Optimization of an Industrial Medium from Molasses for Bioethanol Production Using the Taguchi Statistical Experimental-Design Method
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Strain
2.2. Media and Culture Conditions
2.3. Ethanol Assay
2.4. Measuring Yeast Cell Growth
2.5. Optimization of the Ethanol Production Medium by the Taguchi Experimental-Design Method
2.6. Bioreactor Cultivation
3. Results
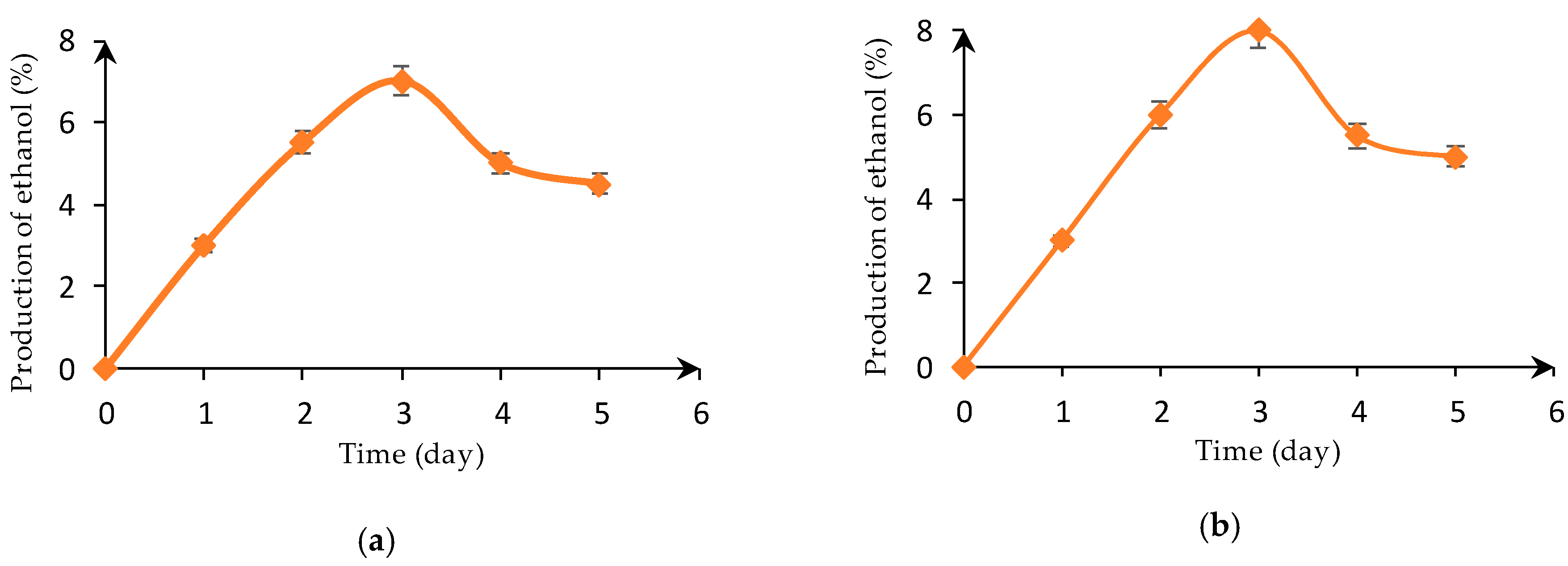
3.1. Yeast Cell Growth and Ethanol Production in the Industrial Medium
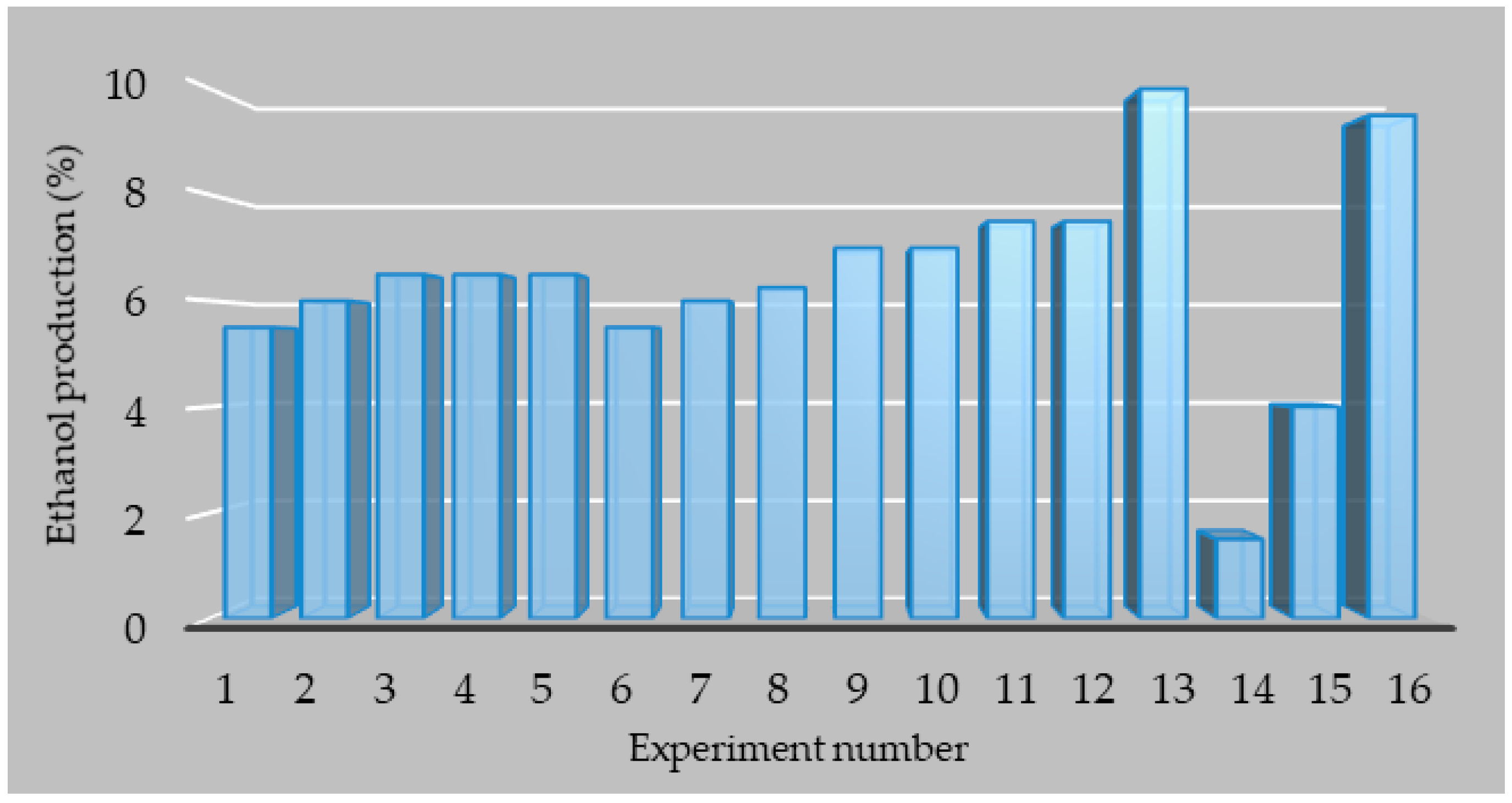
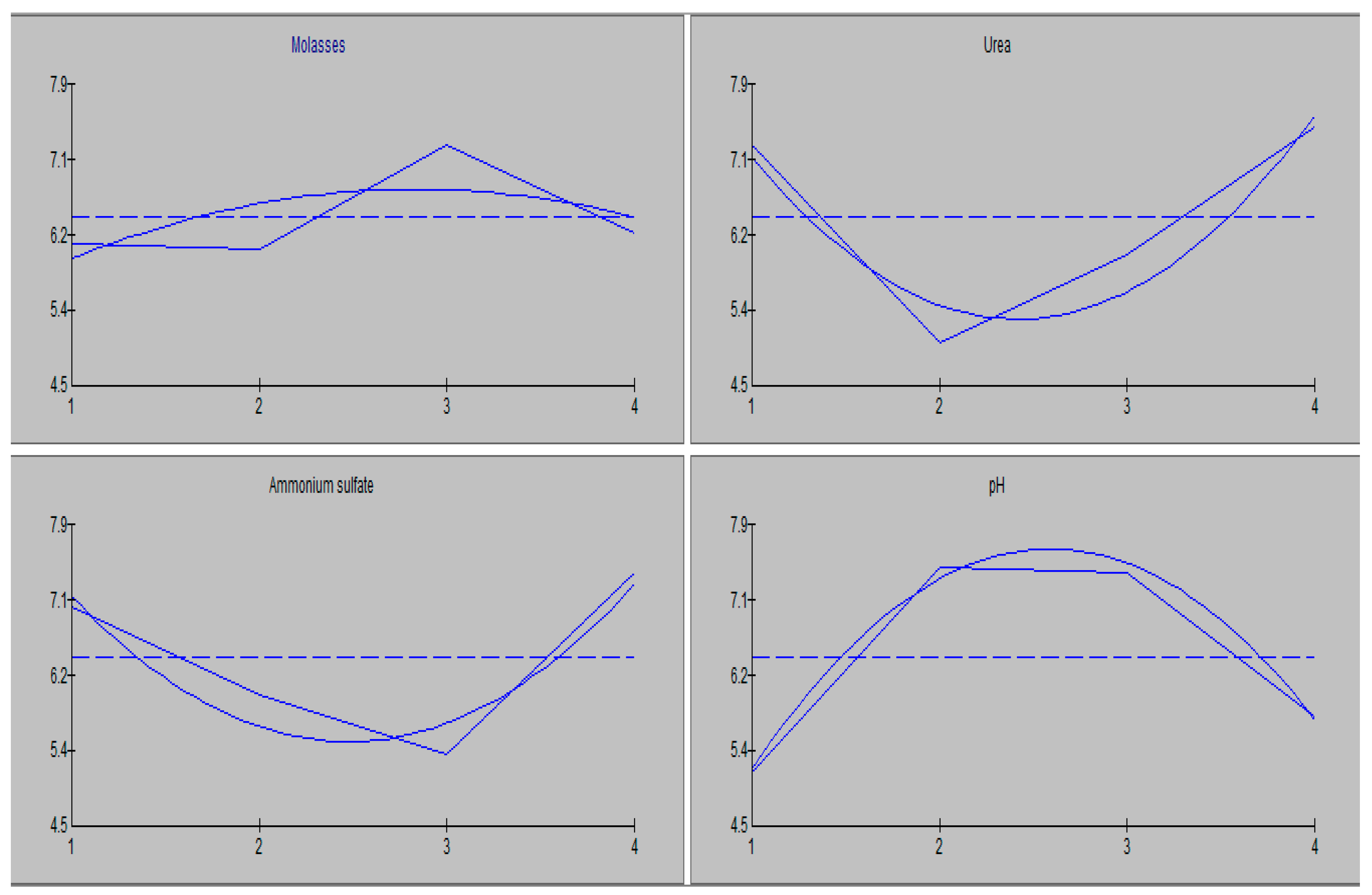
3.2. Optimization of the Industrial Medium for Increasing Ethanol Production
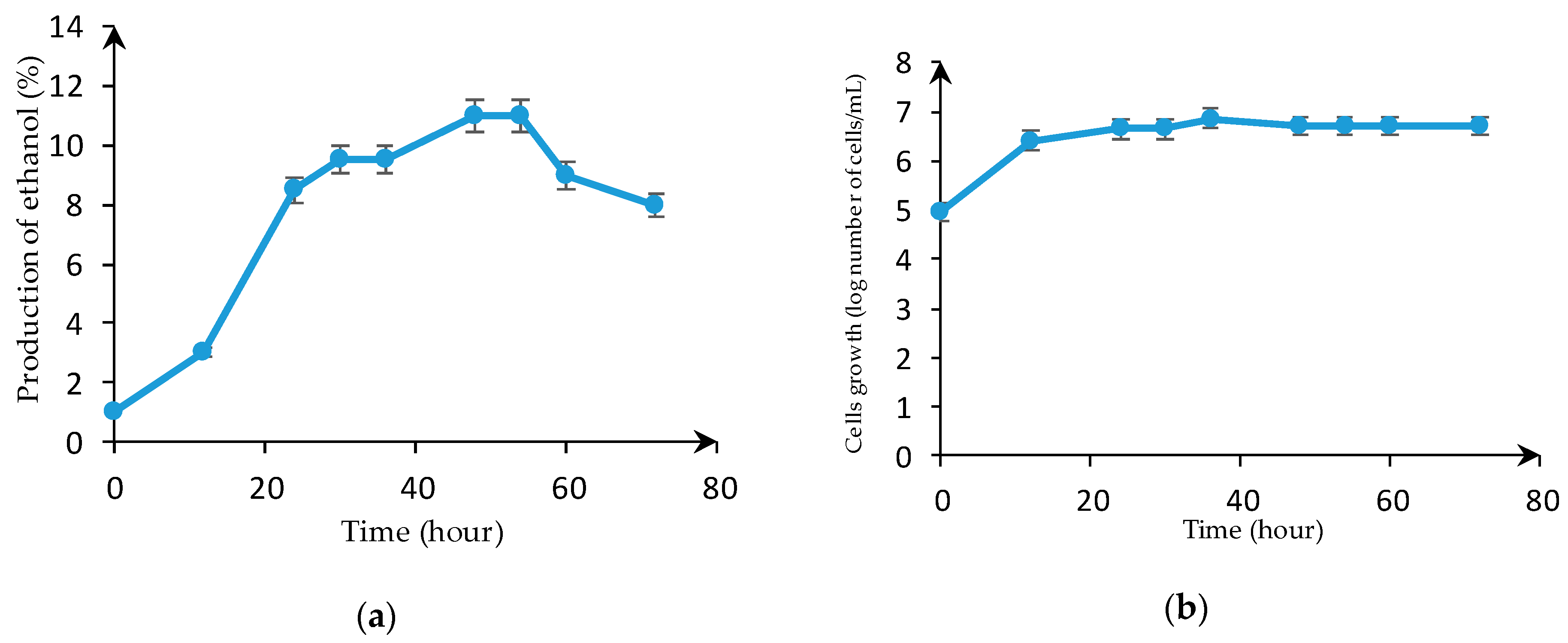
3.3. Ethanol Production in the Bioreactor
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Leung, D.Y.C.; Caramanna, G.; Maroto–Valer, M.M. An overview of current status of carbon dioxide capture and storage technologies. Renew. Sustain. Energy Rev. 2004, 39, 426–443. [Google Scholar] [CrossRef]
- Yu, J.; Corripio, A.B.; Harrison, D.P.; Copeland, R.J. Analysis of the sorbent energy transfer system (SETS) for power generation and CO2 capture. Adv. Environ. Res. 2003, 7, 335–345. [Google Scholar] [CrossRef]
- Sudhakar, M.P.; Merlyn, R.; Arunkumar, R.K.; Perumal, K. Characterization, pretreatment and saccharification of spent seaweed biomass for bioethanol production using baker’s yeast. Biomass Bioenerg. 2016, 90, 148–154. [Google Scholar] [CrossRef]
- Offei, F.; Mensah, M.; Thygesen, A.; Kemausuor, F. Seaweed bioethanol production: A process selection review on hydrolysis and fermentation. Fermentation 2018, 4, 99. [Google Scholar] [CrossRef]
- Antoni, D.; Zverlov, V.V.; Schwarz, W.H. Biofuels from microbes. Appl. Microbiol. Biotechnol. 2007, 77, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Briggs, D.E.; Brookes, P.A.; Stevens, R.; Boulton, C.A. Brewing: Science and Practice, 1st ed.; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Kosaric, N.; Duvnjak, Z.; Farkas, A.; Sahm, H.; Bringer-Meyer, S.; Goebel, O.; Mayer, D. Ethanol. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley: Hoboken, NJ, USA, 2000; pp. 1–72. [Google Scholar]
- Kampen, W.H. Nutritional requirements in fermentation processes. In Fermentation and Biochemical Engineering Handbook, 3rd ed.; Vogel, H.C., Todaro, C.M., Eds.; Elsevier: Norwich, NY, USA, 2014; pp. 37–57. [Google Scholar]
- Springham, D.G.; Moses, V.; Cape, R.E. Biotechnology—The Science and the Business, 2nd ed.; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Khongsay, N.; Laopaiboon, L.; Laopaiboon, P. Growth and batch ethanol fermentation of Saccharomyces cerevisiae on sweet sorghum stem juice under normal and very high gravity conditions. Biotechnology 2010, 9, 9–16. [Google Scholar] [CrossRef]
- Bayrock, D.P.; Ingledew, W.M. Application of multistage continuous fermentation for production of fuel alcohol by very-high-gravity fermentation technology. J. Ind. Microbiol. Biotechnol. 2001, 27, 87–93. [Google Scholar] [CrossRef]
- Khuri, A.I.; Mukhopadhyay, S. Response surface methodology. WIREs Comput. Stat. 2010, 2, 128–149. [Google Scholar] [CrossRef]
- Taguchi, G. System of Experimental Design, 1st ed.; Quality Resources: Milwaukee, WI, USA, 1987. [Google Scholar]
- Wang, K.; Hao, J.; Wang, L.; Li, H. Phase factor sequences algorithm in partial transmit sequence. Trans. Tianjin Univ. 2009, 15, 23–26. [Google Scholar] [CrossRef]
- Yang, L.J.; Qi, Y.M.; Dang, X. Design and optimization of technology and structure parameters for sheet metal drawing by orthogonal experiment. Adv. Mat. Res. 2011, 295, 1714–1717. [Google Scholar] [CrossRef]
- Darvishi, F.; Abolhasan Moghaddami, N. Isolation of industrial strain Saccharomyces cerevisiae with high tolerance to ethanol from Iran’s alcohol manufactures. Iran. J. Biol. 2018, Accepted. [Google Scholar]
- Williams, M.B.; Reese, H.D. Colorimetric determination of ethyl alcohol. Anal. Chem. 1950, 22, 1556–1561. [Google Scholar] [CrossRef]
- Sumbhate, S.V.; Nayak, S.; Goupale, D.; Tiwari, A.; Jadon, R.S. Colorimetric method for the estimation of ethanol in alcoholic-drinks. J Anal. Tech. 2012, 1, 1–6. [Google Scholar]
- Darvishi, F.; Moradi, M.; Madzak, C.; Jolivalt, C. Production of laccase by recombinant Yarrowia lipolytica from molasses: Bioprocess development using statistical modeling and increase productivity in shake-flask and bioreactor cultures. Appl. Biochem. Biotechnol. 2017, 181, 1228–1239. [Google Scholar] [CrossRef] [PubMed]
- Karna, S.K.; Sahai, R. An overview on Taguchi method. Int. J. Eng. Math. 2012, 1, 11–18. [Google Scholar]
- Casey, G.P.; Ingledew, W.M. Ethanol tolerance in yeasts. Crit. Rev. Microbiol. 1986, 13, 219–280. [Google Scholar] [CrossRef] [PubMed]
- Darvishi, F.; Hiligsmann, S. Microbial Fuels: Technologies and Applications, 1st ed.; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Akbas, M.Y.; Stark, B.C. Recent trends in bioethanol production from food processing byproducts. J. Ind. Microbiol. Biotechnol. 2016, 43, 1593–1609. [Google Scholar] [CrossRef]
- Thammasittirong, S.N.; Thirasaktana, T.; Thammasittirong, A.; Srisodsuk, M. Improvement of ethanol production by ethanol-tolerant Saccharomyces cerevisiae UVNR56. Springerplus 2013, 2, 583. [Google Scholar] [CrossRef]
- Shankar, T.; Kumar, R.S.; Anandapandian, K.T.K. Statistical optimization for ethanol production by Saccharomyces cerevisiae (MTCC 170) using response surface methodology. J. Adv. Med. Life Sci. 2015, 2, 1–5. [Google Scholar]
- Barbosa, H.S.; Silveira, E.A.; Miranda, M.; Ernandes, J.R. Efficient very-high-gravity fermentation of sugarcane molasses by industrial yeast strains. J. Inst. Brew. 2016, 122, 329–333. [Google Scholar] [CrossRef]
- Yue, G.; Yu, J. The influence of nitrogen sources on ethanol production by yeast from concentrated sweet sorghum juice. Biomass Bioenerg. 2012, 39, 48–52. [Google Scholar] [CrossRef]
- Dragone, G.; Silva, D.P. Factors influencing ethanol production rates at high-gravity brewing. Food Sci. Technol. 2004, 37, 797–802. [Google Scholar] [CrossRef]
- Lopes, D.H.J.; Sola–Penna, M. Urea increases tolerance of yeast inorganic pyrophosphatase activity to ethanol: The other side of urea interaction with proteins. Arch. Biochem. Biophys. 2001, 394, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Kasemets, K.; Nisamedtinov, I. Growth characteristics of Saccharomyces cerevisiae S288C in changing environmental conditions: Auxo-accelerostat study. Antonie Van Leeuwenhoek 2007, 92, 109–128. [Google Scholar] [CrossRef] [PubMed]
- Eroshin, V.K.; Utkin, I.S.; Ladynichev, S.V.; Samoylov, V.V.; Kuvshinnikov, V.D.; Skryabin, G.K. Influence of pH and temperature on the substrate yield coefficient of yeast growth in a chemostat. Biotechnol. Bioeng. 1976, 18, 289–295. [Google Scholar] [CrossRef]
- Wada, M.; Kato, J.; Chibata, I. A new immobilization of microbial cells. J. Appl. Microbiol. Biotechnol. 1979, 8, 241–247. [Google Scholar] [CrossRef]
- Lin, Y.; Zhang, W.; Li, C.; Sakakibara, K.; Tanaka, S.; Kong, H. Factors affecting ethanol fermentation using Saccharomyces cerevisiae BY4742. Biomass Bioenerg. 2012, 47, 395–401. [Google Scholar] [CrossRef]
- Tahir, A.; Aftab, M.; Farasat, T. Effect of cultural conditions on ethanol production by locally isolated Saccharomyces cerevisiae BIO-07. J. Appl. Pharm. 2010, 3, 72–78. [Google Scholar]
- Bai, F.W.; Anderson, W.A.; Moo–Young, M. Ethanol fermentation technologies from sugar and starch feedstocks. Biotechnol. Adv. 2008, 26, 89–105. [Google Scholar] [CrossRef]

| Factor | Level 1 | Level 2 | Level 3 | Level 4 |
|---|---|---|---|---|
| Brix of molasses (%) | 15 | 18 | 21 | 24 |
| Ammonium sulfate (g/L) | 1 | 2 | 3 | 4 |
| Urea (g/L) | 1 | 2 | 3 | 4 |
| pH | 4 | 5 | 6 | 7 |
| Experiment No. | Molasses (% Brix) | Ammonium Sulfate (g/L) | Urea (g/L) | pH |
|---|---|---|---|---|
| No. 1 | 15 | 1 | 1 | 4 |
| No. 2 | 15 | 2 | 2 | 5 |
| No. 3 | 15 | 3 | 3 | 6 |
| No. 4 | 15 | 4 | 4 | 7 |
| No. 5 | 18 | 1 | 2 | 6 |
| No. 6 | 18 | 2 | 1 | 7 |
| No. 7 | 18 | 3 | 4 | 4 |
| No. 8 | 18 | 4 | 3 | 5 |
| No. 9 | 21 | 1 | 3 | 7 |
| No. 10 | 21 | 2 | 4 | 6 |
| No. 11 | 21 | 3 | 1 | 5 |
| No. 12 | 21 | 4 | 2 | 4 |
| No. 13 | 24 | 1 | 4 | 5 |
| No. 14 | 24 | 2 | 3 | 4 |
| No. 15 | 24 | 3 | 2 | 7 |
| No. 16 | 24 | 4 | 1 | 6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Darvishi, F.; Abolhasan Moghaddami, N. Optimization of an Industrial Medium from Molasses for Bioethanol Production Using the Taguchi Statistical Experimental-Design Method. Fermentation 2019, 5, 14. https://doi.org/10.3390/fermentation5010014
Darvishi F, Abolhasan Moghaddami N. Optimization of an Industrial Medium from Molasses for Bioethanol Production Using the Taguchi Statistical Experimental-Design Method. Fermentation. 2019; 5(1):14. https://doi.org/10.3390/fermentation5010014
Chicago/Turabian StyleDarvishi, Farshad, and Nooshin Abolhasan Moghaddami. 2019. "Optimization of an Industrial Medium from Molasses for Bioethanol Production Using the Taguchi Statistical Experimental-Design Method" Fermentation 5, no. 1: 14. https://doi.org/10.3390/fermentation5010014
APA StyleDarvishi, F., & Abolhasan Moghaddami, N. (2019). Optimization of an Industrial Medium from Molasses for Bioethanol Production Using the Taguchi Statistical Experimental-Design Method. Fermentation, 5(1), 14. https://doi.org/10.3390/fermentation5010014





