Bored Coffee Beans for Production of Hyaluronic Acid by Streptococcus zooepidemicus
Abstract
:1. Introduction
2. Materials and Methods
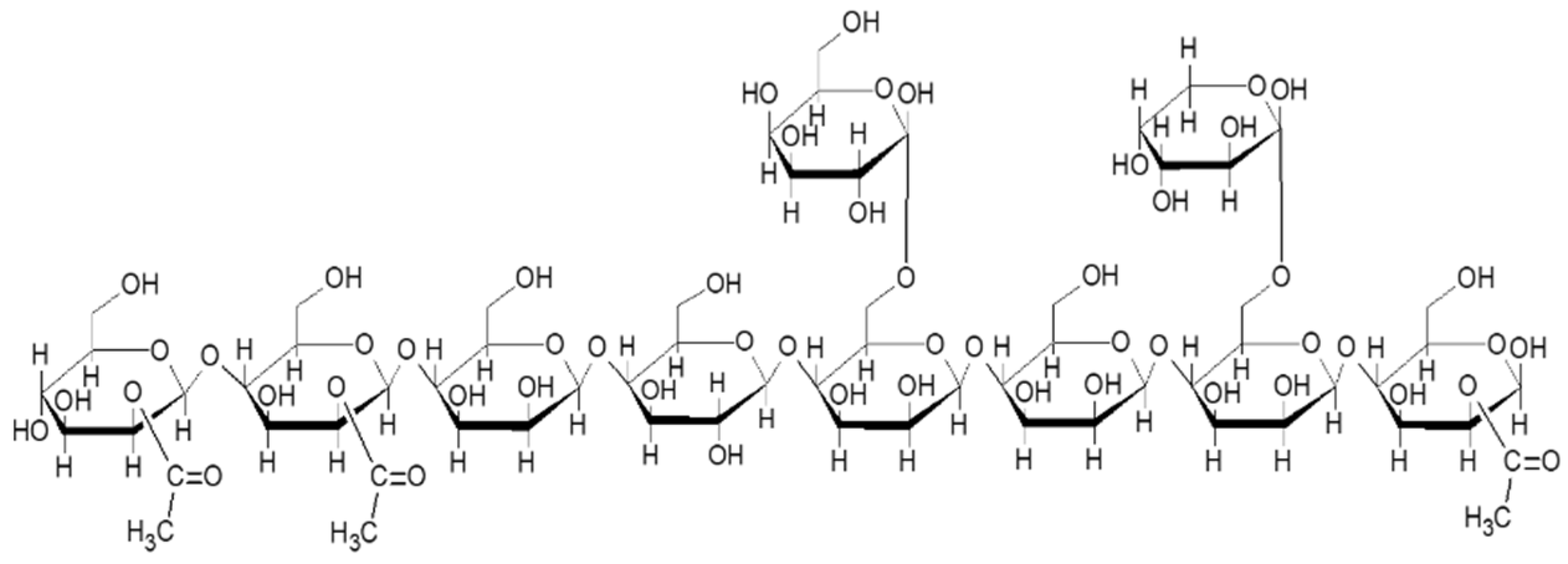
2.1. Chemical Composition of Bored Coffee Beans
2.2. Hydrolysis of Bored Coffee Beans
2.3. Post-Treatment of Acid Hydrolyzates
2.4. Microorganism and Culture Conditions
2.5. Kinetic Parameters
3. Results
3.1. Chemical Composition of Bored Coffee Beans
3.2. Acid Hydrolysis of Bored Coffee Beans and Acetone-Extracted Bored Coffee Beans
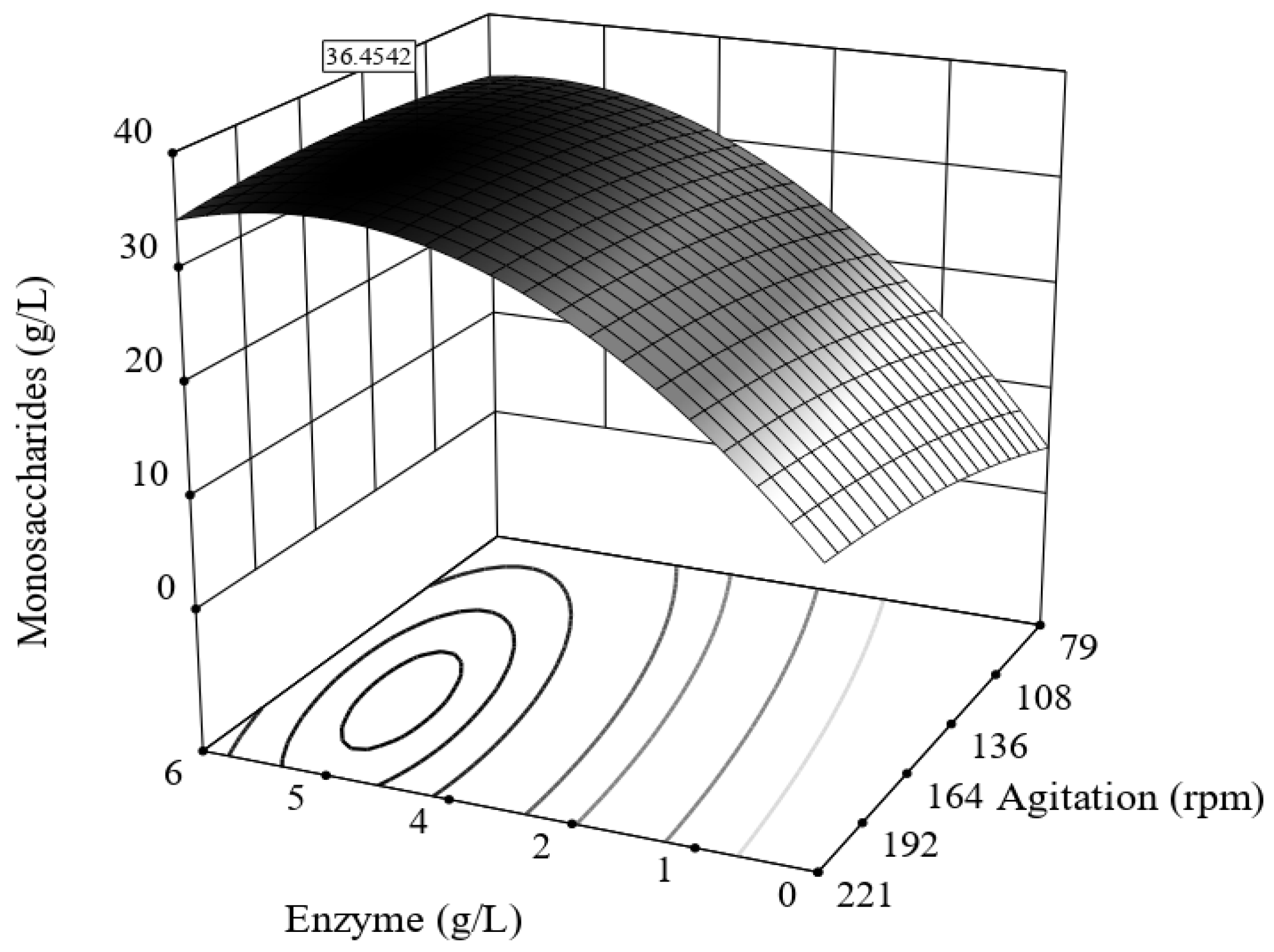
3.3. Enzymatic Hydrolysis of Bored Coffee Beans and Acetone-Extracted Bored Coffee Beans
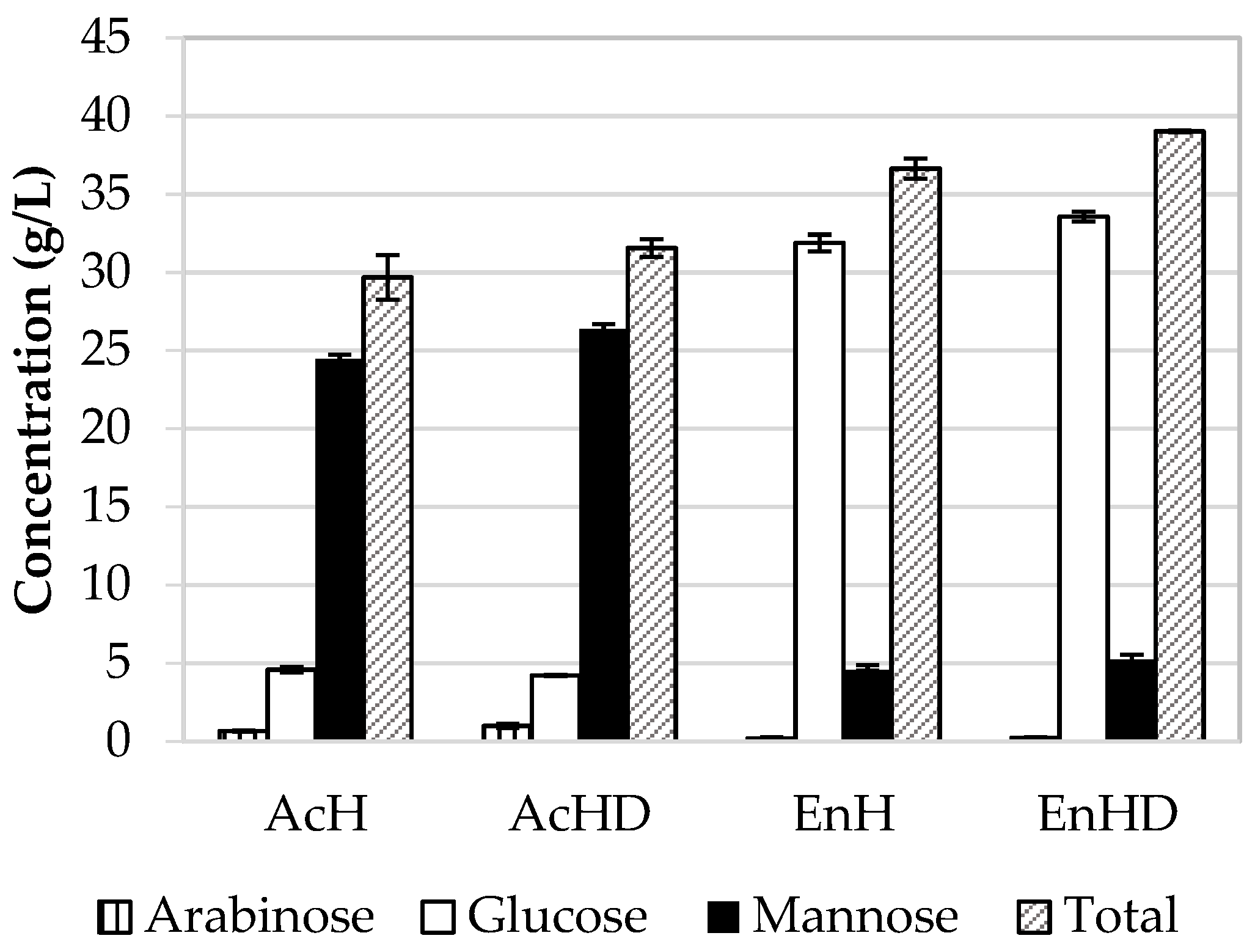
3.4. Comparison between Hydrolytic Treatments
3.5. Post-Treatment of Acid Hydrolyzates
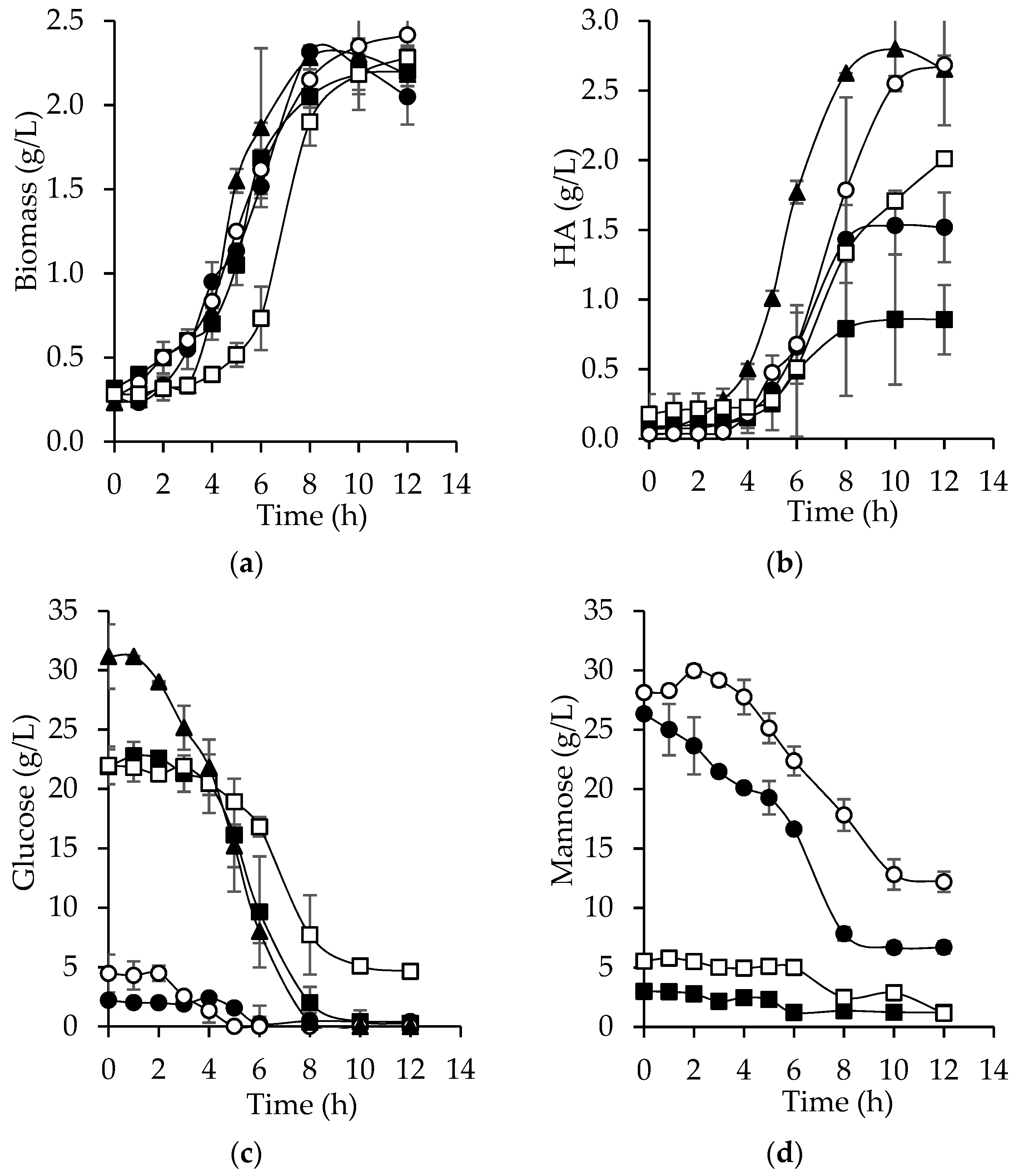
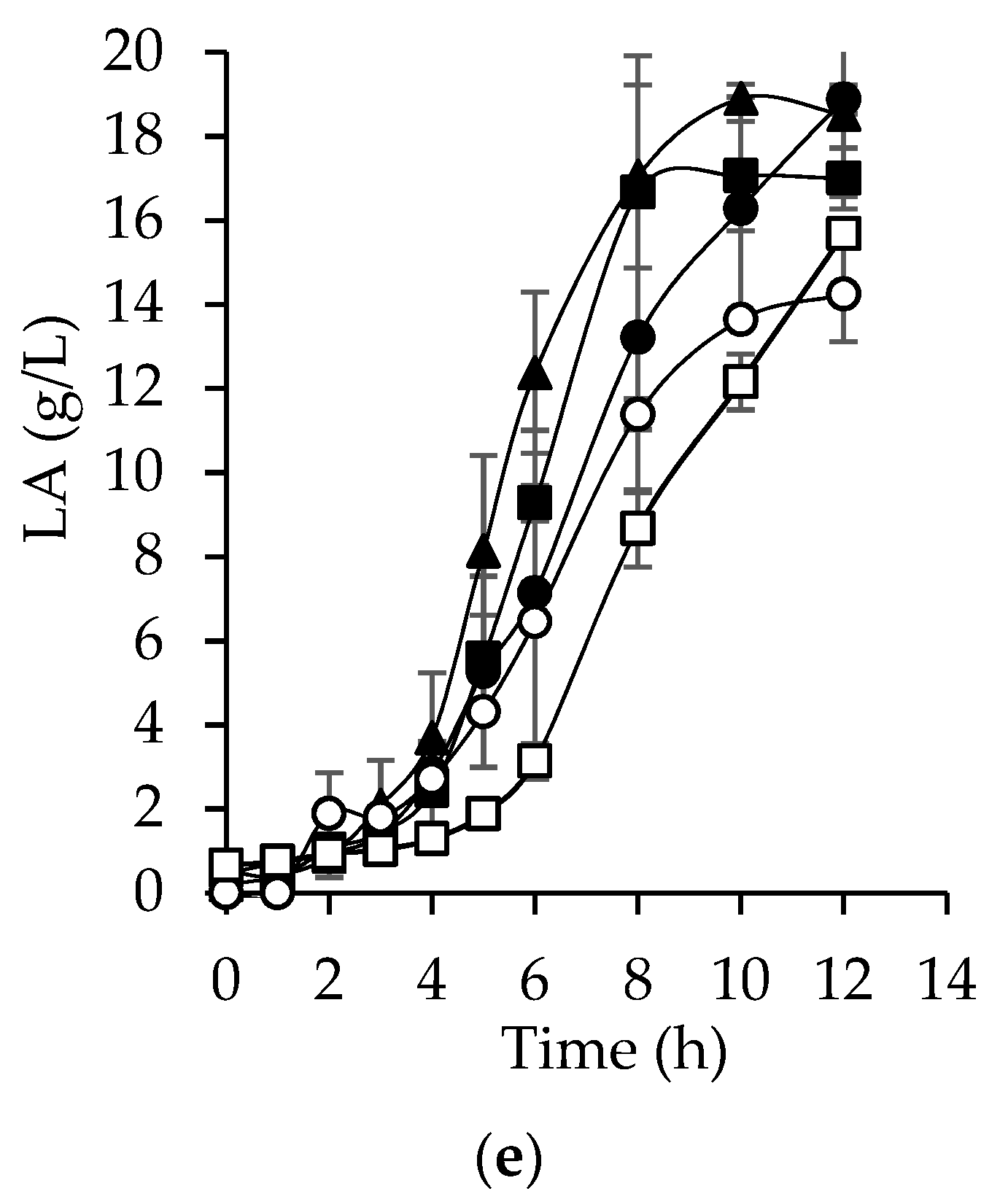
3.6. Hyaluronic Acid Production from Bored Coffee Beans Hydrolyzates
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Farah, A. Coffee Constituents. In Coffee: Emerging Health Effects and Disease Prevention, 1st ed.; Chu, Y., Ed.; John Wiley & Sons, Inc.; IFT Press: Ames, IA, USA, 2012; pp. 21–58. [Google Scholar]
- International Coffee Organization. Available online: http://www.ico.org/prices/new-consumption-table.pdf (accessed on 15 February 2020).
- Pava-ripoll, M.; Posada, F.J.; Momen, B.; Wang, C.; Leger, R. Increased pathogenicity against coffee berry borer, Hypothenemus hampei (Coleoptera: Curculionidae) by Metarhizium anisopliae expressing the scorpion toxin (AaIT) gene. J. Invertebr. Pathol. 2008, 99, 220–226. [Google Scholar] [CrossRef]
- López, E.; Castaño, J. Extracción de aceite a partir de subproductos de la trilla de café pergamino. Cenicafé 1999, 50, 66–77. [Google Scholar]
- Murthy, P.S.; Madhava Naidu, M. Sustainable management of coffee industry by-products and value addition—A review. Resour. Conserv. Recycl. 2012, 66, 45–58. [Google Scholar] [CrossRef]
- Rohit, S.G.; Jyoti, P.K.; Subbi, R.R.T.; Naresh, M.; Senthilkumar, S. Kinetic modeling of hyaluronic acid production in palmyra palm (Borassus flabellifer) based medium by Streptococcus zooepidemicus MTCC 3523. Biochem. Eng. J. 2018, 137, 284–293. [Google Scholar] [CrossRef]
- Pan, N.C.; Vignoli, J.A.; Baldo, C.; Braga, H.C.; Da Silva, R.S.; Pedrine, M.A. Agroindustrial byproducts for the production of hyaluronic acid by Streptococcus zooepidemicus ATCC 39920. Int. J. Sci. Technol. Res. 2015, 4, 114–118. [Google Scholar]
- Zakeri, A.; Rasaee, M.J.; Pourzardosht, N. Enhanced hyluronic acid production in Streptococcus zooepidemicus by over expressing HasA and molecular weight control with Niscin and glucose. Biotechnol. Rep. 2017, 16, 65–70. [Google Scholar] [CrossRef]
- Don, M.M.; Shoparwe, N.F. Kinetics of hyaluronic acid production by Streptococcus zooepidemicus considering the effect of glucose. Biochem. Eng. J. 2010, 49, 95–103. [Google Scholar] [CrossRef]
- Chahuki, F.F.; Aminzadeh, S.; Jafarian, V.; Tabandeh, F.; Khodabandeh, M. Hyaluronic acid production enhancement via genetically modification and culture medium optimization in Lactobacillus acidophilus. Int. J. Biol. Macromol. 2019, 121, 870–881. [Google Scholar] [CrossRef]
- Amado, I.R.; Vázquez, J.A.; Pastrana, L.; Teixeira, J.A. Cheese whey: A cost-effective alternative for hyaluronic acid production by Streptococcus zooepidemicus. Food Chem. 2016, 198, 54–61. [Google Scholar] [CrossRef] [Green Version]
- TAPPI T 264 cm-97. Preparation of Wood for Chemical Analysis; TAPPI Press: Atlanta, GA, USA, 1997. [Google Scholar]
- TAPPI T 211 om-02. Ash in Wood, Pulp, Paper and Paperboard: Combustion at 525 °C; TAPPI Press: Atlanta, GA, USA, 2002. [Google Scholar]
- TAPPI T 207 cm-99. Water Solubility of Wood and Pulp; TAPPI Press: Atlanta, GA, USA, 1999. [Google Scholar]
- TAPPI T 204 Cm-97. Solvent Extractives of Wood and Pulp; TAPPI Press: Atlanta, GA, USA, 2007. [Google Scholar]
- TAPPI T 222 om-02. Lignin in Wood and Pulp; TAPPI Press: Atlanta, GA, USA, 2006. [Google Scholar]
- Wise, L.; Murphy, M.; D’Addieco, A. Chlorite holocellulose, its fractionation and bearing on summative wood analysis and on studies on the hemicellulose. Pap. Trade J. 1946, 122, 35–43. [Google Scholar]
- Cross, C.; Bevan, E. Researches on Cellulose, 2nd ed.; Longmans, green, and Co.: London, UK, 1907; Volume 2. [Google Scholar]
- Shah, M.V.; Badle, S.S.; Ramachandran, K.B. Hyaluronic acid production and molecular weight improvement by redirection of carbon flux towards its biosynthesis pathway. Biochem. Eng. J. 2013, 80, 53–60. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Machado, E.M.S.; Martins, S.; Teixeira, J.A. Production, composition, and application of coffee and its industrial residues. Food Bioprocess Technol. 2011, 4, 661–672. [Google Scholar] [CrossRef] [Green Version]
- Redgwell, R.; Fischer, M. Coffee carbohydrates. Braz. J. Plant Physiol. 2006, 18, 165–174. [Google Scholar] [CrossRef]
- Oliveira, L.; Franca, A.; Mendonça, J.; Barros-Júnior, M. Proximate composition and fatty acids profile of green and roasted defective coffee beans. LWT Food Sci. Technol. 2006, 39, 235–239. [Google Scholar] [CrossRef]
- Cho, E.J.; Thi, L.; Trinh, P.; Song, Y.; Lee, Y.G.; Bae, H. Bioconversion of biomass waste into high value chemicals. Bioresour. Technol. 2020, 298, 122386. [Google Scholar] [CrossRef] [PubMed]
- Constantino, L.V.; Zeffa, D.M.; Koltun, A.; Urbano, M.R.; Santos, A.W.; Nixdorf, S.L. Extraction of soluble sugars from green coffee beans using hot water and quantification by a chromatographic method without an organic solvent. Acta Chromatogr. 2020, 32, 242–246. [Google Scholar] [CrossRef]
- Nuhu, A.A. Bioactive micronutrients in coffee: Recent analytical approaches for characterization and quantification. ISRN Nutr. 2014, 2014, 1–13. [Google Scholar] [CrossRef]
- Juarez, G.F.Y.; Pabiloña, K.B.C.; Manlangit, K.B.L.; Go, A.W. Direct dilute acid hydrolysis of spent coffee grounds: A new approach in sugar and lipid recovery. Waste Biomass Valorization 2018, 9, 235–246. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, R.; Singh, A.; Kuhad, R.C. Detoxification of sugarcane bagasse hydrolysate improves ethanol production by Candida shehatae NCIM 3501. Bioresour. Technol. 2007, 98, 1947–1950. [Google Scholar] [CrossRef]
- Pedroso, G.; Philippsen, M.; Saldanha, L.; Araujo, R.; Martins, A. Strategies for fermentable sugar production by using pressurized acid hydrolysis for rice husks. Rice Sci. 2019, 26, 319–330. [Google Scholar] [CrossRef]
- Kupiainen, L.; Ahola, J.; Tanskanen, J. Kinetics of glucose decomposition in formic acid. Chem. Eng. Res. Des. 2011, 89, 2706–2713. [Google Scholar] [CrossRef]
- Taherzadeh, M.J.; Gustafsson, L.; Niklasson, C.; Lidén, G. Physiological effects of 5-hydroxymethylfurfural on Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 2000, 53, 701–708. [Google Scholar] [CrossRef]
- Kwon, E.E.; Yi, H.; Jeon, Y.J. Sequential co-production of biodiesel and bioethanol with spent coffee grounds. Bioresour. Technol. 2013, 136, 475–480. [Google Scholar] [CrossRef]
- Niglio, S.; Procentese, A.; Russo, M.E. Investigation of enzymatic hydrolysis of coffee silverskin aimed at the production of butanol and succinic acid by fermentative processes. BioEnergy Res. 2019, 12, 312–324. [Google Scholar] [CrossRef]
- Seong, I.; Gon, S.; Kim, S.; Bae, H. Conversion of coffee residue waste into bioethanol with using popping pretreatment. Bioresour. Technol. 2012, 125, 132–137. [Google Scholar] [CrossRef]
- Chaves-Ulate, E.C.; Esquivel-Rodríguez, P. Ácidos clorogénicos presentes en el café: Capacidad antimicrobiana y antioxidante. Agron. Mesoam. 2019, 30, 299–311. [Google Scholar] [CrossRef]
- Berlin, A.; Gilkes, N.; Kilburn, D.; Bura, R.; Markov, A.; Skomarovsky, A.; Okunev, O.; Gusakov, A.; Maximenko, V.; Gregg, D.; et al. Evaluation of novel fungal cellulase preparations for ability to hydrolyze softwood substrates—Evidence for the role of accessory enzymes. Enzym. Microb. Technol. 2005, 37, 175–184. [Google Scholar] [CrossRef]
- Inoue, H.; Yano, S.; Sawayama, S. Effect of β-Mannanase and β-Mannosidase Supplementation on the total hydrolysis of softwood polysaccharides by the Talaromyces cellulolyticus cellulase system. Appl. Biochem. Biotechnol. 2015, 176, 1673–1686. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C. Polysaccharides in coffee and their relationship to health: An overview. In Coffee in Health and Disease Prevention, 1st ed.; Preedy, V.R., Ed.; Elsevier Inc.: San Diego, CA, USA, 2015; pp. 163–171. [Google Scholar]
- Nelson, D.L.; Cox, M.M. Lehninger Principles of Biochemistry, 7th ed.; W. H. Freeman: New York, USA, 2017; pp. 1–1328. [Google Scholar]
- Chen, X.; Shekiro, J.; Franden, M.A.; Wang, W.; Zhang, M.; Kuhn, E.; Johnson, D.K.; Tucker, M.P. The impacts of deacetylation prior to dilute acid pretreatment on the bioethanol process. Biotechnol. Biofuels 2012, 5, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohan, N.; Pavan, S.S.; Achar, A.; Swaminathan, N.; Sivaprakasam, S. Calorespirometric investigation of Streptococcus zooepidemicus metabolism: Thermodynamics of anabolic payload contribution by growth and hyaluronic acid synthesis. Biochem. Eng. J. 2019, 152, 107367. [Google Scholar] [CrossRef]
- Oliveira, A.H.; Ogrodowski, C.C.; De Macedo, A.C.; Santana, M.H.A.; Gonçalves, L.R.B. Cashew apple juice as microbial cultivation medium for non-immunogenic hyaluronic acid production. Braz. J. Microbiol. 2013, 1097–1104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Liu, Y.; Li, J.; Du, G.; Chen, J. Microbial production of hyaluronic acid: Current state, challenges, and perspectives. Microb. Cell Fact. 2011, 10, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eş, I.; Khaneghah, A.M.; Barba, F.J.; Saraiva, J.A.; Sant’Ana, A.S.; Bagher, S.M. Recent advancements in lactic acid production—A review. Food Res. Int. 2018, 107, 763–770. [Google Scholar] [CrossRef] [PubMed]

| Content (% wt) | Bored Coffee Beans |
|---|---|
| Holocellulose | 58.5 ± 1.44 |
| Cellulose | 31.6 ± 3.69 |
| Galactomannans and other hemicelluloses | 26.9 ± 3.28 |
| Lignin | 2.1 ± 0.23 |
| Water soluble extractives (arabinogalactans and pectins) | 25.4 ± 0.77 |
| Organic solvent extractives | 11.9 ± 0.17 |
| Ash | 2.0 ± 0.12 |
| Moisture content | 12.6± 0.21 |
| Run | Acid Hydrolysis | Response | Yield | |
|---|---|---|---|---|
| HCl (% v/v) | Temperature (°C) | Monosaccharides (g/L) | δAcH (%) | |
| 1 | 1 | 110 | 18.2 | 32.0 |
| 2 | 2 | 110 | 22.2 | 39.1 |
| 3 | 1 | 130 | 27.2 | 47.9 |
| 4 | 2 | 130 | 32.5 | 57.2 |
| 5 | 1.5 | 105.85 | 10.9 | 19.2 |
| 6 | 1.5 | 134.14 | 29.4 | 51.7 |
| 7 | 0.79 | 120 | 22.6 | 39.8 |
| 8 | 2.20 | 120 | 29.0 | 51.0 |
| 9 | 1.5 | 120 | 24.7 | 43.5 |
| 10 | 1.5 | 120 | 22.2 | 39.1 |
| 11 | 1.5 | 120 | 23.2 | 40.7 |
| 12 | 1.5 | 120 | 22.1 | 38.9 |
| 13 | 1.5 | 120 | 23.3 | 41.0 |
| Run Number | Enzymatic Hydrolysis | Response | Yield | |
|---|---|---|---|---|
| Enzyme (g/L) | Agitation (rpm) | Monosaccharides * (g/L) | δEnH (%) | |
| 1 | 1 | 100 | 14.4 | 25.4 |
| 2 | 5 | 100 | 36.4 | 64.0 |
| 3 | 1 | 200 | 18.4 | 32.5 |
| 4 | 5 | 200 | 36.1 | 63.5 |
| 5 | 3 | 79.28 | 27.9 | 49.2 |
| 6 | 3 | 220.7 | 34.7 | 61.0 |
| 7 | 0.17 | 150 | 12.0 | 21.2 |
| 8 | 5.8 | 150 | 34.4 | 60.7 |
| 9 | 3 | 150 | 28.3 | 49.9 |
| 10 | 3 | 150 | 29.1 | 51.2 |
| 11 | 3 | 150 | 35.8 | 63.0 |
| 12 | 3 | 150 | 34.7 | 61.1 |
| 13 | 3 | 150 | 32.3 | 56.9 |
| AcH | AcHD | |||||
|---|---|---|---|---|---|---|
| Before | After | Removal | Before | After | Removal | |
| Parameter | Concentration (g/L) | % | Concentration (g/L) | % | ||
| Monosaccharides | 28.6 | 21.7 | 24.1 | 34.5 | 28.3 | 17.9 |
| Formic acid | 0.25 | 0.06 | 76.0 | 1.32 | 0.22 | 83.3 |
| Furfural | 0.24 | 0.02 | 91.6 | 0.23 | 0.01 | 95.6 |
| Hydrometylfurfural | 0.85 | 0.22 | 74.1 | 0.42 | 0.08 | 80.9 |
| pH | 0.86 * | 6.97 * | WR | 0.91 * | 7.04 * | WR |
| Variables | SM | AcH | EnH | AcHD | EnHD |
|---|---|---|---|---|---|
| Biomass (g/L) | 2.3 ± 0.235 | 2.2 ± 0.047 | 2.2 ± 0.141 | 2.4 ± 0.117 | 2.3 ± 0.070 |
| HA (g/L) | 2.8 ± 0.243 | 1.5 ± 0.249 | 0.85 ± 0.467 | 2.7 ± 0.067 | 2.0 ± 0.002 |
| Lactic acid (g/L) | 18.5 ± 1.950 | 16.9 ± 0.727 | 18.8 ± 0.345 | 14.2 ± 1.133 | 15.6 ± 0.252 |
| Glucose (g/L) | 31.1 ± 2.713 | 2.2 ± 0.067 | 21.8 ± 1.468 | 4.4 ± 0.300 | 22.0 ± 1.593 |
| Mannose (g/L) | NP | 26.3 ± 0.302 | 3.0 ± 0.010 | 28.1 ± 0.182 | 5.53 ± 0.780 |
| µmax (1/h) | 0.28 ± 0.001 | 0.27 ± 0.023 | 0.24 ± 0.040 | 0.24 ± 0.005 | 0.26 ± 0.003 |
| qG-max (g/g h) | 3.6 ± 0.087 | 0.17 ± 0.075 | 3.4 ± 0.613 | 0.69 ± 0.017 | 2.4 ± 0.226 |
| qM-max (g/g h) | NC | 2.1 ± 0.036 | 0.36 ± 0.026 | 1.8 ± 0.223 | 0.53 ± 0.045 |
| qLA-max (g/g h) | 2.2 ± 0.333 | 2.1 ± 0.058 | 2.2 ± 0.118 | 1.5 ± 0.127 | 1.1 ± 0.017 |
| qHA-max (g/g h) | 0.34 ± 0.022 | 0.20 ± 0.007 | 0.11 ± 0.060 | 0.27 ± 0.119 | 0.21 ± 0.033 |
| rLA (g/L h) | 1.9 ± 0.034 | 1.6 ± 1.710 | 1.6 ± 0.267 | 1.4 ± 0.016 | 1.3 ± 0.021 |
| rHA (g/L h) | 0.28 ± 0.024 | 0.15 ± 0.021 | 0.09 ± 0.047 | 0.25 ± 0.005 | 0.17 ± 0.004 |
| YX/S (g/g) | 0.06 ± 0.009 | 0.09 ± 0.007 | 0.08 ± 0.011 | 0.10 ± 0.001 | 0.09 ± 0.009 |
| YLA/S (g/g) | 0.60 ± 0.006 | 0.79 ± 0.030 | 0.68 ± 0.150 | 0.69 ± 0.070 | 0.59 ± 0.015 |
| YHA/S (g/g) | 0.09 ± 0.009 | 0.07 ± 0.014 | 0.04 ± 0.019 | 0.13 ± 0.009 | 0.08 ± 0.003 |
| Microorganism | Culture Medium | Sugars (g/L) | HA (g/L) | Reference |
|---|---|---|---|---|
| S. zooepidemicus 39920 | Soy molasses | 30 | ND | [7] |
| Sugarcane molasses | 30 | 0.37 | ||
| Sugarcane juice | 30 | 0.26 | ||
| S. zooepidemicus MTCC 3523 | Palmyra palm sugar | 30 | 1.2 | [6] |
| S. zooepidemicus 39920 | Cashew apple juice | 90 | 1.8 | [41] |
| S. zooepidemicus 35246 | Cheese whey | 100 | 2.1 | [11] |
| Cheese whey hydrolyzate | 100 | 2.4 | ||
| S. zooepidemicus 35246 | AcH | 28 | 1.5 | This work |
| EnH | 24 | 0.85 | ||
| AcHD | 32 | 2.7 | ||
| EnHD | 27 | 2.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores-Méndez, D.A.; Ramos-Ibarra, J.R.; Toriz, G.; Arriola-Guevara, E.; Guatemala-Morales, G.; Corona-González, R.I. Bored Coffee Beans for Production of Hyaluronic Acid by Streptococcus zooepidemicus. Fermentation 2021, 7, 121. https://doi.org/10.3390/fermentation7030121
Flores-Méndez DA, Ramos-Ibarra JR, Toriz G, Arriola-Guevara E, Guatemala-Morales G, Corona-González RI. Bored Coffee Beans for Production of Hyaluronic Acid by Streptococcus zooepidemicus. Fermentation. 2021; 7(3):121. https://doi.org/10.3390/fermentation7030121
Chicago/Turabian StyleFlores-Méndez, David Antonio, José Roberto Ramos-Ibarra, Guillermo Toriz, Enrique Arriola-Guevara, Guadalupe Guatemala-Morales, and Rosa Isela Corona-González. 2021. "Bored Coffee Beans for Production of Hyaluronic Acid by Streptococcus zooepidemicus" Fermentation 7, no. 3: 121. https://doi.org/10.3390/fermentation7030121
APA StyleFlores-Méndez, D. A., Ramos-Ibarra, J. R., Toriz, G., Arriola-Guevara, E., Guatemala-Morales, G., & Corona-González, R. I. (2021). Bored Coffee Beans for Production of Hyaluronic Acid by Streptococcus zooepidemicus. Fermentation, 7(3), 121. https://doi.org/10.3390/fermentation7030121








