The Production of Bioactive Hydroxytyrosol in Fermented Beverages: The Role of Must Composition and a Genetically Modified Yeast Strain
Abstract
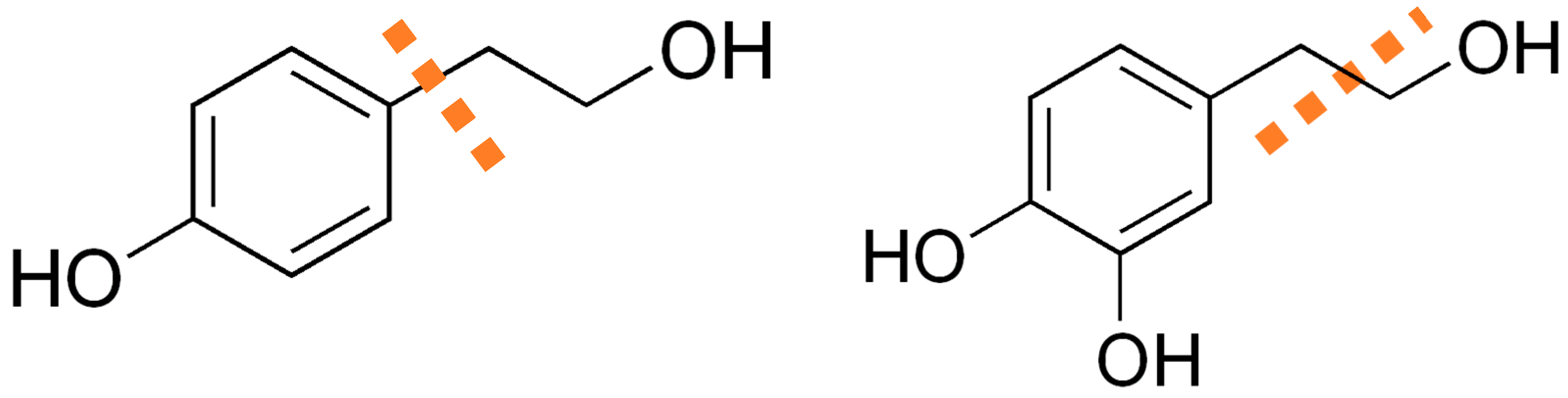
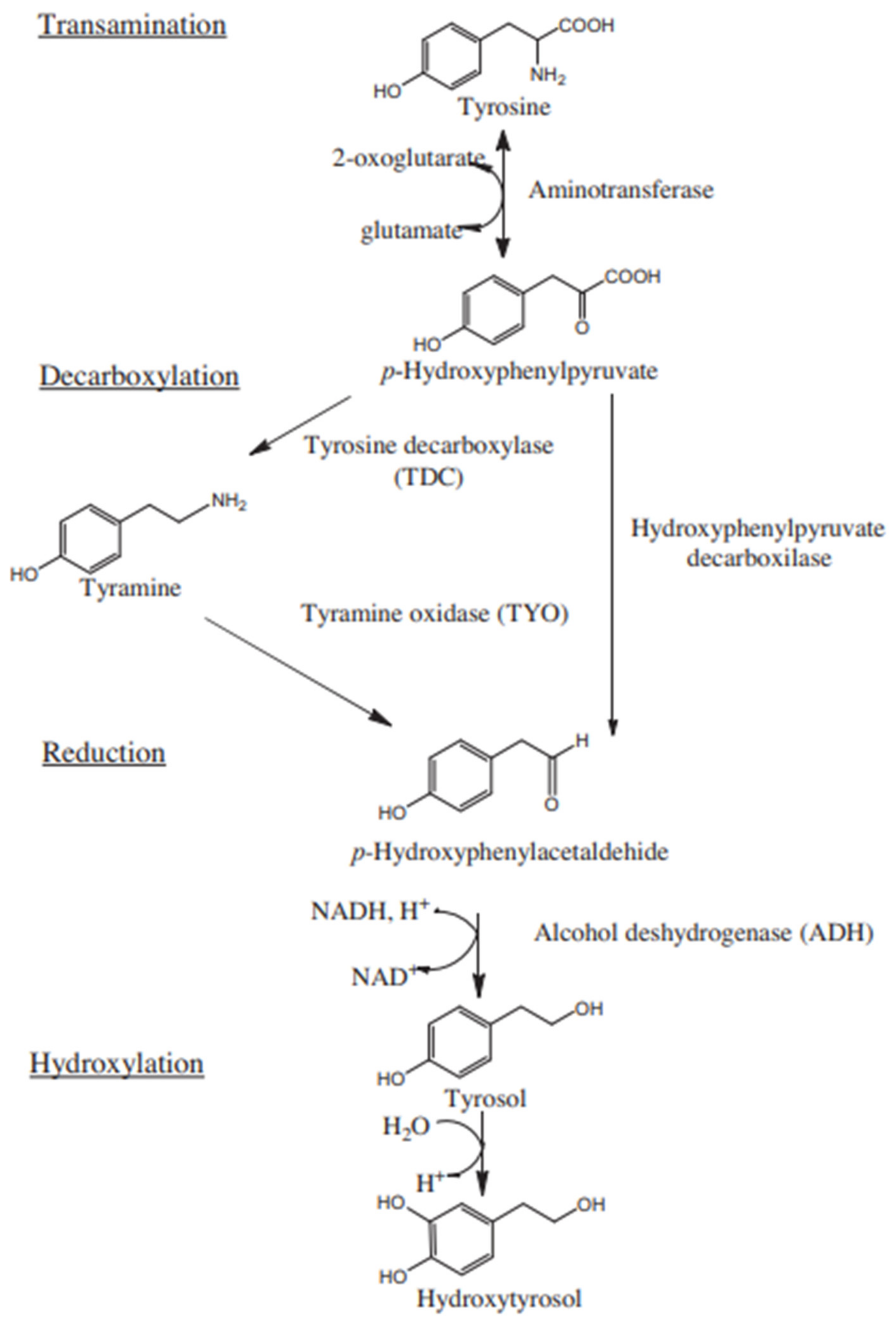
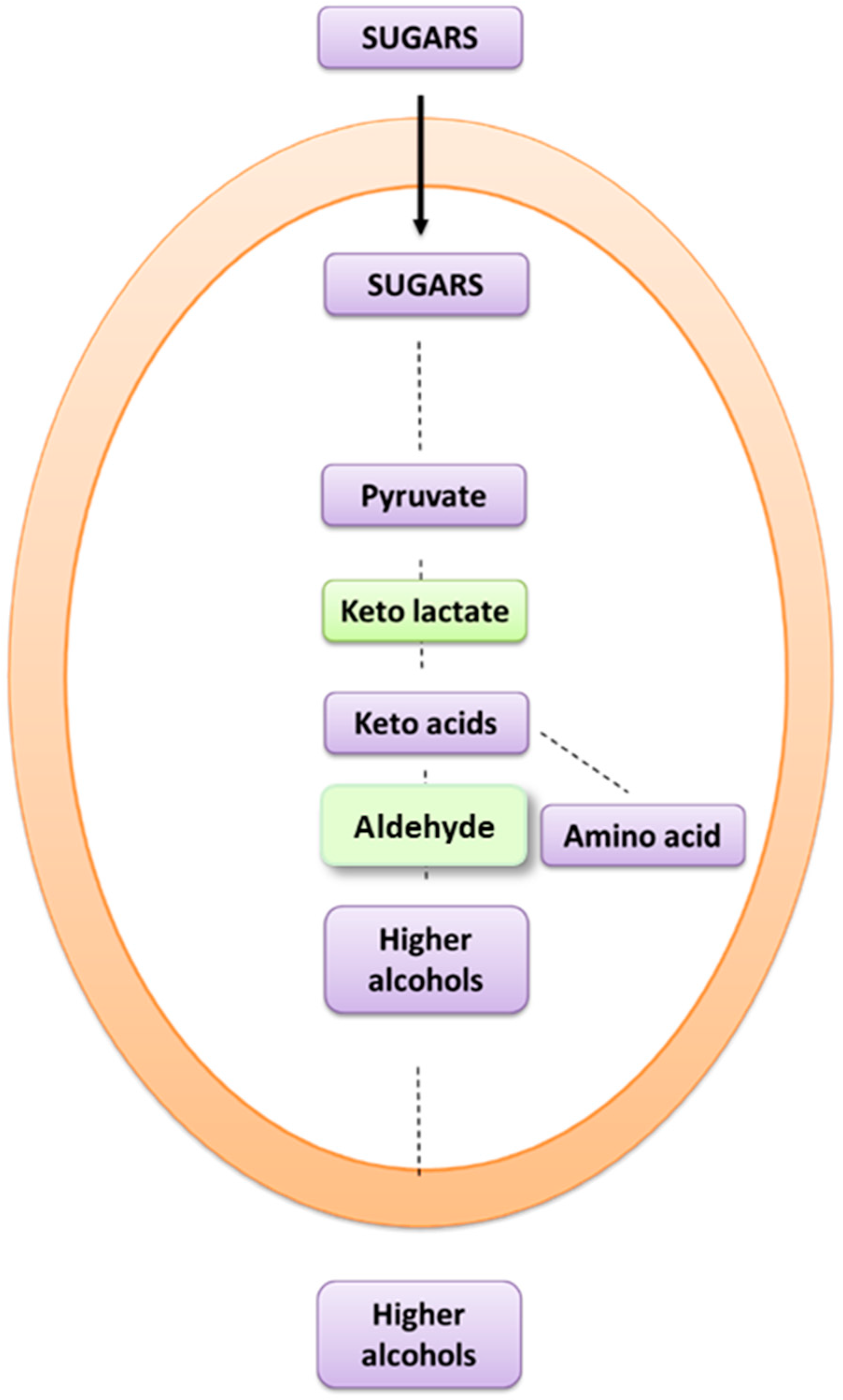
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Yeast Strains
2.3. Synthetic Must
2.4. Inoculation
2.5. Sampling
2.6. Sample Treatment
2.7. UHPLC-MS Parameters
2.8. Statistical Analysis
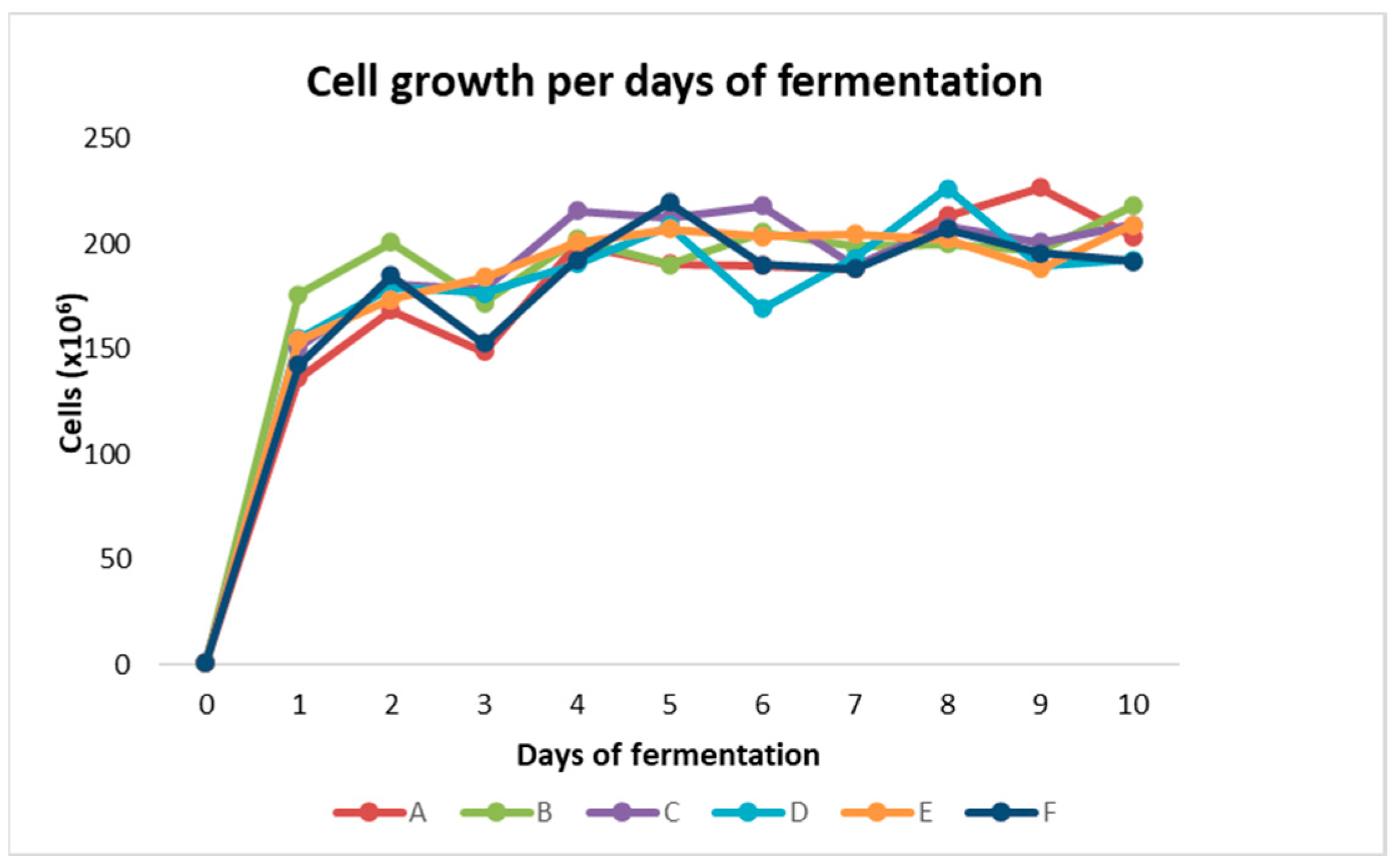
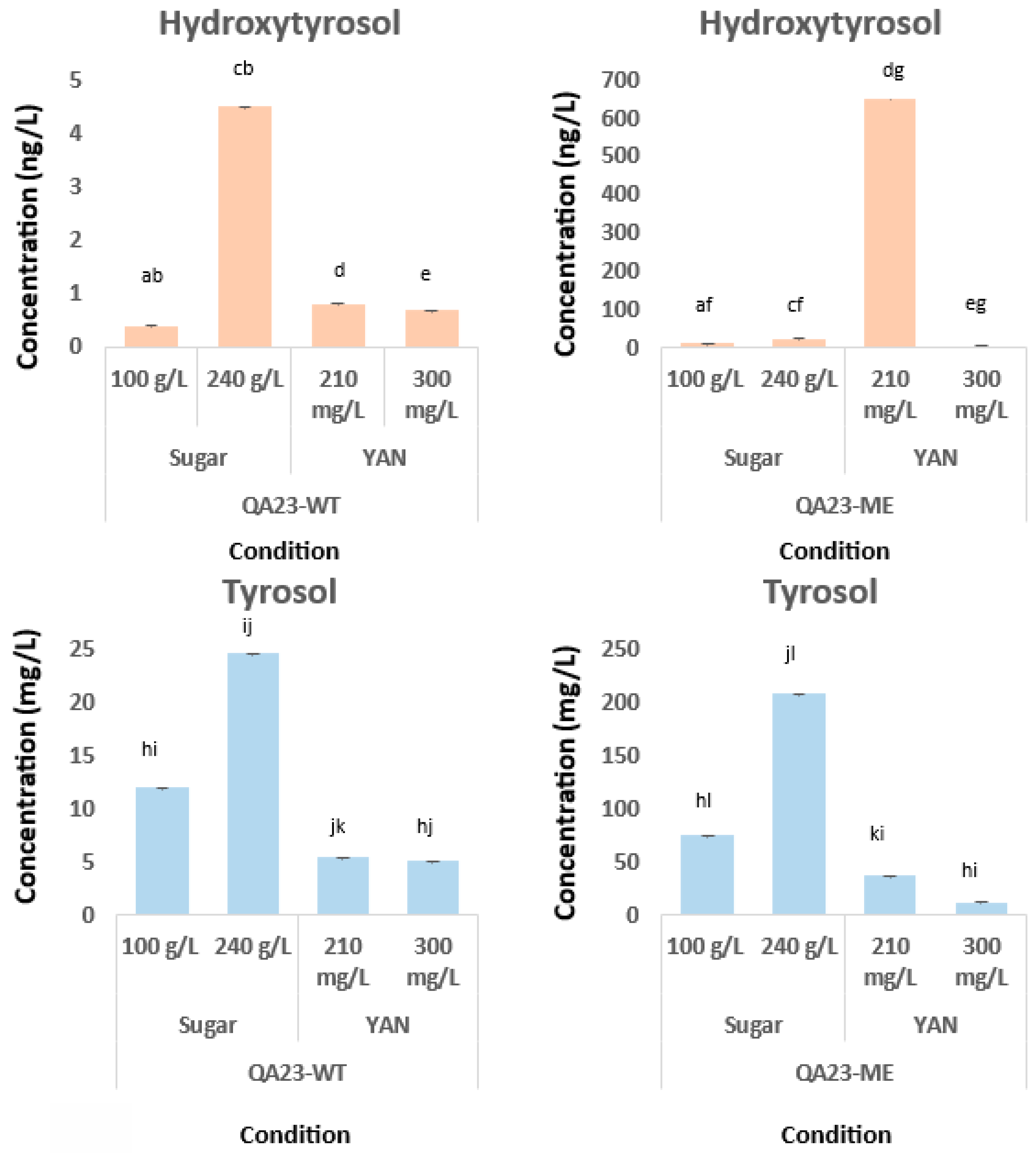
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gallardo-Fernández, M.; Gonzalez-Ramirez, M.; Cerezo, A.B.; Troncoso, A.M.; Garcia-Parrilla, M.C. Hydroxytyrosol in Foods: Analysis, Food Sources, EU Dietary Intake, and Potential Uses. Foods 2022, 11, 2355. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Fernández, M.A.; Fernández-Cruz, E.; Cantos-Villar, E.; Troncoso, A.M.; García-Parrilla, M.C. Determination of hydroxytyrosol produced by winemaking yeasts during alcoholic fermentation using a validated UHPLC-HRMS method. Food chem. 2018, 242, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Cordente, A.G.; Schmidt, S.; Beltran, G.; Torija, M.J.; Curtin, C.D. Harnessing yeast metabolism of aromatic amino acids for fermented beverage bioflavouring and bioproduction. Appl. Microbiol. Biotechnol. 2019, 103, 4325–4336. [Google Scholar] [CrossRef] [PubMed]
- Achmon, Y.; Ayelet, F. The antioxidant hydroxytyrosol: Biotechnological production challenges and opportunities. Appl. Microbiol. Biotechnol. 2015, 99, 1119–1130. [Google Scholar] [CrossRef]
- Hazelwood, L.A.; Daran, J.M.; van Maris, A.J.; Pronk, J.T.; Dickinson, J.R. The Ehrlich pathway for fusel alcohol production: A century of research on Saccharomyces cerevisiae metabolism. Appl. Environ. Microbiol. 2008, 74, 2259–2266. [Google Scholar] [CrossRef] [PubMed]
- Nisbet, M.A.; Tobias, H.J.; Brenna, J.T.; Sacks, G.L.; Mansfield, A.K. Quantifying the contribution of grape hexoses to wine volatiles by high-precision [U¹³C]-glucose tracer studies. J. Agric. Food Chem. 2014, 62, 6820–6827. [Google Scholar] [CrossRef] [PubMed]
- Gallardo-Fernández, M.; Valls-Fonayet, J.; Valero, E.; Hornedo-Ortega, R.; Richard, T.; Troncoso, A.M.; Garcia-Parrilla, M.C. Isotopic labelling-based analysis elucidates biosynthesis pathways in Saccharomyces cerevisiae for Melatonin, Serotonin and Hydroxytyrosol formation. Food Chem. 2022, 374, 131742. [Google Scholar] [CrossRef] [PubMed]
- Robles-Almazan, M.; Pulido-Moran, M.; Moreno-Fernandez, J.; Ramirez-Tortosa, C.; Rodríguez-García, C.; Quiles, J.; Ramirez-Tortosa, M. Hydroxytyrosol: Bioavailability, toxicity, and clinical applications. Food Res. Int. 2018, 105, 654–667. [Google Scholar] [CrossRef]
- de Las Hazas, M.C.L.; Rubio, L.; Macia, A.; Motilva, M.J. Hydroxytyrosol: Emerging trends in potential therapeutic applications. Curr. Pharm. Des. 2018, 24, 2157–2179. [Google Scholar] [CrossRef]
- Vilaplana-Pérez, C.; Auñón, D.; García-Flores, L.A.; Gil-Izquierdo, A. Hydroxytyrosol and potential uses in cardiovascular diseases, cancer, and AIDS. Front. Nutr. 2014, 1, 18. [Google Scholar] [CrossRef]
- González-Correa, J.A.; Navas, M.D.; Lopez-Villodres, J.A.; Trujillo, M.; Espartero, J.L.; De La Cruz, J.P. Neuroprotective effect of hydroxytyrosol and hydroxytyrosol acetate in rat brain slices subjected to hypoxia-reoxygenation. Neurosci. Lett. 2008, 446, 143–146. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific opinion on the substantiation of health claims related to polyphenols in olive and protection of LDL particles from oxidative damage (ID 1333, 1638, 1639, 1696, 2865), maintenance of normal blood HDL cholesterol concentrations (ID 1639), maintenance of normal blood pressure (ID 3781), “anti-inflammatory properties” (ID 1882), “contributes to the upper respiratory tract health” (ID 3468), “can help to maintain a normal function of gastrointestinal tract” (3779), and “contributes to body defences against external agents” (ID 3467) pursuant to Article 13(1) of Regulation (EC) No 1924/2006. EFSA J. 2011, 9, 2033. Available online: https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2011.2033 (accessed on 4 March 2022).
- Karković Marković, A.; Torić, J.; Barbarić, M.; Jakobušić Brala, C. Hydroxytyrosol, tyrosol and derivatives and their potential effects on human health. Molecules 2019, 24, 2001. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; He, X.W.; Jiang, J.G.; Xu, X.L. Hydroxytyrosol and its potential therapeutic effects. J. Agric. Food Chem. 2014, 62, 1449–1455. [Google Scholar] [CrossRef]
- Turck, D.; Bresson, J.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; Naska, A. Safety of hydroxytyrosol as a novel food pursuant to Regulation (EC) No 258/97. EFSA J. 2017, 15, 4728. [Google Scholar] [CrossRef]
- European Commission. Commission Implementing Decision (EU) 2017/2373 of 14 December 2017 authorising the placing on the market of hydroxytyrosol as a novel food ingredient under Regulation (EC) No 258/97 of the European Parliament and of the Council. Off. J. Eur. Union 2017, 337, 56–59. Available online: https://eur-lex.europa.eu/eli/dec_impl/2017/2373/oj (accessed on 5 March 2022).
- Rebollo-Romero, I.; Fernandez-Cruz, E.; Carrasco-Galán, F.; Valero, E.; Cantos-Villar, E.; Cerezo, A.; Troncoso, A.; Garcia-Parrilla, M.C. Factors influencing the production of the antioxidant hydroxytyrosol during alcoholic fermentation: Yeast strain, initial tyrosine concentration and initial must. LWT 2020, 130, 109631. [Google Scholar] [CrossRef]
- Houtman, A.C.; Du Plessis, C.S. The effect of juice clarity and several conditions promoting yeast growth or fermentation rate, the production of aroma components and wine quality. S. Afr. J. Enol. Vitic. 1981, 2, 71–81. [Google Scholar] [CrossRef]
- Houtman, A.C.; Marais, J.; Du Plessis, C.S. The possibilities of applying present-day knowledge of wine aroma components: Influence of several juice factors on fermentation rate and ester production during fermentation. S. Afr. J. Enol. Vitic. 1980, 1, 27–33. [Google Scholar] [CrossRef][Green Version]
- Muñiz-Calvo, S.; Bisquert, R.; Puig, S.; Guillamón, J.M. Overproduction of hydroxytyrosol in Saccharomyces cerevisiae by heterologous overexpression of the Escherichia coli 4-hydroxyphenylacetate 3-monooxygenase. Food Chem. 2020, 308, 125646. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, H.; Hu, H.; Ng, K.R.; Yang, R.; Lyu, X. De novo production of hydroxytyrosol by metabolic engineering of Saccharomyces cerevisiae. J. Agric. Food Chem. 2022, 70, 7490–7499. [Google Scholar] [CrossRef]
- Sonni, F.; Chinnici, F.; Natali, N.; Riponi, C. Pre-fermentative replacement of sulphur dioxide by lysozyme and oenological tannins: Effect on the formation and evolution of volatile compounds during the bottle storage of white wines. Food Chem. 2011, 129, 1193–1200. [Google Scholar] [CrossRef]
- Raposo, R.; Ruiz-Moreno, M.J.; Garde-Cerdán, T.; Puertas, B.; Moreno-Rojas, J.M.; Gonzalo-Diago, A.; Guerrero, R.; Ortíz, V.; Cantos-Villar, E. Grapevine-shoot stilbene extract as a preservative in red wine. Food Chem. 2016, 197 Pt B, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Raposo, R.; Ruiz-Moreno, M.J.; Garde-Cerdán, T.; Puertas, B.; Moreno-Rojas, J.M.; Gonzalo-Diago, A.; Guerrero, R.F.; Ortiz, V.; Cantos-Villar, E. Effect of hydroxytyrosol on quality of sulfur dioxide-free red wine. Food Chem. 2016, 192, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Riou, C.; Nicaud, J.M.; Barre, P.; Gaillardin, C. Stationary-phase gene expression in Saccharomyces cerevisiae during wine fermentation. Yeast 1997, 13, 903–915. [Google Scholar] [CrossRef]
- Neveu, V.; Perez-Jimenez, J.; Vos, F.; Crespy, V.; Du Chaffaut, L.; Mennen, L. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database J. Biol. Databases Curation 2010, 2010, bap024. [Google Scholar] [CrossRef] [PubMed]
- Vilela, A. Use of nonconventional yeasts for modulating wine acidity. Fermentation 2019, 5, 27. [Google Scholar] [CrossRef]
- Bordiga, M.; Lorenzo, C.; Pardo, F.; Salinas, M.R.; Travaglia, F.; Arlorio, M.; Coïsson, J.D.; Garde-Cerdán, T. Factors influencing the formation of histaminol, hydroxytyrosol, tyrosol, and tryptophol in wine: Temperature, alcoholic degree, and amino acids concentration. Food Chem. 2016, 197 Pt B, 1038–1045. [Google Scholar] [CrossRef]
- Wuster, A.; Babu, M.M. Transcriptional control of the quorum sensing response in yeast. Mol. BioSystem 2009, 6, 134–141. [Google Scholar] [CrossRef]
- Crépin, L.; Nidelet, T.; Sanchez, I.; Dequin, S.; Camarasa, C. Sequential use of nitrogen compounds by Saccharomyces cerevisiae during wine fermentation: A model based on kinetic and regulation characteristics of nitrogen permeases. Appl. Environ. Microbiol. 2012, 78, 8102–8111. [Google Scholar] [CrossRef]
- Vendramini, C.; Beltran, G.; Nadai, C.; Giacomini, A.; Mas, A.; Corich, V. The role of nitrogen uptake on the competition ability of three vineyard Saccharomyces cerevisiae strains. Int. J. Food Microbiol. 2017, 258, 1–11. [Google Scholar] [CrossRef]
- Vilanova, M.; Ugliano, M.; Varela, C.; Siebert, T.; Pretorius, I.S.; Henschke, P.A. Assimilable nitrogen utilisation and production of volatile and non-volatile compounds in chemically defined medium by Saccharomyces cerevisiae wine yeasts. Appl. Microbiol. Biotechnol. 2007, 77, 145–157. [Google Scholar] [CrossRef]
- González, B.; Vázquez, J.; Morcillo-Parra, M.A.; Mas, A.; Torija, M.J.; Beltran, G. The production of aromatic alcohols in non-Saccharomyces wine yeast is modulated by nutrient availability. Food Microbiol. 2018, 74, 64–74. [Google Scholar] [CrossRef]
- Beltran, G.; Esteve-Zarzoso, B.; Rozès, N.; Mas, A.; Guillamón, J.M. Influence of the timing of nitrogen additions during synthetic grape must fermentations on fermentation kinetics and nitrogen consumption. J. Agric. Food. Chem. 2005, 53, 996–1002. [Google Scholar] [CrossRef]
- Carrau, F.M.; Medina, K.; Farina, L.; Boido, E.; Henschke, P.A.; Dellacassa, E. Production of fermentation aroma compounds by Saccharomyces cerevisiae wine yeasts: Effects of yeast assimilable nitrogen on two model strains. FEMS Yeast Res. 2008, 8, 1196–1207. [Google Scholar] [CrossRef]

| Yeast | Sugar (g/L) | YAN (mg/L) |
|---|---|---|
| QA23-WT | 200 | 210 |
| 200 | 300 | |
| 100 | 210 | |
| 240 | 210 | |
| QA23-ME | 200 | 210 |
| 200 | 300 | |
| 100 | 210 | |
| 240 | 210 |
| 210 mg/L YAN | 300 mg/L YAN | |||||||
|---|---|---|---|---|---|---|---|---|
| QA23-WT | QA23-ME | QA23WT | QA23 ME | |||||
| Day | Hydroxytyrosol | Tyrosol | Hydroxytyrosol | Tyrosol | Hydroxytyrosol | Tyrosol | Hydroxytyrosol | Tyrosol |
| (ng/mL) | (mg/L) | (ng/mL) | (mg/L) | (ng/mL) | (mg/L) | (ng/mL) | (mg/L) | |
| D1 | 0.16 ± 0.00 | 0.02 ± 0.01 | 0.49 ± 0.00 | 0.07 ± 0.03 | 0.20 ± 0.00 | 1.26 ± 0.11 | 5.15 ± 0.00 | 5.9 ± 0.4 |
| D2 | 0.11 ± 0.00 | 0.03 ± 0.02 | 0.78 ± 0.00 | 0.19 ± 0.05 | 0.13 ± 0.05 | 3.56 ± 0.17 | 2.7 ± 1.5 | 12.1 ± 0.6 eg |
| D3 | 0.17 ± 0.00 | 0.04 ± 0.02 | 1.17 ± 0.00 | 0.20 ± 0.10 | 0.7 ± 0.3 b | 3.95 ± 0.13 | 5.8 ± 0.4 bc | 10.9 ± 0.5 |
| D4 | 0.04 ± 0.00 | 0.04 ± 0.02 | 1.30 ± 0.00 | 0.26 ± 0.08 | 0.4 ± 0.4 | 4.32 ± 0.21 | 1.7 ± 0.6 | 11.1 ± 0.8 |
| D5 | 0.04 ± 0.00 | 0.03 ± 0.00 | 1.93 ± 0.12 | 27.0 ± 2.2 | 0.50 ± 0.24 | 5.1 ± 0.4 ef | 1.7 ± 0.6 | 10.8 ± 0.8 |
| D6 | 0.04 ± 0.00 | 0.02 ± 0.01 | 438.18 ± 0.15 | 27.9 ± 1.7 | 0.56 ± 0.25 | 5.06 ± 0.21 | 1.3 ± 0.7 | 8.8 ± 0.5 |
| D7 | 0.80 ± 0.00 | 4.24 ± 0.13 | 490.78 ± 0.18 | 27.6 ± 2.1 | 0.50 ± 0.11 | 5.0 ± 0.5 | 2.4 ± 0.8 | 7.8 ± 0.8 |
| D8 | 0.81 ± 0.00 a | 4.02 ± 0.18 | 509.62 ± 0.01 | 28.2 ± 3.1 | 0.29 ± 0.18 | 4.7 ± 0.4 | 1.4 ± 0.6 | 8.6 ± 0.9 |
| D9 | 0.70 ± 0.00 | 4.1 ± 0.4 | 561.87 ± 0.01 | 32.2 ± 3.8 | 0.37 ± 0.11 | 4.7 ± 0.3 | 0.8 ± 0.7 | 8.8 ± 0.9 |
| D10 | 0.51 ± 0.01 | 5.4 ± 0.7 df | 651.77 ± 0.01 ac | 37.4 ± 3.9 dg | 0.32 ± 0.00 | 4.3 ± 0.4 | 0.6 ± 0.3 | 9.3 ± 2.5 |
| 100 g/L Sugar | 240 g/L Sugar | |||||||
|---|---|---|---|---|---|---|---|---|
| QA23 WT | QA23 ME | QA23 WT | QA23 ME | |||||
| Day | Hydroxytyrosol | Tyrosol | Hydroxytyrosol | Tyrosol | Hydroxytyrosol | Tyrosol | Hydroxytyrosol | Tyrosol |
| (ng/mL) | (mg/L) | (ng/mL) | (mg/L) | (ng/mL) | (mg/L) | (ng/mL) | (mg/L) | |
| D1 | 0.26 ± 0.03 | 2.4 ± 0.5 | 7.55 ± 2.04 | 26.5 ± 2.2 | 0.16 ± 0.03 | 0.35 ± 0.07 | 1.1 ± 0.3 | 10.5 ± 2.5 |
| D2 | 0.35 ± 0.04 | 10.9 ± 2.1 | 12.6 ± 2.6 ad | 72.2 ± 9.5 | 0.33 ± 0.01 | 2.4 ± 0.3 | 1.9 ± 0.5 | 50.4 ± 15.7 |
| D3 | 0.41 ± 0.05 | 12.0 ± 0.5 eg | 8.0 ± 2.6 | 74.5 ± 9.2 eh | 3.8 ± 0.4 | 19.3 ± 0.9 | 23.4 ± 2.1 bd | 129.9 ± 5.7 |
| D4 | 0.35 ± 0.02 | 11.6 ± 1.2 | 2.8 ± 0.5 | 63.8 ± 6.1 | 2.55 ± 0.17 | 20.3 ± 4.2 | 22.2 ± 2.7 | 149.9 ± 15.5 |
| D5 | 0.41 ± 0.07 ac | 12.1 ± 0.5 | 9.4 ± 1.5 | 68.7 ± 9.5 | 4.0 ± 0.5 | 24.59 ± 1.9 fg | 16.8 ± 1.5 | 207.4 ± 6.5 fh |
| D6 | 0.37 ± 0.02 | 10.9 ± 0.4 | 7.3 ± 2.5 | 65.6 ± 10.5 | 2.8 ± 0.3 | 14.94 ± 1.08 | 7.2 ± 1.5 | 77.15 ± 0.34 |
| D7 | 0.41 ± 0.03 | 10.9 ± 1.1 | 4.7 ± 1.6 | 68.7 ± 5.4 | 3.2 ± 0.3 | 14.9 ± 0.8 | 15.1 ± 2.9 | 68.1 ± 7.9 |
| D8 | 3.6 ± 0.3 | 15.2 ± 0.6 | 20.9 ± 3.7 | 72.7 ± 2.5 | ||||
| D9 | 3.3 ± 0.9 | 17.5 ± 1.1 | 6.3 ± 2.1 | 102.9 ± 32.9 | ||||
| D10 | 4.5 ± 1.7 bc | 16.3 ± 1.6 | 2.31 ± 0.22 | 81.6 ± 8.7 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonzalez-Ramirez, M.; Gallardo-Fernandez, M.; Cerezo, A.B.; Bisquert, R.; Valero, E.; Troncoso, A.M.; Garcia-Parrilla, M.C. The Production of Bioactive Hydroxytyrosol in Fermented Beverages: The Role of Must Composition and a Genetically Modified Yeast Strain. Fermentation 2024, 10, 198. https://doi.org/10.3390/fermentation10040198
Gonzalez-Ramirez M, Gallardo-Fernandez M, Cerezo AB, Bisquert R, Valero E, Troncoso AM, Garcia-Parrilla MC. The Production of Bioactive Hydroxytyrosol in Fermented Beverages: The Role of Must Composition and a Genetically Modified Yeast Strain. Fermentation. 2024; 10(4):198. https://doi.org/10.3390/fermentation10040198
Chicago/Turabian StyleGonzalez-Ramirez, Marina, Marta Gallardo-Fernandez, Ana B. Cerezo, Ricardo Bisquert, Eva Valero, Ana M. Troncoso, and M. Carmen Garcia-Parrilla. 2024. "The Production of Bioactive Hydroxytyrosol in Fermented Beverages: The Role of Must Composition and a Genetically Modified Yeast Strain" Fermentation 10, no. 4: 198. https://doi.org/10.3390/fermentation10040198
APA StyleGonzalez-Ramirez, M., Gallardo-Fernandez, M., Cerezo, A. B., Bisquert, R., Valero, E., Troncoso, A. M., & Garcia-Parrilla, M. C. (2024). The Production of Bioactive Hydroxytyrosol in Fermented Beverages: The Role of Must Composition and a Genetically Modified Yeast Strain. Fermentation, 10(4), 198. https://doi.org/10.3390/fermentation10040198






