Adsorption Isotherms
The data of the adsorption equilibrium allows the determination of the amount of compound that can be adsorbed in each activated carbon once the equilibrium time has been reached. In general, once the adsorption isotherms are determined, it is possible to adjust the data to a mathematical model that describes the experimental behavior from chemical, kinetic, or thermodynamic assumptions.
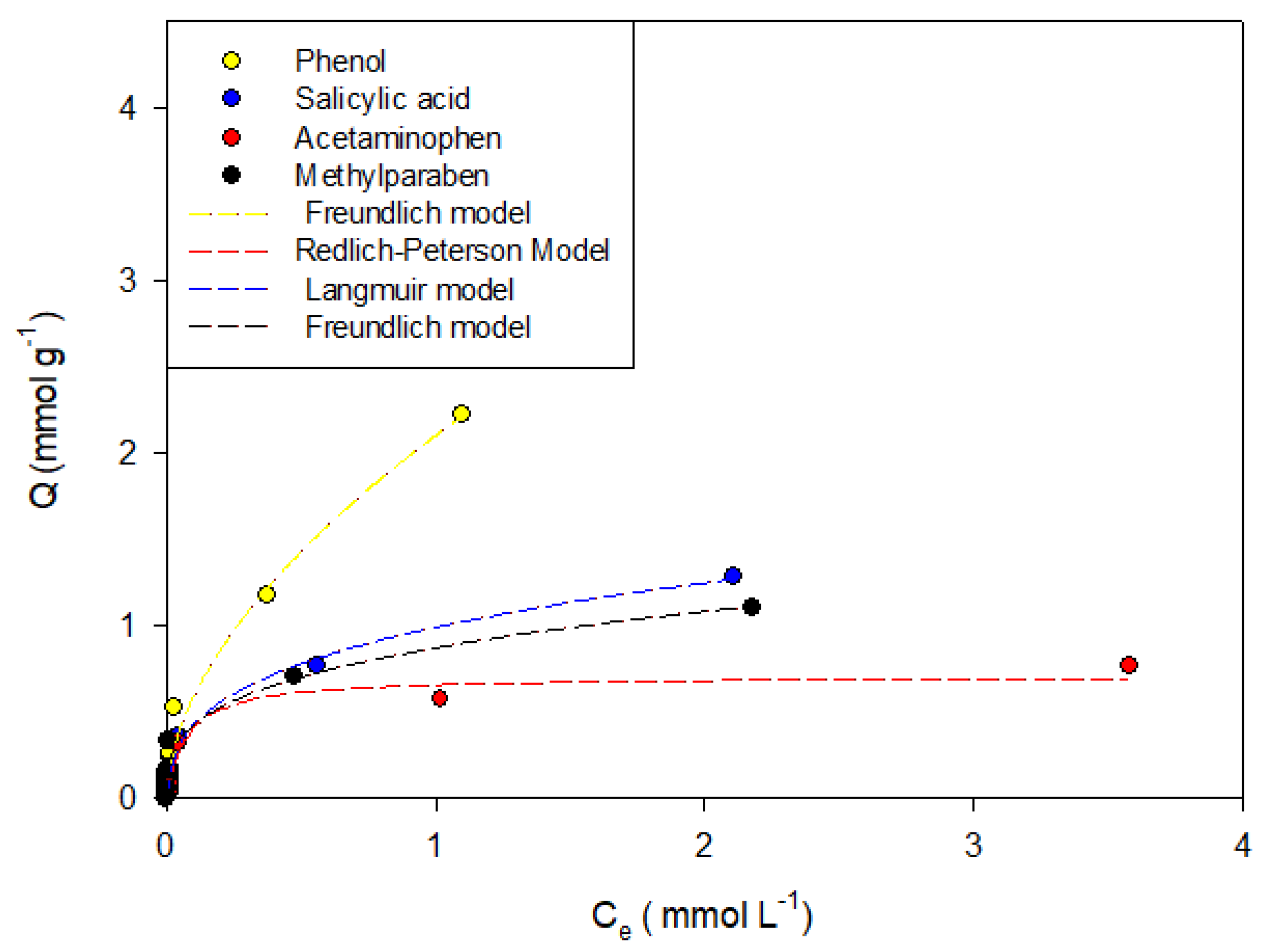
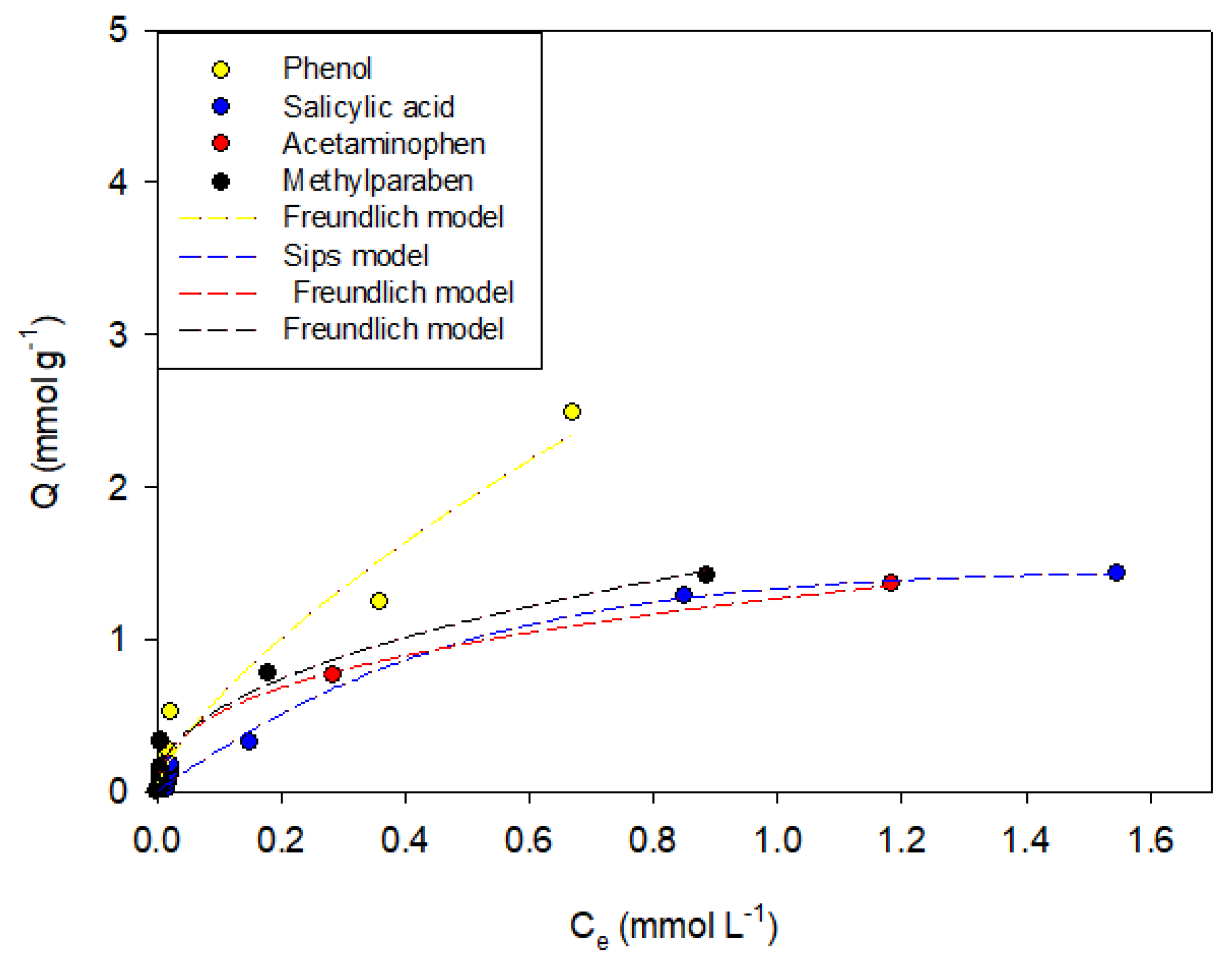
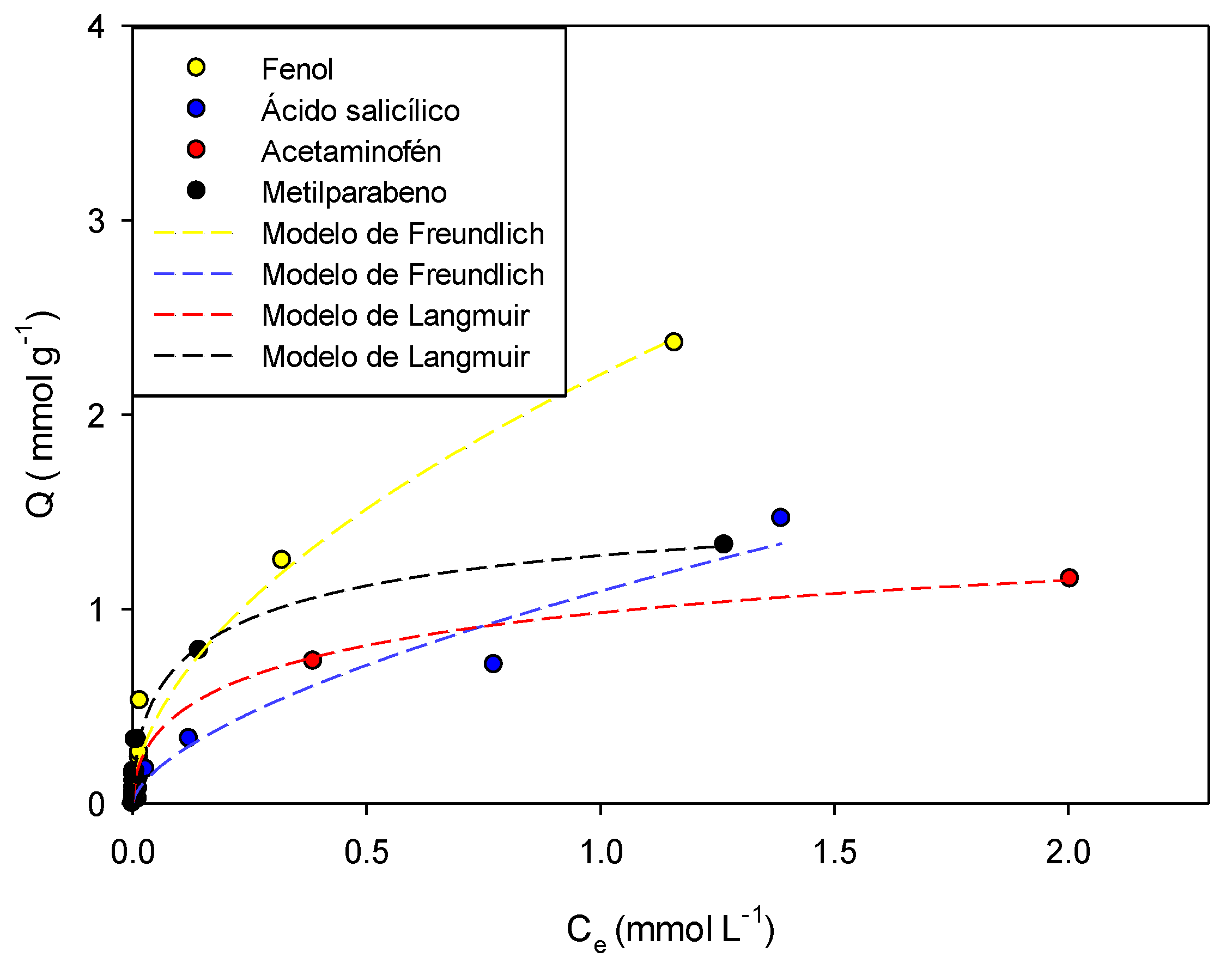
Figure 3,
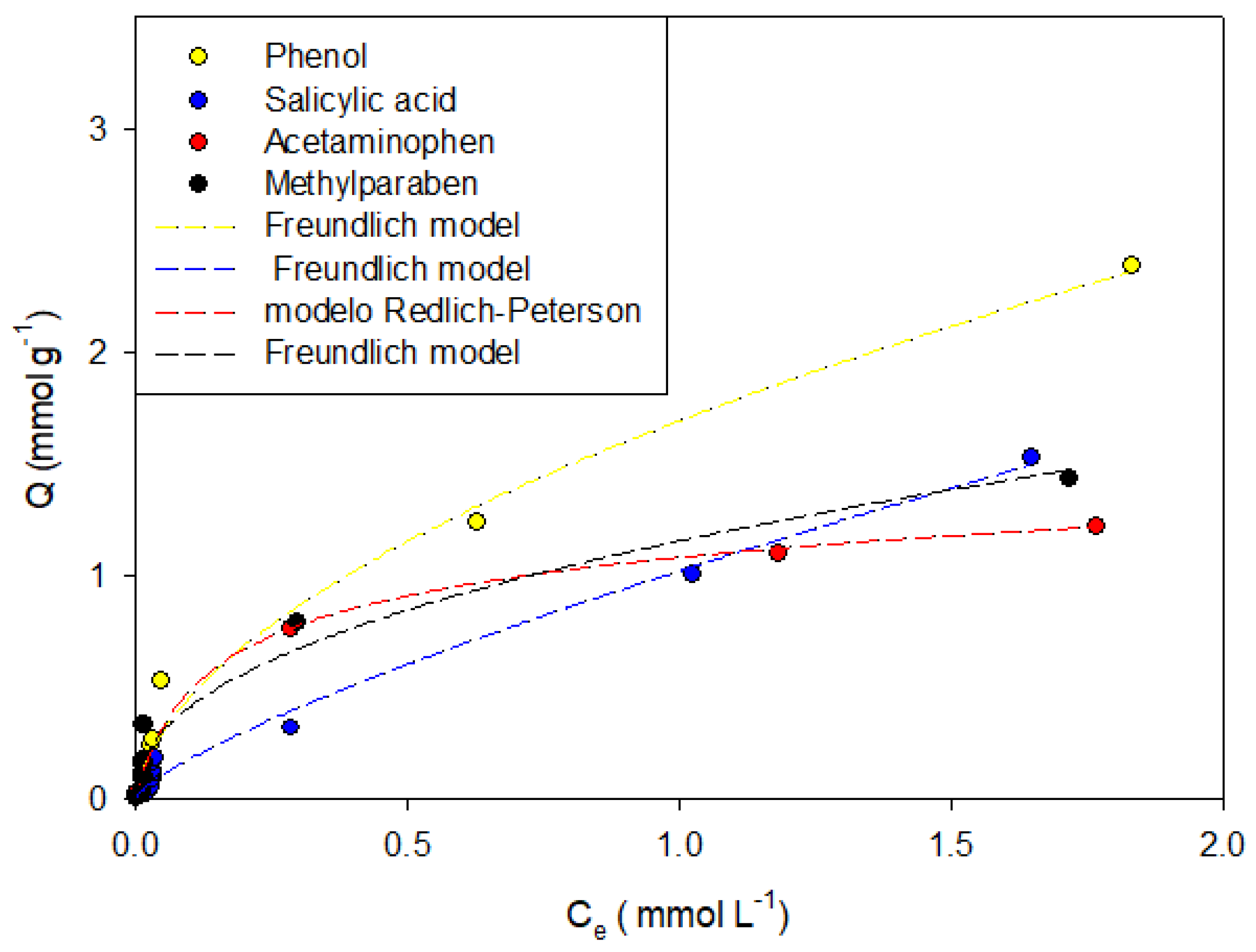
Figure 4,
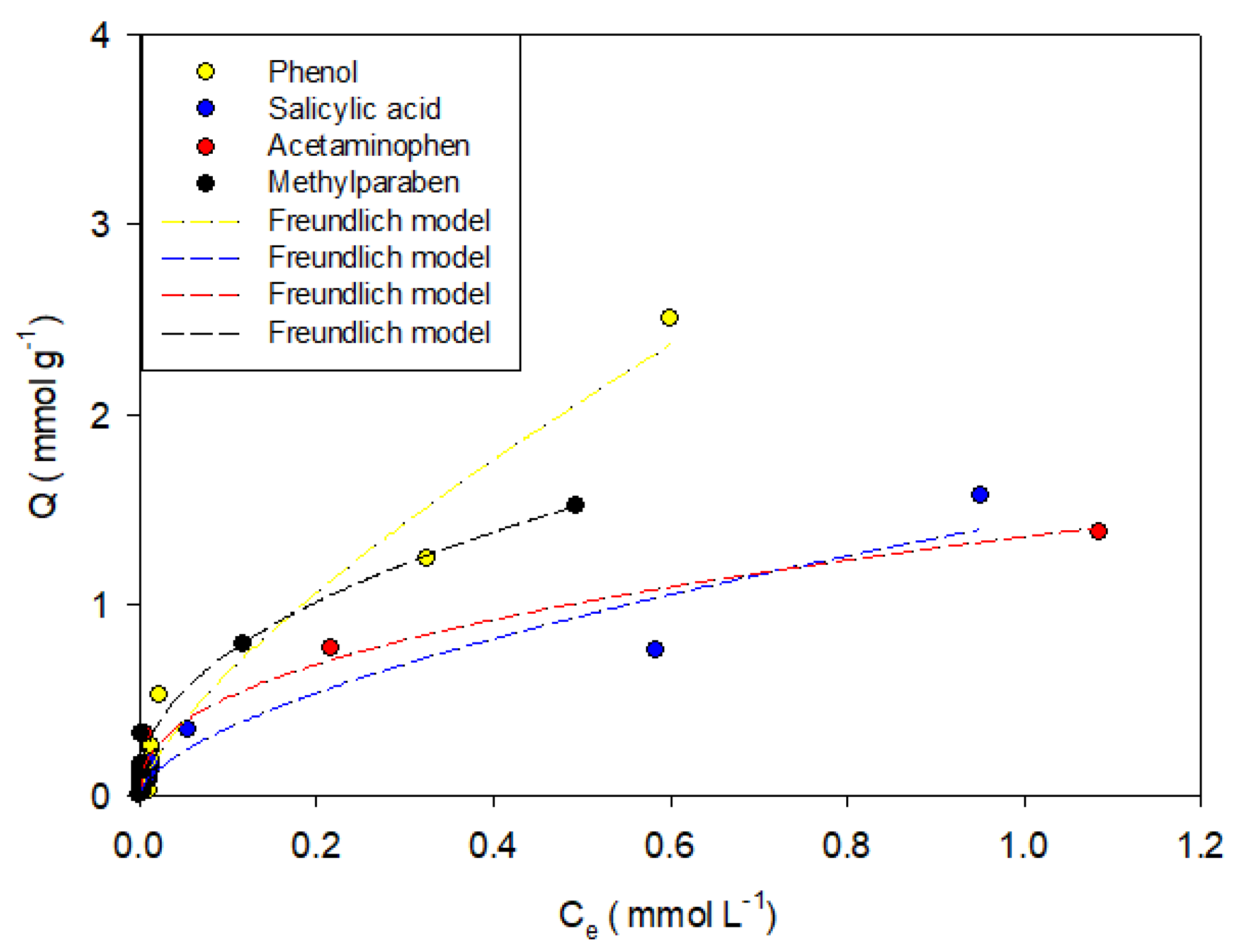
Figure 5,
Figure 6 and
Figure 7 show the adsorption isotherms of phenol, salicylic acid, acetaminophen, and methylparaben in the activated carbons CAO, CAG, CAR1073, CAR1173, and CAR1273.
The selection of the adsorption models used allows the description of different types of systems. The Langmuir model is used for adsorption processes where the adsorbate-adsorbent interactions are specific, and all active sites are energetically equivalent; therefore, the process are considered homogeneous.
The Freundlich, Sips, and Redlich-Peterson models describe systems that present high energetic heterogeneity associated with the formation of nonspecific interactions that include lateral and associative interactions. In the Freundlich model, it is assumed that the adsorbate interacts with different active sites that differ in the energy they possess; therefore, the energy of the process is not constant.
The Sips and Redlich-Peterson models are derived from the Freundlich and Langmuir models, allowing the description of systems that exhibit intermediate behavior [
26,
27,
28].
The models of Freundlich and Redlich–Peterson do not include in their parameters the adsorption capacity; however, by replacing in the mathematical equations the calculated parameters of each model and taking Ce as the higher equilibrium concentration, it is possible to calculate a theoretical capacity of adsorption. The results are shown in
Table 13.
It can be observed that the adsorption isotherms presented in
Figure 3,
Figure 4,
Figure 5,
Figure 6 and
Figure 7 are mainly fitted to the adsorption models that contemplate within their parameters the energetic heterogeneity derived from the physicochemical characteristics of the system. The Freundlich model fits the adsorption data of most systems; this indicates that the process is carried out by the formation of multilayers with heterogeneous distributions of the adsorption enthalpy. According to the model, the energy decreases exponentially and remains constant once the process has reached equilibrium [
29]. From the point of view of statistical thermodynamics, the value of the heterogeneity factor (n) depends on the coordination number of the adsorbate, adsorbate-adsorbate interactions, the Boltzmann constant, and Avogadro’s number; therefore, the heterogeneity of the systems depends on the binding energy of the active sites and the formation of lateral interactions. Values of n that tend to 0 indicate physisorption, while values close to 1 indicate chemisorption or cooperative adsorption [
30]. In the case of Redlich-Peterson and Sips models, when n tends to 0 the equation takes the form of the Freundlich model and when it tends to 1 the Langmuir model is adopted.
Additionally, the energetic heterogeneity of the systems is attributed to the competition between the solvent and the adsorbate for the active sites, which means that the enthalpy of adsorption is not constant throughout the process. Likewise, the change in the adjustment models shows that the adsorption depends on the type of adsorbate–adsorbent interactions, which not only depends on the chemical groups present in the adsorbent.
It was found that phenol adsorption can be described in all systems with the Freundlich models, indicating that the process is highly heterogeneous, which in turn is indicative of the formation of adsorbent-adsorbate, adsorbate–solvent interactions and interactions between molecules that are already adsorbed, which in this case may be hydrogen bonds. This same trend describes the behaviors of salicylic acid, methylparaben, and acetaminophen. However, the adsorption of the latter compound on the activated carbons CAO and CAR1273 are described with the Langmuir models, indicating specific adsorbate–adsorbent interactions, so that the adsorption capacity decreases.
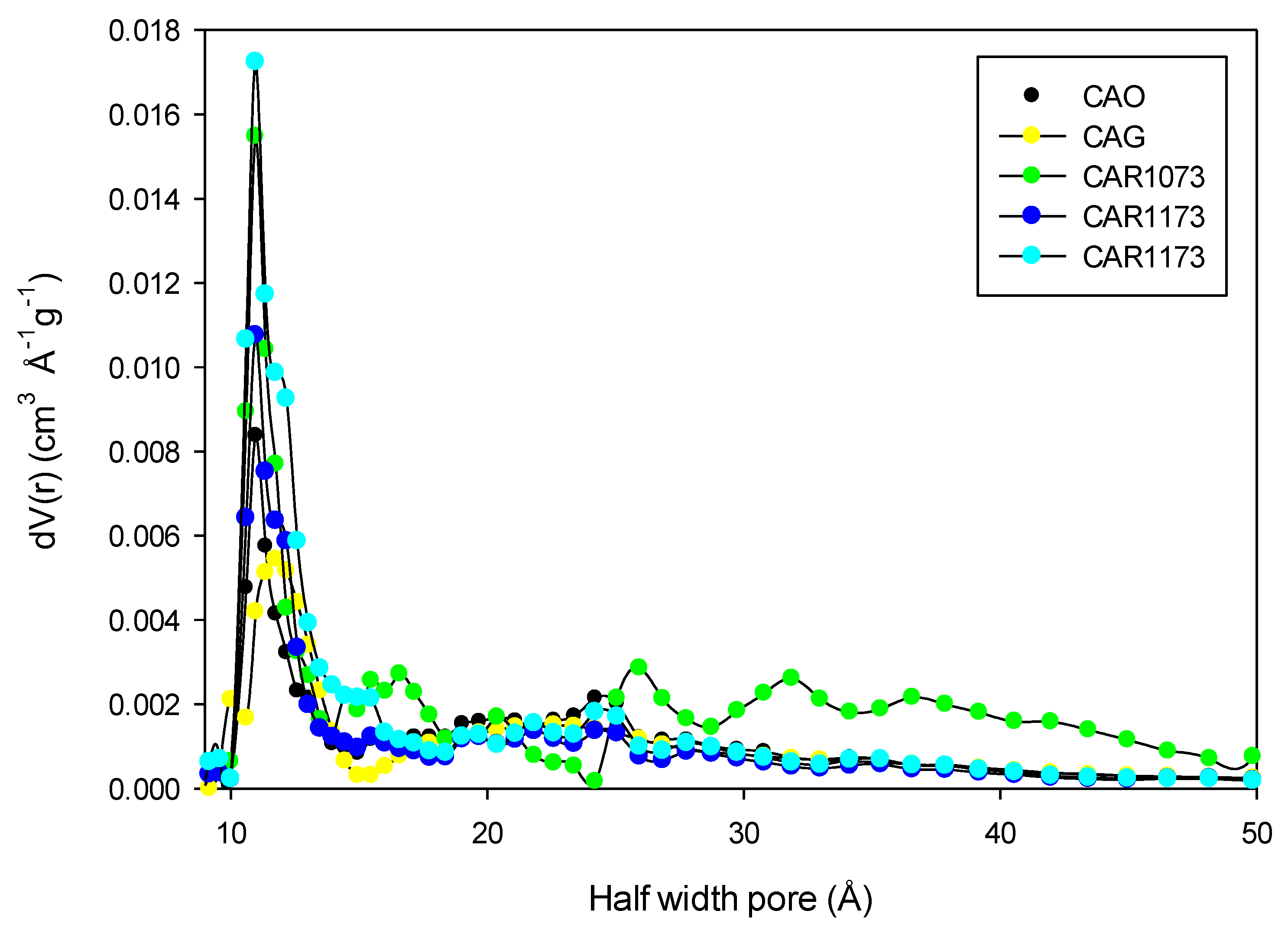
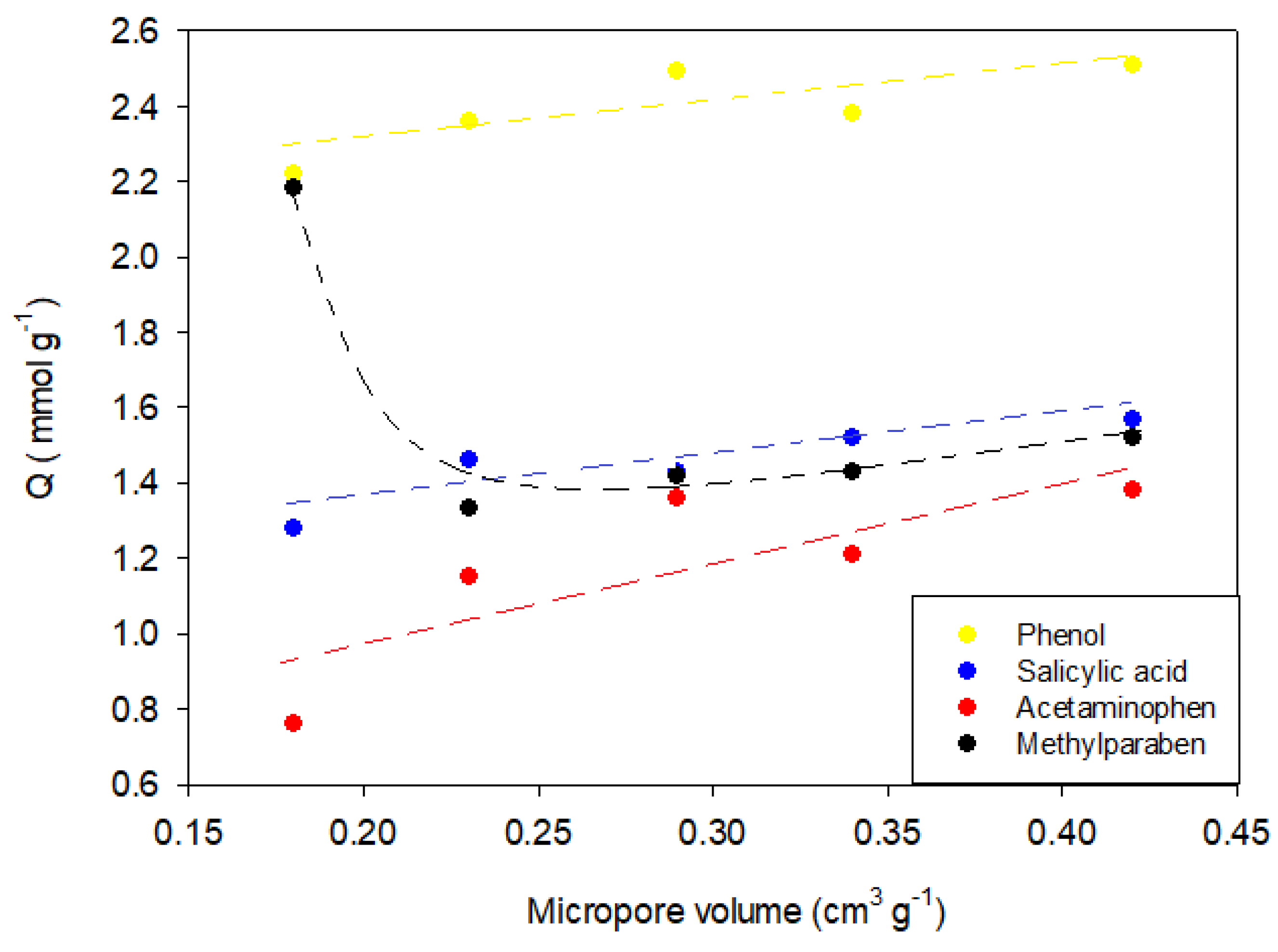
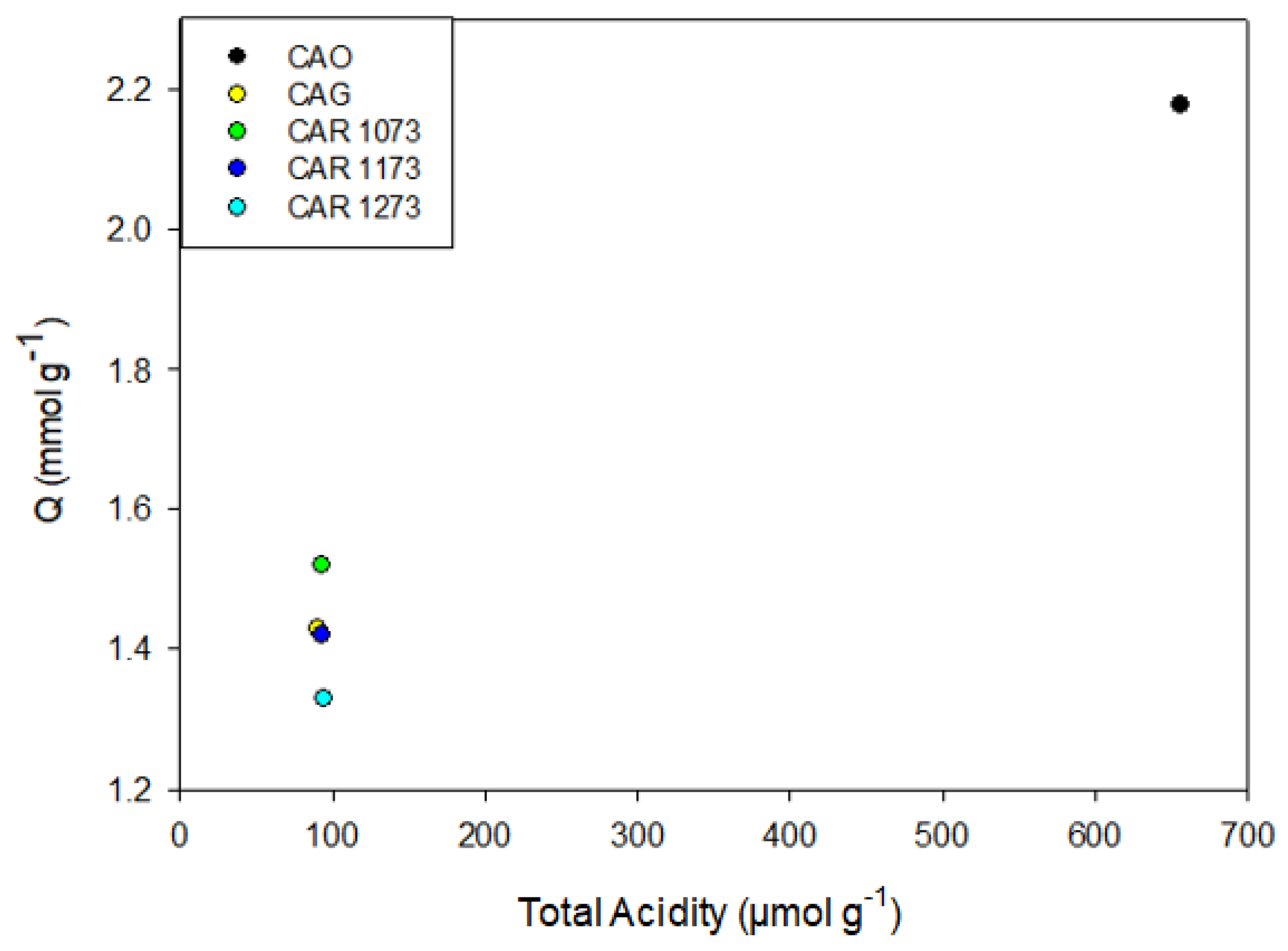
To determine the effect of the textural characteristics on the adsorption capacity, the adsorption capacity was associated with the micropore volume.
Figure 8 shows the results obtained.
Figure 8 shows a directly proportional behavior between the adsorption capacity and the micropore volume. This indicates that the process of adsorption of the study compounds is related to the filling of micropores according to the energy they possess. In the case of acetaminophen and phenol, adsorption on CAR1173 was not found to present this tendency, since the capacity of adsorption in this case was greater than that of activated carbon CAG, even though the latter has a greater surface area and micropore volume (
Table 4). A similar behavior was presented by methylparaben, whose adsorption capacity was greater in the activated carbon CAO, which presented the lowest values of surface area and micropore volume. These deviations from the general behavior were attributed to the effect of surface chemistry on the adsorption capacity.
According to the data in
Table 4, the activated carbons studied are microporous, which could limit the process of adsorption of large molecules, although this is not the case of phenol, salicylic acid, acetaminophen and methylparaben, which have small dimensions. The presence of the aromatic substituents could generate steric hindrance, so the effect of the substituent on the adsorption capacity of the adsorbates on activated carbons CAO, CAG, CAR1073, CAR1173, and CAR1273 was evaluated.
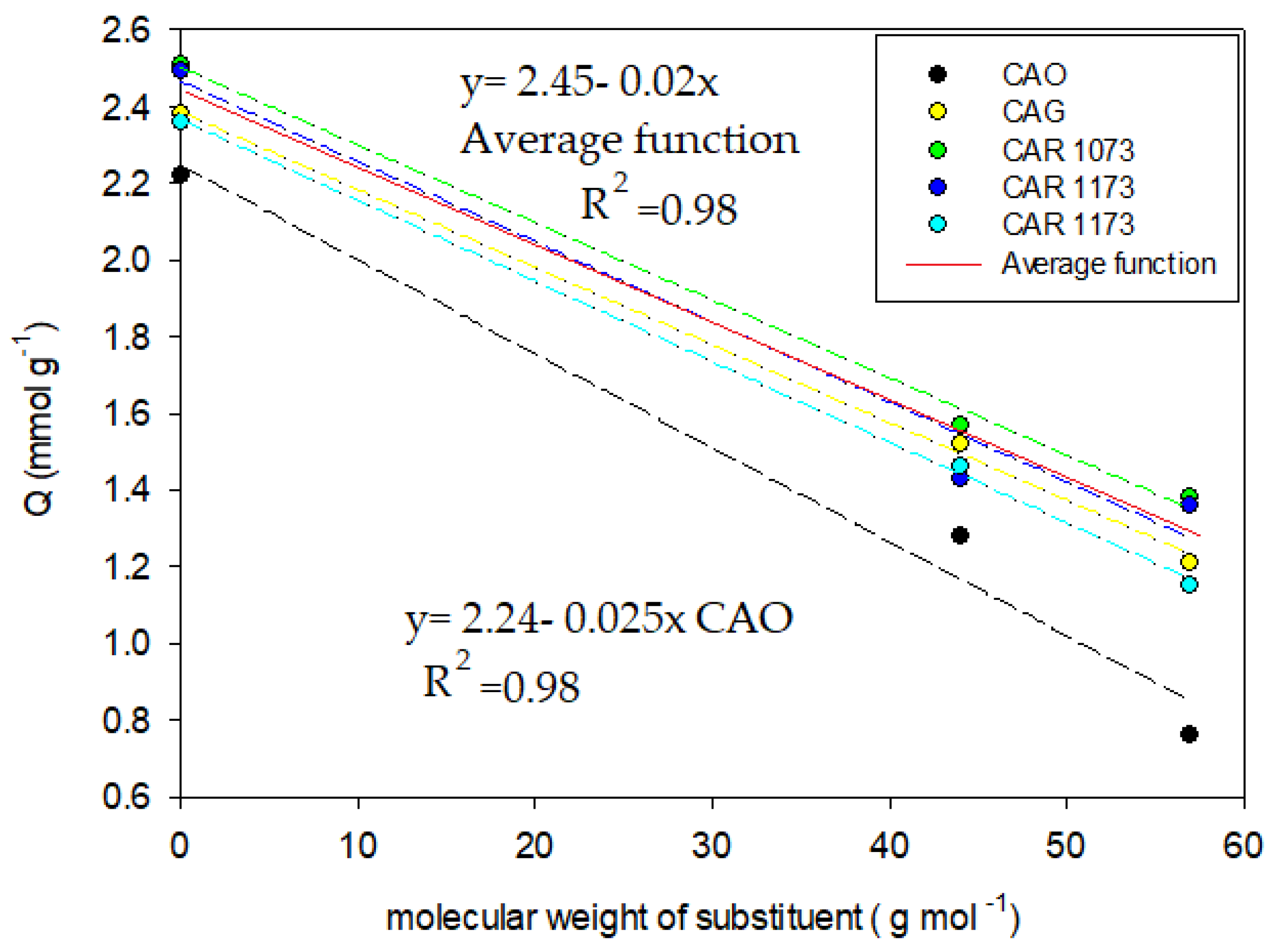
In
Figure 9, it is evident that the behavior between the adsorption capacity and the weight of the substituent is inversely proportional in all activated carbons, as indicated by Lundelius’ rule. For the correlations presented, the slope represents the rate of change in the adsorption capacity for each gram of additional substituent from the phenolic ring. The intercept corresponds to the contribution of the phenolic ring to the adsorption capacity. It is evident that the effect of the weight of the substituent in the adsorption capacity (slope) does not vary between the activated carbons CAG, CAR1073, CAR1173, and CAR1273; therefore, a linear function can be employed to describe the general behavior in the four activated carbons. CAO presents a slope different from that of the other activated carbons; as such, its behavior was described with a different equation.
In
Figure 9 it can be seen that the adsorption capacity (Q) decreases with the molecular weight of the substituent (Q phenol > Q salicylic acid = carboxylic acid > Q acetaminophen = amide ≥ Q methylparaben = ester). Also, according to the slope of the equations, the adsorption capacity varies by 0.02 mmol g
−1 for each gram from the substituent in the phenolic ring. In the activated carbon CAO, the adsorption capacity decreased by 25% with the increase in the molecular weight of the substituent (b = 0.025 mmol g
−1). This may be related to the decrease in the volume of micropores, but also to the presence of oxygenated groups in the entries of this type of porosity, which generates steric hindrance with the substituents in the adsorbates.
The process of adsorption of organic compounds from aqueous solutions can involve two driving forces: a derivative of the hydrophobicity of the solute that induces adsorption by low adsorbate-solvent affinity and the specific affinity of the adsorbate for the surface, which contemplates the adsorption by ionic interactions, by van der Waals forces, and by chemical reactions. Therefore, the adsorbate–adsorbent affinity depends on the surface chemistry of the activated carbon.
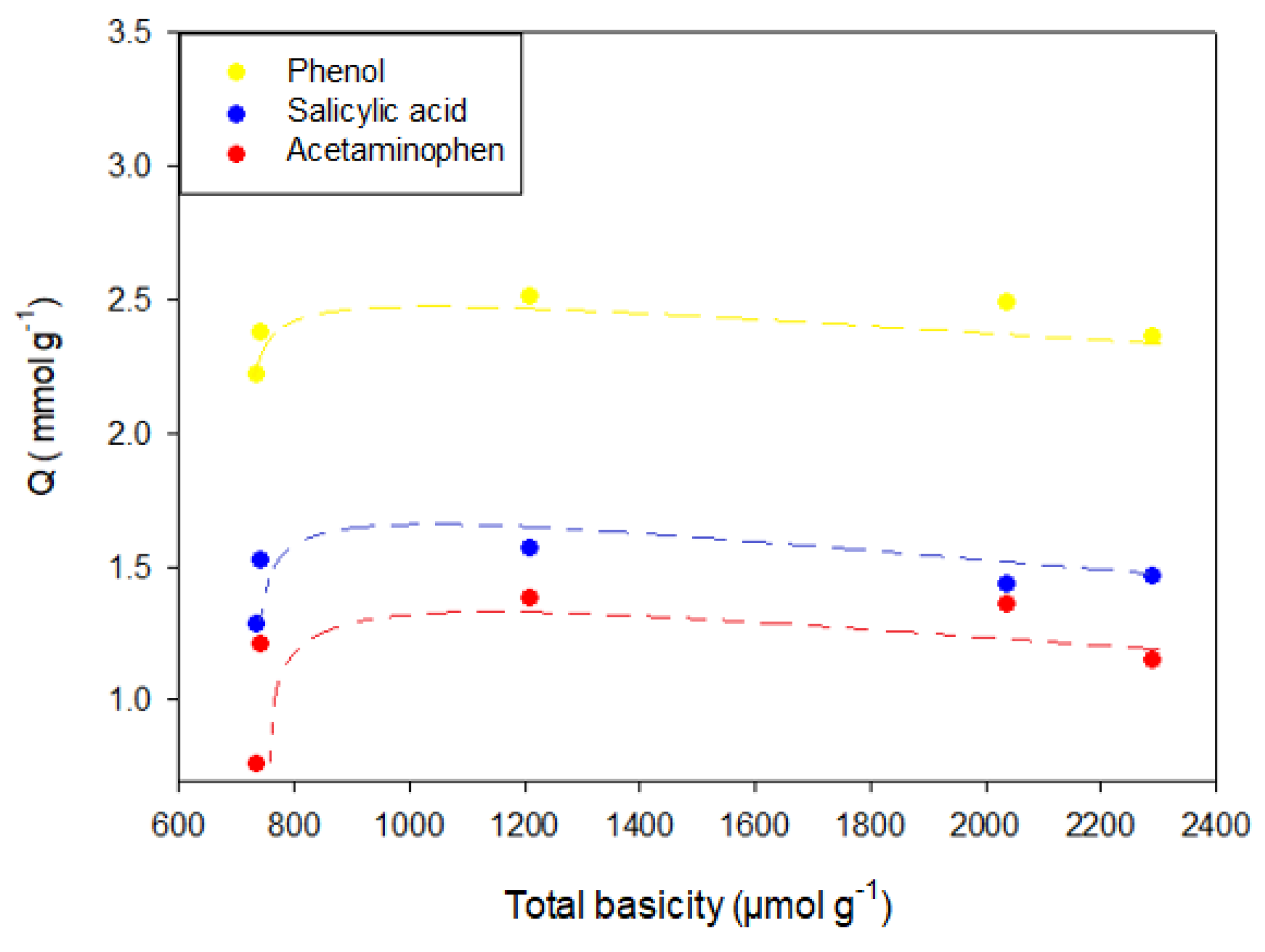
To analyze the correlation between the adsorbed amount and the functional groups present in the activated carbons, it was determined that the adsorbed amount of phenol, salicylic acid, and acetaminophen increases in a manner that is inversely proportional to the total acidity of the activated carbons.
Figure 10 shows the relationship between the total basicity and the adsorbed amount of phenol, salicylic acid, and acetaminophen.
The behavior described in
Figure 10 can be explained by the formation of acid-base interactions and the surface charge of the adsorbent. Surface groups such as amines have a pair of free electrons not compromised in the aromaticity of the aromatic rings of the graphene layers, which allows them to interact with Lewis acids such as phenol that could accept a pair of electrons. In the case of oxidized activated carbon, the adsorbed amount of the compound decreases because this adsorbent in solution yields H
+ ions that decrease the pH, which changes the surface charge of the adsorbent. This implies that when the basic groups are protonated, the electronic pair that was free binds to the hydrogen and therefore decreases the amount of groups available to interact with the adsorbate. Likewise, the amount of H
+ ions in the medium interacts with the π electrons of the graphene layers, preventing the formation of adsorbate-adsorbent interactions by π electrons. On the other hand, if the pH of the medium is above the pH at the point of zero charge, the adsorbent will have a negative charge associated with the deprotonation of the acid groups, implying a greater polarity at the surface, which favors the interaction with water. Therefore, as the activated carbons give less H
+ ions to the medium, the adsorption capacity of the pharmaceutical compounds is increased until it remains constant in the reduced activated carbons, which have the same values in the total acidity.
It was found that the adsorption capacity decreases with the molecular weight of the adsorbate, so molecules with higher molecular size may have problems of dissemination and therefore the adsorption depends on the surface chemistry. In the case of acetaminophen, it was envisaged that the surface chemistry of activated charcoal CAO disfavors the process altogether, in part because of the negative charge of carboxylic acids and the increase in the polarity of the surface, which favors the interactions with the solvent.
Methylparaben and acetaminophen have similar molecular weights; therefore, similar to the latter adsorbate, adsorption depends the interactions between the adsorbate and surface groups. However, contrary to phenol, salicylic acid, and acetaminophen, the adsorption capacity of methylparaben increases with the number of oxygenated groups. To analyze the different correlations between the acid groups and the amount adsorbed, it was determined that there is a directly proportional correlation between the amount of acid groups and the adsorbed amount, which can be observed in
Figure 11.
This behavior was attributed to the hydrolysis of the lactone groups in an acidic medium, which form a carboxylic acid and an alcohol after the reaction. This favors the formation of a greater amount of hydrogen bonds with the phenol in the adsorbate structure; likewise, the formation of phenols prevents the solvent from competing for the adsorption sites created, since specific interactions are formed any carboxylic acid-methylparaben and solvent–phenol interactions. As the pH increases, the hydrolysis of the lactones decreases and therefore the adsorption capacity decreases as well. On the other hand, the increase of pH in the medium favors the formation of the dissociated species, generating repulsions with carboxylic acids on the surface.