Gas Fermentation Enhancement for Chemolithotrophic Growth of Cupriavidus necator on Carbon Dioxide
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Gases
2.2. Strain, Media, and Nutrients
2.3. Bottle Cultures
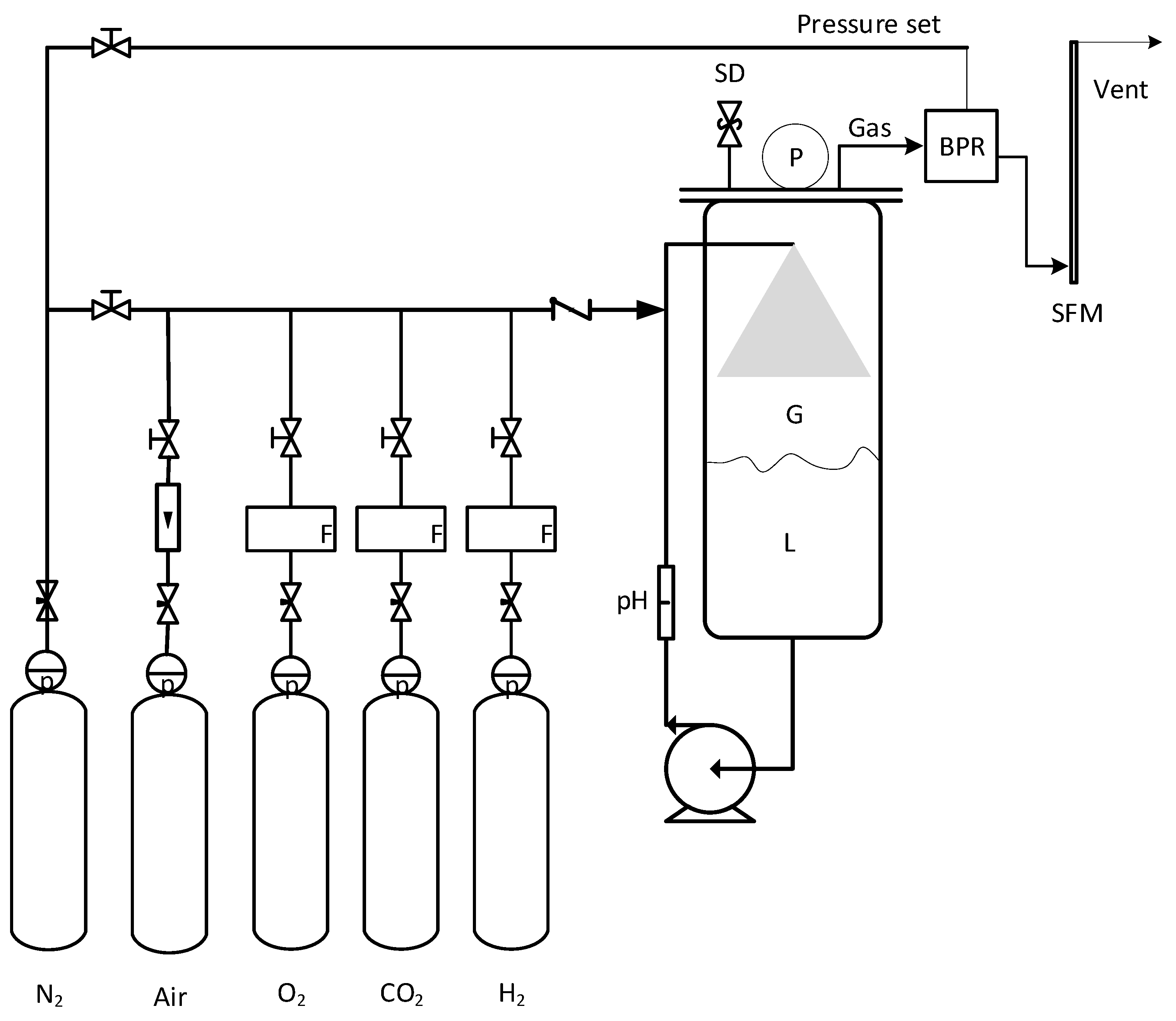
2.4. Bioreactor
2.5. kLa Measurement
2.6. Chemical Analysis
3. Results
3.1. Enhancement by Gas Vector n-Hexadecane
3.1.1. Effect of n-Hexadecane on Oxygen kLa
3.1.2. Effect of n-Hexadecane on Cell Growth
3.2. Enhancement by Gas Pressure
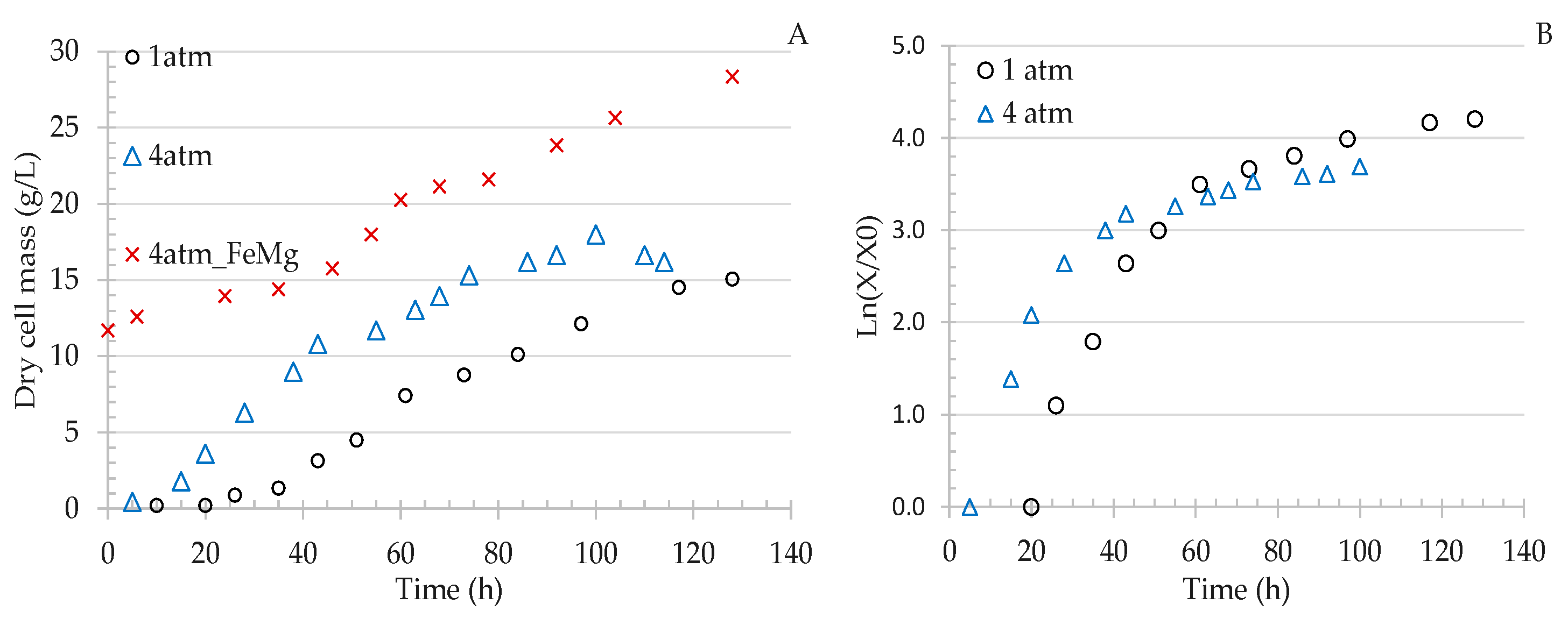
3.2.1. Effect of Gas Pressure on Cell Growth
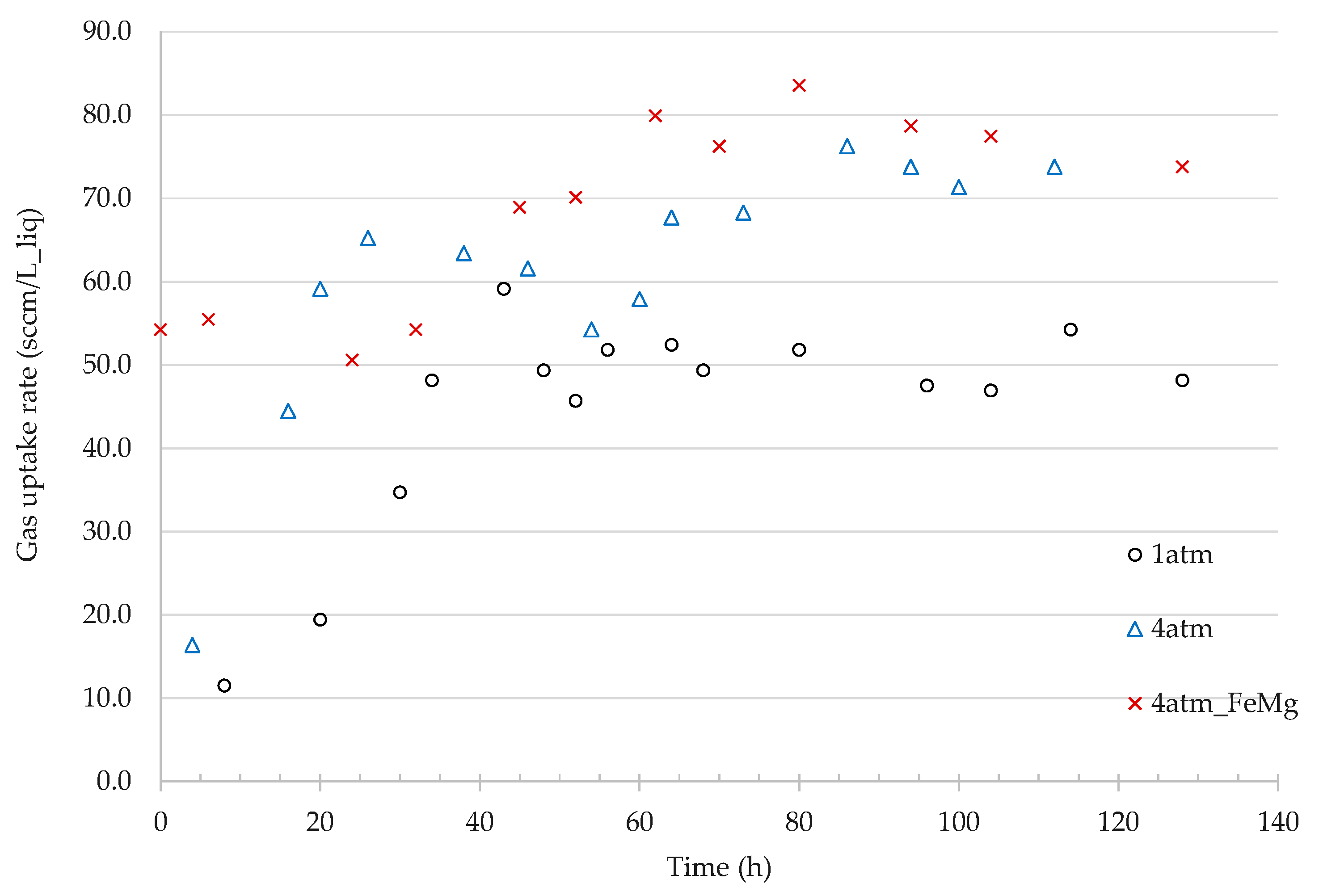
3.2.2. Effect of Pressure on Gas Uptake Rates
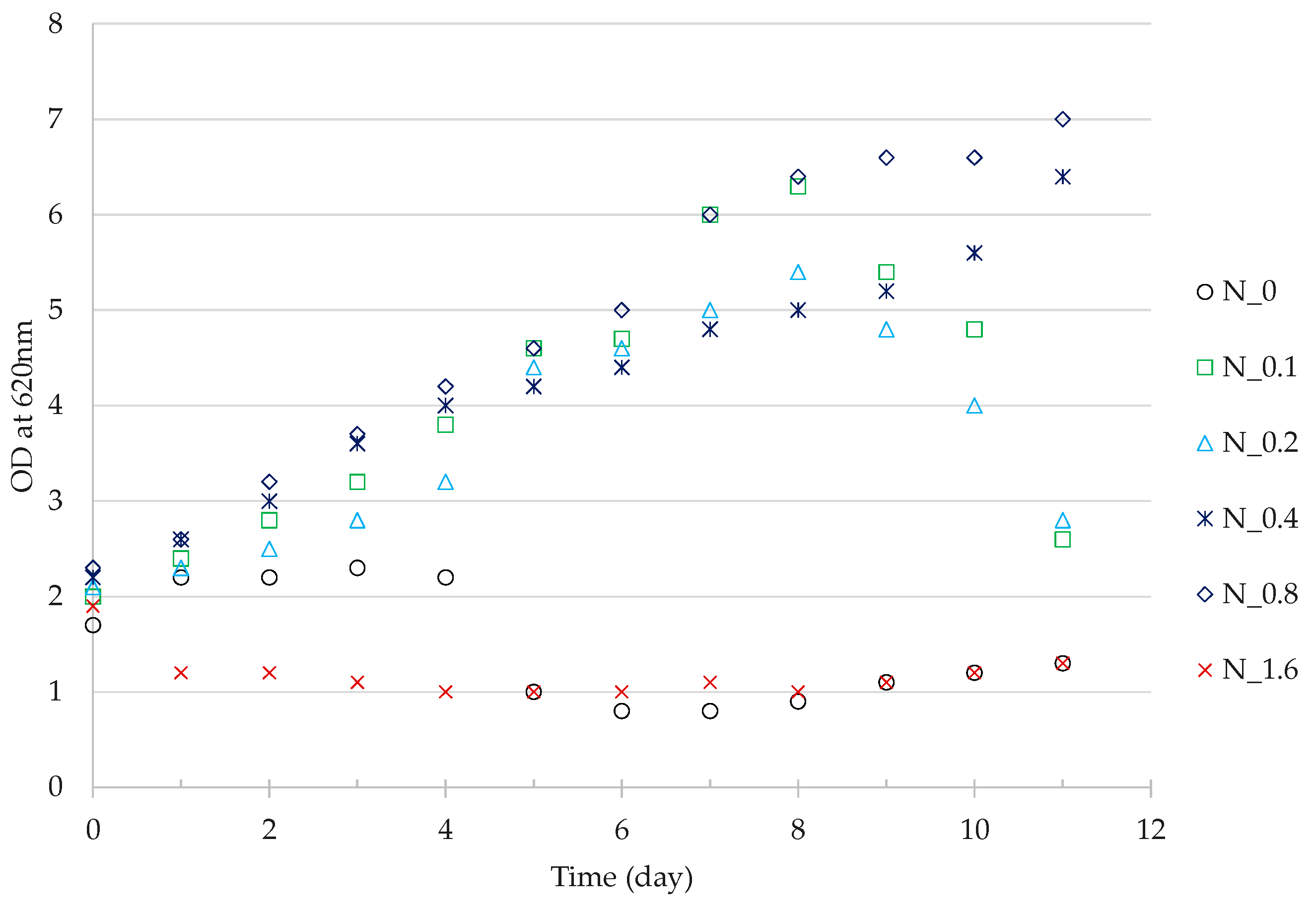
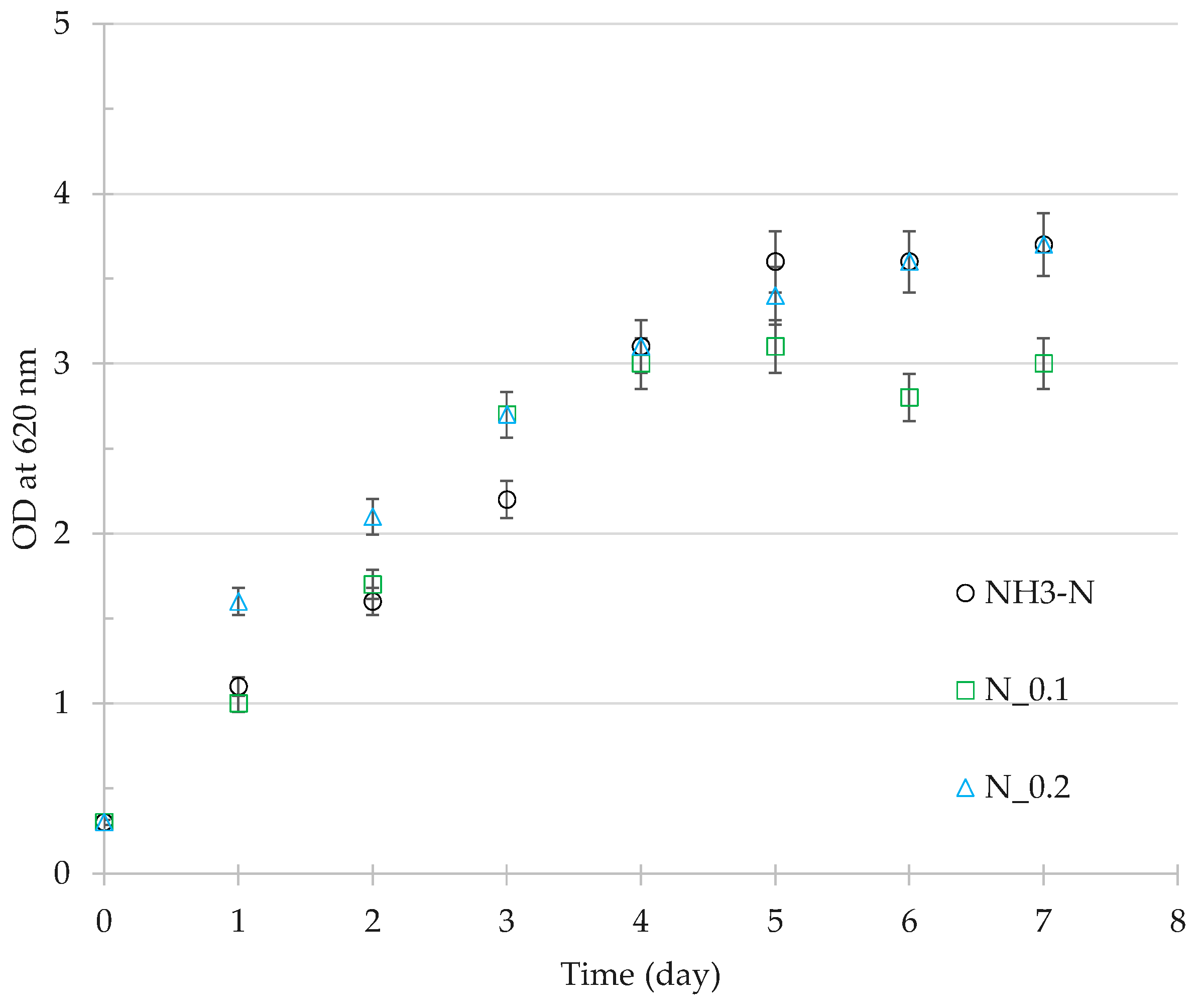
3.3. Residual Cell Mass Hydrolysates as Growth Nutrients
4. Discussion
4.1. Effect of Gas Vector on Gas Fermentation
4.2. Effect of Pressure on Gas Fermentation
4.3. Nutrient Value of Residual Cell Mass
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hunt, A.J.; Sin, E.H.K.; Marriott, R. Generation, capture, and utilization of industrial carbon dioxide. Chem. Sus. Chem. 2010, 3, 306–332. [Google Scholar] [CrossRef] [PubMed]
- Huber, R.; Eder, W. Aquificales. In The Prokaryotes: A Handbook on the Biology of Bacteria, 3rd ed.; Dworkin, M., Falkow, S., Rosenberg, E., Eds.; Springer-Verlag: New York, NY, USA, 2006; Volume 7, pp. 925–928. ISBN 978-0-387-30740-4. [Google Scholar]
- Vandamme, P.; Coeyne, T. Taxonomy of the genus Cupriavidus: A tale of lost and found. Int. J. Syst. Evol. Micr. 2004, 54, 2285–2289. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Ishizaki, A.; Kanamaru, T. Production of poly (D-3-hydroxybutyrate) from CO2, H2, and O2 by high cell density autotrophic cultivation of Alcaligenes eutrophus. Biotech. Bioeng. 1995, 45, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Volova, T.G.; Kiselev, E.G.; Shishatskaya, E.I. Cell growth and accumulation of polyhydroxyalkanoates from CO2 and H2 of a hydrogen-oxidizing bacterium, Cupriavidus eutrophus B.-10646. Bioresour. Technol. 2013, 146, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yu, J. Comparison analysis on the energy efficiencies and biomass yields in microbial CO2 fixation. Process Biochem. 2017, 62, 151–160. [Google Scholar] [CrossRef]
- Yu, J. Fixation of carbon dioxide by a hydrogen-oxidizing bacterium for value-added products. World J. Microbiol. Biotechnol. 2018, 34, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Calloway, D.H.; Kumar, A.M. Protein quality of the bacterium Hydrogenomonas eutropha. Appl. Microbiol. 1969, 17, 176–178. [Google Scholar] [PubMed]
- Waslien, C.I.; Calloway, D.H. Nutritional value of lipids in Hydrogenomonas eutropha as measured in the rat. Appl. Microbiol. 1969, 18, 152–155. [Google Scholar] [PubMed]
- Kang, S.; Yu, J. Hydrophobic organic compounds from hydrothermal liquefaction of bacterial biomass. Biomass Bioenergy 2015, 74, 92–95. [Google Scholar] [CrossRef]
- Nowotny, J.; Bak, T.; Chu, D. Sustainable practices: Solar hydrogen fuel and education program on sustainable energy systems. Int. J. Hydrogen Energy 2014, 39, 4151–4157. [Google Scholar] [CrossRef]
- Garcia-Ochoa, F.; Gomez, E.; Santos, V. Oxygen uptake rate in microbial processes: An overview. Biochem. Eng. J. 2010, 49, 289–307. [Google Scholar] [CrossRef]
- Lu, Y.; Yu, J. Gas mass transfer with microbial CO2 fixation and poly(3-hydroxybutyrate) synthesis in a packed bed bioreactor. Biochem. Eng. J. 2017, 122, 13–21. [Google Scholar] [CrossRef]
- Van Ede, C.J.; Van Houten, R.; Beenackers, A.A.C.M. Enhancement of gas to mass transfer rates by a dispersed organic phase. Chem. Eng. Sci. 1995, 50, 2911–2922. [Google Scholar] [CrossRef]
- Jia, S.; Wang, M.; Kahar, P. Enhancement of yeast fermentation by addition of oxygen vectors in air-lift bioreactor. J. Ferm. Bioeng. 1997, 84, 176–178. [Google Scholar] [CrossRef]
- Preusting, H.; Van Houten, R.; Hoefs, A. High cell density cultivation of Pseudomonas oleovorans: Growth and production of poly(3-hydroxyalkanaotes) in two-liquid phase batch and fed-batch systems. Biotech. Bioeng. 1993, 41, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.S.; Wu, J.Y. Use of n-hexadecane as an oxygen vector to improve Phaffia rhodozyma growth and carotenoid production in shake-flask cultures. J. Appl. Microbiol. 2006, 101, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Knabeen, I.; Regestein, L.; Marquering, F. High cell-density processes in batch mode of a genetically engineered Escherichia coli strain with minimized overflow metabolism using a pressurized bioreactor. J. Biotech. 2010, 150, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Dow, A.; Pingali, S. The energy efficiency of carbon dioxide fixation by a hydrogen-oxidizing bacterium. Int. J. Hydrogen Energy 2013, 38, 8683–8690. [Google Scholar] [CrossRef]
- Tribe, L.A.; Briens, C.L.; Margaritis, A. Determination of the volumetric mass transfer coefficient (kLa) using the dynamic gas-out gas-in method: Analysis of errors caused by dissolved oxygen probes. Biotechnol. Bioeng. 1995, 46, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Ruchti, G.; Dunn, I.J.; Bourne, J.R. Comparison of dynamic oxygen electrode methods for measurement of kLa. Biotechnol. Bioeng. 1981, 23, 277–290. [Google Scholar] [CrossRef]
- Tanadchangsaeng, N.; Yu, J. Microbial synthesis of polyhydroxybutyrate from glycerol: Gluconeogenesis, molecular weight and material properties of biopolyester. Biotechnol. Bioeng. 2012, 109, 2808–2818. [Google Scholar] [CrossRef] [PubMed]
- Bowien, B.; Kusian, B. Genetics and control of CO(2) assimilation in the chemoautotroph Ralstonia eutropha. Arch. Microbiol. 2002, 178, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Berg, I.A. Ecological aspects of the distribution of different autotrophic CO2 fixation pathways. Appl. Environ. Microbiol. 2011, 77, 1925–1936. [Google Scholar] [CrossRef] [PubMed]
- Badger, M.R.; Bek, E.J. Multiple Rubisco forms in proteobacteria: Their functional significance in relation to CO2 acqusition by the CBB cycle. J. Exp. Bot. 2008, 59, 1525–1541. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, H.; Lafferty, R. Novel energy and carbon sources A. The production of biomass from hydrogen and carbon dioxide. In Advances in Biochemical Engineering; Ghose, T.K., Fiechter, A., Eds.; Springer-Verlag: New York, NY, USA, 1971; pp. 143–168. [Google Scholar]
- Zhila, N.; Kalacheva, G.; Volova, T. Fatty acid composition and polyhydroxyalkanoates production by Cupriavidus eutrophus B-10646 cells grown on different carbon sources. Process Biochem. 2015, 50, 69–78. [Google Scholar] [CrossRef]
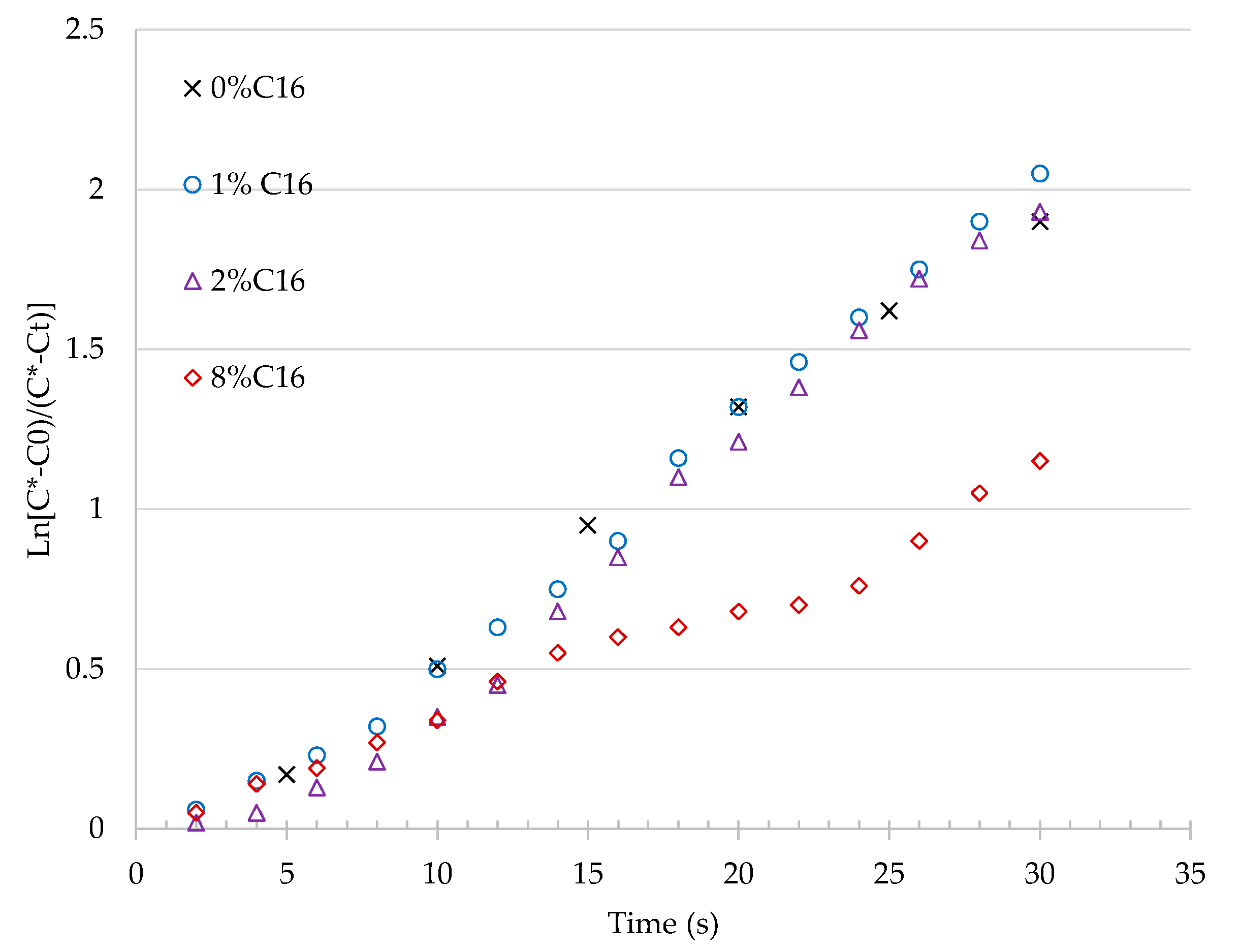
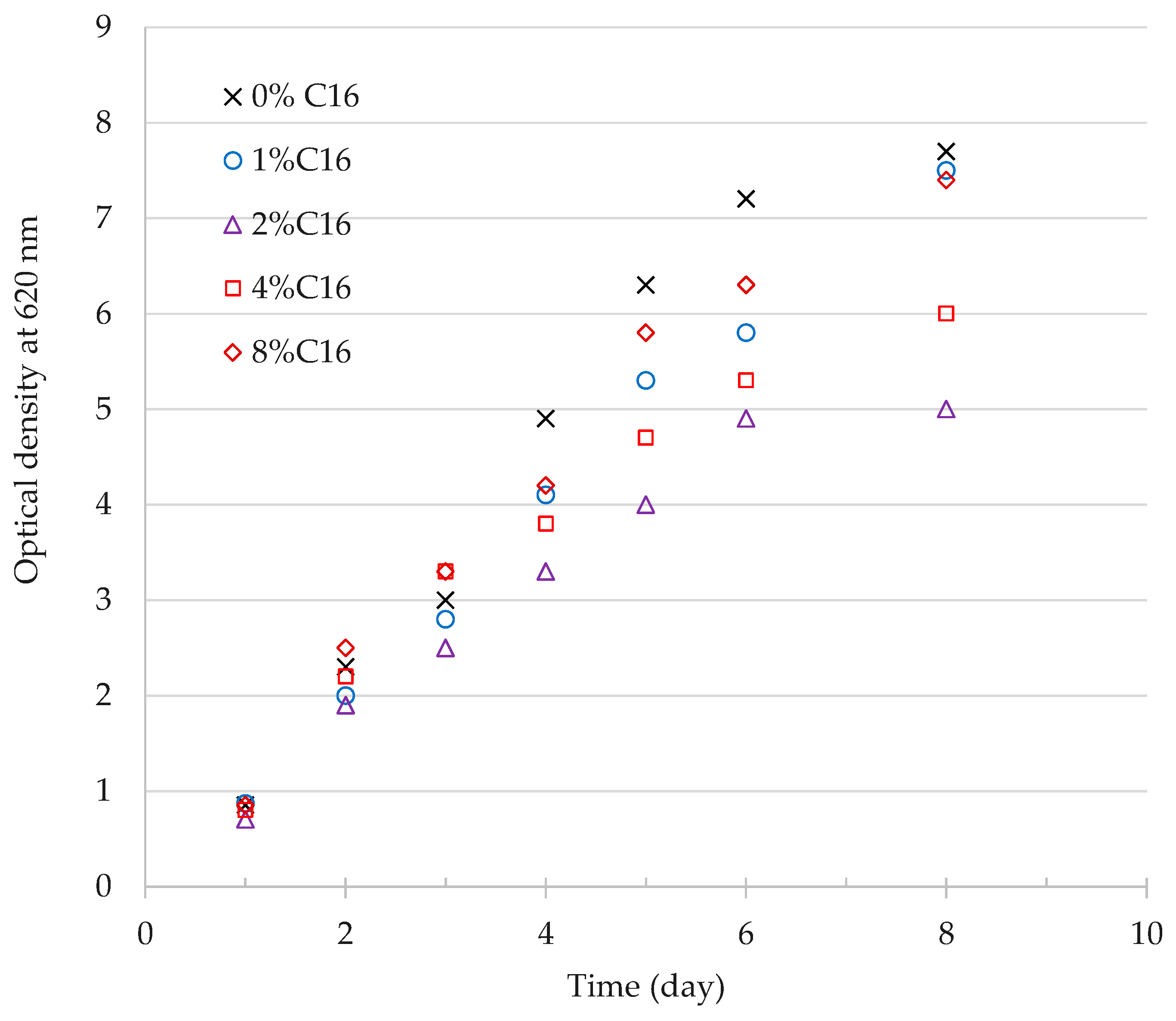
| n-Hexadecane (% Volume) | kLa (h−1) | R2 (-) | kLa Ratio (-) |
|---|---|---|---|
| 0 | 254 | 0.9940 | 1 |
| 1 | 267 | 0.9927 | 1.05 |
| 2 | 273 | 0.9858 | 1.07 |
| 8 | 130 | 0.9783 | 0.51 |
| Run | P a (atm) | FeMg b | X0 c (g L−1) | Xcr d (g L−1) | High Rate | Low Rate | ||
|---|---|---|---|---|---|---|---|---|
| µ e (h−1) | R2 (-) | µ e (h−1) | R2 | |||||
| A | 1 | 1 | 0.5 | 4.5 | 0.094 | 0.960 | 0.011 | 0.975 |
| B | 4 | 1 | 0.45 | 6.3 | 0.12 | 0.976 | 0.010 | 0.954 |
| C | 4 | 2 | 11.7 | None | - | - | 0.0074 | 0.969 |
| Run a | Inlet Gas | Outlet Gas | Uptake Rate (sccm) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Fin b (sccm) | H2 (v %) | O2 (v %) | CO2 (v %) | Fout b (sccm) | H2 (v %) | O2 (v %) | CO2 (v %) | H2 | O2 | CO2 | |
| A | 140 | 60 | 20 | 20 | 67.8 | 60.3 | 15.4 | 24.3 | 43.1 | 17.6 | 11.5 |
| B | 140 | 70 | 20 | 10 | 29.3 | 83.0 | 9.8 | 7.2 | 73.7 | 25.1 | 11.9 |
| C | 140 | 70 | 20 | 10 | 29.3 | 78.0 | 13.0 | 9.0 | 75.1 | 24.2 | 11.4 |
| Residual Cell Mas | Polyhydroxybutyrate (PHB) | |||||
|---|---|---|---|---|---|---|
| C (wt %) | H (wt %) | O (wt %) | N (wt %) | C (wt %) | H (wt %) | O (wt %) |
| 49.0 | 6.9 | 30.1 | 14.0 | 55.8 | 6.9 | 37.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, J.; Munasinghe, P. Gas Fermentation Enhancement for Chemolithotrophic Growth of Cupriavidus necator on Carbon Dioxide. Fermentation 2018, 4, 63. https://doi.org/10.3390/fermentation4030063
Yu J, Munasinghe P. Gas Fermentation Enhancement for Chemolithotrophic Growth of Cupriavidus necator on Carbon Dioxide. Fermentation. 2018; 4(3):63. https://doi.org/10.3390/fermentation4030063
Chicago/Turabian StyleYu, Jian, and Pradeep Munasinghe. 2018. "Gas Fermentation Enhancement for Chemolithotrophic Growth of Cupriavidus necator on Carbon Dioxide" Fermentation 4, no. 3: 63. https://doi.org/10.3390/fermentation4030063
APA StyleYu, J., & Munasinghe, P. (2018). Gas Fermentation Enhancement for Chemolithotrophic Growth of Cupriavidus necator on Carbon Dioxide. Fermentation, 4(3), 63. https://doi.org/10.3390/fermentation4030063






