Ethanol Production by Soy Fiber Treatment and Simultaneous Saccharification and Co-Fermentation in an Integrated Corn-Soy Biorefinery
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Compositional Analyses
2.2.1. Proximate Analyses
2.2.2. Carbohydrate Composition and Lignin Content
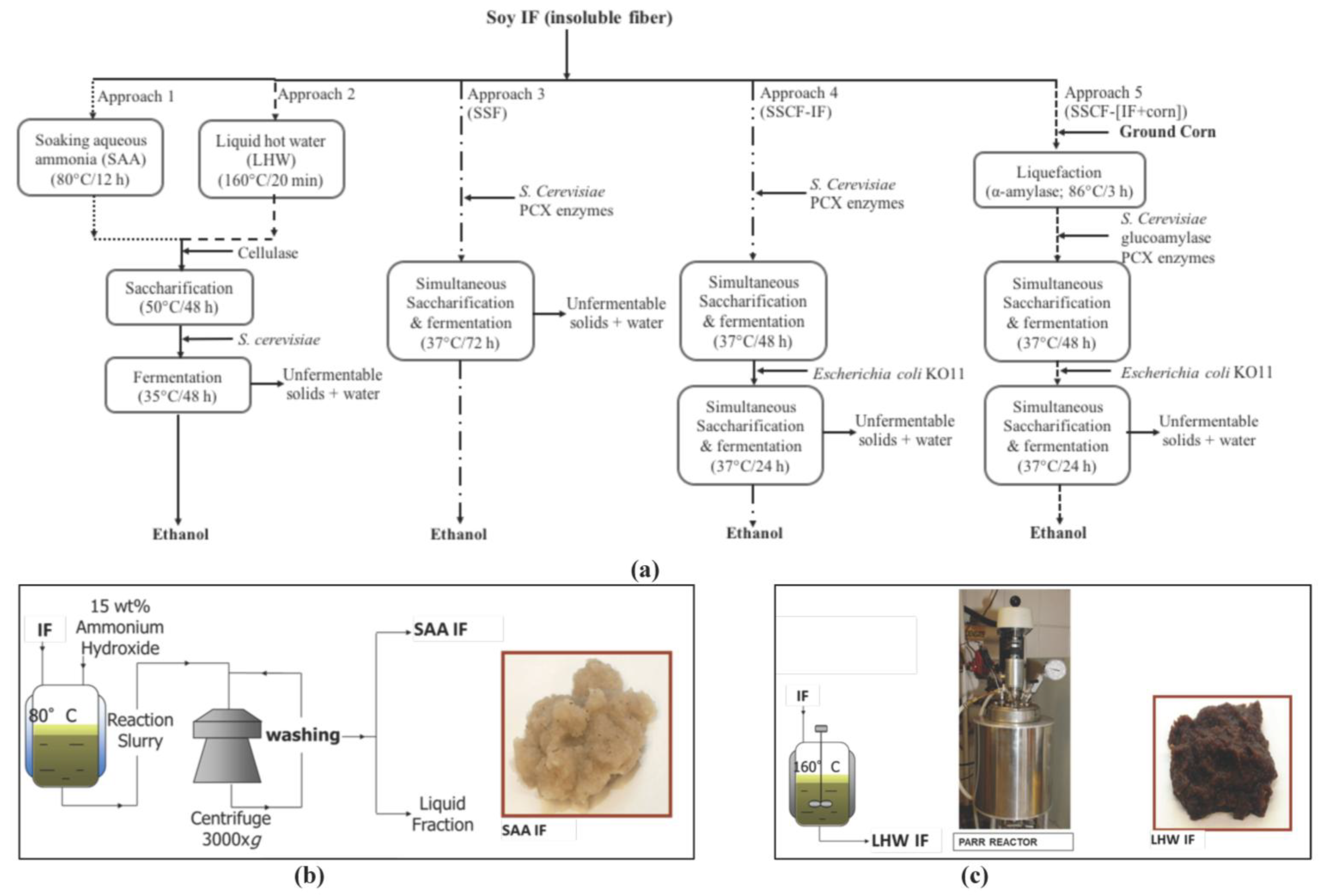
2.3. Insoluble Fiber Treatment
2.3.1. Effect of the Treatment Method on Ethanol Production in Insoluble Fiber Fermentation
Soaking in Aqueous Ammonia Treatment
Liquid Hot Water Treatment
Enzymatic Hydrolysis
2.3.2. Simultaneous Saccharification and Co-Fermentation of Insoluble Fiber
Yeast-Extract Peptone Media
Escherichia coli KO11 Inoculum
Simultaneous Saccharification and Co-Fermentation with S. cerevisiae and E. coli KO11
2.4. Scale-Up
2.5. Integrated Corn-Soy Fermentation
2.5.1. Post-Fermentation Processing
2.5.2. Oil Recovery
2.6. Effect of Ethanol and Sugar Concentration on the Growth of E. coli KO11
2.7. Prediction of E. coli KO11 Kinetics Using Monod Nonlinear Modeling
2.8. HPLC Analysis
2.9. Statistical Analysis
3. Results and Discussion
3.1. Treatment of Insoluble Fiber
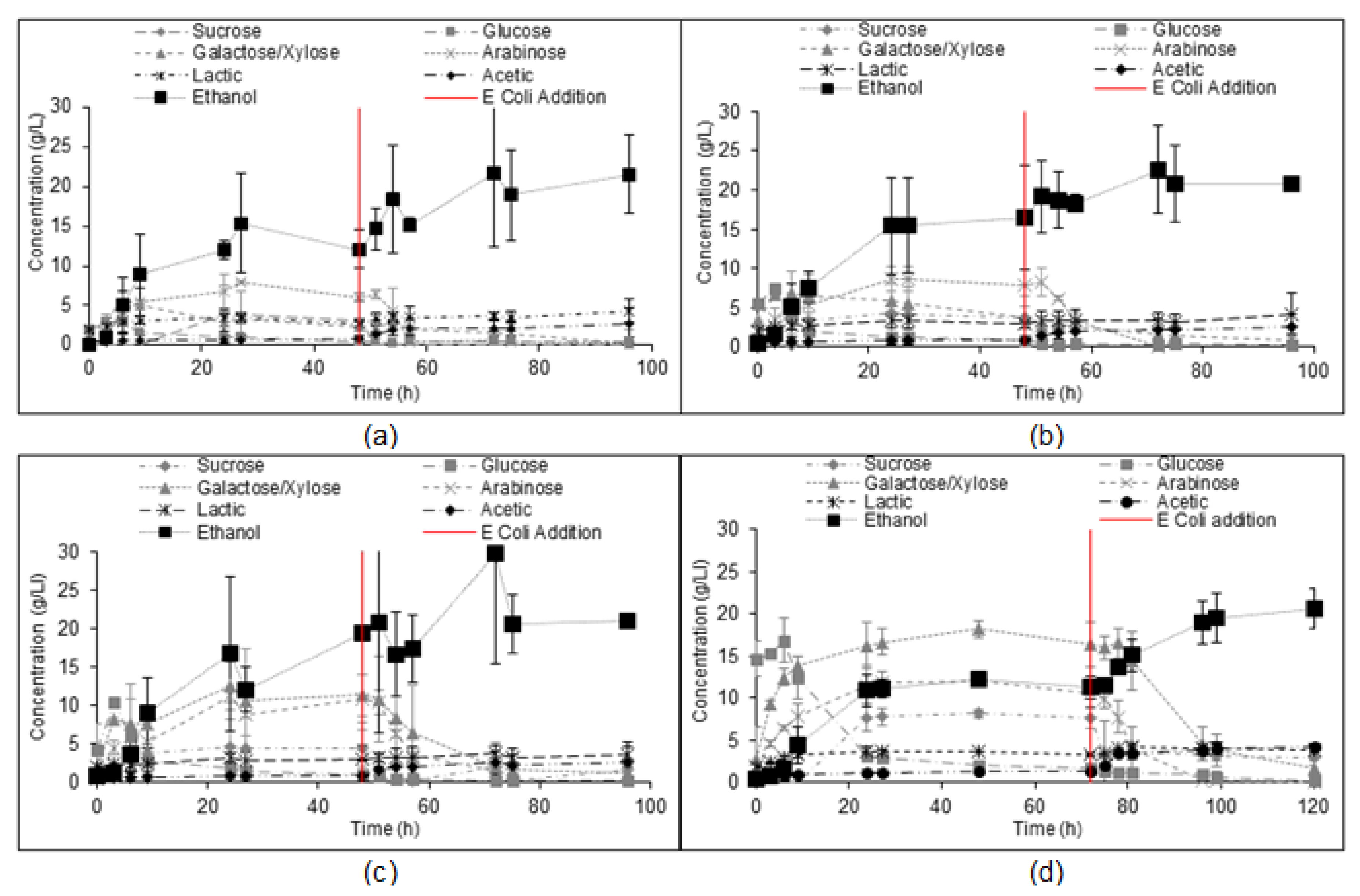
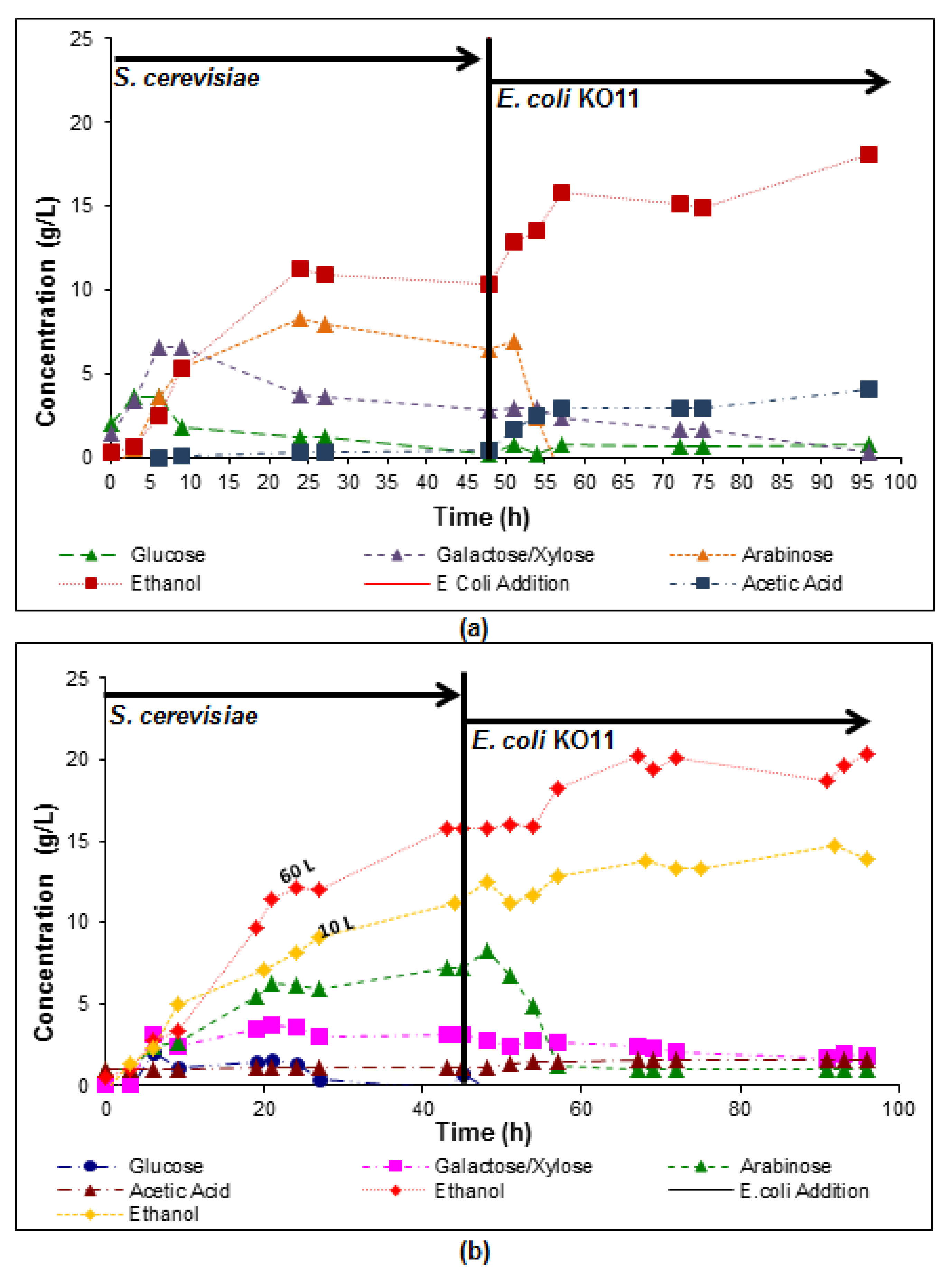
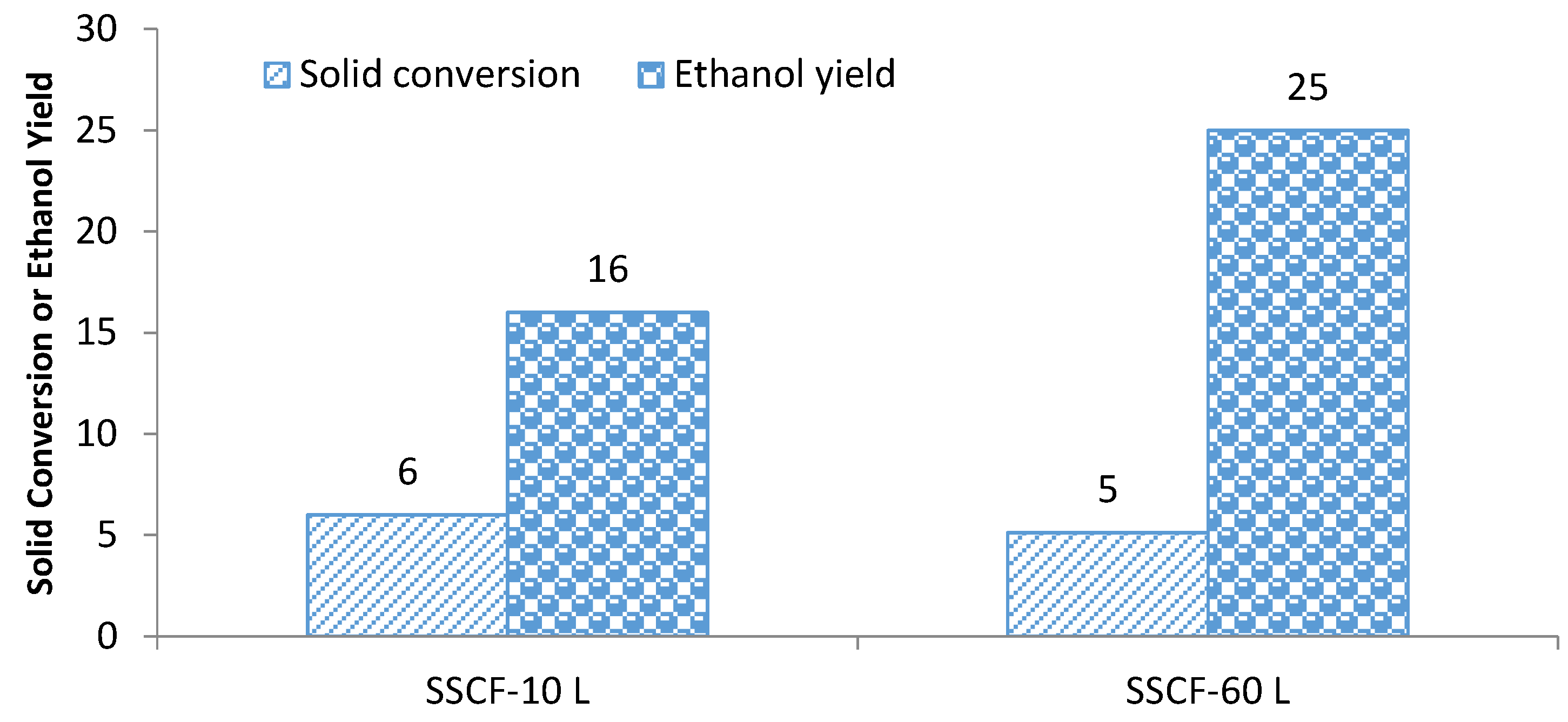
3.2. Scale-Up of the Simultaneous Saccharification and Co-Fermentation of Insoluble Fiber
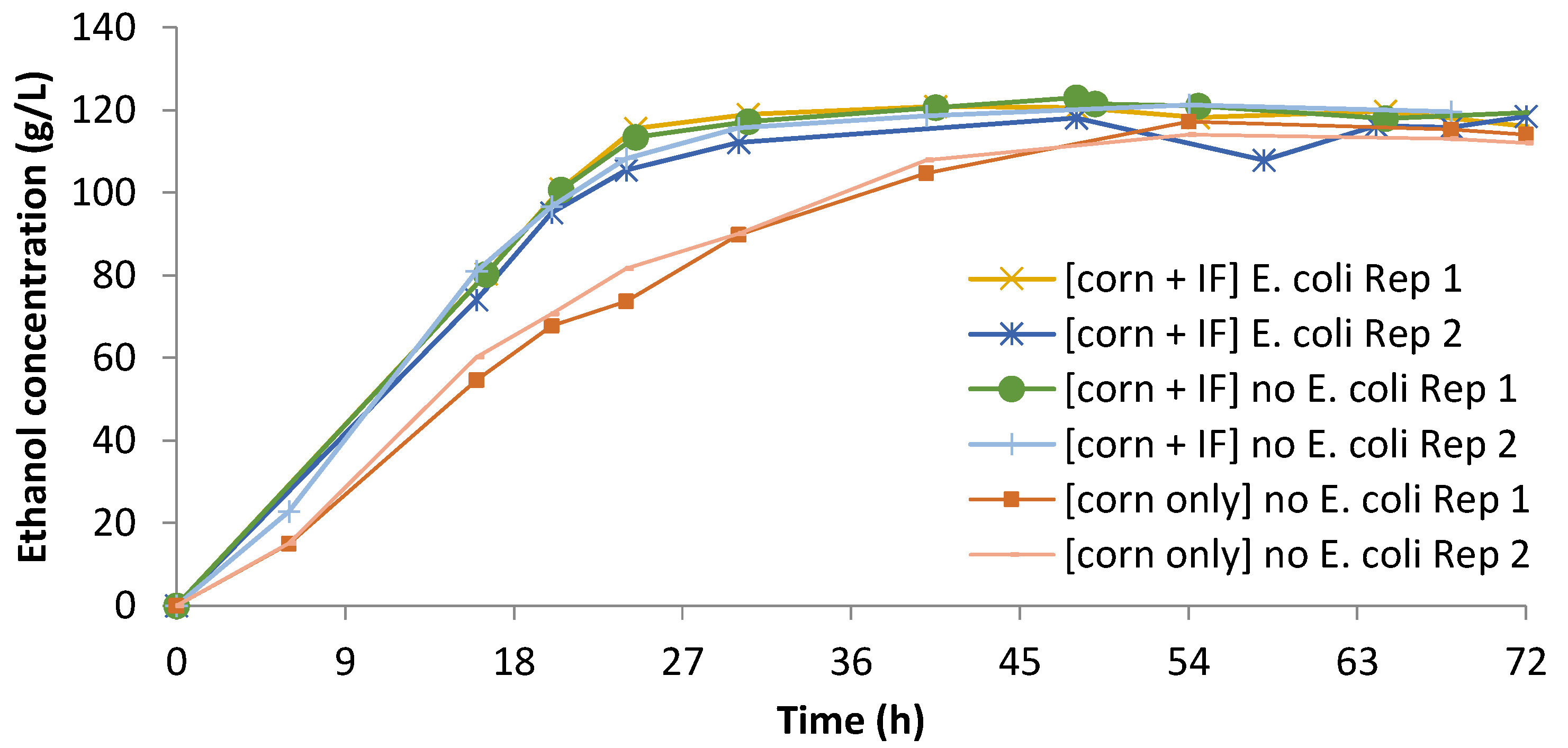
3.3. Integrated Corn-Soy Fermentation
3.4. Effect of Ethanol and Sugar Concentration on E. coli KO11
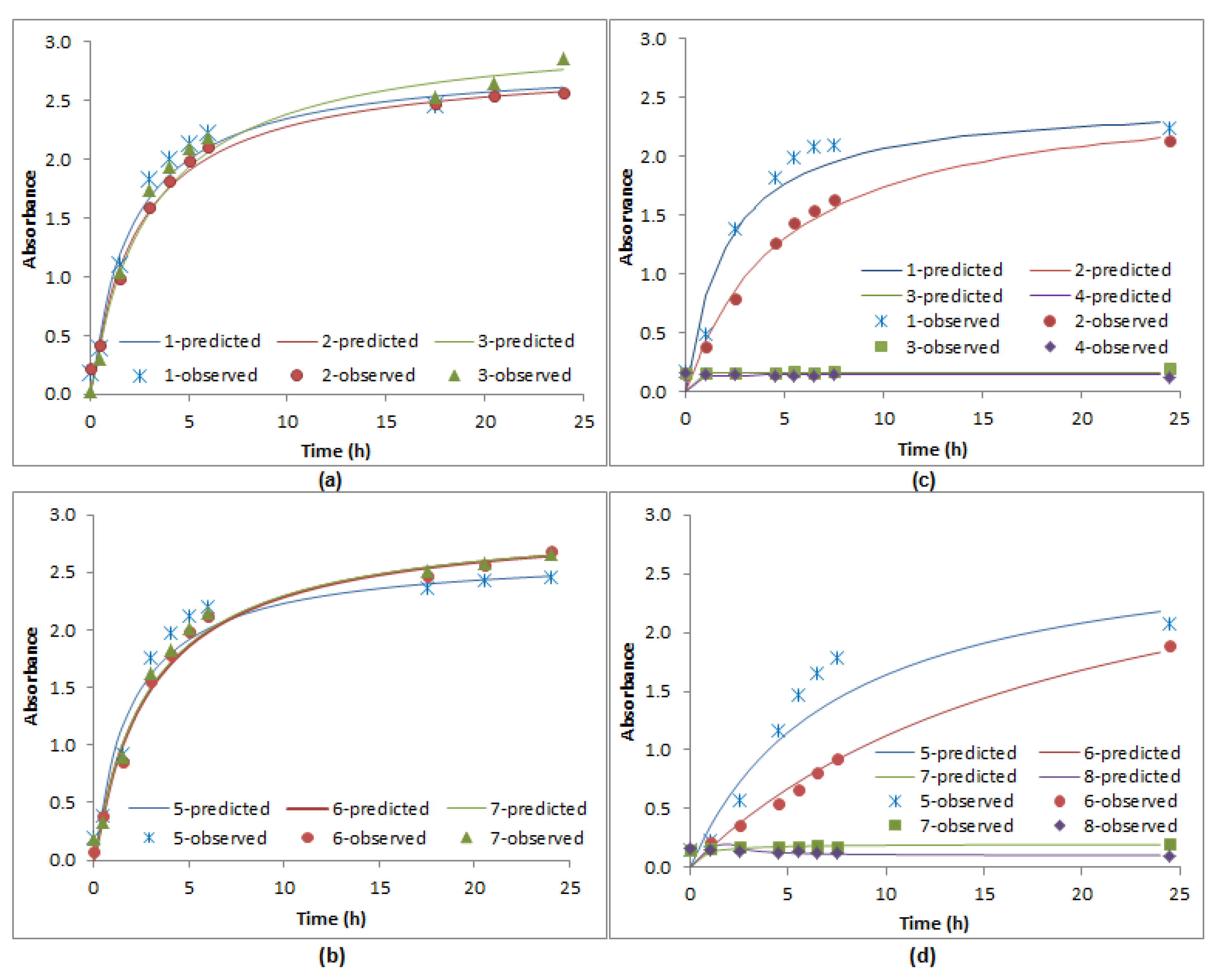
3.5. Prediction of E. coli KO11 Kinetics
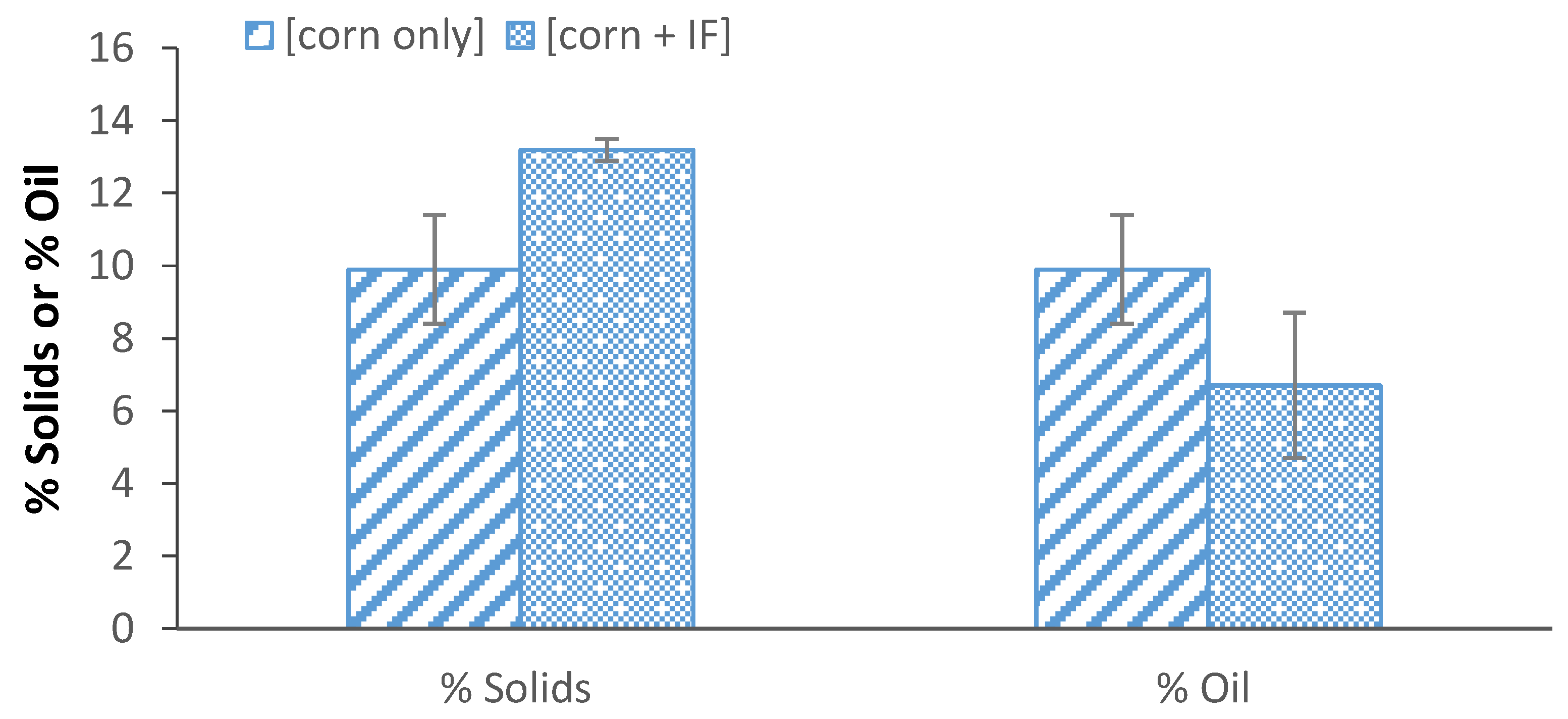
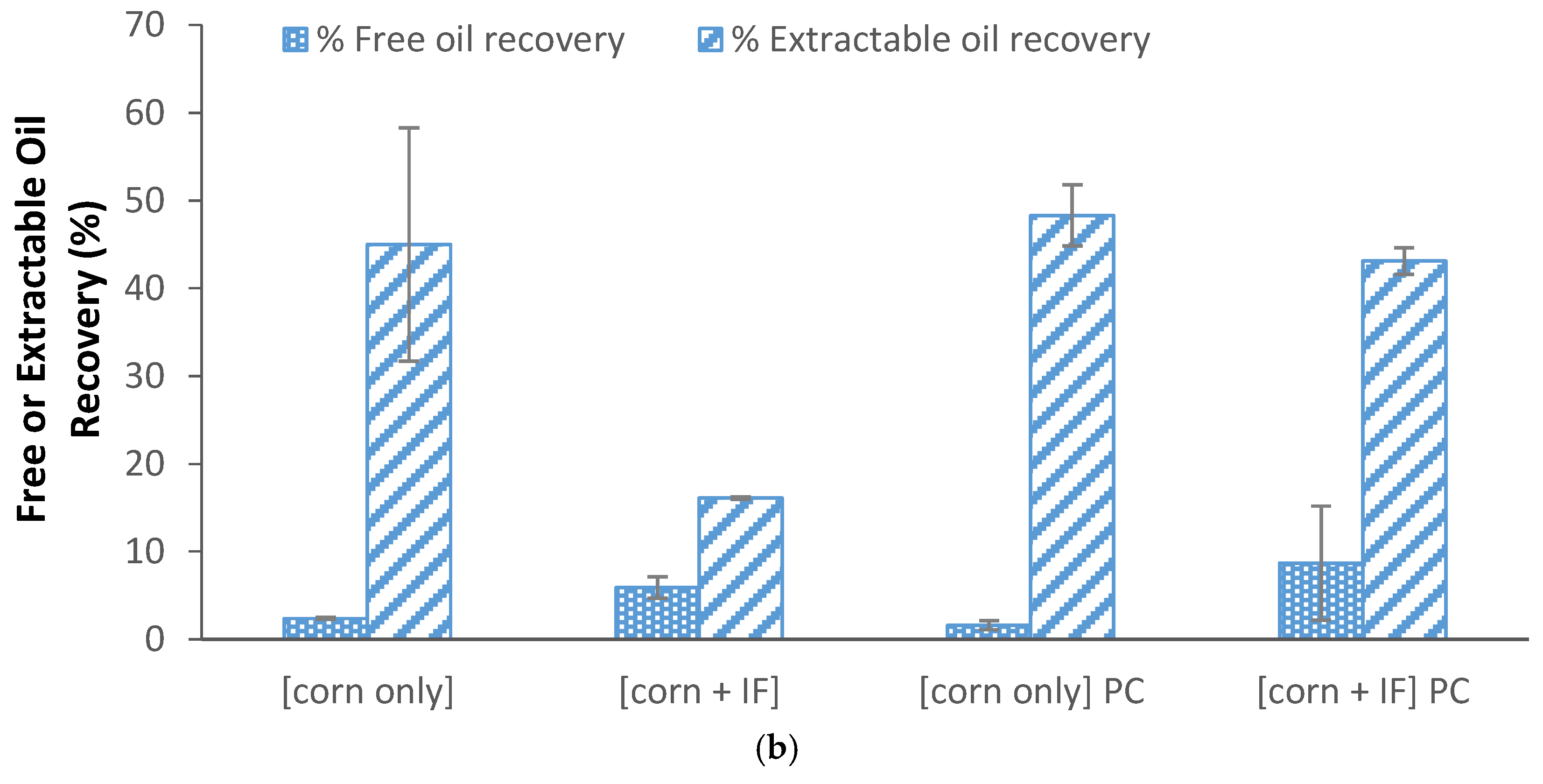
3.6. Effect of Insoluble Fiber on Oil Recovery from Integrated Corn-Soy Fermenation
3.7. Effect of Enzymes on Solids Partition and Oil Recovery
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, Y.; Zhang, Y.; Wang, M.; Jiang, L.; Sui, X. Simplex-centroid mixture design applied to the aqueous enzymatic extraction of fatty acid-balanced oil from mixed seeds. J. Am. Oil Chem. Soc. 2013, 90, 349–357. [Google Scholar] [CrossRef]
- De Moura, J.M.L.N.; Maurer, D.; Jung, S.; Johnson, L.A. Pilot-plant proof-of-concept for integrated, countercurrent, two-stage, enzyme-assisted aqueous extraction of soybeans. J. Am. Oil Chem. Soc. 2011, 88, 1649–1658. [Google Scholar] [CrossRef]
- Jung, S.; Maurer, D.; Johnson, L. Factors affecting emulsion stability and quality of oil recovered from enzyme-assisted aqueous extraction of soybeans. Bioresour. Technol. 2009, 100, 5340–5347. [Google Scholar] [CrossRef] [PubMed]
- USDA/ERS. Fuel Ethanol Supply and Disappearance Calendar Year. 2018. Available online: https://www.ers.usda.gov/data-products/us-bioenergy-statistics/us-bioenergy-statistics/ (accessed on 1 May 2018).
- RFA. U.S. Ethanol Exports and Imports. 2018. Available online: http://www.ethanolrfa.org/wp-content/uploads/2018/02/2017-U.S.-Ethanol-Trade-Statistics-Summary_CORRECTED2.pdf (accessed on 27 April 2018).
- Sekhon, J.K.; Jung, S.; Wang, T.; Rosentrater, K.A.; Johnson, L.A. Effect of co-products of enzyme-assisted aqueous extraction of soybeans on ethanol production in dry-grind corn fermentation. Bioresour. Technol. 2015, 192, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Karki, B.; Maurer, D.; Box, S.; Kim, T.H.; Jung, S. Ethanol production from soybean fiber, a co-product of aqueous oil extraction, using a soaking in aqueous ammonia treatment. J. Am. Oil Chem. Soc. 2012, 89, 1345–1353. [Google Scholar]
- Karki, B.; Maurer, D.; Jung, S. Efficiency of treatments for optimal enzymatic saccharification of soybean fiber. Bioresour. Technol. 2011, 102, 6522–6528. [Google Scholar] [CrossRef] [PubMed]
- Karki, B.; Maurer, D.; Kim, T.H.; Jung, S. Comparison and optimization of enzymatic saccharification of soybean fibers recovered from aqueous extractions. Bioresour. Technol. 2011, 102, 1228–1233. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, K.; Sanchez, E.; El-Halwagi, M.; Kafarov, V. Energy analysis and process integration of bioethanol production from acid pre-treated biomass: Comparison of SHF, SSF and SSCF pathways. Chem. Eng. J. 2011, 176, 195–201. [Google Scholar] [CrossRef]
- Cardona, C.A.; Sanchez, O.J. Fuel ethanol production: Process design trends and integration opportunities. Bioresour. Technol. 2007, 98, 2415–2457. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, I.; Negro, M.J.; Oliva, J.M.; Cabañas, A.; Manzanares, P.; Ballesteros, M. Ethanol production from steam-explosion pretreated wheat straw. In Twenty-Seventh Symposium on Biotechnology for Fuels and Chemicals; Humana Press: New York, NY, USA, 2006; pp. 496–508. [Google Scholar]
- Teixeira, L.C.; Linden, J.C.; Schroeder, H.A. Optimizing peracetic acid treatment conditions for improved simultaneous saccharification and co-fermentation (SSCF) of sugar cane bagasse to ethanol fuel. Renew. Energy 1999, 16, 1070–1073. [Google Scholar] [CrossRef]
- Bokinsky, G.; Peralta-Yahya, P.P.; George, A.; Holmes, B.M.; Steen, E.J.; Dietrich, J.; Lee, T.S.; Tullman-Ercek, D.; Voigt, C.A.; Simmons, B.A.; et al. Synthesis of three advanced biofuels from ionic liquid-pretreated switchgrass using engineered Escherichia coli. Proc. Natl. Acad. Sci. USA 2011, 108, 19949–19954. [Google Scholar] [CrossRef] [PubMed]
- Geddes, C.C.; Mullinnix, M.T.; Nieves, I.U.; Peterson, J.J.; Hoffman, R.W.; York, S.W.; Yomano, L.P.; Miller, E.N.; Shanmugam, K.T.; Ingram, L.O. Simplified process for ethanol production from sugarcane bagasse using hydrolysate-resistant Escherichia coli strain MM160. Bioresour. Technol. 2011, 102, 2702–2711. [Google Scholar] [CrossRef] [PubMed]
- Lau, M.W.; Gunawan, C.; Balan, V.; Dale, B.E. Comparing the fermentation performance of Escherichia coli KO11, Saccharomyces cerevisiae 424A (LNH-ST) and Zymomonas mobilis AX101 for cellulosic ethanol production. Biotechnol. Biofuels 2010, 3, 11. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, A.T.W.M.; Zeeman, G. Treatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, I.; Quintero, R. Recent advancements in treatment technologies of biomass to produce bioenergy. In Bioenergy Research: Advances and Application; Gupta, V.K., Kubicek, C.P., Saddler, J., Xu, F., Tuohy, M.G., Eds.; Elsevier: Oxford, UK, 2014; pp. 57–69. [Google Scholar]
- Yoo, J.; Alavi, S.; Vadlani, P.; Behnke, K.C. Soybean hulls pretreated using thermo-mechanical extrusion-hydrolysis efficiency, fermentation inhibitors, and ethanol yield. Appl. Biochem. Biotechnol. 2012, 166, 576–589. [Google Scholar] [CrossRef] [PubMed]
- Tomas-Pejo, E.; Alvira, P.; Ballesteros, M.; Negro, M.J. Treatment technologies for lignocellulose to bioethanol conversion. In Biofuels: Alternative Feedstocks and Conversion Processes; Pandey, A., Larroche, C., Ricke, S., Dussap, C.G., Gnansounou, E., Eds.; Academic Press: St. Louis, MO, USA, 2011; pp. 149–174. [Google Scholar]
- Wan, C.; Zhou, Y.; Li, Y. Liquid hot water and alkaline treatment of soybean straw for improving cellulose digestibility. Bioresour. Technol. 2011, 102, 6254–6259. [Google Scholar] [CrossRef] [PubMed]
- Mosier, N.; Wyman, C.; Dale, B.; Elander, R.; Lee, Y.Y.; Holtzapple, M.; Ladisch, M. Features of promising technologies for treatment of lignocellulosic biomass. Bioresour. Technol. 2005, 96, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Wyman, C.E.; Dale, B.E.; Elander, R.T.; Holtzapple, M.; Ladisch, M.R.; Lee, Y. Comparative sugar recovery data from laboratory scale application of leading treatment technologies to corn stover. Bioresour. Technol. 2005, 96, 2026–2032. [Google Scholar] [CrossRef] [PubMed]
- Wyman, C.E.; Dale, B.E.; Elander, R.T.; Holtzapple, M.; Ladisch, M.R.; Lee, Y.Y. Coordinated development of leading biomass treatment technologies. Bioresour. Technol. 2005, 96, 1959–1966. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, T.; Johnson, L.A. Effect of low-shear extrusion on corn fermentation and oil partition. J. Agric. Food Chem. 2009, 57, 2302–2307. [Google Scholar] [CrossRef] [PubMed]
- Majoni, S.; Wang, T. Characterization of oil precipitate and oil extracted from condensed corn distillers solubles. J. Am. Oil Chem. Soc. 2010, 87, 205–213. [Google Scholar] [CrossRef]
- Majoni, S.; Wang, T.; Johnson, L.A. Enzyme treatments to enhance oil recovery from condensed corn distillers solubles. J. Am. Oil Chem. Soc. 2011, 88, 523–532. [Google Scholar] [CrossRef]
- Wang, H.; Wang, T.; Johnson, L.A.; Pometto, A.L., III. Effect of the corn breaking method on oil distribution between stillage phases of dry-grind corn ethanol production. J. Agric. Food Chem. 2008, 56, 9975–9980. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ouhida, I.; Pérez, J.F.; Gasa, J. Soybean (Glycine max) cell wall composition and availability to feed enzymes. J. Agric. Food Chem. 2002, 50, 1933–1938. [Google Scholar] [CrossRef] [PubMed]
- Beldman, G.; Rombouts, F.; Voragen, A.; Pilnik, W. Application of cellulase and pectinase from fungal origin for the liquefaction and saccharification of biomass. Enzym. Microb. Technol. 1984, 6, 503–507. [Google Scholar] [CrossRef]
- Marsman, G.J.P.; Gruppen, H.; Mul, A.J.; Voragen, A.G.J. In vitro accessibility of untreated, toasted, and extruded soybean meals for proteases and carbohydrases. J. Agric. Food Chem. 1997, 45, 4088–4095. [Google Scholar] [CrossRef]
- Yomano, L.P.; York, S.W.; Ingram, L.O. Isolation and characterization of ethanol-tolerant mutants of Escherichia coli KO11 for fuel ethanol production. J. Ind. Microbiol. Biotechnol. 1998, 20, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Taylor, F.; Marquez, M.A.; Johnston, D.B.; Goldberg, N.M.; Hicks, K.B. Continuous high-solids corn liquefaction and fermentation with stripping of ethanol. Bioresour. Technol. 2010, 101, 4403–4408. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Balan, V.; Gunawan, C.; Dale, B.E. Quantitatively understanding reduced xylose fermentation performance in AFEX TM treated corn stover hydrolysate using Saccharomyces cerevisiae 424A (LNH-ST) and Escherichia coli KO11. Bioresour. Technol. 2012, 111, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Majoni, S.; Wang, T.; Johnson, L.A. Physical and chemical processes to enhance oil recovery from condensed corn distillers solubles. J. Am. Oil Chem. Soc. 2011, 88, 425–434. [Google Scholar] [CrossRef]
- Johnston, D.; Hicks, K.B.; Moreau, R.A.; Shetty, J.K. Methods for Obtaining Oil from Maize Using Acid Protease and Cell-Wall Polysaccharide-Degrading Enzymes. U.S. Patent Application No. 20,140,134,684, 15 May 2014. [Google Scholar]
- Luangthongkam, P.; Fang, L.; Noomhorm, A.; Lamsal, B. Addition of cellulolytic enzymes and phytase for improving ethanol fermentation performance and oil recovery in corn dry grind process. Ind. Crop. Prod. 2015, 77, 803–808. [Google Scholar] [CrossRef][Green Version]
- Fang, L.; Wang, T.; Lamsal, B. Use of surfactant and enzymes in dry-grind corn ethanol fermentation improves yield of ethanol and distillers corn oil. Ind. Crop. Prod. 2018, 111, 329–335. [Google Scholar] [CrossRef]
- Jung, S.; de Moura, J.M.L.N.; Campbell, K.A.; Johnson, L.A. Enzyme-assisted aqueous extraction of oilseeds. In Enhancing Extraction Processes in the Food Industry; Lebovka, N., Vorobiev, E., Chemat, F., Eds.; CRC Press: Boca Raton, FL, USA, 2011; pp. 477–518. [Google Scholar]
- Bon, E.P.; Ferrara, M.A. Bioethanol Production via Enzymatic Hydrolysis of Cellulosic biomass. In Proceedings of the FAO the Role of Agricultural Biotechnologies for Production of Bioenergy in Developing Countries, Rome, Italy, 12 October 2007. [Google Scholar]
- Talebnia, F.; Karakashev, D.; Angelidaki, I. Production of bioethanol from wheat straw: An overview on treatment, hydrolysis and fermentation. Bioresour. Technol. 2010, 101, 4744–4753. [Google Scholar] [CrossRef] [PubMed]

| Treatment | Total Solids (% db) | Glucan (% db) | Xylan (% db) | Total Lignin (% db) | Glucan Loading (% db) | Ethanol Concentration (g/L) |
|---|---|---|---|---|---|---|
| Control | 14.4 ± 0.6 a | 26.9 ± 6.6 b | 23.2 ± 13.9 a | 4.7 ± 0.4 a | 1% | 2.3 ± 0.1 b |
| SAA | 5.1 ± 1.3 b | 45.8 ± 3.9 a | 18.7 ± 6.2 a | 6.0 ± 2.8 a | 1% | 5.2 ± 0.2 ab |
| LHW | 14.8 ± 1.1 a | 28.4 ± 6.4 b | 27.0 ± 14.8 a | 7.3 ± 1.7 a | 1% | 3.8 ± 0.3 b |
| Enzyme cocktail (PCX) | nd | nd | nd | nd | 2% | 11.8 ± 2.3 a |
| % | IF | SSCF-10 L | SSCF-60 L | RIF | [corn + IF] * |
|---|---|---|---|---|---|
| Solids | 14.4 ± 0.30 a | 8.4 ± 0.10 e | 9.3 ± 0.11 d | 13.6± 0.10 b | 11.2 ± 0.20 c |
| Protein | 11.5 ± 0.50 d | 16.5 ± 0.00 b | 15.3 ± 0.60 c | 16.5 ± 0.20 b | 26.5 ± 0.40 a |
| Oil | 2.5 ± 1.70 c | 6.5 ± 0.40 b | 2.8 ± 0.50 c | 6.5 ± 0.10 b | 12.0 ± 0.10 a |
| Glucan | 27.1 ± 2.70 a | 21.7 ± 0.40 b | 12.4 ± 0.50 d | 21.7 ± 0.40 b | 16.0 ± 0.70 c |
| Galactan | 31.5 ± 7.80 a | 22.3 ± 0.70 b | 16.8 ± 0.20 b | 22.3 ± 0.07 b | 9.6 ± 1.70 c |
| Arabinan | 15.2 ± 1.00 a | na | 7.5 ± 0.20 b | nd | 8.2 ± 0.20 b |
| Xylan | 13.0 ± 2.20 b | na | 8.3 ± 0.00 c | nd | 29.4 ± 0.20 a |
| Lignin | 7.5 ± 2.00 c | 13.8 ± 0.60 a | 9.1 ± 0.20 c | 13.8 ± 0.30 a | 11.8 ± 0.40 b |
| Ash | 4.0 ± 1.00 b | nd | nd | 6.6 ± 0.20 a | 4.5 ± 0.20 b |
| Ethanol Added (g/L) | Time 0 h | Time 24 h | Ethanol Produced (g/L) | ||
|---|---|---|---|---|---|
| Xylose (g/L) | Ethanol (g/L) | Xylose (g/L) | Ethanol (g/L) | ||
| 50 g/L * | |||||
| 0 (no E. coli) | 52 | 0 | 57 | 0 | 0 |
| 0 | 52 | 0 | 21 | 2 | 2 |
| 20 | 50 | 22 | 10 | 30 | 11 |
| 50 | 49 | 46 | 51 | 39 | 0 |
| 100 | 46 | 87 | 49 | 74 | 0 |
| 150 g/L * | |||||
| 0 (no E. coli) | 87 | 0 | 98 | 0 | 0 |
| 0 | 86 | 0 | 87 | 2 | 2 |
| 20 | 86 | 20 | 89 | 21 | 3 |
| 50 | 84 | 49 | 93 | 42 | 0 |
| 100 | 82 | 88 | 91 | 78 | 0 |
| Sugar | Ethanol Conc. in LB Broth (g/L) | Vmax | Km | R2 | Xylose (g/L) | Ethanol Conc. in LB Broth (g/L) | Vmax | Km | R2 |
|---|---|---|---|---|---|---|---|---|---|
| Glucose | 0 | 2.8 | 2.1 | 0.9962 | 50 | 0 | 2.5 | 2.0 | 0.9842 |
| 10 | 2.8 | 2.5 | 0.9989 | 20 | 2.6 | 5.0 | 0.9842 | ||
| 20 | 3.1 | 3.1 | 0.9901 | 50 | 0.2 | 1.1 | 0.9945 | ||
| 100 | 0.1 | −0.9 | 0.9943 | ||||||
| Xylose | 0 | 2.7 | 1.9 | 0.9951 | 150 | 0 | 2.9 | 7.4 | 0.8826 |
| 10 | 2.9 | 2.9 | 0.9968 | 20 | 3.4 | 19.9 | 0.8826 | ||
| 20 | 2.9 | 2.9 | 0.9961 | 50 | 0.2 | 0.6 | 0.8954 | ||
| 100 | 0.1 | −1.0 | 0.9992 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sekhon, J.K.; Maurer, D.; Wang, T.; Jung, S.; Rosentrater, K.A. Ethanol Production by Soy Fiber Treatment and Simultaneous Saccharification and Co-Fermentation in an Integrated Corn-Soy Biorefinery. Fermentation 2018, 4, 35. https://doi.org/10.3390/fermentation4020035
Sekhon JK, Maurer D, Wang T, Jung S, Rosentrater KA. Ethanol Production by Soy Fiber Treatment and Simultaneous Saccharification and Co-Fermentation in an Integrated Corn-Soy Biorefinery. Fermentation. 2018; 4(2):35. https://doi.org/10.3390/fermentation4020035
Chicago/Turabian StyleSekhon, Jasreen K., Devin Maurer, Tong Wang, Stephanie Jung, and Kurt A. Rosentrater. 2018. "Ethanol Production by Soy Fiber Treatment and Simultaneous Saccharification and Co-Fermentation in an Integrated Corn-Soy Biorefinery" Fermentation 4, no. 2: 35. https://doi.org/10.3390/fermentation4020035
APA StyleSekhon, J. K., Maurer, D., Wang, T., Jung, S., & Rosentrater, K. A. (2018). Ethanol Production by Soy Fiber Treatment and Simultaneous Saccharification and Co-Fermentation in an Integrated Corn-Soy Biorefinery. Fermentation, 4(2), 35. https://doi.org/10.3390/fermentation4020035







