Biohydrogen and Methane Production from Sugarcane Leaves Pretreated by Deep Eutectic Solvents and Enzymatic Hydrolysis by Cellulolytic Consortia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sugarcane Leaves Preparation
2.2. Deep Eutectic Solvents Preparations
2.3. Cellulolytic Consortium
2.4. Clostridium butyricum TISTR 1032 Prepartions
2.5. Anaerobic Sludge Preparations
2.6. The Optimization Factors Affecting the Pretreatment of Sugarcane Leaves
2.7. SHF for Two-Stage Hydrogen and Methane Production and One-Stage Methane Production from Pretreated Sugarcane Leaves
2.8. SSF for Two-Stage Hydrogen and Methane Production and One-Stage Methane Production from Pretreated Sugarcane Leaves
2.9. Enzyme acTIVITY Assay
2.10. Analytical Methods
2.11. Statistical Analysis
2.12. Calculations
3. Results
3.1. Effects of Molar Ratio and Type of Deep Eutectic Solvent (DES) on Sugarcane Leaves
3.2. Effects of Pretreatment Time and Pretreatment Temperature
3.3. Effect of the Substrate to DES Solution Ratio
3.4. Enzymatic Hydrolysis of DES Pretreated Sugarcane Leaves by the Cellulolytic Consortium
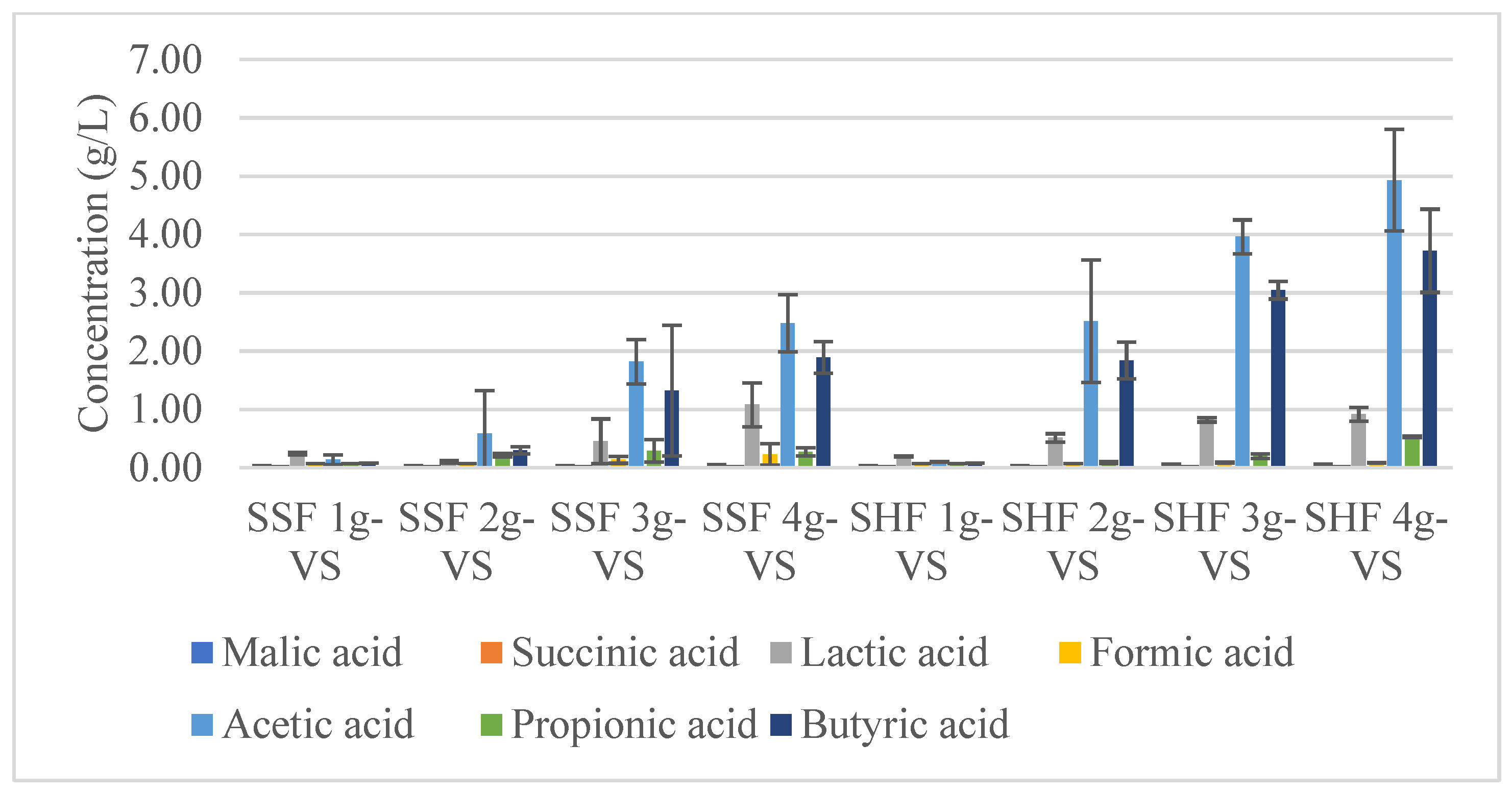
3.5. The Two-Stages Hydrogen and Methane Production by SHF and SSF
3.5.1. Hydrogen Production
3.5.2. Methane Production
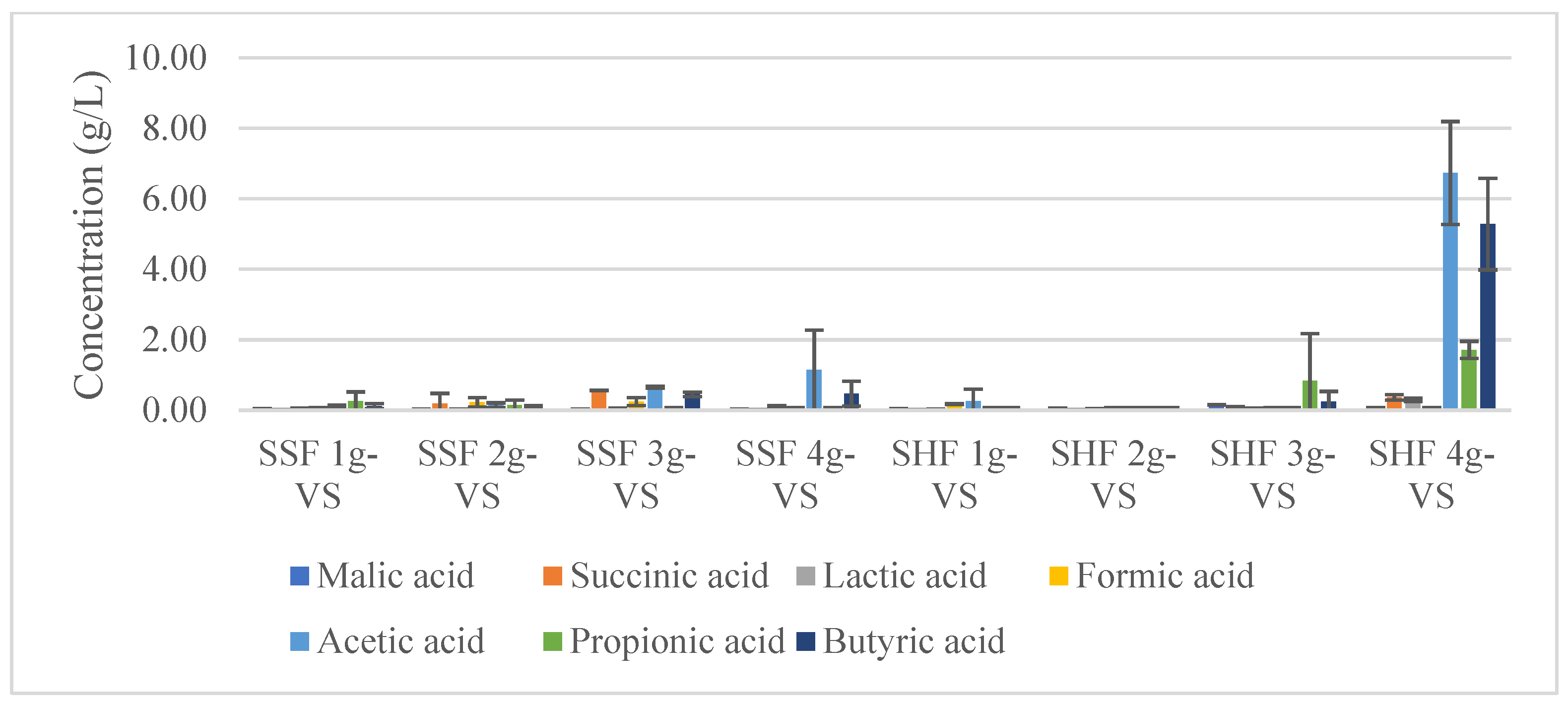
3.6. One-Stage Methane Production by SHF and SSF
4. Discussion
4.1. The Effect of Molar Ratio and Type of Deep Eutectic Solvents (DESs) on Sugarcane Leaves
4.2. The Effect of Pretreatment Time and Pretreatment Temperature
4.3. Effects of the Substrate to DES Solution Ratio
4.4. The Enzymatic Hydrolysis of DES Pretreated Sugarcane Leaves by the Cellulolytic Consortium
4.5. The Two Stages of Hydrogen and Methane Production by SHF and SSF
4.5.1. Hydrogen Production
4.5.2. Methane Production
4.6. One-Stage Methane Production by SHF and SSF
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Olguin-Maciel, E.; Singh, A.; Chable-Villacis, R.; Tapia-Tussell, R.; Ruiz, H.A. Consolidated Bioprocessing, an Innovative Strategy towards Sustainability for Biofuels Production from Crop Residues: An Overview. Agronomy 2020, 10, 1834. [Google Scholar] [CrossRef]
- Wilkie, A.C. Biomethane from Biomass, Biowaste, and Biofuels. In Bioenergy; ASM Press: Washington, DC, USA, 2008; pp. 195–205. [Google Scholar] [CrossRef]
- Zhu, X.; Yellezuome, D.; Liu, R.; Wang, Z.; Liu, X. Effects of co-digestion of food waste, corn straw and chicken manure in two-stage anaerobic digestion on trace element bioavailability and microbial community composition. Bioresour. Technol. 2022, 346, 126625. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Ankaram, S. Waste-to-Energy Model/Tool Presentation. In Current Developments in Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar] [CrossRef]
- Manyi-Loh, C.E.; Mamphweli, S.N.; Meyer, E.L.; Okoh, A.I.; Makaka, G.; Simon, M. Microbial Anaerobic Digestion (Bio-Digesters) as an Approach to the Decontamination of Animal Wastes in Pollution Control and the Generation of Renewable Energy. Int. J. Environ. Res. Public Health 2013, 10, 4390–4417. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Liu, S.; Xin, F.; Zhou, J.; Jia, H.; Xu, J.; Jiang, M.; Dong, W. Biomethane Production from Lignocellulose: Biomass Recalcitrance and Its Impacts on Anaerobic Digestion. Front. Bioeng. Biotechnol. 2019, 7, 191. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.; Wright, M.M. Anaerobic digestion fundamentals, challenges, and technological advances. Phys. Sci. Rev. 2022. [Google Scholar] [CrossRef]
- Ambaye, T.G.; Vaccari, M.; Bonilla-Petriciolet, A.; Prasad, S.; van Hullebusch, E.D.; Rtimi, S. Emerging technologies for biofuel production: A critical review on recent progress, challenges and perspectives. J. Environ. Manag. 2021, 290, 112627. [Google Scholar] [CrossRef]
- Dodo, C.M.; Mamphweli, S.; Okoh, O. Bioethanol production from lignocellulosic sugarcane leaves and tops. J. Energy South. Afr. 2017, 28, 1–11. [Google Scholar] [CrossRef]
- Chandel, A.K.; da Silva, S.S.; Carvalho, W.; Singh, O.V. Sugarcane bagasse and leaves: Foreseeable biomass of biofuel and bio-products. J. Chem. Technol. Biotechnol. 2012, 87, 11–20. [Google Scholar] [CrossRef]
- Moodley, P.; Kana, E.G. Comparative study of three optimized acid-based pretreatments for sugar recovery from sugarcane leaf waste: A sustainable feedstock for biohydrogen production. Eng. Sci. Technol. Int. J. 2018, 21, 107–116. [Google Scholar] [CrossRef]
- Moodley, P.; Kana, E.G. Optimization of xylose and glucose production from sugarcane leaves (Saccharum officinarum) using hybrid pretreatment techniques and assessment for hydrogen generation at semi-pilot scale. Int. J. Hydrogen Energy 2015, 40, 3859–3867. [Google Scholar] [CrossRef]
- Oliveira, D.M.; Mota, T.R.; Grandis, A.; de Morais, G.R.; de Lucas, R.C.; Polizeli, M.L.; Marchiosi, R.; Buckeridge, M.S.; Ferrarese-Filho, O.; dos Santos, W.D. Lignin plays a key role in determining biomass recalcitrance in forage grasses. Renew. Energy 2020, 147, 2206–2217. [Google Scholar] [CrossRef]
- Prasad, R.K.; Chatterjee, S.; Mazumder, P.B.; Gupta, S.K.; Sharma, S.; Vairale, M.G.; Datta, S.; Dwivedi, S.K.; Gupta, D.K. Bioethanol production from waste lignocelluloses: A review on microbial degradation potential. Chemosphere 2019, 231, 588–606. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, A.; Kumar, P.S.; Jeevanantham, S.; Karishma, S.; Vo, D.-V.N. Recent advances and sustainable development of biofuels production from lignocellulosic biomass. Bioresour. Technol. 2022, 344, 126203. [Google Scholar] [CrossRef]
- Yoo, C.G.; Meng, X.; Pu, Y.; Ragauskas, A.J. The critical role of lignin in lignocellulosic biomass conversion and recent pretreatment strategies: A comprehensive review. Bioresour. Technol. 2020, 301, 122784. [Google Scholar] [CrossRef] [PubMed]
- Yoo, C.G.; Li, M.; Meng, X.; Pu, Y.; Ragauskas, A.J. Effects of organosolv and ammonia pretreatments on lignin properties and its inhibition for enzymatic hydrolysis. Green Chem. 2017, 19, 2006–2016. [Google Scholar] [CrossRef]
- Kellock, M.; Maaheimo, H.; Marjamaa, K.; Rahikainen, J.; Zhang, H.; Holopainen-Mantila, U.; Ralph, J.; Tamminen, T.; Felby, C.; Kruus, K. Effect of hydrothermal pretreatment severity on lignin inhibition in enzymatic hydrolysis. Bioresour. Technol. 2019, 280, 303–312. [Google Scholar] [CrossRef]
- Yu, Z.; Gwak, K.-S.; Treasure, T.; Jameel, H.; Chang, H.-M.; Park, S. Effect of Lignin Chemistry on the Enzymatic Hydrolysis of Woody Biomass. ChemSusChem 2014, 7, 1942–1950. [Google Scholar] [CrossRef]
- Zoghlami, A.; Paës, G. Lignocellulosic Biomass: Understanding Recalcitrance and Predicting Hydrolysis. Front. Chem. 2019, 7, 874. [Google Scholar] [CrossRef]
- Gunny, A.A.N.; Arbain, D. Ionic Liquids: Green Solvent for Pretreatment of Lingnocellulosic Biomass. Adv. Mater. Res. 2013, 701, 399–402. [Google Scholar] [CrossRef]
- Chen, Y.; Mu, T. Application of deep eutectic solvents in biomass pretreatment and conversion. Green Energy Environ. 2019, 4, 95–115. [Google Scholar] [CrossRef]
- Wang, B.; Qin, L.; Mu, T.; Xue, Z.; Gao, G. Are Ionic Liquids Chemically Stable? Chem. Rev. 2017, 117, 7113–7131. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Chen, Y.; Lu, L.; Xue, Z.; Mu, T. Water Sorption in Functionalized Ionic Liquids: Kinetics and Intermolecular Interactions. Ind. Eng. Chem. Res. 2013, 52, 2073–2083. [Google Scholar] [CrossRef]
- Cao, Y.; Mu, T. Comprehensive Investigation on the Thermal Stability of 66 Ionic Liquids by Thermogravimetric Analysis. Ind. Eng. Chem. Res. 2014, 53, 8651–8664. [Google Scholar] [CrossRef]
- Ishizaki, H.; Hasumi, K. Ethanol Production from Biomass. In Sustainable Biomass Systems; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar] [CrossRef]
- Shamurad, B.; Sallis, P.; Petropoulos, E.; Tabraiz, S.; Ospina, C.; Leary, P.; Dolfing, J.; Gray, N. Stable biogas production from single-stage anaerobic digestion of food waste. Appl. Energy 2020, 263, 114609. [Google Scholar] [CrossRef]
- Van, D.P.; Fujiwara, T.; Tho, B.L.; Toan, P.P.S.; Minh, G.H. A review of anaerobic digestion systems for biodegradable waste: Configurations, operating parameters, and current trends. Environ. Eng. Res. 2020, 25, 1–17. [Google Scholar] [CrossRef]
- Holdeman, L.V.; Moore, W.E.C. Anaerobe Laboratory Manual; Virginia Polytechnic Institute and State University, Anaerobe Laboratory: Blacksburg, VA, USA, 1975. [Google Scholar]
- Feng, Y.; Yu, Y.; Wang, X.; Qu, Y.; Li, D.; He, W.; Kim, B.H. Degradation of raw corn stover powder (RCSP) by an enriched microbial consortium and its community structure. Bioresour. Technol. 2011, 102, 742–747. [Google Scholar] [CrossRef]
- Owen, W.F.; Stuckey, D.C.; Healy, J.B., Jr.; Young, L.Y.; McCarty, P.L. Bioassay for Monitoring Biochemical Methane Potential And Anaerobic Toxicity. Water Res. 1979, 13, 485–492. [Google Scholar] [CrossRef]
- Ghose, T.K. Measurement of Cellulase Activities. Pure Appl. Chem. 1987, 59, 257–268. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017. [Google Scholar]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D.L. Determination of Structural Carbohydrates and Lignin in Biomass. Natl. Renew. Energy Lab. 2012, 17, 1–13. [Google Scholar]
- Miller, G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Sitthikitpanya, S.; Reungsang, A.; Prasertsan, P. Two-stage thermophilic bio-hydrogen and methane production from lime-pretreated oil palm trunk by simultaneous saccharification and fermentation. Int. J. Hydrogen Energy 2018, 43, 4284–4293. [Google Scholar] [CrossRef]
- Jomnonkhaow, U.; Sittijunda, S.; Reungsang, A. Enhanced simultaneous saccharification and fermentation of Napier grass and Napier silage for two stage bio-hydrogen and methane production using organosolv and hydrothermal. Mater. Chem. Phys. 2021, 267, 124614. [Google Scholar] [CrossRef]
- Zheng, X.-J.; Yu, H.-Q. Inhibitory effects of butyrate on biological hydrogen production with mixed anaerobic cultures. J. Environ. Manag. 2005, 74, 65–70. [Google Scholar] [CrossRef]
- Khanal, S. Biological hydrogen production: Effects of pH and intermediate products. Int. J. Hydrogen Energy 2004, 29, 1123–1131. [Google Scholar] [CrossRef]
- Zhao, Z.; Chen, X.; Ali, M.F.; Abdeltawab, A.A.; Yakout, S.M.; Yu, G. Pretreatment of wheat straw using basic ethanolamine-based deep eutectic solvents for improving enzymatic hydrolysis. Bioresour. Technol. 2018, 263, 325–333. [Google Scholar] [CrossRef]
- Galbe, M.; Wallberg, O. Pretreatment for biorefineries: A review of common methods for efficient utilisation of lignocellulosic materials. Biotechnol. Biofuels 2019, 12, 294. [Google Scholar] [CrossRef]
- Kwon, G.-J.; Bandi, R.; Yang, B.-S.; Park, C.-W.; Han, S.-Y.; Park, J.-S.; Lee, E.-A.; Kim, N.-H.; Lee, S.-H. Choline chloride based deep eutectic solvents for the lignocellulose nanofibril production from Mongolian oak (Quercus mongolica). Cellulose 2021, 28, 9169–9185. [Google Scholar] [CrossRef]
- Ling, J.K.U.; Hadinoto, K. Deep Eutectic Solvent as Green Solvent in Extraction of Biological Macromolecules: A Review. Int. J. Mol. Sci. 2022, 23, 3381. [Google Scholar] [CrossRef]
- Triwahyuni, E.; Muryanto; Sudiyani, Y.; Abimanyu, H. The Effect of Substrate Loading on Simultaneous Saccharification and Fermentation Process for Bioethanol Production from Oil Palm Empty Fruit Bunches. Energy Procedia 2015, 68, 138–146. [Google Scholar] [CrossRef]
- Loow, Y.-L.; New, E.K.; Yang, G.H.; Ang, L.Y.; Foo, L.Y.W.; Wu, T.Y. Potential use of deep eutectic solvents to facilitate lignocellulosic biomass utilization and conversion. Cellulose 2017, 24, 3591–3618. [Google Scholar] [CrossRef]
- Sun, Y.; Cheng, J. Hydrolysis of lignocellulosic materials for ethanol production: A review. Bioresour. Technol. 2002, 83, 1–11. [Google Scholar] [CrossRef]
- Xia, Q.; Liu, Y.; Meng, J.; Cheng, W.; Chen, W.; Liu, S.; Liu, Y.; Li, J.; Yu, H. Multiple hydrogen bond coordination in three-constituent deep eutectic solvents enhances lignin fractionation from biomass. Green Chem. 2018, 20, 2711–2721. [Google Scholar] [CrossRef]
- Amin, F.R.; Khalid, H.; Zhang, H.; Rahman, S.U.; Zhang, R.; Liu, G.; Chen, C. Pretreatment methods of lignocellulosic biomass for anaerobic digestion. AMB Express 2017, 7, 72. [Google Scholar] [CrossRef]
- Baruah, J.; Nath, B.K.; Sharma, R.; Kumar, S.; Deka, R.C.; Baruah, D.C.; Kalita, E. Recent Trends in the Pretreatment of Lignocellulosic Biomass for Value-Added Products. Front. Energy Res. 2018, 6, 141. [Google Scholar] [CrossRef]
- Niju, S.; Swathika, M.; Balajii, M. Pretreatment of lignocellulosic sugarcane leaves and tops for bioethanol production. In Lignocellulosic Biomass to Liquid Biofuels; Academic Press: Cambridge, MA, USA, 2019. [Google Scholar] [CrossRef]
- Nhuchhen, D.; Basu, P.; Acharya, B. A Comprehensive Review on Biomass Torrefaction. Int. J. Renew. Energy Biofuels 2014, 2014, 506376. [Google Scholar] [CrossRef]
- Lismeri, L.; Darni, Y.; Sanjaya, M.D.; Immadudin, M.I. Pengaruh Suhu Dan Waktu Pretreatment Alkali Pada Isolasi Selulosa Limbah Batang Pisang. J. Chem. Process Eng. 2019, 4, 18–22. [Google Scholar] [CrossRef]
- Modenbach, A.A.; Nokes, S.E. The use of high-solids loadings in biomass pretreatment—A review. Biotechnol. Bioeng. 2012, 109, 1430–1442. [Google Scholar] [CrossRef]
- Lay, J.-J.; Li, Y.-Y.; Noike, T. Influences of pH and moisture content on the methane production in high-solids sludge digestion. Water Res. 1997, 31, 1518–1524. [Google Scholar] [CrossRef]
- Taconi, K.A.; Zappi, M.E.; French, W.T.; Brown, L.R. Methanogenesis under acidic pH conditions in a semi-continuous reactor system. Bioresour. Technol. 2008, 99, 8075–8081. [Google Scholar] [CrossRef]
- Magdalena, J.A.; Greses, S.; González-Fernández, C. Impact of Organic Loading Rate in Volatile Fatty Acids Production and Population Dynamics Using Microalgae Biomass as Substrate. Sci. Rep. 2019, 9, 18374. [Google Scholar] [CrossRef]
- Luo, J.; Meng, H.; Yao, Z.; Wachemo, A.C.; Yuan, H.; Zhang, L.; Li, X. Anaerobic co-digestion of sodium hydroxide pretreated sugarcane leaves with pig manure and dairy manure. Int. J. Agric. Biol. Eng. 2018, 11, 197–204. [Google Scholar] [CrossRef]
- Mokomele, T.; Da CostaSousa, L.; Balan, V.; van Rensburg, E.; Dale, B.E.; Görgens, J.F. Incorporating anaerobic co-digestion of steam exploded or ammonia fiber expansion pretreated sugarcane residues with manure into a sugarcane-based bioenergy-livestock nexus. Bioresour. Technol. 2019, 272, 326–336. [Google Scholar] [CrossRef]
- Ketsub, N.; Latif, A.; Kent, G.; Doherty, W.O.; O’Hara, I.M.; Zhang, Z.; Kaparaju, P. A systematic evaluation of biomethane production from sugarcane trash pretreated by different methods. Bioresour. Technol. 2021, 319, 124137. [Google Scholar] [CrossRef]
- Ellacuriaga, M.; Cascallana, J.G.; González, R.; Gómez, X. High-Solid Anaerobic Digestion: Reviewing Strategies for Increasing Reactor Performance. Environments 2021, 8, 80. [Google Scholar] [CrossRef]
- Wang, B.; Strömberg, S.; Li, C.; Nges, I.A.; Nistor, M.; Deng, L.; Liu, J. Effects of substrate concentration on methane potential and degradation kinetics in batch anaerobic digestion. Bioresour. Technol. 2015, 194, 240–246. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhao, J.; Xu, F.; Li, Y. Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog. Energy. Combust. Sci. 2014, 42, 35–53. [Google Scholar] [CrossRef]
| DES | Molar Ratio | pH | Pretreatment | Composition of Residues | ||||
|---|---|---|---|---|---|---|---|---|
| Cellulose Recovery (%) | Hemicellulose Removal (%) | Lignin Removal (%) | Cellulose (%) | Hemicellulose (%) | Lignin (%) | |||
| Untreated | - | - | - | - | - | 36.18 ± 0.73 f | 25.23 ± 0.02 a | 27.68 ± 0.35 a |
| ChCl/MEA | 1:6 | 14.08 | 71.86 ± 0.37 c,d | 69.88 ± 2.93 b,c | 77.62 ± 0.79 a | 57.62 ± 0.30 b,c | 16.84 ± 1.64 d | 13.73 ± 0.48 e |
| 1:8 | 14.12 | 73.49 ± 0.53 c | 71.07 ± 0.54 b | 77.50 ± 0.33 a | 58.92 ± 0.42 a | 16.21 ± 0.30 d | 13.80 ± 0.30 e | |
| 1:10 | 14.18 | 70.96 ± 2.10 d | 68.21 ± 1.45 c | 76.41 ± 0.85 a | 55.33 ± 1.64 d | 17.28 ± 0.79 d | 14.07 ± 0.51 e | |
| ChCl/G | 1:2 | 7.93 | 82.37 ± 0.71 b | 33.60 ± 1.16 e | 28.98 ± 1.23 d | 39.40 ± 0.34 e | 22.15 ± 0.45 b | 25.99 ± 0.45 b |
| 1:4 | 6.40 | 85.08 ± 0.81 a | 33.57 ± 1.67 e | 25.11 ± 0.61 e | 38.72 ± 0.53 e | 21.09 ± 0.53 b,c | 26.08 ± 0.21 b | |
| 1:6 | 6.60 | 82.14 ± 1.49 b | 37.35 ± 2.39 d | 29.41 ± 0.80 d | 39.12 ± 0.71 e | 20.81 ± 0.79 c | 25.72 ± 0.29 b | |
| ChCl/G/AlCl3 | 1:2:0.33 | 0.40 | 65.65 ± 0.23 f | 87.00 ± 0.07 a | 68.57 ± 0.26 c | 59.28 ± 0.20 a | 8.19 ± 0.05 e | 21.72 ± 0.18 c |
| 1:4:0.33 | 0.38 | 66.61 ± 0.70 e,f | 88.54 ± 0.48 a | 70.27 ± 0.54 b | 58.74 ± 0.62 a,b | 7.05 ± 0.29 f | 20.06 ± 0.37 d | |
| 1:6:0.33 | 0.36 | 68.11 ± 0.81 e | 88.21 ± 0.22 a | 68.39 ± 0.19 c | 57.37 ± 0.68 c | 6.92 ± 0.13 f | 20.37 ± 0.13 d | |
| DES | Temp (°C)/Time (h) | Pretreatment | Composition of Residues | ||||
|---|---|---|---|---|---|---|---|
| Cellulose Recovery (%) | Hemicellulose Removal (%) | Lignin Removal (%) | Cellulose (%) | Hemicellulose (%) | Lignin (%) | ||
| Untreated | 36.56 ± 0.77 k | 27.31 ± 0.03 a | 27.68 ± 0.35 a | ||||
| ChCl/MEA (1:6) | 80/3 | 69.25 ± 0.97 g | 57.76 ± 0.72 h | 73.49 ± 0.86 i | 53.37 ± 0.75 j | 24.32 ± 0.41 b | 15.47 ± 0.50 b |
| 80/6 | 68.69 ± 1.51 g | 66.60 ± 1.90 g | 78.67 ± 0.52 h | 57.61 ± 1.27 h | 20.93 ± 1.19 c | 13.55 ± 0.33 c | |
| 80/9 | 68.37 ± 1.13 g | 70.23 ± 1.40 f | 80.81 ± 0.61 g | 60.93 ± 1.01 g | 19.82 ± 0.93 d | 12.95 ± 0.41 d | |
| 100/3 | 71.54 ± 0.30 f | 70.29 ± 0.30 f | 83.45 ± 0.02 e,f | 64.25 ± 0.27 f | 19.93 ± 0.20 d | 11.25 ± 0.01 e | |
| 100/6 | 72.81 ± 0.42 e | 75.05 ± 0.96 a,b | 85.96 ± 0.28 d | 67.52 ± 0.39 d | 17.28 ± 0.66 f,g | 9.86 ± 0.20 f | |
| 100/9 | 72.04 ± 0.40 e,f | 76.57 ± 0.19 a | 87.06 ± 0.01 c | 69.64 ± 0.39 b,c | 16.92 ± 0.14 g,h | 9.47 ± 0.01 f,g | |
| 120/3 | 78.75 ± 0.22 a | 70.64 ± 0.51 e,f | 86.73 ± 0.22 c | 70.17 ± 0.20 b | 19.54 ± 0.34 d | 8.95 ± 0.15 h,i | |
| 120/6 | 78.32 ± 0.38 a | 72.57 ± 0.37 d | 87.95 ± 0.53 b | 71.91 ± 0.34 a | 18.12 ± 0.24 e,f | 8.07 ± 0.35 j | |
| 120/9 | 78.80 ± 0.89 a | 74.54 ± 0.79 b,c | 89.18 ± 0.29 a | 71.33 ± 0.81 a | 17.22 ± 0.54 f,gh | 7.42 ± 0.20 k | |
| The Substrate to DES Solution Ratio | Pretreatment | Composition of Residues | ||||
|---|---|---|---|---|---|---|
| Cellulose Recovery (%) | Hemicellulose Removal (%) | Lignin Removal (%) | Cellulose (%) | Hemicellulose (%) | Lignin (%) | |
| Untreated | 36.56 ± 0.77 d | 27.31 ± 0.03 a | 27.68 ± 0.35 a | |||
| 1:8 | 81.84 ± 2.23 b | 72.40 ± 0.68 c | 86.86 ± 0.16 b | 69.81 ± 0.62 c | 17.59 ± 0.43 b | 8.49 ± 0.10 b,c |
| 1:12 | 84.13 ± 0.77 a | 73.98 ± 0.42 b | 86.37 ± 0.36 b | 72.54 ± 0.67 b | 16.76 ± 0.27 c | 8.90 ± 0.23 b |
| 1:16 | 81.44 ± 1.21 b | 74.62 ± 0.41 b | 88.23 ± 0.83 a | 74.27 ± 1.11 a | 17.29 ± 0.28 b | 8.13 ± 0.57 c |
| 1:20 | 81.31 ± 1.11 b | 75.78 ± 0.45 a | 88.06 ± 0.48 a | 73.34 ± 1.00 a,b | 16.32 ± 0.30 c | 8.15 ± 0.33 c |
| 1:24 | 84.38 ± 0.15 a | 76.29 ± 0.10 a | 86.42 ± 0.09 b | 74.27 ± 0.13 a | 15.59 ± 0.07 d | 9.05 ± 0.06 b |
| Substrate Loading (g-VSadded) | Reducing Sugar Concentration (g/L) | |||||||
|---|---|---|---|---|---|---|---|---|
| Day 0 | Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 | Day 7 | |
| U-1 | 0.15 ± 0.02 a | 0.40 ± 0.01 e | 0.28 ± 0.01 e | 0.40 ± 0.04 d | 0.36 ± 0.01 e | 0.44 ± 0.01 d | 0.37 ± 0.01 f | 0.26 ± 0.24 f |
| U-2 | 0.16 ± 0.02 a | 0.21 ± 0.01 g | 0.43 ± 0.00 e | 0.50 ± 0.00 d | 0.78 ± 0.05 d | 0.88 ± 0.05 c | 0.96 ± 0.02 d | 0.85 ± 0.04 e |
| U-3 | 0.17 ± 0.02 a | 0.81 ± 0.03 c | 0.50 ± 0.02 d | 0.72 ± 0.02 c | 0.90 ± 0.02 c,d | 0.98 ± 0.00 c | 1.70 ± 0.01 d | 1.13 ± 0.01 d |
| U-4 | 0.17 ± 0.02 a | 0.35 ± 0.02 f | 0.36 ± 0.00 f | 0.74 ± 0.02 c | 1.12 ± 0.20 b | 1.66 ± 0.33 a | 1.64 ± 0.29 c | 1.49 ± 0.28 c |
| P-1 | 0.17 ± 0.02 a | 0.41 ± 0.02 e | 0.36 ± 0.03 f | 0.49 ± 0.02 d | 0.47 ± 0.09 e | 0.60 ± 0.14 d | 0.61 ± 0.11 e | 0.35 ± 0.07 f |
| P-2 | 0.17 ± 0.02 a | 0.70 ± 0.01 d | 0.57 ± 0.02 c | 0.86 ± 0.15 b | 0.95 ± 0.11 c | 1.43 ± 0.02 b | 1.65 ± 0.06 c | 1.85 ± 0.15 b |
| P-3 | 0.16 ± 0.00 a | 1.18 ± 0.03 a | 1.52 ± 0.01 a | 1.56 ± 0.02 a | 1.71 ± 0.02 a | 1.78 ± 0.02 a | 1.97 ± 0.08 b | 1.95 ± 0.07 b |
| P-4 | 0.17 ± 0.02 a | 0.95 ± 0.03 b | 1.10 ± 0.03 b | 0.92 ± 0.03 b | 1.25 ± 0.03 b | 1.88 ± 0.01 a | 2.18 ± 0.02 a | 2.35 ± 0.02 a |
| Substrate Loading (g-VSadded) | SSF-Two Stages Hydrogen Production | SHF-Two Stages Hydrogen Production | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Hydrogen Production (mL/L) | Hydrogen Production Rate (mL/L d) | Lag Phase (λ) (d) | Hydrogen Yield (mL/g-VSadded) | R2 | Hydrogen Production (mL/L) | Hydrogen Production Rate (mL/L d) | Lag Phase (λ) (d) | Hydrogen Yield (mL/g-VSadded) | R2 | |
| U_1 | 21 ± 4 e | 1.8 ± 0.8 h | 0.0 | 1.0 ± 0.1 f | 0.9826 | 17 ± 1 e | 1.5 ± 0.2 h | 0.5 | 0.8 ± 0.1 f | 0.9844 |
| U_2 | 66 ± 5 e | 10.0 ± 2.0 g,h | 2.0 | 1.7 ± 0.1 f | 0.9935 | 32 ± 15 e | 6.2 ± 6.4 g,h | 4.0 | 0.8 ± 0.4 f | 0.9814 |
| U_3 | 133 ± 14 e | 22.0 ± 3.3 f,g | 2.0 | 2.2 ± 0.2 f | 0.9958 | 52 ± 10 e | 14.2 ± 5.2 g,h | 5.0 | 0.9 ± 0.2 f | 0.9892 |
| U_4 | 229 ± 20 e | 34.7 ± 3.9 e,f | 2.0 | 2.9 ± 0.2 f | 0.9962 | 99 ± 81 e | 20.2 ± 15.4 f,g,h | 5.0 | 1.2 ± 1.0 f | 0.9885 |
| P_1 | 152 ± 7.0 e | 23.0 ± 2.2 f,g | 0.5 | 7.6 ± 0.3 e | 0.9947 | 48 ± 19 e | 7.9 ± 3.6 g,h | 3.0 | 2.4 ± 1.0 f | 0.9902 |
| P_2 | 821 ± 48 d | 85.9 ± 5.1 d | 4.0 | 20.5 ± 1.2 c | 0.9956 | 682 ± 154 d | 40.9 ± 10.7 e | 4.0 | 17.0 ± 3.8 d | 0.9916 |
| P_3 | 1840 ± 201 c | 211.2 ± 14.7 b | 4.0 | 30.7 ± 3.3 b | 0.9978 | 1700 ± 136 c | 112.4 ± 7.2 c | 5.0 | 28.1 ± 1.9 b | 0.9962 |
| P_4 | 3187 ± 209 a | 317.5 ± 20.5 a | 5.0 | 39.8 ± 2.6 a | 0.9941 | 2135 ± 315 b | 197.2 ± 20.5 b | 7.0 | 26.7 ± 3.9 a | 0.9952 |
| Substrates Loading (g-VSadded) | SSF Two-Stages Methane Production | SHF Two-Stages Methane Production | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Methane Production (mL/L) | Methane Production Rate (mL/L d) | Lag Phase (λ) (d) | Methane Yield (mL/g-VSadded) | R2 | Methane Production (mL/L) | Methane Production Rate (mL/L d) | Lag Phase (λ) (d) | Methane Yield (mL/g-VSadded) | R2 | |
| U_1 | 1448 ± 354 f | 29.9 ± 10.4 e | 14 | 115.9 ± 28.3 c,d,e,f | 0.9931 | 1468 ± 441 f | 33.3 ± 13.9 d,e | 13 | 117.4 ± 35.3 c,d,e,f | 0.9921 |
| U_2 | 2633 ± 390 c,d,e | 64.7 ± 9.7 c,d,e | 11 | 105.3 ± 15.6 d,e,f | 0.9968 | 1974 ± 545 e,f | 43.1 ± 15.6 d,e | 12 | 91.1 ± 21.4 e,f | 0.9972 |
| U_3 | 3009 ± 516 b,c | 63.1 ± 15.6 c,d,e | 12 | 80.3 ± 13.8 e,f | 0.9949 | 2847 ± 530 b,c,d | 53.8 ± 3.1 d,e | 10 | 75.9 ± 14.1 e,f | 0.9950 |
| U_4 | 3311 ± 806 b,c | 76.8 ± 18.8 b,c,d,e | 19 | 66.2 ± 16.1 e,f | 0.9954 | 3097 ± 244 b,c | 70.1 ± 3.6 b,c,d,e | 18 | 61.9 ± 4.9 f | 0.9926 |
| P_1 | 2654 ± 277 c,d,e | 101.8 ± 27.7 b,c | 6 | 212.3 ± 22.2 a | 0.9950 | 2077 ± 308 d,e,f | 68.4 ± 25.4 b,c,d,e | 6 | 166.1 ± 24.6 a,b,c,d | 0.9873 |
| P_2 | 3402 ± 760 b,c | 109.7 ± 68.4 b,c | 3 | 178.5 ± 72.0 a,b,c | 0.9660 | 3644 ± 473 b | 77.5 ± 25.3 b,c,d,e | 16 | 201.2 ± 113.8 a,b | 0.9795 |
| P_3 | 5179 ± 291 a | 160.1 ± 15.2 a | 1 | 138.1 ± 7.8 b,c,d,e | 0.9751 | 3221 ± 417 b,c | 113.8 ± 30.9 b | 1 | 85.9 ± 11.1 e,f | 0.9797 |
| P_4 | 5923 ± 251 a | 159.3 ± 19.0 a | 1 | 118.5 ± 5.0 c,d,e,f | 0.9828 | 3583 ± 128 b | 79.3 ± 9.8 b,c,d | 2 | 71.7 ± 2.6 e,f | 0.9778 |
| Substrates Loading (g-VSadded) | SSF One-Stage Methane Production | SHF One-Stage Methane Production | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Methane Production (mL/L) | Methane Production Rate (mL/L d) | Lag Phase (λ) (d) | Methane Yield (mL/g-VSadded) | R2 | Methane Production (mL/L) | Methane Production Rate (ml/L d) | Lag Phase (λ) (d) | Methane Yield (mL/g-VSadded) | R2 | |
| U_1 | 1632 ± 481 f,g | 27.4 ± 14.0 e | 9 | 130.5 ± 38.5 a,b,c | 0.9848 | 1597 ± 12 f,g | 21.5 ± 6.3 e | 10 | 127.8 ± 33.5 a,b,c | 0.9918 |
| U_2 | 2015 ± 370 e,f,g | 30.0 ± 7.0 d,e | 9 | 80.6 ± 14.8 | 0.9816 | 2179 ± 405 e,f,g | 33.4 ± 5.1 d,e | 18 | 87.2 ± 16.2 d,e,f,g | 0.9938 |
| U_3 | 2735 ± 186 c,d,e | 42.6 ± 3.1 c,d,e | 16 | 76.3 ± 5.5 d,e,f,g | 0.9907 | 1941 ± 558 e,f,g | 29.2 ± 11.3 e | 16 | 51.7 ± 14.9 e,f,g | 0.9629 |
| U_4 | 2988 ± 112 b,c,d | 46.8 ± 4.9 c,d,e | 12 | 59.8 ± 2.2 e,f,g | 0.9940 | 2304 ± 304 d,e,f | 35.9 ± 7.5 c,d,e | 15 | 46.1 ± 6.1 g | 0.9867 |
| P_1 | 2029 ± 104 e,f,g | 41.8 ± 3.7 c,d,e | 4 | 162.4 ± 8.3 a | 0.9948 | 1853 ± 242 f,g | 34.5 ± 7.9 d,e | 9 | 148.3 ± 19.3 a,b | 0.9948 |
| P_2 | 2155 ± 449 e,f,g | 59.3 ± 31.2 b,c | 2 | 113.0 ± 44.9 b,c,d | 0.9905 | 1478 ± 796 g | 44.6 ± 17.7 c,d,e | 2 | 74.7 ± 34.3 d,e,f,g | 0.9600 |
| P_3 | 3474 ± 613 a,b,c | 79.8 ± 15.8 b | 1 | 92.6 ± 16.3 c,d,e,f | 0.9875 | 3723 ± 340 a,b | 60.3 ± 13.4 b,c | 12 | 99.3 ± 9.1 c,d,e | 0.9965 |
| P_4 | 4067 ± 319 a | 120.5 ± 9.5 a | 4 | 81.3 ± 6.4 d,e,f,g | 0.9936 | 3349 ± 415 a,b,c | 54.7 ± 16.6 c,d | 3 | 67.0 ± 8.3 e,f,g | 0.9793 |
| Substrate | Pretreatment Method | Pretreatment Conditions | Fermentation Mode | Methane Yield | References |
|---|---|---|---|---|---|
| Sugarcane leaves | Ammonium fiber explosion (AFEX) pretreatment | 80–120 °C, 60 min | N/A-One-stage | 336 mL/g-VSadded | [58] |
| Sodium hydroxide (6% NaOH) pretreatment | 25 °C, 3 days | N/A-One-stage | 287 mL/g-TSadded | [57] | |
| Potassium hydroxide (KOH) pretreatment | 170 °C, 60 min | N/A-One-stage | 205 mL/g-TSadded | [59] | |
| Liquid hot water (LHW) pretreatment | 190 °C, 60 min | N/A-One-stage | 162 mL/g-TSadded | [59] | |
| Dilute acid (DA) pretreatment | 170 °C, 15 min | N/A-One-stage | 156 mL/g-TSadded | [59] | |
| DES Pretreatment | 120 °C, 3 h | SSF-Two-stage | 118 mL/g-VSadded | This study | |
| DES Pretreatment | 120 °C, 3 h | SSF-One-stage | 81 mL/g-VSadded | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miftah, A.K.; Sittijunda, S.; Imai, T.; Salakkam, A.; Reungsang, A. Biohydrogen and Methane Production from Sugarcane Leaves Pretreated by Deep Eutectic Solvents and Enzymatic Hydrolysis by Cellulolytic Consortia. Fermentation 2022, 8, 396. https://doi.org/10.3390/fermentation8080396
Miftah AK, Sittijunda S, Imai T, Salakkam A, Reungsang A. Biohydrogen and Methane Production from Sugarcane Leaves Pretreated by Deep Eutectic Solvents and Enzymatic Hydrolysis by Cellulolytic Consortia. Fermentation. 2022; 8(8):396. https://doi.org/10.3390/fermentation8080396
Chicago/Turabian StyleMiftah, Apik Khautsart, Sureewan Sittijunda, Tsuyoshi Imai, Apilak Salakkam, and Alissara Reungsang. 2022. "Biohydrogen and Methane Production from Sugarcane Leaves Pretreated by Deep Eutectic Solvents and Enzymatic Hydrolysis by Cellulolytic Consortia" Fermentation 8, no. 8: 396. https://doi.org/10.3390/fermentation8080396
APA StyleMiftah, A. K., Sittijunda, S., Imai, T., Salakkam, A., & Reungsang, A. (2022). Biohydrogen and Methane Production from Sugarcane Leaves Pretreated by Deep Eutectic Solvents and Enzymatic Hydrolysis by Cellulolytic Consortia. Fermentation, 8(8), 396. https://doi.org/10.3390/fermentation8080396








