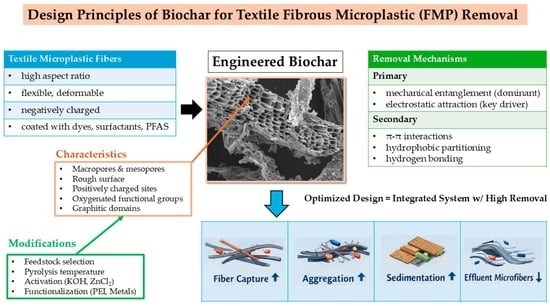
Engineered Biochar for the Sequestration of Textile Fibrous Microplastics: From Mechanistic Insights to Rational Functional Design
Abstract
1. Introduction
2. Physicochemical and Morphological Determinants of Textile FMPs
2.1. Compositional Heterogeneity and Reactive Surfaces
2.2. Geometric Anisotropy and Anomalous Hydrodynamics
2.3. Weathering Dynamics and Variable Buoyancy
3. Mechanisms of Biochar-Mediated FMP Sequestration
3.1. Hierarchical Interception and Mechanical Bridging
3.2. Electrostatic Coupling and Charge-Reversal Strategies
3.3. Aromatic Partitioning and π–π Interactions
3.4. Longitudinal Multi-Point Attachment and Cooperative Binding
3.5. Hetero-Aggregation, Flocculation, and Ballasting Effects
4. Rational Design of Biochar for Targeted FMP Sequestration
4.1. Hierarchical Porosity and Morphological Complementarity
4.2. Surface Charge Engineering and Cationic Functionalization
4.3. Graphitic Degree and π–π Affinity Optimization
4.4. Amphiphilicity Modulation and Surface Free Energy Alignment
4.5. Ballasting Potential and Skeletal Density Modulation
5. Critical Barriers and Future Research Frontiers
5.1. The Matrix Effect and Competitive Adsorption Dynamics
5.1.1. Competitive Sorption and Pore Blockage by DOM
5.1.2. Eco-Corona and Interfacial Masking
5.1.3. Strategic Research Imperatives
5.2. Hydraulic Scalability and Contact Time Limitations
5.2.1. Kinetic Mismatch and Collision Efficiency
5.2.2. The Granulometry Dilemma Between Permeability and Interfacial Area
5.2.3. Strategic Engineering for Hydraulic Integration
5.3. Lifecycle Management and the Spent Sorbent Dilemma
5.3.1. Secondary Contamination and Leaching Kinetics
5.3.2. Thermochemical Regeneration and Toxicological Risks
5.3.3. Future Directions: Circularity and Encapsulation
5.4. Standardization of Analytical Metrics and Reporting Protocols
5.4.1. The Mass-Count Duality and Fragmentation Paradox
5.4.2. Methodological Inconsistency and Inter-Laboratory Comparability
5.4.3. Future Direction: Toward a Unified Reporting Framework
- (1)
- Influent and effluent characterization: reporting both mass concentration (µg/L or mg/L) and particle counts (fibers/L), along with polymer identification. Such information enables a comparison between studies using both gravimetric and count-based metrics and captures the environmental relevance of small fibers that contribute little mass but large counts.
- (2)
- Fiber morphology metrics: including length distribution, diameter range, and aspect ratio before and after treatment. Such information provides insight into the capture mechanisms (e.g., entanglement, interception, adsorption) and enables the evaluation of treatment-induced fragmentation.
- (3)
- Operational parameters: detailed documentation of hydraulic retention time, mixing intensity, contact time, sorbent dosage (g/L), temperature, pH, and ionic strength. Such information ensures reproducibility and allows performance comparisons across reactor configurations and water chemistry conditions.
- (4)
- Analytical pretreatment methods: explicit reporting of organic matter digestion methods, filtration pore size, and contamination control procedures. Such information prevents analytical artifacts such as fiber degradation or loss during sample preparation.
- (5)
- Polymer identification: spectroscopic confirmation of polymer composition using techniques such as µ-FTIR or Raman spectroscopy. Such information confirms polymer identity and distinguishes synthetic fibers from natural or semi-synthetic materials.
- (6)
- Reporting of detection limits and size thresholds: specifying the smallest detectable fiber size and associated analytical uncertainty.
- (7)
- Breakthrough and fragmentation analysis: characterization of whether treatment leads to complete capture, partial retention, or mechanical fragmentation of fibers.
5.5. Economic Feasibility of Designer Biochars
5.5.1. Cost-Performance Trade-Offs in Advanced Synthesis
5.5.2. Scarcity of Robust Techno-Economic Analyses (TEA)
5.5.3. Future Direction: Valorization of Industrial Byproducts
6. Conclusions and Outlook
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nihei, Y.; Ota, H.; Tanaka, M.; Kataoka, T.; Kashiwada, J. Comparison of concentration, shape, and polymer composition between microplastics and mesoplastics in Japanese river waters. Water Res. 2024, 249, 120979. [Google Scholar] [CrossRef]
- Zhao, S.; Kvale, K.F.; Zhu, L.; Zettler, E.R.; Egger, M.; Mincer, T.J.; Amaral-Zettler, L.A.; Lebreton, L.; Niemann, H.; Nakajima, R. The distribution of subsurface microplastics in the ocean. Nature 2025, 641, 51–61. [Google Scholar] [CrossRef]
- Wang, T.; Li, B.; Shi, H.; Ding, Y.; Chen, H.; Yuan, F.; Liu, R.; Zou, X. The processes and transport fluxes of land-based macroplastics and microplastics entering the ocean via rivers. J. Hazard. Mater. 2024, 466, 133623. [Google Scholar] [CrossRef]
- Chen, L.; Zhou, S.; Zhang, Q.; Su, B.; Yin, Q.; Zou, M. Global occurrence characteristics, drivers, and environmental risk assessment of microplastics in lakes: A meta-analysis. Environ. Pollut. 2024, 344, 123321. [Google Scholar] [CrossRef]
- Chowdhury, S.R.; Razzak, S.A.; Hassan, I.; Hossain, S.Z.; Hossain, M.M. Microplastics in freshwater and drinking water: Sources, impacts, detection, and removal strategies. Water Air Soil Pollut. 2023, 234, 673. [Google Scholar] [CrossRef]
- Kumar, A.; Mishra, S.; Pandey, R.; Yu, Z.G.; Kumar, M.; Khoo, K.S.; Thakur, T.K.; Show, P.L. Microplastics in terrestrial ecosystems: Un-ignorable impacts on soil characterises, nutrient storage and its cycling. TrAC Trends Anal. Chem. 2023, 158, 116869. [Google Scholar] [CrossRef]
- Aves, A.R.; Revell, L.E.; Gaw, S.; Ruffell, H.; Schuddeboom, A.; Wotherspoon, N.E.; LaRue, M.; McDonald, A.J. First evidence of microplastics in Antarctic snow. Cryosphere Discuss. 2022, 16, 2127–2145. [Google Scholar] [CrossRef]
- Leonard, S.V.; Liddle, C.R.; Atherall, C.A.; Chapman, E.; Watkins, M.; Calaminus, S.D.; Rotchell, J.M. Microplastics in human blood: Polymer types, concentrations and characterisation using μFTIR. Environ. Int. 2024, 188, 108751. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.C.; Saha, G. Effect of microplastics deposition on human lung airways: A review with computational benefits and challenges. Heliyon 2024, 10, e24355. [Google Scholar] [CrossRef]
- Ragusa, A.; Svelato, A.; Santacroce, C.; Catalano, P.; Notarstefano, V.; Carnevali, O.; Papa, F.; Rongioletti, M.C.A.; Baiocco, F.; Draghi, S. Plasticenta: First evidence of microplastics in human placenta. Environ. Int. 2021, 146, 106274. [Google Scholar] [CrossRef]
- Li, H.; Yang, Z.; Jiang, F.; Li, L.; Li, Y.; Zhang, M.; Qi, Z.; Ma, R.; Zhang, Y.; Fang, J. Detection of microplastics in domestic and fetal pigs’ lung tissue in natural environment: A preliminary study. Environ. Res. 2023, 216, 114623. [Google Scholar] [CrossRef]
- Di Fiore, C.; Ishikawa, Y.; Wright, S.L. A review on methods for extracting and quantifying microplastic in biological tissues. J. Hazard. Mater. 2024, 464, 132991. [Google Scholar] [CrossRef]
- Yang, J.; Monnot, M.; Sun, Y.; Asia, L.; Wong-Wah-Chung, P.; Doumenq, P.; Moulin, P. Microplastics in different water samples (seawater, freshwater, and wastewater): Removal efficiency of membrane treatment processes. Water Res. 2023, 232, 119673. [Google Scholar] [CrossRef]
- Kwon, H.J.; Hidayaturrahman, H.; Peera, S.G.; Lee, T.G. Elimination of microplastics at different stages in wastewater treatment plants. Water 2022, 14, 2404. [Google Scholar] [CrossRef]
- Sol, D.; Laca, A.; Laca, A.; Díaz, M. Microplastics in wastewater and drinking water treatment plants: Occurrence and removal of microfibres. Appl. Sci. 2021, 11, 10109. [Google Scholar] [CrossRef]
- Acharya, S.; Rumi, S.S.; Hu, Y.; Abidi, N. Microfibers from synthetic textiles as a major source of microplastics in the environment: A review. Text. Res. J. 2021, 91, 2136–2156. [Google Scholar] [CrossRef]
- Herzke, D.; Ghaffari, P.; Sundet, J.H.; Tranang, C.A.; Halsband, C. Microplastic fiber emissions from wastewater effluents: Abundance, transport behavior and exposure risk for biota in an arctic fjord. Front. Environ. Sci. 2021, 9, 662168. [Google Scholar] [CrossRef]
- Xu, C.; Zhou, G.; Lu, J.; Shen, C.; Dong, Z.; Yin, S.; Li, F. Spatio-vertical distribution of riverine microplastics: Impact of the textile industry. Environ. Res. 2022, 211, 112789. [Google Scholar] [CrossRef]
- Akyildiz, S.H.; Bellopede, R.; Sezgin, H.; Yalcin-Enis, I.; Yalcin, B.; Fiore, S. Detection and analysis of microfibers and microplastics in wastewater from a textile company. Microplastics 2022, 1, 572–586. [Google Scholar] [CrossRef]
- Cui, H.; Xu, C. Study on the relationship between textile microplastics shedding and fabric structure. Polymers 2022, 14, 5309. [Google Scholar] [CrossRef]
- Zhang, Y.-Q.; Lykaki, M.; Alrajoula, M.T.; Markiewicz, M.; Kraas, C.; Kolbe, S.; Klinkhammer, K.; Rabe, M.; Klauer, R.; Bendt, E. Microplastics from textile origin–emission and reduction measures. Green Chem. 2021, 23, 5247–5271. [Google Scholar] [CrossRef]
- Jabbar, A.; Tausif, M. Investigation of ring, airjet and rotor spun yarn structures on the fragmented fibers (microplastics) released from polyester textiles during laundering. Text. Res. J. 2023, 93, 5017–5028. [Google Scholar] [CrossRef]
- Militký, J.; Novotná, J.; Wiener, J.; Křemenáková, D.; Venkataraman, M. Microplastics and Fibrous Fragments Generated during the Production and Maintenance of Textiles. Fibers 2024, 12, 51. [Google Scholar] [CrossRef]
- Sait, S.T.L.; Sørensen, L.; Kubowicz, S.; Vike-Jonas, K.; Gonzalez, S.V.; Asimakopoulos, A.G.; Booth, A.M. Microplastic fibres from synthetic textiles: Environmental degradation and additive chemical content. Environ. Pollut. 2021, 268, 115745. [Google Scholar] [CrossRef]
- Akarsu, C.; Kumbur, H.; Kideys, A.E. Removal of microplastics from wastewater through electrocoagulation-electroflotation and membrane filtration processes. Water Sci. Technol. 2021, 84, 1648–1662. [Google Scholar] [CrossRef]
- Sembiring, E.; Fajar, M.; Handajani, M. Performance of rapid sand filter–single media to remove microplastics. Water Supply 2021, 21, 2273–2284. [Google Scholar] [CrossRef]
- De Somer, T.; Van Melkebeke, M.; Goethals, B.; Gusev, S.; Van der Meeren, P.; Van Geem, K.M.; De Meester, S. Modelling and application of dissolved air flotation for efficient separation of microplastics from sludges and sediments. J. Environ. Chem. Eng. 2024, 12, 112864. [Google Scholar] [CrossRef]
- Li, C.; Busquets, R.; Moruzzi, R.B.; Campos, L.C. Preliminary study on low-density polystyrene microplastics bead removal from drinking water by coagulation-flocculation and sedimentation. J. Water Process Eng. 2021, 44, 102346. [Google Scholar] [CrossRef]
- Shen, M.; Zhang, Y.; Almatrafi, E.; Hu, T.; Zhou, C.; Song, B.; Zeng, Z.; Zeng, G. Efficient removal of microplastics from wastewater by an electrocoagulation process. Chem. Eng. J. 2022, 428, 131161. [Google Scholar] [CrossRef]
- Huang, Y.; Hu, T.; Lin, B.; Ke, Y.; Li, J.; Ma, J. Microplastics-biofilm interactions in biofilm-based wastewater treatment processes: A review. Environ. Pollut. 2024, 361, 124836. [Google Scholar] [CrossRef]
- Li, S. Selecting the front-runners: Comparative evaluation of emerging technologies for microplastic removal from drinking water. Processes 2025, 13, 3943. [Google Scholar] [CrossRef]
- Wang, Z.; Sedighi, M.; Lea-Langton, A. Filtration of microplastic spheres by biochar: Removal efficiency and immobilisation mechanisms. Water Res. 2020, 184, 116165. [Google Scholar] [CrossRef] [PubMed]
- Selim, M.M.; Tounsi, A.; Gomaa, H.; Shenashen, M. Biochar-based adsorption technologies for microplastic remediation in aquatic ecosystems. AIP Adv. 2025, 15, 030701. [Google Scholar] [CrossRef]
- Lin, J.; Cheng, Q.; Kumar, A.; Zhang, W.; Yu, Z.; Hui, D.; Zhang, C.; Shan, S. Effect of degradable microplastics, biochar and their coexistence on soil organic matter decomposition: A critical review. TrAC Trends Anal. Chem. 2025, 183, 118082. [Google Scholar] [CrossRef]
- Mohsenzadeh, A.; Persson, M.; Pettersson, A.; Frandsen, F.J. Biochar for the Removal of Microplastics from Water: A Comprehensive Scoping Review. Microplastics 2025, 4, 99. [Google Scholar] [CrossRef]
- Li, S.; Harris, S.; Anandhi, A.; Chen, G. Predicting biochar properties and functions based on feedstock and pyrolysis temperature: A review and data syntheses. J. Clean. Prod. 2019, 215, 890–902. [Google Scholar] [CrossRef]
- Mota, L.S.O.; de Oliveira, P.C.O.; Peixoto, B.S.; Bezerra, E.S.; de Moraes, M.C. Biochar applications in microplastic and nanoplastic removal: Mechanisms and integrated approaches. Environ. Sci. Water Res. Technol. 2025, 11, 222–241. [Google Scholar] [CrossRef]
- Ji, G.; Xing, Y.; You, T. Biochar as adsorbents for environmental microplastics and nanoplastics removal. J. Environ. Chem. Eng. 2024, 12, 113377. [Google Scholar] [CrossRef]
- Schirrmeister, S.; Kurzweg, L.; Gjashta, X.; Socher, M.; Fery, A.; Harre, K. Regression analysis for the determination of microplastics in sediments using differential scanning calorimetry. Environ. Sci. Pollut. Res. 2024, 31, 31001–31014. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.P.d.; Avellan, A.; Mouneyrac, C.; Duarte, A.; Rocha-Santos, T. Plastic additives and microplastics as emerging contaminants: Mechanisms and analytical assessment. TrAC Trends Anal. Chem. 2023, 158, 116898. [Google Scholar] [CrossRef]
- Khanum, R.; Shoukat Ali, R.A.; Rangaswamy, H.R.; Santhosh Kumar, S.R.; Prashantha, A.G.; Jagadisha, A.S. Recent review on Synthesis, spectral Studies, versatile applications of azo dyes and its metal complexes. Results Chem. 2023, 5, 100890. [Google Scholar] [CrossRef]
- Su, Z.-H.; Wang, C.; Zhou, X.; He, M.-J. Organophosphate esters and phthalate esters in marine fishes from a coastal area of China: Occurrence, tissue distribution, trophic transfer, and human exposure. Mar. Environ. Res. 2025, 208, 107135. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.W.; Gunderson, K.G.; Green, L.A.; Rediske, R.R.; Steinman, A.D. Perfluoroalkylated Substances (PFAS) Associated with Microplastics in a Lake Environment. Toxics 2021, 9, 106. [Google Scholar] [CrossRef]
- Li, S.; Liu, H.; Gao, R.; Abdurahman, A.; Dai, J.; Zeng, F. Aggregation kinetics of microplastics in aquatic environment: Complex roles of electrolytes, pH, and natural organic matter. Environ. Pollut. 2018, 237, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Tong, M.; Li, T.; Li, M.; He, L.; Ma, Z. Cotransport and deposition of biochar with different sized-plastic particles in saturated porous media. Sci. Total Environ. 2020, 713, 136387. [Google Scholar] [CrossRef]
- Wang, B.; Wu, L.; Pang, K.; Zhang, G.; Xu, D.; Sun, H.; Yin, X. Transport of reduced PBAT microplastics in saturated porous media: Synergistic effects of enhanced surface energy and roughness. Water Res. 2024, 267, 122514. [Google Scholar] [CrossRef]
- Tatsii, D.; Bucci, S.; Bhowmick, T.; Guettler, J.; Bakels, L.; Bagheri, G.; Stohl, A. Shape Matters: Long-Range Transport of Microplastic Fibers in the Atmosphere. Environ. Sci. Technol. 2024, 58, 671–682. [Google Scholar] [CrossRef]
- Malchiodi, B.; Cedillo-González, E.I.; Siligardi, C.; Pozzi, P. A Practical Valorization Approach for Mitigating Textile Fibrous Microplastics in the Environment: Collection of Textile-Processing Waste Microfibers and Direct Reuse in Green Thermal-Insulating and Mechanical-Performing Composite Construction Materials. Microplastics 2022, 1, 393–405. [Google Scholar] [CrossRef]
- Schutte, M.M.; Kteeba, S.M.; Guo, L. Photochemical reactivity of water-soluble dissolved organic matter from microplastics and microfibers. Sci. Total Environ. 2024, 911, 168616. [Google Scholar] [CrossRef]
- Frost, H.; Bond, T.; Sizmur, T.; Felipe-Sotelo, M. A review of microplastic fibres: Generation, transport, and vectors for metal(loid)s in terrestrial environments. Environ. Sci. Process. Impacts 2022, 24, 504–524. [Google Scholar] [CrossRef]
- Rostampour, S.; Jhang, S.S.; Hsu, J.-K.; Cook, R.; Li, Y.; Fan, C.; Sung, L.-P. Evaluating the Environmental Factors on Microplastic Generation: An Accelerated Weathering Study. Microplastics 2025, 4, 13. [Google Scholar] [CrossRef]
- Stark, N.M.; Matuana, L.M. Surface chemistry and mechanical property changes of wood-flour/high-density-polyethylene composites after accelerated weathering. J. Appl. Polym. Sci. 2004, 94, 2263–2273. [Google Scholar] [CrossRef]
- Bond, T.; Morton, J.; Al-Rekabi, Z.; Cant, D.; Davidson, S.; Pei, Y. Surface properties and rising velocities of pristine and weathered plastic pellets. Environ. Sci. Process. Impacts 2022, 24, 794–804. [Google Scholar] [CrossRef]
- Li, M.; Ou, J.; Chen, Z. Settling behavior of microplastic hetero-aggregates in aquatic environments with varying salinity. Int. J. Sediment Res. 2025, 40, 561–572. [Google Scholar] [CrossRef]
- Li, S.; Chen, G. Thermogravimetric, thermochemical, and infrared spectral characterization of feedstocks and biochar derived at different pyrolysis temperatures. Waste Manag. 2018, 78, 198–207. [Google Scholar] [CrossRef]
- Fryda, L.; Visser, R. Biochar for Soil Improvement: Evaluation of Biochar from Gasification and Slow Pyrolysis. Agriculture 2015, 5, 1076–1115. [Google Scholar] [CrossRef]
- Chaudhary, R.; Sangwan, G.; Kumar, S.; Sharma, V. Turning Trash into Tools: Agricultural Waste-Derived Biochar and Composites for Microplastic Removal from Wastewater. RSC Sustain. 2026, 4, 650–676. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Ghosh, P.K.; Mandal, B.; Mallik, A.; Mukherjee, A. Microplastic in Mangrove Water: Occurrence and Possible Remediation Through Biological and Technological Advancement. In Water Crises and Sustainable Management in the Global South; Izah, S.C., Ogwu, M.C., Loukas, A., Hamidifar, H., Eds.; Springer Nature: Singapore, 2024; pp. 279–312. [Google Scholar]
- Seyfi, S.; Karimpour, S.; Balachandar, R.; Shnapp, R.; Liberzon, A. Response of microplastic particles to turbulent flow: An experimental study. In River Flow 2024; CRC Press: Boca Raton, FL, USA, 2025; pp. 717–724. [Google Scholar]
- Das, K.P.; Chauhan, P.; Staudinger, U.; Satapathy, B.K. Exploring sustainable adsorbents to mitigate micro-/nano-plastic contamination: Perspectives on electrospun fibrous constructs, biochar, and aerogels. Environ. Sci. Adv. 2024, 3, 1217–1243. [Google Scholar] [CrossRef]
- Duan, X.; Chen, X.; Shi, L.; Cao, Y.; Liang, Y.; Wang, T.; Huang, C.; Cao, Y. Functionality-dependent removal efficiency and mechanisms of polystyrene microplastics by a robust magnetic biochar. J. Environ. Chem. Eng. 2025, 13, 115509. [Google Scholar] [CrossRef]
- Changlor, N.; Inchana, C.; Sabar, M.A.; Suyamud, B.; Lohwacharin, J. Effects of relative microplastic–biochar sizes and biofilm formation on fragmental microplastic retention in biochar filters. Environ. Res. 2025, 268, 120834. [Google Scholar] [CrossRef]
- Subair, A.; Krishnamoorthy Lakshmi, P.; Chellappan, S.; Chinghakham, C. Removal of polystyrene microplastics using biochar-based continuous flow fixed-bed column. Environ. Sci. Pollut. Res. 2024, 31, 13753–13765. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, S.; Paciência, D.; Rodrigues, J.M.M.; Lindman, B.; Alves, L.; Medronho, B.; Rasteiro, M.d.G. Insights on Microplastic Contamination from Municipal and Textile Industry Effluents and Their Removal Using a Cellulose-Based Approach. Polymers 2024, 16, 2803. [Google Scholar] [CrossRef]
- Li, S.; Tasnady, D.; Skelley, S.; Calderon, B.; Jiang, S. Enhancing Organic Contaminant Removal from Wool Scouring Wastewater Using Chemically Modified Biochars. C 2024, 10, 6. [Google Scholar] [CrossRef]
- Ohshima, H. The Derjaguin–Landau–Verwey–Overbeek (DLVO) Theory of Colloid Stability. In Electrical Phenomena at Interfaces and Biointerfaces; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Long, X.; Zhang, R.; Rong, R.; Wu, P.; Chen, S.; Ao, J.; An, L.; Fu, Y.; Xie, H. Adsorption characteristics of heavy metals Pb2+ and Zn2+ by magnetic biochar obtained from modified AMD sludge. Toxics 2023, 11, 590. [Google Scholar] [CrossRef]
- Long, L.; Xue, Y.; Hu, X.; Zhu, Y. Study on the influence of surface potential on the nitrate adsorption capacity of metal modified biochar. Environ. Sci. Pollut. Res. 2019, 26, 3065–3074. [Google Scholar] [CrossRef]
- Li, X.; Cheng, Q.; Kang, Y.; Gu, Z.; Bao, H.; Wang, N.; Liu, Y.; Hu, C.; Qu, J. Synergistic adsorption and removal of nitrate in an electrically polarized column packed with AlCl3-modified activated carbon. Sep. Purif. Technol. 2025, 364, 132570. [Google Scholar] [CrossRef]
- Chai, Y.; Li, Y.; Han, W.; Zhang, Y.; Du, Y.; Wang, B.; Chen, M.; Li, N.; Zha, X.; Hu, Z. Magnetic biochar conjugated quaternary ammonium salt-constructed hemoperfusion adsorbent for viral removal with rapid and high-performance. Sep. Purif. Technol. 2025, 354, 129366. [Google Scholar] [CrossRef]
- Wang, B.; He, G.; Xiao, J.; Shi, H.; Li, Y. Cationic wood-derived anisotropic biochar: Monolithic design enables high-efficiency capture of microplastic. Chem. Eng. J. 2025, 525, 169959. [Google Scholar] [CrossRef]
- Liu, F.; Pignatello, J.J.; Sun, R.; Guan, X.; Xiao, F. A comprehensive review of novel adsorbents for per-and polyfluoroalkyl substances in water. ACS ES T Water 2024, 4, 1191–1205. [Google Scholar] [CrossRef]
- Zhao, J.; Jiang, Y.; Chen, X.; Wang, C.; Nan, H. Unlocking the potential of element-doped biochar: From tailored synthesis to multifunctional applications in environment and energy. Biochar 2025, 7, 77. [Google Scholar] [CrossRef]
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Johir, M.A.H.; Sun, L.; Asadullah, M.; Belhaj, D. Sorption of hydrophobic organic contaminants on functionalized biochar: Protagonist role of π-π electron-donor-acceptor interactions and hydrogen bonds. J. Hazard. Mater. 2018, 360, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Melki, S.; Biguenet, F.; Dupuis, D. Hydrophobic properties of textile materials: Robustness of hydrophobicity. J. Text. Inst. 2019, 110, 1221–1228. [Google Scholar] [CrossRef]
- Pusceddu, E.; Montanaro, A.; Fioravanti, G.; Santilli, S.; Foscolo, P.; Criscuoli, I.; Raschi, A.; Miglietta, F. Comparison between ancient and fresh biochar samples, a study on the recalcitrance of carbonaceous structures during soil incubation. Int. J. New Technol. Res. 2017, 3, 39–46. [Google Scholar]
- Wang, L.; Liang, L.; Li, N.; Chen, G.; Guo, H.; Hou, L.a. A Mini-Review of Sludge-Derived Biochar (SDB) for Wastewater Treatment: Recent Advances in 2020–2025. Appl. Sci. 2025, 15, 6173. [Google Scholar] [CrossRef]
- Li, S.; Galoustian, T.; Trejo, H. Biochar pyrolyzed with concentrated solar radiation for enhanced nitrate adsorption. J. Anal. Appl. Pyrolysis 2023, 174, 106131. [Google Scholar] [CrossRef]
- Keiluweit, M.; Kleber, M. Molecular-Level Interactions in Soils and Sediments: The Role of Aromatic π-Systems. Environ. Sci. Technol. 2009, 43, 3421–3429. [Google Scholar] [CrossRef]
- Zhang, Q.; Cai, H.; Yi, W.; Lei, H.; Liu, H.; Wang, W.; Ruan, R. Biocomposites from organic solid wastes derived biochars: A review. Materials 2020, 13, 3923. [Google Scholar] [CrossRef]
- Bashir, M.; Ahanger, M.A.; Gani, K.M. Investigations on adsorptive removal of PVC microplastics from aqueous solutions using Pinus roxburghii–derived biochar. Environ. Sci. Pollut. Res. 2024, 31, 59416–59429. [Google Scholar] [CrossRef]
- Siipola, V.; Pflugmacher, S.; Romar, H.; Wendling, L.; Koukkari, P. Low-Cost Biochar Adsorbents for Water Purification Including Microplastics Removal. Appl. Sci. 2020, 10, 788. [Google Scholar] [CrossRef]
- Liu, J.; Pan, Z.; Sun, K.; Chen, Y.; Yang, Y.; Gao, B.; Xing, B. The preferential preservation of both different minerals and polyethylene microplastics on aromatic or aliphatic carbon fractions within low or high pyrolysis temperature biochar under mineralization. Renew. Sustain. Energy Rev. 2022, 170, 112963. [Google Scholar] [CrossRef]
- Kaur, K.; Saleem, M.; Kaur, H. Biochar and biopolymer based nanomaterials for microplastic remediation: A sustainable approach for environment. Top. Catal. 2025, 69, 825–852. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhou, S.; Fei, J.; Qin, Z.; Yin, X.; Sun, H.; Sun, Y. Transport of different microplastics in porous media: Effect of the adhesion of surfactants on microplastics. Water Res. 2022, 215, 118262. [Google Scholar] [CrossRef]
- Tubić, A.; Vujić, M.; Gvoić, V.; Agbaba, J.; Vasiljević, S.; Cveticanin, L.; Vukelić, Đ.; Prica, M. Sorption potential of microplastics for azo- and phthalocyanine printing dyes. Dye. Pigment. 2023, 209, 110884. [Google Scholar] [CrossRef]
- Li, S.; Barreto, V.; Li, R.; Chen, G.; Hsieh, Y.P. Nitrogen retention of biochar derived from different feedstocks at variable pyrolysis temperatures. J. Anal. Appl. Pyrolysis 2018, 133, 136–146. [Google Scholar] [CrossRef]
- Li, J.; Zhou, Y.; Liu, W.; Zhang, M. Molecular dynamics simulation of microplastics adsorption mechanisms on balsa biochar. Sep. Purif. Technol. 2026, 383, 136214. [Google Scholar] [CrossRef]
- Xiao, S.; Cui, Y.; Brahney, J.; Mahowald, N.M.; Li, Q. Long-distance atmospheric transport of microplastic fibres influenced by their shapes. Nat. Geosci. 2023, 16, 863–870. [Google Scholar] [CrossRef]
- Yaseen, Z.M.; Zigale, T.T.; Tiyasha; Salih, S.Q.; Awasthi, S.; Tung, T.M.; Al-Ansari, N.; Bhagat, S.K. Laundry wastewater treatment using a combination of sand filter, bio-char and teff straw media. Sci. Rep. 2019, 9, 18709. [Google Scholar] [CrossRef] [PubMed]
- Downie, A.; Crosky, A.; Munroe, P. Physical properties of biochar. In Biochar for Environmental Management; Routledge: Oxfordshire, UK, 2012; pp. 45–64. [Google Scholar]
- Bianco, V.; Pirone, D.; Memmolo, P.; Merola, F.; Ferraro, P. Identification of microplastics based on the fractal properties of their holographic fingerprint. ACS Photonics 2021, 8, 2148–2157. [Google Scholar] [CrossRef]
- Jiang, H.; Bu, J.; Bian, K.; Su, J.; Wang, Z.; Sun, H.; Wang, H.; Zhang, Y.; Wang, C. Surface change of microplastics in aquatic environment and the removal by froth flotation assisted with cationic and anionic surfactants. Water Res. 2023, 233, 119794. [Google Scholar] [CrossRef] [PubMed]
- Dai, G.; Zhang, L.; Liao, Y.; Shi, Y.; Xie, J.; Lei, F.; Fan, L. Multi-scale model for describing the effect of pore structure on carbon-based electric double layer. J. Phys. Chem. C 2020, 124, 3952–3961. [Google Scholar] [CrossRef]
- Akhil, D.; Lakshmi, D.; Kartik, A.; Vo, D.-V.N.; Arun, J.; Gopinath, K.P. Production, characterization, activation and environmental applications of engineered biochar: A review. Environ. Chem. Lett. 2021, 19, 2261–2297. [Google Scholar] [CrossRef]
- Sadegh, F.; Sadegh, N.; Wongniramaikul, W.; Apiratikul, R.; Choodum, A. Adsorption of volatile organic compounds on biochar: A review. Process Saf. Environ. Prot. 2024, 182, 559–578. [Google Scholar] [CrossRef]
- Chen, C.; Sun, K.; Huang, C.; Yang, M.; Fan, M.; Wang, A.; Zhang, G.; Li, B.; Jiang, J.; Xu, W. Investigation on the mechanism of structural reconstruction of biochars derived from lignin and cellulose during graphitization under high temperature. Biochar 2023, 5, 51. [Google Scholar] [CrossRef]
- Jang, M.-H.; Kim, M.-S.; Han, M.; Kwak, D.-H. Experimental application of a zero-point charge based on pH as a simple indicator of microplastic particle aggregation. Chemosphere 2022, 299, 134388. [Google Scholar] [CrossRef]
- Jetsrisuparb, K.; Jeejaila, T.; Saengthip, C.; Kasemsiri, P.; Ngernyen, Y.; Chindaprasirt, P.; Knijnenburg, J.T. Tailoring the phosphorus release from biochar-based fertilizers: Role of magnesium or calcium addition during co-pyrolysis. RSC Adv. 2022, 12, 30539–30548. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, Y.-G.; Kim, H.; Chon, K.; Phae, C. One-step synthesis of magnetic biochar via co-pyrolysis of walnut shells and Fe-rich mine tails for adsorption capacity improvement of polystyrene sulfonate microplastics: Role of microplastic size. Environ. Technol. Innov. 2024, 34, 103624. [Google Scholar] [CrossRef]
- Qin, L.; Qian, S.; Zhou, X.; Al-Dhabi, N.A.; Lai, C.; Ma, D.; Huo, X.; Wu, L.; Tang, W. Co-pyrolyzed biochar derived from microplastics and microalgae as peroxymonosulfate activator: Influence of microplastic types and analysis of systemic causes. J. Environ. Chem. Eng. 2024, 12, 114647. [Google Scholar] [CrossRef]
- Sun, J.; He, F.; Pan, Y.; Zhang, Z. Effects of pyrolysis temperature and residence time on physicochemical properties of different biochar types. Acta Agric. Scand. Sect. B—Soil Plant Sci. 2017, 67, 12–22. [Google Scholar] [CrossRef]
- Tong, Y.; Mayer, B.K.; McNamara, P.J. Adsorption of organic micropollutants to biosolids-derived biochar: Estimation of thermodynamic parameters. Environ. Sci. Water Res. Technol. 2019, 5, 1132–1144. [Google Scholar] [CrossRef]
- Xiao, G.; Li, R.; Zhu, D.; Vakili, M.; Zhu, X.; Liu, X.; Zhou, S.; Wang, W. Adsorption of Pb (II) on covalent organic frameworks: Overlooked sorption sites formed in the post-synthetic modification process. Colloids Surf. A Physicochem. Eng. Asp. 2024, 681, 132791. [Google Scholar] [CrossRef]
- Gęca, M.; Khalil, A.M.; Tang, M.; Bhakta, A.K.; Snoussi, Y.; Nowicki, P.; Wiśniewska, M.; Chehimi, M.M. Surface treatment of biochar—Methods, surface analysis and potential applications: A comprehensive review. Surfaces 2023, 6, 179–213. [Google Scholar] [CrossRef]
- Tadesse, M.G.; Abate, M.T.; Lübben, J.F. Polymeric Materials in Textile Finishing. In Sustainable Finishing Techniques in Textiles; Springer: Berlin/Heidelberg, Germany, 2025; pp. 129–154. [Google Scholar]
- Bélanger, N.; Prasher, S.; Dumont, M.-J. Tailoring biochar production for use as a reinforcing bio-based filler in rubber composites: A review. Polym.-Plast. Technol. Mater. 2023, 62, 54–75. [Google Scholar] [CrossRef]
- Li, S.-K.; Wen, Y.-J.; Wang, Y.-F.; Liu, M.; Peng, Z.-j.; Su, L.-z.; Zhou, Z.; Zhou, N. Biochar rich in amino ligand for copper selective recollection in wastewater. Sep. Purif. Technol. 2025, 356, 129981. [Google Scholar] [CrossRef]
- Leksungnoen, P.; Wisawapipat, W.; Ketrot, D.; Aramrak, S.; Nookabkaew, S.; Rangkadilok, N.; Satayavivad, J. Biochar and ash derived from silicon-rich rice husk decrease inorganic arsenic species in rice grain. Sci. Total Environ. 2019, 684, 360–370. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Li, X.; Wu, M.; Lin, K.; Xu, L.; Zeng, T.; Shi, H.; Zhang, M. Synergistic role of inherent calcium and iron minerals in paper mill sludge biochar for phosphate adsorption. Sci. Total Environ. 2022, 834, 155193. [Google Scholar] [CrossRef] [PubMed]
- Brewer, C.E.; Chuang, V.J.; Masiello, C.A.; Gonnermann, H.; Gao, X.; Dugan, B.; Driver, L.E.; Panzacchi, P.; Zygourakis, K.; Davies, C.A. New approaches to measuring biochar density and porosity. Biomass Bioenergy 2014, 66, 176–185. [Google Scholar] [CrossRef]
- Xu, Q.; Zhou, Q.; Pan, M.; Dai, L. Interaction between chlortetracycline and calcium-rich biochar: Enhanced removal by adsorption coupled with flocculation. Chem. Eng. J. 2020, 382, 122705. [Google Scholar] [CrossRef]
- Yin, P.; Luo, Y.; Lan, D.; Wang, J.; Feng, X.; Jia, Z.; Wu, G.; Zhang, Y. Structural engineering of porous biochar loaded with ferromagnetic/anti-ferromagnetic NiCo2O4/CoO for excellent electromagnetic dissipation with flexible and self-cleaning properties. J. Mater. Sci. Technol. 2024, 180, 12–22. [Google Scholar] [CrossRef]
- Zhang, L.; Dong, Y.; Liu, J.; Liu, C.; Liu, W.; Lin, H. The effect of co-pyrolysis temperature for iron-biochar composites on their adsorption behavior of antimonite and antimonate in aqueous solution. Bioresour. Technol. 2022, 347, 126362. [Google Scholar] [CrossRef]
- Li, S.; Zhao, H.; Bai, P.; Meng, Y.; Tian, Y. A multifunctional magnetic biochar composites for advanced magnetorheological fluid applications. Colloids Surf. A Physicochem. Eng. Asp. 2025, 711, 136346. [Google Scholar] [CrossRef]
- Zhang, L.; Yu, Q.; Li, W.; Zhou, Y.; Peng, Q.; Wang, Y. Plane trees’ bark–derived porous biochar for efficient methylene blue biosorption. Biomass Convers. Biorefin. 2025, 15, 23411–23422. [Google Scholar] [CrossRef]
- Patra, B.R.; Mukherjee, A.; Nanda, S.; Dalai, A.K. Biochar production, activation and adsorptive applications: A review. Environ. Chem. Lett. 2021, 19, 2237–2259. [Google Scholar] [CrossRef]
- Alharbi, H.A.; Hameed, B.H.; Alotaibi, K.D.; Al-Oud, S.S.; Al-Modaihsh, A.S. Recent methods in the production of activated carbon from date palm residues for the adsorption of textile dyes: A review. Front. Environ. Sci. 2022, 10, 996953. [Google Scholar] [CrossRef]
- Wu, W.; Liu, J.; Zhang, G.; Wang, Y.; Wu, C.; Li, G.; Zhao, Y. Adsorption of CO2 by sludge/bamboo shoot shell hybrid biochar prepared by a single-step K2CO3 activation. Fuel 2025, 381, 133555. [Google Scholar] [CrossRef]
- Tan, M.-M.; Feng, L.-J.; Bian, S.-Z.; Duan, J.-L.; Li, X.-H.; Sun, X.-D.; Sun, Y.-C.; Wang, S.-G.; Yuan, X.-Z. Interaction of Dissolved Organic Matters and Microplastics Regulates the Transport of Microplastics in Saturated Porous Media. ACS ES T Eng. 2024, 4, 1230–1239. [Google Scholar] [CrossRef]
- Wang, H.; Tian, Z.; Jiang, L.; Luo, W.; Wei, Z.; Li, S.; Cui, J.; Wei, W. Highly efficient adsorption of Cr (VI) from aqueous solution by Fe3+ impregnated biochar. J. Dispers. Sci. Technol. 2017, 38, 815–825. [Google Scholar] [CrossRef]
- Savić, A.; Vuksanović, M.M.; Savić, M.; Knežević, N.; Šaponjić, A.; Ilić, S.; Egelja, A. Modified Silica Particles Coated with Cu-Al Layered Double Hydroxide for Phosphate and Arsenate Removal in Water Treatment. Molecules 2025, 30, 2138. [Google Scholar] [CrossRef] [PubMed]
- Alshahrani, H.; Prakash, V.A. Effect of silane-grafted orange peel biochar and areca fibre on mechanical, thermal conductivity and dielectric properties of epoxy resin composites. Biomass Convers. Biorefinery 2024, 14, 8081–8089. [Google Scholar] [CrossRef]
- Zhang, M.; Zhu, H.; Xi, B.; Tian, Y.; Sun, X.; Zhang, H.; Wu, B. Surface hydrophobic modification of biochar by silane coupling agent KH-570. Processes 2022, 10, 301. [Google Scholar] [CrossRef]
- Navarathna, C.M.; Bombuwala Dewage, N.; Keeton, C.; Pennisson, J.; Henderson, R.; Lashley, B.; Zhang, X.; Hassan, E.B.; Perez, F.; Mohan, D. Biochar adsorbents with enhanced hydrophobicity for oil spill removal. ACS Appl. Mater. Interfaces 2020, 12, 9248–9260. [Google Scholar] [CrossRef]
- Tujjohra, F.; Hoque, E.; Kader, M.A.; Rahman, M.M. Sustainable valorization of textile industry cotton waste through pyrolysis for biochar production. Clean. Chem. Eng. 2025, 11, 100161. [Google Scholar] [CrossRef]
- Xia, X.; Wang, X.; Riaz, M.; Babar, S.; El-Desouki, Z.; Wang, J.; Yan, B.; Jiang, C. Nitric acid-modified biochar enhances saline-alkali soil remediation and cotton growth via regulating soil-plant homeostasis. Plant Physiol. Biochem. 2025, 229, 110722. [Google Scholar] [CrossRef]
- Arunmugan, M.; Rupasinghe, C.; Manoharan, T.; Kannan, N. The effect of hydrogen peroxide treatment on functional properties of paddy husk biochar and its application for wastewater treatment. Trop. Agric. Res. Ext. 2023, 26, 318–330. [Google Scholar] [CrossRef]
- Verheijen, F.G.; Zhuravel, A.; Silva, F.C.; Amaro, A.; Ben-Hur, M.; Keizer, J.J. The influence of biochar particle size and concentration on bulk density and maximum water holding capacity of sandy vs sandy loam soil in a column experiment. Geoderma 2019, 347, 194–202. [Google Scholar] [CrossRef]
- Zhou, X.; Lai, C.; Almatrafi, E.; Liu, S.; Yan, H.; Qian, S.; Li, H.; Qin, L.; Yi, H.; Fu, Y. Unveiling the roles of dissolved organic matters derived from different biochar in biochar/persulfate system: Mechanism and toxicity. Sci. Total Environ. 2023, 864, 161062. [Google Scholar] [CrossRef] [PubMed]
- Goswami, L.; Kushwaha, A.; Kafle, S.R.; Kim, B.-S. Surface modification of biochar for dye removal from wastewater. Catalysts 2022, 12, 817. [Google Scholar] [CrossRef]
- Yao, S.; Li, X.; Wang, T.; Jiang, X.; Song, Y.; Arp, H.P.H. Soil metabolome impacts the formation of the eco-corona and adsorption processes on microplastic surfaces. Environ. Sci. Technol. 2023, 57, 8139–8148. [Google Scholar] [CrossRef]
- Yang, H.; Chen, Z.; Kong, L.; Xing, H.; Yang, Q.; Wu, J. A Review of Eco-Corona Formation on Micro/Nanoplastics and Its Effects on Stability, Bioavailability, and Toxicity. Water 2025, 17, 1124. [Google Scholar] [CrossRef]
- Dharmarathna, S.P.; Priyantha, N. Investigation of boundary layer effect of intra-particle diffusion on methylene blue adsorption on activated carbon. Energy Nexus 2024, 14, 100294. [Google Scholar] [CrossRef]
- Sarsour, J.; Ewert, B.; Janisch, B.; Stegmaier, T.; Gresser, G.T. Efficient Filtration Systems for Microplastic Elimination in Wastewater. Microplastics 2025, 4, 36. [Google Scholar] [CrossRef]
- Othman, M.; Roddick, F.; Snow, R. Removal of dissolved organic compounds in fixed-bed columns: Evaluation of low-rank coal adsorbents. Water Res. 2001, 35, 2943–2949. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.I.; Farghali, M.; Rashwan, A.K. Life cycle assessment of biochar as a green sorbent for soil remediation. Curr. Opin. Green Sustain. Chem. 2024, 46, 100882. [Google Scholar] [CrossRef]
- Mpeta, M.; Wenga, T.; Marondedze, K.A.; Sadondo, P. Regeneration, recycling, and disposal of spent biochars. In Biochar for Environmental Remediation; Elsevier: Amsterdam, The Netherlands, 2025; pp. 379–393. [Google Scholar]
- Tan, Z.; Yuan, S.; Hong, M.; Zhang, L.; Huang, Q. Mechanism of negative surface charge formation on biochar and its effect on the fixation of soil Cd. J. Hazard. Mater. 2020, 384, 121370. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, T.A.; Gomes Filho, R.R.; Wisniewski, A., Jr.; Mašek, O. Biochar physical degradation: Long-term effects as soil amendments. Biomass Bioenergy 2025, 203, 108284. [Google Scholar] [CrossRef]
- Xu, Z.; He, M.; Xu, X.; Cao, X.; Tsang, D.C. Impacts of different activation processes on the carbon stability of biochar for oxidation resistance. Bioresour. Technol. 2021, 338, 125555. [Google Scholar] [CrossRef]
- Xiong, P.; Yan, X.; Zhu, Q.; Qu, G.; Shi, J.; Liao, C.; Jiang, G. A review of environmental occurrence, fate, and toxicity of novel brominated flame retardants. Environ. Sci. Technol. 2019, 53, 13551–13569. [Google Scholar] [CrossRef]
- Li, S.; Tasnady, D. Biochar for Soil Carbon Sequestration: Current Knowledge, Mechanisms, and Future Perspectives. C 2023, 9, 67. [Google Scholar] [CrossRef]
- Zhu, X.; Labianca, C.; He, M.; Luo, Z.; Wu, C.; You, S.; Tsang, D.C. Life-cycle assessment of pyrolysis processes for sustainable production of biochar from agro-residues. Bioresour. Technol. 2022, 360, 127601. [Google Scholar] [CrossRef]
- Dassanayake, C.; Mashaan, N.S. Biochar as additive and modifier in bitumen and asphalt mixtures. Eng 2025, 6, 341. [Google Scholar] [CrossRef]
- Rathinamoorthy, R.; Balasaraswathi, S.R. Impact of quantification method on microfiber assessment–A comparative analysis between mass and count based methods. J. Environ. Manag. 2023, 347, 119040. [Google Scholar] [CrossRef]
- Adeel, M.; Frois, C.F.G.; Berruti, I.; Sirtori, C.; Oller, I.; Malato, S.; Rizzo, L. Effect of microplastics on tertiary/quaternary treatment of urban wastewater: Fe-biochar/peroxymonosulfate/sunlight vs solar photo-Fenton. J. Environ. Manag. 2025, 384, 125555. [Google Scholar] [CrossRef]
- Maroušek, J.; Kolář, L.; Strunecký, O.; Kopecký, M.; Bartoš, P.; Maroušková, A.; Cudlínová, E.; Konvalina, P.; Šoch, M.; Moudrý, J., Jr. Modified biochars present an economic challenge to phosphate management in wastewater treatment plants. J. Clean. Prod. 2020, 272, 123015. [Google Scholar] [CrossRef]
- Chandra, A.; Singh, A.A.; Prasad, S.; Andersson, M.R.; Gedefaw, D. Crosslinking Approaches for Polyethylene Imine (PEI) and Its Uses in Adsorption of Heavy Metals, Dyes, and Carbon Dioxide. Appl. Sci. 2025, 15, 4767. [Google Scholar] [CrossRef]
- Hadadi, A.; Imessaoudene, A.; Bollinger, J.-C.; Assadi, A.A.; Amrane, A.; Mouni, L. Comparison of four plant-based bio-coagulants performances against alum and ferric chloride in the turbidity improvement of bentonite synthetic water. Water 2022, 14, 3324. [Google Scholar] [CrossRef]
- Anosike-Francis, E.N.; Obianyo, I.I.; Kaoje, H.A.; Oyinloye, O.B.; Ogedengbe, T.S.; Adeleke, A.A. Artificial intelligence-driven techno-economic and life cycle assessment of biochar-based catalysts for contaminant removal and waste recycling: A comparative study of developing and developed nations. In Biochar-Based Catalysts for Removal of Environmental Contaminants; Elsevier: Amsterdam, The Netherlands, 2026; pp. 281–306. [Google Scholar]
- Labianca, C.; DeGisi, S.; Notarnicola, M.; Zhu, X.; Tsang, D.W. Environmental and economic evaluation of biochar application in wastewater and sludge treatment. In Biochar Applications for Wastewater Treatment; John Wiley & Sons: Hoboken, NJ, USA, 2023; pp. 289–308. [Google Scholar]
- Nayak, K.C.; Pathania, A.; Pathania, A.R. Red mud: Characteristics, utilization, and environmental remediation strategies in the aluminium industry. Mater. Today Proc. 2024, in press. [Google Scholar]
- Truong, H.B.; Ike, I.A.; Ok, Y.S.; Hur, J. Polyethyleneimine modification of activated fly ash and biochar for enhanced removal of natural organic matter from water via adsorption. Chemosphere 2020, 243, 125454. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Lai, D.Y.F.; Abid, A.A.; Neogi, S.; Xu, X.; Wang, C. Effects of steel slag and biochar incorporation on active soil organic carbon pools in a subtropical paddy field. Agronomy 2018, 8, 135. [Google Scholar] [CrossRef]





| Mechanism | Primary Feedstock | Pyrolysis Temperature | Residence Time | Activation/Surface Modification | Rational Contribution to FMP Removal | References |
|---|---|---|---|---|---|---|
| Physical interception & mechanical entanglement | Lignocellulosic (e.g., bamboo, wood, maize stalks) | High (600–900 °C) | Moderate (1–2 h) | K2CO3, KOH, ZnCl2 | Maintains internal pathways for the fibers to settle into and uses a highly textured surface to hook and hold them in place. | [117,118,119,120] |
| Electrostatic attraction (charge neutralization) | OFG-rich (e.g., agricultural waste, low-temperature residues) | Low (<400 °C) for anchoring sites | Short to moderate (0.5–1 h) | Cationization: Fe3+, Al3+, Mg2+ impregnation or Amination: PEI/Chitosan grafting | Overcomes the electrostatic repulsion barrier by reversing surface charge ( > +30 mV); reduces Debye length. | [121,122,123] |
| π–π electron donor-acceptor (EDA) | High-carbon (e.g., hardwoods, nut shells) | Ultra-high (>700 °C) | Extended (2–4 h) | Catalytic graphitization: Pre-impregnation with Fe, Ni, Co | Promotes -hybridization and graphitic crystallinity for high-energy overlap with aromatic polymer rings (e.g., PET). | [74,83,102] |
| Hydrophobic partitioning | Low-ash (e.g., (woody biomass, crop residues) | High (>700 °C) | Moderate (1–2 h) | Surface tailoring: Organosilane grafting (e.g., octadecyltri-chlorosilane) | Decreases H/C and O/C ratios to match surface energy of non-polar polymers; maximizes entropic gain (∆S) via water displacement. | [124,125,126] |
| Hydrogen bonding | High-cellulose (e.g., cotton waste, softwoods) | Low (<450 °C) | Short (0.5–1 h) | Oxidative modification: HNO3, H2O2, or aminosilane grafting | Preserves polar moieties (carboxyl, hydroxyl) to engage with amide linkages in Nylon or functional finishes. | [127,128,129] |
| Ballasting & hetero-aggregation | High-ash (e.g., sewage sludge, manure, rice husks) | Moderate to High (500–700 °C) | Moderate (1–1.5 h) | Magnetic Functionalization: In-situ Fe3O4 precipitation | Increases skeletal density (1.5–2.1 g/cm3) to circumvent buoyancy traps and accelerate sedimentation. | [112,130] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cruz, K.; Li, S. Engineered Biochar for the Sequestration of Textile Fibrous Microplastics: From Mechanistic Insights to Rational Functional Design. C 2026, 12, 31. https://doi.org/10.3390/c12020031
Cruz K, Li S. Engineered Biochar for the Sequestration of Textile Fibrous Microplastics: From Mechanistic Insights to Rational Functional Design. C. 2026; 12(2):31. https://doi.org/10.3390/c12020031
Chicago/Turabian StyleCruz, Kiara, and Simeng Li. 2026. "Engineered Biochar for the Sequestration of Textile Fibrous Microplastics: From Mechanistic Insights to Rational Functional Design" C 12, no. 2: 31. https://doi.org/10.3390/c12020031
APA StyleCruz, K., & Li, S. (2026). Engineered Biochar for the Sequestration of Textile Fibrous Microplastics: From Mechanistic Insights to Rational Functional Design. C, 12(2), 31. https://doi.org/10.3390/c12020031