Outgrowth Inhibition of Clostridium sporogenes Spores by a Bacteriocin-Producing Streptococcus thermophilus Strain, Under Conditions Simulating Graviera Cheese Manufacture
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
2.2. Spore Preparation
2.3. Fermentation
2.4. Estimation of Thermophilin-T Activity
2.5. Enumeration of Microbial Populations
2.6. HPLC Analysis
2.7. Statistical Analysis
3. Results
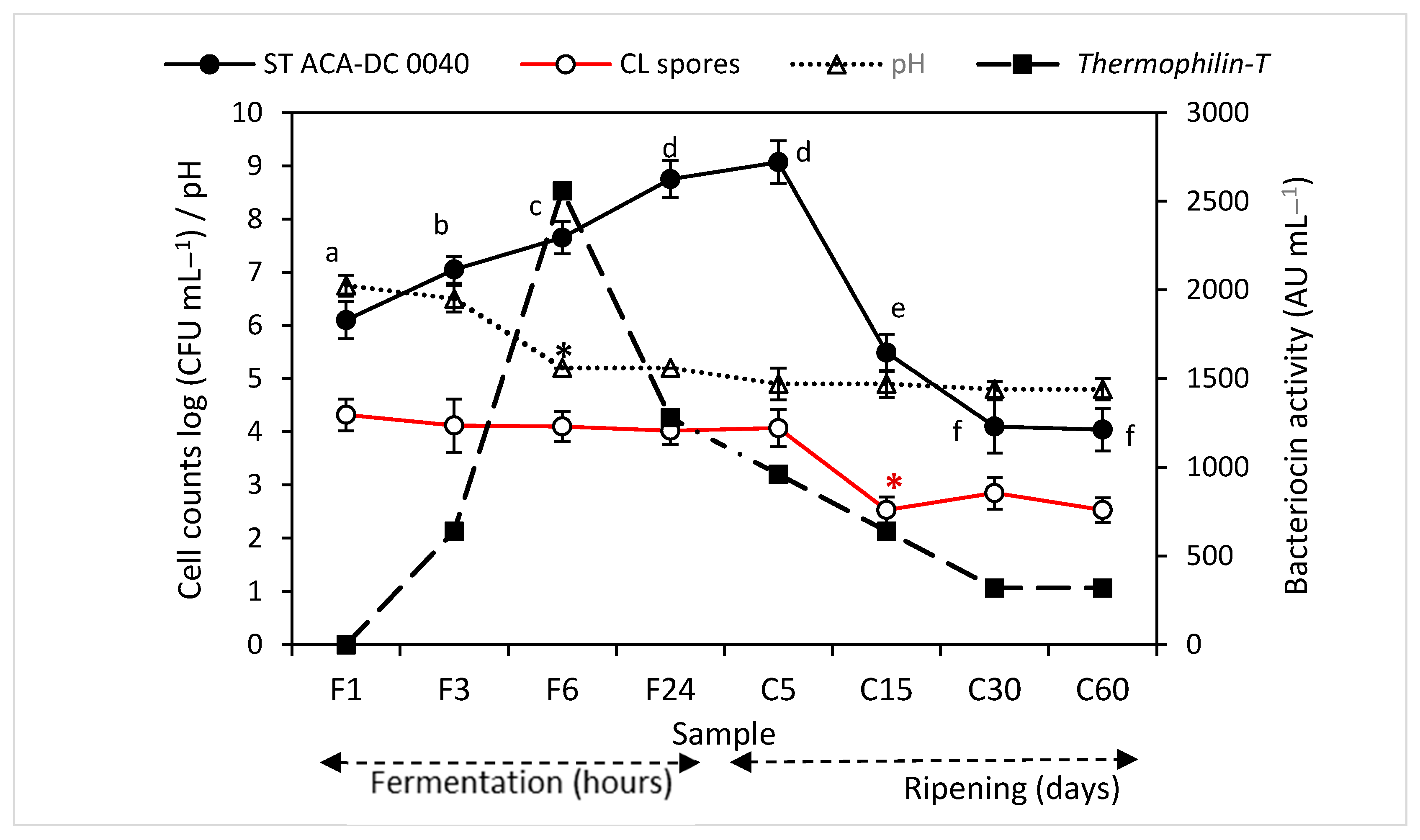
3.1. Evolution of Microbiological Parameters
3.2. Evolution of pH
3.3. Evolution of Bacteriocin Activity
3.4. Organic Acid Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACA-DC | Agricultural College of Athens-Dairy Collection |
| LBD | Late Blowing Defect |
| BAF | Butyric Acid Fermentation |
| AU | Arbitrary Units |
| BP | Bacteriocin Producing |
| NBP | Non Bacteriocin Producing |
References
- Le Bourhis, A.-G.; Doré, J.; Carlier, J.-P.; Chamba, J.-F.; Popoff, M.-R.; Tholozan, J.-L. Contribution of C. beijerinckii and C. sporogenes in Association with C. tyrobutyricum to the Butyric Fermentation in Emmental Type Cheese. Int. J. Food Microbiol. 2007, 113, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Torres, N.; Garde, S.; Peirotén, Á.; Ávila, M. Impact of Clostridium spp. on Cheese Characteristics: Microbiology, Color, Formation of Volatile Compounds and off-Flavors. Food Control 2015, 56, 186–194. [Google Scholar] [CrossRef]
- Podrzaj, L.; Burtscher, J.; Küller, F.; Domig, K.J. Strain-Dependent Cheese Spoilage Potential of Clostridium tyrobutyricum. Microorganisms 2020, 8, 1836. [Google Scholar] [CrossRef]
- Ávila, M.; Gómez-Torres, N.; Gaya, P.; Garde, S. Effect of a Nisin-Producing Lactococcal Starter on the Late Blowing Defect of Cheese Caused by Clostridium tyrobutyricum. Int. J. Food Sci. Technol. 2020, 55, 3343–3349. [Google Scholar] [CrossRef]
- Cocolin, L.; Innocente, N.; Biasutti, M.; Comi, G. The Late Blowing in Cheese: A New Molecular Approach Based on PCR and DGGE to Study the Microbial Ecology of the Alteration Process. Int. J. Food Microbiol. 2004, 90, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.L.; Tran-dinh, N.; Chapman, B. Clostridium sporogenes PA 3679 and Its Uses in the Derivation of Thermal Processing Schedules for Low-Acid Shelf-Stable Foods and as a Research Model for Proteolytic Clostridium botulinum. J. Food Prot. 2012, 75, 779–792. [Google Scholar] [CrossRef] [PubMed]
- Diao, M.M.; André, S.; Membré, J.-M. Meta-Analysis of D-Values of Proteolytic Clostridium botulinum and Its Surrogate Strain Clostridium sporogenes PA 3679. Int. J. Food Microbiol. 2014, 174, 23–30. [Google Scholar] [CrossRef]
- Taylor, R.H.; Dunn, M.L.; Ogden, L.V.; Jefferies, L.K.; Eggett, D.L.; Steele, F.M. Conditions Associated with Clostridium sporogenes Growth as a Surrogate for Clostridium botulinum in Nonthermally Processed Canned Butter. J. Dairy Sci. 2013, 96, 2754–2764. [Google Scholar] [CrossRef] [PubMed]
- Driehuis, F.; Elferink, S.J.W.H.O. The Impact of the Quality of Silage on Animal Health and Food Safety: A Review. Vet. Q. 2000, 22, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Carminati, D.; Perrone, A.; Neviani, E. Inhibition of Clostridium sporogenes Growth in Mascarpone Cheese by Co-Inoculation with Streptococcus thermophilus under Conditions of Temperature Abuse. Food Microbiol. 2001, 18, 571–579. [Google Scholar] [CrossRef]
- Loessner, M.J.; Maier, S.K.; Schiwek, P.; Scherer, S. Long-Chain Polyphosphates Inhibit Growth of Clostridium tyrobutyricum in Processed Cheese Spreads. J. Food Prot. 1997, 60, 493–498. [Google Scholar] [CrossRef]
- Regulation—1333/2008—EN—Additives—EUR-Lex. Available online: https://eur-lex.europa.eu/eli/reg/2008/1333/oj/eng (accessed on 17 September 2025).
- Current Publications of the Articles of the Food and Drinks Code. 2009. Available online: https://www.aade.gr/en/chemical-laboratories/businesses/food-materials-contact-food/current-publications-articles-food-and-drinks-code (accessed on 7 January 2025).
- Abee, T.; Krockel, L.; Hill, C. Bacteriocins: Modes of Action and Potentials in Food Preservation and Control of Food Poisoning. Int. J. Food Microbiol. 1995, 28, 169–185. [Google Scholar] [CrossRef]
- Silva, C.C.G.; Silva, S.P.M.; Ribeiro, S.C. Application of Bacteriocins and Protective Cultures in Dairy Food Preservation. Front. Microbiol. 2018, 9, 594. [Google Scholar] [CrossRef]
- Ribeiro, S.C.; O’Connor, P.M.; Ross, R.P.; Stanton, C.; Silva, C.C.G. An Anti-Listerial Lactococcus lactis Strain Isolated from Azorean Pico Cheese Produces Lacticin 481. Int. Dairy J. 2016, 63, 18–28. [Google Scholar] [CrossRef]
- Burgos, M.J.G.; Pulido, R.P.; Aguayo, M.C.L.; Gálvez, A.; Lucas, R. The Cyclic Antibacterial Peptide Enterocin AS-48: Isolation, Mode of Action, and Possible Food Applications. Int. J. Mol. Sci. 2014, 15, 22706–22767. [Google Scholar] [CrossRef]
- González, L.; Zárate, V. Inhibitory activity of Lactobacillus plantarum TF711 against Clostridium sporogenes when used as adjunct culture in cheese manufacture. J. Dairy Res. 2015, 82, 236–241. [Google Scholar] [CrossRef]
- Aktypis, A.; Kalantzopoulos, G.; Huis in’t Veld, J.H.J.; Ten Brink, B. Purification and Characterization of Thermophilin-T, a Novel Bacteriocin Produced by Streptococcus thermophilus ACA-DC 0040. J. Appl. Microbiol. 1998, 84, 568–576. [Google Scholar] [CrossRef]
- Klijn, N.; Nieuwenhof, F.F.; Hoolwerf, J.D.; van der Waals, C.B.; Weerkamp, A.H. Identification of Clostridium Tyrobutyricum as the Causative Agent of Late Blowing in Cheese by Species-Specific PCR Amplification. Appl. Environ. Microbiol. 1995, 61, 2919–2924. [Google Scholar] [CrossRef] [PubMed]
- Akbal1, S.; Öner, Z. Investigation of Some Properties and Late Blowing Defect of Model White Cheeses Contaminated with Clostridium sporogenes Strains. Turk. J. Agric. Food Sci. Technol. 2025, 13, 958–963. [Google Scholar] [CrossRef]
- Mathot, A.G.; Beliard, E.; Thuault, D. Streptococcus thermophilus 580 Produces a Bacteriocin Potentially Suitable for Inhibition of Clostridium tyrobutyricum in Hard Cheese. J. Dairy Sci. 2003, 86, 3068–3074. [Google Scholar] [CrossRef] [PubMed]
- Tagg, J.R.; McGiven, A.R. Assay System for Bacteriocins. Appl. Microbiol. 1971, 21, 943. [Google Scholar] [CrossRef] [PubMed]
- Anastasiou, R.; Aktypis, A.; Georgalaki, M.; Papadelli, M.; De Vuyst, L.; Tsakalidou, E. Inhibition of Clostridium tyrobutyricum by Streptococcus macedonicus ACA-DC 198 under Conditions Mimicking Kasseri Cheese Production and Ripening. Int. Dairy J. 2009, 19, 330–335. [Google Scholar] [CrossRef]
- Benech, R.-O.; Kheadr, E.E.; Laridi, R.; Lacroix, C.; Fliss, I. Inhibition of Listeria innocua in Cheddar Cheese by Addition of Nisin Z in Liposomes or by In Situ Production in Mixed Culture. Appl. Environ. Microbiol. 2002, 68, 3683–3690. [Google Scholar] [CrossRef]
- Garde, S.; Gómez-Torres, N.; Hernández, M.; Ávila, M. Susceptibility of Clostridium perfringens to Antimicrobials Produced by Lactic Acid Bacteria: Reuterin and Nisin. Food Control 2014, 44, 22–25. [Google Scholar] [CrossRef]
- Foulquié Moreno, M.R.; Callewaert, R.; Devreese, B.; Van Beeumen, J.; De Vuyst, L. Isolation and Biochemical Characterisation of Enterocins Produced by Enterococci from Different Sources. J. Appl. Microbiol. 2003, 94, 214–229. [Google Scholar] [CrossRef]
- Rodríguez, E.; Calzada, J.; Arqués, J.L.; Rodríguez, J.M.; Nuñez, M.; Medina, M. Antimicrobial Activity of Pediocin-Producing Lactococcus lactis on Listeria monocytogenes, Staphylococcus aureus and Escherichia coli O157:H7 in Cheese. Int. Dairy J. 2005, 15, 51–57. [Google Scholar] [CrossRef]
- Egan, K.; Field, D.; Rea, M.C.; Ross, R.P.; Hill, C.; Cotter, P.D. Bacteriocins: Novel Solutions to Age-Old Spore-Related Problems? Front. Microbiol. 2016, 7, 461. [Google Scholar] [CrossRef] [PubMed]
- Gut, I.M.; Prouty, A.M.; Ballard, J.D.; van der Donk, W.A.; Blanke, S.R. Inhibition of Bacillus anthracis spore outgrowth by nisin. Antimicrob. Agents Chemother. 2008, 52, 4281–4288. [Google Scholar] [CrossRef]
- Rilla, N.; Martínez, B.; Delgado, T.; Rodríguez, A. Inhibition of Clostridium tyrobutyricum in Vidiago Cheese by Lactococcus lactis Ssp. Lactis IPLA 729, a Nisin Z Producer. Int. J. Food Microbiol. 2003, 85, 23–33. [Google Scholar] [CrossRef]
- Carmen Martínez-Cuesta, M.; Bengoechea, J.; Bustos, I.; Rodríguez, B.; Requena, T.; Peláez, C. Control of Late Blowing in Cheese by Adding Lacticin 3147-Producing Lactococcus lactis IFPL 3593 to the Starter. Int. Dairy J. 2010, 20, 18–24. [Google Scholar] [CrossRef]
- Abhyankar, W.; de Koning, L.J.; Brul, S.; de Koster, C.G. Spore proteomics: The past, present and the future. FEMS Microbiol. Lett. 2014, 358, 137–144. [Google Scholar] [CrossRef][Green Version]
- Garde, S.; Arias, R.; Gaya, P.; Nuñez, M. Occurrence of Clostridium spp. in Ovine Milk and Manchego Cheese with Late Blowing Defect: Identification and Characterization of Isolates. Int. Dairy J. 2011, 21, 272–278. [Google Scholar] [CrossRef]
- Bogovič Matijašić, B.; Koman Rajšp, M.; Perko, B.; Rogelj, I. Inhibition of Clostridium tyrobutyricum in Cheese by Lactobacillus gasseri. Int. Dairy J. 2007, 17, 157–166. [Google Scholar] [CrossRef]
- Curioni, P.M.G.; Bosset, J.O. Key Odorants in Various Cheese Types as Determined by Gas Chromatography-Olfactometry. Int. Dairy J. 2002, 12, 959–984. [Google Scholar] [CrossRef]
- Garde, S.; Ávila, M.; Gaya, P.; Arias, R.; Nuñez, M. Sugars and Organic Acids in Raw and Pasteurized Milk Manchego Cheeses with Different Degrees of Late Blowing Defect. Int. Dairy J. 2012, 25, 87–91. [Google Scholar] [CrossRef]
- Zhang, F.; Tang, X.; Xiong, B. Optimal Calcium Propionate Supplementation in Early-Lactation Dairy Cows Improves Milk Yield and Alters Milk Composition. Animals 2025, 15, 2995. [Google Scholar] [CrossRef]
- Montville, T.J.; Parris, N.; Conway, L.K. Influence of pH on Organic Acid Production by Clostridium sporogenes in Test Tube and Fermentor Cultures. Appl. Environ. Microbiol. 1985, 49, 733–736. [Google Scholar] [CrossRef] [PubMed]
- Canganella, F.; Kuk, S.-U.; Morgan, H.; Wiegel, J. Clostridium thermobutyricum: Growth Studies and Stimulation of Butyrate Formation by Acetate Supplementation. Microbiol. Res. 2002, 157, 149–156. [Google Scholar] [CrossRef]
- Tao, Y.; Hu, X.; Zhu, X.; Jin, H.; Xu, Z.; Tang, Q.; Li, X. Production of Butyrate from Lactate by a Newly Isolated Clostridium sp. BPY5. Appl. Biochem. Biotechnol. 2016, 179, 361–374. [Google Scholar] [CrossRef]
- Flythe, M.D.; Russell, J.B. Fermentation acids inhibit amino acid deamination by Clostridium sporogenes MD1 via a mechanism involving a decline in intracellular glutamate rather than protonmotive force. Microbiology 2006, 152, 2619–2624. [Google Scholar] [CrossRef][Green Version]



| Sample | Sampling Time | Graviera Cheese Manufacturing Phase |
|---|---|---|
| F1 | 1 h | Starter culture activation at 37 °C, 1 h |
| F3 | 3 h | Curd scalding at 52 °C, 20 min |
| F6 | 6 h | Curd drainage at 16 °C |
| F24 | 24 h | Salting at 16 °C |
| C5 | 5th d | Ripening at 16 °C |
| C15 | 15th d | Ripening at 16 °C |
| C30 | 30th d | Ripening at 16 °C |
| C60 | 60th d | Ripening at 16 °C |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Aktypis, A.; Baklagi, S.; Georgalaki, M.; Misyrli, G.; Georgala, A. Outgrowth Inhibition of Clostridium sporogenes Spores by a Bacteriocin-Producing Streptococcus thermophilus Strain, Under Conditions Simulating Graviera Cheese Manufacture. Fermentation 2026, 12, 125. https://doi.org/10.3390/fermentation12030125
Aktypis A, Baklagi S, Georgalaki M, Misyrli G, Georgala A. Outgrowth Inhibition of Clostridium sporogenes Spores by a Bacteriocin-Producing Streptococcus thermophilus Strain, Under Conditions Simulating Graviera Cheese Manufacture. Fermentation. 2026; 12(3):125. https://doi.org/10.3390/fermentation12030125
Chicago/Turabian StyleAktypis, Anastasios, Stellina Baklagi, Marina Georgalaki, Georgia Misyrli, and Aikaterini Georgala. 2026. "Outgrowth Inhibition of Clostridium sporogenes Spores by a Bacteriocin-Producing Streptococcus thermophilus Strain, Under Conditions Simulating Graviera Cheese Manufacture" Fermentation 12, no. 3: 125. https://doi.org/10.3390/fermentation12030125
APA StyleAktypis, A., Baklagi, S., Georgalaki, M., Misyrli, G., & Georgala, A. (2026). Outgrowth Inhibition of Clostridium sporogenes Spores by a Bacteriocin-Producing Streptococcus thermophilus Strain, Under Conditions Simulating Graviera Cheese Manufacture. Fermentation, 12(3), 125. https://doi.org/10.3390/fermentation12030125






