Processes, Challenges and Optimisation of Rum Production from Molasses—A Contemporary Review
Abstract
1. Introduction
1.1. Definition of Rum
- (1)
- ‘a spirit drink produced exclusively by alcoholic fermentation and distillation, either from molasses or syrup produced in the manufacture of cane sugar or from sugar-cane juice itself and distilled at less than 96% volume so that the distillate has the discernible specific organoleptic characteristics of rum.’
- (2)
- ‘a spirit drink produced exclusively by alcoholic fermentation and distillation of sugar-cane juice which has the aromatic characteristics specific to rum and a volatile substances content equal to or exceeding 225 g per hectolitre of 100% vol. alcohol. This spirit may be placed on the market with the word ‘agricultural’ qualifying the sales denomination ‘rum’ accompanied by any of the geographical indications of the French Overseas Departments and the Autonomous Region of Madeira as registered in Annex III.’ [11]
- (1)
- ‘a spirit obtained exclusively by alcoholic fermentation and distillation of sugar cane molasses, sugar cane syrups, sugar cane juices or cane sugar produced during the processing of sugar cane.’
- (2)
- ‘a spirit drink distilled at an alcohol content of less than 96.0% alcohol by volume at 20 °C.’
- (3)
- ‘a spirit drink produced in such a way that the product has the organoleptic characteristics derived from the natural volatile elements contained in the above raw materials or formed during the fermentation or distillation process of the named raw materials; and which includes mixtures solely of the above distillate.’ [12]
1.2. Types of Rum
1.3. Global Rum Production Statistics
1.4. Review Aim
2. Molasses: The Feedstock for Rum Production
2.1. Production of Molasses
2.2. Physical and Chemical Properties of Sugar Cane Molasses
2.3. Impact of Sugarcane Cultivation on Molasses Quality
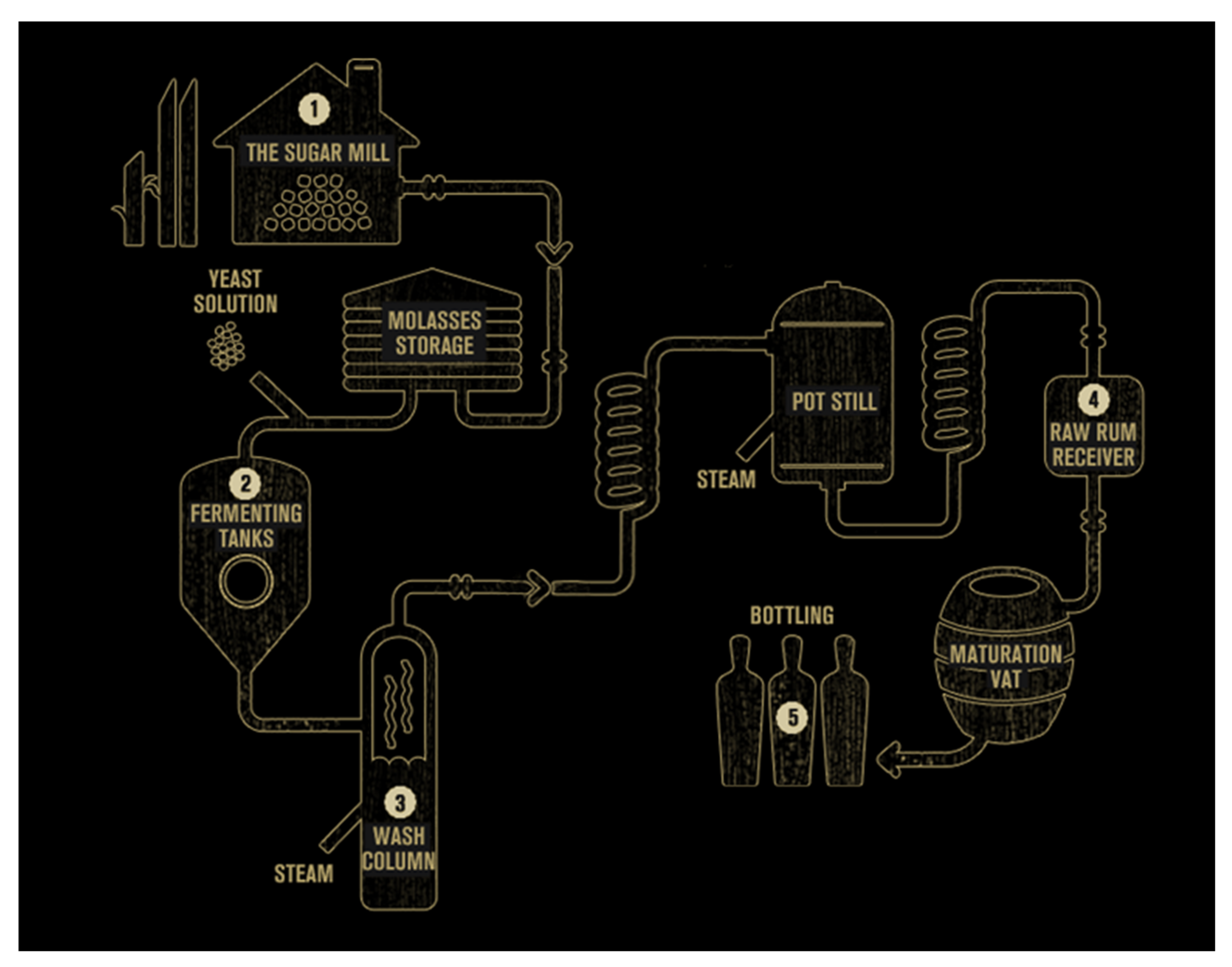
3. Rum Production Processes
3.1. Pre-Treatment of Molasses
3.2. Yeast Propagation
3.3. Rum Fermentation Processes
3.4. Distillation
3.5. Post-Fermentation Processes: Maturation and Aging
4. Composition of Rum
4.1. General Composition
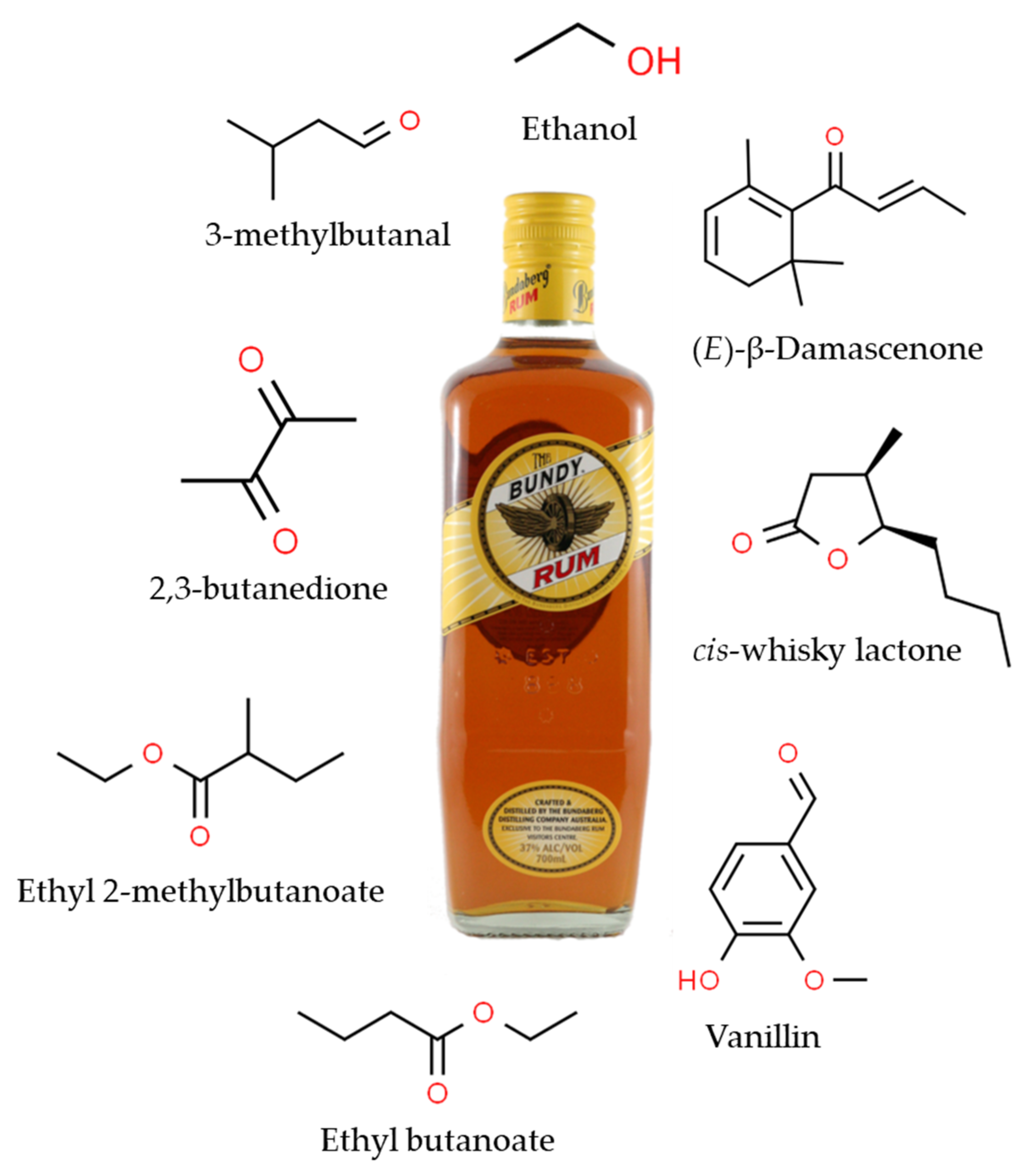
4.2. Flavour-Active Compounds
4.3. Other Constituents
5. Quality Management in Rum Production
6. Current Challenges in the Fermentation Process—Globally
6.1. Feedstock Variation
6.2. Incomplete Fermentation
6.3. Yeast Strains and Health
6.4. Microbial Contamination
6.5. Process Control and Optimization
7. Future Directions and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pounder, P. Branding: A Caribbean perspective on rum manufacturing competitiveness. Int. J. Entrep. Small Bus. 2010, 9, 394–406. [Google Scholar] [CrossRef]
- Pino, J.A. Characterization of rum using solid-phase microextraction with gas chromatography-mass spectrometry. Food Chem. 2007, 104, 421–428. [Google Scholar] [CrossRef]
- Piggot, R. Treatment and fermentation of molasses when making rum-type spirits. In The Alcohol Textbook, 4th ed.; Kelsall, D., Jacques, K., Lyons, T., Eds.; Nottingham University Press: Nottingham, UK, 2003; pp. 75–84. [Google Scholar]
- Leal, M. Ethanol production from cane resources. In Bioenergy for Sustainable Development and International Competitiveness: The Role of Sugar Cane in Africa; Johnson, F.X., Seebaluck, V., Eds.; Routledge: New York, NY, USA, 2012; pp. 126–157. [Google Scholar]
- Lisle, P. Rum beginnings: Towards a new perspective of the Grose years. J. R. Aust. Hist. Soc. 2005, 91, 15–28. [Google Scholar]
- Nicol, D. Rum. In Fermented Beverage Production, 2nd ed.; Lea, A.G., Piggott, J.R., Eds.; Springer: Boston, MA, USA, 2003. [Google Scholar]
- Liberman, A. The Rum History of the Word “Rum”. Available online: https://blog.oup.com/2010/10/rum/ (accessed on 17 November 2020).
- FSANZ (Food Standards Australia & New Zealand). Standard 2.7.5—Spirits; Technical Report No. 2.7.5; FSANZ: Canberra, Australia, 2000.
- Australian Distillers Association. Guidance Summary: Legal Definitions of Brandy, Rum, Whisky; Australian Distillers Association: Sydney, Australia, 2017. [Google Scholar]
- US Government. 27 CFR and 5.22—The Standards of Identity; US Government: Washington, WA, USA, 2010.
- The Council of European Communities. Appellations of Origin (Spirit Drinks) Council Regulations; Technical Report No. 1576/89; The Council of European Communities: Brussels, Belgium, 1989. [Google Scholar]
- CROSQ (CARICOM Regional Organisation for Standards and Quality). CARICOM Regional Standard for Rum: Specifications; Technical Report No. CRS 25: 2008; CARICOM: Belleville, St Michael, Barbados, 2008. [Google Scholar]
- Murtagh, J.E. Feedstocks, fermentation and distillation for production of heavy and light rums. In The Alcohol Textbook, 4th ed.; Kelsall, D., Jacques, K., Lyons, T., Eds.; Nottingham University Press: Nottingham, UK, 2003; pp. 243–255. [Google Scholar]
- Fahrasmane, L.; Parfait, A. Rum. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Caballero, B., Ed.; Academic Press: Oxford, UK, 2003; pp. 5021–5027. [Google Scholar] [CrossRef]
- Medeiros, A.B.P.; de Matos, M.E.; de Pinho Monteiro, A.; de Carvalho, J.C.; Soccol, C.R. 16—Cachaça and Rum. In Current Developments in Biotechnology and Bioengineering; Pandey, A., Sanromán, M.Á., Du, G., Soccol, C.R., Dussap, C.-G., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 451–468. [Google Scholar] [CrossRef]
- Furiassi, C. Towards a glossary of rum making and rum tasting. In Proceedings of the XVIII EURALEX International Congress, Ljubljana, Slovenia, 17–21 July 2018; p. 139. [Google Scholar]
- Statista. Rum: Worldwide. Available online: https://www.statista.com/outlook/10020300/100/rum/worldwide (accessed on 8 December 2020).
- Hamrick, D.; Fernandez-Stark, K. Barbados in the Rum Global Value Chain; Duke Global Value Chain Center, Duke University: Durham, NC, USA, 2017. [Google Scholar]
- Pandey, V.C.; Bajpai, O.; Pandey, D.N.; Singh, N. Saccharum spontaneum: An underutilized tall grass for revegetation and restoration programs. Genet. Resour. Crop Evol. 2015, 62, 443–450. [Google Scholar] [CrossRef]
- Castro, S.G.Q.D.; Franco, H.C.J.; Mutton, M.Â. Harvest managements and cultural practices in sugarcane. Rev. Bras. Ciência Solo 2014, 38, 299–306. [Google Scholar] [CrossRef]
- Park, M.J.; Jo, J.H.; Park, D.; Lee, D.S.; Park, J.M. Comprehensive study on a two-stage anaerobic digestion process for the sequential production of hydrogen and methane from cost-effective molasses. Int. J. Hydrog. Energy 2010, 35, 6194–6202. [Google Scholar] [CrossRef]
- Coelho, C.; Brottier, C.; Beuchet, F.; Elichiry-Ortiz, P.; Bach, B.; Lafarge, C.; Tourdot-Maréchal, R. Effect of ageing on lees and distillation process on fermented sugarcane molasses for the production of rum. Food Chem. 2020, 303, 125405. [Google Scholar] [CrossRef]
- Fleet, G.H.; Green, V. The microbiology and biotechnology of rum production. In Distilled Spirits, Volume 3: New Horizons: Energy, Environment and Enlightenment; Nottingham University Press: Nottingham, UK, 2010; Volume 3, p. 203. [Google Scholar]
- Jevtić-Mučibabić, R.; Grbić, J.; Mišljenović, N.; Koprivica, G.; Kuljanin, T.; Radivojević, S. Nitrogen compounds in the molasses. J. Process. Energy Agric. 2011, 15, 169–172. [Google Scholar]
- Schoonees, B.; Pillay, V. A New Filtration system for gum analysis in raw sugar and molasses. Proc. South Afr. Sugar Technol. Assoc. 2004, 78, 565–568. [Google Scholar]
- Kampen, W.H. Chapter 4—Nutritional Requirements in fermentation processes. In Fermentation and Biochemical Engineering Handbook, 3rd ed.; Vogel, H.C., Todaro, C.M., Eds.; William Andrew Publishing: Boston, MA, USA, 2014; pp. 37–57. [Google Scholar] [CrossRef]
- Olbrich, H. The Molasses; Biotechnologie-Kempe GmbH: Berlin, Germany, 1963; p. 128. [Google Scholar]
- Jones, A.M.; Thomas, K.C.; Ingledew, W.M. Ethanolic Fermentation of Blackstrap molasses and sugarcane juice using very high gravity technology. J. Agric. Food Chem. 1994, 42, 1242–1246. [Google Scholar] [CrossRef]
- Quesada-Granados, J.J.; Samaniego-Sánchez, C.; Blanca-Herrera, R.M. Rhum-ron-rum: Technology and tradition. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Oxford, UK, 2016; pp. 618–627. [Google Scholar] [CrossRef]
- Abubaker, H.O.; Sulieman, A.M.E.; Elamin, H.B. Utilization of Schizosaccharomyces pombe for production of ethanol from cane molasses. J. Microbiol. Res. 2012, 2, 36–40. [Google Scholar] [CrossRef]
- Paturau, J.M. By-Products of the Cane Sugar Industry. An Introduction to Their Industrial Utilization; Elsevier Science Publishers BV: Amsterdam, The Netherlands, 1989. [Google Scholar]
- Wiebe, H.H.; Kidambi, R.N.; Richardson, G.H.; Ernstrom, C.A. A Rapid Psychrometric procedure for water activity measurement of foods in the intermediate moisture range. J. Food Prot. 1981, 44, 892–895. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani, F.; Younesi, H.; Esmaeili Sari, A.; Najafpour, G. Cane molasses fermentation for continuous ethanol production in an immobilized cells reactor by Saccharomyces cerevisiae. Renew. Energy 2011, 36, 503–509. [Google Scholar] [CrossRef]
- Li, H.; Jiang, Z.; Yang, X.; Yu, L.; Zhang, G.; Wu, J.; Liu, X. Sustainable resource opportunity for cane molasses: Use of cane molasses as a grinding aid in the production of Portland cement. J. Clean. Prod. 2015, 93, 56–64. [Google Scholar] [CrossRef]
- Clarke, M. Syrups. In Encyclopedia of Food Science; Sugar Processing Research Institute Incorporated: New Orleans, LA, USA, 1993; pp. 5711–5717. [Google Scholar]
- Nelson, E.K. Some organic acids of sugar cane molasses. J. Am. Chem. Soc. 1929, 51, 2808–2810. [Google Scholar] [CrossRef]
- Yoder, P.A. Notes on the determination of acids in sugar cane juice. J. Ind. Eng. Chem. 1911, 3, 640–646. [Google Scholar] [CrossRef][Green Version]
- Binkley, W.W.; Wolform, M.L. Composition of Cane juice and cane final molasses. In Advances in Carbohydrate Chemistry; Hudson, C.S., Wolfrom, M.L., Eds.; Academic Press: Cambridge, MA, USA, 1953; Volume 8, pp. 291–314. [Google Scholar]
- Lehtonen, M.; Suomalainen, H. Rum. In Economic Microbiology; Rose, A., Ed.; Academic Press: Cambridge, MA, USA, 1977; Volume 1, pp. 595–633. [Google Scholar]
- Lea, A.G.; Piggott, J.R. Fermented Beverage Production, 2nd ed.; Kluwer Academic: New York, NY, USA, 2003. [Google Scholar]
- Walker, G.M. Metals in yeast fermentation processes. In Advances in Applied Microbiology; Laskin, A.I., Bennett, J.W., Gadd, G.M., Eds.; Elsevier Academic Press: San Diego, CA, USA, 2004; pp. 197–230. [Google Scholar]
- Siqueira, P.F.; Karp, S.G.; Carvalho, J.C.; Sturm, W.; Rodríguez-León, J.A.; Tholozan, J.-L.; Singhania, R.R.; Pandey, A.; Soccol, C.R. Production of bio-ethanol from soybean molasses by Saccharomyces cerevisiae at laboratory, pilot and industrial scales. Bioresour. Technol. 2008, 99, 8156–8163. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Haque, S.; Niwas, R.; Srivastava, A.; Pasupuleti, M.; Tripathi, C.K.M. Strategies for fermentation Medium optimization: An in-depth review. Front. Microbiol. 2017, 7. [Google Scholar] [CrossRef]
- Bluhm, L. Distilled beverages. In Biotechnology, a Comprehensive Treatise in 8 Volumes; Rehm, H.-J., Reed, G., Eds.; Verlag Chemie: Weinheim, Germany, 1983; Volume 5, pp. 447–475. [Google Scholar]
- Walker, G.M.; Stewart, G.G. Saccharomyces cerevisiae in the Production of fermented beverages. Beverages 2016, 2, 30. [Google Scholar] [CrossRef]
- Goksel, M.; Dogan, M.; Toker, O.S.; Ozgen, S.; Sarioglu, K.; Oral, R.A. The Effect of starch concentration and temperature on Grape molasses: Rheological and Textural properties. Food Bioprocess Technol. 2013, 6, 259–271. [Google Scholar] [CrossRef]
- Greetham, D.; Hart, A.J.; Tucker, G.A. Presence of low concentrations of acetic acid improves yeast tolerance to hydroxymethylfurfural (HMF) and furfural. Biomass Bioenergy 2016, 85, 53–60. [Google Scholar] [CrossRef]
- Casey, E.; Sedlak, M.; Ho, N.W.Y.; Mosier, N.S. Effect of acetic acid and pH on the cofermentation of glucose and xylose to ethanol by a genetically engineered strain of Saccharomyces cerevisiae. Fems Yeast Res. 2010, 10, 385–393. [Google Scholar] [CrossRef]
- Ferreira, M.M.; Loureiro-Dias, M.C.; Loureiro, V. Weak acid inhibition of fermentation by Zygosaccharomyces bailii and Saccharomyces cerevisiae. Int. J. Food Microbiol. 1997, 36, 145–153. [Google Scholar] [CrossRef]
- Laluce, C.; Leite, G.R.; Zavitoski, B.Z.; Zamai, T.T.; Ventura, R. Fermentation of sugarcane juice and molasses for ethanol production. In Sugarcane-Based Biofuels and Bioproducts; Wiley: Hoboken, NJ, USA, 2016; pp. 53–86. [Google Scholar]
- Bortolussi, G.; O’Neill, C.J. Variation in molasses composition from eastern Australian sugar mills. Aust. J. Exp. Agric. 2006, 46, 1455–1463. [Google Scholar] [CrossRef]
- Lingle, S.E.; Wiegand, C.L. Soil salinity and sugarcane juice quality. Field Crop. Res. 1997, 54, 259–268. [Google Scholar] [CrossRef]
- Shukla, S.K.; Solomon, S.; Sharma, L.; Jaiswal, V.P.; Pathak, A.D.; Singh, P. Green technologies for improving cane sugar productivity and sustaining soil fertility in sugarcane-based cropping system. Sugar Tech. 2019, 21, 186–196. [Google Scholar] [CrossRef]
- Alamilla-Magaña, J.C.; Carrillo-Ávila, E.; Obrador-Olán, J.J.; Landeros-Sánchez, C.; Vera-Lopez, J.; Juárez-López, J.F. Soil moisture tension effect on sugar cane growth and yield. Agric. Water Manag. 2016, 177, 264–273. [Google Scholar] [CrossRef]
- Franitza, L.; Granvogl, M.; Schieberle, P. Influence of the Production process on the key aroma compounds of rum: From molasses to the spirit. J. Agric. Food Chem. 2016, 64, 9041–9053. [Google Scholar] [CrossRef] [PubMed]
- Murtagh, J.E. Molasses as a feedstock for alcohol production. In The Alcohol Textbook; Lyons, T., Kelsall, D., Murtagh, J.E., Eds.; Nottingham University Press: Nottingham, UK, 1995. [Google Scholar]
- Hill, A.E. Deciphering dunder: An examination of rum fermentation. In Proceedings of the Worldwide Distilled Spirits Conference: Local Roots; Global Reach: Delivering Distilling Expertise to the World, Glasgow, UK, 29 May–1 June 2017. [Google Scholar]
- Hutzler, M.; Koob, J.; Riedl, R.; Schneiderbanger, H.; Mueller-Auffermann, K.; Jacob, F. 5-yeast identification and characterization. In Brewing Microbiology; Hill, A.E., Ed.; Woodhead Publishing: Cambridge, UK, 2015; pp. 65–104. [Google Scholar] [CrossRef]
- Fahrasmane, L.; Ganou-Parfait, B. Microbial flora of rum fermentation media. J. Appl. Microbiol. 1998, 84, 921–928. [Google Scholar] [CrossRef]
- Suomalainen, H. Some general aspects of the composition of alcoholic beverages [Quelques aspects généraux de la composition des boissons alcooliques]. Ann. Technol. Agric. 1975, 24, 453–467. [Google Scholar]
- Broom, D. Rum; Abbeville Press: New York, NY, USA, 2003. [Google Scholar]
- Piggott, J. Distillation of brandies. In The Alcohol Textbook, 5th ed.; Nottingham University Press: Nottingham, UK, 2009; pp. 491–506. [Google Scholar]
- Jacques, K.; Lyon, T.; Kelsall, D. The Alcohol Textbook, 4th ed.; Nottingham University Press: Nottingham, UK, 2003. [Google Scholar]
- Berry, D.R.; Slaughter, J.C. Alcoholic Beverage fermentations. In Fermented Beverage Production; Lea, A.G.H., Piggott, J.R., Eds.; Springer: Boston, MA, USA, 2003; pp. 25–39. [Google Scholar] [CrossRef]
- Madrera, R.R.; Gomis, D.B.; Alonso, J.J.M. Influence of Distillation system, oak wood type, and aging time on volatile compounds of cider brandy. J. Agric. Food Chem. 2003, 51, 5709–5714. [Google Scholar] [CrossRef] [PubMed]
- Reche, R.V.; Leite Neto, A.F.; Da Silva, A.A.; Galinaro, C.A.; De Osti, R.Z.; Franco, D.W. Influence of Type of distillation apparatus on chemical profiles of Brazilian cachaças. J. Agric. Food Chem. 2007, 55, 6603–6608. [Google Scholar] [CrossRef] [PubMed]
- Zitting, A.; Savolainen, H.; Nickels, J. Biochemical and toxicological effects of single and repeated exposures to polyacetal thermodegradation products. Environ. Res. 1982, 29, 287–296. [Google Scholar] [CrossRef]
- Spaho, N.; Dürr, P.; Grba, S.; Velagić-Habul, E.; Blesić, M. Effects of distillation cut on the distribution of higher alcohols and esters in brandy produced from three plum varieties. J. Inst. Brew. 2013, 119, 48–56. [Google Scholar] [CrossRef]
- Bamforth, C.W.; Ward, R.E. The Oxford Handbook of Food Fermentations; Oxford University Press: Oxford, UK, 2014. [Google Scholar]
- Clutton, D. Rum. Flavour Ind. 1974, 5, 286–288. [Google Scholar]
- Mosedale, J.R. Effects of oak wood on the maturation of alcoholic beverages with particular reference to whisky. For. Int. J. For. Res. 1995, 68, 203–230. [Google Scholar] [CrossRef]
- Mosedale, J.R.; Puech, J.L. Wood maturation of distilled beverages. Trends Food Sci. Technol. 1998, 9, 95–101. [Google Scholar] [CrossRef]
- De Rosso, M.; Cancian, D.; Panighel, A.; Dalla Vedova, A.; Flamini, R. Chemical compounds released from five different woods used to make barrels for aging wines and spirits: Volatile compounds and polyphenols. Wood Sci. Technol. 2009, 43, 375–385. [Google Scholar] [CrossRef]
- Coldea, T.E.; Mudura, E.; Socaciu, C. Advances in distilled beverages authenticity and quality testing. In Ideas and Applications toward Sample Preparation for Food and Beverage Analysis; Stauffer, M.T., Ed.; IntechOpen: London, UK, 2017; pp. 109–130. [Google Scholar] [CrossRef]
- Quesada Granados, J.; Merelo Guervós, J.J.; Oliveras López, M.J.; González Peñalver, J.; Olalla Herrera, M.; Blanca Herrera, R.; López Martinez, M.C. Application of Artificial aging techniques to samples of rum and comparison with traditionally aged rums by analysis with artificial neural nets. J. Agric. Food Chem. 2002, 50, 1470–1477. [Google Scholar] [CrossRef]
- Fernández de Simón, B.; Cadahía, E. Volatile compounds in a Rioja wine aged in Spanish, French and American oak barrels. In Proceedings of the Congresos y Jornadas-Gobierno de La Rioja (España), Logroño, Spain, 21–23 May 2003. [Google Scholar]
- Remmer, C. Differences between Swedish, French and American Oak when Storing Whisky Analysed by GC-MS. Bachelor’s Thesis, Örebro University, Örebro, Sweden, 2015. [Google Scholar]
- Pino, J.A.; Tolle, S.; Gök, R.; Winterhalter, P. Characterisation of odour-active compounds in aged rum. Food Chem. 2012, 132, 1436–1441. [Google Scholar] [CrossRef] [PubMed]
- Franitza, L.; Granvogl, M.; Schieberle, P. Characterization of the Key aroma compounds in two commercial rums by means of the sensomics approach. J. Agric. Food Chem. 2016, 64, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Herranz, A.; de la Serna, P.; Barro, C.; Martin-Alvarez, P.J. Multivariate statistical methods applied to the differentiation of rum brands. J. Sci. Food Agric. 1990, 51, 555–560. [Google Scholar] [CrossRef]
- Nykänen, L.; Puputti, E.; Suomalainen, H. Volatile Fatty acids in some brands of whisky, cognac and rum. J. Food Sci. 1968, 33, 88–92. [Google Scholar] [CrossRef]
- Piggott, J. Alcoholic Beverages: Sensory Evaluation and Consumer Research; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Kourtis, L.K.; Arvanitoyannis, I.S. Implementation of hazard analysis critical control point (HACCP) system to the alcoholic beverages industry. Food Rev. Int. 2001, 17, 1–44. [Google Scholar] [CrossRef]
- Chikhoune, A.; Bedjou, F.; Oubouzid, S.; Boukefoussa, R.; Bechri, B.; Tarmoul, H.; Abdeladim, T.; Tounsi, A.; Hamitri, M.; Chikh, S.; et al. Development of sugar cane molasses in formulations of madeleines, mini croissants, and buns incorporated with interesterified oil. J. Chem. 2014, 2014, 936780. [Google Scholar] [CrossRef]
- Deseo, M.A.; Elkins, A.; Rochfort, S.; Kitchen, B. Antioxidant activity and polyphenol composition of sugarcane molasses extract. Food Chem. 2020, 314, 126180. [Google Scholar] [CrossRef] [PubMed]
- Palmonari, A.; Cavallini, D.; Sniffen, C.J.; Fernandes, L.; Holder, P.; Fagioli, L.; Fusaro, I.; Biagi, G.; Formigoni, A.; Mammi, L. Short communication: Characterization of molasses chemical composition. J. Dairy Sci. 2020, 103, 6244–6249. [Google Scholar] [CrossRef] [PubMed]
- Pierce, J.S. Horace Brown memorial lecture the role of nitrogen in brewing. J. Inst. Brew. 1987, 93, 378–381. [Google Scholar] [CrossRef]
- Hohmann, S. Osmotic Stress signaling and osmoadaptation in yeasts. Microbiol. Mol. Biol. Rev. 2002, 66, 300–372. [Google Scholar] [CrossRef] [PubMed]
- O’Connor-Cox, E.S.C.; Ingledew, W.M. Wort nitrogenous sources—Their use by brewing yeasts: A review. J. Am. Soc. Brew. Chem. 1989, 47, 102–108. [Google Scholar] [CrossRef]
- Almeida, T.; Marques, M.; Mojzita, D.; Amorim, M.A.; Silva, R.D.; Almeida, B.; Rodrigues, P.; Ludovico, P.; Hohmann, S.; Moradas-Ferreira, P.; et al. Isc1p Plays a key role in hydrogen peroxide resistance and chronological lifespan through modulation of iron levels and apoptosis. Mol. Biol. Cell 2008, 19, 865–876. [Google Scholar] [CrossRef] [PubMed]
- Walker, G.M. Yeast Physiology and Biotechnology; John Wiley & Sons: Hoboken, NJ, USA, 1998. [Google Scholar]
- Eide, D.J. Multiple Regulatory mechanisms maintain zinc homeostasis in Saccharomyces cerevisiae. J. Nutr. 2003, 133, 1532S–1535S. [Google Scholar] [CrossRef] [PubMed]
- Ingledew, W. Alcohol production by Saccharomyces cerevisiae: A yeast primer. In The Alcohol Textbook; Lyons, T., Kelsall, D., Eds.; Nottingham University Press: Nottingham, UK, 1999; Volume 3, pp. 49–87. [Google Scholar]
- Barbour, E.; Priest, F. Some effects of Lactobacillus contamination in scotch whisky fermentations. J. Inst. Brew. 1988, 94, 89–92. [Google Scholar] [CrossRef]
- Dolan, T.C.S. Some Aspects of the impact of brewing science on scotch malt whisky production. J. Inst. Brew. 1976, 82, 177–181. [Google Scholar] [CrossRef]
- Makanjuola, D.B.; Tymon, A.; Springham, D.G. Some effects of lactic acid bacteria on laboratory-scale yeast fermentations. Enzym. Microb. Technol. 1992, 14, 350–357. [Google Scholar] [CrossRef]
- Meilgaard, M.C. Flavor chemistry of beer. II. Flavor and threshold of 239 aroma volatiles. Tech. Quart. Master. Brew. Assoc. Am. 1975, 12, 151–168. [Google Scholar]
- Uselmann, V.; Schieberle, P. Decoding the combinatorial aroma code of a commercial cognac by application of the sensomics concept and first insights into differences from a German brandy. J. Agric. Food Chem. 2015, 63, 1948–1956. [Google Scholar] [CrossRef] [PubMed]
- Czerny, M.; Christlbauer, M.; Christlbauer, M.; Fischer, A.; Granvogl, M.; Hammer, M.; Hartl, C.; Hernandez, N.M.; Schieberle, P. Re-investigation on odour thresholds of key food aroma compounds and development of an aroma language based on odour qualities of defined aqueous odorant solutions. Eur. Food Res. Technol. 2008, 228, 265–273. [Google Scholar] [CrossRef]
- Poisson, L.; Schieberle, P. Characterization of the key aroma compounds in an American bourbon whisky by quantitative measurements, aroma recombination, and omission studies. J. Agric. Food Chem. 2008, 56, 5820–5826. [Google Scholar] [CrossRef] [PubMed]
- Willner, B.; Granvogl, M.; Schieberle, P. Characterization of the key aroma compounds in Bartlett pear brandies by means of the sensomics concept. J. Agric. Food Chem. 2013, 61, 9583–9593. [Google Scholar] [CrossRef]
- Suomalainen, H.; Nykänen, L. Investigations on the aroma of alcoholic beverages. Naeringsmiddelindustrien 1970, 23, 15–30. [Google Scholar]
- Otsuka, K.-I.; Zenibayashi, Y.; Itoh, M.; Totsuka, A. Presence and significance of two diastereomers of β-methyl-γ-octalactone in aged distilled liquors. Agric. Biol. Chem. 1974, 38, 485–490. [Google Scholar] [CrossRef]
| Rum Type | Aging | Principal Production Region(s) | Ethanol Content (% ABV) | Additional Notes |
|---|---|---|---|---|
| White | Stainless steel casks (1–2 yrs); often aged less than other rums | Puerto Rico | 37–43 | Lighter bodied; filtered before sale |
| Dark | Charred oak barrels (2 yrs) | Jamaica, La Martinica | 37–43 | Darker, fuller flavour |
| Amber/Gold | Charred oak barrels (1.5 yrs) | Cuba, Puerto Rico | 37–43 | Flavour not as complex as dark rums |
| Over proof/ Naval | Variable; can be sold with no aging in some countries | Jamaica | 70–80 | |
| Spiced | Charred oak barrels (1–2 yrs) | Jamaica, Puerto Rico | 37–43 | Most are darker in colour and based on gold rums |
| Demerara Rum | Longer aging | Guyana | 37–43 | Distilled in old stills; complex flavour similar to Jamaican rum |
| Constituents | Typical Range | References |
|---|---|---|
| Physical Parameters | ||
| Moisture Content (%) | 17–25 | [28] |
| pH | 2.5–5.5 | [23,28,29] |
| Ash (% w/w) | 10–16 | [28,30,31] |
| Water activity (Aw) | 0.76 | [32] |
| Total dissolved solids (TDS) (% w/w) | 76.6 | [28] |
| °Brix | 79.5–89.5 | [28] |
| Volatile matter (%) | 86.3 | [33] |
| Protein (%) | 0.145 | [33] |
| Gums (% w/w) | 6 | [25,26] |
| Colloidal substances (% w/w) | 9.2 | [34] |
| Sugars (% w/w) | ||
| Sucrose | 30–57 | [15,28] |
| Fructose | 5–13 | [28] |
| Glucose | 4–10 | [28] |
| Maltose | 0.11 | [28] |
| Maltotriose | 0.43 | [28] |
| Isomaltose | 0.020 | [28] |
| Non-fermentable sugars (% w/w) | 5.1 | [34] |
| Total fermentable sugars (% w/w) | 54 | [34] |
| Nitrogenous substances (% w/w) | ||
| Total nitrogen | 0.36–1.97 | [24,28] |
| Free amino nitrogen (FAN) | 0.10 | [28] |
| Minerals (mg L−1) | ||
| Sodium | 1600 | [28] |
| Phosphorus | 600 | [28] |
| Potassium | 27200 | [28] |
| Sulphur | 3800 | [28] |
| Calcium | 10600 | [28] |
| Magnesium | 4200 | [28] |
| Copper | 17 | [28] |
| Iron | 150–1170 | [28,30] |
| Manganese | 53 | [28] |
| Zinc | 19 | [28] |
| Vitamins (mg L−1) | ||
| Thiamin (B1) | 2–10 | [35] |
| Riboflavin (B2) | 1–6 | [35] |
| Pyridoxine (B6) | 1–10 | [35] |
| Nicotinamide | 1–25 | [35] |
| Pantothenic acid | 2–25 | [35] |
| Folic acid | 10–50 | [35] |
| Biotin | 0.1–2 | [35] |
| Organic acids (% w/w) | ||
| Acetic acid | 0.2–1 | [36] |
| Aconitic acid | 0.05–0.8 | [36,37] |
| Formic acid | 0.097–0.12 | [36,38] |
| Valeric acid | <0.1 | [39] |
| Lactic acid | ~0.05 | [36] |
| Citric acid | ~0.05 | [36] |
| Malic acid | 0.001 | [37] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mangwanda, T.; Johnson, J.B.; Mani, J.S.; Jackson, S.; Chandra, S.; McKeown, T.; White, S.; Naiker, M. Processes, Challenges and Optimisation of Rum Production from Molasses—A Contemporary Review. Fermentation 2021, 7, 21. https://doi.org/10.3390/fermentation7010021
Mangwanda T, Johnson JB, Mani JS, Jackson S, Chandra S, McKeown T, White S, Naiker M. Processes, Challenges and Optimisation of Rum Production from Molasses—A Contemporary Review. Fermentation. 2021; 7(1):21. https://doi.org/10.3390/fermentation7010021
Chicago/Turabian StyleMangwanda, Tinashe, Joel B. Johnson, Janice S. Mani, Steve Jackson, Shaneel Chandra, Tyryn McKeown, Simon White, and Mani Naiker. 2021. "Processes, Challenges and Optimisation of Rum Production from Molasses—A Contemporary Review" Fermentation 7, no. 1: 21. https://doi.org/10.3390/fermentation7010021
APA StyleMangwanda, T., Johnson, J. B., Mani, J. S., Jackson, S., Chandra, S., McKeown, T., White, S., & Naiker, M. (2021). Processes, Challenges and Optimisation of Rum Production from Molasses—A Contemporary Review. Fermentation, 7(1), 21. https://doi.org/10.3390/fermentation7010021








