Fungal Solid-State and Submerged Fermentation of Blueberry Bagasse: Extraction Strategies, Phenolic Profiling, and Cellular Immunomodulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of Blueberry Bagasse Powder
2.3. Preparation of Fungal Inoculum
2.4. Submerged Fermentation (SmF)
2.5. Solid State Fermentation (SSF)
2.6. Extraction Procedures
2.7. Determination of Total Phenolic Content, Total Anthocyanin Content and Antioxidant Capacity
2.8. UHPLC Analysis of Individual Phenolic Compounds
2.9. In Vitro Cellular Bioactivity of Fermented Blueberry Bagasse Extracts
2.9.1. Cell Cultures, Extract Preparation, and Viability
2.9.2. Intracellular Antioxidant Activity Assay
2.9.3. Cellular Anti-Inflammatory Activity
2.9.4. Cytokine Production
2.10. Statistical Analysis
3. Results
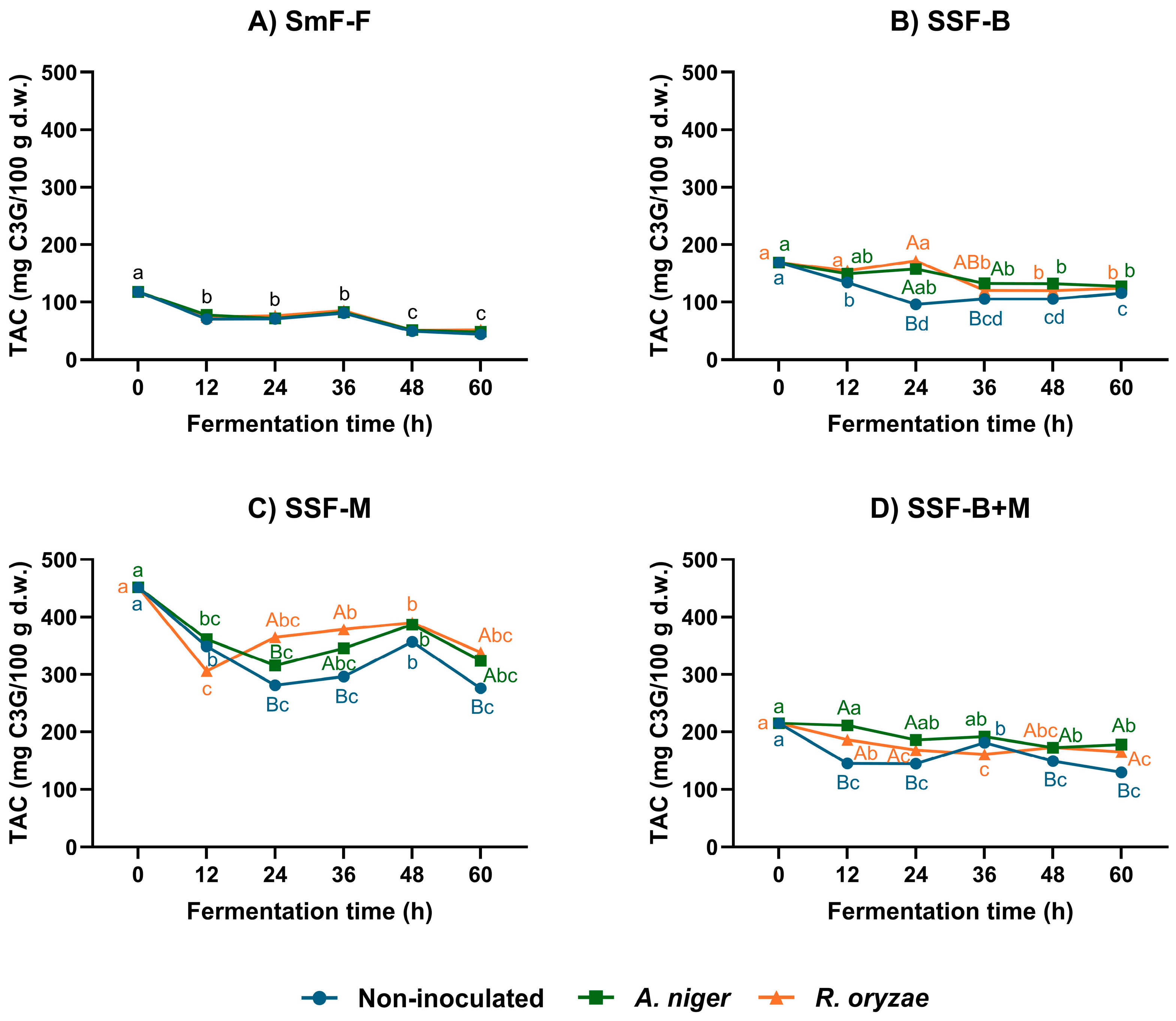
3.1. Effect of Fermentation and Extraction Strategy on Phenolic Content and Antioxidant Capacity
3.2. UHPLC Profiling of Individual Phenolic Compounds in SSF-M Extracts
3.3. Cellular Antioxidant Activity
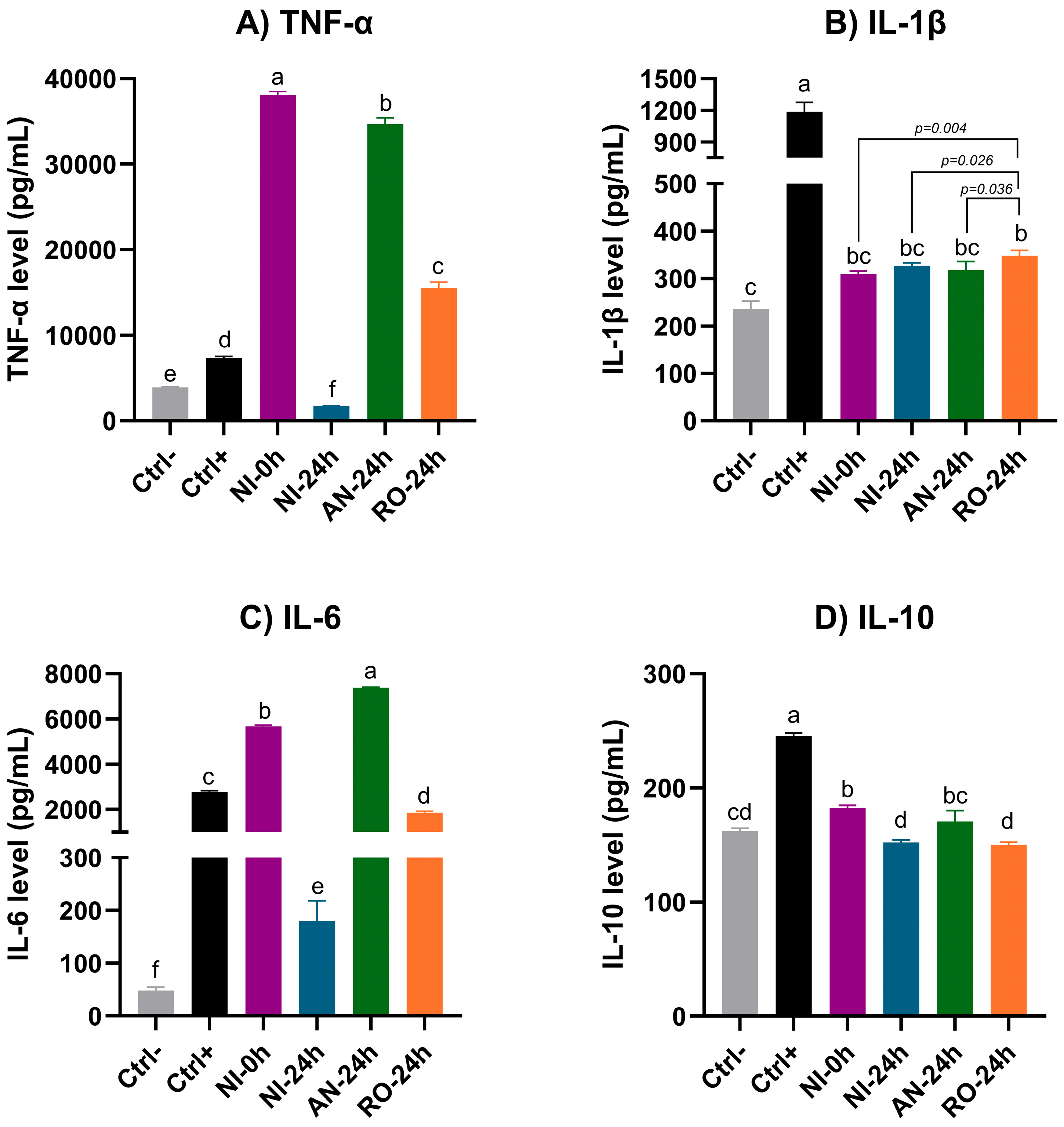
3.4. Cellular Immunomodulatory Activity: NO Production and Cytokine Profiling
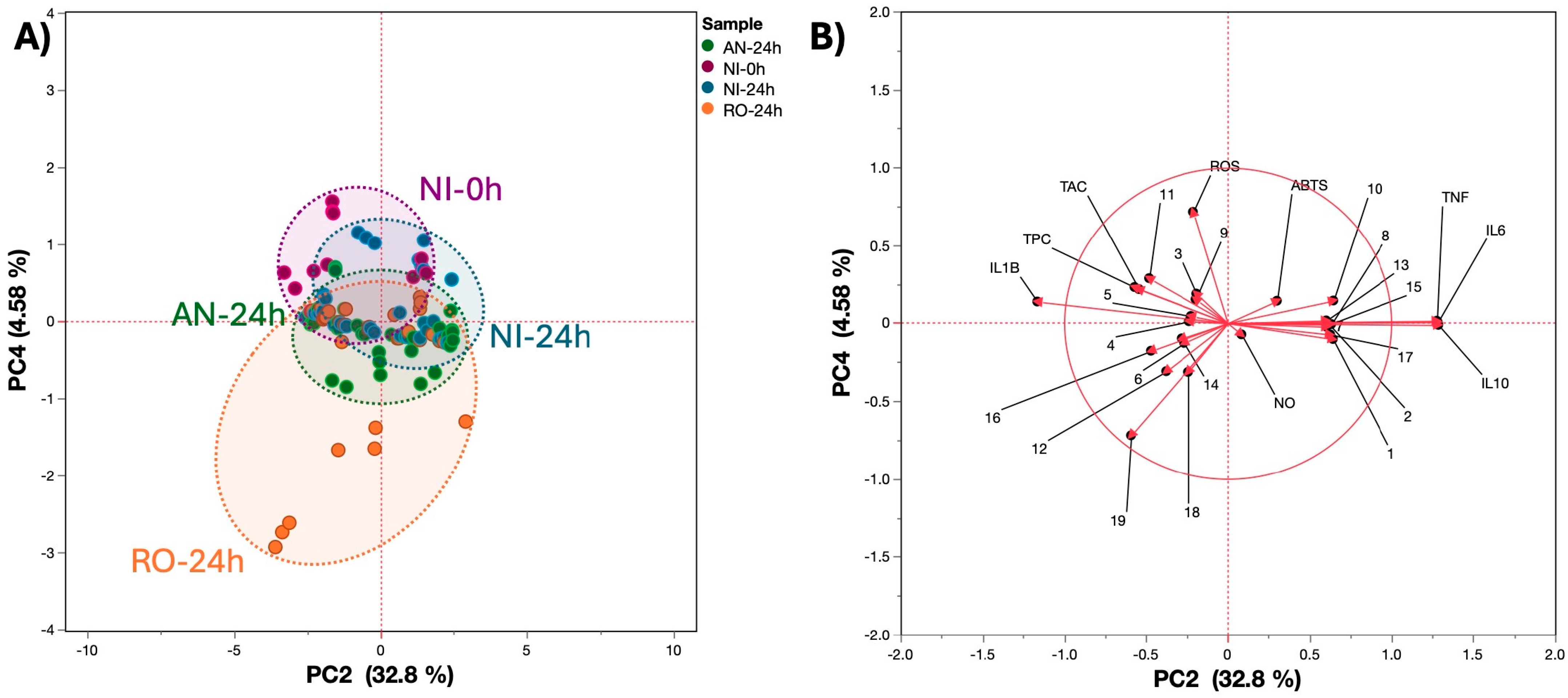
3.5. Multivariate and Correlation Analysis
4. Discussion
4.1. Fermentation–Extraction Interactions Affecting Phenolic Recovery and Antioxidant Capacity
4.2. Effects of Fungal Fermentation on the Phenolic Profile of Blueberry Bagasse
4.3. Cellular Antioxidant and Immunomodulation Properties of Fermented Blueberry Bagasse Extracts
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAPH | 2,2′-azobis(2-amidinopropane) dihydrochloride |
| ABTS | 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) |
| AN-24h | Blueberry bagasse fermented with Aspergillus niger ATCC 6275 for 24 h |
| ATCC | American Type Culture Collection |
| C3G | Cyanidin-3-glucoside equivalents |
| Ctrl+ | LPS-treated control |
| Ctrl− | Unstimulated control |
| DCFH-DA | Dichloro-dihydro-fluorescein diacetate |
| DMEM | Dulbecco Modified Eagle’s Medium |
| d.w. | Dry weight |
| ELISA | Enzyme-linked immunosorbent assay |
| FBS | Fetal bovine serum |
| GAE | Gallic acid equivalents |
| IL-1β | Interleukin-1 beta |
| IL-6 | Interleukin-6 |
| IL-10 | Interleukin-10 |
| LPS | Lipopolysaccharide |
| NI-0h | Non-inoculated blueberry bagasse at 0 h |
| NI-24h | Non-inoculated blueberry bagasse after 24 h |
| NO | Nitric oxide |
| PBS | Phosphate-buffered saline |
| PC | Principal component |
| PCA | Principal component analysis |
| Pen-Strep | Penicillin–streptomycin |
| RO-24h | Blueberry bagasse fermented with Rhizopus oryzae BIOTEC018 for 24 h |
| ROS | Reactive oxygen species |
| SmF | Submerged fermentation |
| SmF-F | Filtered extract from submerged fermentation |
| SSF | Solid-state fermentation |
| SSF-B | Buffer extract from SSF |
| SSF-B+M | Buffer–methanol extract from SSF |
| SSF-M | Methanol extract from SSF |
| TAC | Total anthocyanin content |
| TE | Trolox equivalents |
| TNF-α | Tumor necrosis factor alpha |
| TPC | Total phenolic content |
| UHPLC | Ultra-high-performance liquid chromatography |
References
- Huynh, N.K.; Wilson, M.D.; Eyles, A.; Stanley, R.A. Recent Advances in Postharvest Technologies to Extend the Shelf Life of Blueberries (Vaccinium sp.), Raspberries (Rubus idaeus L.) and Blackberries (Rubus sp.). J. Berry Res. 2019, 9, 687–707. [Google Scholar] [CrossRef]
- Horvitz, S. Postharvest Handling of Berries. In Postharvest Handling; Kahramanoglu, I., Ed.; InTech: Rijeka, Croatia, 2017; pp. 107–120. [Google Scholar]
- Stanca, L.; Bilteanu, L.; Bujor, O.C.; Ion, V.A.; Petre, A.C.; Bădulescu, L.; Geicu, O.I.; Pisoschi, A.M.; Serban, A.I.; Ghimpeteanu, O.-M. Development of Functional Foods: A Comparative Study on the Polyphenols and Anthocyanins Content in Chokeberry and Blueberry Pomace Extracts and Their Antitumor Properties. Foods 2024, 13, 2552. [Google Scholar] [CrossRef]
- Diez-Sánchez, E.; Quiles, A.; Hernando, I. Use of Berry Pomace to Design Functional Foods. Food Rev. Int. 2023, 39, 3204–3224. [Google Scholar] [CrossRef]
- Hurtado-Romero, A.; Garcia-Amezquita, L.E.; Carrillo-Nieves, D.; Montilla, A.; Villamiel, M.; Requena, T.; García-Cayuela, T. Characterization of Berry By-Products as Fermentable Substrates: Proximate and Phenolic Composition, Antimicrobial Activity, and Probiotic Growth Dynamics. LWT 2024, 204, 116468. [Google Scholar] [CrossRef]
- Fogarasi, M.; Socaciu, M.-I.; Sălăgean, C.-D.; Ranga, F.; Fărcaș, A.C.; Socaci, S.A.; Socaciu, C.; Țibulcă, D.; Fogarasi, S.; Semeniuc, C.A. Comparison of Different Extraction Solvents for Characterization of Antioxidant Potential and Polyphenolic Composition in Boletus edulis and Cantharellus cibarius Mushrooms from Romania. Molecules 2021, 26, 7508. [Google Scholar] [CrossRef] [PubMed]
- De Moraes Crizel, T.; Hermes, V.S.; De Oliveira Rios, A.; Flôres, S.H. Evaluation of Bioactive Compounds, Chemical and Technological Properties of Fruits Byproducts Powder. J. Food Sci. Technol. 2016, 53, 4067–4075. [Google Scholar] [CrossRef]
- Jiang, Y.; Subbiah, V.; Wu, H.; Bk, A.; Sharifi-Rad, J.; Suleria, H.A.R. Phenolic Profiling of Berries Waste and Determination of Their Antioxidant Potential. J. Food Qual. 2022, 2022, 5605739. [Google Scholar] [CrossRef]
- Tian, Z.-X.; Li, Y.-F.; Long, M.-X.; Liang, Q.; Chen, X.; Huang, D.-M.; Ran, Y.-Q. Effects of Six Different Microbial Strains on Polyphenol Profiles, Antioxidant Activity, and Bioaccessibility of Blueberry Pomace with Solid-State Fermentation. Front. Nutr. 2023, 10, 1282438. [Google Scholar] [CrossRef] [PubMed]
- Sarıtaş, S.; Portocarrero, A.C.M.; Miranda López, J.M.; Lombardo, M.; Koch, W.; Raposo, A.; El-Seedi, H.R.; De Brito Alves, J.L.; Esatbeyoglu, T.; Karav, S.; et al. The Impact of Fermentation on the Antioxidant Activity of Food Products. Molecules 2024, 29, 3941. [Google Scholar] [CrossRef] [PubMed]
- Cano y Postigo, L.O.; Jacobo-Velázquez, D.A.; Guajardo-Flores, D.; Garcia Amezquita, L.E.; García-Cayuela, T. Solid-State Fermentation for Enhancing the Nutraceutical Content of Agrifood by-Products: Recent Advances and Its Industrial Feasibility. Food Biosci. 2021, 41, 100926. [Google Scholar] [CrossRef]
- Bhanja Dey, T.; Chakraborty, S.; Jain, K.K.; Sharma, A.; Kuhad, R.C. Antioxidant Phenolics and Their Microbial Production by Submerged and Solid State Fermentation Process: A Review. Trends Food Sci. Technol. 2016, 53, 60–74. [Google Scholar] [CrossRef]
- Prado Barragán, L.A.; Figueroa, J.J.B.; Rodríguez Durán, L.V.; Aguilar González, C.N.; Hennigs, C. Fermentative Production Methods. In Biotransformation of Agricultural Waste and By-Products; Poltronieri, P., D’Urso, O.F., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 189–217. [Google Scholar]
- Ge, X.; Vasco-Correa, J.; Li, Y. Solid-State Fermentation Bioreactors and Fundamentals. In Current Developments in Biotechnology and Bioengineering; Larroche, C., Sanromán, M.A., Du, G., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 381–402. [Google Scholar]
- Pathania, S.; Sharma, N.; Handa, S. Utilization of Horticultural Waste (Apple Pomace) for Multiple Carbohydrase Production from Rhizopus delemar F2 under Solid State Fermentation. J. Genet. Eng. Biotechnol. 2018, 16, 181–189. [Google Scholar] [CrossRef]
- Šelo, G.; Planinić, M.; Tišma, M.; Martinović, J.; Perković, G.; Bucić-Kojić, A. Bioconversion of Grape Pomace with Rhizopus oryzae under Solid-State Conditions: Changes in the Chemical Composition and Profile of Phenolic Compounds. Microorganisms 2023, 11, 956. [Google Scholar] [CrossRef]
- Ortega-Hernández, E.; Martinez-Alvarado, L.; Acosta-Estrada, B.A.; Antunes-Ricardo, M. Solid-State Fermented Pineapple Peel: A Novel Food Ingredient with Antioxidant and Anti-Inflammatory Properties. Foods 2023, 12, 4162. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Zhang, F.; Chai, Z.; Liu, M.; Battino, M.; Meng, X. Mixed Fermentation of Blueberry Pomace with L. rhamnosus GG and L. plantarum-1: Enhance the Active Ingredient, Antioxidant Activity and Health-Promoting Benefits. Food Chem. Toxicol. 2019, 131, 110541. [Google Scholar] [CrossRef]
- Méndez-Galarraga, M.P.; Hurtado-Romero, A.; Antunes-Ricardo, M.; Garcia-Amezquita, L.E.; Pirovani, M.É.; Vinderola, G.; Van De Velde, F.; García-Cayuela, T. Enhancing Safety and Bioactivity of Blueberry-Watermelon Smoothies through Combined Ultrasound and Lactic Acid Fermentation with Potential Probiotics. Food Biosci. 2025, 69, 106991. [Google Scholar] [CrossRef]
- Hurtado-Romero, A.; Zepeda-Hernández, A.; Uribe-Velázquez, T.; Rosales-De La Cruz, M.F.; Raygoza-Murguía, L.V.; Garcia-Amezquita, L.E.; García-Cayuela, T. Utilization of Blueberry-Based Ingredients for Formulating a Synbiotic Petit Suisse Cheese: Physicochemical, Microbiological, Sensory, and Functional Characterization during Cold Storage. LWT 2023, 183, 114955. [Google Scholar] [CrossRef]
- Kaminskyj, S.G.W. Fundamentals of Growth, Storage, Genetics and Microscopy of Aspergillus Nidulans. Fungal Genet. Rep. 2001, 48, 25–31. [Google Scholar] [CrossRef]
- Sánchez-Rangel, J.C.; Benavides, J.; Heredia, J.B.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. The Folin–Ciocalteu Assay Revisited: Improvement of Its Specificity for Total Phenolic Content Determination. Anal. Methods 2013, 5, 5990. [Google Scholar] [CrossRef]
- Martín-Gómez, J.; Varo, M.Á.; Mérida, J.; Serratosa, M.P. Influence of Drying Processes on Anthocyanin Profiles, Total Phenolic Compounds and Antioxidant Activities of Blueberry (Vaccinium corymbosum). LWT 2020, 120, 108931. [Google Scholar] [CrossRef]
- García-Cayuela, T.; Gómez-Maqueo, A.; Guajardo-Flores, D.; Welti-Chanes, J.; Cano, M.P. Characterization and Quantification of Individual Betalain and Phenolic Compounds in Mexican and Spanish Prickly Pear (Opuntia ficus-indica L. Mill) Tissues: A Comparative Study. J. Food Compos. Anal. 2019, 76, 1–13. [Google Scholar] [CrossRef]
- Ibarruri, J.; Cebrián, M.; Hernández, I. Valorisation of Fruit and Vegetable Discards by Fungal Submerged and Solid-State Fermentation for Alternative Feed Ingredients Production. J. Food Compos. Anal. 2021, 281, 111901. [Google Scholar] [CrossRef]
- Erskine, E.; Ozkan, G.; Lu, B.; Capanoglu, E. Effects of Fermentation Process on the Antioxidant Capacity of Fruit Byproducts. ACS Omega 2023, 8, 4543–4553. [Google Scholar] [CrossRef]
- Vijayalaxmi, S.; Jayalakshmi, S.K.; Sreeramulu, K. Polyphenols from Different Agricultural Residues: Extraction, Identification and Their Antioxidant Properties. J. Food Sci. Technol. 2015, 52, 2761–2769. [Google Scholar] [CrossRef]
- Amaya-Chantaca, D.; Flores-Gallegos, A.C.; Iliná, A.; Aguilar, C.N.; Sepúlveda-Torre, L.; Ascacio-Vadlés, J.A.; Chávez-González, M.L. Comparative Extraction Study of Grape Pomace Bioactive Compounds by Submerged and Solid-state Fermentation. J. Chem. Technol. Biotechnol. 2022, 97, 1494–1505. [Google Scholar] [CrossRef]
- Troncoso Mesa, S.; Flórez-Méndez, J.; López, J.; Bustos, R. Optimization of Conventional Solid-Liquid Extraction and Microwave-Assisted Extraction of Polyphenols and Antioxidant Compounds of Blueberry (Vaccinium corymbosum) Pomace through Response Surface Methodology. J. Berry Res. 2021, 11, 649–668. [Google Scholar] [CrossRef]
- Benoit, I.; Navarro, D.; Marnet, N.; Rakotomanomana, N.; Lesage-Meessen, L.; Sigoillot, J.-C.; Asther, M.; Asther, M. Feruloyl Esterases as a Tool for the Release of Phenolic Compounds from Agro-Industrial by-Products. Carbohydr. Res. 2006, 341, 1820–1827. [Google Scholar] [CrossRef]
- Denardi De Souza, T.; Leal, C.A.; Massarolo, K.C.; Badiale-Fulong, E. Profile of Phenolic Compounds Released from Rice Bran by Rhizopus oryzae and Trichoderma reesei: Their Relation with Hydrolases Activity. J. Food Sci. 2019, 84, 1382–1389. [Google Scholar] [CrossRef]
- Kang, S. Production of Cellulases and Hemicellulases by Aspergillus niger KK2 from Lignocellulosic Biomass. Bioresour. Technol. 2004, 91, 153–156. [Google Scholar] [CrossRef]
- Rezaei, F.; Joh, L.D.; Kashima, H.; Reddy, A.P.; VanderGheynst, J.S. Selection of conditions for cellulase and xylanase extraction from switchgrass colonized by Acidothermus cellulolyticus. Appl. Biochem. Biotechnol. 2011, 164, 793–803. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Thakur, C.; Ahlawat, Y.K.; Mehta, H.; Chauhan, V.; Sharma, S.; Bisht, K. Maximizing enzyme production by standardizing process parameters through one factor at a time approach (OFAT) subjected to a statistical technique: Response surface methodology (RSM). Plant Sci. Today 2025, 12, 4. [Google Scholar] [CrossRef]
- Yuan, K.; Wu, G.; Li, X.; Zeng, Y.; Wen, X.; Liu, R.; Bai, W. Anthocyanins degradation mediated by β-glycosidase contributes to the color loss during alcoholic fermentation in a structure-dependent manner. Food Res. Int. 2024, 175, 113732. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, D.; Zhang, J.; Shen, J.; Cao, J.; Gu, H.; Cui, M.; He, L.; Chen, G.; Liu, S.; et al. Improving Soluble Phenolic Profile and Antioxidant Activity of Grape Pomace Seeds through Fungal Solid-State Fermentation. Foods 2024, 13, 1158. [Google Scholar] [CrossRef] [PubMed]
- López-Cárdenas, F.; Ochoa-Reyes, E.; Baeza-Jiménez, R.; Tafolla-Arellano, J.C.; Ascacio-Valdés, J.A.; Buenrostro-Figueroa, J.J. Solid-State Fermentation as a Sustainable Tool for Extracting Phenolic Compounds from Cascalote Pods. Fermentation 2023, 9, 823. [Google Scholar] [CrossRef]
- Da Costa Maia, I.; Thomaz Dos Santos D’Almeida, C.; Guimarães Freire, D.M.; d’Avila Costa Cavalcanti, E.; Cameron, L.C.; Furtado Dias, J.; Simões Larraz Ferreira, M. Effect of Solid-State Fermentation over the Release of Phenolic Compounds from Brewer’s Spent Grain Revealed by UPLC-MSE. LWT 2020, 133, 110136. [Google Scholar] [CrossRef]
- Yepes-Betancur, D.P.; Márquez-Cardozo, C.J.; Cadena-Chamorro, E.M.; Martinez-Saldarriaga, J.; Torres-León, C.; Ascacio-Valdes, A.; Aguilar, C.N. Solid-State Fermentation—Assisted Extraction of Bioactive Compounds from Hass Avocado Seeds. Food Bioprod. Process. 2021, 126, 155–163. [Google Scholar] [CrossRef]
- Valencia-Hernández, L.J.; Wong-Paz, J.E.; Ascacio-Valdés, J.A.; Prado-Barragan, A.; Chávez-González, M.L.; Aguilar, C.N. Fungal Biodegradation of Procyanidin in Submerged Fermentation. Fermentation 2025, 11, 75. [Google Scholar] [CrossRef]
- Leonard, W.; Zhang, P.; Ying, D.; Adhikari, B.; Fang, Z. Fermentation Transforms the Phenolic Profiles and Bioactivities of Plant-Based Foods. Biotechnol. Adv. 2021, 49, 107763. [Google Scholar] [CrossRef]
- Rupasinghe, H.P.V.; Parmar, I.; Neir, S.V. Biotransformation of Cranberry Proanthocyanidins to Probiotic Metabolites by Lactobacillus rhamnosus Enhances Their Anticancer Activity in HepG2 Cells In Vitro. Oxid. Med. Cell. Longev. 2019, 2019, 4750795. [Google Scholar] [CrossRef] [PubMed]
- Zang, Z.; Tang, S.; Li, Z.; Chou, S.; Shu, C.; Chen, Y.; Chen, W.; Yang, S.; Yang, Y.; Tian, J.; et al. An Updated Review on the Stability of Anthocyanins Regarding the Interaction with Food Proteins and Polysaccharides. Compr. Rev. Food Sci. Food Saf. 2022, 21, 4378–4401. [Google Scholar] [CrossRef]
- Mallet, J.-F.; Shahbazi, R.; Alsadi, N.; Saleem, A.; Sobiesiak, A.; Arnason, J.T.; Matar, C. Role of a Mixture of Polyphenol Compounds Released after Blueberry Fermentation in Chemoprevention of Mammary Carcinoma: In Vivo Involvement of miR-145. Int. J. Mol. Sci. 2023, 24, 3677. [Google Scholar] [CrossRef]
- Ryu, J.; Kang, H.R.; Cho, S.K. Changes Over the Fermentation Period in Phenolic Compounds and Antioxidant and Anticancer Activities of Blueberries Fermented by Lactobacillus plantarum. J. Food Sci. 2019, 84, 2347–2356. [Google Scholar] [CrossRef]
- Sivapragasam, N.; Neelakandan, N.; Rupasinghe, H.P.V. Potential Health Benefits of Fermented Blueberry: A Review of Current Scientific Evidence. Trends Food Sci. Technol. 2023, 132, 103–120. [Google Scholar] [CrossRef]
- Huang, W.; Zhu, Y.; Li, C.; Sui, Z.; Min, W. Effect of Blueberry Anthocyanins Malvidin and Glycosides on the Antioxidant Properties in Endothelial Cells. Oxidative Med. Cell. Longev. 2016, 2016, 1591803. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Deng, R.; Hua, X.; Zhang, L.; Lu, F.; Coursey, T.G.; Pflugfelder, S.C.; Li, D.-Q. Blueberry Component Pterostilbene Protects Corneal Epithelial Cells from Inflammation via Anti-Oxidative Pathway. Sci. Rep. 2016, 6, 19408. [Google Scholar] [CrossRef] [PubMed]
- Lau, F.C.; Bielinski, D.F.; Joseph, J.A. Inhibitory Effects of Blueberry Extract on the Production of Inflammatory Mediators in Lipopolysaccharide-activated BV2 Microglia. J. Neurosci. Res. 2007, 85, 1010–1017. [Google Scholar] [CrossRef]
- Vuong, T.; Martin, L.; Matar, C. Antioxidant Activity of Fermented Berry Juices and Their Effects on Nitric Oxide and Tumor Necrosis Factor-Alpha Production in Macrophages 264.7 Gamma NO(−) Cell Line. J. Food Biochem. 2006, 30, 249–268. [Google Scholar] [CrossRef]
- Felgus-Lavefve, L.; Howard, L.; Adams, S.H.; Baum, J.I. The Effects of Blueberry Phytochemicals on Cell Models of Inflammation and Oxidative Stress. Adv. Nutr. 2022, 13, 1279–1309. [Google Scholar] [CrossRef]
- Son, T.G.; Camandola, S.; Mattson, M.P. Hormetic Dietary Phytochemicals. Neuromol. Med. 2008, 10, 236–246. [Google Scholar] [CrossRef]
- Youdim, K.A.; McDonald, J.; Kalt, W.; Joseph, J.A. Potential Role of Dietary Flavonoids in Reducing Microvascular Endothelium Vulnerability to Oxidative and Inflammatory Insults. J. Nutr. Biochem. 2002, 13, 282–288. [Google Scholar] [CrossRef]
- Sahakyan, N. Plant-Derived Phenolics as Regulators of Nitric Oxide Production in Microglia: Mechanisms and Therapeutic Potential. Med. Gas Res. 2026, 16, 169–175. [Google Scholar] [CrossRef]
- Roy, S.; Dickerson, R.; Khanna, S.; Collard, E.; Gnyawali, U.; Gordillo, G.M.; Sen, C.K. Particulate β-glucan Induces TNF-α Production in Wound Macrophages via a Redox-sensitive NF-κβ-dependent Pathway. Wound Repair Regen. 2011, 19, 411–419. [Google Scholar] [CrossRef]
- Ben Lagha, A.; Dudonné, S.; Desjardins, Y.; Grenier, D. Wild Blueberry (Vaccinium angustifolium Ait.) Polyphenols Target Fusobacterium Nucleatum and the Host Inflammatory Response: Potential Innovative Molecules for Treating Periodontal Diseases. J. Agric. Food Chem. 2015, 63, 6999–7008. [Google Scholar] [CrossRef]
- Vendrame, S.; Daugherty, A.; Kristo, A.S.; Riso, P.; Klimis-Zacas, D. Wild Blueberry (Vaccinium angustifolium) Consumption Improves Inflammatory Status in the Obese Zucker Rat Model of the Metabolic Syndrome. J. Nutr. Biochem. 2013, 24, 1508–1512. [Google Scholar] [CrossRef] [PubMed]
- Gu, I.; Brownmiller, C.; Stebbins, N.B.; Mauromoustakos, A.; Howard, L.; Lee, S.O. Berry Phenolic and Volatile Extracts Inhibit Pro-Inflammatory Cytokine Secretion in LPS-Stimulated RAW264.7 Cells through Suppression of NF-κB Signaling Pathway. Antioxidants 2020, 9, 871. [Google Scholar] [CrossRef] [PubMed]
- Jeelani, M.; Fouotsa, H.; Mohammed, O.A.; Alfaifi, J.; Adebayo, S.; Ahmed, M.M.; Isa, A.I. Naturally Occurring Benzophenones and Xanthones from Garcinia smeathmannii (Planch. & Triana) Oliv. Displayed Anti-Inflammatory Effects by Modulating the Activities of Inflammatory Mediators in LPS-Stimulated RAW 264.7 Macrophages. Front. Pharmacol. 2024, 15, 1370073. [Google Scholar] [CrossRef]
- Yoon, S.B.; Lee, Y.J.; Park, S.K.; Kim, H.C.; Bae, H.; Kim, H.M.; Park, W. Anti-Inflammatory Effects of Scutellaria baicalensis Water Extract on LPS-Activated RAW 264.7 Macrophages. J. Ethnopharmacol. 2009, 125, 286–290. [Google Scholar] [CrossRef]






| Compound | NI-0h | NI-24h | AN-24h | RO-24h |
|---|---|---|---|---|
| Hydroxybenzoic acids | ||||
| Gallic acid | 23.26 ± 0.22 B | 19.40 ± 0.03 C | 28.38 ± 2.13 A | 29.72 ± 1.62 A |
| Protocatechuic acid | 20.68 ± 0.08 A | 17.75 ± 0.17 B | 21.06 ± 1.02 A | 22.32 ± 0.83 A |
| p-Hydroxybenzoic acid | 18.46 ± 0.20 A | 15.99 ± 1.51 A | 15.65 ± 0.18 A | 16.95 ± 1.58 A |
| Hydroxycinnamic acids | ||||
| Sinapic acid | 16.28 ± 0.04 A | 15.35 ± 0.10 A | 16.29 ± 0.32 A | 16.90 ± 0.57 A |
| Caffeic acid | 18.78 ± 0.12 A | 16.67 ± 0.06 B | 18.28 ± 0.67 A | 18.99 ± 0.54 A |
| Chlorogenic acid | 29.39 ± 0.28 A | 19.89 ± 0.36 B | 26.33 ± 1.88 A | 28.73 ± 1.58 A |
| p-Coumaric acid | 18.47 ± 0.05 A | 15.51 ± 0.02 C | 16.96 ± 0.38 B | 17.52 ± 0.38 AB |
| 4-hydroxyferulic acid | 21.22 ± 0.14 A | 20.40 ± 0.22 AB | 18.55 ± 0.72 C | 18.76 ± 0.42 BC |
| Ferulic acid | 5.48 ± 0.11 | ND | ND | ND |
| Flavan-3-ols | ||||
| (+)-Catechin | 43.90 ± 1.47 A | 16.95 ± 0.64 B | 36.47 ± 6.93 A | 45.86 ± 6.17 A |
| Epicatechin | 133.41 ± 2.36 A | 49.96 ± 1.67 B | 97.34 ± 17.10 A | 117.98 ± 13.52 A |
| Flavonols | ||||
| Quercetin | 3.71 ± 0.12 A | ND | 1.11 ± 1.05 B | 1.41 ± 0.70 B |
| Syringetin | 16.06 ± 0.22 A | 15.34 ± 0.00 A | 16.48 ± 0.41 A | 17.16 ± 1.25 A |
| Myricetin | 1.74 ± 0.13 A | ND | 2.09 ± 0.16 A | 3.11 ± 0.92 A |
| Kaempferol | 3.91 ± 0.22 A | ND | 1.61 ± 0.56 A | 5.09 ± 1.04 A |
| Anthocyanins | ||||
| Cyanidin-3-galactoside | 10.08 ± 0.09 A | 5.86 ± 0.65 D | 7.07 ± 0.24 C | 8.20 ± 0.84 B |
| Petunidin-3-galactoside | 80.98 ± 2.37 A | 39.54 ± 2.27 C | 58.29 ± 6.56 B | 66.59 ± 6.70 B |
| Peonidin-3-glucoside | 42.26 ± 1.14 A | 20.75 ± 0.90 B | 35.99 ± 3.56 B | 37.95 ± 1.17 B |
| Malvidin-3-glucoside | 140.97 ± 4.19 A | 68.19 ± 1.86 C | 111.15 ± 13.11 B | 136.10 ± 13.10 A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hurtado-Romero, A.; Luzardo-Ocampo, I.; Antunes-Ricardo, M.; López-Pacheco, F.; Garcia-Amezquita, L.E.; Carrillo-Nieves, D.; García-Cayuela, T. Fungal Solid-State and Submerged Fermentation of Blueberry Bagasse: Extraction Strategies, Phenolic Profiling, and Cellular Immunomodulation. Fermentation 2026, 12, 128. https://doi.org/10.3390/fermentation12030128
Hurtado-Romero A, Luzardo-Ocampo I, Antunes-Ricardo M, López-Pacheco F, Garcia-Amezquita LE, Carrillo-Nieves D, García-Cayuela T. Fungal Solid-State and Submerged Fermentation of Blueberry Bagasse: Extraction Strategies, Phenolic Profiling, and Cellular Immunomodulation. Fermentation. 2026; 12(3):128. https://doi.org/10.3390/fermentation12030128
Chicago/Turabian StyleHurtado-Romero, Alejandra, Ivan Luzardo-Ocampo, Marilena Antunes-Ricardo, Felipe López-Pacheco, Luis Eduardo Garcia-Amezquita, Danay Carrillo-Nieves, and Tomás García-Cayuela. 2026. "Fungal Solid-State and Submerged Fermentation of Blueberry Bagasse: Extraction Strategies, Phenolic Profiling, and Cellular Immunomodulation" Fermentation 12, no. 3: 128. https://doi.org/10.3390/fermentation12030128
APA StyleHurtado-Romero, A., Luzardo-Ocampo, I., Antunes-Ricardo, M., López-Pacheco, F., Garcia-Amezquita, L. E., Carrillo-Nieves, D., & García-Cayuela, T. (2026). Fungal Solid-State and Submerged Fermentation of Blueberry Bagasse: Extraction Strategies, Phenolic Profiling, and Cellular Immunomodulation. Fermentation, 12(3), 128. https://doi.org/10.3390/fermentation12030128











