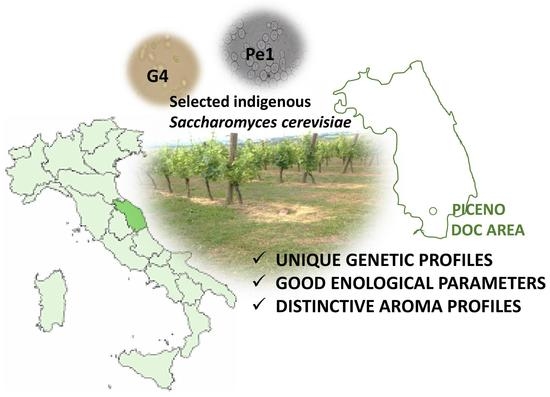
Fitness of Selected Indigenous Saccharomyces cerevisiae Strains for White Piceno DOC Wines Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Strains
2.2. Molecular Identification and Characterization of the S. cerevisiae Isolates
2.3. Fermentation Power Assay (Maximum Ethanol Production)
2.4. Microfermentation Trials
2.5. Analytical Procedures
2.6. Statistical Analysis
3. Results
3.1. Biotyping of the Selected Cultures
3.2. Fermentation Power (FP) and Fermentation Vigour(FV) Test Assay
3.3. Microfermentation Trials Carried Out at 16 °C and 22 °C
3.4. Main Analytical Characteristics
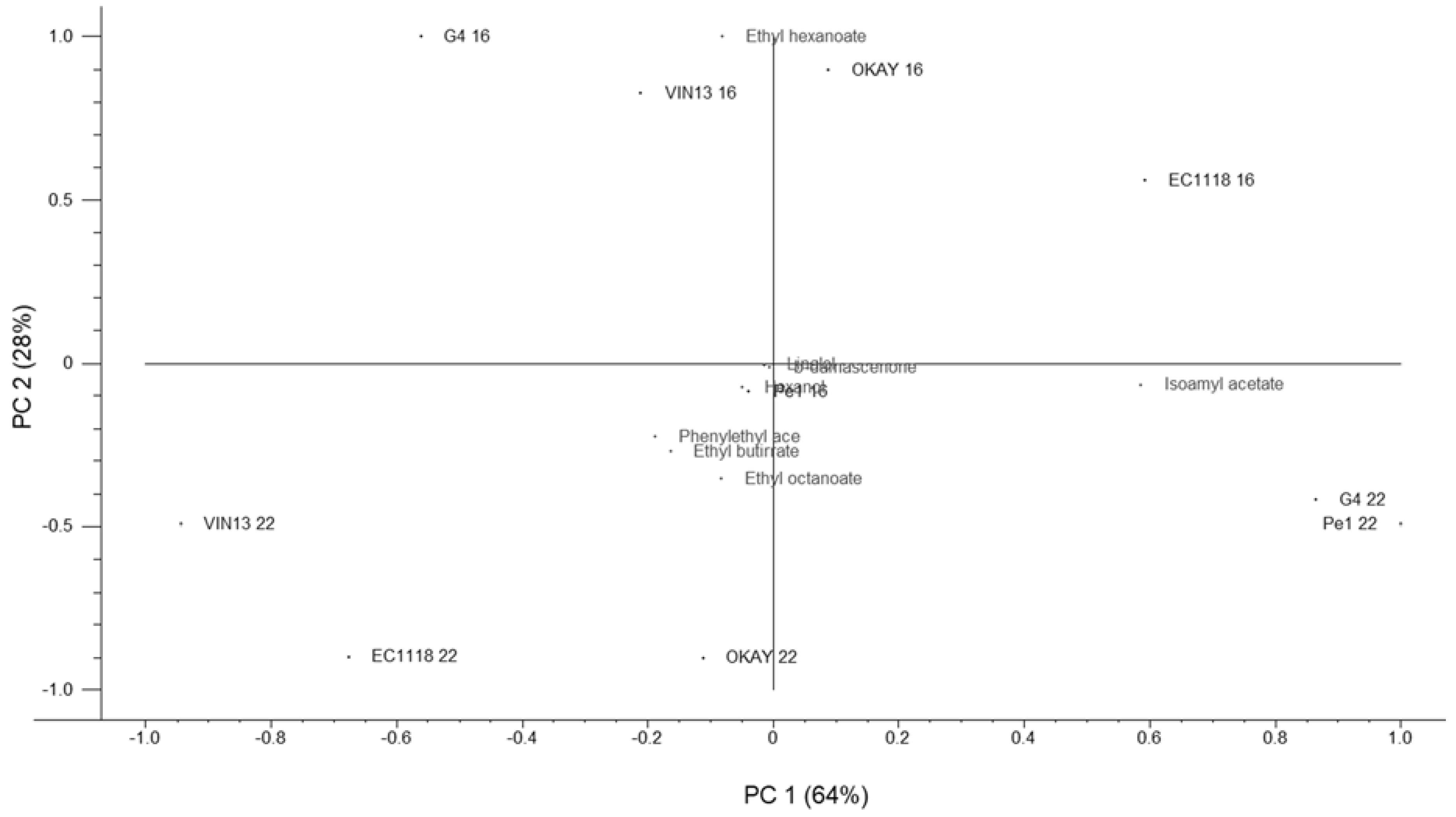
3.5. Main Volatile Compounds
4. Discussion
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Consorziovinipiceni. Available online: http://www.consorziovinipiceni.com (accessed on 8 February 2018).
- Padilla, B.; Gil, J.V.; Manzanares, P. Past and future of non-Saccharomyces yeasts: From spoilage microorganisms to biotechnological tools for improving wine aroma complexity. Front. Microbiol. 2016, 7, 411. [Google Scholar] [CrossRef] [PubMed]
- Callejon, R.M.; Clavijo, A.; Ortigueira, P.; Troncoso, A.M.; Paneque, P.; Morales, M.L. Volatile and sensory profile of organic red wines produced by different selected autochthonous and commercial Saccharomyces cerevisiae strains. Anal. Chim. Acta 2010, 660, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Molina, A.M.; Swiegers, J.H.; Varela, C.; Pretorius, I.S.; Agosin, E. Influence of wine fermentation temperature on the synthesis of yeast-derived volatile aroma compounds. Appl. Microbiol. Biotechnol. 2007, 77, 675–687. [Google Scholar] [CrossRef] [PubMed]
- Torija, M.J.; Beltran, G.; Novo, M.; Poblet, M.; Guillamo, J.M.; Mas, A.; Roze, N. Effects of fermentation temperature and Saccharomyces species on the cell fatty acid composition and presence of volatile compounds in wine. Int. J. Food Microbiol. 2003, 85, 127–136. [Google Scholar] [CrossRef]
- Jolly, N.P.; Varela, C.; Pretorius, I.S. Not your ordinary yeast: Non-Saccharomyces yeasts in wine production uncovered. FEMS Yeast Res. 2014, 14, 215–237. [Google Scholar] [CrossRef] [PubMed]
- Torrens, J.; Urpí, P.; Riu-Aumatell, M.; Vichi, S.; López-Tamames, E.; Buxaderas, S. Different commercial yeast strains affecting the volatile and sensory profile of cava base wine. Int. J. Food Microbiol. 2008, 124, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Pérez, J.M.; Campo, E.; San-Juan, F.; Coque, J.J.R.; Ferreira, V.; Hernández-Orte, P. Sensory and chemical characterization of the aroma of Prieto Picudo rosé wines: The differential role of autochthonous yeast strains on aroma profiles. Food Chem. 2012, 133, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Capece, A.; Granchi, L.; Guerrini, S.; Mangani, S.; Romaniello, R.; Vincenzini, M.; Romano, P. Diversity of Saccharomyces cerevisiae strains isolated from two Italian wine-producing regions. Front. Microbiol. 2016, 7, 1018. [Google Scholar] [CrossRef] [PubMed]
- Capozzi, V.; Garofalo, C.; Chiriatti, M.A.; Grieco, F.; Spano, G. Microbial terroir and food innovation: The case of yeast biodiversity in wine. Microbiol. Res. 2015, 181, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Schüller, D.; Casal, M. The use of genetically modified Saccharomyces cerevisiae strains in the wine industry. Appl. Microbiol. Biot. 2005, 68, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Tofalo, R.; Perpetuini, G.; Fasoli, G.; Schirone, M.; Corsetti, A.; Suzzi, G. Biodiversity study of wine yeasts belonging to the “terroir” of Montepulciano d’Abruzzo “Colline Teramane” revealed Saccharomyces cerevisiae strains exhibiting atypical and unique 5.8 S-ITS restriction patterns. Food Microbiol. 2014, 39, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Valero, A.; Marín, S.; Rarnos, A.J.; Sanchis, V. Ochratoxin A-producing species in grapes and sun-dried grapes and their relation to ecophysiological factors. Lett. Appl. Microbiol. 2005, 41, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Comitini, F.; Gobbi, M.; Domizio, P.; Romani, C.; Lencioni, L.; Mannazzu, I.; Ciani, M. Selected non-Saccharomyces wine yeasts in controlled multistarter fermentations with Saccharomyces cerevisiae. Food Microbiol. 2011, 28, 873–888. [Google Scholar] [CrossRef] [PubMed]
- Bokulich, N.A.; Thorngate, J.H.; Richardson, P.M.; Mills, D.A. Microbial biogeography of wine grapes is conditioned by cultivar, vintage, and climate. Proc. Natl. Acad. Sci. USA 2014, 111, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Legras, J.L.; Karst, F. Optimisation of interdelta analysis for Saccharomyces cerevisiae strain characterisation. FEMS microbiol. Lett. 2003, 221, 249–255. [Google Scholar] [CrossRef]
- EEC. Council Regulation 2870/00 laying down Community reference methods for the analysis of spirit drinks. Off. J. Eur. Comm. 2000, L333, 20–46. [Google Scholar]
- Dukes, B.C.; Butzke, C.E. Rapid determination of primary amino acids in grape juice using an o-phthaldialdehyde/Nacetyl-l-cysteine spectrophotometric assay. Am. J. Enol. Vitic. 1998, 49, 125–134. [Google Scholar]
- Vigentini, I.; Barrera Cardenas, S.; Valdetara, F.; Faccincani, M.; Panont, C.A.; Picozzi, C.; Foschino, R. Use of native yeast strains for in-bottle fermentation to face the uniformity in sparkling wine production. Front. Microbiol. 2017, 8, 1225. [Google Scholar] [CrossRef] [PubMed]
- Canonico, L.; Comitini, F.; Ciani, M. Influence of vintage and selected starter on Torulaspora delbrueckii/Saccharomyces cerevisiae sequential fermentation. Eur. Food Res. Technol. 2015, 241, 827–833. [Google Scholar] [CrossRef]
- Gayevskiy, V.; Goddard, M.R. Geographic delineations of yeast communities and populations associated with vines and wines in New Zealand. ISME J. 2012, 6, 1281. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.W.; Turner, E.; Townsend, J.P.; Dettman, J.R.; Jacobson, D. Eukaryotic microbes, species recognition and the geographic limits of species: Examples from the Kingdom Fungi. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2006, 361, 1947–1963. [Google Scholar] [CrossRef] [PubMed]
- Milanović, V.; Comitini, F.; Ciani, M. Grape berry yeast communities: Influence of fungicide treatments. Int. J. Food Microbiol. 2013, 161, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Vigentini, I.; De Lorenzis, G.; Fabrizio, V.; Valdetara, F.; Faccincani, M.; Panont, C.A.; Foschino, R. The vintage effect overcomes the terroir effect: A three year survey on the wine yeast biodiversity in Franciacorta and Oltrepò Pavese, two northern Italian vine-growing areas. Microbiology 2015, 161, 362–373. [Google Scholar] [CrossRef] [PubMed]
- Bokulich, N.A.; Lewis, Z.T.; Boundy-Mills, K.; Mills, D.A. A new perspective on microbial landscapes within food production. Curr. Opin. Biotechnol. 2016, 37, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Knight, S.; Klaere, S.; Fedrizzi, B.; Goddard, M.R. Regional microbial signatures positively correlate with differential wine phenotypes: Evidence for a microbial aspect to terroir. Sci. Rep. 2015, 5, 14233. [Google Scholar] [CrossRef] [PubMed]
- Knight, S.; Goddard, M.R. Quantifying separation and similarity in a Saccharomyces cerevisiae metapopulation. ISME J. 2015, 9, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Lopandic, K.; Gangl, H.; Wallner, E.; Tscheik, G.; Leitner, G.; Querol, A.; Borth, N.; Breitenbach, M.; Prillinger, H.; Tiefenbrunner, W. Genetically different wine yeasts isolated from Austrian vine-growing regions influence wine aroma differently and contain putative hybrids between Saccharomyces cerevisiae and Saccharomyces kudriavzevii. FEMS Yeast Res. 2007, 7, 953–965. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.W.; Tsai, P.; Anfang, N.; Ross, H.A.; Goddard, M.R. Pyrosequencing reveals regional differences in fruit-associated fungal communities. Environ. Microbiol. 2014, 16, 2848–2858. [Google Scholar] [CrossRef] [PubMed]
- Schüller, D.; Casal, M. The genetic structure of fermentative vineyard-associated Saccharomyces cerevisiae populations revealed by microsatellite analysis. Antonie Van Leeuwenhoek 2007, 91, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Cordero-Bueso, G.; Esteve-Zarzoso, B.; Gil-Díaz, M.; García, M.; Cabellos, J.M.; Arroyo, T. Improvement of Malvar wine quality by use of locally-selected Saccharomyces cerevisiae strains. Fermentation 2016, 2, 7. [Google Scholar] [CrossRef]
- Suzzi, G.; Arfelli, G.; Schirone, M.; Corsetti, A.; Perpetuini, G.; Tofalo, R. Effect of grape indigenous Saccharomyces cerevisiae strains on Montepulciano d’Abruzzo red wine quality. Food Res. Int. 2012, 46, 22–29. [Google Scholar] [CrossRef]
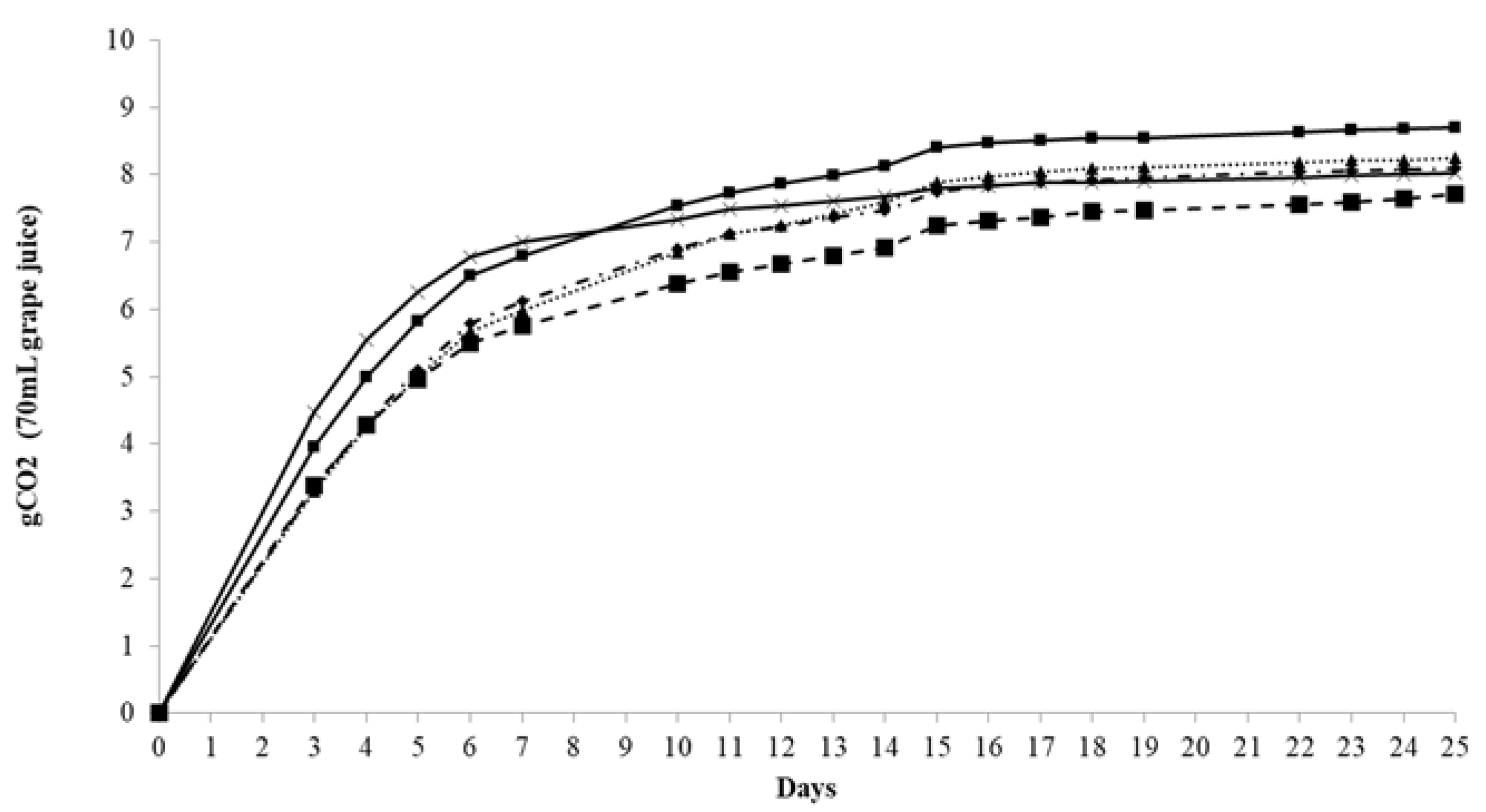
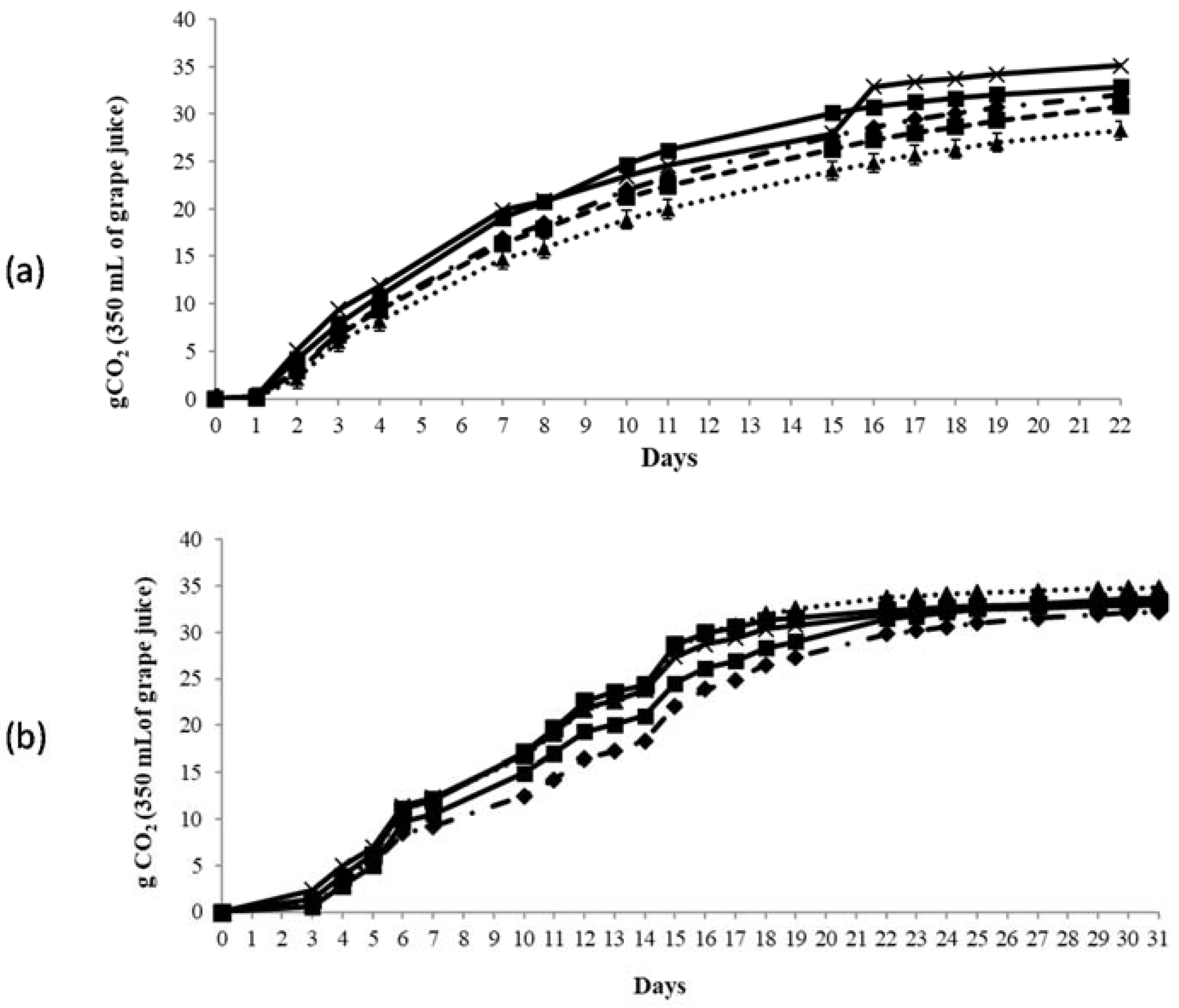
 ); VIN13 (
); VIN13 (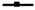 ); EC1118 (
); EC1118 ( ); Pe1 (
); Pe1 ( ); and G4 (
); and G4 ( ).
).
 ); VIN13 (
); VIN13 ( ); EC1118 (
); EC1118 ( ); Pe1 (
); Pe1 ( ); and G4 (
); and G4 ( )).
)).
 ); VIN13 (
); VIN13 ( ); EC1118 (
); EC1118 ( ); Pe1 (
); Pe1 ( ); and G4 (
); and G4 ( )).
)).
| Strains | Ethanol (% v/v) | Residual Sugar (g/L) | * Fermentation Vigour (g CO2/3 Days) |
|---|---|---|---|
| OKAY | 13.05 ± 0.27 c | 24.04 ± 2.75 b,c | 3.34 ± 0.26 b |
| VIN13 | 14.34 ± 0.10 a | 7.36 ± 2.92 d | 3.95 ± 0.34 a,b |
| EC1118 | 14.23 ± 0.07 a | 21.09 ± 0.80 c | 3.30 ± 0.16 a |
| Pe1 | 13.78 ± 0.10 b | 27.38 ± 2.34 b | 4.47 ± 0.01 a |
| G4 | 12.15 ± 0.06 d | 35.90 ± 0.21 a | 3.38 ± 0.40 b |
| 22 °C | 16 °C | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Strains | Ethanol (% v/v) | Volatile Acidity (Acetic Acid g/L) | Glycerol (g/L) | SO2 (mg/L) | Residual Sugar (g/L) | Fermentation Rate (g CO2/3 Days) | Ethanol (% v/v) | Volatile Acidity (Acetic Acid g/L) | Glycerol (g/L) | SO2 (mg/L) | Residual Sugar (g/L) | Fermentation Rate (g CO2/3 Days) |
| OKAY | 12.26 ± 0.00 a | 0.38 ± 0.03 b | 4.77 ± 0.39 a | 5.76 ± 1.35 ab | 0.04 ± 0.00 a | 6.97 ± 1.36 b | 11.77 ± 0.07 b,c | 0.53 ± 0.04 a | 4.56 ± 0.08 e | 14.12 ± 1.30 b | 0.03 ± 0.02 a | 1.28 ± 0.04 b,c |
| VIN13 | 12.00 ± 0.06 a | 0.38 ± 0.03 b | 3.81 ± 0.05 b | 7.67 ± 0.91 a | 0.01 ± 0.00 c | 7.97 ± 0.46 a,b | 12.45 ± 0.21 a | 0.41 ± 0.04 b | 5.47 ± 0.13 c | 14.88 ± 0.22 b | 0.01 ± 0.00 a | 1.39 ± 0.34 b |
| EC1118 | 11.56 ± 0.27 b | 0.44 ± 0.03 b | 4.46 ± 0.13 a | 0.16 ± 0.22 c | 0.01 ± 0.01 b,c | 6.04 ± 0.88 b | 11.33 ± 0.23 c | 0.38 ± 0.01 b | 4.99 ± 0.04 d | 16.39 ± 0.09 a,b | 0.04 ± 0.03 a | 0.71 ± 0.19 b,c |
| Pe1 | 11.96 ± 0.05 a | 0.40 ± 0.05 b | 4.57 ± 0.10 a | 3.36 ± 0.67 b | 0.04 ± 0.01 a,b | 9.41 ± 0.40 a | 11.50 ± 0.21 b,c | 0.44 ± 0.04 b | 6.42 ± 0.12 a | 16.48 ± 2.50 a,b | 0.02 ± 0.01 a | 2.37 ± 0.46 a |
| G4 | 11.96 ± 0.00 a | 0.64 ± 0.02 a | 4.93 ± 0.07 a | 9.44 ± 2.94 a | 0.02 ± 0.02 a,b,c | 6.75 ± 0.48 b | 11.92 ± 0.11 b | 0.54 ± 0.01 a | 5.82 ± 0.02 b | 18.56 ± 0.45 a | 0.01 ± 0.00 a | 0.57 ± 0.19 c |
| Volatile Compounds (mg L−1) | 22 °C | 16 °C | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Esters | OKAY | VIN13 | EC1118 | Pe1 | G4 | OKAY | VIN13 | EC1118 | Pe1 | G4 |
| Ethyl acetate | 23.46 ± 0.51 b | 20.51 ± 0.10 c | 33.25 ± 0.01 a | 14.47 ± 0.33 e | 16.74 ± 0.15 d | 25.53 ± 0.15 b | 25.62 ± 0.46 b | 31.03 ± 0.37 a | 15.92 ± 0.12 c | 15.95 ± 0.53 c |
| Alcohols | ||||||||||
| n-propanol | 40.54 ± 0.20 a | 18.41 ± 0.16 c | 19.62 ± 0.06 b | 13.76 ± 0.04 e | 15.705 ± 0.20 d | 41.70 ± 0.24 a | 24.58 ± 0.01 b | 21.34 ± 0.31 c | 16.00 ± 0.49 e | 19.95 ± 0.56 d |
| Isobutanol | 5.166 ± 0.17 d | 7.049 ± 0.08 c | 13.681 ± 0.02 a | 6.777 ± 0.20 c | 9.548 ± 0.30 b | 10.20 ± 0.50 b | 7.41 ± 0.37 c | 11.88 ± 0.30 a | 8.44 ± 0.46 c | 11.60 ± 0.58 a |
| Amilyc alcohol | 13.35 ± 0.05 d | 14.70 ± 0.31 c | 18.82 ± 0.53 a | 11.45 ± 0.02 e | 16.71 ± 0.04 b | 10.67 ± 0.40 c | 12.34 ± 0.30 b | 10.77 ± 0.07 c | 12.43 ± 0.51 b | 14.14 ± 0.62 a |
| Isoamilyc alcohol | 94.43 ± 0.47 c | 96.04 ± 0.40 b | 105.49 ± 0.30 a | 80.38 ± 0.48 e | 82.54 ± 0.25 d | 74.55 ± 0.37 d | 80.82 ± 0.74 c | 112.43 ± 2.87 a | 61.91 ± 0.59 e | 88.64 ± 0.36 b |
| β-Phenyl ethanol | 34.95 ± 7.02 a | 11.32 ± 0.32 b | 13.03 ± 0.36 b | 11.04 ± 1.12 b | 12.13 ± 0.77 b | 11.25 ± 0.11 c | 14.3 ± 0.09 b | 16.7 ± 0.05 a | 10.82 ± 0.01 c | 13.2 ± 0.03 b |
| Carbonyl compounds | ||||||||||
| Acetaldehyde | 10.57 ± 0.14 e | 20.38 ± 0.48 d | 91.33 ± 0.29 a | 53.77 ± 0.12 b | 32.355 ± 0.24 c | 13.07 ± 0.44 d | 19.56 ± 0.40 c | 134.901 ± 0.80 a | 30.86 ± 2.67 b | 9.90 ± 0.13 d |
| Acetoin | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agarbati, A.; Canonico, L.; Ciani, M.; Comitini, F. Fitness of Selected Indigenous Saccharomyces cerevisiae Strains for White Piceno DOC Wines Production. Fermentation 2018, 4, 37. https://doi.org/10.3390/fermentation4020037
Agarbati A, Canonico L, Ciani M, Comitini F. Fitness of Selected Indigenous Saccharomyces cerevisiae Strains for White Piceno DOC Wines Production. Fermentation. 2018; 4(2):37. https://doi.org/10.3390/fermentation4020037
Chicago/Turabian StyleAgarbati, Alice, Laura Canonico, Maurizio Ciani, and Francesca Comitini. 2018. "Fitness of Selected Indigenous Saccharomyces cerevisiae Strains for White Piceno DOC Wines Production" Fermentation 4, no. 2: 37. https://doi.org/10.3390/fermentation4020037
APA StyleAgarbati, A., Canonico, L., Ciani, M., & Comitini, F. (2018). Fitness of Selected Indigenous Saccharomyces cerevisiae Strains for White Piceno DOC Wines Production. Fermentation, 4(2), 37. https://doi.org/10.3390/fermentation4020037