An Overview of the Recent Progress in the Synthesis and Applications of Carbon Nanotubes
Abstract
1. Introduction
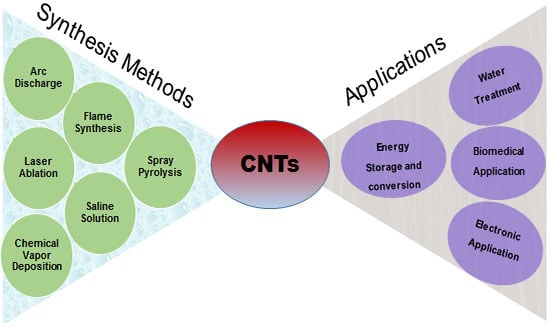
2. Synthesis of CNTs
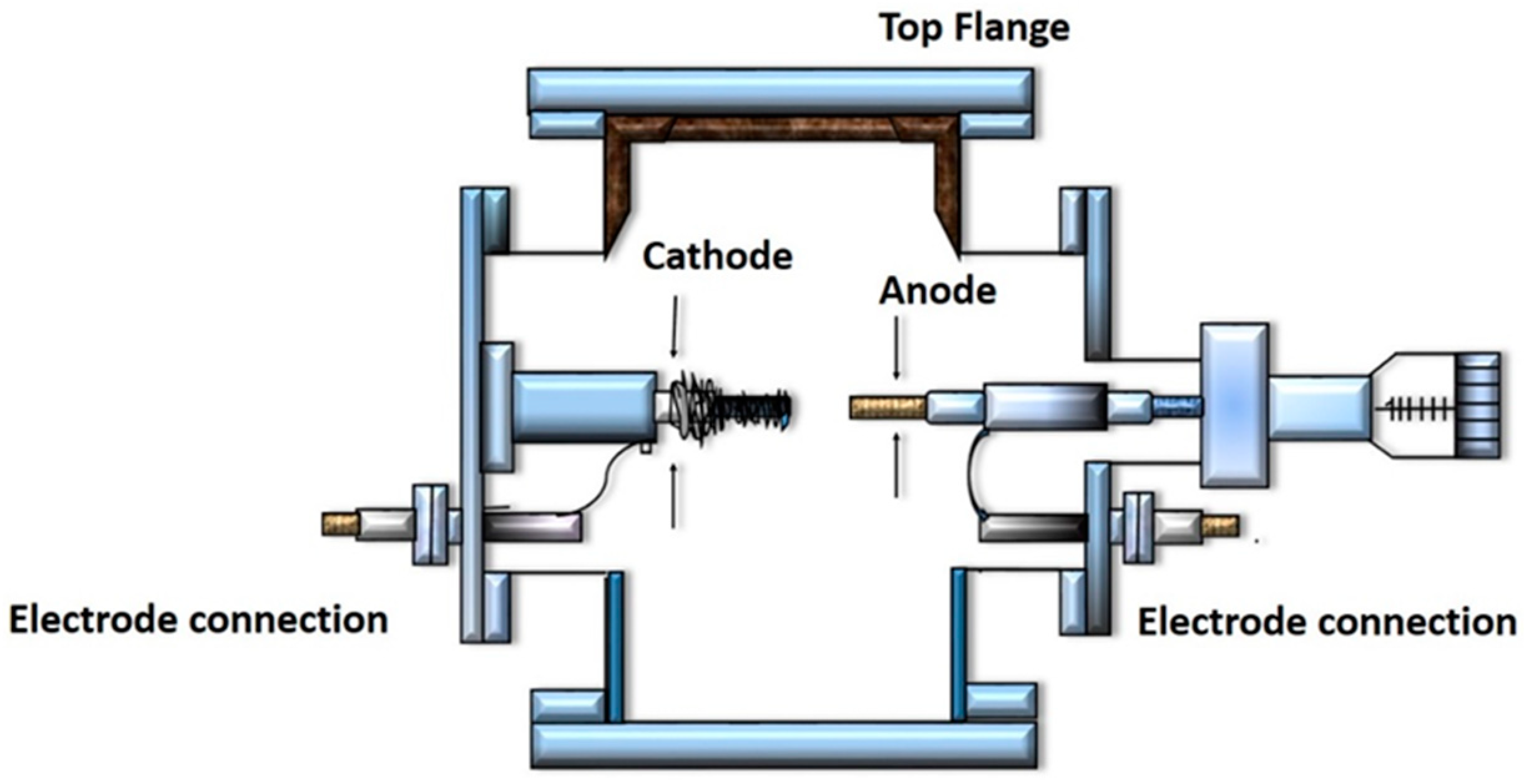
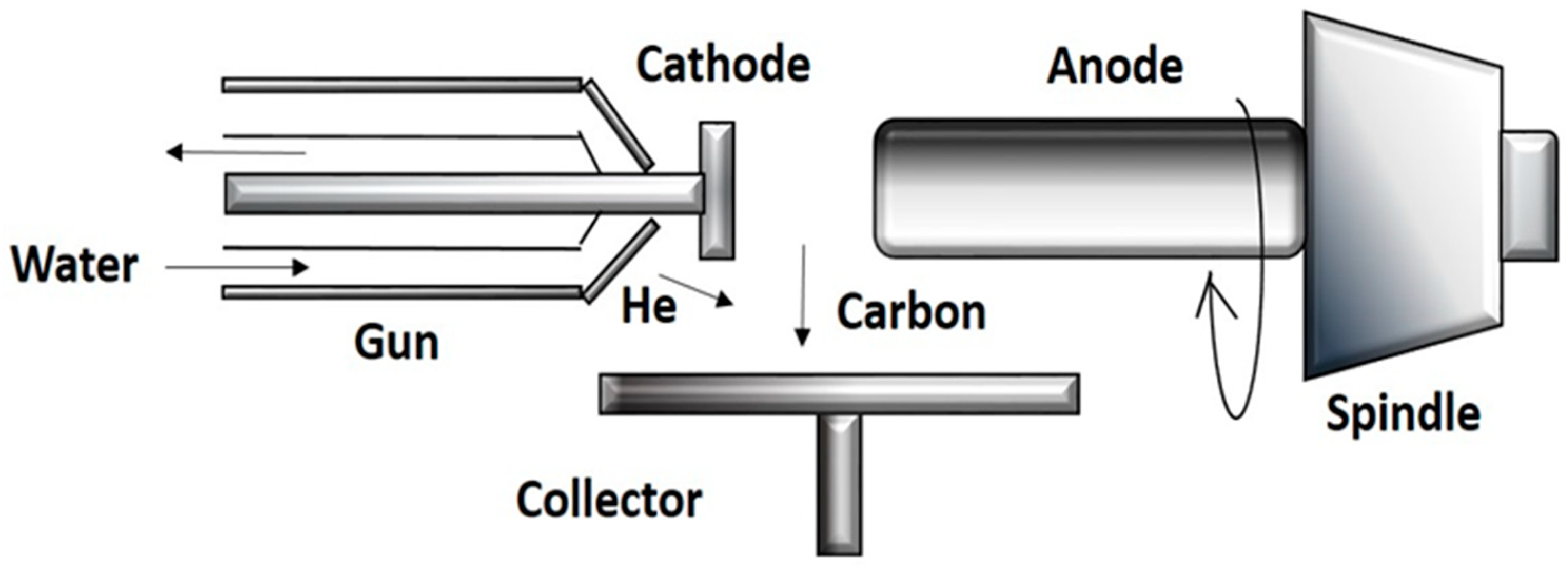
2.1. Arc Discharge Method
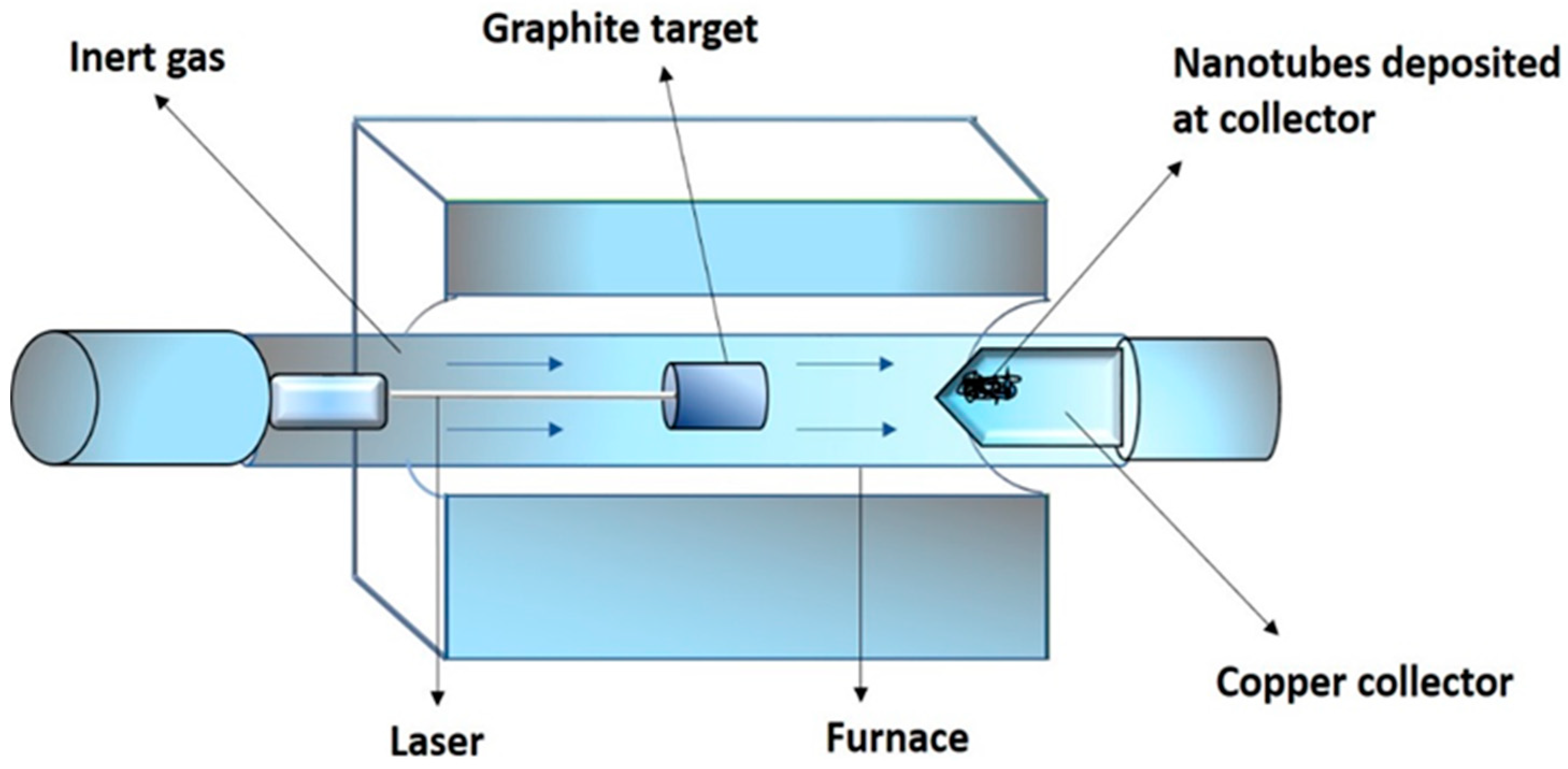
2.2. Laser Ablation Method
2.3. Chemical Vapor Deposition (CVD)
2.4. Flame Synthesis Method
2.5. Saline Solution Method
2.6. Spray Pyrolysis Method
3. Characterization Techniques for CNTs
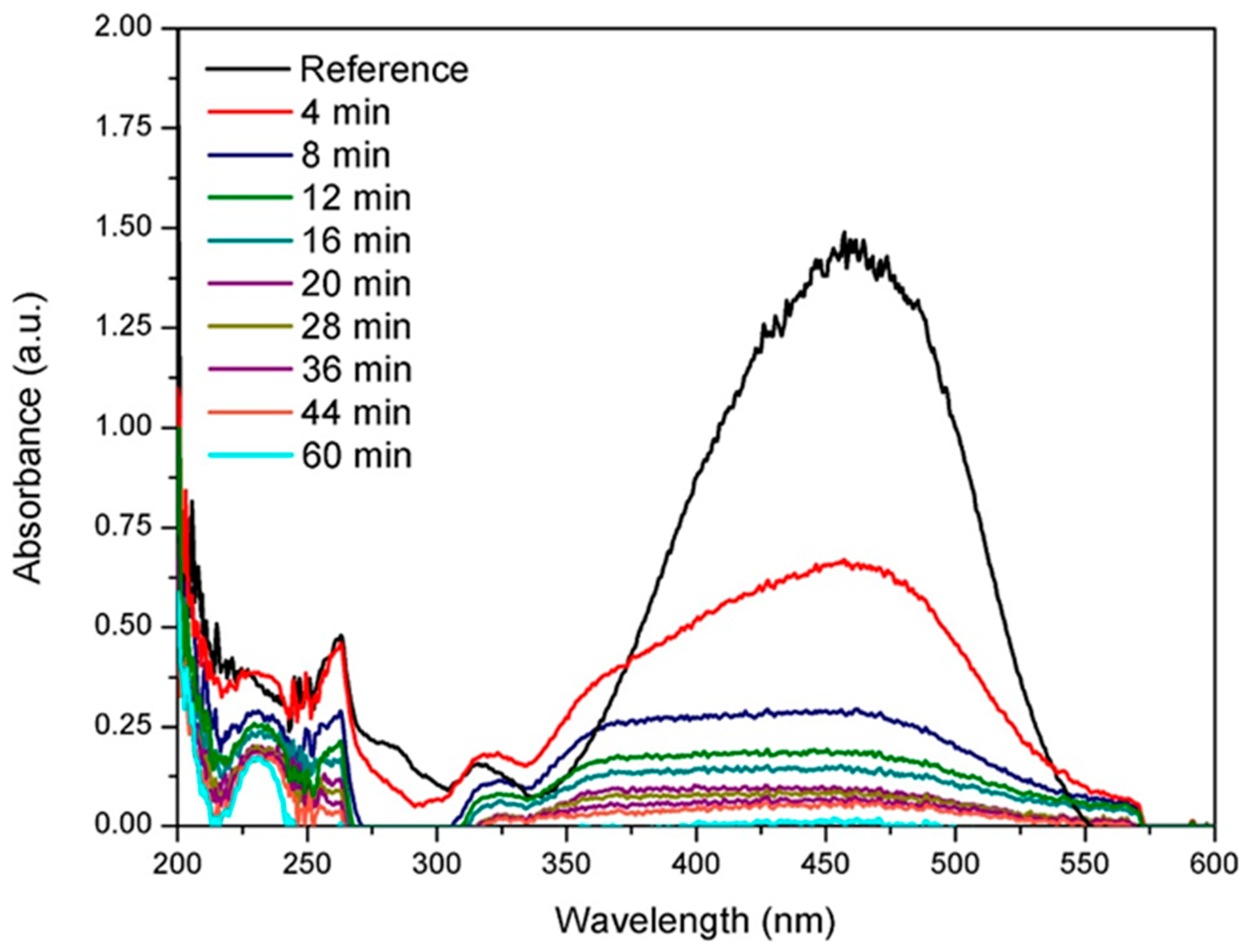
3.1. Raman Spectroscopy
3.2. Transmission Electron Microscopy
3.3. Atomic Force Microscopy
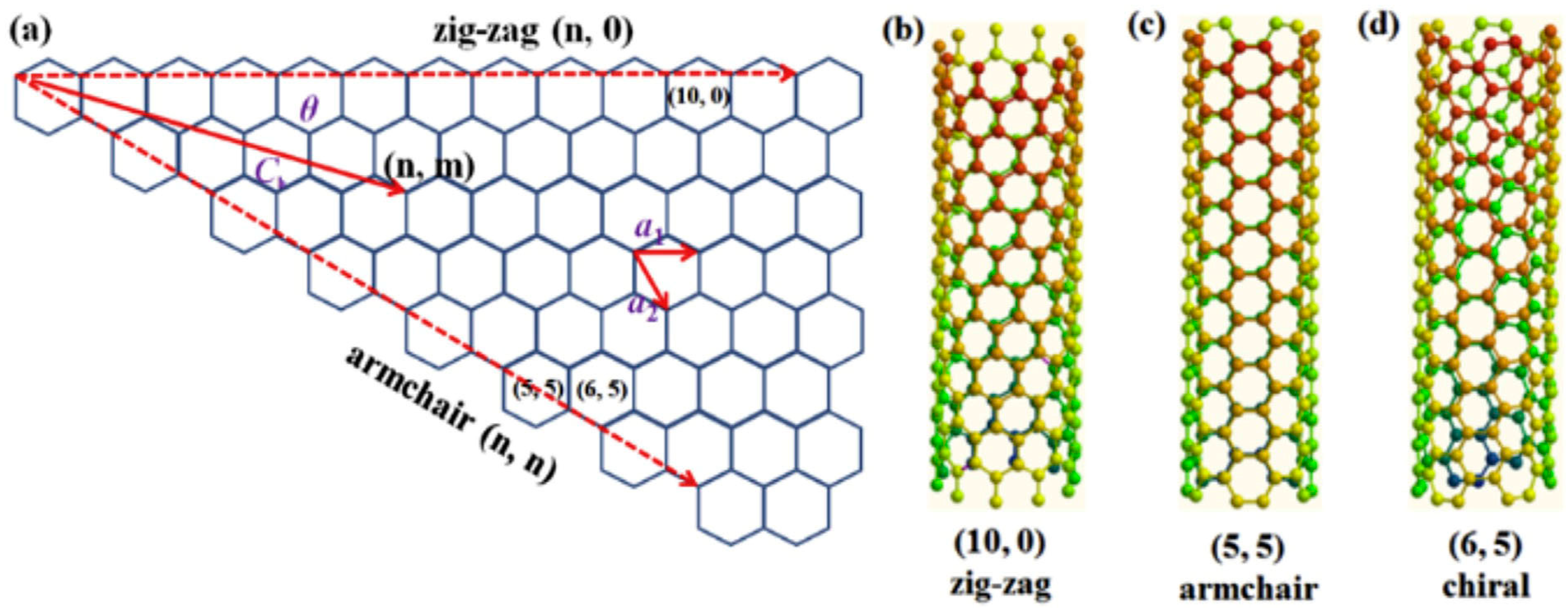
4. Shapes of CNTs
5. Applications of CNTs
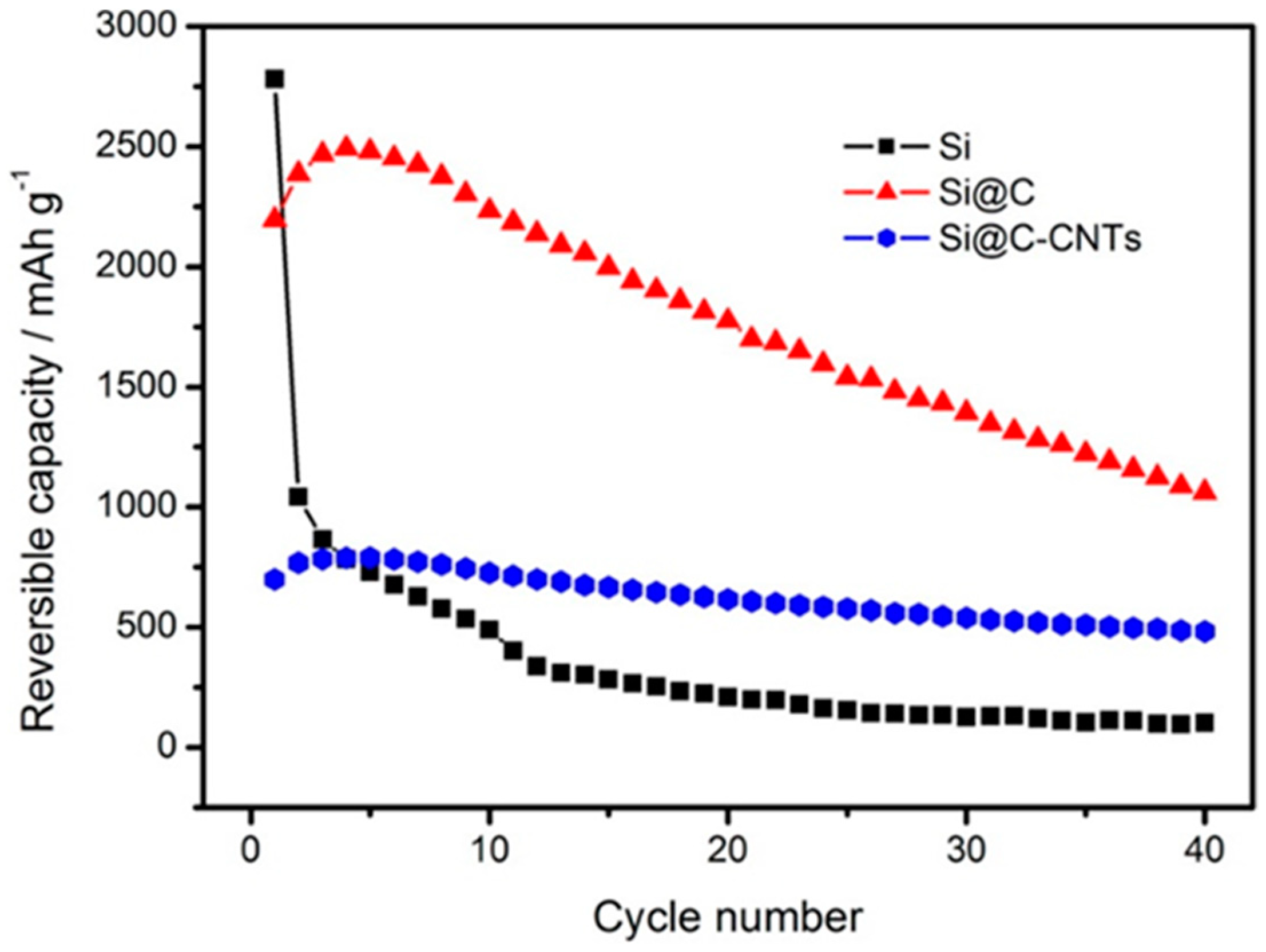
5.1. Energy Storage and Conversion
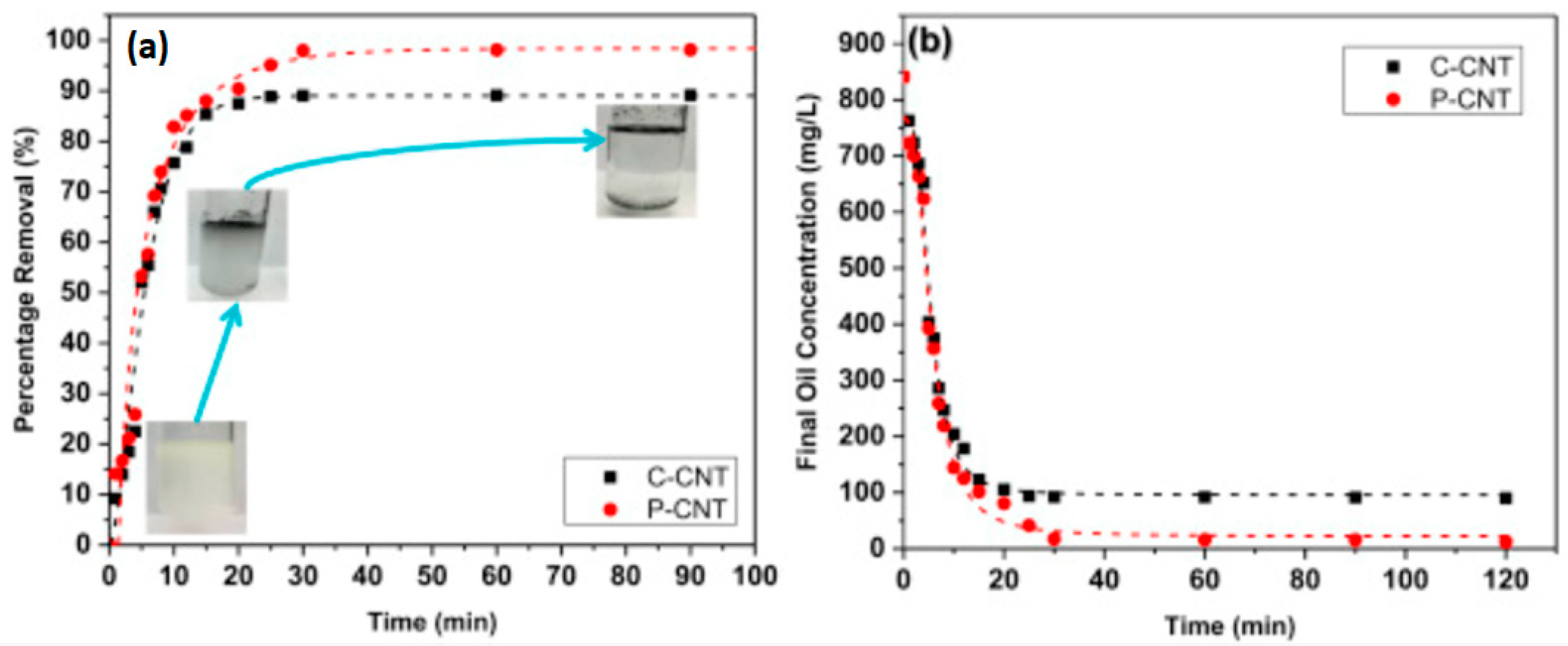
5.2. Water Treatment
5.3. Biomedical Applications
5.4. Electronic Applications
5.4.1. Carbon Nanotube-Based Diodes, Field-Effect Transistors, and Logic Circuits
5.4.2. CNT-Based Sensors
5.4.3. Field Emission
5.4.4. Transparent Electrodes
5.4.5. CNTs as Probes in Atomic Force Microscopy
5.5. Buckypaper
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Coville, N.J.; Mhlanga, S.D.; Nxumalo, E.N.; Shaikjee, A. A review of shaped carbon nanomaterials. S. Afr. J. Sci. 2011, 107, 1–15. [Google Scholar] [CrossRef]
- Rius, G. Technologies of Carbon Materials. Syntheses and Preparations. In Carbon for Sensing Devices; Springer: Cham, Switzerland, 2015; pp. 15–42. [Google Scholar]
- Lee, J.; Kim, T.; Jung, Y.; Jung, K.; Park, J.; Lee, D.-M.; Jeong, H.S.; Hwang, J.Y.; Park, C.R.; Lee, K.-H. High-strength carbon nanotube/carbon composite fibers via chemical vapor infiltration. Nanoscale 2016, 8, 18972–18979. [Google Scholar] [CrossRef] [PubMed]
- Chua, M.; Chui, C.-K.; Chng, C.-B.; Lau, D. Carbon nanotube-based artificial tracheal prosthesis: Carbon nanocomposite implants for patient-specific ENT care. IEEE Nanotechnol. Mag. 2013, 7, 27–31. [Google Scholar] [CrossRef]
- Ketabi, S.; Rahmani, L. Carbon nanotube as a carrier in drug delivery system for carnosine dipeptide: A computer simulation study. Mater. Sci. Eng. C 2017, 73, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Arunachalam, S.; Gupta, A.A.; Izquierdo, R.; Nabki, F. Suspended Carbon Nanotubes for Humidity Sensing. Sensors 2018, 18, 1655. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Nehra, M.; Kedia, D.; Dilbaghi, N.; Tankeshwar, K.; Kim, K.-H. Carbon nanotubes: A potential material for energy conversion and storage. Prog. Energy Combust. Sci. 2018, 64, 219–253. [Google Scholar] [CrossRef]
- Puett, C.; Inscoe, C.; Hartman, A.; Calliste, J.; Franceschi, D.K.; Lu, J.; Zhou, O.; Lee, Y.Z. An update on carbon nanotube-enabled X-ray sources for biomedical imaging. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2018, 10, e1475. [Google Scholar] [CrossRef]
- Sun, Y.; Yun, K.N.; Leti, G.; Lee, S.H.; Song, Y.-H.; Lee, C.J. High-performance field emission of carbon nanotube paste emitters fabricated using graphite nanopowder filler. Nanotechnology 2017, 28, 065201. [Google Scholar] [CrossRef]
- Zhao, T.; Ji, X.; Jin, W.; Yang, W.; Li, T. Hydrogen storage capacity of single-walled carbon nanotube prepared by a modified arc discharge. Fuller. Nanotubes Carbon Nanostruct. 2017, 25, 355–358. [Google Scholar] [CrossRef]
- Xu, J.-L.; Dai, R.-X.; Xin, Y.; Sun, Y.-L.; Li, X.; Yu, Y.-X.; Xiang, L.; Xie, D.; Wang, S.-D.; Ren, T.-L. Efficient and reversible electron doping of semiconductor-enriched single-walled carbon nanotubes by using decamethylcobaltocene. Sci. Rep. 2017, 7, 6751. [Google Scholar] [CrossRef]
- Choi, J.; Park, B.C.; Ahn, S.J.; Kim, D.-H.; Lyou, J.; Dixson, R.G.; Orji, N.G.; Fu, J.; Vorburger, T.V. Evaluation of carbon nanotube probes in critical dimension atomic force microscopes. J. Micro/Nanolithogr. MEMS MOEMS 2016, 15, 034005. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Shan, B.; Yang, Y.; Yuan, G.; Huang, S.; Lu, X.; Zhang, Y.; Fu, Y.; Ye, L.; Liu, J. An overview of carbon nanotubes based interconnects for microelectronic packaging. In Proceedings of the 2017 IMAPS Nordic Conference on Microelectronics Packaging (NordPac), Gothenburg, Sweden, 18–20 June 2017; pp. 113–119. [Google Scholar]
- Lukyanovich, V. The structure of carbon forming in thermal decomposition of carbon monoxide on an iron catalyst. Sov. J. Phys. Chem. 1952, 26, 88–95. [Google Scholar]
- Oberlin, A.; Endo, M.; Koyama, T. Filamentous growth of carbon through benzene decomposition. J. Cryst. Growth 1976, 32, 335–349. [Google Scholar] [CrossRef]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- Dresselhaus, M.; Dresselhaus, G.; Saito, R. Physics of carbon nanotubes. Carbon 1995, 33, 883–891. [Google Scholar] [CrossRef]
- Saito, R.; Dresselhaus, G.; Dresselhaus, M.S. Physical Properties of Carbon Nanotubes; World Scientific: Singapore, 1998; Vol. 35. [Google Scholar]
- Zhang, F.; Hou, P.-X.; Liu, C.; Cheng, H.-M. Epitaxial growth of single-wall carbon nanotubes. Carbon 2016, 102, 181–197. [Google Scholar] [CrossRef]
- Ding, R.; Lu, G.; Yan, Z.; Wilson, M. Recent advances in the preparation and utilization of carbon nanotubes for hydrogen storage. J. Nanosci. Nanotechnol. 2001, 1, 7–29. [Google Scholar] [CrossRef]
- Tang, Z.; Zhang, L.; Wang, N.; Zhang, X.; Wen, G.; Li, G.; Wang, J.; Chan, C.; Sheng, P. Superconductivity in 4 angstrom single-walled carbon nanotubes. Science 2001, 292, 2462–2465. [Google Scholar] [CrossRef]
- Al Moustafa, A.-E.; Mfoumou, E.; Roman, D.E.; Nerguizian, V.; Alazzam, A.; Stiharu, I.; Yasmeen, A. Impact of single-walled carbon nanotubes on the embryo: A brief review. Int. J. Nanomed. 2016, 11, 349. [Google Scholar] [CrossRef]
- Iijima, S.; Ichihashi, T. Single-shell carbon nanotubes of 1-nm diameter. Nature 1993, 363, 603. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, L.; Liu, J.; Fang, X.; Zhang, Z. Effect of morphology of carbon nanomaterials on thermo-physical characteristics, optical properties and photo-thermal conversion performance of nanofluids. Renew. Energy 2016, 99, 888–897. [Google Scholar] [CrossRef]
- Eatemadi, A.; Daraee, H.; Karimkhanloo, H.; Kouhi, M.; Zarghami, N.; Akbarzadeh, A.; Abasi, M.; Hanifehpour, Y.; Joo, S.W. Carbon nanotubes: Properties, synthesis, purification, and medical applications. Nanoscale Res. Lett. 2014, 9, 393. [Google Scholar] [CrossRef] [PubMed]
- Hirlekar, R.; Yamagar, M.; Garse, H.; Vij, M.; Kadam, V. Carbon nanotubes and its applications: A review. Asian J. Pharm. Clin. Res. 2009, 2, 17–27. [Google Scholar]
- Hou, P.-X.; Liu, C.; Cheng, H.-M. Purification of carbon nanotubes. Carbon 2008, 46, 2003–2025. [Google Scholar] [CrossRef]
- Wang, H.; Chhowalla, M.; Sano, N.; Jia, S.; Amaratunga, G. Large-scale synthesis of single-walled carbon nanohorns by submerged arc. Nanotechnology 2004, 15, 546. [Google Scholar] [CrossRef]
- Anazawa, K.; Shimotani, K.; Manabe, C.; Watanabe, H.; Shimizu, M. High-purity carbon nanotubes synthesis method by an arc discharging in magnetic field. Appl. Phys. Lett. 2002, 81, 739–741. [Google Scholar] [CrossRef]
- José-Yacamán, M.; Miki-Yoshida, M.; Rendon, L.; Santiesteban, J. Catalytic growth of carbon microtubules with fullerene structure. Appl. Phys. Lett. 1993, 62, 202–204. [Google Scholar] [CrossRef]
- Thess, A.; Lee, R.; Nikolaev, P.; Dai, H. Crystalline ropes of metallic carbon nanotubes. Science 1996, 273, 483. [Google Scholar] [CrossRef]
- Seo, J.W.; Magrez, A.; Milas, M.; Lee, K.; Lukovac, V.; Forro, L. Catalytically grown carbon nanotubes: From synthesis to toxicity. J. Phys. D Appl. Phys. 2007, 40, R109. [Google Scholar] [CrossRef]
- Li, Y.; Mann, D.; Rolandi, M.; Kim, W.; Ural, A.; Hung, S.; Javey, A.; Cao, J.; Wang, D.; Yenilmez, E. Preferential growth of semiconducting single-walled carbon nanotubes by a plasma enhanced CVD method. Nano Lett. 2004, 4, 317–321. [Google Scholar] [CrossRef]
- Ebbesen, T.; Ajayan, P. Large-scale synthesis of carbon nanotubes. Nature 1992, 358, 220. [Google Scholar] [CrossRef]
- Szabó, A.; Perri, C.; Csató, A.; Giordano, G.; Vuono, D.; Nagy, J.B. Synthesis methods of carbon nanotubes and related materials. Materials 2010, 3, 3092–3140. [Google Scholar] [CrossRef]
- Teo, K.B.; Singh, C.; Chhowalla, M.; Milne, W.I. Catalytic synthesis of carbon nanotubes and nanofibers. Encycl. Nanosci. Nanotechnol. 2003, 10, 1–22. [Google Scholar]
- Paradise, M.; Goswami, T. Carbon nanotubes–production and industrial applications. Mater. Des. 2007, 28, 1477–1489. [Google Scholar] [CrossRef]
- Journet, C.; Bernier, P. Production of carbon nanotubes. Appl. Phys. A Mater. Sci. Process. 1998, 67, 1–9. [Google Scholar] [CrossRef]
- Landi, B.J.; Raffaelle, R.P.; Castro, S.L.; Bailey, S.G. Single-wall carbon nanotube–polymer solar cells. Prog. Photovolt. Res. Appl. 2005, 13, 165–172. [Google Scholar] [CrossRef]
- Terrones-Maldonado, M. Production and Characterisation of Novel Fullerene-Related Materials: Nanotubes, Nanofibres and Giant Fullerenes. Ph.D. Thesis, University of Sussex, Brighton, UK, 1997. [Google Scholar]
- Pandey, P.; Dahiya, M. Carbon nanotubes: Types, methods of preparation and applications. IJPSR 2016, 1, 15–21. [Google Scholar]
- Yang, Q.H.; Bai, S.; Sauvajol, J.L.; Bai, J.B. Large-diameter single-walled carbon nanotubes synthesized by chemical vapor deposition. Adv. Mater. 2003, 15, 792–795. [Google Scholar] [CrossRef]
- Zhang, B.; Chen, Q.; Tang, H.; Xie, Q.; Ma, M.; Tan, L.; Zhang, Y.; Yao, S. Characterization of and biomolecule immobilization on the biocompatible multi-walled carbon nanotubes generated by functionalization with polyamidoamine dendrimers. Colloids Surf. B Biointerfaces 2010, 80, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Couteau, E.; Hernadi, K.; Seo, J.W.; Thien-Nga, L.; Mikó, C.; Gaal, R.; Forro, L. CVD synthesis of high-purity multiwalled carbon nanotubes using CaCO3 catalyst support for large-scale production. Chem. Phys. Lett. 2003, 378, 9–17. [Google Scholar] [CrossRef]
- Ren, Z.; Huang, Z.; Xu, J.; Wang, J.; Bush, P.; Siegal, M.; Provencio, P. Synthesis of large arrays of well-aligned carbon nanotubes on glass. Science 1998, 282, 1105–1107. [Google Scholar] [CrossRef] [PubMed]
- Fan, S.; Chapline, M.G.; Franklin, N.R.; Tombler, T.W.; Cassell, A.M.; Dai, H. Self-oriented regular arrays of carbon nanotubes and their field emission properties. Science 1999, 283, 512–514. [Google Scholar] [CrossRef] [PubMed]
- Patole, S.; Alegaonkar, P.; Lee, H.-C.; Yoo, J.-B. Optimization of water assisted chemical vapor deposition parameters for super growth of carbon nanotubes. Carbon 2008, 46, 1987–1993. [Google Scholar] [CrossRef]
- Chen, J.; Gao, X. Recent Advances in the Flame Synthesis of Carbon Nanotubes. Am. J. Mater. Synth. Process. 2017, 2, 71. [Google Scholar] [CrossRef]
- Merchan-Merchan, W.; Saveliev, A.V.; Kennedy, L.; Jimenez, W.C. Combustion synthesis of carbon nanotubes and related nanostructures. Prog. Energy Combust. Sci. 2010, 36, 696–727. [Google Scholar] [CrossRef]
- Xu, Z.; Zhao, H. Simultaneous measurement of internal and external properties of nanoparticles in flame based on thermophoresis. Combust. Flame 2015, 162, 2200–2213. [Google Scholar] [CrossRef]
- Yuan, L.; Saito, K.; Pan, C.; Williams, F.; Gordon, A. Nanotubes from methane flames. Chem. Phys. Lett. 2001, 340, 237–241. [Google Scholar] [CrossRef]
- Yuan, L.; Saito, K.; Hu, W.; Chen, Z. Ethylene flame synthesis of well-aligned multi-walled carbon nanotubes. Chem. Phys. Lett. 2001, 346, 23–28. [Google Scholar] [CrossRef]
- Lee, G.W.; Jurng, J.; Hwang, J. Formation of Ni-catalyzed multiwalled carbon nanotubes and nanofibers on a substrate using an ethylene inverse diffusion flame. Combust. Flame 2004, 139, 167–175. [Google Scholar] [CrossRef]
- Vander Wal, R.L.; Berger, G.M.; Hall, L.J. Single-walled carbon nanotube synthesis via a multi-stage flame configuration. J. Phys. Chem. B 2002, 106, 3564–3567. [Google Scholar] [CrossRef]
- Vander Wal, R.L.; Ticich, T.M. Flame and furnace synthesis of single-walled and multi-walled carbon nanotubes and nanofibers. J. Phys. Chem. B 2001, 105, 10249–10256. [Google Scholar] [CrossRef]
- Digge, M.; Moon, R.; Gattani, S. Applications of carbon nanotubes in drug delivery: A review. Int. J. PharmTech Res. 2012, 4, 839–847. [Google Scholar]
- Bhaskar, A.; Deshmukh, V.; Prajapati, L. Carbon nanotube as a drug delivery system: A review. Int. J. Pharm. Technol. 2013, 5, 2695–2711. [Google Scholar]
- Annu, A.; Bhattacharya, B.; Singh, P.K.; Shukla, P.K.; Rhee, H.-W. Carbon nanotube using spray pyrolysis: Recent scenario. J. Alloys Compd. 2017, 691, 970–982. [Google Scholar] [CrossRef]
- Yuan, D.; Ding, L.; Chu, H.; Feng, Y.; McNicholas, T.P.; Liu, J. Horizontally aligned single-walled carbon nanotube on quartz from a large variety of metal catalysts. Nano Lett. 2008, 8, 2576–2579. [Google Scholar] [CrossRef] [PubMed]
- Wasel, W.; Kuwana, K.; Reilly, P.T.; Saito, K. Experimental characterization of the role of hydrogen in CVD synthesis of MWCNTs. Carbon 2007, 45, 833–838. [Google Scholar] [CrossRef]
- Jorio, A.; Pimenta, M.; Souza Filho, A.; Saito, R.; Dresselhaus, G.; Dresselhaus, M. Characterizing carbon nanotube samples with resonance Raman scattering. New J. Phys. 2003, 5, 139. [Google Scholar] [CrossRef]
- Dresselhaus, M.; Dresselhaus, G.; Jorio, A.; Souza Filho, A.; Saito, R. Raman spectroscopy on isolated single wall carbon nanotubes. Carbon 2002, 40, 2043–2061. [Google Scholar] [CrossRef]
- Fantini, C.; Jorio, A.; Souza, M.; Ladeira, L.; Souza Filho, A.; Saito, R.; Samsonidze, G.G.; Dresselhaus, G.; Dresselhaus, M.; Pimenta, M. One-dimensional character of combination modes in the resonance Raman scattering of carbon nanotubes. Phys. Rev. Lett. 2004, 93, 087401. [Google Scholar] [CrossRef]
- Jorio, A.; Saito, R.; Hafner, J.; Lieber, C.; Hunter, D.; McClure, T.; Dresselhaus, G.; Dresselhaus, M. Structural (n, m) determination of isolated single-wall carbon nanotubes by resonant Raman scattering. Phys. Rev. Lett. 2001, 86, 1118. [Google Scholar] [CrossRef]
- Milnera, M.; Kürti, J.; Hulman, M.; Kuzmany, H. Periodic resonance excitation and intertube interaction from quasicontinuous distributed helicities in single-wall carbon nanotubes. Phys. Rev. Lett. 2000, 84, 1324. [Google Scholar] [CrossRef] [PubMed]
- Dresselhaus, M.S.; Dresselhaus, G.; Saito, R.; Jorio, A. Raman spectroscopy of carbon nanotubes. Phys. Rep. 2005, 409, 47–99. [Google Scholar] [CrossRef]
- Souza Filho, A.; Jorio, A.; Swan, A.K.; Ünlü, M.; Goldberg, B.; Saito, R.; Hafner, J.; Lieber, C.; Pimenta, M.; Dresselhaus, G. Anomalous two-peak G′-band Raman effect in one isolated single-wall carbon nanotube. Phys. Rev. B 2002, 65, 085417. [Google Scholar] [CrossRef]
- Souza Filho, A.; Jorio, A.; Samsonidze, G.G.; Dresselhaus, G.; Dresselhaus, M.; Swan, A.K.; Ünlü, M.; Goldberg, B.; Saito, R.; Hafner, J. Probing the electronic trigonal warping effect in individual single-wall carbon nanotubes using phonon spectra. Chem. Phys. Lett. 2002, 354, 62–68. [Google Scholar] [CrossRef]
- Wilhelm, H.; Lelaurain, M.; McRae, E.; Humbert, B. Raman spectroscopic studies on well-defined carbonaceous materials of strong two-dimensional character. J. Appl. Phys. 1998, 84, 6552–6558. [Google Scholar] [CrossRef]
- Branca, C.; Frusteri, F.; Magazu, V.; Mangione, A. Characterization of carbon nanotubes by TEM and infrared spectroscopy. J. Phys. Chem. B 2004, 108, 3469–3473. [Google Scholar] [CrossRef]
- Safarova, K.; Dvorak, A.; Kubinek, R.; Vujtek, M.; Rek, A. Usage of AFM, SEM and TEM for the research of carbon nanotubes. Mod. Res. Educ. Top. Microsc. 2007, 2, 513–519. [Google Scholar]
- Oh, S.; Hyon, C.; Sull, S.; Hwang, S.; Park, Y. Detection and volume estimation of semiconductor quantum dots from atomic force microscope images. Rev. Sci. Instrum. 2003, 74, 4687–4695. [Google Scholar] [CrossRef]
- Hata, K.; Futaba, D.N.; Mizuno, K.; Namai, T.; Yumura, M.; Iijima, S. Water-assisted highly efficient synthesis of impurity-free single-walled carbon nanotubes. Science 2004, 306, 1362–1364. [Google Scholar] [CrossRef]
- Huczko, A. Synthesis of aligned carbon nanotubes. Appl. Phys. A 2002, 74, 617–638. [Google Scholar] [CrossRef]
- Kong, J.; Soh, H.T.; Cassell, A.M.; Quate, C.F.; Dai, H. Synthesis of individual single-walled carbon nanotubes on patterned silicon wafers. Nature 1998, 395, 878–881. [Google Scholar] [CrossRef]
- Ural, A.; Li, Y.; Dai, H. Electric-field-aligned growth of single-walled carbon nanotubes on surfaces. Appl. Phys. Lett. 2002, 81, 3464–3466. [Google Scholar] [CrossRef]
- Huang, S.; Cai, X.; Liu, J. Growth of millimeter-long and horizontally aligned single-walled carbon nanotubes on flat substrates. J. Am. Chem. Soc. 2003, 125, 5636–5637. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Chu, H.; Wang, J.; Hong, J.; Tan, W.; Li, Y. Ultralow feeding gas flow guiding growth of large-scale horizontally aligned single-walled carbon nanotube arrays. Nano Lett. 2007, 7, 2073–2079. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Liu, X.; Zhou, C. Template-free directional growth of single-walled carbon nanotubes on a-and r-plane sapphire. J. Am. Chem. Soc. 2005, 127, 5294–5295. [Google Scholar] [CrossRef] [PubMed]
- Louie, S.G. Electronic properties, junctions, and defects of carbon nanotubes. In Carbon Nanotubes; Springer: Berlin/Heidelberg, Germany, 2001; pp. 113–145. [Google Scholar]
- Baughman, R.H.; Zakhidov, A.A.; De Heer, W.A. Carbon nanotubes—The route toward applications. Science 2002, 297, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Bachtold, A.; Hadley, P.; Nakanishi, T.; Dekker, C.; Kuzmany, H.; Fink, J.; Mehring, M.; Roth, S. Logic circuits with carbon nanotubes. AIP Conf. Proc. 2002, 633, 502–507. [Google Scholar]
- AuBuchon, J.F.; Chen, L.-H.; Gapin, A.I.; Kim, D.-W.; Daraio, C.; Jin, S. Multiple sharp bendings of carbon nanotubes during growth to produce zigzag morphology. Nano Lett. 2004, 4, 1781–1784. [Google Scholar] [CrossRef]
- Zhang, M.; Li, J. Carbon nanotube in different shapes. Mater. Today 2009, 12, 12–18. [Google Scholar] [CrossRef]
- Iijima, S.; Brabec, C.; Maiti, A.; Bernholc, J. Structural flexibility of carbon nanotubes. J. Chem. Phys. 1996, 104, 2089–2092. [Google Scholar] [CrossRef]
- Yakobson, B.I.; Brabec, C.; Bernholc, J. Nanomechanics of carbon tubes: Instabilities beyond linear response. Phys. Rev. Lett. 1996, 76, 2511. [Google Scholar] [CrossRef] [PubMed]
- Gao, R.; Wang, Z.L.; Fan, S. Kinetically controlled growth of helical and zigzag shapes of carbon nanotubes. J. Phys. Chem. B 2000, 104, 1227–1234. [Google Scholar] [CrossRef]
- Dunlap, B.I. Connecting carbon tubules. Phys. Rev. B 1992, 46, 1933. [Google Scholar] [CrossRef]
- Itoh, S.; Ihara, S. Toroidal forms of graphitic carbon. II. Elongated tori. Phys. Rev. B 1993, 48, 8323. [Google Scholar] [CrossRef]
- Zhang, M.; Nakayama, Y.; Pan, L. Synthesis of carbon tubule nanocoils in high yield using iron-coated indium tin oxide as catalyst. Jpn. J. Appl. Phys. 2000, 39, L1242. [Google Scholar] [CrossRef]
- Hou, H.; Jun, Z.; Weller, F.; Greiner, A. Large-scale synthesis and characterization of helically coiled carbon nanotubes by use of Fe(CO)5 as floating catalyst precursor. Chem. Mater. 2003, 15, 3170–3175. [Google Scholar] [CrossRef]
- Lu, M.; Li, H.-L.; Lau, K.-T. Formation and growth mechanism of dissimilar coiled carbon nanotubes by reduced-pressure catalytic chemical vapor deposition. J. Phys. Chem. B 2004, 108, 6186–6192. [Google Scholar] [CrossRef]
- Wang, J.N.; Su, L.F.; Wu, Z.P. Growth of highly compressed and regular coiled carbon nanotubes by a spray-pyrolysis method. Cryst. Growth Des. 2008, 8, 1741–1747. [Google Scholar] [CrossRef]
- Shaikjee, A.; Coville, N.J. The synthesis, properties and uses of carbon materials with helical morphology. J. Adv. Res. 2012, 3, 195–223. [Google Scholar] [CrossRef]
- Zhou, D.; Seraphin, S. Complex branching phenomena in the growth of carbon nanotubes. Chem. Phys. Lett. 1995, 238, 286–289. [Google Scholar] [CrossRef]
- Scuseria, G.E. Negative curvature and hyperfullerenes. Chem. Phys. Lett. 1992, 195, 534–536. [Google Scholar] [CrossRef]
- Chernozatonskii, L. Carbon nanotube connectors and planar jungle gyms. Phys. Lett. A 1992, 172, 173–176. [Google Scholar] [CrossRef]
- Li, J.; Papadopoulos, C.; Xu, J. Nanoelectronics: Growing Y-junction carbon nanotubes. Nature 1999, 402, 253–254. [Google Scholar] [CrossRef]
- Nakayama, Y.; Zhang, M. Synthesis of carbon nanochaplets by catalytic thermal chemical vapor deposition. Jpn. J. Appl. Phys. 2001, 40, L492. [Google Scholar] [CrossRef]
- De Heer, W.A.; Poncharal, P.; Berger, C.; Gezo, J.; Song, Z.; Bettini, J.; Ugarte, D. Liquid carbon, carbon-glass beads, and the crystallization of carbon nanotubes. Science 2005, 307, 907–910. [Google Scholar] [CrossRef] [PubMed]
- Monthioux, M.; Allouche, H.; Jacobsen, R.L. Chemical vapour deposition of pyrolytic carbon on carbon nanotubes: Part 3: Growth mechanisms. Carbon 2006, 44, 3183–3194. [Google Scholar] [CrossRef]
- Ting, J.-M.; Lan, J.B. Beaded carbon tubes. Appl. Phys. Lett. 1999, 75, 3309–3311. [Google Scholar] [CrossRef]
- Sharma, P.; Ahuja, P. Recent advances in carbon nanotube-based electronics. Mater. Res. Bull. 2008, 43, 2517–2526. [Google Scholar] [CrossRef]
- Wildoer, J.; Venema, L.; Rinzler, A.; Smalley, R.; Dekker, C. Electronic structure of carbon nanotubes investigated by scanning tunneling spectroscopy. Nature 1998, 391, 59–62. [Google Scholar]
- Zhu, J.; Holmen, A.; Chen, D. Carbon nanomaterials in catalysis: Proton affinity, chemical and electronic properties, and their catalytic consequences. ChemCatChem 2013, 5, 378–401. [Google Scholar] [CrossRef]
- Karami, M.; Bahabadi, M.A.; Delfani, S.; Ghozatloo, A. A new application of carbon nanotubes nanofluid as working fluid of low-temperature direct absorption solar collector. Sol. Energy Mater. Sol. Cells 2014, 121, 114–118. [Google Scholar] [CrossRef]
- Esfe, M.H.; Saedodin, S.; Yan, W.-M.; Afrand, M.; Sina, N. Study on thermal conductivity of water-based nanofluids with hybrid suspensions of CNTs/Al2O3 nanoparticles. J. Therm. Anal. Calorim. 2016, 124, 455–460. [Google Scholar] [CrossRef]
- Mauter, M.S.; Elimelech, M. Environmental applications of carbon-based nanomaterials. Environ. Sci. Technol. 2008, 42, 5843–5859. [Google Scholar] [CrossRef] [PubMed]
- Thirumal, V.; Pandurangan, A.; Jayavel, R.; Krishnamoorthi, S.; Ilangovan, R. Synthesis of nitrogen doped coiled double walled carbon nanotubes by chemical vapor deposition method for supercapacitor applications. Curr. Appl. Phys. 2016, 16, 816–825. [Google Scholar] [CrossRef]
- Hone, J. Carbon Nanotube, Phonons and Thermal Properties; Springer: Berlin/Heidelberg, Germany, 2001; Volume 80. [Google Scholar]
- Robertson, D.; Brenner, D.; Mintmire, J. Energetics of nanoscale graphitic tubules. Phys. Rev. B 1992, 45, 12592. [Google Scholar] [CrossRef]
- Yu, M.-F.; Files, B.S.; Arepalli, S.; Ruoff, R.S. Tensile loading of ropes of single wall carbon nanotubes and their mechanical properties. Phys. Rev. Lett. 2000, 84, 5552. [Google Scholar] [CrossRef] [PubMed]
- Saifuddin, N.; Raziah, A.; Junizah, A. Carbon nanotubes: A review on structure and their interaction with proteins. J. Chem. 2012, 2013, 676815. [Google Scholar] [CrossRef]
- Tans, S.J.; Verschueren, A.R.; Dekker, C. Room-temperature transistor based on a single carbon nanotube. Nature 1998, 393, 49–52. [Google Scholar] [CrossRef]
- Bandyopadhyaya, R.; Nativ-Roth, E.; Regev, O.; Yerushalmi-Rozen, R. Stabilization of individual carbon nanotubes in aqueous solutions. Nano Lett. 2002, 2, 25–28. [Google Scholar] [CrossRef]
- Kharissova, O.V.; Kharisov, B.I.; de Casas Ortiz, E.G. Dispersion of carbon nanotubes in water and non-aqueous solvents. Rsc Adv. 2013, 3, 24812–24852. [Google Scholar] [CrossRef]
- Sondi, I.; Goia, D.V.; Matijević, E. Preparation of highly concentrated stable dispersions of uniform silver nanoparticles. J. Colloid Interface Sci. 2003, 260, 75–81. [Google Scholar] [CrossRef]
- Yu, W.; Xie, H. A review on nanofluids: Preparation, stability mechanisms, and applications. J. Nanomater. 2012, 2012, 1. [Google Scholar] [CrossRef]
- Bilalis, P.; Katsigiannopoulos, D.; Avgeropoulos, A.; Sakellariou, G. Non-covalent functionalization of carbon nanotubes with polymers. Rsc Adv. 2014, 4, 2911–2934. [Google Scholar] [CrossRef]
- Sayes, C.M.; Liang, F.; Hudson, J.L.; Mendez, J.; Guo, W.; Beach, J.M.; Moore, V.C.; Doyle, C.D.; West, J.L.; Billups, W.E. Functionalization density dependence of single-walled carbon nanotubes cytotoxicity in vitro. Toxicol. Lett. 2006, 161, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Sehrawat, P.; Julien, C.; Islam, S.S. Carbon nanotubes in Li-ion batteries: A review. Mater. Sci. Eng. B 2016, 213, 12–40. [Google Scholar] [CrossRef]
- Ma, L.; Hendrickson, K.E.; Wei, S.; Archer, L.A. Nanomaterials: Science and applications in the lithium–sulfur battery. Nano Today 2015, 10, 315–338. [Google Scholar] [CrossRef]
- Shende, R.C.; Ramaprabhu, S. Thermo-optical properties of partially unzipped multiwalled carbon nanotubes dispersed nanofluids for direct absorption solar thermal energy systems. Sol. Energy Mater. Sol. Cells 2016, 157, 117–125. [Google Scholar] [CrossRef]
- Muhammad, M.J.; Muhammad, I.A.; Sidik, N.A.C.; Yazid, M.N.A.W.M.; Mamat, R.; Najafi, G. The use of nanofluids for enhancing the thermal performance of stationary solar collectors: A review. Renew. Sustain. Energy Rev. 2016, 63, 226–236. [Google Scholar] [CrossRef]
- Mesgari, S.; Taylor, R.A.; Hjerrild, N.E.; Crisostomo, F.; Li, Q.; Scott, J. An investigation of thermal stability of carbon nanofluids for solar thermal applications. Sol. Energy Mater. Sol. Cells 2016, 157, 652–659. [Google Scholar] [CrossRef]
- Li, X.; Zou, C.; Chen, W.; Lei, X. Experimental investigation of β-cyclodextrin modified carbon nanotubes nanofluids for solar energy systems: Stability, optical properties and thermal conductivity. Sol. Energy Mater. Sol. Cells 2016, 157, 572–579. [Google Scholar] [CrossRef]
- Dürkop, T.; Getty, S.; Cobas, E.; Fuhrer, M. Extraordinary mobility in semiconducting carbon nanotubes. Nano Lett. 2004, 4, 35–39. [Google Scholar] [CrossRef]
- Wang, F.; Kozawa, D.; Miyauchi, Y.; Hiraoka, K.; Mouri, S.; Ohno, Y.; Matsuda, K. Considerably improved photovoltaic performance of carbon nanotube-based solar cells using metal oxide layers. Nat. Commun. 2015, 6, 6305. [Google Scholar] [CrossRef] [PubMed]
- Alturaif, H.; ALOthman, Z.; Shapter, J.; Wabaidur, S. Use of carbon nanotubes (CNTs) with polymers in solar cells. Molecules 2014, 19, 17329–17344. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Jia, Y.; Shu, Q.; Gu, Z.; Wang, K.; Zhuang, D.; Zhang, G.; Wang, Z.; Luo, J.; Cao, A. Double-walled carbon nanotube solar cells. Nano Lett. 2007, 7, 2317–2321. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Wei, J.; Wang, K.; Cao, A.; Shu, Q.; Gui, X.; Zhu, Y.; Zhuang, D.; Zhang, G.; Ma, B. Nanotube–silicon heterojunction solar cells. Adv. Mater. 2008, 20, 4594–4598. [Google Scholar] [CrossRef]
- Liang, C.-W.; Roth, S. Electrical and optical transport of GaAs/carbon nanotube heterojunctions. Nano Lett. 2008, 8, 1809–1812. [Google Scholar] [CrossRef] [PubMed]
- Signorelli, R.; Ku, D.C.; Kassakian, J.G.; Schindall, J.E. Electrochemical double-layer capacitors using carbon nanotube electrode structures. Proc. IEEE 2009, 97, 1837–1847. [Google Scholar] [CrossRef]
- Xu, X.; Tan, H.; Xi, K.; Ding, S.; Yu, D.; Cheng, S.; Yang, G.; Peng, X.; Fakeeh, A.; Kumar, R.V. Bamboo-like amorphous carbon nanotubes clad in ultrathin nickel oxide nanosheets for lithium-ion battery electrodes with long cycle life. Carbon 2015, 84, 491–499. [Google Scholar] [CrossRef]
- Wang, W.; Kumta, P.N. Nanostructured hybrid silicon/carbon nanotube heterostructures: Reversible high-capacity lithium-ion anodes. ACS Nano 2010, 4, 2233–2241. [Google Scholar] [CrossRef]
- Wu, H.; Chan, G.; Choi, J.W.; Yao, Y.; McDowell, M.T.; Lee, S.W.; Jackson, A.; Yang, Y.; Hu, L.; Cui, Y. Stable cycling of double-walled silicon nanotube battery anodes through solid-electrolyte interphase control. Nature Nanotechnol. 2012, 7, 310–315. [Google Scholar] [CrossRef]
- Xue, L.; Xu, G.; Li, Y.; Li, S.; Fu, K.; Shi, Q.; Zhang, X. Carbon-coated Si nanoparticles dispersed in carbon nanotube networks as anode material for lithium-ion batteries. ACS Appl. Mater. Interfaces 2012, 5, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Thepsuparungsikul, N.; Phonthamachai, N.; Ng, H. Multi-walled carbon nanotubes as electrode material for microbial fuel cells. Water Sci. Technol. 2012, 65, 1208–1214. [Google Scholar] [CrossRef] [PubMed]
- Fraiwan, A.; Adusumilli, S.; Han, D.; Steckl, A.; Call, D.; Westgate, C.; Choi, S. Microbial Power-Generating Capabilities on Micro-/Nano-Structured Anodes in Micro-Sized Microbial Fuel Cells. Fuel Cells 2014, 14, 801–809. [Google Scholar] [CrossRef]
- Ren, H.; Pyo, S.; Lee, J.-I.; Park, T.-J.; Gittleson, F.S.; Leung, F.C.; Kim, J.; Taylor, A.D.; Lee, H.-S.; Chae, J. A high power density miniaturized microbial fuel cell having carbon nanotube anodes. J. Power Sources 2015, 273, 823–830. [Google Scholar] [CrossRef]
- Wang, Y.-Q.; Huang, H.-X.; Li, B.; Li, W.-S. Novelly developed three-dimensional carbon scaffold anodes from polyacrylonitrile for microbial fuel cells. J. Mater. Chem. A 2015, 3, 5110–5118. [Google Scholar] [CrossRef]
- Xie, X.; Hu, L.; Pasta, M.; Wells, G.F.; Kong, D.; Criddle, C.S.; Cui, Y. Three-dimensional carbon nanotube−textile anode for high-performance microbial fuel cells. Nano Lett. 2011, 11, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Ye, M.; Hu, L.; Liu, N.; McDonough, J.R.; Chen, W.; Alshareef, H.N.; Criddle, C.S.; Cui, Y. Carbon nanotube-coated macroporous sponge for microbial fuel cell electrodes. Energy Environ. Sci. 2012, 5, 5265–5270. [Google Scholar] [CrossRef]
- He, Y.; Liu, Z.; Xing, X.-H.; Li, B.; Zhang, Y.; Shen, R.; Zhu, Z.; Duan, N. Carbon nanotubes simultaneously as the anode and microbial carrier for up-flow fixed-bed microbial fuel cell. Biochem. Eng. J. 2015, 94, 39–44. [Google Scholar] [CrossRef]
- Shen, Y.; Zhou, Y.; Chen, S.; Yang, F.; Zheng, S.; Hou, H. Carbon nanofibers modified graphite felt for high performance anode in high substrate concentration microbial fuel cells. Sci. World J. 2014, 2014, 130185. [Google Scholar] [CrossRef]
- Manickam, S.S.; Karra, U.; Huang, L.; Bui, N.-N.; Li, B.; McCutcheon, J.R. Activated carbon nanofiber anodes for microbial fuel cells. Carbon 2013, 53, 19–28. [Google Scholar] [CrossRef]
- Pokhrel, L.R.; Ettore, N.; Jacobs, Z.L.; Zarr, A.; Weir, M.H.; Scheuerman, P.R.; Kanel, S.R.; Dubey, B. Novel carbon nanotube (CNT)-based ultrasensitive sensors for trace mercury(II) detection in water: A review. Sci. Total Environ. 2017, 574, 1379–1388. [Google Scholar] [CrossRef] [PubMed]
- Anitha, K.; Namsani, S.; Singh, J.K. Removal of Heavy Metal Ions Using a Functionalized Single-Walled Carbon Nanotube: A Molecular Dynamics Study. J. Phys. Chem. A 2015, 119, 8349–8358. [Google Scholar] [CrossRef] [PubMed]
- Tofighy, M.A.; Mohammadi, T. Nickel ions removal from water by two different morphologies of induced CNTs in mullite pore channels as adsorptive membrane. Ceram. Int. 2015, 41, 5464–5472. [Google Scholar] [CrossRef]
- Zhu, X.; Cui, Y.; Chang, X.; Wang, H. Selective solid-phase extraction and analysis of trace-level Cr (III), Fe (III), Pb (II), and Mn (II) Ions in wastewater using diethylenetriamine-functionalized carbon nanotubes dispersed in graphene oxide colloids. Talanta 2016, 146, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Mubarak, N.M.; Sahu, J.N.; Abdullah, E.C.; Jayakumar, N.S. Rapid adsorption of toxic Pb (II) ions from aqueous solution using multiwall carbon nanotubes synthesized by microwave chemical vapor deposition technique. J. Environ. Sci. 2016, 45, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-H.; Wang, S.; Wei, J.; Zhang, X.; Xu, C.; Luan, Z.; Wu, D.; Wei, B. Lead adsorption on carbon nanotubes. Chem. Phys. Lett. 2002, 357, 263–266. [Google Scholar] [CrossRef]
- Lu, C.; Liu, C. Removal of nickel (II) from aqueous solution by carbon nanotubes. J. Chem. Technol. Biotechnol. 2006, 81, 1932–1940. [Google Scholar] [CrossRef]
- Abbas, A.; Al-Amer, A.M.; Laoui, T.; Al-Marri, M.J.; Nasser, M.S.; Khraisheh, M.; Atieh, M.A. Heavy metal removal from aqueous solution by advanced carbon nanotubes: Critical review of adsorption applications. Sep. Purif. Technol. 2016, 157, 141–161. [Google Scholar]
- Santhosh, C.; Velmurugan, V.; Jacob, G.; Jeong, S.K.; Grace, A.N.; Bhatnagar, A. Role of nanomaterials in water treatment applications: A review. Chem. Eng. J. 2016, 306, 1116–1137. [Google Scholar] [CrossRef]
- Wu, C.-H. Adsorption of reactive dye onto carbon nanotubes: Equilibrium, kinetics and thermodynamics. J. Hazard. Mater. 2007, 144, 93–100. [Google Scholar] [CrossRef]
- Bahgat, M.; Farghali, A.A.; El Rouby, W.M.A.; Khedr, M.H. Synthesis and modification of multi-walled carbon nano-tubes (MWCNTs) for water treatment applications. J. Anal. Appl. Pyrolysis 2011, 92, 307–313. [Google Scholar] [CrossRef]
- Cano, O.A.; González, C.R.; Paz, J.H.; Madrid, P.A.; Casillas, P.G.; Hernández, A.M.; Pérez, C.M. Catalytic activity of palladium nanocubes/multiwalled carbon nanotubes structures for methyl orange dye removal. Catal. Today 2017, 282, 168–173. [Google Scholar] [CrossRef]
- Ma, J.; Yu, F.; Zhou, L.; Jin, L.; Yang, M.; Luan, J.; Tang, Y.; Fan, H.; Yuan, Z.; Chen, J. Enhanced adsorptive removal of methyl orange and methylene blue from aqueous solution by alkali-activated multiwalled carbon nanotubes. ACS Appl. Mater. Interfaces 2012, 4, 5749–5760. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ng, C.W.; Wang, W.; Li, Q.; Hao, Z. Synergistic and competitive adsorption of organic dyes on multiwalled carbon nanotubes. Chem. Eng. J. 2012, 197, 34–40. [Google Scholar] [CrossRef]
- Lou, J.C.; Jung, M.J.; Yang, H.W.; Han, J.Y.; Huang, W.H. Removal of dissolved organic matter (DOM) from raw water by single-walled carbon nanotubes (SWCNTs). J. Environ. Sci. Health Part A 2011, 46, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Jiang, R.; Xiao, L.; Zeng, G. Preparation, characterization, adsorption kinetics and thermodynamics of novel magnetic chitosan enwrapping nanosized γ-Fe2O3 and multi-walled carbon nanotubes with enhanced adsorption properties for methyl orange. Bioresour. Technol. 2010, 101, 5063–5069. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Lu, Y.; Zheng, F.; Xue, X.; Li, N.; Liu, D. Adsorption behavior and mechanisms of norfloxacin onto porous resins and carbon nanotube. Chem. Eng. J. 2012, 179, 112–118. [Google Scholar] [CrossRef]
- Ncibi, M.C.; Sillanpää, M. Optimized removal of antibiotic drugs from aqueous solutions using single, double and multi-walled carbon nanotubes. J. Hazard. Mater. 2015, 298, 102–110. [Google Scholar] [CrossRef]
- Yang, Q.; Chen, G.; Zhang, J.; Li, H. Adsorption of sulfamethazine by multi-walled carbon nanotubes: Effects of aqueous solution chemistry. Rsc Adv. 2015, 5, 25541–25549. [Google Scholar] [CrossRef]
- Fard, A.K.; Mckay, G.; Manawi, Y.; Malaibari, Z.; Hussien, M.A. Outstanding adsorption performance of high aspect ratio and super-hydrophobic carbon nanotubes for oil removal. Chemosphere 2016, 164, 142–155. [Google Scholar] [CrossRef]
- Qiu, W.; Xu, H.; Takalkar, S.; Gurung, A.S.; Liu, B.; Zheng, Y.; Guo, Z.; Baloda, M.; Baryeh, K.; Liu, G. Carbon nanotube-based lateral flow biosensor for sensitive and rapid detection of DNA sequence. Biosens. Bioelectron. 2015, 64, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Sinha, N.; Yeow, J.-W. Carbon nanotubes for biomedical applications. IEEE Trans. NanoBiosci. 2005, 4, 180–195. [Google Scholar] [CrossRef]
- Joseph, H. MEMS in the medical world. Sens. J. Appl. Sens. Technol. 1997, 14, 47–51. [Google Scholar]
- Bernal-Martínez, J.; Seseña-Rubfiaro, A.; Godínez-Fernández, R.; Aguilar-Elguezabal, A. Electrodes made of multi-wall carbon nanotubes on PVDF-filters have low electrical resistance and are able to record electrocardiograms in humans. Microelectron. Eng. 2016, 166, 10–14. [Google Scholar] [CrossRef]
- Vohrer, U.; Kolaric, I.; Haque, M.; Roth, S.; Detlaff-Weglikowska, U. Carbon nanotube sheets for the use as artificial muscles. Carbon 2004, 42, 1159–1164. [Google Scholar] [CrossRef]
- Zeng, Z.; Jin, H.; Zhang, L.; Zhang, H.; Chen, Z.; Gao, F.; Zhang, Z. Low-voltage and high-performance electrothermal actuator based on multi-walled carbon nanotube/polymer composites. Carbon 2015, 84, 327–334. [Google Scholar] [CrossRef]
- Foroughi, J.; Spinks, G.M.; Aziz, S.; Mirabedini, A.; Jeiranikhameneh, A.; Wallace, G.G.; Kozlov, M.E.; Baughman, R.H. Knitted carbon-nanotube-sheath/spandex-core elastomeric yarns for artificial muscles and strain sensing. ACS Nano 2016, 10, 9129–9135. [Google Scholar] [CrossRef] [PubMed]
- Inganäs, O.; Lundstrüm, I. Carbon nanotube muscles. Science 1999, 284, 1281–1282. [Google Scholar]
- Wu, L.; Qu, X. Cancer biomarker detection: Recent achievements and challenges. Chem. Soc. Rev. 2015, 44, 2963–2997. [Google Scholar] [CrossRef]
- Hong, H.; Gao, T.; Cai, W. Molecular imaging with single-walled carbon nanotubes. Nano Today 2009, 4, 252–261. [Google Scholar] [CrossRef]
- Patel, A.; Prajapati, P.; Chaudhari, K.; Boghra, R.; Jadhav, A. Carbon nanotubes production, characterisation and its applications. J. Adv. Pharm. Sci. 2010, 1, 187–195. [Google Scholar]
- Marquis, F.D. Fully integrated hybrid polymeric carbon nanotube composites. In Materials Science Forum; Trans Tech Publications Ltd.: Zurich-Uetikon, Switzerland, 2003; Volume 437, pp. 85–88. [Google Scholar]
- Bian, Z.; Wang, R.J.; Wang, W.H.; Zhang, T.; Inoue, A. Carbon-Nanotube-Reinforced Zr-Based Bulk Metallic Glass Composites and Their Properties. Adv. Funct. Mater. 2004, 14, 55–63. [Google Scholar] [CrossRef]
- Costa, P.M.; Bourgognon, M.; Wang, J.T.; Al-Jamal, K.T. Functionalised carbon nanotubes: From intracellular uptake and cell-related toxicity to systemic brain delivery. J. Control. Release 2016, 241, 200–219. [Google Scholar] [CrossRef] [PubMed]
- Flahaut, E.; Rul, S.; Lefévre-Schlick, F.; Laurent, C.; Peigney, A. Carbon Nanotubes-Ceramic Composites. Ceram. Nanomater. Nanotechnol. II 2004, 148, 69–82. [Google Scholar]
- Yanagi, H.; Kawai, Y.; Kita, T.; Fujii, S.; Hayashi, Y.; Magario, A.; Noguchi, T. Carbon nanotube/aluminum composites as a novel field electron emitter. Jpn. J. Appl. Phys. 2006, 45, L650. [Google Scholar] [CrossRef]
- Baughman, R.H.; Cui, C.; Zakhidov, A.A.; Iqbal, Z.; Barisci, J.N.; Spinks, G.M.; Wallace, G.G.; Mazzoldi, A.; De Rossi, D.; Rinzler, A.G. Carbon nanotube actuators. Science 1999, 284, 1340–1344. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Sichel, E.K.; Hoch, R.; Moy, D.; Tennent, H. High power electrochemical capacitors based on carbon nanotube electrodes. Appl. Phys. Lett. 1997, 70, 1480–1482. [Google Scholar] [CrossRef]
- Dai, H.; Hafner, J.H.; Rinzler, A.G.; Colbert, D.T.; Smalley, R.E. Nanotubes as nanoprobes in scanning probe microscopy. Nature 1996, 384, 147–150. [Google Scholar] [CrossRef]
- Tibbetts, G.G.; Meisner, G.P.; Olk, C.H. Hydrogen storage capacity of carbon nanotubes, filaments, and vapor-grown fibers. Carbon 2001, 39, 2291–2301. [Google Scholar] [CrossRef]
- Wei, J.; Zhu, H.; Wu, D.; Wei, B. Carbon nanotube filaments in household light bulbs. Appl. Phys. Lett. 2004, 84, 4869–4871. [Google Scholar] [CrossRef]
- Yu, X.; Munge, B.; Patel, V.; Jensen, G.; Bhirde, A.; Gong, J.D.; Kim, S.N.; Gillespie, J.; Gutkind, J.S.; Papadimitrakopoulos, F. Carbon nanotube amplification strategies for highly sensitive immunodetection of cancer biomarkers. J. Am. Chem. Soc. 2006, 128, 11199–11205. [Google Scholar] [CrossRef] [PubMed]
- Okuno, J.; Maehashi, K.; Kerman, K.; Takamura, Y.; Matsumoto, K.; Tamiya, E. Label-free immunosensor for prostate-specific antigen based on single-walled carbon nanotube array-modified microelectrodes. Biosens. Bioelectron. 2007, 22, 2377–2381. [Google Scholar] [CrossRef] [PubMed]
- Ou, C.; Yuan, R.; Chai, Y.; Tang, M.; Chai, R.; He, X. A novel amperometric immunosensor based on layer-by-layer assembly of gold nanoparticles–multi-walled carbon nanotubes-thionine multilayer films on polyelectrolyte surface. Anal. Chim. Acta 2007, 603, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.P.; Lee, B.Y.; Lee, J.; Hong, S.; Sim, S.J. Enhancement of sensitivity and specificity by surface modification of carbon nanotubes in diagnosis of prostate cancer based on carbon nanotube field effect transistors. Biosens. Bioelectron. 2009, 24, 3372–3378. [Google Scholar] [CrossRef] [PubMed]
- Bi, S.; Zhou, H.; Zhang, S. Multilayers enzyme-coated carbon nanotubes as biolabel for ultrasensitive chemiluminescence immunoassay of cancer biomarker. Biosens. Bioelectron. 2009, 24, 2961–2966. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-H.; Wu, C.-C.; Zhong, Z. A fully tunable single-walled carbon nanotube diode. Nano Lett. 2011, 11, 1782–1785. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.U. Photovoltaic effect in ideal carbon nanotube diodes. Appl. Phys. Lett. 2005, 87, 073101. [Google Scholar] [CrossRef]
- Jilili, J.; Abdurahman, A.; Gülseren, O.; Schwingenschlögl, U. Non-covalent functionalization of single wall carbon nanotubes and graphene by a conjugated polymer. Appl. Phys. Lett. 2014, 105, 013103. [Google Scholar] [CrossRef]
- Chen, C.; Jin, T.; Wei, L.; Li, Y.; Liu, X.; Wang, Y.; Zhang, L.; Liao, C.; Hu, N.; Song, C. High-work-function metal/carbon nanotube/low-work-function metal hybrid junction photovoltaic device. NPG Asia Mater. 2015, 7, e220. [Google Scholar] [CrossRef]
- Miyata, Y.; Shiozawa, K.; Asada, Y.; Ohno, Y.; Kitaura, R.; Mizutani, T.; Shinohara, H. Length-sorted semiconducting carbon nanotubes for high-mobility thin film transistors. Nano Res. 2011, 4, 963–970. [Google Scholar] [CrossRef]
- Martel, R.; Derycke, V.; Appenzeller, J.; Wind, S.; Avouris, P. Carbon nanotube field-effect transistors and logic circuits. In Proceedings of the 39th Annual Design Automation Conference, New Orleans, LA, USA, 10–14 June 2002; pp. 94–98. [Google Scholar]
- Peng, L.-M.; Zhang, Z.; Wang, S. Carbon nanotube electronics: Recent advances. Mater. Today 2014, 17, 433–442. [Google Scholar] [CrossRef]
- Appenzeller, J.; Martel, R.; Avouris, P.; Stahl, H.; Lengeler, B. Optimized contact configuration for the study of transport phenomena in ropes of single-wall carbon nanotubes. Appl. Phys. Lett. 2001, 78, 3313–3315. [Google Scholar] [CrossRef]
- Chen, Z.; Appenzeller, J.; Knoch, J.; Lin, Y.-m.; Avouris, P. The role of metal−nanotube contact in the performance of carbon nanotube field-effect transistors. Nano Lett. 2005, 5, 1497–1502. [Google Scholar] [CrossRef] [PubMed]
- Sinha, N.; Ma, J.; Yeow, J.T. Carbon nanotube-based sensors. J. Nanosci. Nanotechnol. 2006, 6, 573–590. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, C.B.; Peairs, M.J.; Venton, B.J. Carbon nanotube based electrochemical sensors for biomolecules. Anal. Chim. Acta 2010, 662, 105–127. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Chen, X.; Ren, T.; Zhang, P.; Yang, D. Carbon nanotube based biosensors. Sens. Actuators B Chem. 2015, 207, 690–715. [Google Scholar] [CrossRef]
- Zhu, Z.; Song, W.; Burugapalli, K.; Moussy, F.; Li, Y.-L.; Zhong, X.-H. Nano-yarn carbon nanotube fiber based enzymatic glucose biosensor. Nanotechnology 2010, 21, 165501. [Google Scholar] [CrossRef]
- Fatoni, A.; Numnuam, A.; Kanatharana, P.; Limbut, W.; Thammakhet, C.; Thavarungkul, P. A highly stable oxygen-independent glucose biosensor based on a chitosan-albumin cryogel incorporated with carbon nanotubes and ferrocene. Sens. Actuators B Chem. 2013, 185, 725–734. [Google Scholar] [CrossRef]
- Kan, X.; Zhou, H.; Li, C.; Zhu, A.; Xing, Z.; Zhao, Z. Imprinted electrochemical sensor for dopamine recognition and determination based on a carbon nanotube/polypyrrole film. Electrochim. Acta 2012, 63, 69–75. [Google Scholar] [CrossRef]
- Wang, Y.; Yeow, J.T. A review of carbon nanotubes-based gas sensors. J. Sens. 2009, 2009, 493904. [Google Scholar] [CrossRef]
- Jeong, H.Y.; Lee, D.-S.; Choi, H.K.; Lee, D.H.; Kim, J.-E.; Lee, J.Y.; Lee, W.J.; Kim, S.O.; Choi, S.-Y. Flexible room-temperature NO2 gas sensors based on carbon nanotubes/reduced graphene hybrid films. Appl. Phys. Lett. 2010, 96, 213105. [Google Scholar] [CrossRef]
- Abdulla, S.; Mathew, T.L.; Pullithadathil, B. Highly sensitive, room temperature gas sensor based on polyaniline-multiwalled carbon nanotubes (PANI/MWCNTs) nanocomposite for trace-level ammonia detection. Sens. Actuators B Chem. 2015, 221, 1523–1534. [Google Scholar] [CrossRef]
- Saito, Y.; Uemura, S. Field emission from carbon nanotubes and its application to electron sources. Carbon 2000, 38, 169–182. [Google Scholar] [CrossRef]
- Lee, N.; Chung, D.; Han, I.; Kang, J.; Choi, Y.; Kim, H.; Park, S.; Jin, Y.; Yi, W.; Yun, M. Application of carbon nanotubes to field emission displays. Diamond Relat. Mater. 2001, 10, 265–270. [Google Scholar] [CrossRef]
- De Heer, W.A.; Chatelain, A.; Ugarte, D. A carbon nanotube field-emission electron source. Science 1995, 270, 1179–1181. [Google Scholar] [CrossRef]
- Rosen, R.; Simendinger, W.; Debbault, C.; Shimoda, H.; Fleming, L.; Stoner, B.; Zhou, O. Application of carbon nanotubes as electrodes in gas discharge tubes. Appl. Phys. Lett. 2000, 76, 1668–1670. [Google Scholar] [CrossRef]
- Sugie, H.; Tanemura, M.; Filip, V.; Iwata, K.; Takahashi, K.; Okuyama, F. Carbon nanotubes as electron source in an X-ray tube. Appl. Phys. Lett. 2001, 78, 2578–2580. [Google Scholar] [CrossRef]
- Neupane, S.; Lastres, M.; Chiarella, M.; Li, W.; Su, Q.; Du, G. Synthesis and field emission properties of vertically aligned carbon nanotube arrays on copper. Carbon 2012, 50, 2641–2650. [Google Scholar] [CrossRef]
- Huang, Z.; Carnahan, D.; Rybczynski, J.; Giersig, M.; Sennett, M.; Wang, D.; Wen, J.; Kempa, K.; Ren, Z. Growth of large periodic arrays of carbon nanotubes. Appl. Phys. Lett. 2003, 82, 460–462. [Google Scholar] [CrossRef]
- Teo, K.; Lee, S.; Chhowalla, M.; Semet, V.; Binh, V.T.; Groening, O.; Castignolles, M.; Loiseau, A.; Pirio, G.; Legagneux, P. Plasma enhanced chemical vapour deposition carbon nanotubes/nanofibres—How uniform do they grow? Nanotechnology 2003, 14, 204. [Google Scholar] [CrossRef]
- Kim, D.-H.; Cho, D.-S.; Jang, H.-S.; Kim, C.-D.; Lee, H.-R. The growth of freestanding single carbon nanotube arrays. Nanotechnology 2003, 14, 1269. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Eastman, M.; Gutu, T.; Wyse, M.; Jiao, J.; Kim, S.-M.; Mann, M.; Zhang, Y.; Teo, K. Fabrication of carbon nanotube-based nanodevices using a combination technique of focused ion beam and plasma-enhanced chemical vapor deposition. Appl. Phys. Lett. 2007, 91, 173122. [Google Scholar] [CrossRef]
- Lee, C.J.; Kim, D.W.; Lee, T.J.; Choi, Y.C.; Park, Y.S.; Lee, Y.H.; Choi, W.B.; Lee, N.S.; Park, G.-S.; Kim, J.M. Synthesis of aligned carbon nanotubes using thermal chemical vapor deposition. Chem. Phys. Lett. 1999, 312, 461–468. [Google Scholar] [CrossRef]
- Dionne, M.; Coulombe, S.; Meunier, J.-L. Screening effects between field-enhancing patterned carbon nanotubes: A numerical study. IEEE Trans. Electron Devices 2008, 55, 1298–1305. [Google Scholar] [CrossRef]
- Wright, A.W. ART. VII.—On the production of Transparent Metallic Films by the Electrical Discharge in exhausted tubes. Am. J. Sci. Arts 1877, 13, 49. [Google Scholar] [CrossRef]
- Baedeker, K. Über die elektrische Leitfähigkeit und die thermoelektrische Kraft einiger Schwermetallverbindungen. Ann. Phys. 1907, 327, 749–766. [Google Scholar] [CrossRef]
- Hecht, D.S.; Hu, L.; Irvin, G. Emerging transparent electrodes based on thin films of carbon nanotubes, graphene, and metallic nanostructures. Adv. Mater. 2011, 23, 1482–1513. [Google Scholar] [CrossRef]
- Kolosov, O.; Gruverman, A.; Hatano, J.; Takahashi, K.; Tokumoto, H. Nanoscale visualization and control of ferroelectric domains by atomic force microscopy. Phys. Rev. Lett. 1995, 74, 4309. [Google Scholar] [CrossRef]
- Lanza, M.; Bayerl, A.; Gao, T.; Porti, M.; Nafria, M.; Jing, G.; Zhang, Y.; Liu, Z.; Duan, H. Graphene-Coated Atomic Force Microscope Tips for Reliable Nanoscale Electrical Characterization. Adv. Mater. 2013, 25, 1440–1444. [Google Scholar] [CrossRef]
- Stevens, R.M. New carbon nanotube AFM probe technology. Mater. Today 2009, 12, 42–45. [Google Scholar] [CrossRef]
- Slattery, A.D.; Shearer, C.J.; Gibson, C.T.; Shapter, J.G.; Lewis, D.A.; Stapleton, A.J. Carbon nanotube modified probes for stable and high sensitivity conductive atomic force microscopy. Nanotechnology 2016, 27, 475708. [Google Scholar] [CrossRef] [PubMed]
- Slattery, A.D.; Blanch, A.J.; Quinton, J.S.; Gibson, C.T. Efficient attachment of carbon nanotubes to conventional and high-frequency AFM probes enhanced by electron beam processes. Nanotechnology 2013, 24, 235705. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Haldane, D.; Liang, R.; Smithyman, J.; Zhang, C.; Wang, B. Nanoscale infiltration behaviour and through-thickness permeability of carbon nanotube buckypapers. Nanotechnology 2012, 24, 015704. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Kröger, M. A theoretical evaluation of the effects of carbon nanotube entanglement and bundling on the structural and mechanical properties of buckypaper. Carbon 2012, 50, 1793–1806. [Google Scholar] [CrossRef]
- He, S.; Wei, J.; Guo, F.; Xu, R.; Li, C.; Cui, X.; Zhu, H.; Wang, K.; Wu, D. A large area, flexible polyaniline/buckypaper composite with a core–shell structure for efficient supercapacitors. J. Mater. Chem. A 2014, 2, 5898–5902. [Google Scholar] [CrossRef]
- Lu, H.; Gou, J. Study on 3-D high conductive graphene buckypaper for electrical actuation of shape memory polymer. Nanosci. Nanotechnol. Lett. 2012, 4, 1155–1159. [Google Scholar] [CrossRef]
- Che, J.; Chen, P.; Chan-Park, M.B. High-strength carbon nanotube buckypaper composites as applied to free-standing electrodes for supercapacitors. J. Mater. Chem. A 2013, 1, 4057–4066. [Google Scholar] [CrossRef]
- Liu, Q.; Nayfeh, M.H.; Yau, S.-T. Brushed-on flexible supercapacitor sheets using a nanocomposite of polyaniline and carbon nanotubes. J. Power Sources 2010, 195, 7480–7483. [Google Scholar] [CrossRef]
- Chen, I.-W.P.; Yang, M.-C.; Yang, C.-H.; Zhong, D.-X.; Hsu, M.-C.; Chen, Y. Newton Output Blocking Force under Low-Voltage Stimulation for Carbon Nanotube–Electroactive Polymer Composite Artificial Muscles. ACS Appl. Mater. Interfaces 2017, 9, 5550–5555. [Google Scholar] [CrossRef]
- Schwengber, A.; Prado, H.J.; Zilli, D.A.; Bonelli, P.R.; Cukierman, A.L. Carbon nanotubes buckypapers for potential transdermal drug delivery. Mater. Sci. Eng. C 2015, 57, 7–13. [Google Scholar] [CrossRef]
- Aqil, A.; Vlad, A.; Piedboeuf, M.-L.; Aqil, M.; Job, N.; Melinte, S.; Detrembleur, C.; Jérôme, C. A new design of organic radical batteries (ORBs): Carbon nanotube buckypaper electrode functionalized by electrografting. Chem. Commun. 2015, 51, 9301–9304. [Google Scholar] [CrossRef] [PubMed]
- Giubileo, F.; Iemmo, L.; Luongo, G.; Martucciello, N.; Raimondo, M.; Guadagno, L.; Passacantando, M.; Lafdi, K.; Di Bartolomeo, A. Transport and field emission properties of buckypapers obtained from aligned carbon nanotubes. J. Mater. Sci. 2017, 52, 6459–6468. [Google Scholar] [CrossRef]
- Wu, Q.; Zhu, W.; Zhang, C.; Liang, Z.; Wang, B. Study of fire retardant behavior of carbon nanotube membranes and carbon nanofiber paper in carbon fiber reinforced epoxy composites. Carbon 2010, 48, 1799–1806. [Google Scholar] [CrossRef]
- Yang, X.; Lee, J.; Yuan, L.; Chae, S.-R.; Peterson, V.K.; Minett, A.I.; Yin, Y.; Harris, A.T. Removal of natural organic matter in water using functionalised carbon nanotube buckypaper. Carbon 2013, 59, 160–166. [Google Scholar] [CrossRef]
- Rashid, M.H.-O.; Pham, S.Q.; Sweetman, L.J.; Alcock, L.J.; Wise, A.; Nghiem, L.D.; Triani, G.; in het Panhuis, M.; Ralph, S.F. Synthesis, properties, water and solute permeability of MWNT buckypapers. J. Membr. Sci. 2014, 456, 175–184. [Google Scholar] [CrossRef]
- Wen, G.; Yu, H.; Huang, X. Synthesis of carbon microtube buckypaper by a gas pressure enhanced chemical vapor deposition method. Carbon 2011, 49, 4067–4069. [Google Scholar] [CrossRef]
- LeMieux, M.C.; Roberts, M.; Barman, S.; Jin, Y.W.; Kim, J.M.; Bao, Z. Self-sorted, aligned nanotube networks for thin-film transistors. Science 2008, 321, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.U.; Kausar, A.; Ullah, H.; Badshah, A.; Khan, W.U. A review of graphene oxide, graphene buckypaper, and polymer/graphene composites: Properties and fabrication techniques. J. Plast. Film Sheeting 2016, 32, 336–379. [Google Scholar] [CrossRef]






| SWNT | MWNT |
|---|---|
| Single graphene layer | Multiple graphene layers |
| Synthesis requires catalyst | No catalyst is required |
| Difficult bulk synthesis due to the requirement of appropriate growth and atmospheric condition. | Easy bulk synthesis |
| Poor purity | High purity |
| Greater chances of defects during functionalization | Lesser defect chances but when this occurs, it is hard to recover |
| Aggregation in the body is less | Aggregation in the body is greater |
| Easy assessment and characterization | Structure is complicated |
| More pliable and easily twisted | Twisting is not easy |
| Adsorbents | Metal Ions | Adsorption Capacity (mg g−1) | Reference |
|---|---|---|---|
| CNTs | Pb2+ | 17.44 (pH 7.0) | [151] |
| CNTs (HNO3) | Pb2+ | 49.95 (pH 7.0) | [152] |
| MWCNTs | Ni2+ | 7.53 (pH 7.0) | [153] |
| MWCNTs (HNO3) | Pb2+ | 97.08 (pH 5.0) | [154] |
| CNT–OH | Cu2+ | 1.342 | [148] |
| CNT–COO− | Cd2+ | 3.325 | [148] |
| CNT–CONH | Hg2+ | 1.658 | [148] |
| CNT–COO− | Hg2+ | 3.300 | [148] |
| Type of CNTs | Organic Pollutants | Adsorption Capacity (mg g−1) | Reference |
|---|---|---|---|
| Alkali-activated MWCNTs | Methylene blue | 399 | [159] |
| Untreated MWCNTs | Methylene blue | 59.7 | [160] |
| Untreated SWCNTs | Dissolved organic matter (DOM) | 26.1–20.8 | [161] |
| Calcium alginate/MWCNTs | Methyl orange | 12.5 | [162] |
| Carboxylated multiwalled carbon nanotubes | Norfloxacin | 90.3 | [163] |
| Single-, double-, and multiwalled carbon nanotubes | Ciprofloxacin | 933.8, 901.2, 651.4 | [164] |
| Pristine and hydroxylated MWCNTs | Sulfamethazine | 24.78, 13.31 | [165] |
| Type of CNT | Materials Type | Type of Cell or Tissue | Properties | Reference |
|---|---|---|---|---|
| Porous SWCNT | Polycarbonate membrane | Osteoblast-like cells | Enhances lamellipoda (cytoskeletal) extensions, and lamellipodia extensions | [181] |
| SWCNT-incorporated | Chitosan scaffolds | C2C12 cells/C2 myogenic cell line | Improvement in cell growth | [182] |
| MWCNT | Collagen sponge honeycomb scaffold | MC3T3-E1 cells, a mouse osteoblast-like cell line | Enhances proliferation and cellular adhesion | [183] |
| MWCNT | Polyurethane | Fibroblast cells | Improve cellular interactions and the polyurethane surface | [184] |
| MWCNT | Alginate | Rat heart endothelial cell | Improvement in cellular adhesion and proliferation | [185] |
| MWCNT | Poly(acrylic acid) | Human embryonic stem cells | Increase cellular differentiation toward neurons | [186] |
| SWCNT | Propylene fumarate | Rabbit tibia | Support the attachment of cells and proliferation | [187] |
| Carbon nanotube | Biomarker | Form of Cancer | Reference |
|---|---|---|---|
| Multilabel secondary antibody-nanotube bioconjugates | Prostate-specific antigen (PSA) | prostate cancer | [188] |
| Microelectrode arrays modified with single-walled carbon nanotubes (SWNTs) | Total prostate-specific antigen (T-PSA) | Prostate cancer | [188] |
| Multiwalled carbon nanotubes–thionine–chitosan (MWCNTs–THI–CHIT) nanocomposite film | Chlorpyrifos residues | Many forms | [189] |
| Tris(2,2′-bipyridyl)cobalt(III) (Co(bpy)33+)–MWNTs–Nafion composite film | Carcinoma antigen-125 (CA125) | Carcinoma | [190] |
| Carbon nanotube field effect transistor (CNT-FET) | Prostate-specific antigen (PSA) | Prostate cancer | [191] |
| Multiple enzyme layers assembled multiwall carbon nanotubes (MWCNTs) | Alpha-fetoprotein (AFP) | Many forms | [192] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahman, G.; Najaf, Z.; Mehmood, A.; Bilal, S.; Shah, A.u.H.A.; Mian, S.A.; Ali, G. An Overview of the Recent Progress in the Synthesis and Applications of Carbon Nanotubes. C 2019, 5, 3. https://doi.org/10.3390/c5010003
Rahman G, Najaf Z, Mehmood A, Bilal S, Shah AuHA, Mian SA, Ali G. An Overview of the Recent Progress in the Synthesis and Applications of Carbon Nanotubes. C. 2019; 5(1):3. https://doi.org/10.3390/c5010003
Chicago/Turabian StyleRahman, Gul, Zainab Najaf, Asad Mehmood, Salma Bilal, Anwar ul Haq Ali Shah, Shabeer Ahmad Mian, and Ghulam Ali. 2019. "An Overview of the Recent Progress in the Synthesis and Applications of Carbon Nanotubes" C 5, no. 1: 3. https://doi.org/10.3390/c5010003
APA StyleRahman, G., Najaf, Z., Mehmood, A., Bilal, S., Shah, A. u. H. A., Mian, S. A., & Ali, G. (2019). An Overview of the Recent Progress in the Synthesis and Applications of Carbon Nanotubes. C, 5(1), 3. https://doi.org/10.3390/c5010003