Bovine Serum Albumin Molecularly Imprinted Electrochemical Sensors Modified by Carboxylated Multi-Walled Carbon Nanotubes/CaAlg Hydrogels
Abstract
:1. Introduction
2. Results and Discussion
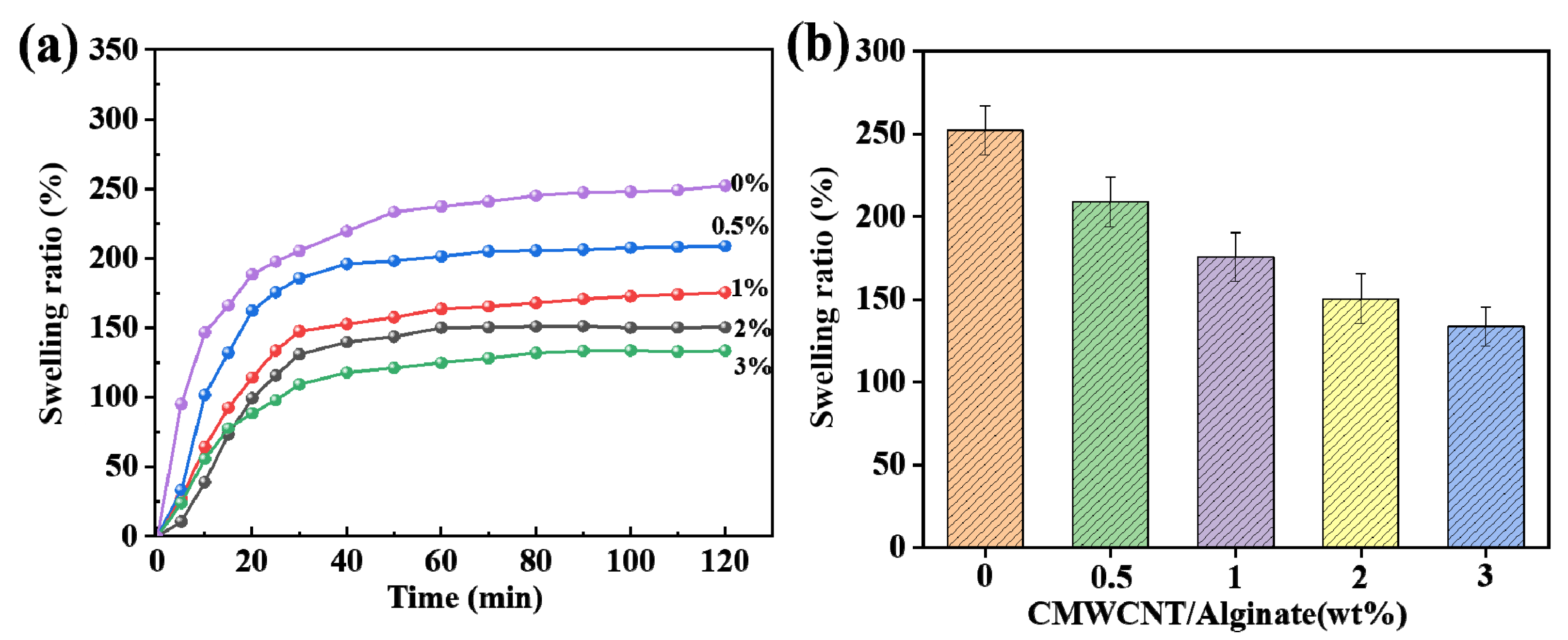
2.1. Characterizations of BSA Molecularly Imprinted CMWCNT/CaAlg Hydrogel Films
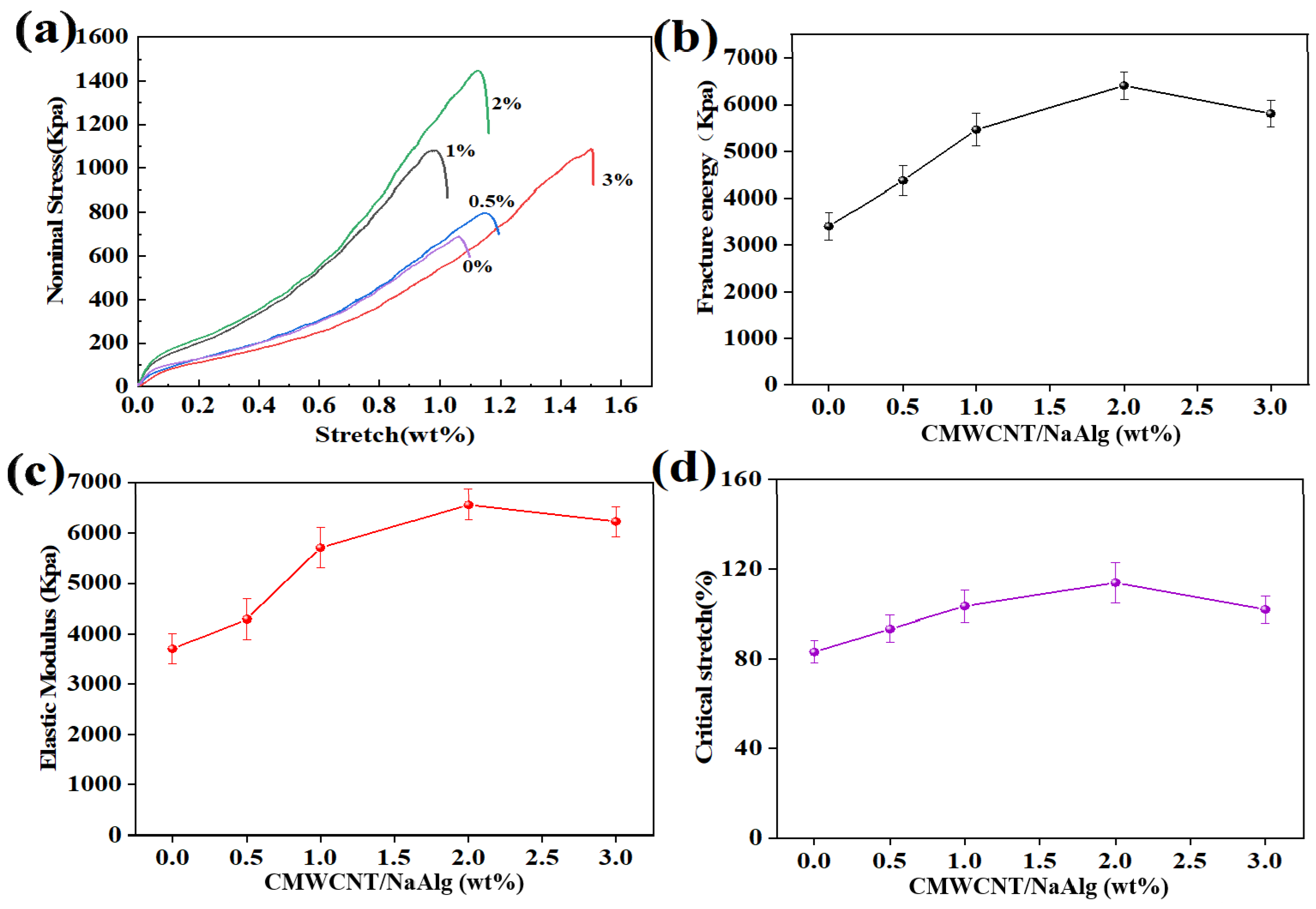
2.2. Mechanical Properties of BSA Molecularly Imprinted CMWCNT/CaAlg Hydrogel Films
2.3. Adsorption Properties of BSA Molecularly Imprinted CMWCNT/CaAlg Hydrogel Films
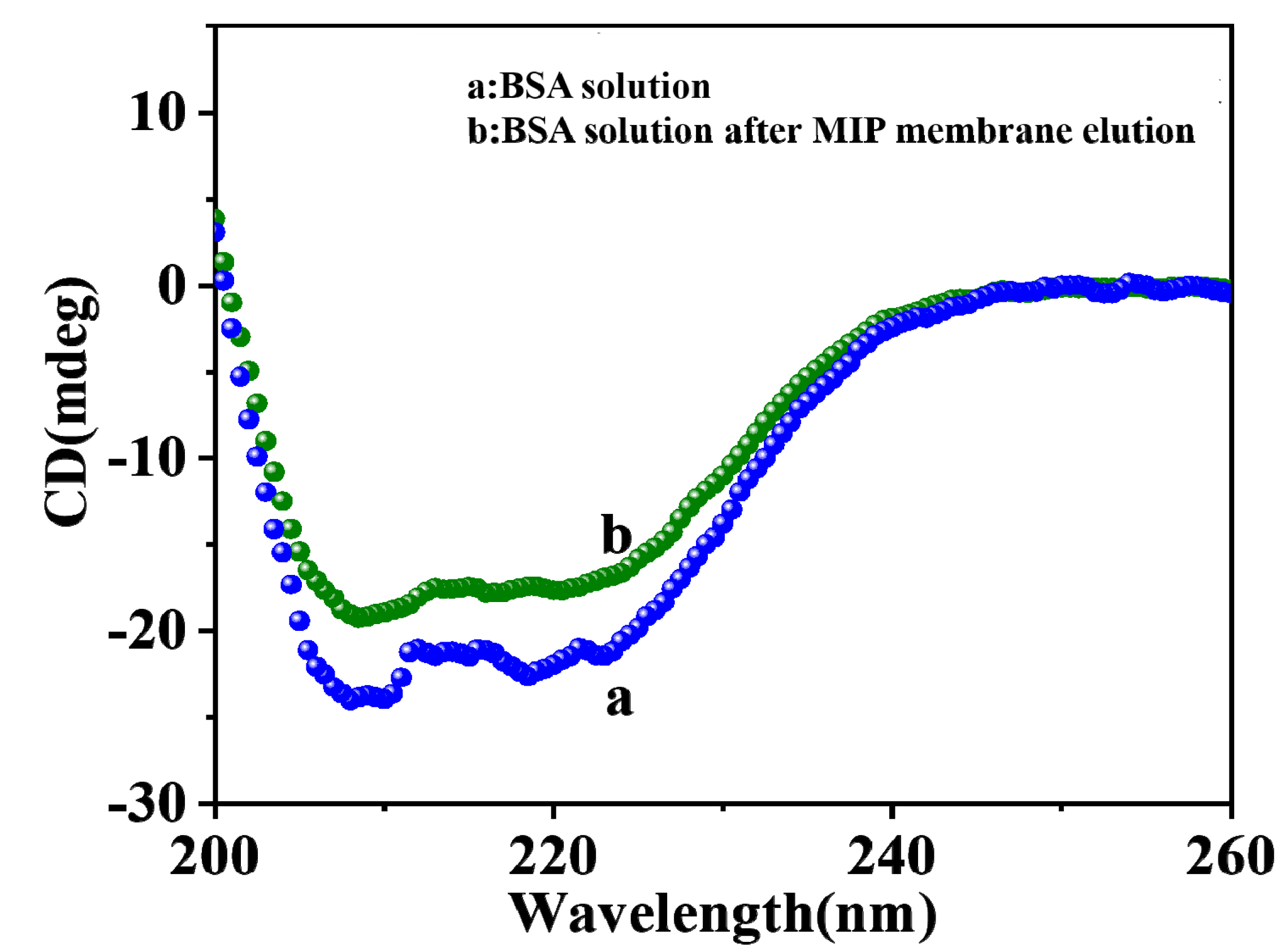
2.4. Effect of Alginate on BSA Conformational
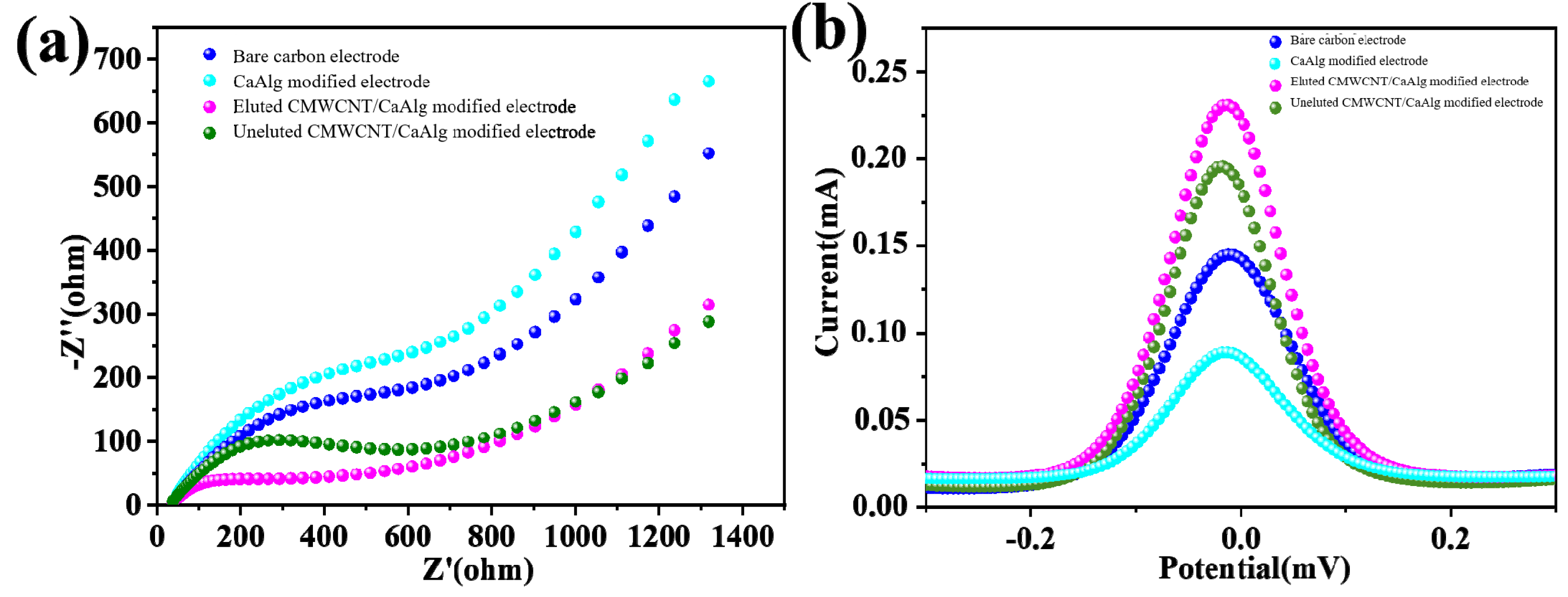
2.5. Electrochemical Characterization of Different Electrode Surfaces
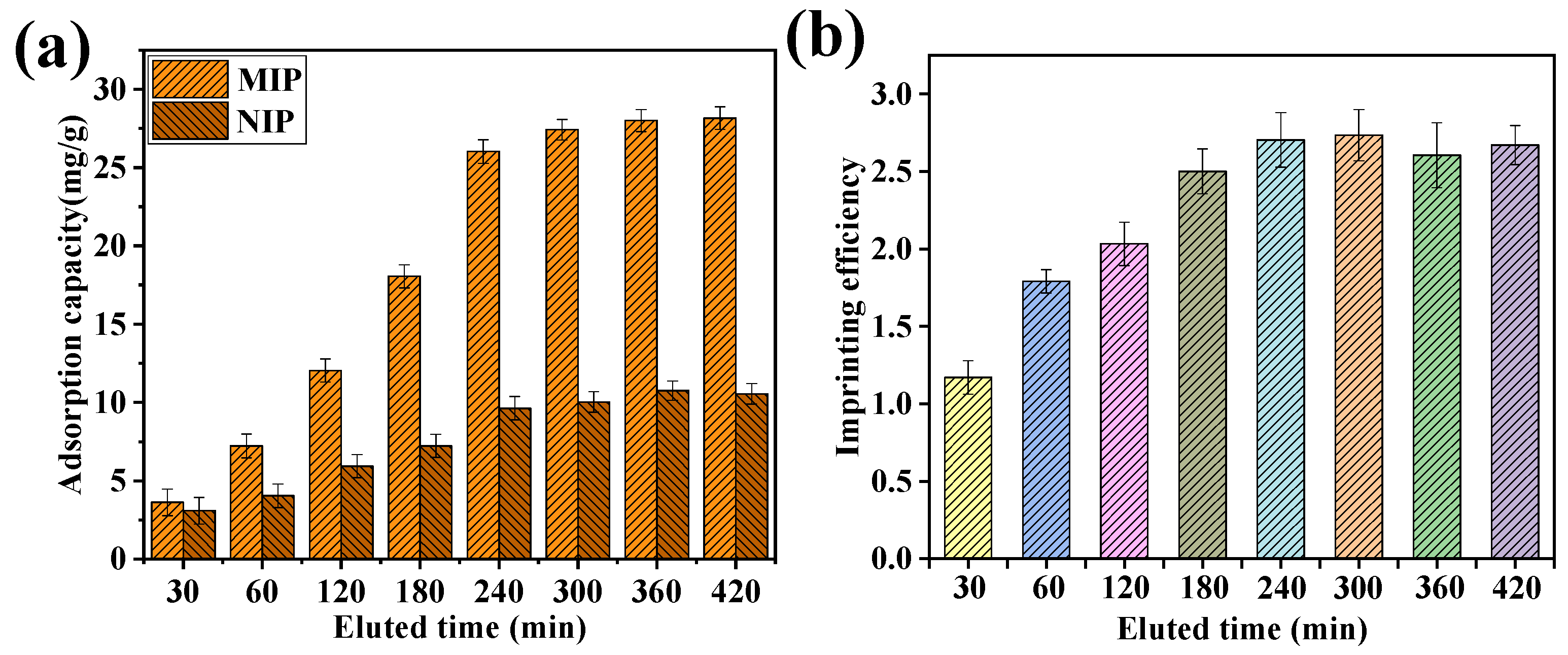
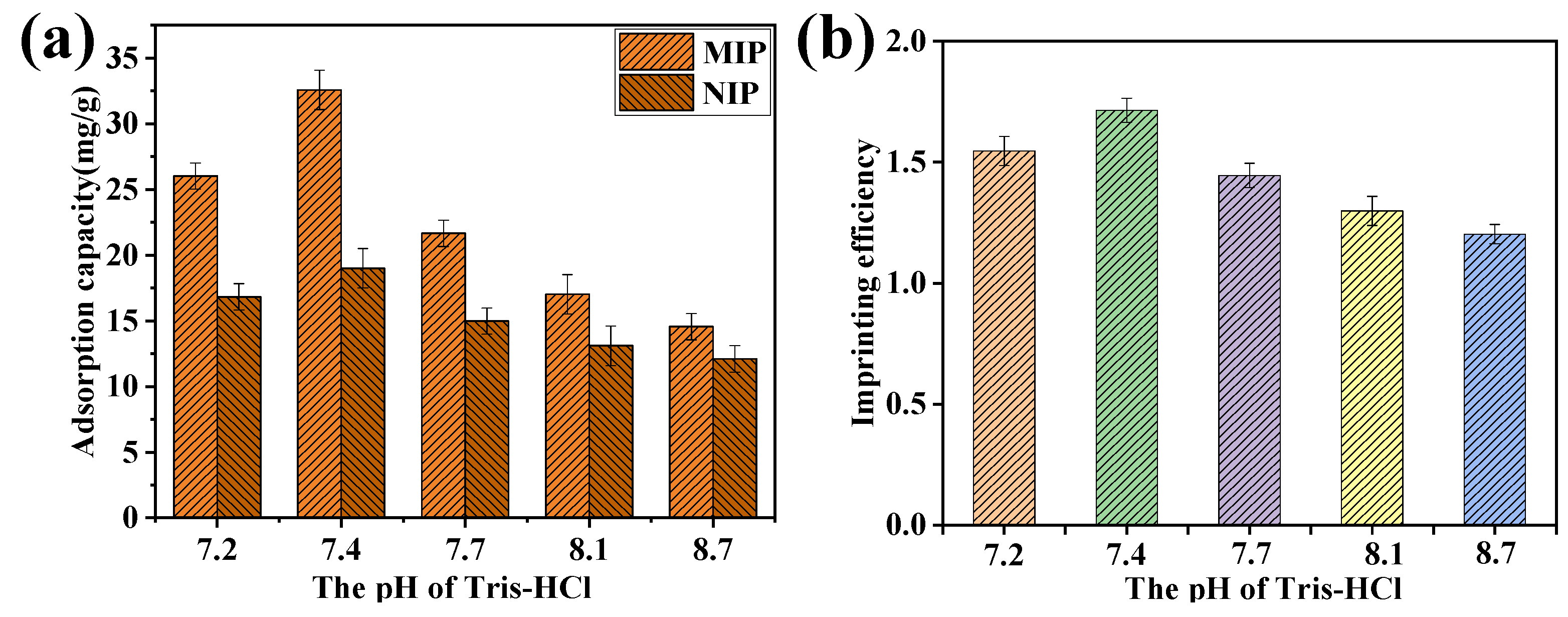
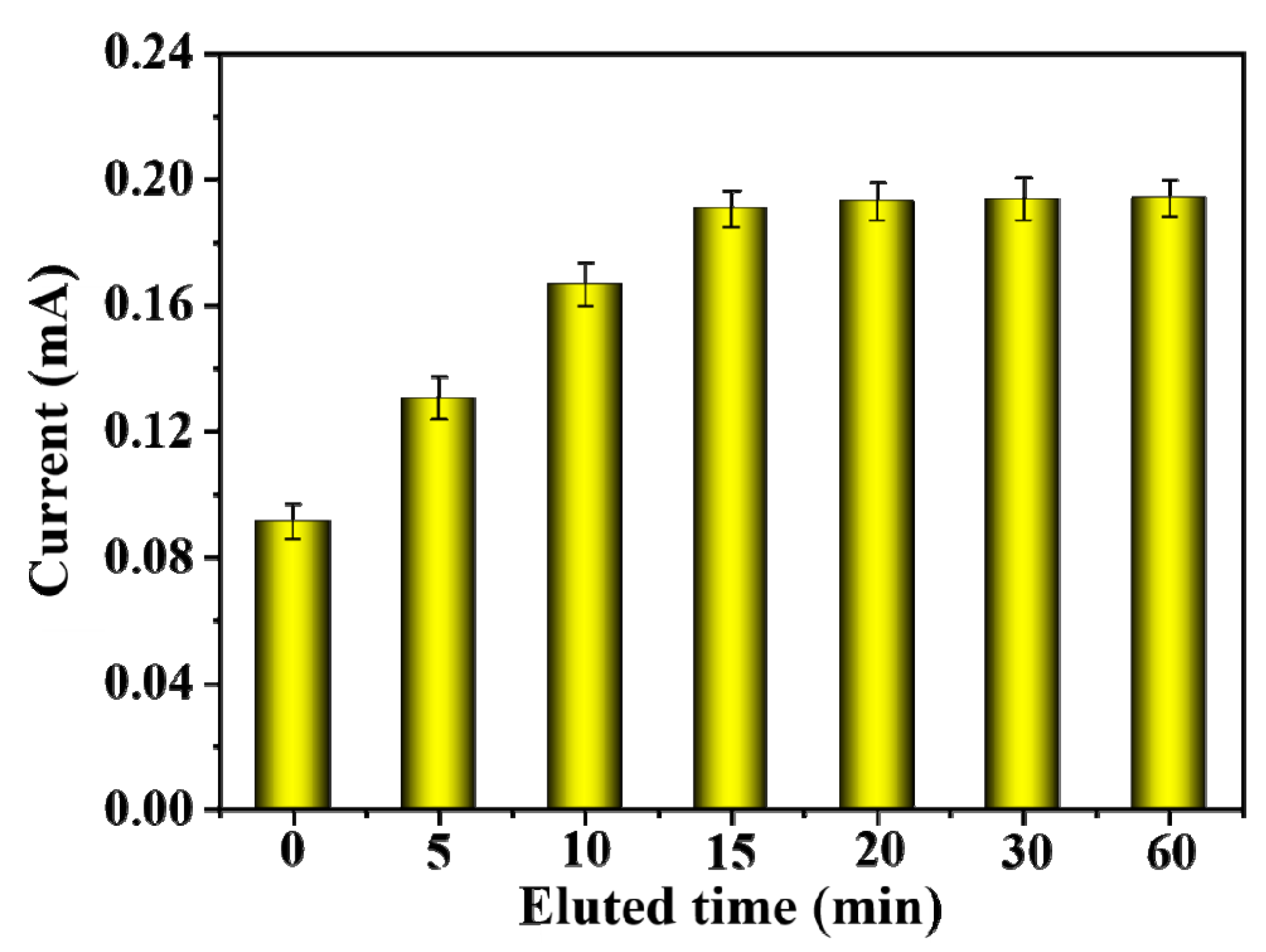
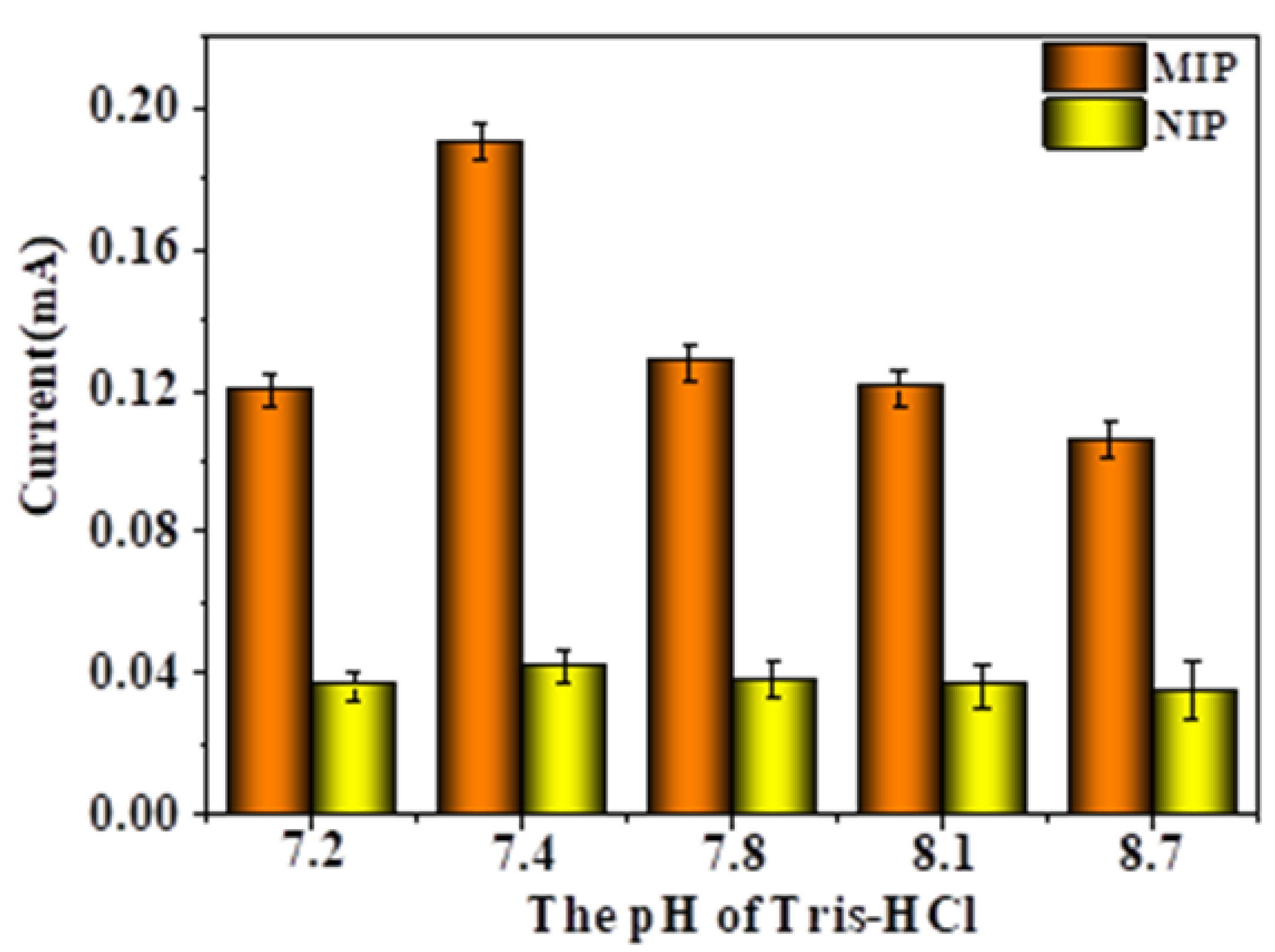
2.6. Effect of Eluent Times, Adsorption Times, and Eluent pH on DPV Response Currents of the MIP-Modified Electrodes
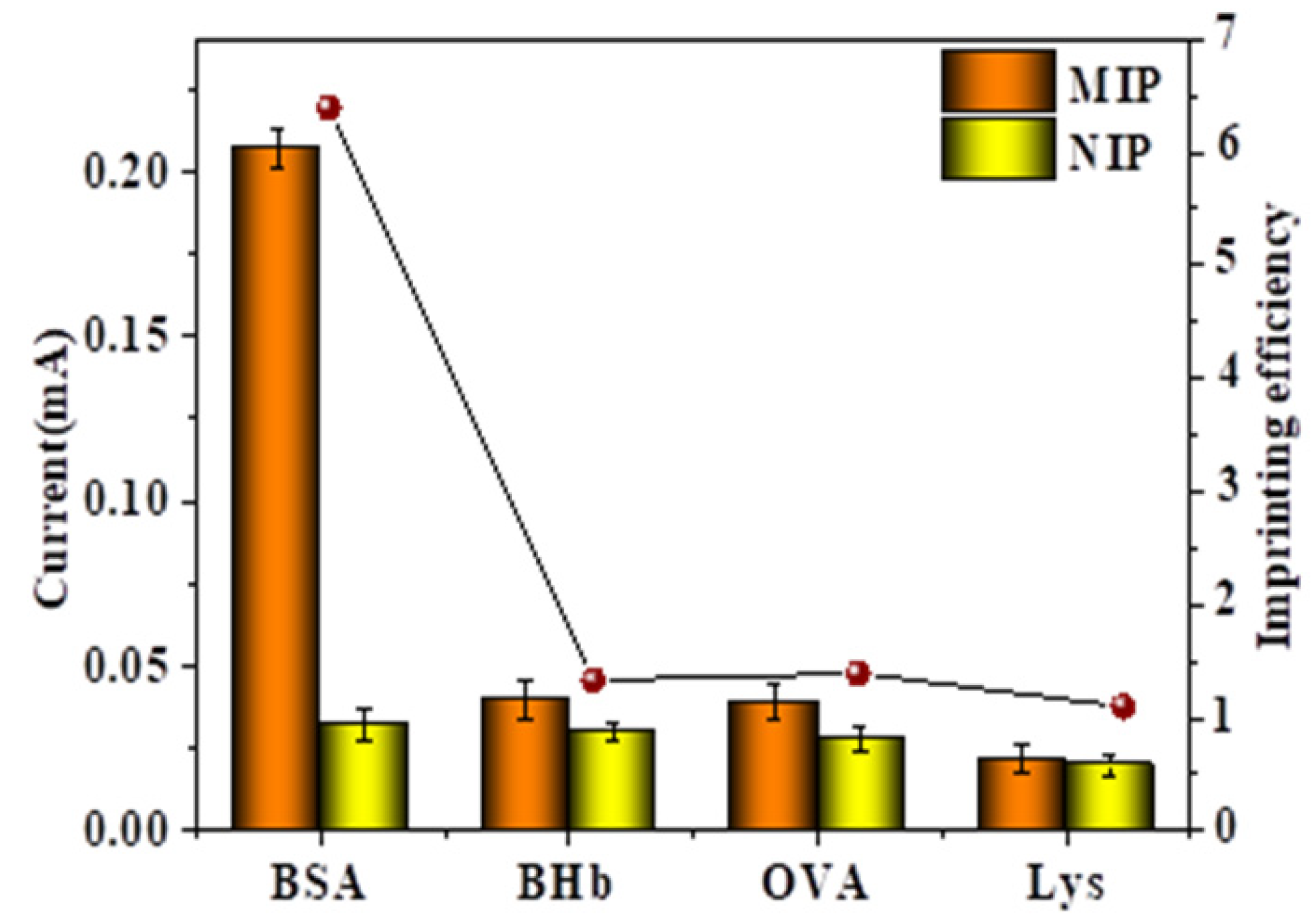
2.7. Selective Detection and Imprinting Efficiency of the MIP- and NIP-Modified Electrodes
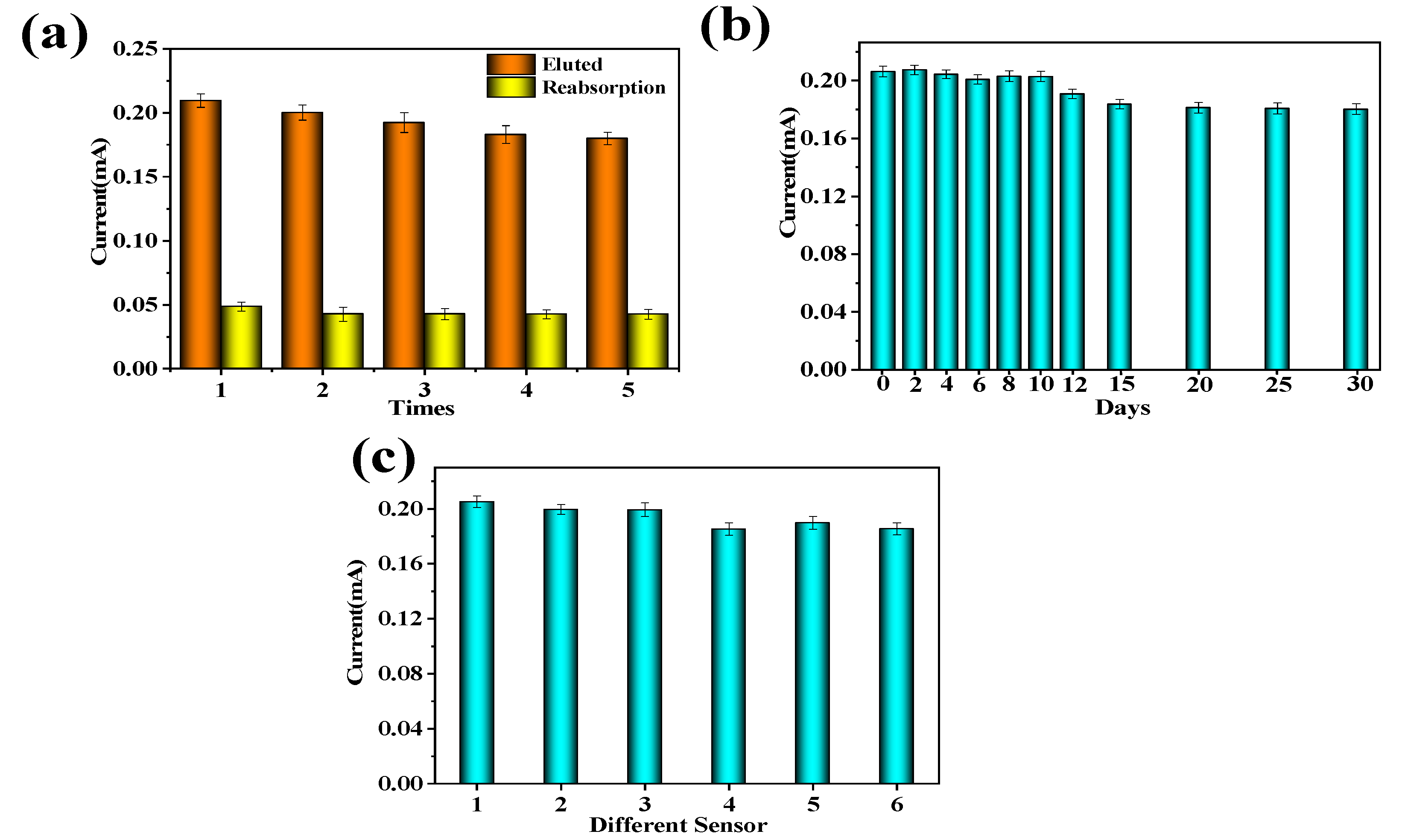
2.8. Reusability, Stability, and Reproducibility of MIP-Modified Electrodes for BSA Detection
2.9. Linear Range, Detection Limit of the Sensor, and Comparison with Other Materials
3. Conclusions
4. Materials and Methods
4.1. Materials
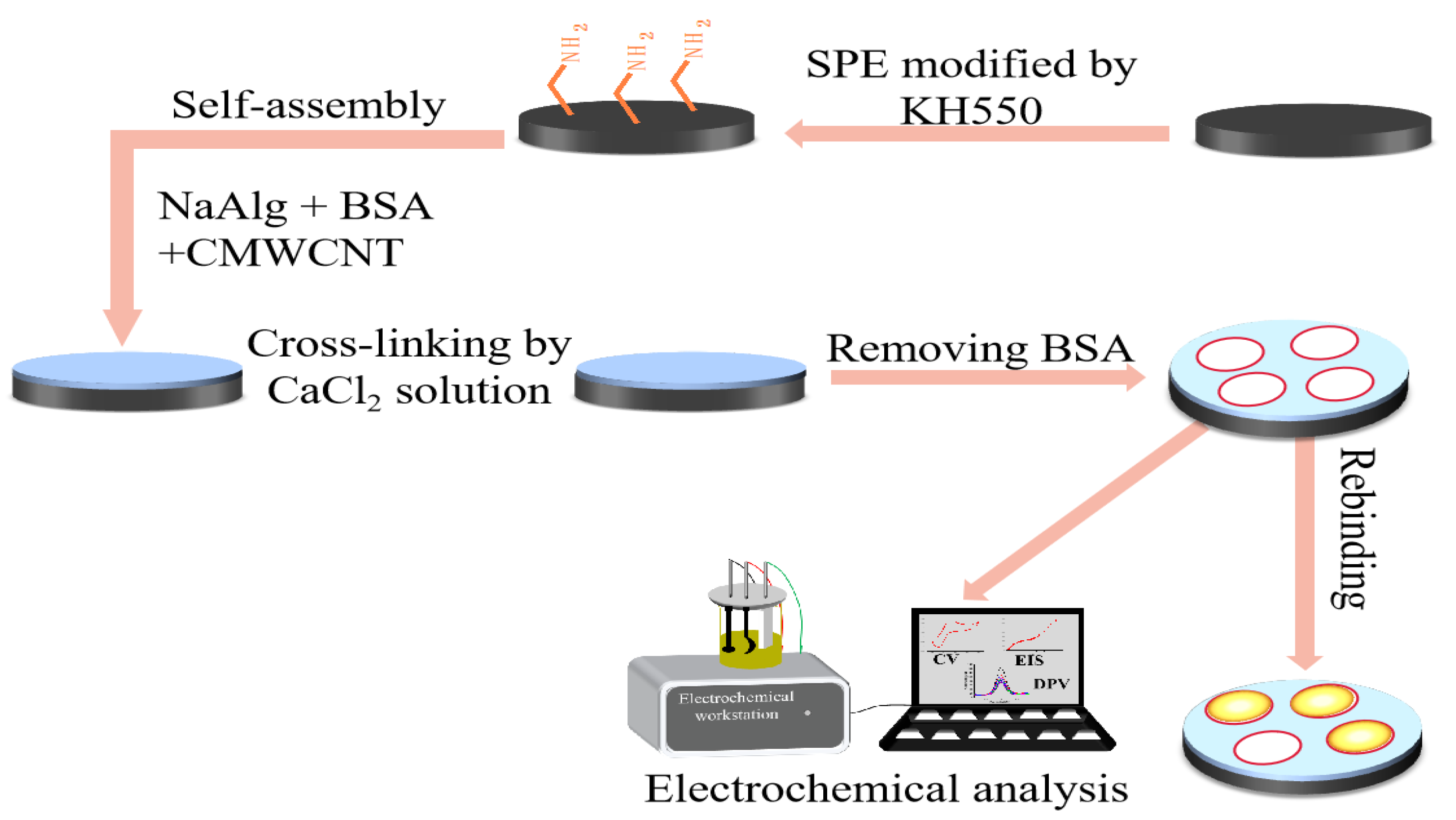
4.2. Preparation of BSA Molecularly Imprinted CMWCNT/CaAlg Hydrogel Films
4.3. Preparation of BSA Molecularly Imprinted CMWCNT/CaAlg Hydrogel-Modified Electrochemical Sensors
4.4. Characterizations
4.5. Electrochemical Testing
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| NaAlg | sodium alginate |
| BSA | bovine serum albumin |
| CMWCNT | carboxylated multi-wall carbon nanotube |
| CaAlg | calcium alginate |
| MIP | molecularly imprinted polymer |
| NIP | non-imprinted polymer |
| EIS | electrochemical impedance spectroscopy |
| DPV | differential pulse voltammetry |
| CD | circular dichroism |
| Tris | three (hydroxymethyl) aminomethane |
| BHb | bovine hemoglobin |
| OVA | ovalbumin |
| Lys | lysozyme |
| IF | imprinting efficiency |
| ΔI | current change value |
References
- Shrivastav, A.M.; Sharma, G.; Jha, R. Hypersensitive and selective biosensing based on microfiber interferometry and molecular imprinted nanoparticles. Biosens. Bioelectron. 2019, 141, 111347. [Google Scholar] [CrossRef] [PubMed]
- Mackay, D.; Bliek, J.; Kagami, M.; Tenorio-Castano, J.; Pereda, A.; Brioude, F.; Netchine, I.; Papingi, D.; de Franco, E.; Lever, M.; et al. First step towards a consensus strategy for multi-locus diagnostic testing of imprinting disorders. Clin. Epigenetics 2022, 14, 143. [Google Scholar] [CrossRef]
- Zhang, G.Y.; Ali, M.M.; Feng, X.; Zhou, J.T.; Hu, L.H. Mesoporous molecularly imprinted materials: From preparation to biorecognition and analysis. Trac-Trends Anal. Chem. 2021, 144, 116426. [Google Scholar] [CrossRef]
- Korde, B.A.; Mankar, J.S.; Phule, S.; Krupadam, R.J. Nanoporous imprinted polymers (nanoMIPs) for controlled release of cancer drug. Mat. Sci. Eng. C-Mater. 2019, 99, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Baker, M. Antibody anarchy: A call to order. Nature 2015, 527, 545–551. [Google Scholar] [CrossRef]
- Bradbury, A.; Pluckthun, A. Standardize antibodies used in research. Nature 2015, 518, 27–29. [Google Scholar] [CrossRef] [PubMed]
- BelBruno, J.J. Molecularly imprinted polymers. Chem. Rev. 2019, 119, 94–119. [Google Scholar] [CrossRef]
- Haupt, K.; Rangel, P.X.M.; Bui, B.T.S. Molecularly imprinted polymers: Antibody mimics for bioimaging and therapy. Chem. Rev. 2020, 120, 9554–9582. [Google Scholar] [CrossRef] [PubMed]
- Gopalan, A.I.; Komathi, S.; Muthuchamy, N.; Lee, K.P.; Whitcombe, M.J.; Dhana, L.; Sai-Anand, G. Functionalized conjugated polymers for sensing and molecular imprinting applications. Prog. Polym. Sci. 2019, 88, 1–129. [Google Scholar]
- Yan, M.; Wu, Y.L.; Zhang, K.C.; Lin, R.X.; Jia, S.H.; Lu, J.; Xing, W.D. Multifunctional-imprinted nanocomposite films with thermo-responsive biocompatibility for selective/controllable recognition and separation application. J. Colloid Interface Sci. 2021, 582, 991–1002. [Google Scholar] [CrossRef]
- Wright, K.M.; Bowyer, M.C.; McCluskey, A.; Holdsworth, C.I. Molecular imprinting of benzylpiperazine: A comparison of the self-assembly and semi-covalent approaches. Int. J. Mol. Sci. 2023, 24, 5117. [Google Scholar] [CrossRef]
- Xu, J.S.; Shang, M.; Liu, J.; Chen, X.; Cao, Y.H. Simultaneous self-assembly of molecularly imprinted magnetic nanoparticles to construct a magnetically responsive photonic crystals sensor for bisphenol A. Sens. Actuator B-Chem. 2021, 338, 129858. [Google Scholar] [CrossRef]
- Liang, W.X.; Hu, H.W.; Guo, P.R.; Ma, Y.F.; Li, P.Y.; Zheng, W.R.; Zhang, M. Combining pickering emulsion polymerization with molecular imprinting to prepare polymer microspheres for selective solid-phase extraction of malachite green. Polymers 2017, 9, 344. [Google Scholar] [CrossRef]
- Kryscio, D.R.; Fleming, M.Q.; Peppas, N.A. Protein conformational studies for macromolecularly imprinted polymers. Macromol. Biosci. 2012, 12, 1137–1144. [Google Scholar] [CrossRef]
- Qian, L.W.; Hu, X.L.; Guan, P.; Wang, D.; Li, J.; Du, C.B.; Song, R.Y.; Wang, C.L.; Song, W.Q. The effectively specific recognition of bovine serum albumin imprinted silica nanoparticles by utilizing a macromolecularly functional monomer to stabilize and imprint template. Anal. Chim. Acta 2015, 884, 97–105. [Google Scholar] [CrossRef]
- Xing, R.R.; Wang, S.S.; Bie, Z.J.; He, H.; Liu, Z. Preparation of molecularly imprinted polymers specific to glycoproteins, glycans and monosaccharides via boronate affinity controllable-oriented surface imprinting. Nat. Protoc. 2017, 12, 964–987. [Google Scholar] [CrossRef]
- Li, S.W.; Yang, K.G.; Deng, N.; Min, Y.; Liu, L.K.; Zhang, L.H.; Zhang, Y.K. Thermoresponsive epitope surface-imprinted nanoparticles for specific capture and release of target protein from human plasma. ACS Appl. Mater. Interfaces 2016, 8, 5747–5751. [Google Scholar] [CrossRef]
- Liang, R.N.; Ding, J.W.; Gao, S.S.; Qin, W. Mussel-inspired surface-imprinted sensors for potentiometric label-free detection of biological species. Angew. Chem. Int. Ed. 2017, 56, 6833–6837. [Google Scholar] [CrossRef]
- Shi, H.Q.; Tsai, W.B.; Garrison, M.D.; Ferrari, S.; Ratner, B.D. Template-imprinted nanostructured surfaces for protein recognition. Nature 1999, 398, 593–597. [Google Scholar] [CrossRef]
- Yarman, A.; Kurbanoglu, S.; Zebger, I.; Scheller, F.W. Simple and robust: The claims of protein sensing by molecularly imprinted polymers. Sens. Actuat. B-Chem. 2021, 330, 129369. [Google Scholar] [CrossRef]
- Qi, M.; Zhao, K.Y.; Bao, Q.W.; Pan, P.; Zhao, Y.W.; Yang, Z.C.; Wang, H.Q.; Wei, J.F. Adsorption and electrochemical detection of bovine serum albumin imprinted calcium alginate hydrogel membrane. Polymers 2019, 11, 622. [Google Scholar] [CrossRef]
- Sun, J.Y.; Zhao, X.H.; Illeperuma, W.R.K.; Chaudhuri, O.; Oh, K.H.; Mooney, D.J.; Vlassak, J.J.; Suo, Z.G. Highly stretchable and tough hydrogels. Nature 2012, 489, 133–136. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.Y.; Chen, T.; Lin, B.B.; Cui, W.K.; Kan, B.H.; Yang, N.; Zhou, X.Y.; Zhang, X.X.; Wei, J.F. Adsorption and recognition of protein molecular imprinted calcium alginate/polyacrylamide hydrogel film with good regeneration performance and high toughness. React. Funct. Polym. 2015, 87, 7–14. [Google Scholar] [CrossRef]
- Xu, L.J.; Xie, W.B.; Zhao, K.Y.; Shi, W.X.; Jiang, J.; Lin, L.G. Chlorine-resistance, reswelling after drying and molecule/ions separation properties of carboxyl multi-walled carbon nanotubes/calcium alginate composite hydrogel membrane. Compos. Commun. 2023, 37, 101412. [Google Scholar] [CrossRef]
- Bi, H.N.; Tang, L.; Gao, X.; Jia, J.J.; Lv, H.H. Spectroscopic analysis on the binding interaction between tetracycline hydrochloride and bovine proteins beta-casein, alpha-lactalbumin. J. Lumin. 2016, 178, 72–83. [Google Scholar] [CrossRef]
- Raja, S.T.K.; Thiruselvi, T.; Mandal, A.B.; Gnanamani, A. pH and redox sensitive albumin hydrogel: A self-derived biomaterial. Sci. Rep. 2015, 5, 15977. [Google Scholar] [CrossRef]
- Ojha, H.; Mishra, K.; Hassan, M.I.; Chaudhury, N.K. Spectroscopic and isothermal titration calorimetry studies of binding interaction of ferulic acid with bovine serum albumin. Thermochim. Acta 2012, 548, 56–64. [Google Scholar] [CrossRef]
- Banerjee, M.; Pal, U.; Subudhhi, A.; Chakrabarti, A.; Basu, S. Interaction of Merocyanine 540 with serum albumins: Photophysical and binding studies. J. Photochem. Photobiol. B 2012, 108, 23–33. [Google Scholar] [CrossRef]
- Zhou, X.M.; Yue, Y.Y.; Yang, Q.; Yan, N.; Chen, X.G. Complexes between CI Acid Orange 6 and human serum albumin, a multi-spectroscopic approach to investigate the binding behavior. J. Lumin. 2011, 131, 1222–1228. [Google Scholar] [CrossRef]
- Wang, Q.; Ju, J.P.; Tan, Y.Q.; Hao, L.Y.; Ma, Y.L.; Wu, Y.; Zhang, H.W.; Xia, Y.Z.; Sui, K.Y. Controlled synthesis of sodium alginate electrospun nanofiber membranes for multi-occasion adsorption and separation of methylene blue. Carbohydr. Polym. 2019, 205, 125–134. [Google Scholar] [CrossRef]
- Zhao, W.; Li, B.; Xu, S.; Zhu, Y.; Liu, X.Y. A fabrication strategy for protein sensors based on an electroactive molecularly imprinted polymer: Cases of bovine serum albumin and trypsin sensing. Anal. Chim. Acta 2020, 1117, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.H.; Wan, F.W.; Zhang, C.C.; Yan, M.; Zhang, X.N.; Wang, S.W. Molecularly imprinted polymeric microspheres for determination of bovine serum albumin based on flow injection chemiluminescence sensor. Biosens. Bioelectron. 2010, 26, 632–637. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hu, Q.Z.; Tian, T.T.; Gao, Y.A.; Yu, L. A nonionic surfactant-decorated liquid crystal sensor for sensitive and selective detection of proteins. Anal. Chim. Acta 2016, 937, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.J.; Chen, Y.J.; Fang, M.Y.; Tian, Y.B.; Bai, G.Y.; Zhuo, K.L. Silanized carbon dot-based thermo-sensitive molecularly imprinted fluorescent sensor for bovine hemoglobin detection. Anal. Bioanal. Chem. 2020, 412, 5811–5817. [Google Scholar] [PubMed]


| Materials | Linear Range (mg mL−1) | Detection Limit (mg mL−1) | References |
|---|---|---|---|
| MIP microspheres | 1.0 × 10−5–5.0 × 10−3 | 1.5 × 10−6 | [32] |
| MIP/Cd Te quantum dots | 3.3 × 10−2–0.66 | 1.0 × 10−3 | [33] |
| Carbon dot | 2.0 × 10−2–0.1 | 8.5 × 10−4 | [34] |
| CaAlg/CMWCNT hydrogel MIP | 1.0 × 10−6–1.15 × 10−3 | 5.6 × 10−6 | This method |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, L.; Guo, Z.; Lin, Y.; Wei, X.; Zhao, K.; Yang, Z. Bovine Serum Albumin Molecularly Imprinted Electrochemical Sensors Modified by Carboxylated Multi-Walled Carbon Nanotubes/CaAlg Hydrogels. Gels 2023, 9, 673. https://doi.org/10.3390/gels9080673
Cheng L, Guo Z, Lin Y, Wei X, Zhao K, Yang Z. Bovine Serum Albumin Molecularly Imprinted Electrochemical Sensors Modified by Carboxylated Multi-Walled Carbon Nanotubes/CaAlg Hydrogels. Gels. 2023; 9(8):673. https://doi.org/10.3390/gels9080673
Chicago/Turabian StyleCheng, Letian, Zhilong Guo, Yuansheng Lin, Xiujuan Wei, Kongyin Zhao, and Zhengchun Yang. 2023. "Bovine Serum Albumin Molecularly Imprinted Electrochemical Sensors Modified by Carboxylated Multi-Walled Carbon Nanotubes/CaAlg Hydrogels" Gels 9, no. 8: 673. https://doi.org/10.3390/gels9080673
APA StyleCheng, L., Guo, Z., Lin, Y., Wei, X., Zhao, K., & Yang, Z. (2023). Bovine Serum Albumin Molecularly Imprinted Electrochemical Sensors Modified by Carboxylated Multi-Walled Carbon Nanotubes/CaAlg Hydrogels. Gels, 9(8), 673. https://doi.org/10.3390/gels9080673







