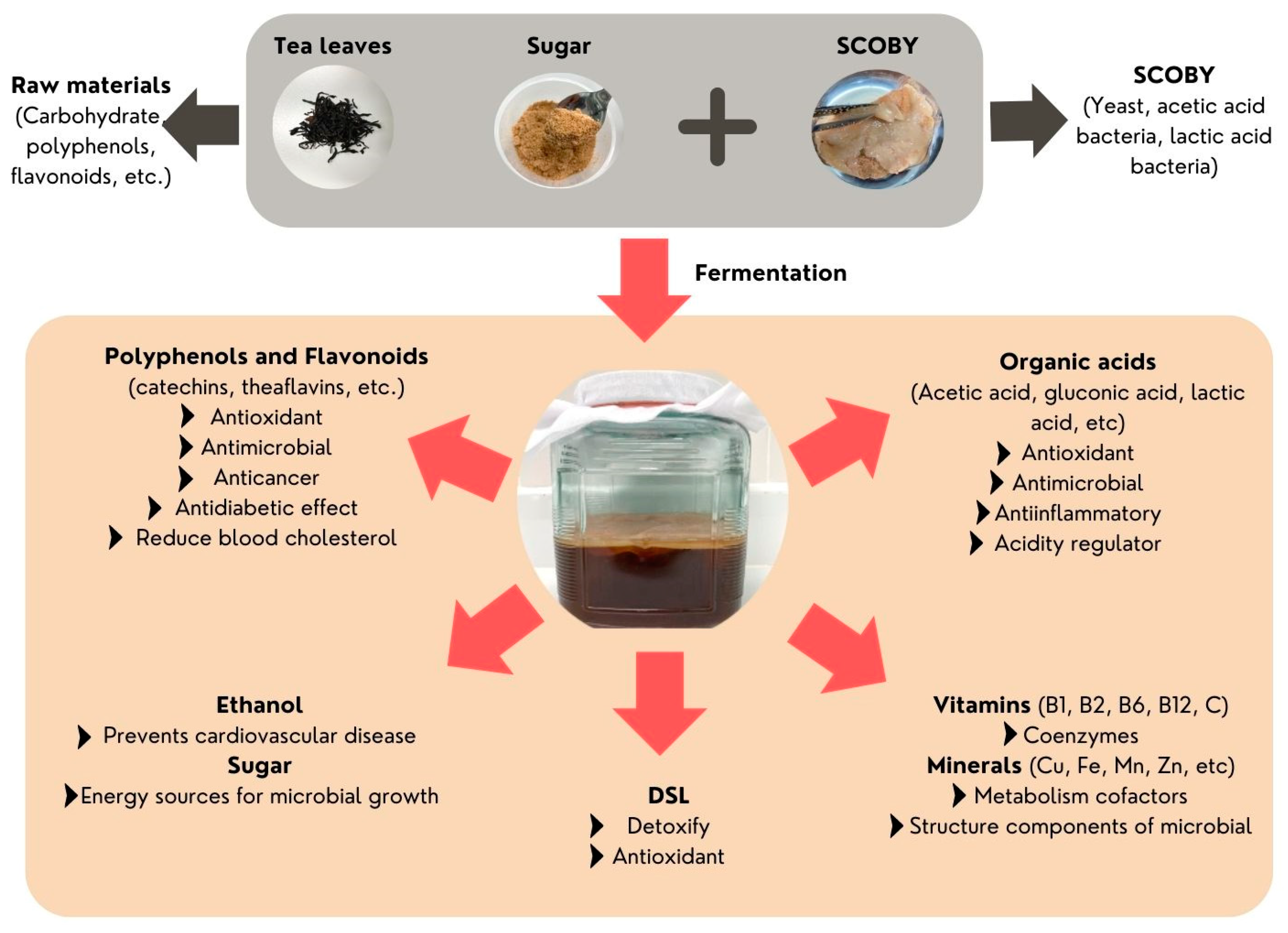
4. Chemical Compositions of Kombucha
| Component | Chemical | References |
|---|---|---|
| Organic acids | acetic acid, lactic acid, citric acid, gluconic acid, glucuronic acid, malic acid, tartaric acid, succinic acid, pyruvic acid, oxalic acid, butyric acid | [31,43,73,74] |
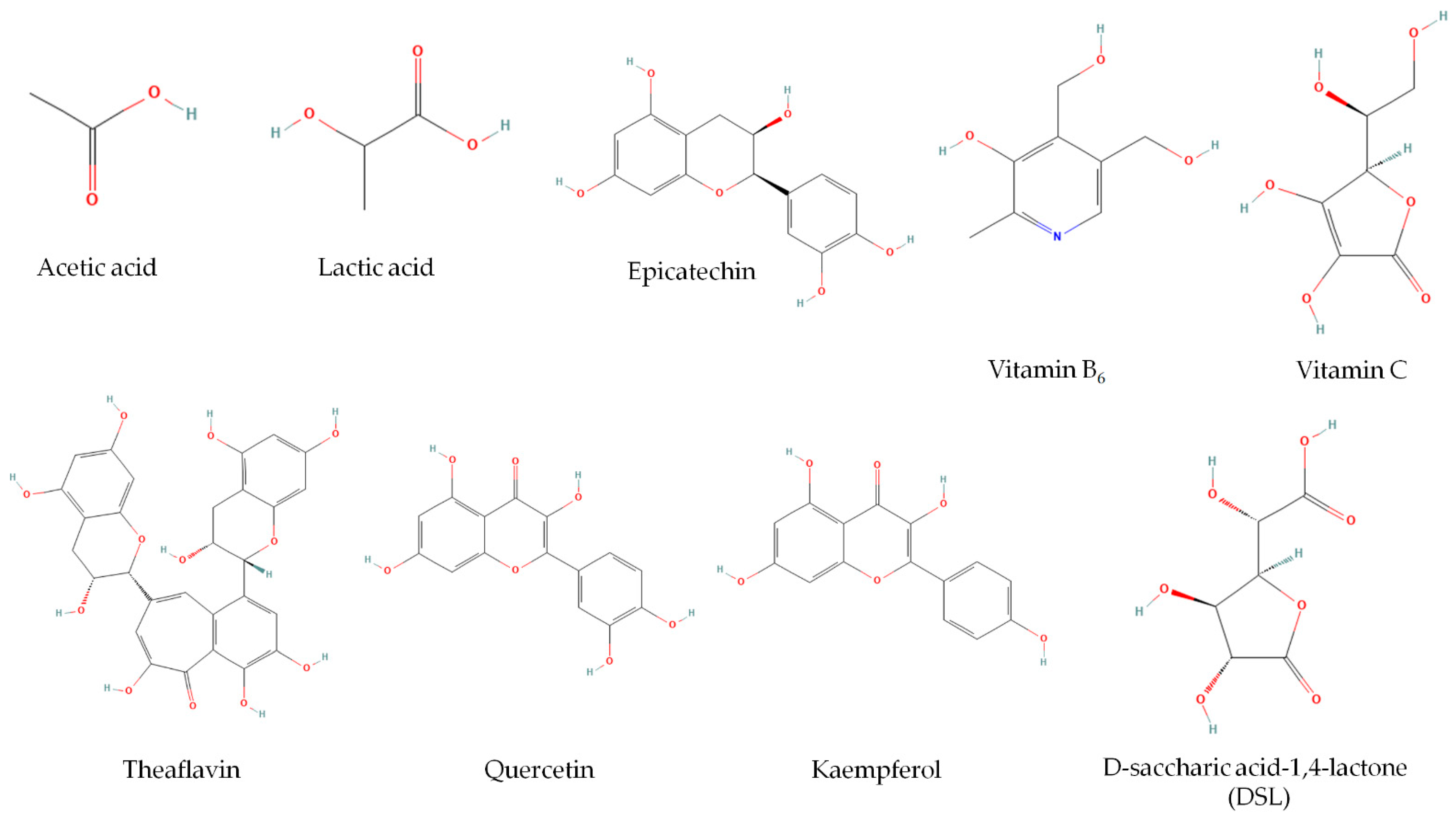
| Polyphenols | catechins [α-epicatechin, α-epicatechin-3-gallate (ECG), α-epigallocatechin (EGC), α-epigallocatechin-3-gallate (EGCG), α-gallocatechin, β-catechin], caffeine, theaflavin, thearubigins, gallotannins | [73,75,76] |
| Vitamins | vitamin B1, B2, B6, B9, B12, C | [74,77,78] |
| Amino acids | isoleucine, leucine, lysine, methionine, phenylalanine, threonine, valine, tryptophan, alanine, arginine, aspartic acid, cysteine, glutamic acid, glycine, histidine, proline, serine, tyrosine | [79] |
| Proteins/organic nitrogen/hydrolytic enzymes | peptides, biogenic amines (ethylamine, choline, adenine, histamine tyramine, putrescine, cadaverine, β-phenylethylamine, tryptamine, spermidine, spermine), proteinase, peptidase, catalase | [80,81,82] |
| Minerals/anions | manganese, iron, nickel, copper, zinc, cobalt, nitrate, phosphate, sulfate, bromide, chloride, iodide, fluoride | [77,83,84] |
| Alcohol | Ethanol | [85] |
| Other substances | Flavonoids (quercetin, kaempferol, myricetin, rutin), alkaloids, purines, lipids, D-saccharic acid-1,4-lactone (DSL), carbon dioxide | [4,7] |
5. Health Benefits of Kombucha
5.1. Antioxidant Activity
5.2. Antimicrobial Activity
5.3. Anti-inflammatory and Anticancer Activities
5.4. Other Benefits
6. Adverse Effects of Kombucha
7. Clinical Trials of Kombucha Health Benefits
8. Conclusions and Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, B.; Rutherfurd-Markwick, K.; Zhang, X.X.; Mutukumira, A.N. Kombucha: Production and microbiological research. Foods 2022, 11, 3456. [Google Scholar] [CrossRef]
- Srihari, T.; Satyanarayana, U. Changes in free radical scavenging activity of kombucha during fermentation. J. Pharm. Sci. Res. 2012, 4, 1978–1981. [Google Scholar]
- Sreeramulu, G.; Zhu, Y.; Knol, W. Kombucha fermentation and its antimicrobial activity. J. Agric. Food Chem. 2000, 48, 2589–2594. [Google Scholar] [CrossRef]
- Jayabalan, R.; Malbaša, R.V.; Loncar, E.S.; Vitas, J.S.; Sathishkumar, M. A review on kombucha tea–microbiology, composition, fermentation, beneficial effects, toxicity, and tea fungus. Compr. Rev. Food Sci. 2014, 13, 538–550. [Google Scholar] [CrossRef] [PubMed]
- Dutta, H.; Paul, S.K. Kombucha drink: Production, quality, and safety aspects. In Production and Management of Beverages; Grumezescu, A.M., Holban, A.M., Eds.; Woodhead Publishing: Sawston, UK, 2019; pp. 259–288. [Google Scholar]
- Kim, J.; Adhikari, K. Current trends in kombucha: Marketing perspectives and the need for improved sensory research. Beverages 2020, 6, 15. [Google Scholar] [CrossRef]
- Sen, G.; Sarkar, N.; Nath, M.; Maity, S. Bioactive components of tea. Arch. Food Nutr. Sci. 2020, 4, 001–009. [Google Scholar]
- Villarreal-Soto, S.A.; Beaufort, S.; Bouajila, J.; Souchard, J.P.; Taillandier, P. Understanding kombucha tea fermentation: A review. J. Food Sci. 2018, 83, 580–588. [Google Scholar] [CrossRef]
- Leal, J.M.; Suárez, L.V.; Jayabalan, R.; Oros, J.H.; Escalante-Aburto, A. A review on health benefits of kombucha nutritional compounds and metabolites. CYTA—J. Food 2018, 16, 390–399. [Google Scholar] [CrossRef]
- Applegate, K.B.; Cheek, P.R.; Inlow, J.K. Analysis of kombucha to teach biochemical concepts and techniques to undergraduate students. Biochem. Mol. Biol. Educ. 2019, 47, 459–467. [Google Scholar] [CrossRef]
- Chakravorty, S.; Bhattacharya, S.; Bhattacharya, D.; Sarkar, S.; Gachhui, R. Kombucha: A promising functional beverage prepared from tea. In Nonalcoholic Beverages; Grumezescu, A.M., Holban, A.M., Eds.; Woodhead Publishing: Sawston, UK, 2019; pp. 285–327. [Google Scholar]
- Villarreal-Soto, S.A.; Beaufort, S.; Bouajila, J.; Souch, J.P.; Renard, T.; Rollan, S.; Taillandier, P. Impact of fermentation conditions on the production of bioactive compounds with anticancer, anti-inflammatory and antioxidant properties in kombucha tea extracts. Process Biochem. 2019, 83, 44–54. [Google Scholar] [CrossRef]
- Leal, J.M.; Garcia, N.P.; Escalante-Aburto, A. Recent evidence of the beneficial effects associated with glucuronic acid contained in kombucha beverages. Curr. Nutr. Rep. 2020, 9, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Kaewkod, T.; Bovonsombut, S.; Tragoolpua, Y. Efficacy of kombucha obtained from green, oolong, and black teas on inhibition of pathogenic bacteria, antioxidation, and toxicity on colorectal cancer cell line. Microorganisms 2019, 7, 700. [Google Scholar] [CrossRef] [PubMed]
- Jakubczyk, K.; Kalduñska, J.; Kochman, J.; Janda, K. Chemical profile and antioxidant activity of the kombucha beverage derived from white, green, black and red tea. Antioxidants 2020, 9, 447. [Google Scholar] [CrossRef]
- Watawana, M.I.; Jayawardena, N.; Gunawardhana, C.B.; Waisundara, V.Y. Health, wellness, and safety aspects of the consumption of kombucha. J. Chem. 2015, 2015, 591869. [Google Scholar] [CrossRef]
- Coton, M.; Pawtowski, A.; Taminiau, B.; Burgaud, G.; Deniel, F.; Coulloumme-Labarthe, L.; Fall, A.; Daube, G.; Coton, E. Unraveling microbial ecology of industrial-scale kombucha fermentations by metabarcoding and culture-based methods. FEMS Microbiol. Ecol. 2017, 1, 93. [Google Scholar] [CrossRef]
- de Oliveira, I.A.C.L.; Rolim, V.A.O.; Gaspar, R.P.L.; Rossini, D.Q.; de Souza, R.; Bogsan, C.S.B. The technological perspectives of kombucha and its implications for production. Fermentation 2022, 8, 185. [Google Scholar] [CrossRef]
- Villarreal-Soto, S.A.; Bouajila, J.; Pace, M.; Leech, J.; Cotter, P.D.; Souchard, J.P.; Taillandier, P.; Beaufort, S. Metabolome-microbiome signatures in the fermented beverage, kombucha. Int. J. Food Microbiol. 2020, 333, 108778. [Google Scholar] [CrossRef]
- Ivanišová, E.; Menhartová, K.; Terentjeva, M.; Harangozo, L.; Kántor, A.; Kacaniová, M. The evaluation of chemical, antioxidant, antimicrobial and sensory properties of kombucha tea beverage. J. Food Sci. Technol. 2020, 57, 1840–1846. [Google Scholar] [CrossRef]
- Gaggia, F.; Baffoni, L.; Galiano, M.; Nielsen, D.S.; Jakobsen, R.R.; Castro-Mejia, J.L.; Bosi, S.; Truzzi, F.; Musumeci, F.; Dinelli, G.; et al. Kombucha beverage from green, black and rooibos teas: A comparative study looking at microbiology, chemistry and antioxidant activity. Nutrients 2018, 11, 1. [Google Scholar] [CrossRef]
- Ahmed, R.F.; Hikal, M.S.; Abou-Taleb, K.A. Biological, chemical and antioxidant activities of different types kombucha. Ann. Agric. Sci. 2020, 65, 35–41. [Google Scholar] [CrossRef]
- Salvadó, Z.; Arroyo-López, F.N.; Guillamón, J.M.; Salazar, G.; Querol, A.; Barrio, E. Temperature adaptation markedly determines evolution within the genus Saccharomyces. Appl. Environ. Microbiol. 2011, 77, 2292–2302. [Google Scholar] [CrossRef] [PubMed]
- Mamlouk, D.; Gullo, M. Acetic acid bacteria: Physiology and carbon sources oxidation. Indian J. Microbiol. 2013, 53, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Matejceková, Z.; Liptáková, D.; Spodniaková, S.; Valik, L. Characterization of the growth of Lactobacillus plantarum in milk in dependence on temperature. Acta Chim. Slovaca 2016, 9, 104–108. [Google Scholar] [CrossRef]
- Thavasi, V.; Bettens, R.P.A.; Leong, L.P. Temperature and solvent effects on radical scavenging ability of phenols. J. Phys. Chem. A 2009, 113, 3068–3077. [Google Scholar] [CrossRef]
- De Filippis, F.; Troise, A.D.; Vitaglione, P.; Ercolini, D. Different temperatures select distinctive acetic acid bacteria species and promotes organic acids production during kombucha tea fermentation. Food Microbiol. 2018, 73, 11–16. [Google Scholar] [CrossRef]
- Hur, S.J.; Lee, S.Y.; Kim, Y.C.; Choi, I.; Kim, G.B. Effect of fermentation on the antioxidant activity in plant-based foods. Food Chem. 2014, 160, 346–356. [Google Scholar] [CrossRef]
- Šaponjac, V.T.T.; Vulic, J.J. Antioxidant and antibacterial activity of the beverage obtained by fermentation of sweetened lemon balm (Melissa officinalis L.) tea with symbiotic consortium of bacteria and yeasts. Food Technol. Biotechnol. 2014, 52, 420–429. [Google Scholar]
- Yavari, N.; Assadi, M.M.; Larijani, K.; Moghadam, M.B. Response surface methodology for optimization of glucuronic acid production using kombucha layer on sour cherry juice. Aust. J. Basic Appl. Sci. 2010, 4, 3250–3256. [Google Scholar]
- Yavari, N.; Assadi, M.M.; Moghadam, M.B.; Larijani, K. Optimizing glucuronic acid production using tea fungus on grape juice by response surface methodology. Aust. J. Basic Appl. Sci. 2011, 5, 1788–1794. [Google Scholar]
- Battikh, H.; Bakhrouf, A.; Ammar, E. Antimicrobial effect of kombucha analogues. LWT—Food Sci. Technol. 2012, 47, 71–77. [Google Scholar] [CrossRef]
- Sun, T.Y.; Li, J.S.; Chen, C. Effects of blending wheatgrass juice on enhancing phenolic compounds and antioxidant activities of traditional kombucha beverage. J. Food Drug Anal. 2015, 23, 709–718. [Google Scholar] [CrossRef] [PubMed]
- Watawana, M.I.; Jayawardena, N.; Gunawardhana, C.B.; Waisundara, V.Y. Enhancement of the antioxidant and starch hydrolase inhibitory activities of king coconut water (Cocos nucifera var. Aurantiaca) by fermentation with kombucha “tea fungus”. Int. J. Food Sci. Technol. 2016, 51, 490–498. [Google Scholar] [CrossRef]
- Pure, A.E.; Pure, M.E. Antioxidant and antibacterial activity of kombucha beverages prepared using banana peel, common nettles and black tea infusions. Appl. Food Biotechnol. 2016, 3, 125–130. [Google Scholar]
- Pure, A.E.; Pure, M.E. Antioxidant, antibacterial and color analysis of garlic fermented in kombucha and red grape vinegar. Appl. Food Biotechnol. 2016, 3, 246–252. [Google Scholar]
- Vazquez-Cabral, B.D.; Larrosa-Pérez, M.; Gallegos-Infante, J.A.; Moreno-Jiménez, M.R.; González-Laredo, R.F.; Rutiaga-Quiñones, J.G.; Gamboa-Gómez, C.I.; Rocha-Guzmán, N.E. Oak kombucha protects against oxidative stress and inflammatory processes. Chem. Biol. Interact. 2017, 272, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Jiménez, M.R.; Rocha-Guzmán, N.E.; Rutiaga-Quiñones, G.J.; Medrano-Núñez, D.; Rojas-Contreras, J.A.; González-Laredo, R.F.; Gallegos-Infante, J.A. Polyphenolic profile, sugar consumption and organic acids generation along fermentation of infusions from guava (Pisidium guajava) by the kombucha consortium. Recent Res. Sci. Technol. 2018, 10, 16–22. [Google Scholar]
- Degirmencioglu, N.; Yildiz, E.; Guldas, M.; Gurbuz, O. Health benefits of kombucha tea enriched with olive leaf and honey. J. Obes. Chronic Dis. 2020, 4, 1–5. [Google Scholar] [CrossRef]
- Agnolucci, M.; Tirelli, A.; Cocolin, L.; Toffanin, A. Brettanomyces bruxellensis yeasts: Impact on wine and winemaking. World J. Microbiol. Biotechnol. 2017, 33, 180. [Google Scholar] [CrossRef]
- Loira, I.; Morata, A.; Palomero, F.; González, C.; Suárez-Lepe, J.A. Schizosaccharomyces pombe: A promising biotechnology for modulating wine composition. Fermentation 2018, 4, 70. [Google Scholar] [CrossRef]
- Laureys, D.; Britton, S.J.; De Clippeleer, J. Kombucha tea fermentation: A review. J. Am. Soc. Brew. Chem. 2020, 78, 165–174. [Google Scholar] [CrossRef]
- Yang, Z.; Zhou, F.; Ji, B.; Li, B.; Luo, Y.; Yang, L.; Li, T. Symbiosis between microorganisms from kombucha and kefir: Potential significance to the enhancement of kombucha function. Appl. Biochem. Biotechnol. 2010, 160, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Dufresne, C.; Farnworth, E. Tea, kombucha, and health: A review. Food Res. Int. 2000, 33, 409–421. [Google Scholar] [CrossRef]
- Sievers, M.; Lanini, C.; Weber, A.; Schuler-Schmid, U.; Teuber, M. Microbiology and fermentation balance in kombucha beverage obtained from a tea fungus fermentation. Syst. Appl. Microbiol. 1995, 18, 590–594. [Google Scholar] [CrossRef]
- Zhao, Z.J.; Sui, Y.C.; Wu, H.W.; Zhou, C.B.; Hu, X.C.; Zhang, J. Flavour chemical dynamics during fermentation of kombucha tea. Emir. J. Food Agric. 2018, 30, 732–741. [Google Scholar]
- Goh, W.N.A.; Rosma, A.; Kaur, B.; Fazilah, B.; Karim, A.A.; Bhat, R. Fermentation of black tea broth (kombucha): I. Effects of sucrose concentration and fermentation time on yield of microbial cellulose. Int. Food Res. J. 2012, 19, 109–117. [Google Scholar]
- Boesch, T.; Trcek, J.; Sievers, M.; Teuber, M. Acetobacter intermedius, sp. nov. Syst. Appl. Microbiol. 1998, 21, 220. [Google Scholar] [CrossRef]
- Harrison, K.; Curtin, C. Microbial composition of SCOBY starter cultures used by commercial kombucha brewers in North America. Microorganisms 2021, 9, 1060. [Google Scholar] [CrossRef]
- Dutta, D.; Gachhui, R. Novel nitrogen-fixing Acetobacter nitrogenifigens sp. nov., isolated from kombucha tea. Int. J. Syst. Evol. Microbiol. 2006, 56, 1899–1903. [Google Scholar] [CrossRef]
- Dutta, D.; Gachhui, R. Nitrogen-fixing and cellulose-producing Gluconacetobacter kombuchae sp. nov., isolated from kombucha tea. Int. J. Syst. Evol. Microbiol. 2007, 57, 353–357. [Google Scholar] [CrossRef]
- Marsh, A.J.; O’Sullivan, O.; Hill, C.; Ross, R.P.; Cotter, P.D. Sequence-based analysis of the bacterial and fungal compositions of multiple kombucha (tea fungus) samples. Food Microbiol. 2014, 38, 171–178. [Google Scholar] [CrossRef]
- Yang, J.; Lagishetty, V.; Kurnia, P.; Henning, S.M.; Ahdoot, A.I.; Jacobs, J.P. Microbial and chemical profiles of commercial kombucha products. Nutrients 2022, 14, 670. [Google Scholar] [CrossRef] [PubMed]
- Pei, J.; Jin, W.; Abd El-Aty, A.; Baranenko, D.A.; Gou, X.; Zhang, H.; Geng, J.; Jiang, L.; Chen, D.; Yue, T. Isolation, purification, and structural identification of a new bacteriocin made by Lactobacillus plantarum found in conventional kombucha. Food Control 2020, 110, 106923. [Google Scholar] [CrossRef]
- Safak, S.; Mercan, N.; Aslim, B.; Beyatli, Y. A study on the production of poly-beta-hydroxybutyrate by some eukaryotic microorganisms. Turk. Electron. J. Biotechnol. 2002, 11–17. [Google Scholar]
- Markov, S.L.; Malbaša, R.V.; Hauk, M.J.; Cvetkovic, D.D. Investigation of tea fungus microbe associations. I. The yeast. Acta Period. Technol. 2001, 32, 133–138. [Google Scholar]
- Ramadani, A.S.; Abulreesh, H.H. Isolation and identification of yeast flora in local kombucha sample: Al Nabtah. Umm Al-Qura Univ. J. App. Sci. 2010, 2, 42–51. [Google Scholar]
- Teoh, A.L.; Heard, G.; Cox, J. Yeast ecology of kombucha fermentation. Int. J. Food Microbiol. 2004, 95, 119–126. [Google Scholar] [CrossRef]
- Herrera, T.; Calderon-Villagomez, A. Species of yeasts isolated in Mexico from the tea fungus. Rev. Mex. Micol. 1989, 5, 205–210. [Google Scholar]
- Bellut, K.; Michel, M.; Zarnkow, M.; Hutzler, M.; Jacob, F.; De Schutter, D.P.; Daenen, L.; Lynch, K.M.; Zannini, E.; Arendt, E.K. Application of non-Saccharomyces yeasts isolated from kombucha in the production of alcohol-free beer. Fermentation 2018, 4, 66. [Google Scholar] [CrossRef]
- Kurtzman, C.P.; Robnett, C.J.; Basehoar-Powers, E. Zygosaccharomyces kombuchaensis, a new ascosporogenous yeast from “Kombucha tea”. FEMS Yeast Res. 2001, 1, 133–138. [Google Scholar] [CrossRef]
- Jayabalan, R.; Subathradevi, P.; Marimuthu, S.; Sathishkumar, M.; Swaminathan, K. Changes in free-radical scavenging ability of kombucha tea during fermentation. Food Chem. 2008, 109, 227–234. [Google Scholar] [CrossRef]
- Tu, C.; Tang, S.; Azi, F.; Hu, W.; Dong, M. Use of kombucha consortium to transform soy whey into a novel functional beverage. J. Funct. Foods. 2019, 52, 81–89. [Google Scholar] [CrossRef]
- Reva, O.N.; Zaets, I.E.; Ovcharenko, L.P.; Kukharenko, O.E.; Shpylova, S.P.; Podolich, O.V.; de Vera, J.P.; Kozyrovska, N.O. Metabarcoding of the kombucha microbial community grown in different microenvironments. AMB Express 2015, 5, 124. [Google Scholar] [CrossRef] [PubMed]
- Andrés-Barrao, C.; Barja, F.; Pérez, R.O.; Chappuis, M.L.; Braito, S. Identification techniques of acetic acid bacteria: Comparison between MALDI-TOF MS and molecular biology technique. In Acetic Acid Bacteria; CRC Press: Boca Raton, FL, USA, 2017; pp. 162–192. [Google Scholar]
- Shade, A.; Caporaso, G.; Handelsman, J.; Knight, R.; Fierer, N. A meta-analysis of changes in bacterial and archaeal communities with time. ISME J. 2013, 7, 1493–1506. [Google Scholar] [CrossRef]
- Chakravorty, S.; Bhattacharya, S.; Chatzinotas, A.; Chakraborty, W.; Bhattacharya, D.; Gachhui, R. Kombucha tea fermentation: Microbial and biochemical dynamics. Int. J. Food Microbiol. 2016, 220, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Arikan, M.; Mitchell, A.; Finn, R.D.; Gürel, F. Microbial composition of kombucha determined using amplicon sequencing and shotgun metagenomics. J. Food Sci. 2020, 85, 455–464. [Google Scholar] [CrossRef]
- De Filippis, F.; Parente, E.; Ercolini, D. Metagenomics insights into food fermentations. Microb. Biotechnol. 2017, 10, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Unban, K.; Khatthongngam, N.; Pattananandecha, T.; Saenjum, C.; Shetty, K.; Khanongnuch, C. Microbial community dynamics during the non-filamentous fungi growth-based fermentation process of miang, a traditional fermented tea of north Thailand and their product characterizations. Front. Microbiol. 2020, 11, 1515. [Google Scholar] [CrossRef] [PubMed]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Elsevier: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Jayabalan, R.; Marimuthu, S.; Swaminathan, K. Changes in content of organic acids and tea polyphenols during kombucha tea fermentation. Food Chem. 2007, 102, 392–398. [Google Scholar] [CrossRef]
- Malbasa, R.V.; Loncar, E.S.; Vitas, J.S.; Canadanovic-Brunet, J.M. Influence of starter cultures on the antioxidant activity of kombucha beverage. Food Chem. 2011, 127, 1727–1731. [Google Scholar] [CrossRef]
- Morshedi, A.; Dashti-Rahmatabadi, M.H. Chronic consumption of kombucha and black tea prevents weight loss in diabetic rats. Iran. J. Diabetes Obes. 2010, 2, 23–26. [Google Scholar]
- Sharma, V.K.; Bhattacharya, A.; Kumar, A.; Sharma, H.K. Health benefits of tea consumption. Trop. J. Pharm. Res. 2007, 6, 785–792. [Google Scholar] [CrossRef]
- Bauer-Petrovska, B.; Petrushevska-Tozi, L. Mineral and water soluble vitamin content in the kombucha drink. Int. J. Food Sci. Technol. 2000, 35, 201–205. [Google Scholar] [CrossRef]
- Vitas, J.S.; Malbasa, R.V.; Grahovac, J.A.; Loncar, E.S. The antioxidant activity of kombucha fermented milk products with stinging nettle and winter savory. Chem. Ind. Chem. Eng. Q. 2013, 19, 129–139. [Google Scholar] [CrossRef]
- Jayabalan, R.; Baskaran, S.; Marimuthu, S.; Swaminathan, K.; Yun, S.E. Effect of kombucha tea on aflatoxin B1 induced acute hepatotoxicity in albino rats-prophylatic and curative studies. J. Korean Soc. Appl. Biol. Chem. 2010, 53, 407–416. [Google Scholar] [CrossRef]
- Gonzalez-Gonzalez, C.R.; Tuohy, K.M.; Jauregi, P. Production of angiotensin-I-converting enzyme (ACE) inhibitory activity in milk fermented with probiotic strains: Effects of calcium, pH and peptides on the ACE-inhibitory activity. Int. Dairy J. 2011, 21, 615–622. [Google Scholar] [CrossRef]
- Elkhtab, E.; El-Alfy, M.; Shenana, M.; Mohamed, A.; Yousef, A.E. New potentially antihypertensive peptides liberated in milk during fermentation with selected lactic acid bacteria and kombucha cultures. J. Dairy Sci. 2017, 100, 9508–9520. [Google Scholar] [CrossRef]
- Vinci, G.; Maddaloni, L. Biogenic amines in alcohol-free beverages. Beverages 2020, 6, 17. [Google Scholar] [CrossRef]
- Chu, S.C.; Chen, C. Effects of origins and fermentation time on the antioxidant activities of kombucha. Food Chem. 2006, 98, 502–507. [Google Scholar] [CrossRef]
- Kumar, S.D.; Narayan, G.; Hassarajani, S. Determination of anionic minerals in black and kombucha tea using ion chromatography. Food Chem. 2008, 111, 784–788. [Google Scholar] [CrossRef]
- Chen, C.; Liu, B.Y. Changes in major components of tea fungus metabolites during prolonged fermentation. J. Appl. Microbiol. 2000, 89, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Bishop, P.; Pitts, E.R.; Budner, D.; Thompson-Witrick, K.A. Chemical composition of kombucha. Beverages 2022, 8, 45. [Google Scholar] [CrossRef]
- Kim, S.; Chen, J.; Cheng, T.; Gindulyte, A.; He, J.; He, S.; Li, Q.; Shoemaker, B.A.; Thiessen, P.A.; Yu, B.; et al. PubChem in 2021: New data content and improved web interfaces. Nucleic Acids Res. 2021, 49, D1388–D1395. [Google Scholar] [CrossRef]
- Bhattacharya, D.; Bhattacharya, S.; Patra, M.M.; Chakravorty, S.; Sarkar, S.; Chakraborty, W.; Koley, H.; Gachhui, R. Antibacterial activity of polyphenolic fraction of kombucha against enteric bacterial pathogens. Curr. Microbiol. 2016, 73, 885–896. [Google Scholar] [CrossRef] [PubMed]
- Jafari, R.; Naghavi, N.S.; Khosravi-Darani, K.; Doudi, M.; Shahanipour, K. Kombucha microbial starter with enhanced production of antioxidant compounds and invertase. Biocatal. Agric. Biotechnol. 2020, 29, 101789. [Google Scholar] [CrossRef]
- Cardoso, R.R.; Neto, R.O.; dos Santos D’Almeida, C.T.; do Nascimento, T.P.; Pressete, C.G.; Azevedo, L.; Martino, H.S.D.; Cameron, L.C.; Ferreira, M.S.L.; Barros, F.A.R. Kombuchas from green and black teas have different phenolic profile, with impacts their antioxidant capacities, antibacterial and antiproliferative activities. Food Res. Int. 2020, 128, 108782. [Google Scholar] [CrossRef]
- Nguyen, N.K.; Nguyen, P.B.; Nguyen, H.T.; Le, P.H. Screening the optimal ratio of symbiosis between isolated yeast and acetic acid bacteria strain from traditional kombucha for high-level production of glucuronic acid. LWT Food Sci. Technol. 2015, 64, 1149–1155. [Google Scholar] [CrossRef]
- Ruiz-Capillas, C.; Herrero, A. Impact of biogenic amines on food quality and safety. Foods 2019, 8, 62. [Google Scholar] [CrossRef]
- Utoiu, E.; Matei, F.; Toma, A.; Diguta, C.; Stefan, L.; Manoiu, S.; Vrajmasu, V.V.; Moraru, I.; Oancea, A.; Israel-Roming, F.; et al. Bee collected pollen with enhanced health benefits, produced by fermentation with a kombucha consortium. Nutrients 2018, 10, 1365. [Google Scholar] [CrossRef]
- Cao, Q.Q.; Zou, C.; Zhang, Y.H.; Du, Q.Z.; Yin, J.F.; Shi, J.; Xue, S.; Xu, Y.Q. Improving the taste of autumn green tea with tannase. Food Chem. 2019, 277, 432–437. [Google Scholar] [CrossRef]
- Zhou, D.D.; Saimaiti, A.; Luo, M.; Huang, S.Y.; Xiong, R.G.; Shang, A.; Gan, R.Y. Fermentation with tea residues enhances antioxidant activities and polyphenol contents in kombucha beverages. Antioxidants 2022, 11, 155. [Google Scholar] [CrossRef] [PubMed]
- Al-Suod, H.; Gadzaia-Kopeiuch, R.; Buszewski, B. Simultaneous HPLC-ELSD determination of sugars and cyclitols in different parts of Phacelia tanacelifolia Benth. Biochem. Syst. Ecol. 2018, 80, 32–38. [Google Scholar] [CrossRef]
- Jiang, L.; Shen, X.; Shoji, T.; Kanda, T.; Zhou, J.; Zhao, L. Characterization and activity of anthocyanins in Zijuan tea (Camellia sinensis var. Kitamura). J. Agri. Food Chem. 2013, 61, 3306–3310. [Google Scholar] [CrossRef] [PubMed]
- Shebis, Y.; Iluz, D.; Kinel-Tahan, Y.; Dubinsky, Z.; Yehoshua, Y. Natural antioxidants: Function and sources. Food Nutr. Sci. 2013, 4, 643–649. [Google Scholar] [CrossRef]
- Vohra, B.M.; Fazry, S.; Sairi, F.; Babul-Airianah, O. Effects of medium variation and fermentation time on the antioxidant and antimicrobial properties of kombucha. Malays. J. Fund. Appl. Sci. 2019, 15, 298–302. [Google Scholar] [CrossRef]
- Oliveira, J.T.; da Costa, F.M.; da Silva, T.G.; Simoes, G.D.; Pereira, E.S.; da Costa, P.Q.; Andreazza, R.; Schenkel, P.C.; Pieniz, S. Green tea and kombucha characterization: Phenolic composition, antioxidant capacity and enzymatic inhibition potential. Food Chem. 2023, 408, 135206. [Google Scholar] [CrossRef]
- Antony, J.I.X.; Shankaranaryana, M.L. Polyphenols of green tea. Int. Food Ingred. 1997, 5, 47–50. [Google Scholar]
- Yen, G.C.; Chen, H.Y.; Peng, H.H. Antioxidant and prooxidant effects of various tea extracts. J. Agri. Food Chem. 1997, 45, 30–34. [Google Scholar] [CrossRef]
- Battikh, H.; Chaieb, K.; Bakhrouf, A.; Ammar, E. Antibacterial and antifungal activities of black and green kombucha teas. J. Food Biochem. 2013, 37, 231–236. [Google Scholar] [CrossRef]
- Cetojevic-Simin, D.D.; Velicanski, A.S.; Cvetkovic, D.D.; Markov, S.L.; Mrdanovic, J.Z.; Bogdanovic, V.V.; Solajic, S.V. Bioactivity of lemon balm kombucha. Food Bioprocess Technol. 2012, 5, 1756–1765. [Google Scholar] [CrossRef]
- Deghrigue, M.; Chriaa, J.; Battikh, H.; Abid, K.; Bakhrouf, A. Antiproliferative and antimicrobial activities of kombucha tea. Afr. J. Microbiol. Res. 2013, 7, 3466–3470. [Google Scholar]
- Xia, X.; Dai, Y.; Wu, H.; Liu, X.; Wang, Y.; Yin, L.; Wang, Z.; Li, X.; Zhou, J. Kombucha fermentation enhances the health-promoting properties of soymilk beverage. J. Func. Food. 2019, 62, 103549. [Google Scholar] [CrossRef]
- Jayabalan, R.; Chen, P.N.; Hsieh, Y.S.; Prabhakaran, K.; Pitchai, P.; Marimuthu, S.; Thangaraj, P.; Swaminathan, K.; Yun, S.E. Effect of solvent fractions of kombucha tea on viability and invasiveness of cancer cells—Characterization of dimethyl 2-(2-hydroxy-2-methoxypropylidine) malonate and vitexin. Indian J. Biotechnol. 2011, 10, 75–82. [Google Scholar]
- Yapar, K.; Cavusoglu, K.; Oruc, E.; Yalcin, E. Protective effect of kombucha mushroom (KM) tea on phenol-induced cytotoxicity in albino mice. J. Environ. Biol. 2010, 31, 615–621. [Google Scholar]
- Coelho, R.M.D.; de Almeida, A.L.; do Amaral, R.Q.G.; da Mota, R.N.; de Sousa, P.H.M. Kombucha: Review. Int. J. Gastron. Food Sci. 2020, 22, 100272. [Google Scholar] [CrossRef]
- Antolak, H.; Piechota, D.; Kucharska, A. Kombucha tea-a double power of bioactive compounds from tea and symbiotic culture of bacteria and yeasts (SCOBY). Antioxidants 2012, 10, 1541. [Google Scholar] [CrossRef]
- Lee, C.; Kim, J.; Wang, S.; Sung, S.; Kim, N.; Lee, H.H.; Seo, Y.S.; Jung, Y. Hepatoprotective effect of kombucha tea in rodent model of nanalcoholic fatty liver disease/nonalcoholic steatohepatitis. Int. J. Mol. Sci. 2019, 20, 2369. [Google Scholar] [CrossRef]
- Jung, Y.; Kim, I.; Mannaa, M.; Kim, J.; Wang, S.; Park, I.; Kim, J.; Seo, Y.S. Effect of kombucha on gut-microbiota in mouse having nonalcoholic fatty liver disease. Food Sci. Biotechnol. 2019, 28, 261–267. [Google Scholar] [CrossRef]
- Kozyrovska, N.O.; Reva, O.M.; Goginyan, V.B.; Devera, J.P. Kombucha microbiome as a probiotic: A view from the perspective of post-genomics and synthetic ecology. Biopolym. Cell 2012, 28, 103–113. [Google Scholar] [CrossRef]
- Greenwalt, C.J.; Steinkraus, K.H.; Ledford, R.A. Kombucha, the fermented tea: Microbiology, composition, and claimed health effects. J. Food Prot. 2000, 63, 976–981. [Google Scholar] [CrossRef]
- Murphy, T.E.; Walia, K.; Farber, J.M. Safety aspects and guidance for consumers on the safe preparation, handling and storage of kombucha—A fermented tea beverage. Food Prot. Trends 2018, 38, 329–337. [Google Scholar]
- Kapp, J.M.; Summer, W. Kombucha: A systematic review of the empirical evidence of human health benefit. Ann. Epidemiol. 2019, 30, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Bailowitz, Z.; Grams, R.; Teeple, D.; Hew-Butler, T. Exercise-associated hyponatremia in a lactating female. Clin. J. Sport Med. 2017, 27, e55–e57. [Google Scholar] [CrossRef] [PubMed]
- Kovacevic, Z.; Davidovic, G.; Vuckovic-Filipovic, J.; Janicijevic-Petrovic, M.A.; Janicijevic, K.; Popovic, A. A toxic hepatitis caused the kombucha tea—Case report. Maced. J. Med. Sci. 2014, 2, 128–131. [Google Scholar] [CrossRef]
- Derk, C.T.; Sandorfi, N.; Curtis, M.T. A case of anti-Jo1 myositis with pleural effusions and pericardial tamponade developing after exposure to a fermented kombucha beverage. Clin. Rheumatol. 2004, 23, 355–357. [Google Scholar] [CrossRef]
- Gedela, M.; Potu, K.C.; Gali, V.L.; Alyamany, K.; Jha, L.K. A case of hepatotoxicity related to kombucha tea consumption. SD Med. 2016, 69, 26–28. [Google Scholar]
- Mallmann, M.M.; Valderramas, S.; Garcia, A.C.; Petterle, R.R.; Duarte, M.L.; Junior, O.R. Kombucha: A systemtic review and meta-analysis of experimental evidence of its effects on blood glucose, dyslipidemia and body weight in diabetes mellitus. Res. Soc. Dev. 2022, 11, e49011629278. [Google Scholar] [CrossRef]
| Microorganisms | Name | References |
|---|---|---|
| Acetic acid bacteria | Acetobacter xylinum | [45] |
| Acetobacter aceti | [46] | |
| Acetobacter pausterianus, Acetobacter intermedius | [19,47,48] | |
| Acetobacter peroxydans | [17] | |
| Acetobacter pomorum, Acetobacter malorum | [19] | |
| Acetobacter tropicalis, Acetobacter senegalensis | [19,49] | |
| Acetobacter nitrogenifigens Gluconobacter oxydans Gluconacetobacter entanii Komagataeibacter europaeus Komagataeibacter kombuchae | [50,51] [17,49] [21] [17,46,49] [51] | |
| Komagataeibacter medellinensis | [49] | |
| Komagataeibacter nataicola Komagataeibacter rhaeticus | [49] [19,49] | |
| Komagataeibacter saccharivorans | [46,49] | |
| Komagataeibacter xylinus | [49] | |
| Lactic acid bacteria | Bifidobacterium sp., Lactococcus sp., Leuconostoc sp., Lactobacillus kefiranofaciens Lactobacillus mali Lactiplantibacillus plantarum | [52] [53] [54] |
| Liquorilactobacillus nagelii | [49,52] | |
| Liquorilactobacillus satsumensis | [52] | |
| Limosilactobacillus fermentum | [46] | |
| Oenococcus oeni | [17] | |
| Yeasts | Arxula adeninivorans | [46] |
| Saccharomyces cerevisiae | [46,49,55] | |
| Saccharomyces bisporus | [56] | |
| Saccharomycodes ludwigii | [49,57] | |
| Schizosaccharomyces pombe | [19,49,58] | |
| Starmerella davenpoortii, Zygosaccharomyces bisporus | [49] | |
| Zygosaccharomyces rouxii | [59] | |
| Zygosaccharomyces bailii | [17,19,47,60] | |
| Zygosaccharomyces parabailii | [21] | |
| Zygosaccharomyces kombuchaensis | [60,61] | |
| Brettanomyces intermedius | [59] | |
| Brettanomyces bruxellensis | [17,19,49,58] | |
| Brettanomyces anomalus | [17,49] | |
| Brettanomyces claussenii Brettanomyces/Dekkera sp. | [62] [63] | |
| Candida arabinofermentans | [19] | |
| Candida guilliermondii, Candida colleculosa, Candida kefyr, Candida krusei, Candida stellata | [57,58] | |
| Hanseniaspora valbyensis, Hanseniaspora vineae | [17,60] | |
| Kloeckera apiculata, Kluyeromyces africanus | [55] | |
| Pichia membranefaciens | [59] | |
| Torulaspora delbrueckii | [58,60] |
| Component | Techniques or Platforms | References |
|---|---|---|
| Organic acids | High-performance liquid chromatography (HPLC) equipped with an Agilent ZORBAX® SB-C18 column and a UV detector | [93] |
| HPLC equipped with a C18 column and a UV detector | [14] | |
| Polyphenols (catechins), caffeine | HPLC equipped with a Waters Symmetry C18 column and a UV detector | [94] |
| HPLC coupled with a photodiode array detector (HPLC-PDAD) and an Agilent Zorbax Eclipse XDB-C18 column | [95] | |
| Sugars (sucrose, glucose, fructose) | HPLC equipped with a Waters XBridgeTM Amdie column and an evaporative light-scattering detector | [96] |
| Pigments (anthocyanins) | HPLC equipped with an Agilent ZORBAX® SB-C18 column and a UV detector | [97] |
| Alcohol | Ebuliometer | [14] |
| Alcoholometer | [15] | |
| Ultra-performance liquid chromatography (UPLC) | [12] | |
| Volatile compounds | Gas chromatography–mass spectrometry (GC-MS) | [19,46] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kitwetcharoen, H.; Phung, L.T.; Klanrit, P.; Thanonkeo, S.; Tippayawat, P.; Yamada, M.; Thanonkeo, P. Kombucha Healthy Drink—Recent Advances in Production, Chemical Composition and Health Benefits. Fermentation 2023, 9, 48. https://doi.org/10.3390/fermentation9010048
Kitwetcharoen H, Phung LT, Klanrit P, Thanonkeo S, Tippayawat P, Yamada M, Thanonkeo P. Kombucha Healthy Drink—Recent Advances in Production, Chemical Composition and Health Benefits. Fermentation. 2023; 9(1):48. https://doi.org/10.3390/fermentation9010048
Chicago/Turabian StyleKitwetcharoen, Haruthairat, Ly Tu Phung, Preekamol Klanrit, Sudarat Thanonkeo, Patcharaporn Tippayawat, Mamoru Yamada, and Pornthap Thanonkeo. 2023. "Kombucha Healthy Drink—Recent Advances in Production, Chemical Composition and Health Benefits" Fermentation 9, no. 1: 48. https://doi.org/10.3390/fermentation9010048
APA StyleKitwetcharoen, H., Phung, L. T., Klanrit, P., Thanonkeo, S., Tippayawat, P., Yamada, M., & Thanonkeo, P. (2023). Kombucha Healthy Drink—Recent Advances in Production, Chemical Composition and Health Benefits. Fermentation, 9(1), 48. https://doi.org/10.3390/fermentation9010048