Mathematical Analysis and Update of ADM1 Model for Biomethane Production by Anaerobic Digestion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mathematical Model of Anaerobic Digestion
2.2. Variables and Constant Description
2.2.1. Variables
2.2.2. Constants
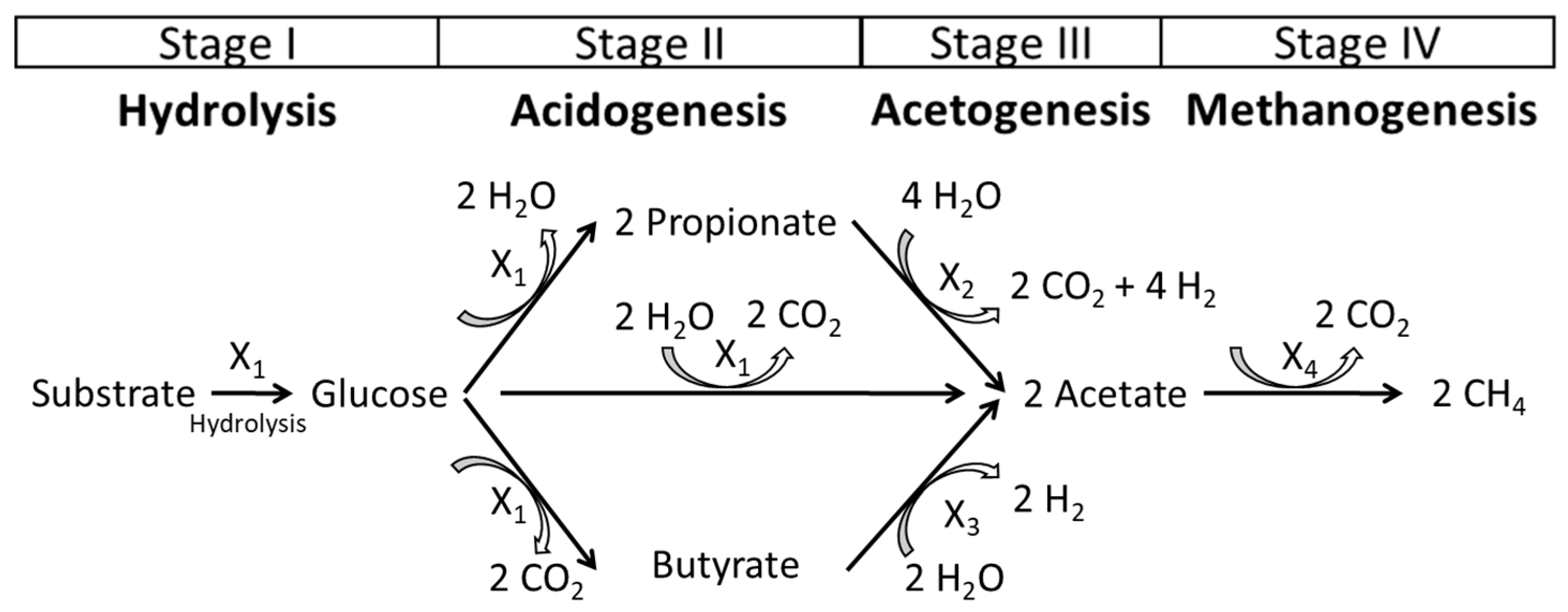
2.3. Balancing the Reaction Equations of Anaerobic Digestion Mathematical Model
2.4. Simulation Studies
3. Results and Discussion
3.1. Biochemical Description of the Model
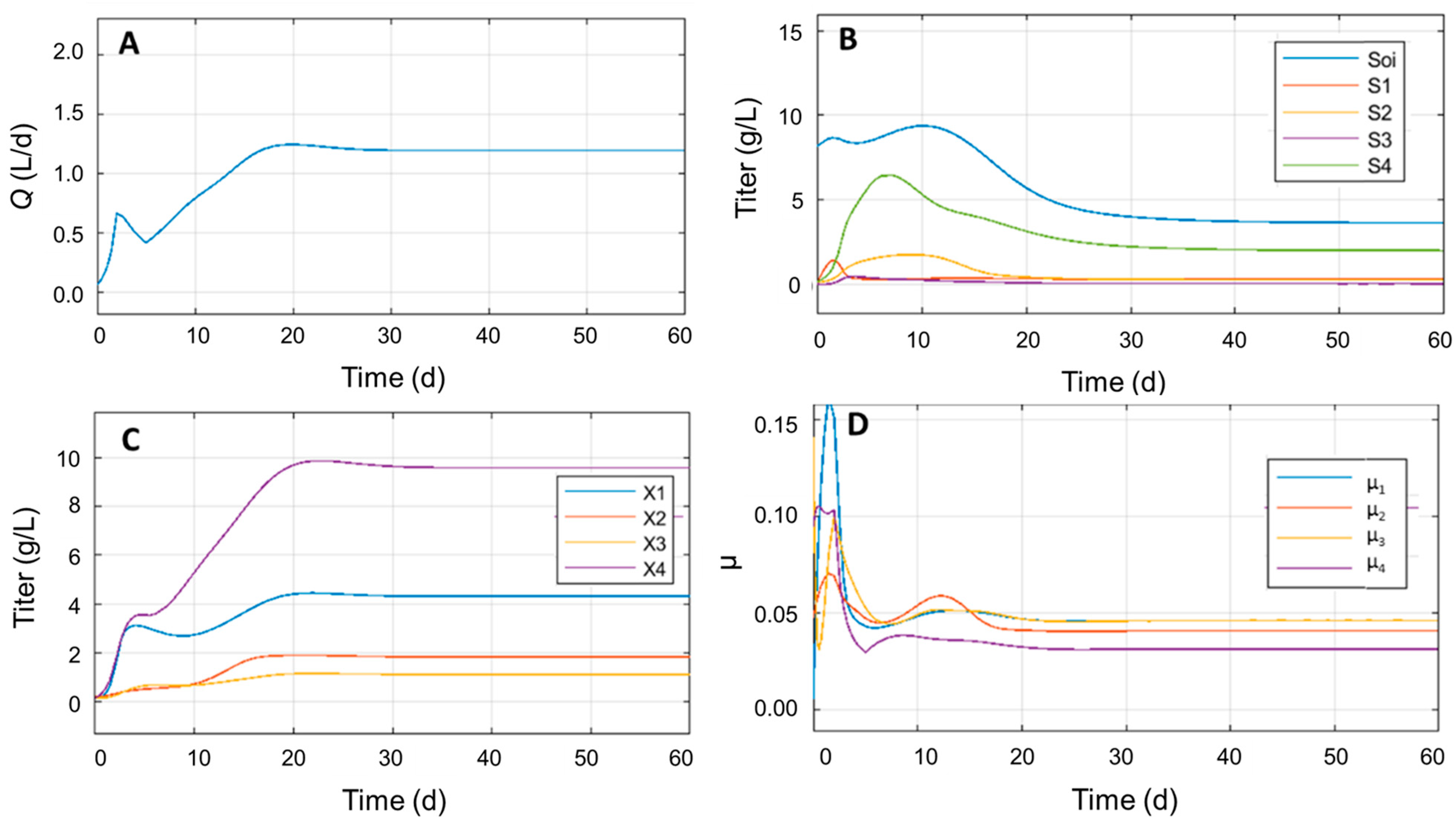
3.2. Microbiological Analysis of ADM1 Model
3.3. Update to ADM1 Model Structure
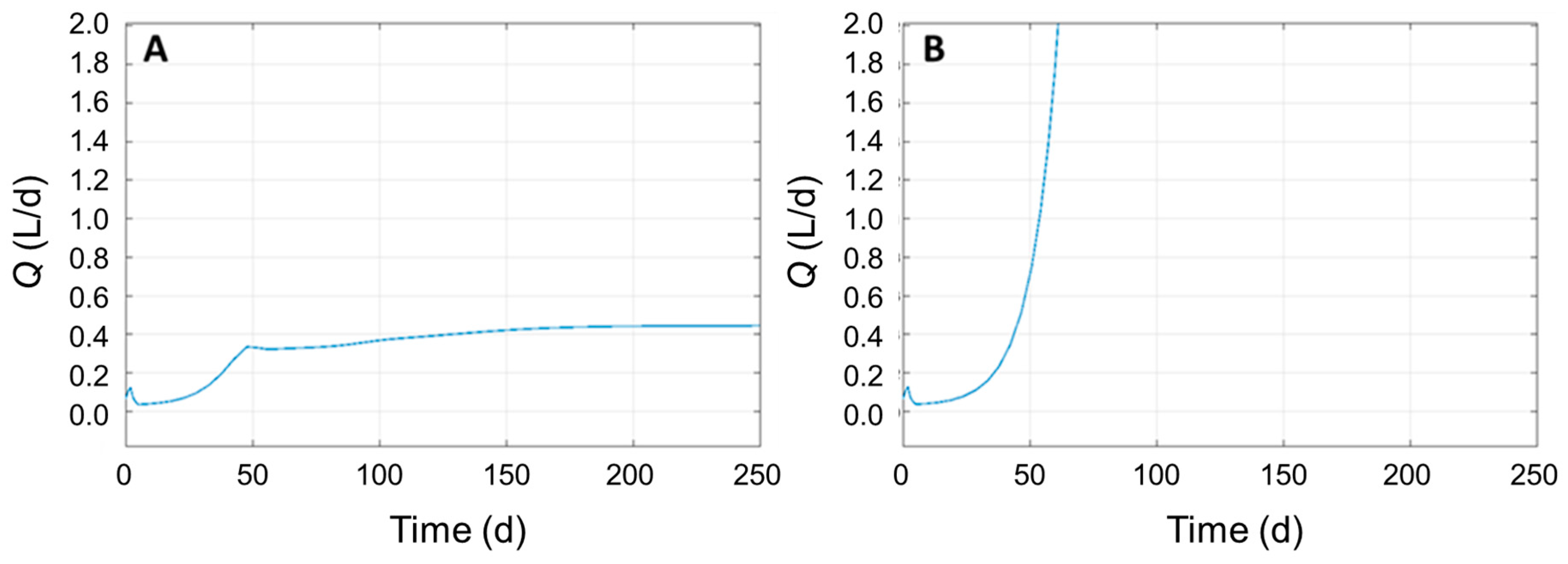
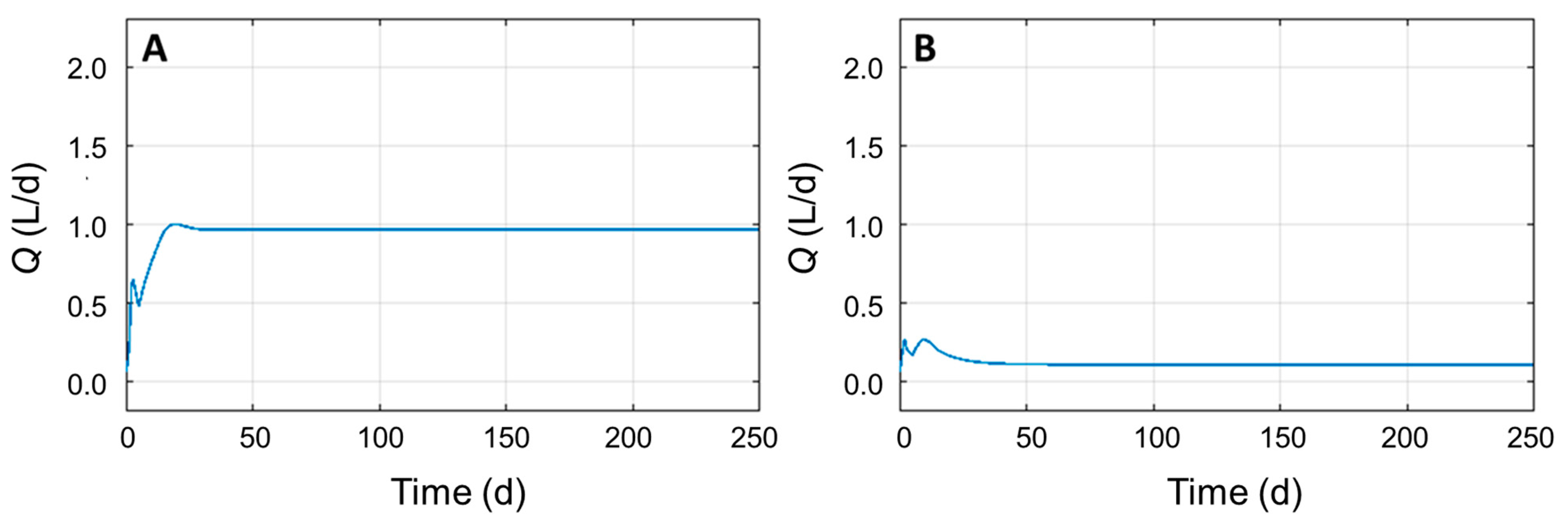
3.4. Identifying Significant Process Parameters
4. Practical Applications of This Work and Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- The European Parliament and the Council of the European Union. Directive 2009/28/EC on the Promotion of the Use of Energy from Renewable Sources and Amending and Subsequently Repealing Directives 2001/77/EC and 2003/30/EC; Official Journal of the European Union, European Union: Luxembourg, 2009. [Google Scholar]
- D’Adamo, I.; Falcone, P.M.; Ferella, F. A socio-economic analysis of biomethane in the transport sector: The case of Italy. Waste Manag. 2019, 95, 102–115. [Google Scholar] [CrossRef]
- Barbera, E.; Menegon, S.; Banzato, D.; D’Alpaos, C.; Bertucco, A. From biogas to biomethane: A process simulation-based techno-economic comparison of different upgrading technologies in the Italian context. Renew. Energy 2019, 135, 663–673. [Google Scholar] [CrossRef]
- Adnan, A.I.; Ong, M.Y.; Nomanbhay, S.; Chew, K.W.; Show, P.L. Technologies for biogas upgrading to biomethane: A review. Bioengineering 2019, 6, 92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, C.; Feng, Y.; Wang, X.; Ren, G. Review on research achievements of biogas from anaerobic digestion. Renew. Sustain. Energy Rev. 2015, 45, 540–555. [Google Scholar] [CrossRef]
- Paolini, V.; Petracchini, F.; Segreto, M.; Tomassetti, L.; Naja, N.; Cecinato, A. Environmental impact of biogas: A short review of current knowledge. J. Environ. Sci Heal. Part A 2018, 53, 899–906. [Google Scholar] [CrossRef] [Green Version]
- Zhu, T.; Curtis, J.; Clancy, M. Promoting agricultural biogas and biomethane production: Lessons from cross-country studies. Renew. Sustain. Energy Rev. 2019, 114, 109332. [Google Scholar] [CrossRef]
- Jiang, L.L.; Zhou, J.J.; Quan, C.S.; Xiu, Z.L. Advances in industrial microbiome based on microbial consortium for biorefinery. Bioresour. Bioprocess. 2017, 4, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Batstone, D.J.; Keller, J.; Angelidaki, I.; Kalyuzhnyi, S.V.; Pavlostathis, S.G.; Rozzi, A.; Sanders, W.T.M.; Siegrist, H.; Vavilin, V.A. The IWA Anaerobic Digestion Model No 1 (ADM1). Water Sci. Technol. 2002, 45, 65–73. [Google Scholar] [CrossRef]
- Simeonov, I.; Kroumov, A. A Mathematical Study of the Impact of Methanogenic and Hydrogenotrophic Steps on Biomethane Production from Organic Wastes. Effic Énergétique—Sources D’énergies Renouvelables—Prot L’environnement COFRET’12, Sozopol, Bulg. 2012. Available online: https://hal-riip.archives-ouvertes.fr/pasteur-00755456 (accessed on 9 July 2021).
- Simeonov, I.; Karakashev, D. Mathematical Modelling of the Anaerobic Digestion Including the Syntrophic Acetate Oxidation. IFAC Proc. Vol. 2012, 45, 309–314. [Google Scholar] [CrossRef] [Green Version]
- Giovannini, G.; Sbarciog, M.; Steyer, J.P.; Chamy, R.; Vande Wouwer, A. On the derivation of a simple dynamic model of anaerobic digestion including the evolution of hydrogen. Water Res. 2018, 134, 209–225. [Google Scholar] [CrossRef]
- Borisov, M.; Dimitrova, N.; Simeonov, I. Mathematical modeling and stability analysis of a two-phase biosystem. Processes 2020, 8, 791. [Google Scholar] [CrossRef]
- Weinrich, S.; Nelles, M. Systematic simplification of the Anaerobic Digestion Model No. 1 (ADM1)—Model development and stoichiometric analysis. Bioresour. Technol. 2021, 333, 125124. [Google Scholar] [CrossRef]
- Li, X.; Yang, Z.; Liu, G.; Ma, Z.; Wang, W. Modified anaerobic digestion model No.1 (ADM1) for modeling anaerobic digestion process at different ammonium concentrations. Water Environ. Res. 2019, 91, 700–714. [Google Scholar] [CrossRef]
- Fezzani, B.; Cheikh, R.B. Implementation of IWA anaerobic digestion model No. 1 (ADM1) for simulating the thermophilic anaerobic co-digestion of olive mill wastewater with olive mill solid waste in a semi-continuous tubular digester. Chem. Eng. J. 2008, 141, 75–88. [Google Scholar] [CrossRef]
- Li, J.; Zhou, P.; Liu, H.; Xiong, C.; Lin, J.; Xiao, W.; Gong, Y.; Liu, Z. Synergism of cellulase, xylanase, and pectinase on hydrolyzing sugarcane bagasse resulting from different pretreatment technologies. Bioresour. Technol. 2014, 155, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Lübken, M.; Gehring, T.; Wichern, M. Microbiological fermentation of lignocellulosic biomass: Current state and prospects of mathematical modeling. Appl. Microbiol. Biotechnol. 2010, 85, 1643–1652. [Google Scholar] [CrossRef] [PubMed]
- Bułkowska, K.; Białobrzewski, I.; Gusiatin, Z.M.; Klimiuk, E.; Pokój, T. ADM1-based modeling of anaerobic codigestion of maize silage and cattle manure—Calibration of parameters and model verification (part II). Arch. Environ. Prot. 2015, 41, 20–27. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Chen, Z.; Fu, D.; Wang, Y.; Zheng, Y.; Li, Q. Improved ADM1 for modelling C, N, P fates in anaerobic digestion process of pig manure and optimization approaches to biogas production. Renew. Energy 2020, 146, 2330–2336. [Google Scholar] [CrossRef]
- Biernacki, P.; Steinigeweg, S.; Borchert, A.; Uhlenhut, F. Application of Anaerobic Digestion Model No. 1 for describing anaerobic digestion of grass, maize, green weed silage, and industrial glycerine. Bioresour. Technol. 2013, 127, 188–194. [Google Scholar] [CrossRef]
- Flamholz, A.; Noor, E.; Bar-Even, A.; Milo, R. Equilibrator—The biochemical thermodynamics calculator. Nucleic Acids Res. 2012, 40, D770–D775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Guo, R.B.; Qiu, Y.L.; Qiao, J.T.; Yuan, X.Z.; Shi, X.S.; Wang, C.S. Bioaugmentation with an acetate-type fermentation bacterium Acetobacteroides hydrogenigenes improves methane production from corn straw. Bioresour Technol. 2015, 179, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Ngo, H.H.; Guo, W.S.; Liu, Y.; Nghiem, L.D.; Hai, F.I.; Deng, L.J.; Wang, J.; Wu, Y. Optimization of process parameters for production of volatile fatty acid, biohydrogen and methane from anaerobic digestion. Bioresour Technol. 2016, 219, 738–748. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Luo, K.; Li, X.M.; Wang, D.B.; Zheng, W.; Zeng, G.M.; Liu, J.J. Enhanced efficiency of biological excess sludge hydrolysis under anaerobic digestion by additional enzymes. Bioresour Technol. 2010, 101, 2924–2930. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, J.A.; Harman-Ware, A.E.; Sandgren, M.; Schnürer, A. Biomass recalcitrance in willow under two biological conversion paradigms: Enzymatic hydrolysis and anaerobic digestion. Bioenergy Res. BioEnergy Res. 2020, 13, 260–270. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Cheng, S.; Li, Z.; Men, Y.; Wu, J. Impacts of cellulase and amylase on enzymatic hydrolysis and methane production in the anaerobic digestion of corn straw. Sustainability 2020, 12, 5453. [Google Scholar] [CrossRef]
- Filer, J.; Ding, H.H.; Chang, S. Biochemical Methane Potential (BMP) Assay Method for Anaerobic Digestion Research. Water 2019, 11.5, 921. [Google Scholar] [CrossRef] [Green Version]
- IEA Bioenergy Task40. Cascading of Woody Biomass: Definitions, Policies and Effects on International Trade; IEA Bioenergy: Stockholm, Sweden, 2016; p. 71. [Google Scholar]
- Mlaik, N.; Khoufi, S.; Hamza, M.; Masmoudi, M.A.; Sayadi, S. Enzymatic pre-hydrolysis of organic fraction of municipal solid waste to enhance anaerobic digestion. Biomass Bioenergy 2019, 127, 105286. [Google Scholar] [CrossRef]
| Reaction | Estimated ΔrG’° (KJ/Mol) | K’eq |
|---|---|---|
| (1) Glucose + 2 H2O <=> 4 CO2 + 2 CH4 + 4 H2 | −148.6 ± 35.0 | 1.1 × 1026 |
| (2) Glucose + 2 NAD+ + 2 H2O <=> 4 CO2 + 2 CH4 + 2 H2 + 2 NADH | −217.7 ± 28.6 | 1.5 × 1038 |
| (3) Glucose + 4 NAD+ + 2 H2O <=> 4 CO2 + 2 CH4 + 4 NADH | −286.9 ± 26.3 | 1.9 × 1050 |
| Parameter | Value | Ratio | Q Value | Q Ratio | Q Ratio/Parameter Value Ratio |
|---|---|---|---|---|---|
| Yacet/X1 | 20 | 1.198 | |||
| 40 | 2 | 1.933 | 1.62 | 0.81 | |
| 60 | 3 | 2.548 | 2.13 | 0.71 | |
| S1i (g/L) | 5.1 | 1.198 | |||
| 10.2 | 2 | 1.504 | 1.26 | 0.63 | |
| 15.3 | 3 | 1.807 | 1.51 | 0.50 | |
| S0i (g/L) | 30.6 | 1.198 | |||
| 61.2 | 2 | 2.001 | 1.67 | 0.84 | |
| 91.8 | 3 | 2.796 | 2.33 | 0.78 | |
| Ye | 0.55 | 1.198 | |||
| 1.1 | 2 | 2.001 | 1.67 | 0.84 | |
| 1.65 | 3 | 2.796 | 2.33 | 0.78 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertacchi, S.; Ruusunen, M.; Sorsa, A.; Sirviö, A.; Branduardi, P. Mathematical Analysis and Update of ADM1 Model for Biomethane Production by Anaerobic Digestion. Fermentation 2021, 7, 237. https://doi.org/10.3390/fermentation7040237
Bertacchi S, Ruusunen M, Sorsa A, Sirviö A, Branduardi P. Mathematical Analysis and Update of ADM1 Model for Biomethane Production by Anaerobic Digestion. Fermentation. 2021; 7(4):237. https://doi.org/10.3390/fermentation7040237
Chicago/Turabian StyleBertacchi, Stefano, Mika Ruusunen, Aki Sorsa, Anu Sirviö, and Paola Branduardi. 2021. "Mathematical Analysis and Update of ADM1 Model for Biomethane Production by Anaerobic Digestion" Fermentation 7, no. 4: 237. https://doi.org/10.3390/fermentation7040237
APA StyleBertacchi, S., Ruusunen, M., Sorsa, A., Sirviö, A., & Branduardi, P. (2021). Mathematical Analysis and Update of ADM1 Model for Biomethane Production by Anaerobic Digestion. Fermentation, 7(4), 237. https://doi.org/10.3390/fermentation7040237







