Ice-Template Crosslinked PVA Aerogels Modified with Tannic Acid and Sodium Alginate
Abstract
:1. Introduction
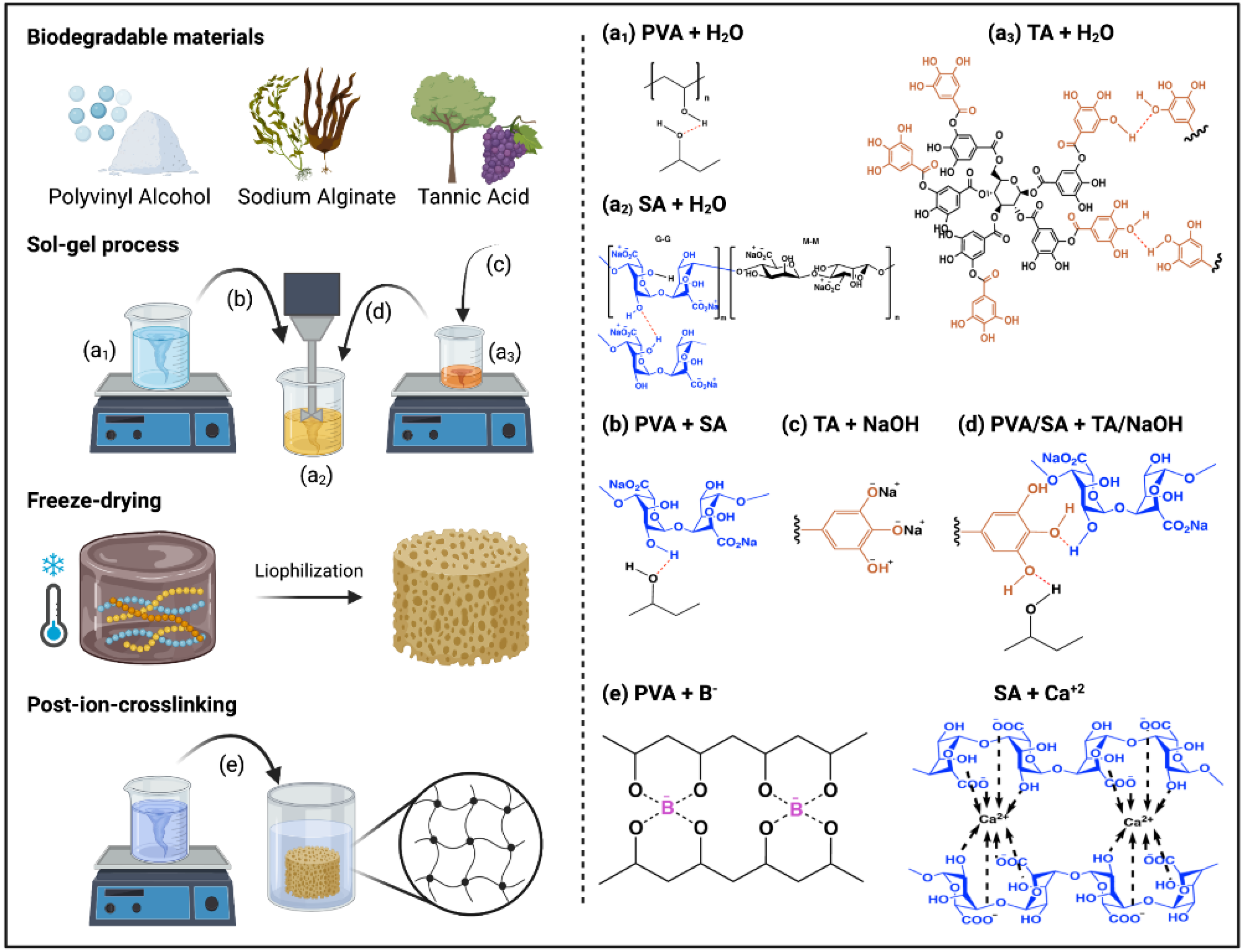
2. Results and Discussion
2.1. Density and Porosity
2.2. Morphology
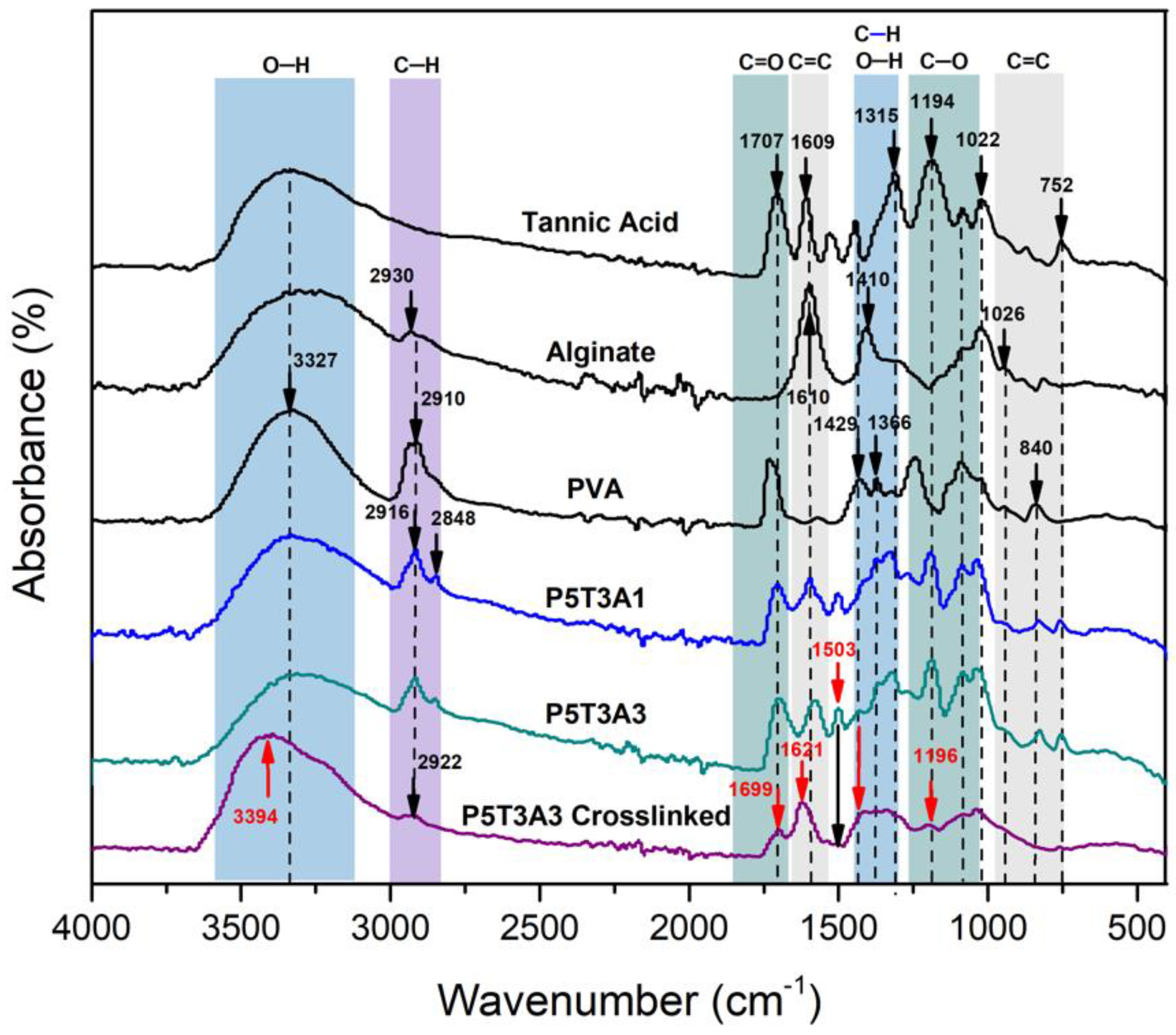
2.3. FTIR-ATR Spectroscopy
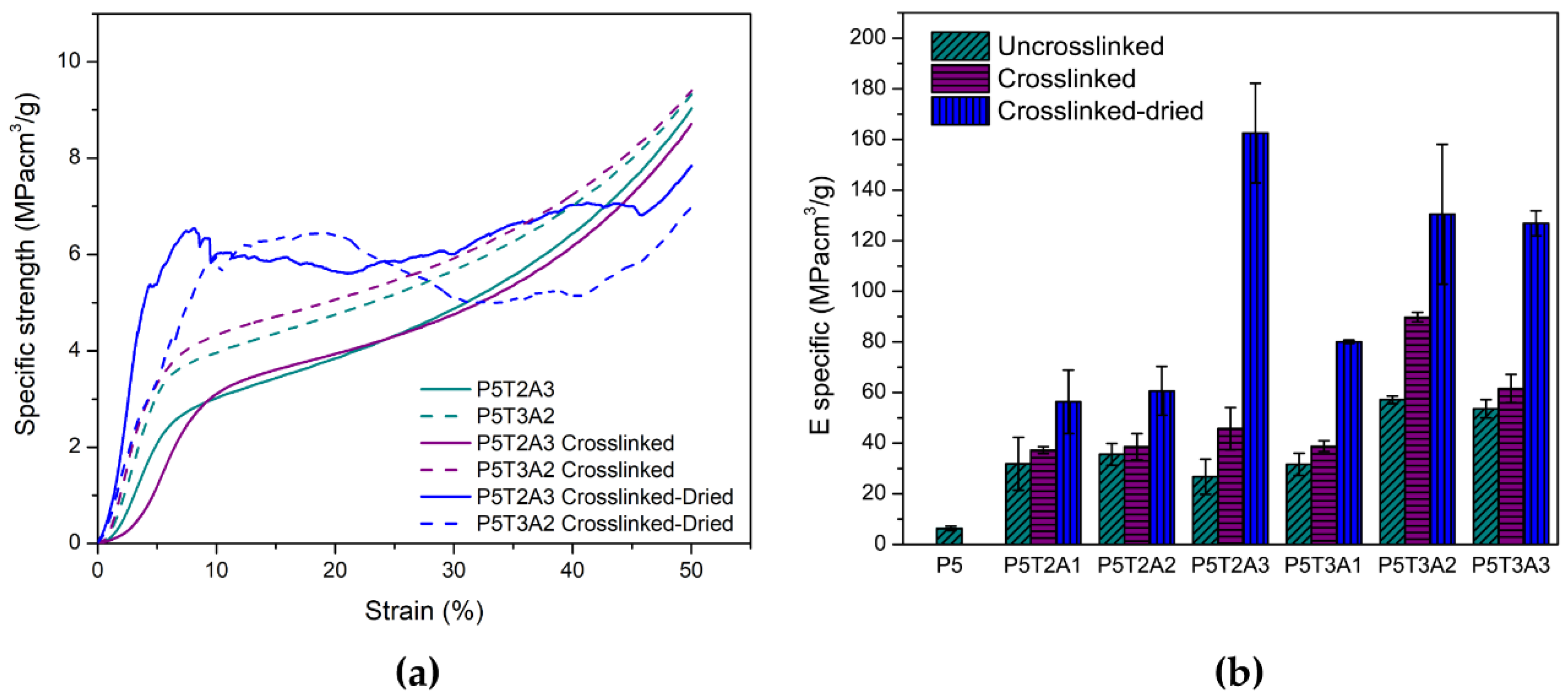
2.4. Mechanical Properties
2.5. Thermal Conductivity
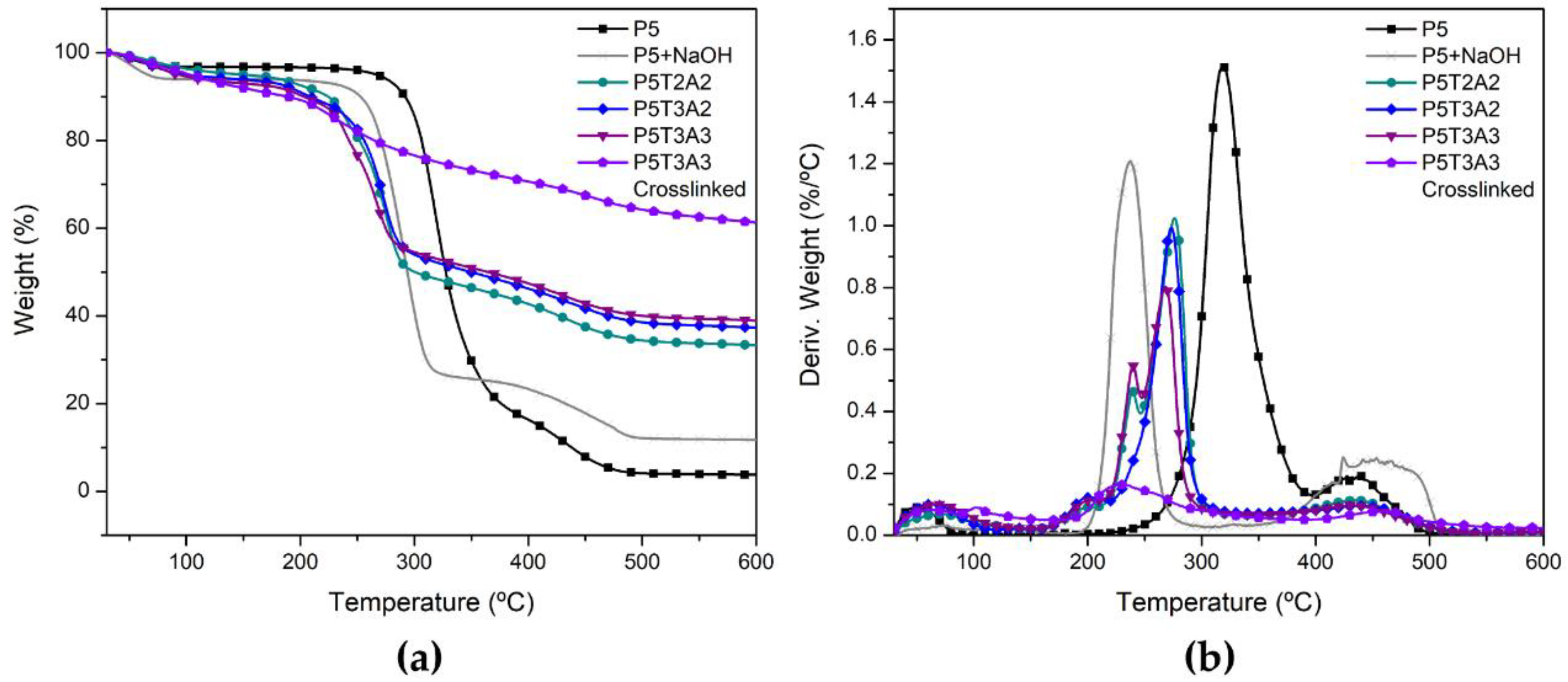
2.6. Thermal Degradation
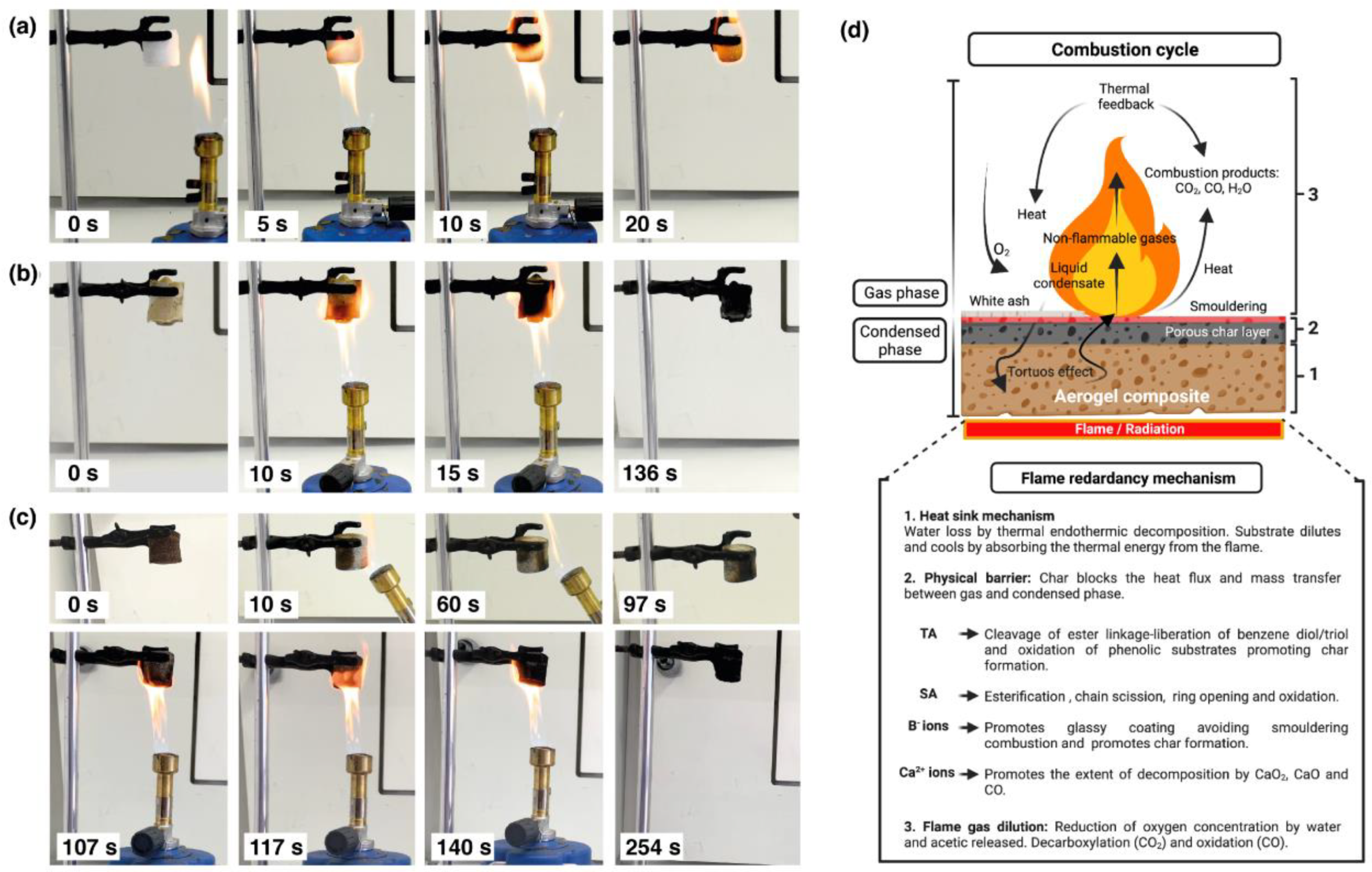
2.7. Combustion Behavior
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Aerogel Preparation
4.2.1. Post-Ion-Crosslinking Process
4.2.2. Characterization
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Verma, A.; Thakur, S.; Goel, G.; Raj, J.; Gupta, V.K.; Roberts, D.; Thakur, V.K. Bio-Based Sustainable Aerogels: New Sensation in CO2 Capture. Curr. Res. Green Sustain. Chem. 2020, 3, 100027. [Google Scholar] [CrossRef]
- Mao, J.; Iocozzia, J.; Huang, J.; Meng, K.; Lai, Y.; Lin, Z. Graphene Aerogels for Efficient Energy Storage and Conversion. Energy Environ. Sci. 2018, 11, 772–799. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, X.; Liu, L.; Zhang, Z.; Shen, J. Alumina-Doped Silica Aerogels for High-Temperature Thermal Insulation. Gels 2021, 7, 122. [Google Scholar] [CrossRef]
- Wang, X.; Xie, P.; Wan, K.; Miao, Y.; Liu, Z.; Li, X.; Wang, C. Mechanically Strong, Low Thermal Conductivity and Improved Thermal Stability Polyvinyl Alcohol-Graphene-Nanocellulose Aerogel. Gels 2021, 7, 170. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Sánchez-Soto, M.; Maspoch, M.L. Polymer/Clay Aerogel Composites with Flame Retardant Agents: Mechanical, Thermal and Fire Behavior. Mater. Des. 2013, 52, 609–614. [Google Scholar] [CrossRef]
- Wang, H.; Cao, M.; Zhao, H.-B.; Liu, J.-X.; Geng, C.-Z.; Wang, Y.-Z. Double-Cross-Linked Aerogels towards Ultrahigh Mechanical Properties and Thermal Insulation at Extreme Environment. Chem. Eng. J. 2020, 399, 125698. [Google Scholar] [CrossRef]
- Zhou, L.; Zhai, S.; Chen, Y.; Xu, Z. Anisotropic Cellulose Nanofibers/Polyvinyl Alcohol/Graphene Aerogels Fabricated by Directional Freeze-Drying as Effective Oil Adsorbents. Polymers 2019, 11, 712. [Google Scholar] [CrossRef] [Green Version]
- Li, X.-L.; Chen, M.-J.; Chen, H.-B. Facile Fabrication of Mechanically-Strong and Flame Retardant Alginate/Clay Aerogels. Compos. Part B Eng. 2019, 164, 18–25. [Google Scholar] [CrossRef]
- Chen, H.-B.; Wang, Y.-Z.; Sánchez-Soto, M.; Schiraldi, D.A. Low Flammability, Foam-like Materials Based on Ammonium Alginate and Sodium Montmorillonite Clay. Polymer 2012, 53, 5825–5831. [Google Scholar] [CrossRef]
- Kabir, I.I.; Sorrell, C.C.; Mofarah, S.S.; Yang, W.; Yuen, A.C.Y.; Nazir, M.T.; Yeoh, G.H. Alginate/Polymer-Based Materials for Fire Retardancy: Synthesis, Structure, Properties, and Applications. Polym. Rev. 2021, 61, 357–414. [Google Scholar] [CrossRef]
- Nam, S.; Condon, B.D.; Xia, Z.; Nagarajan, R.; Hinchliffe, D.J.; Madison, C.A. Intumescent Flame-Retardant Cotton Produced by Tannic Acid and Sodium Hydroxide. J. Anal. Appl. Pyrolysis 2017, 126, 239–246. [Google Scholar] [CrossRef]
- Chen, Y.-N.; Peng, L.; Liu, T.; Wang, Y.; Shi, S.; Wang, H. Poly(Vinyl Alcohol)–Tannic Acid Hydrogels with Excellent Mechanical Properties and Shape Memory Behaviors. ACS Appl. Mater. Interfaces 2016, 8, 27199–27206. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Shao, L.; Dong, D.; Wang, F.; Zhang, Y.; Wang, Y. Bio-Inspired Natural Polyphenol Cross-Linking Poly(Vinyl Alcohol) Films with Strong Integrated Strength and Toughness. RSC Adv. 2016, 6, 69966–69972. [Google Scholar] [CrossRef]
- Cheng, Z.; DeGracia, K.; Schiraldi, D.A. Sustainable, Low Flammability, Mechanically-Strong Poly(Vinyl Alcohol) Aerogels. Polymers 2018, 10, 1102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grant, G.T.; Morris, E.R.; Rees, D.A.; Smith, P.J.C.; Thom, D. Biological Interactions between Polysaccharides and Divalent Cations: The Egg-Box Model. FEBS Lett. 1973, 32, 195–198. [Google Scholar] [CrossRef] [Green Version]
- Ochiai, H.; Shimizu, S.; Tadokoro, Y.; Murakami, I. Complex Formation between Poly(Vinyl Alcohol) and Borate Ion. Polymer 1981, 22, 1456–1458. [Google Scholar] [CrossRef]
- Erel-Unal, I.; Sukhishvili, S.A. Hydrogen-Bonded Multilayers of a Neutral Polymer and a Polyphenol. Macromolecules 2008, 41, 3962–3970. [Google Scholar] [CrossRef]
- Ni, X.; Ke, F.; Xiao, M.; Wu, K.; Kuang, Y.; Corke, H.; Jiang, F. The Control of Ice Crystal Growth and Effect on Porous Structure of Konjac Glucomannan-Based Aerogels. Int. J. Biol. Macromol. 2016, 92, 1130–1135. [Google Scholar] [CrossRef]
- Sun, M.; Sun, H.; Wang, Y.; Sánchez-Soto, M.; Schiraldi, D.A. The Relation between the Rheological Properties of Gels and the Mechanical Properties of Their Corresponding Aerogels. Gels 2018, 4, 33. [Google Scholar] [CrossRef] [Green Version]
- Joseph, J.; Jemmis, E.D. Red-, Blue-, or No-Shift in Hydrogen Bonds: A Unified Explanation. J. Am. Chem. Soc. 2007, 129, 4620–4632. [Google Scholar] [CrossRef]
- Kumar, A.; Lee, Y.; Kim, D.; Rao, K.; Kim, J.; Park, S.; Haider, A.; Lee, D.; Han, S. Effect of Crosslinking Functionality on Microstructure, Mechanical Properties, and in Vitro Cytocompatibility of Cellulose Nanocrystals Reinforced Poly(Vinyl Alcohol)/Sodium Alginate Hybrid Scaffolds. Int. J. Biol. Macromol. 2017, 95, 962–973. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Yang, X.; Liu, W.; Xi, G.; Wang, M.; Liang, B.; Ma, Z.; Feng, Y.; Chen, H.; Shi, C. Tannic Acid Cross-Linked Polysaccharide-Based Multifunctional Hemostatic Microparticles for the Regulation of Rapid Wound Healing. Macromol. Biosci. 2018, 18, 1800209. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kwak, S.; Lee, S.; Cho, W.K.; Lee, J.K.; Kang, S.M. One-Step Functionalization of Zwitterionic Poly[(3-(Methacryloylamino)Propyl)Dimethyl(3-Sulfopropyl)Ammonium Hydroxide] Surfaces by Metal–Polyphenol Coating. Chem. Commun. 2015, 51, 5340–5342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Helmiyati; Aprilliza, M. Characterization and Properties of Sodium Alginate from Brown Algae Used as an Ecofriendly Superabsorbent. IOP Conf. Ser. Mater. Sci. Eng. 2017, 188, 012019. [Google Scholar] [CrossRef]
- Harahap, M.R.; Mauliza, N.; Asmara, A.P.; Lestari, E.C.; Afriani, W. The Effect of Seaweed Combination on the Extract of Robusta Coffee (Coffea Robusta) Waste Extract in Producing Facial Mask Products. Biomedika 2020, 13, 15–22. [Google Scholar] [CrossRef]
- Hu, T.; Liu, Q.; Gao, T.; Dong, K.; Wei, G.; Yao, J. Facile Preparation of Tannic Acid–Poly(Vinyl Alcohol)/Sodium Alginate Hydrogel Beads for Methylene Blue Removal from Simulated Solution. ACS Omega 2018, 3, 7523–7531. [Google Scholar] [CrossRef]
- Liu, R.; Ge, H.; Wang, X.; Luo, J.; Li, Z.; Liu, X. Three-Dimensional Ag–Tannic Acid–Graphene as an Antibacterial Material. New J. Chem. 2016, 40, 6332–6339. [Google Scholar] [CrossRef]
- Wu, N.; Niu, F.; Lang, W.; Xia, M. Highly Efficient Flame-Retardant and Low-Smoke-Toxicity Poly(Vinyl Alcohol)/Alginate/Montmorillonite Composite Aerogels by Two-Step Crosslinking Strategy. Carbohydr. Polym. 2019, 221, 221–230. [Google Scholar] [CrossRef]
- Abulateefeh, S.R.; Taha, M.O. Enhanced Drug Encapsulation and Extended Release Profiles of Calcium–Alginate Nanoparticles by Using Tannic Acid as a Bridging Cross-Linking Agent. J. Microencapsul. 2015, 32, 96–105. [Google Scholar] [CrossRef]
- Schmidt, M.P.; Siciliano, S.D.; Peak, D. The Role of Monodentate Tetrahedral Borate Complexes in Boric Acid Binding to a Soil Organic Matter Analogue. Chemosphere 2021, 276, 130150. [Google Scholar] [CrossRef]
- Sun, L.; Wang, J.; Liang, J.; Li, G. Boric Acid Cross-Linked 3D Polyvinyl Alcohol Gel Beads by NaOH-Titration Method as a Suitable Biomass Immobilization Matrix. J. Polym. Environ. 2020, 28, 532–541. [Google Scholar] [CrossRef] [Green Version]
- Abureesh, M.A.; Oladipo, A.A.; Gazi, M. Facile Synthesis of Glucose-Sensitive Chitosan–Poly(Vinyl Alcohol) Hydrogel: Drug Release Optimization and Swelling Properties. Int. J. Biol. Macromol. 2016, 90, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Konidari, M.V.; Papadokostaki, K.G.; Sanopoulou, M. Moisture-Induced Effects on the Tensile Mechanical Properties and Glass-Transition Temperature of Poly(Vinyl Alcohol) Films. J. Appl. Polym. Sci. 2011, 120, 3381–3386. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, X.; Tao, X.; Li, Z.; Li, X.; Zhang, Z. Polyvinyl Alcohol Composite Aerogel with Remarkable Flame Retardancy, Chemical Durability and Self-Cleaning Property. Compos. Commun. 2019, 15, 96–102. [Google Scholar] [CrossRef]
- Toledo, P.V.O.; Petri, D.F.S. Hydrophilic, Hydrophobic, Janus and Multilayer Xanthan Based Cryogels. Int. J. Biol. Macromol. 2019, 123, 1180–1188. [Google Scholar] [CrossRef]
- Zhang, L.; Ruesch, M.; Zhang, X.; Bai, Z.; Liu, L. Tuning Thermal Conductivity of Crystalline Polymer Nanofibers by Interchain Hydrogen Bonding. RSC Adv. 2015, 5, 87981–87986. [Google Scholar] [CrossRef]
- Mu, L.; He, J.; Li, Y.; Ji, T.; Mehra, N.; Shi, Y.; Zhu, J. Molecular Origin of Efficient Phonon Transfer in Modulated Polymer Blends: Effect of Hydrogen Bonding on Polymer Coil Size and Assembled Microstructure. J. Phys. Chem. C 2017, 121, 14204–14212. [Google Scholar] [CrossRef]
- Cheng, Z.-H.; Guo, M.-L.; Chen, X.-Y.; Wang, T.; Wang, Y.-Z.; Schiraldi, D.A. Reduction of PVA Aerogel Flammability by Incorporation of an Alkaline Catalyst. Gels 2021, 7, 57. [Google Scholar] [CrossRef]
- Kang, A.-H.; Shang, K.; Ye, D.-D.; Wang, Y.-T.; Wang, H.; Zhu, Z.-M.; Liao, W.; Xu, S.-M.; Wang, Y.-Z.; Schiraldi, D.A. Rejuvenated Fly Ash in Poly(Vinyl Alcohol)-Based Composite Aerogels with High Fire Safety and Smoke Suppression. Chem. Eng. J. 2017, 327, 992–999. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, S.-D.; Huang, Z.-Q.; Tang, G.; Hu, Y. Self-Assembly of Hydroxyapatite with Polyelectrolyte as a Green Flame Retardant for Poly(Vinyl Alcohol). J. Fire Sci. 2017, 35, 507–520. [Google Scholar] [CrossRef]
- Chen, H.-B.; Wang, Y.-Z.; Schiraldi, D.A. Preparation and Flammability of Poly(Vinyl Alcohol) Composite Aerogels. ACS Appl. Mater. Interfaces 2014, 6, 6790–6796. [Google Scholar] [CrossRef] [PubMed]
- Shang, K.; Yang, J.-C.; Cao, Z.-J.; Liao, W.; Wang, Y.-Z.; Schiraldi, D.A. Novel Polymer Aerogel toward High Dimensional Stability, Mechanical Property, and Fire Safety. ACS Appl. Mater. Interfaces 2017, 9, 22985–22993. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, R.L.; Morse, J.G.; Morse, K.W. Main Group Elements. In Encyclopedia of Physical Science and Technology, 3rd ed.; Meyers, R.A., Ed.; Academic Press: New York, NY, USA, 2003; pp. 1–30. ISBN 978-0-12-227410-7. [Google Scholar]


| Sample | Uncrosslinked | Crosslinked | ||||
|---|---|---|---|---|---|---|
| P5 | 0.075 ± 0.005 | 94.5 | 94.1 | - | - | - |
| P5T2A1 | 0.157 ± 0.023 | 89.2 | 88.9 | 0.314 ± 0.048 | 0.244 ± 0.047 | 69.5 |
| P5T2A2 | 0.163 ± 0.019 | 88.9 | 88.7 | 0.415 ± 0.020 | 0.300 ± 0.004 | 75.2 |
| P5T2A3 | 0.169 ± 0.012 | 88.6 | 88.5 | 0.359 ± 0.002 | 0.294 ± 0.008 | 79.1 |
| P5T3A1 | 0.158 ± 0.034 | 89.1 | 89.1 | 0.258 ± 0.012 | 0.217 ± 0.001 | 83.4 |
| P5T3A2 | 0.164 ± 0.023 | 88.9 | 88.8 | 0.247 ± 0.031 | 0.211 ± 0.020 | 81.2 |
| P5T3A3 | 0.167 ± 0.006 | 88.9 | 88.7 | 0.283 ± 0.005 | 0.233 ± 0.005 | 84.3 |
| Sample | Uncrosslinked | Crosslinked | Crosslinked-Dried | |||
|---|---|---|---|---|---|---|
| Esp (MPa cm3/g) | σysp (MPa cm3/g) | Esp (MPa cm3/g) | σysp (MPa cm3/g) | Esp (MPa cm3/g) | σysp (MPa cm3/g) | |
| P5 | 6.3 ± 0.8 | 0.25 ± 0.01 | - | - | - | - |
| P5T2A1 | 31.9 ± 5.4 | 1.77 ± 0.67 | 37.2 ± 1.4 | 2.02 ± 0.36 | 56.3 ± 3.5 | 2.17 ± 0.04 |
| P5T2A2 | 35.6 ± 4.3 | 1.86 ± 0.55 | 38.7 ± 5.2 | 2.65 ± 0.60 | 60.7 ± 5.6 | 5.42 ± 0.78 |
| P5T2A3 | 26.7 ± 6.9 | 2.17 ± 0.61 | 45.7 ± 5.3 | 2.42 ± 0.38 | 162.5 ± 9.5 | 5.87 ± 0.76 |
| P5T3A1 | 31.6 ± 4.3 | 1.50 ± 0.31 | 38.8 ± 2.1 | 1.96 ± 0.26 | 80.1 ± 6.6 | 5.75 ± 0.88 |
| P5T3A2 | 57.1 ± 1.5 | 2.07 ± 0.18 | 89.7 ± 1.8 | 2.61 ± 0.76 | 130.4 ± 1.6 | 5.85 ± 0.03 |
| P5T3A3 | 53.6 ± 3.6 | 2.45 ± 0.10 | 61.6 ± 5.6 | 2.58 ± 0.01 | 127.0 ± 5.1 | 5.15 ± 0.67 |
| Sample | Uncrosslinked | Crosslinked |
|---|---|---|
| λ (W/m·K) | λ (W/m·K) | |
| P5 | 0.030 ± 0.002 | - |
| P5T2A1 | 0.043 ± 0.001 | 0.054 ± 0.003 |
| P5T2A2 | 0.044 ± 0.001 | 0.059 ± 0.010 |
| P5T2A3 | 0.046 ± 0.002 | 0.060 ± 0.009 |
| P5T3A1 | 0.043 ± 0.001 | 0.049 ± 0.001 |
| P5T3A2 | 0.044 ± 0.001 | 0.051 ± 0.004 |
| P5T3A3 | 0.046 ± 0.002 | 0.057 ± 0.004 |
| Sample | Uncrosslinked | Crosslinked | ||||||
|---|---|---|---|---|---|---|---|---|
(°C) | (°C) | (%/°C) | WR (%) | (°C) | (°C) | (%/°C) | WR (%) | |
| P5 | 297.3 | 317.4 | 1.52 | 3.8 | - | - | - | - |
| P5 + NaOH | 218.6 | 237.5 | 1.21 | 27.1 | - | - | - | - |
| P5T2A1 | 178.9 | 277.1 | 1.07 | 33.2 | 214.2 | 231.6 | 0.158 | 52.3 |
| P5T2A2 | 177.1 | 273.8 | 1.05 | 34.1 | 217.3 | 235.7 | 0.160 | 50.5 |
| P5T2A3 | 177.3 | 273.6 | 0.92 | 34.2 | 211.7 | 232.0 | 0.208 | 59.9 |
| P5T3A1 | 187.1 | 271.2 | 0.90 | 39.3 | 225.6 | 274.4 | 0.146 | 56.1 |
| P5T3A2 | 181.5 | 272.7 | 0.99 | 37.3 | 215.4 | 239.1 | 0.145 | 61.1 |
| P5T3A3 | 186.4 | 269.0 | 0.83 | 38.9 | 206.2 | 229.5 | 0.164 | 61.3 |
| Sample | Uncrosslinked | Crosslinked | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TTI (s) | PHRR (kW/m2) | TTPHRR (s) | THReff (MJ/m2·g) | FIGRA (kW/s·m2) | WR (%) | TTI (s) | PHRR (kW/m2) | TTPHRR (s) | THReff (MJ/m2·g) | FIGRA (kW/s·m2) | WR (%) | |
| P5 | 0 | 328.6 | 30 | 2.53 | 10.96 | 1.3 | 0 | - | - | - | - | - |
| P5 + NaOH | 0 | 287.4 | 30 | 2.08 | 10.04 | 18.4 | 0 | - | - | - | - | - |
| P5T2A1 | 0 | 210.2 | 24 | 2.39 | 8.76 | 10.8 | 16 | 141.3 | 48 | 2.25 | 2.94 | 39.1 |
| P5T2A2 | 0 | 186.6 | 24 | 2.12 | 7.78 | 12.9 | 8 | 132.7 | 33 | 1.81 | 4.02 | 46.3 |
| P5T2A3 | 1 | 192.8 | 24 | 1.99 | 8.03 | 14.4 | 5 | 110.1 | 55 | 1.70 | 2.00 | 38.1 |
| P5T3A1 | 0 | 128.4 | 24 | 2.10 | 5.35 | 13.0 | 6 | 93.3 | 33 | 1.83 | 2.83 | 40.4 |
| P5T3A2 | 0 | 119.6 | 21 | 2.09 | 5.70 | 14.5 | 5 | 68.1 | 27 | 1.51 | 2.52 | 39.3 |
| P5T3A3 | 1 | 112.5 | 24 | 2.05 | 4.69 | 16.4 | 5 | 83.7 | 30 | 1.62 | 2.79 | 37.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De la Cruz, L.G.; Abt, T.; León, N.; Wang, L.; Sánchez-Soto, M. Ice-Template Crosslinked PVA Aerogels Modified with Tannic Acid and Sodium Alginate. Gels 2022, 8, 419. https://doi.org/10.3390/gels8070419
De la Cruz LG, Abt T, León N, Wang L, Sánchez-Soto M. Ice-Template Crosslinked PVA Aerogels Modified with Tannic Acid and Sodium Alginate. Gels. 2022; 8(7):419. https://doi.org/10.3390/gels8070419
Chicago/Turabian StyleDe la Cruz, Lucía G., Tobias Abt, Noel León, Liang Wang, and Miguel Sánchez-Soto. 2022. "Ice-Template Crosslinked PVA Aerogels Modified with Tannic Acid and Sodium Alginate" Gels 8, no. 7: 419. https://doi.org/10.3390/gels8070419
APA StyleDe la Cruz, L. G., Abt, T., León, N., Wang, L., & Sánchez-Soto, M. (2022). Ice-Template Crosslinked PVA Aerogels Modified with Tannic Acid and Sodium Alginate. Gels, 8(7), 419. https://doi.org/10.3390/gels8070419








