Impact of Wort Amino Acids on Beer Flavour: A Review
Abstract
1. Introduction
2. Wort Amino Acid Composition
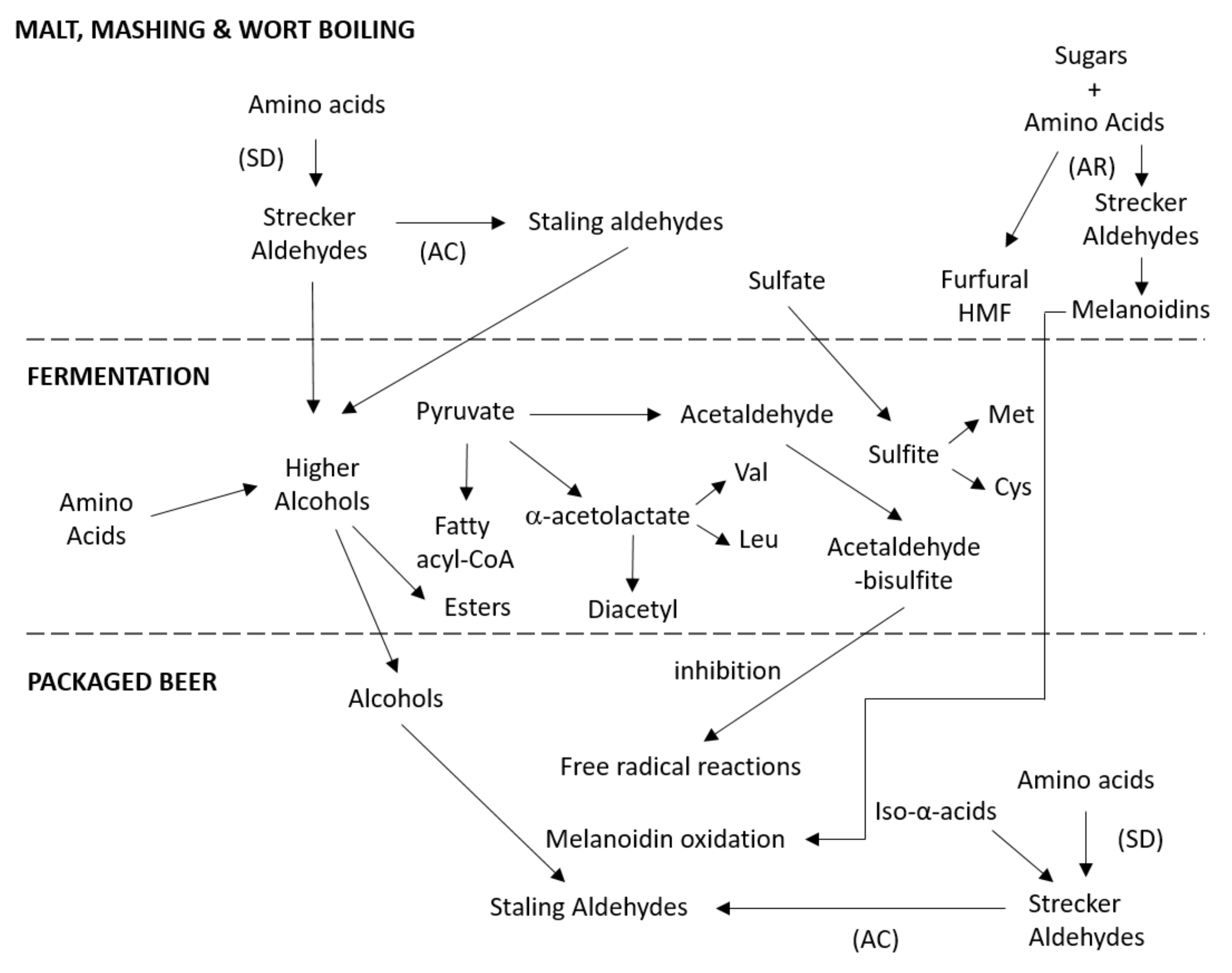
3. Amino Acid Degradation during Mashing
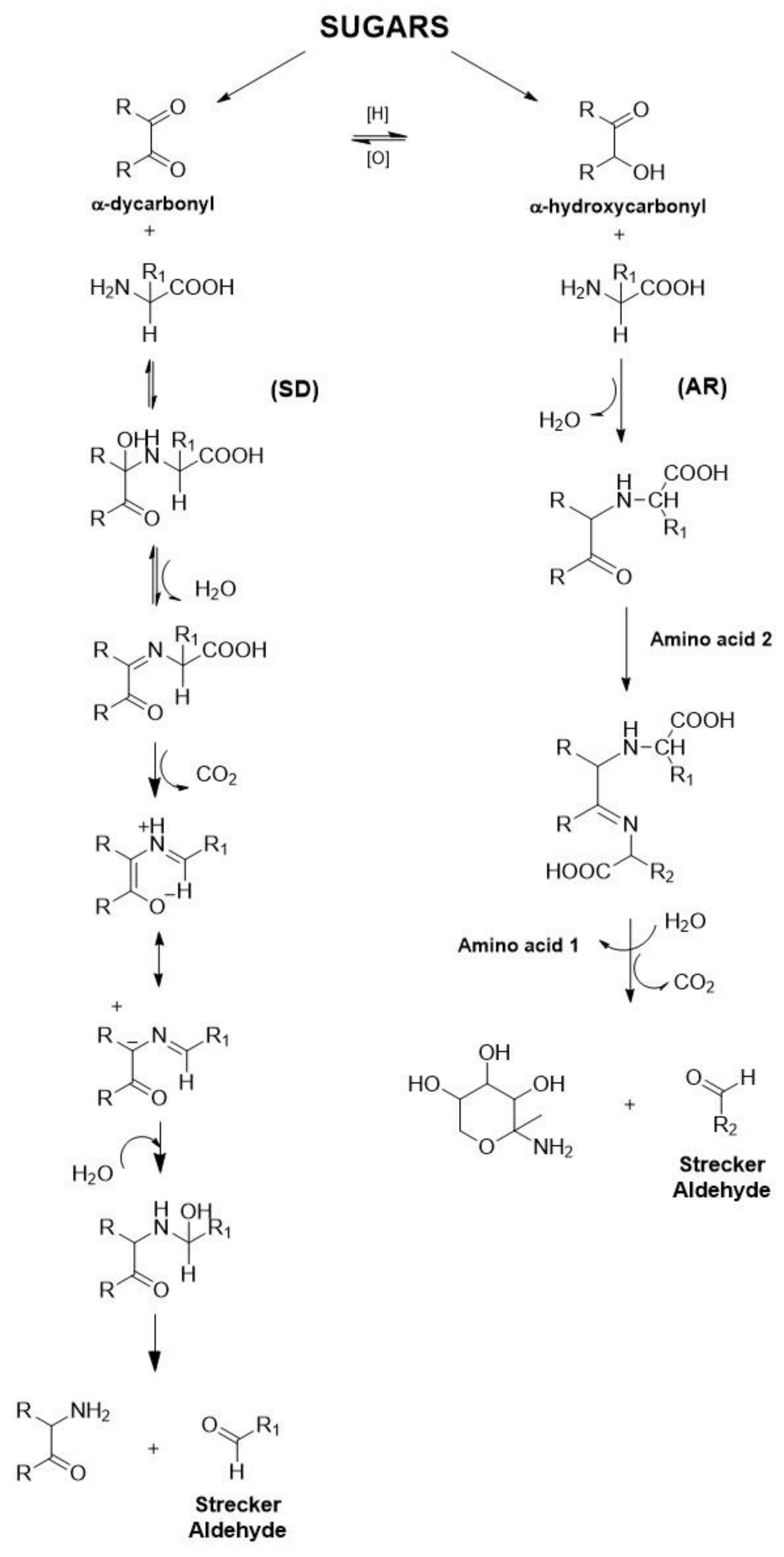
3.1. Maillard Reaction
- The first starts with a condensation between the amino group and the reducing sugar, leading to an N-glycosamine in the case of an aldose sugar that rearranges into the so called Amadori product—that is degraded to 1,2-dicarbonyl compounds in the second stage of this complex reaction. These compounds are responsible for the formation and stability of off-flavours in beer.
- The second or intermediate stage starts with the Amadori product, leading to sugar fragmentation products and the release of the amino group.
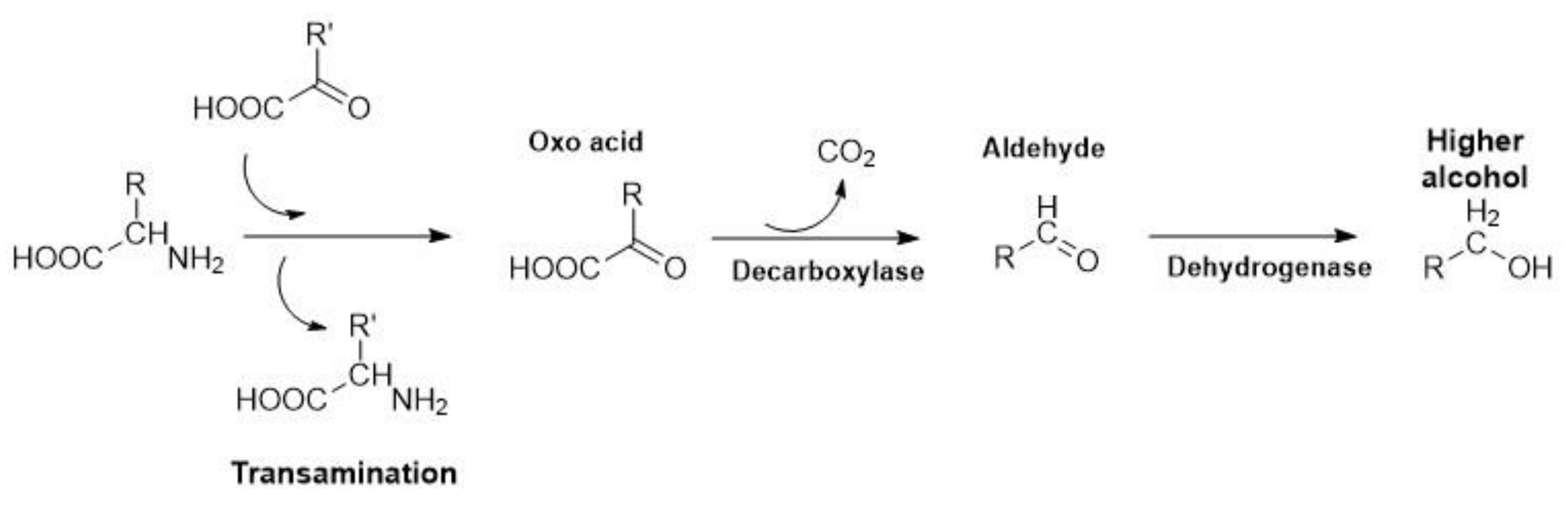
3.2. Strecker Degradation of Amino Acids
4. Amino Acids Metabolism during Fermentation
4.1. Higher Alcohols
4.2. Esters
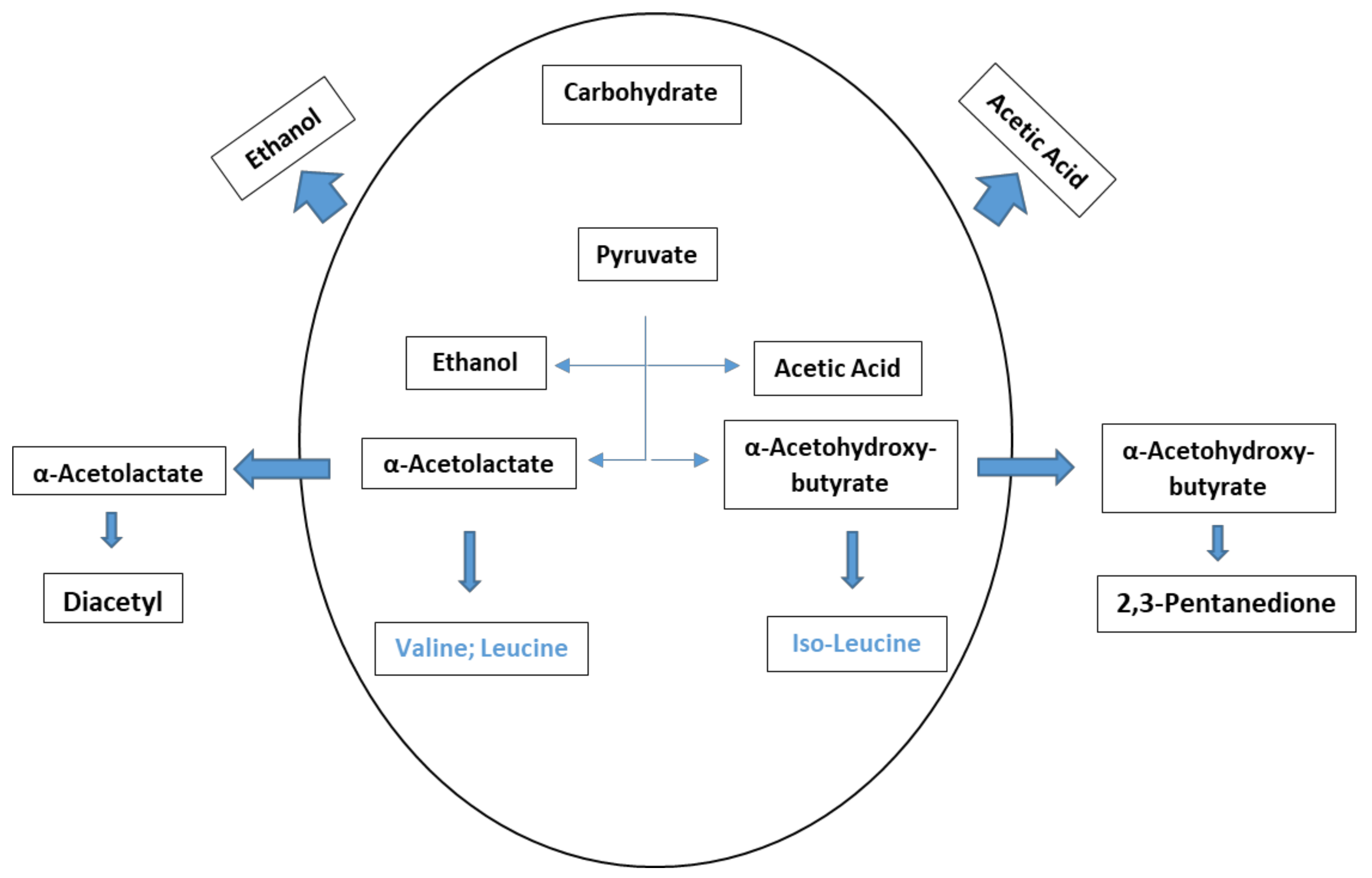
4.3. Vicinal Diketones (VDKs)
4.4. Sulfur Compounds
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lekkas, C.; Stewart, G.G.; Hill, A.; Taidi, B.; Hodgson, J. The Importance of Free Amino Nitrogen in Wort and Beer. Master Brew. Assoc. Am. Tech. Q. 2005, 42, 113–116. [Google Scholar]
- Reed, G.; Nagodawithana, T.W. Brewers Yeast. In Yeast Technology; Avi Publishing: New York, NY, USA, 1991; pp. 89–105. [Google Scholar]
- Hough, J.S.; Briggs, D.E.; Stevens, R.; Young, T.W. Malting and Brewing Science—Hopped Wort and Beer, 2nd ed.; Chapman & Hall: London, UK, 1982; Volume 2. [Google Scholar]
- Garza-Ulloa, H.; Cantú, R.H.; Gajá, A.M.C. Determination of Amino Acids in Wort and Beer by Reverse-Phase High-Performance Liquid Chromatography. Am. Soc. Brew. Chem. 1985, 44, 47–51. [Google Scholar]
- Hough, J.S.; Briggs, D.E.; Stevens, R.; Young, T.W. Metabolism of wort by yeast. In Malting and Brewing Science: Hopped Wort and Beer; Chapman & Hall: London, UK, 1982; pp. 566–612. [Google Scholar]
- He, Y.; Dong, J.; Yin, H.; Zhao, Y.; Chen, R.; Wan, X.; Chen, P.; Hou, X.; Liu, J.; Chen, L. Wort composition and its impact on theflavour-active higher alcohol and esterformation of beer—A review. J. Inst. Brew. 2014, 120, 157–163. [Google Scholar] [CrossRef]
- Dack, R.E.; Black, G.W.; Koutsidis, G.; Usher, S.J. The effect of Maillard reaction products and yeast strain on the synthesis of key higher alcohols and esters in beer fermentations. Food Chem. 2017, 232, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Hellwig, M.; Witte, S.; Henle, T. Free and Protein-Bound Maillard Reaction Products in Beer: Method Development and a Survey of Different Beer Types. J. Agric. Food Chem. 2016, 64, 7234–7243. [Google Scholar] [CrossRef] [PubMed]
- Vanderhaegen, B.; Neven, H.; Verachtert, H.; Derdelinckx, G. The chemistry of beer aging—A critical review. Food Chem. 2006, 9, 357–381. [Google Scholar] [CrossRef]
- Van Boekel, M.A. Formation of flavour compounds in the Maillard reaction. Biotechnol. Adv. 2006, 24, 230–233. [Google Scholar] [CrossRef] [PubMed]
- Oliver, G. The Oxford Companion to Beer; Oxford University Press: New, York, NY, USA, 2012. [Google Scholar]
- Briggs, D.E.; Boulton, C.A.; Brookes, P.A. Malts, adjuncts and supplementary enzymes. In Brewing Science and Practice; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Cameiro, J.R.; Guido, L.F.; Almeida, P.J.; Rodrigues, J.A.; Barros, A.A. The impact of sulphur dioxide and oxygen on the behaviour of 2-furaldehyde in beer: An industrial approach. Int. J. Food Sci. Technol. 2006, 41, 545–552. [Google Scholar]
- De Clippeleer, J.; Van Opstaele, F.; De Cooman, L.; Aerts, G. The contribution of aldehyed to the staling taste and odour of pale large beer. In Proceedings of the 33rd International Congress of the European Brewery Convention, Glasgow, UK, 22–26 May 2011. [Google Scholar]
- De la Cueva, S.P.; Álvarez, J.; Végvári, Á.; Montilla-Gómez, J.; Cruz-López, O.; Delgado-Andrade, C.; Rufián-Henares, J.A. Relationship between HMF intake and SMF formation in vivo: An animal and human study. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Modig, T.; Liden, G.; Taherzadeh, M.J. Inhibition effects of furfural on alcohol dehydrogenase, aldehyde dehydrogenase and pyruvate dehydrogenase. Biochem. J. 2002, 363, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.L.; Slininger, P.J.; Dien, B.S.; Berhow, M.A.; Kurtzman, C.P.; Gorsich, S.W. Adaptive response of Yeasts to furfural and 5-hydroxymethylfurfural and new chemical evidence for HMF conversion to 2,5-bis-hydroxymethylfuran. J. Ind. Microbiol. Biotechnol. 2004, 31, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Palamand, S.R.; Hardwick, W.A. Studies on the relative flavour importance of some beer constituents. Master Brew. Assoc. Am. Tech. Q. 1969, 6, 117–127. [Google Scholar]
- Saison, D.; De Schutter, D.P.; Uyttenhove, B.; Delvaux, F.; Delvaux, F.R. Contribution of staling compounds to the aged flavour of lager beer by studying their flavour thresholds. Food Chem. 2009, 114, 1206–1215. [Google Scholar] [CrossRef]
- De Pinho, M.P.G.; Silva Ferreira, A.C. Role of Strecker aldehydes on beer flavour stability. Dev. Food Sci. 2006, 43, 529–532. [Google Scholar]
- Baert, J.J.; De Clippeleer, J.; Hughes, P.S.; De Cooman, L.; Aerts, G. On the origin of free and bound staling aldehydes in beer. J. Agric. Food Chem. 2012, 60, 11449–11472. [Google Scholar] [CrossRef] [PubMed]
- Yaylayan, V.A. Recent Advances in the Chemistry of Strecker Degradation and Amadori Rearrangement Implications to Aroma and Color Formation. Food Sci. Technol. Res. 2003, 9, 1–6. [Google Scholar] [CrossRef]
- Rizzi, G.P. The Strecker Degradation of Amino Acids: Newer Avenues for Flavor Formation. Food Rev. Int. 2008, 24, 416–435. [Google Scholar] [CrossRef]
- Thum, B.; Miedaner, H.; Narziss, L.; Back, W. Bildung von “Alterungscarbonylen”—Mogliche Mechanismen un Bedeutung bei der Bierlagerung. In Proceedings of the 25th European Brewery Convention Congress, Brussels, Belgium, 14–18 May 1995; pp. 491–498. [Google Scholar]
- Heggart, H.M.; Margaritis, A.; Pilkington, H.; Stewart, R.J.; Dowhanick, T.M.; Russell, I. Factores Affecting Yeast Viability and Vitality Characteristics: A Review. Master Brew. Assoc. Am. Tech. Q. 1999, 36, 383–406. [Google Scholar]
- Hammond, J.R.M. Brewer’s Yeasts. In The Yeasts; Academic Press: London, UK, 1993; pp. 7–67. [Google Scholar]
- Hiralal, L.; Olaniran, A.O.; Pillay, B. Aroma-active ester profile of ale beer produced under different fermentation and nutritional conditions. J. Biosci. Bioeng. 2014, 117, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.; Kopecká, J.; Meier-Dörnberg, T.; Zarnkow, M.; Jacob, F.; Hutzler, M. Screening for new brewing yeasts in the non-Saccharomyces sector with Torulaspora delbrueckii as model. Yeast 2016, 33, 129–144. [Google Scholar] [CrossRef] [PubMed]
- Boekhout, T.; Robert, V. Yeasts in Food—Beneficial and Detrimental Aspects; Woodhead Publishing: Sawston, UK, 2003; pp. 363–370. [Google Scholar]
- Jones, M.; Pierce, J.S. Absorption of Amino Acids from Worst by Yeasts. J. Inst. Brew. 1964, 70, 307–315. [Google Scholar] [CrossRef]
- Enari, T.-M. Amino acids, peptides and proteins. European Brewery Convention. Wort Symposium Zeit, Monograph 1. 1974; 73–88. [Google Scholar]
- Meier-Dörnberg, T.; Hutzler, M.; Michel, M.; Methner, F.-J.; Jacob, F. The Importance of a Comparative Characterization of Saccharomyces Cerevisiae and Saccharomyces Pastorianus Strains for Brewing. Fermentation 2017, 3, 41. [Google Scholar] [CrossRef]
- Dufour, J.P.; Malcorps, P. Ester synthesis during fermentation: Enzymes characterization and modulation mechanism. In Proceedings of the 4th Aviemore Conference on Malting, Brewing and Distilling, Aviemore, UK, 23–26 May 1994; Campbell, I., Priest, F.G., Eds.; The Institute of Brewing: London, UK, 1994; pp. 137–151. [Google Scholar]
- Engan, S. Beer composition: Volatile substances. In Brewing Science; Pollock, J.R.A., Ed.; Academic Press: London, UK, 1981; pp. 93–165. [Google Scholar]
- Meilgaard, M.C. Flavour chemistry of beer. Part II: Flavour and threshold of 239 aroma volatiles. Master Brew. Assoc. Am. Tech. Q. 1975, 28, 132–141. [Google Scholar]
- Engan, S. Organoleptic threshold values of some alcohols and esters inbeer. J. Inst. Brew. 1972, 78, 33–36. [Google Scholar] [CrossRef]
- Reed, G. Brewer’s Yeast—Biochemistry of Brewing. In Yeast Technology; Nagodawithana, T.W., Ed.; Avi Publishing: New York, NY, USA, 1991; pp. 130–131. [Google Scholar]
- Renger, R.S.; van Hateren, S.H.; Luyben, K.C.A.M. The formation of esters and higher alcohols during brewery fermentation. The effect of carbon dioxide pressure. J. Inst. Brew. 1992, 98, 509–513. [Google Scholar] [CrossRef]
- Olaniran, A.O.; Hiralal, L.; Mokoena, M.P.; Pillay, B. Flavour-active volatile compounds in beer: Production, regulation and control. J. Inst. Brew. 2017, 123, 13–23. [Google Scholar] [CrossRef]
- Hughes, P.S.; Baxter, E.D. Flavour Determinants of Beer Quality. In Beer: Quality, Safety and Nutritional Aspects; RSC Publishing: Cambridge, UK, 2001; pp. 48–64. [Google Scholar]
- Willaert, R.; Nedovic, V.A. Primary beer fermentation by immobilised yeast—A review on flavour formation and control strategies. J. Chem. Technol. Biotechnol. 2006, 81, 1353–1367. [Google Scholar] [CrossRef]
- Ehrlich, F. Uber die Bedingungen der Fuselolbildung und uber ihren Zusammenhang mit dem Eiweissaufbau der Hefe. Ber. Dtch. Chem. Ges. 1907, 40, 1027–1047. [Google Scholar] [CrossRef]
- Chen, E.C.H. The Relative Contribution of Ehrlich and Biosynthetic Pathways to the Formation of Fusel Alcohols. J. Am. Soc. Brew. Chem. 1978, 36, 39. [Google Scholar] [CrossRef]
- Lei, H.; Li, H.; Mo, F.; Zheng, L.; Zhao, H.; Zhao, M. Effects of Lys and His supplementations on the regulation of nitrogen metabolism in lager yeast. Appl. Microbiol. Biotechnol. 2013, 97, 8913–8921. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, J.R.; Salgado, L.E.; Hewlins, M.J. The catabolism of amino acids to long chain and complex alcohols in Saccharomyces cerevisiae. J. Biol. Chem. 2003, 278, 8028–8034. [Google Scholar] [CrossRef] [PubMed]
- Avalos, J.L.; Fink, G.R.; Stephanopoulos, G. Compartmentalization of metabolic pathways in yeast mitochondria improves the production of branched-chain alcohols. Nat. Biotechnol. 2013, 31, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Guido, L.; Rajendram, R.; Barros, A.A. Pitching Yeast and Beer Flavour. In Beer in Health and Disease Prevention; Preedy, V.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 23–32. [Google Scholar]
- Pires, E.J.; Teixeira, J.A.; Brányik, T.; Vicente, A.A. Yeast: The soul of beer’s aroma—A review of flavour-active esters and higher alcohols produced by the brewing yeast. Appl. Microbiol. Biotechnol. 2014, 98, 1937–1949. [Google Scholar] [CrossRef] [PubMed]
- Engan, S. Wort composition and beer flavour. I. The influence of some amino acids on the formation of higher aliphatic alcohols and esters. J. Inst. Brew. 1969, 76, 254–261. [Google Scholar] [CrossRef]
- Pavsier, A.; Bulatti, S. Lager Beer. In Beer in Health and Disease Prevention; Preedy, V., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; p. 35. [Google Scholar]
- James, N.; Stahl, U. Amino Acid Permeases and their Influence on Flavour Compounds in Beer. Brew. Sci. 2014, 67, 120–127. [Google Scholar]
- Donalies, U.E.; Stahl, U. Increasing sulphite formation in Saccharomyces cerevisiae by overexpression of MET14 and SSU1. Yeast 2002, 1, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Guido, L.F. Sulfites in beer: Reviewing regulation, analysis and role. Sci. Agric. 2016, 73, 189–197. [Google Scholar] [CrossRef]
- Duan, W.; Roddick, F.A.; Higgins, V.J.; Rogers, P.J. A parallel analysis of H2S and SO2 formation by brewing yeast in response to sulfur-containing amino acids and ammonium ions. J. Am. Soc. Brew. Chem. 2004, 62, 35–41. [Google Scholar]
| Nitrogen and Amino Acids | Concentration in Wort (mg.100 cm−3) |
|---|---|
| Total nitrogen | 88.0 a |
| Low molecular nitrogen alcohol-soluble | 63.4 a |
| Total α-amino nitrogen | 42.7 a |
| Alcohol-soluble α-amino nitrogen | 37.6 a |
| Alanine | 9.8 a; 6.5 b |
| ϒ-amino-butyric-acid | 8.3 a; 11.15 b |
| Arginine | 13.8 a; 6.12 b |
| Aspartic acid | 7.0 a; 4.99 b |
| Glutamic acid | 6.4 a; 3.87 b |
| Glycine | 2.3 a; 2.82 b |
| Histidine | 5.7 a; 2.06 b |
| Isoleucine | 6.2 a; 6.4 b |
| Leucine | 18.1 a; 12.25 b |
| Lysine | 14.9 a; 10.78 b |
| Phenylalanine | 13.7 a; 9.21 b |
| Proline | 45.7 a |
| Threonine | 5.9 a; 5.21 b |
| Tyrosine | 10.6 a; 6.21 b |
| Valine | 11.9 a; 9.51 b |
| Serine + Asparagine (mg in 100 cm3) | 168.6 a |
| Ammonia | 2.4 a |
| Compound | Threshold (mg L−1) | Aroma Impression |
|---|---|---|
| Maillard Reaction Products | ||
| Furfural | 25,000–50,000 a; 15157 b | Bitter, winey |
| 5-Hydroxymethy lfurfural (HMF) | 35784 b | Cardboard, papery, cucumber |
| Compound | Threshold (mg L−1) | Aroma Impression |
|---|---|---|
| Strecker Degradation Products | ||
| 2-methylpropanal | 65 a; 86 b | Grainy, varnish, fruity |
| 2-methylbutanal | 35 a; 45 b | Almond, apple-like, malty |
| 3-methylbutanal | 46 a; 56 b | Malty, chocolate, cherry, almond |
| Methional | 4.2 b | Cooked potatoes, worty |
| Phenylacetaldehyde | 100 a; 105 b | Hyacinth, flowery, roses |
| Benzaldehyde | 515 b | Almond, cherry, stone |
| Groups | Jones and Pierce (1964) [30] | Enari et al. (1970s) [31] |
|---|---|---|
| Fast Absorption Group A | Asparagine | Asparagine |
| Serine | Serine | |
| Threonine | Threonine | |
| Lysine | Lysine | |
| Arginine | ||
| Glutamic Acid | ||
| Aspartic Acid | ||
| Glutamine | ||
| Intermediate Absorption Group B | Valine | Arginine |
| Methionine | Aspartic Acid | |
| Leucine | Glutamic Acid | |
| Isoleucine | Valine | |
| Histidine | Methionine | |
| Leucine | ||
| Isoleucine | ||
| Slow Absorption Group C | Glycine | Histidine |
| Phenylalanine | Glycine | |
| Tyrosine | Phenylalanine | |
| Tryptophan | Tyrosine | |
| Ammonia | Tryptophan | |
| Alanine | Ammonia | |
| Little or no absorption Group D | Proline | Alanine |
| Proline |
| Compound | Threshold (mg L−1) | Aroma Impression |
|---|---|---|
| Esters | ||
| Ethyl acetate | 20–30 a; 25–30 b; 30 c,g,33 f | Fruity, solvent-like |
| Isoamyl acetate | 0.6–1.2 a; 1.2–2 b; 1.2 c,g,1.6 f | Banana, pear |
| Phenylethyl acetate | 3.8 a; 0.2–3.8 b,f,g | Roses, honey, sweet |
| Ethyl hexanoate | 0.2–0.23 b,f | Apple, fruity |
| Ethyl caproate | 0.17–0.21 a; 0.21 c,g | Apple, aniseed |
| Ethyl caprylate | 0.3–0.9 a; 0.9 c,g | Apple |
| Ethyl octanoate | 0.9–1.0 b, 0.9 f | Apple, aniseed |
| Higher Alcohols | ||
| Propanol | 600 d, 700 h, 800 f,g | Alcohol, solvent-like |
| Isobutanol | 100 d; 80–100 e, 200 f,g | Alcohol, solvent-like |
| Isoamyl alcohol | 50–65 b; 50 d; 50–60 e, 70 f,g | Alcohol, banana, vinous |
| Amyl alcohol | 50–70 b; 50 d; 50–60 e, 65 f,g | Alcohol, solvent-like |
| 2-Phenylethanol | 40 b,d; 45–50 e, 125 f,g | Roses, sweet |
| Tyrosol | 200 c,100 e | Bitter, chemical |
| Sulfur Compound | Typical Levels (μg L−1) | Flavour Threshold (μg L−1) | Flavour Descriptors |
|---|---|---|---|
| Sulfite | - | 10000 a | Pungent a |
| Hydrogen sulfide | 1–20 b | 8 a,b | Sulfidic, rotten eggs |
| Sulfur dioxide | 200–20,000 b | >25,000 b | Sulfitic, burnt match |
| Carbon disulfide | 0.01–0.3 b | >50 b | - |
| Methanethiol | 0.2–15 b | 2.0 b | Putrefaction, drains |
| Ethylene sulfide | 0.3–2.0 b | >20 b | - |
| Ethanethiol | 0–20 b | 1.7 b | Putrefaction |
| Propanethiol | 0.1–0.2 b | 0.15 b | Putrefaction, rubber |
| Dimethyl sulfide | 10–100 b | 30 a,b | Sweetcorn, tin tomatoes |
| Diethyl sulfide | 0.1–1.0 b | 1.2 b | Cooked vegetables |
| Dimethyl disulfide | 0.1–3.0 b | 7.5 b | Rotten vegetables |
| Diethyl disulfide | 0–0.01 b | 0.4 b | Garlic, burnt rubber |
| Dimethyl trisulfide | 0.01–0.8 b | 0.1 b | Rotten vegetables, onion |
| Methyl thioacetate | 5–20 b | 50 b | Cabbage |
| Ethyl thioacetate | 0–2 b | 10 b | Cabbage |
| Methionol | 50–1300 b | 2000 b | Raw potatoes |
| Methional | 20–50 b | 250 b | Mash potatoes, soup-like |
| 3-methyl-2-butene-1-thiol | 0.001–0.1 b | 0.01 b | Skunk, leek-like, lightstruck |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, I.M.; Guido, L.F. Impact of Wort Amino Acids on Beer Flavour: A Review. Fermentation 2018, 4, 23. https://doi.org/10.3390/fermentation4020023
Ferreira IM, Guido LF. Impact of Wort Amino Acids on Beer Flavour: A Review. Fermentation. 2018; 4(2):23. https://doi.org/10.3390/fermentation4020023
Chicago/Turabian StyleFerreira, Inês M., and Luís F. Guido. 2018. "Impact of Wort Amino Acids on Beer Flavour: A Review" Fermentation 4, no. 2: 23. https://doi.org/10.3390/fermentation4020023
APA StyleFerreira, I. M., & Guido, L. F. (2018). Impact of Wort Amino Acids on Beer Flavour: A Review. Fermentation, 4(2), 23. https://doi.org/10.3390/fermentation4020023






