Golden Berry Waste for Electricity Generation
Abstract
:1. Introduction
2. Materials and Methods
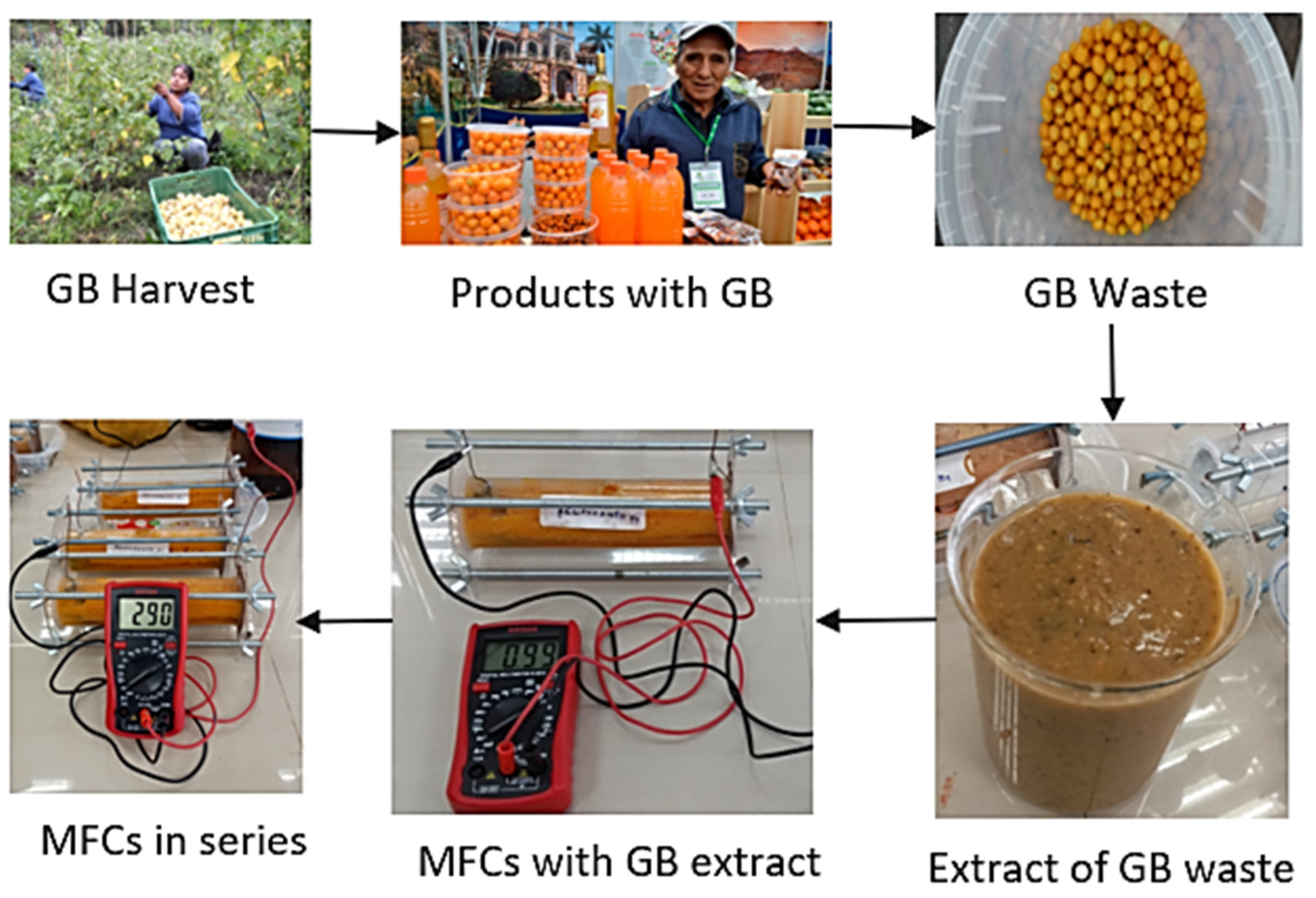
2.1. Construction of Microbial Fuel Cells
2.2. Collection and Preparation of Golden Berry Waste
2.3. Isolation of Electrogenic Microorganisms from the Anode Chamber
2.4. Molecular Identification of Bacteria
2.5. Physicochemical Characterization of MFCs
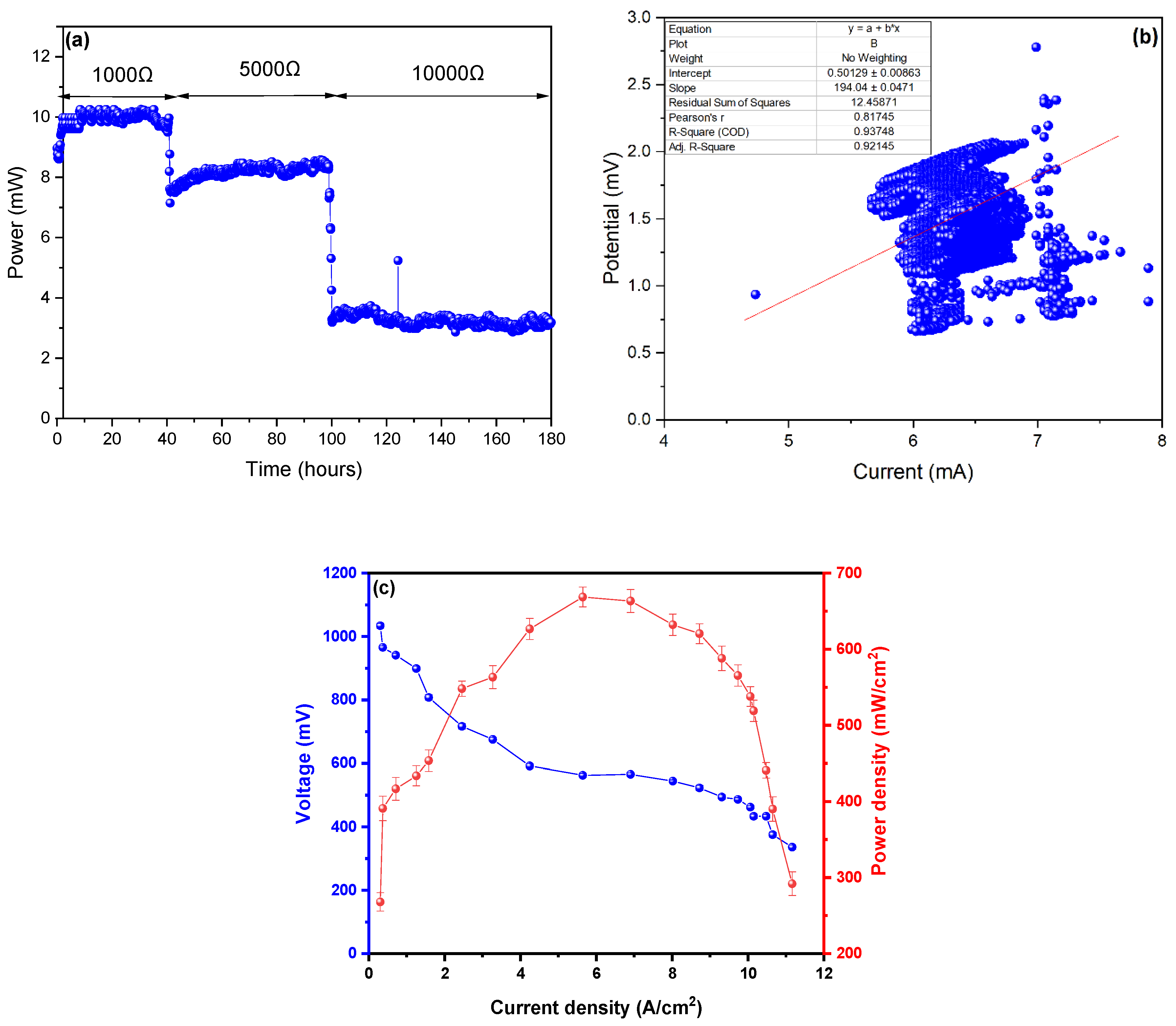
3. Results and Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Naseer, M.N.; Zaidi, A.A.; Khan, H.; Kumar, S.; bin Owais, M.T.; Jaafar, J.; Suhaimin, N.S.; Wahab, Y.A.; Dutta, K.; Asif, M.; et al. Mapping the field of microbial fuel cell: A quantitative literature review (1970–2020). Energy Rep. 2021, 7, 4126–4138. [Google Scholar] [CrossRef]
- Qian, X.; Lee, S.; Chandrasekaran, R.; Yang, Y.; Caballes, M.; Alamu, O.; Chen, G. Electricity evaluation and emission characteristics of poultry litter co-combustion process. Appl. Sci. 2019, 9, 4116. [Google Scholar] [CrossRef] [Green Version]
- Abavisani, F.; Mahdavi, M.A.; Gheshlaghi, R. Energy Harvesting from Microbial Fuel Cell Using a Power Management System: A Review. In Proceedings of the 3rd International Conference on Innovation and Research in Engineering Science, Tbilisi, Georgia, 21 July 2019; pp. 1–5. [Google Scholar]
- Song, H.L.; Zhu, Y.; Li, J. Electron transfer mechanisms, characteristics and applications of biological cathode microbial fuel cells–A mini review. Arab. J. Chem. 2019, 12, 2236–2243. [Google Scholar] [CrossRef] [Green Version]
- Kaur, R.; Marwaha, A.; Chhabra, V.A.; Kim, K.H.; Tripathi, S.K. Recent developments on functional nanomaterial-based electrodes for microbial fuel cells. Renew. Sustain. Energy Rev. 2020, 119, 109551. [Google Scholar] [CrossRef]
- Zhao, Q.; Yu, H.; Zhang, W.; Kabutey, F.T.; Jiang, J.; Zhang, Y.; Wang, K.; Ding, J. Microbial fuel cell with high content solid wastes as substrates: A review. Front. Environ. Sci. Eng. 2017, 11, 13. [Google Scholar] [CrossRef]
- Palanisamy, G.; Jung, H.Y.; Sadhasivam, T.; Kurkuri, M.D.; Kim, S.C.; Roh, S.H. A comprehensive review on microbial fuel cell technologies: Processes, utilization, and advanced developments in electrodes and membranes. J. Clean. Prod. 2019, 221, 598–621. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; La Cruz-Noriega, D.; Nazario-Naveda, R.; Benites, S.M.; Delfín-Narciso, D.; Angelats-Silva, L.; Murga-Torres, E. Use of Banana Waste as a Source for Bioelectricity Generation. Processes 2022, 10, 942. [Google Scholar] [CrossRef]
- Pereda, M.S.B.; Nazareno, M.A.; Viturro, C.I. Nutritional and antioxidant properties of Physalis peruviana L. fruits from the Argentinean northern Andean region. Plant Foods Hum. Nutr. 2019, 74, 68–75. [Google Scholar] [CrossRef]
- Shenstone, E.; Lippman, Z.; Van Eck, J. A review of nutritional properties and health benefits of Physalis species. Plant Foods Hum. Nutr. 2020, 75, 316–325. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; Santiago, B.; Quiñones, R.A.; Enríquez-León, R.; Luis, A.S. Bioelectricity through Microbial Fuel Cells from Decomposed Fruits Using Lead and Copper Electrodes. Available online: http://www.laccei.org/LACCEI2020-VirtualEdition/full_papers/FP17.pdf (accessed on 2 May 2022).
- Hidayat, D.D.; Luthfiyanti, R.; Iwansyah, A.C.; Herminiati, A.; Rahman, T.; Rahman, N.; Andriansyah, R.C.E. Identification and Evaluation of Physical and Mechanical Properties of Physalis peruviana L. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Jember, Indonesia, 7–8 November 2020; IOP Publishing: Bristol, UK, 2021; Volume 672, p. 012056. [Google Scholar]
- Vega, J.C.D.L.; Olmedo, V.; Ortega, C.G.; Lara, M.V.; Espín, R.D.C. Conservation advances on Physalis peruviana L. and Spondia purpurea: A review. Food Sci. Technol. 2020, 42. [Google Scholar] [CrossRef]
- Rahman, W.; Yusup, S.; Mohammad, S.A. Screening of fruit waste as substrate for microbial fuel cell (MFC). AIP Conf. Proc. 2021, 2332, 020003. [Google Scholar]
- Kebaili, H.; Kameche, M.; Innocent, C.; Ziane, F.Z.; Sabeur, S.A.; Sahraoui, T.; Ouis, M.; Zerrouki, A.; Charef, M.A. Treatment of fruit waste leachate using microbial fuel cell: Preservation of agricultural environment. Acta Ecol. Sin. 2021, 41, 97–105. [Google Scholar] [CrossRef]
- Dhulipala, V.R.; Gurjar, R.; Behera, M. Bioelectricity generation from kitchen waste in a low-cost earthenware microbial fuel cell. In Recent Developments in Waste Management; Springer: Singapore, 2020; pp. 309–322. [Google Scholar]
- Ramadan, M.A.; Abd-Alla, M.H.; Abdul-Raouf, U.M. Bioelectricity generation from agro-industrial waste water using dual-chambered microbial fuel cell. Enzyme Microb. Technol. 2013, 52, 352–357. [Google Scholar]
- Rojas-Flores, S.; Benites, S.M.; De La Cruz-Noriega, M.; Cabanillas-Chirinos, L.; Valdiviezo-Dominguez, F.; Quezada Álvarez, M.A.; Vega-Ybañez, V.; Angelats-Silva, L. Bioelectricity Production from Blueberry Waste. Processes 2021, 9, 1301. [Google Scholar] [CrossRef]
- Del Rosario Rodicio, M.; del Carmen Mendoza, M. Identificación bacteriana mediante secuenciación del ARNr 16S: Fundamento, metodología y aplicaciones en microbiología clínica. Enferm. Infecc. Microbiol. Clín. 2004, 22, 238–245. [Google Scholar] [CrossRef]
- Ma’arof MIN, M.I.N.; Chala, G.T.; Ravichanthiran, S. A study on microbial fuel cell (MFC) with graphite electrode to power underwater monitoring devices. Int. J. Mech. Technol. 2018, 9, 98–105. [Google Scholar]
- Ivanov, R.; Bratkova, S.; Angelov, A. Analysis of the efficiency of microbial fuel cells based on sulfate-reduction process, integrated in anaerobic wetlands. Ann. Univ. Sofia Fac. Biol. 2017, 102, 248–260. [Google Scholar]
- Ma, H.; Peng, C.; Jia, Y.; Wang, Q.; Tu, M.; Gao, M. Effect of fermentation stillage of food waste on bioelectricity production and microbial community structure in microbial fuel cells. R. Soc. Open Sci. 2018, 5, 180457. [Google Scholar] [CrossRef] [Green Version]
- Prasidha, W. Electricity Production from Food Waste Leachate (Fruit and Vegetable Waste) using Double Chamber Microbial Fuel Cell: Comparison between Non-aerated and Aerated Configuration. ROTASI 2020, 22, 162–168. [Google Scholar]
- Yang, N.; Zhan, G.; Wu, T.; Zhang, Y.; Jiang, Q.; Li, D.; Xiang, Y. Effect of air-exposed biocathode on the performance of a Thauera-dominated membraneless single-chamber microbial fuel cell (SCMFC). J. Environ. Sci. 2018, 66, 216–224. [Google Scholar] [CrossRef]
- Tamboli, E.; Eswari, J.S. Microbial fuel cell configurations: An overview. In Microbial Electrochemical Technology; Elsevier: Amsterdam, The Netherlands, 2019; pp. 407–435. [Google Scholar]
- Hindatu, Y.; Annuar, M.S.M.; Gumel, A.M. Mini-review: Anode modification for improved performance of microbial fuel cell. Renew. Sustain. Energy Rev. 2017, 73, 236–248. [Google Scholar] [CrossRef]
- Santoro, C.; Kodali, M.; Shamoon, N.; Serov, A.; Soavi, F.; Merino-Jimenez, I.; Gajda, I.; Greenman, J.; Ieropoulos, I.; Atanassov, P. Increased power generation in supercapacitive microbial fuel cell stack using FeNC cathode catalyst. J. Power Sources 2019, 412, 416–424. [Google Scholar] [CrossRef] [PubMed]
- Borjas, Z.; Esteve-Núñez, A.; Ortiz, J.M. Strategies for merging microbial fuel cell technologies in water desalination processes: Start-up protocol and desalination efficiency assessment. J. Power Sources 2017, 356, 519–528. [Google Scholar] [CrossRef]
- Fischer, G.; Almanza-Merchán, P.J.; Miranda, D. Importancia y cultivo de la uchuva (Physalis peruviana L.). Rev. Bras. Frutic. 2014, 36, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Brooke, J.S.; Di Bonaventura, G.; Berg, G.; Martinez, J.L. A multidisciplinary look at Stenotrophomonas maltophilia: An emerging multi-drug-resistant global opportunistic pathogen. Front. Microbiol. 2017, 8, 1511. [Google Scholar] [CrossRef]
- Margaria, V.; Tommasi, T.; Pentassuglia, S.; Agostino, V.; Sacco, A.; Armato, C.; Chiodoni, A.; Schilirò, T.; Quaglio, M. Effects of pH variations on anodic marine consortia in a dual chamber microbial fuel cell. Int. J. Hydrogen Energy 2017, 42, 1820–1829. [Google Scholar] [CrossRef]
- Embaby, H.E.S.; Mokhtar, S.M. Impact of adding goldenberry (Physalis peruviana L.) on some quality characteristics and bio-functional properties of pasteurized carrot (Daucus carota L.) nectar. J. Food Sci. Technol. 2019, 56, 966–975. [Google Scholar] [CrossRef]
- Kamau, J.M.; Mbui, D.N.; Mwaniki, J.M.; Mwaura, F.B.; Kamau, G.N. Microbial fuel cells: Influence of external resistors on power, current and power density. J. Thermodyn. Catal. 2017, 8, 1–5. [Google Scholar]
- Christwardana, M.; Hadiyanto, H.; Motto, S.A.; Sudarno, S.; Haryani, K. Performance evaluation of yeast-assisted microalgal microbial fuel cells on bioremediation of cafeteria wastewater for electricity generation and microalgae biomass production. Biomass Bioenergy 2020, 139, 105617. [Google Scholar] [CrossRef]
- Zu, B.; Ma, L.; Liu, B.; Lu, P.L.; Xu, J. Effects of Organic Substrates on ANAMMOX-MFC Denitrification Electrogenesis Performance. Huan Jing Ke Xue 2018, 39, 3937–3945. [Google Scholar]
- Flores, S.J.R.; Benites, S.M.; Rosa, A.L.R.A.L.; Zoilita, A.L.Z.A.L.; Luis, A.S.L. The Using Lime (Citrus × aurantiifolia), Orange (Citrus × sinensis), and Tangerine (Citrus reticulata) Waste as a Substrate for Generating Bioelectricity: Using lime (Citrus × aurantiifolia), orange (Citrus × sinensis), and tangerine (Citrus reticulata) waste as a substrate for generating bioelectricity. Environ. Res. Eng. Manag. 2020, 76, 24–34. [Google Scholar]
- Kamau, J.M.; Mbui, D.N.; Mwaniki, J.M.; Mwaura, F.B. Utilization of rumen fluid in production of bio–energy from market waste using microbial fuel cells technology. J. Appl. Biotechnol. Bioeng. 2018, 5, 227–231. [Google Scholar]
- Nogales-Bueno, J.; Baca-Bocanegra, B.; Rooney, A.; Hernández-Hierro, J.M.; Byrne, H.J.; Heredia, F.J. Study of phenolic extractability in grape seeds by means of ATR-FTIR and Raman spectroscopy. Food Chem. 2017, 232, 602–609. [Google Scholar] [CrossRef]
- Mahesar, S.A.; Lucarini, M.; Durazzo, A.; Santini, A.; Lampe, A.I.; Kiefer, J. Application of infrared spectroscopy for functional compounds evaluation in olive oil: A current snapshot. J. Spectrosc. 2019, 2019, 5319024. [Google Scholar] [CrossRef] [Green Version]
- Ricci, A.; Olejar, K.J.; Parpinello, G.P.; Kilmartin, P.A.; Versari, A. Application of Fourier transform infrared (FTIR) spectroscopy in the characterization of tannins. Appl. Spectrosc. Rev. 2015, 50, 407–442. [Google Scholar] [CrossRef]
- Bose, D.; Gopinath, M.; Vijay, P. Sustainable power generation from wastewater sources using microbial fuel cell. Biofuels Bioprod. Biorefining 2018, 12, 559–576. [Google Scholar] [CrossRef]
- Zafar, Z.; Ayaz, K.; Nasir, M.H.; Yousaf, S.; Sharafat, I.; Ali, N. Electrochemical performance of biocathode microbial fuel cells using petroleum-contaminated soil and hot water spring. Int. J. Environ. Sci. Technol. 2019, 16, 1487–1500. [Google Scholar] [CrossRef]
- Adegoke, A.; Stenström, T.; Okoh, A. Stenotrophomonas maltophilia as an Emerging Ubiquitous Pathogen: Looking Beyond Contemporary Antibiotic Therapy. Front. Microbiol. 2017, 8, 2276. [Google Scholar] [CrossRef] [Green Version]
- An, S.; Berg, G. Stenotrophomonas maltophilia. Trends Microbiol. 2018, 26, 637–638. [Google Scholar] [CrossRef] [Green Version]
- Sharma, P.; Mutnuri, S. Nutrient recovery and microbial diversity in human urine fed microbial fuel cell. Water Sci. Technol. 2019, 79, 718–730. [Google Scholar] [CrossRef]





| BLAST Characterization | Consensus Sequence Length (nt) | % Maximum Identity | Accession Number | Phylogeny |
|---|---|---|---|---|
| Stenotrophomonas maltophilia | 1474 | 99.93% | CP040434.1 | Cellular organisms; Bacteria; Proteobacteria; Gammaproteobacteria; Xanthomonadales; Xanthomonadaceae; Stenotrophomonas; Stenotrophomonas maltophilia group |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Segundo, R.-F.; De La Cruz-Noriega, M.; Nazario-Naveda, R.; Benites, S.M.; Delfín-Narciso, D.; Angelats-Silva, L.; Díaz, F. Golden Berry Waste for Electricity Generation. Fermentation 2022, 8, 256. https://doi.org/10.3390/fermentation8060256
Segundo R-F, De La Cruz-Noriega M, Nazario-Naveda R, Benites SM, Delfín-Narciso D, Angelats-Silva L, Díaz F. Golden Berry Waste for Electricity Generation. Fermentation. 2022; 8(6):256. https://doi.org/10.3390/fermentation8060256
Chicago/Turabian StyleSegundo, Rojas-Flores, Magaly De La Cruz-Noriega, Renny Nazario-Naveda, Santiago M. Benites, Daniel Delfín-Narciso, Luis Angelats-Silva, and Felix Díaz. 2022. "Golden Berry Waste for Electricity Generation" Fermentation 8, no. 6: 256. https://doi.org/10.3390/fermentation8060256
APA StyleSegundo, R.-F., De La Cruz-Noriega, M., Nazario-Naveda, R., Benites, S. M., Delfín-Narciso, D., Angelats-Silva, L., & Díaz, F. (2022). Golden Berry Waste for Electricity Generation. Fermentation, 8(6), 256. https://doi.org/10.3390/fermentation8060256







