Swelling and Mechanical Characterization of Polyelectrolyte Hydrogels as Potential Synthetic Cartilage Substitute Materials
Abstract
:1. Introduction
2. Results and Discussion
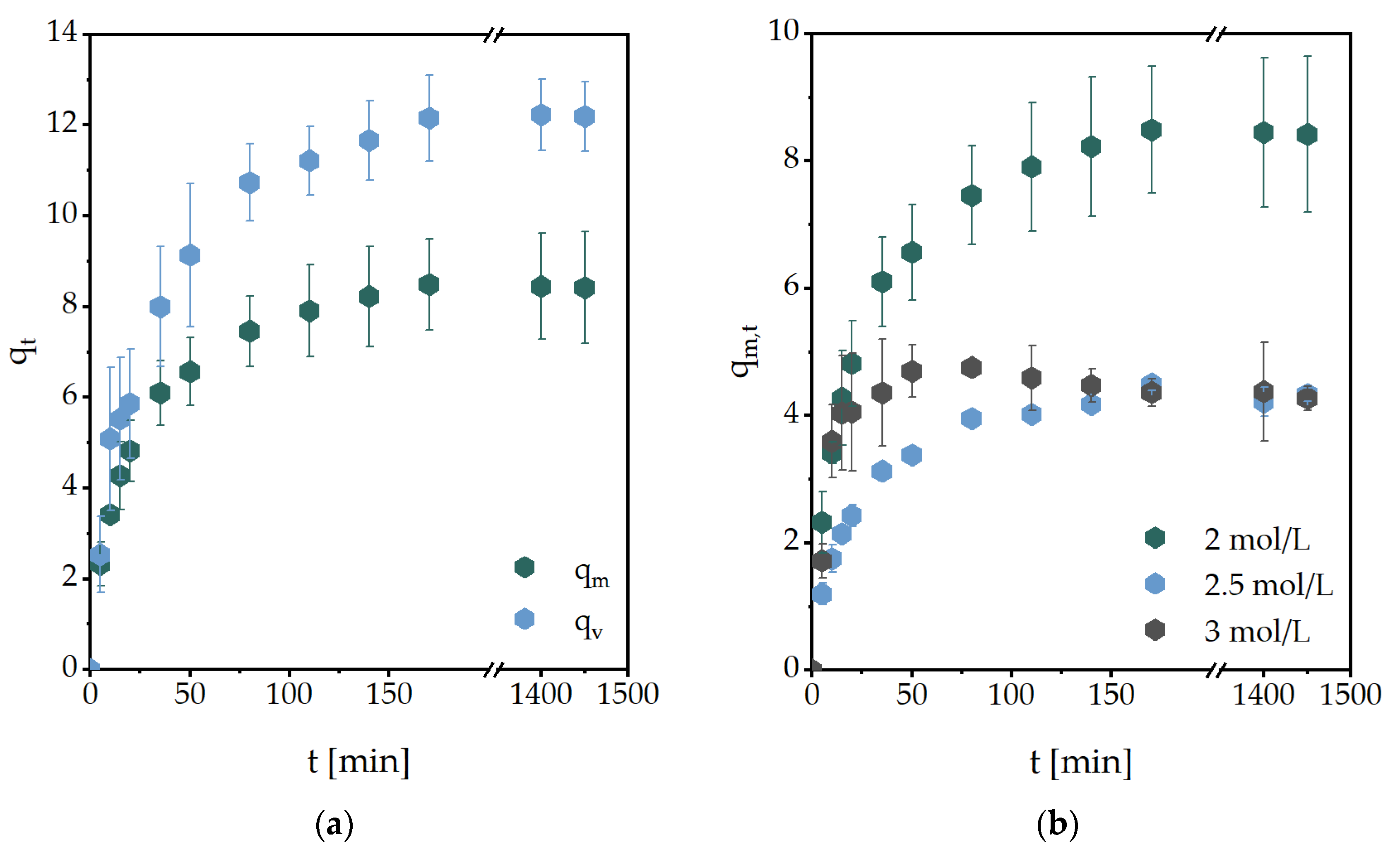
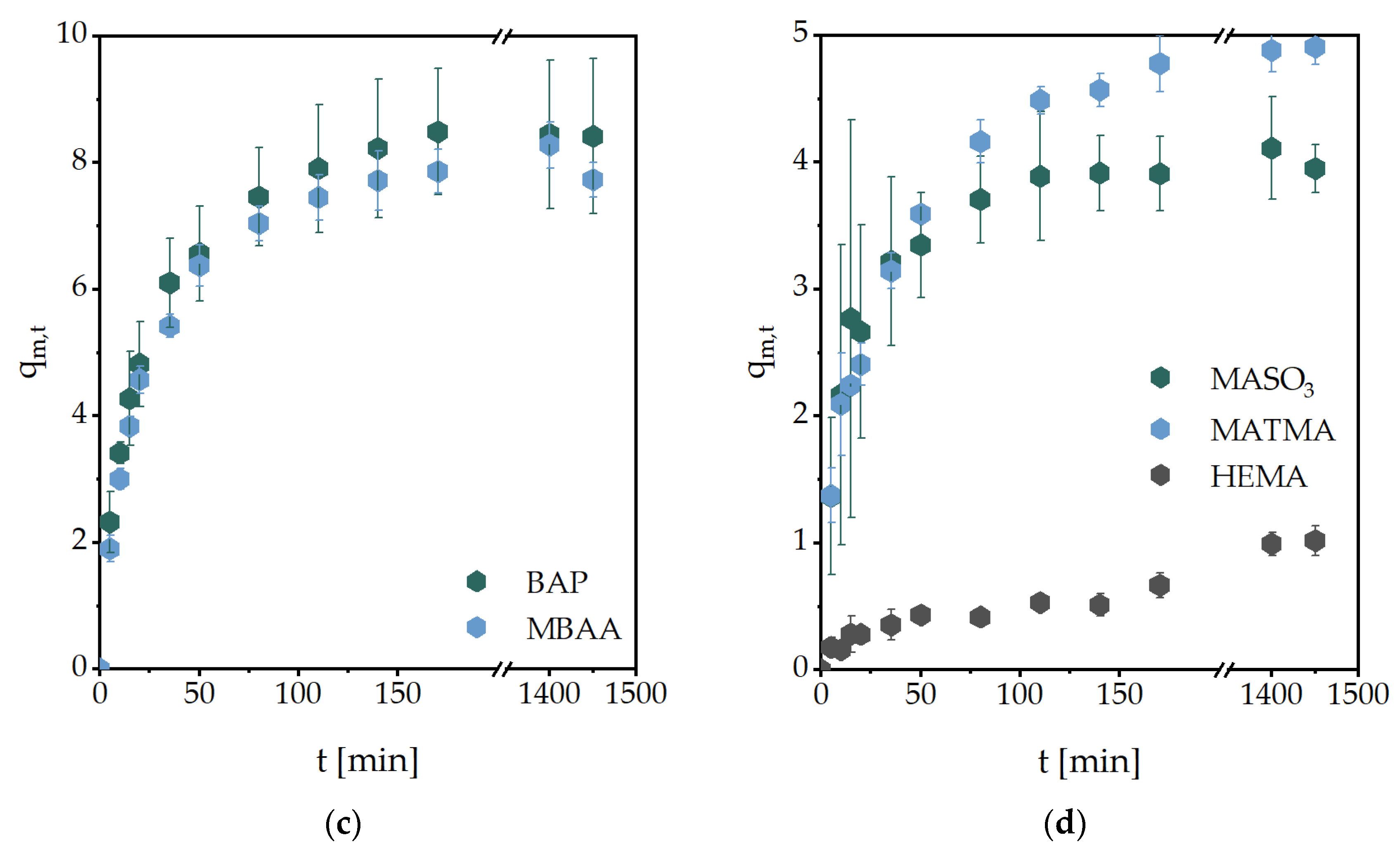
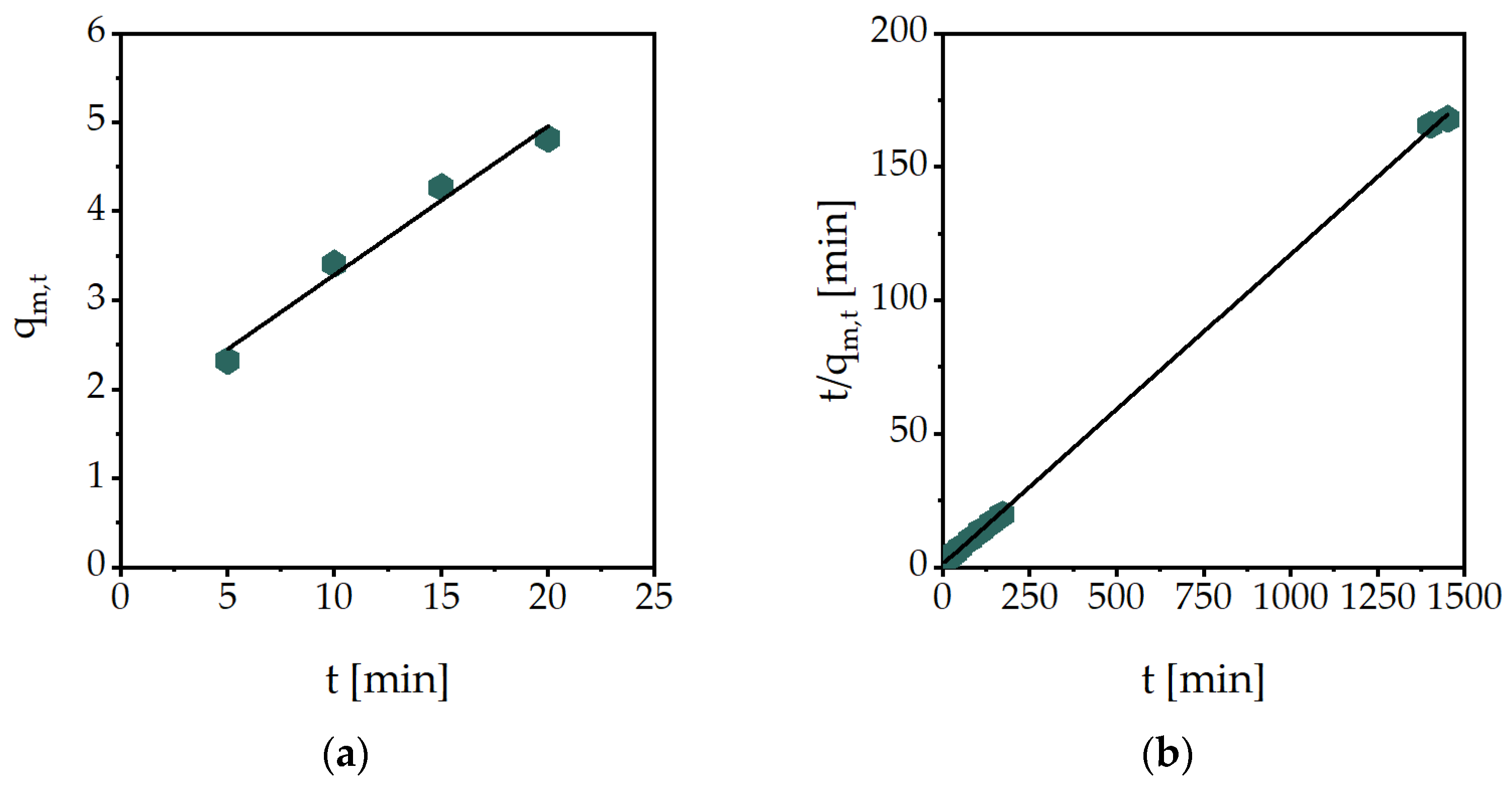
2.1. Gravimetric and Volumetric Swelling Experiments
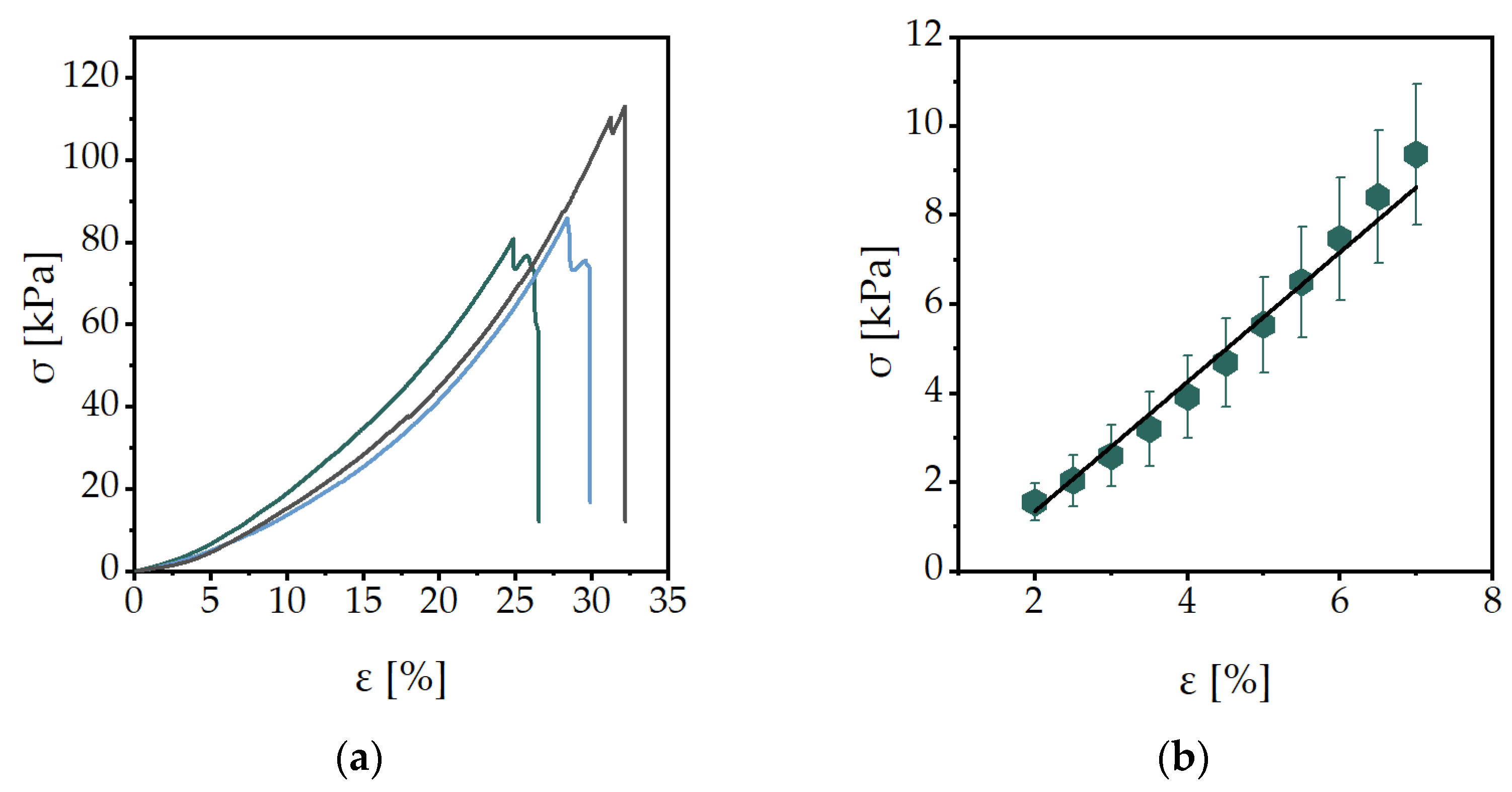
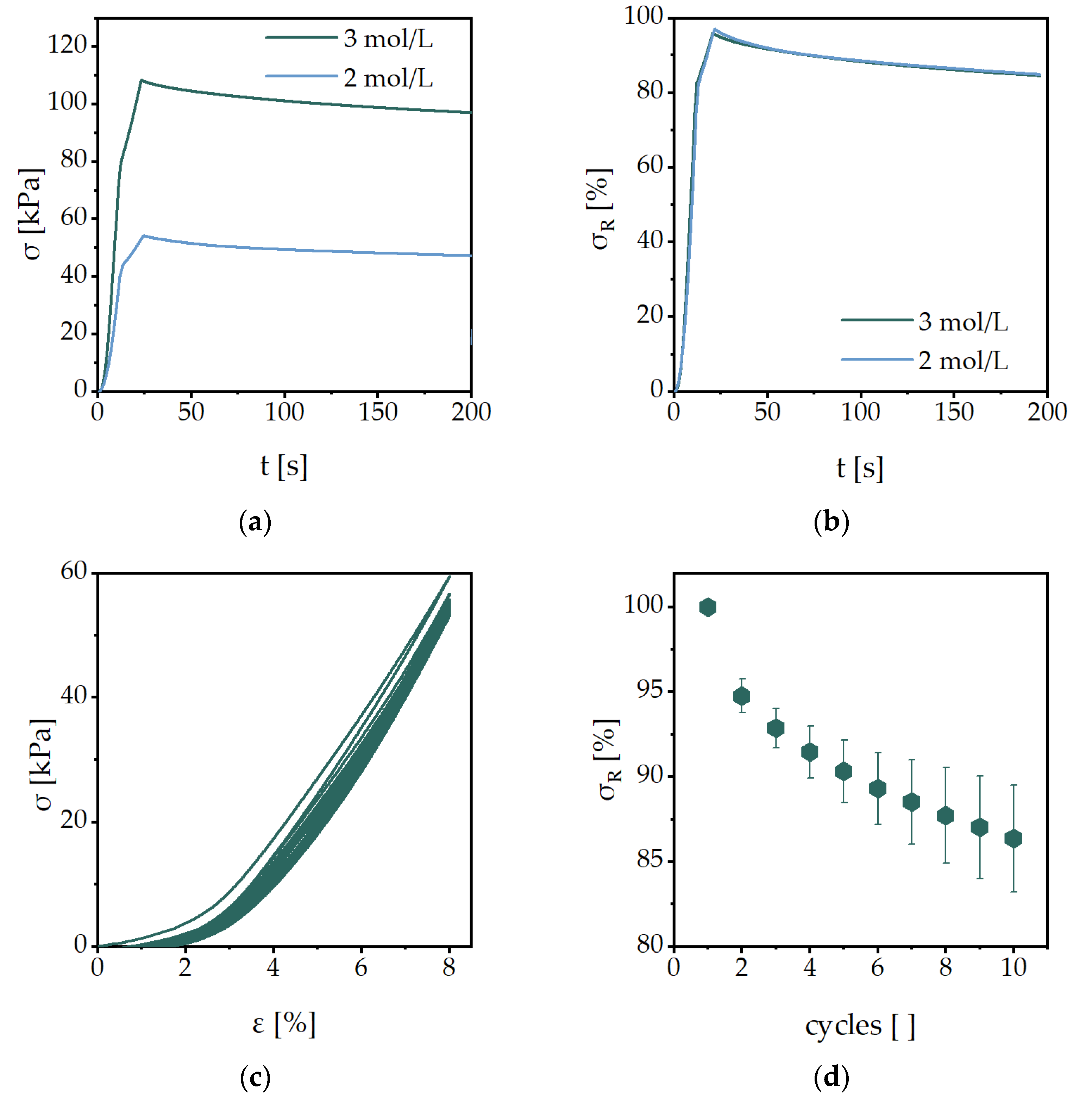
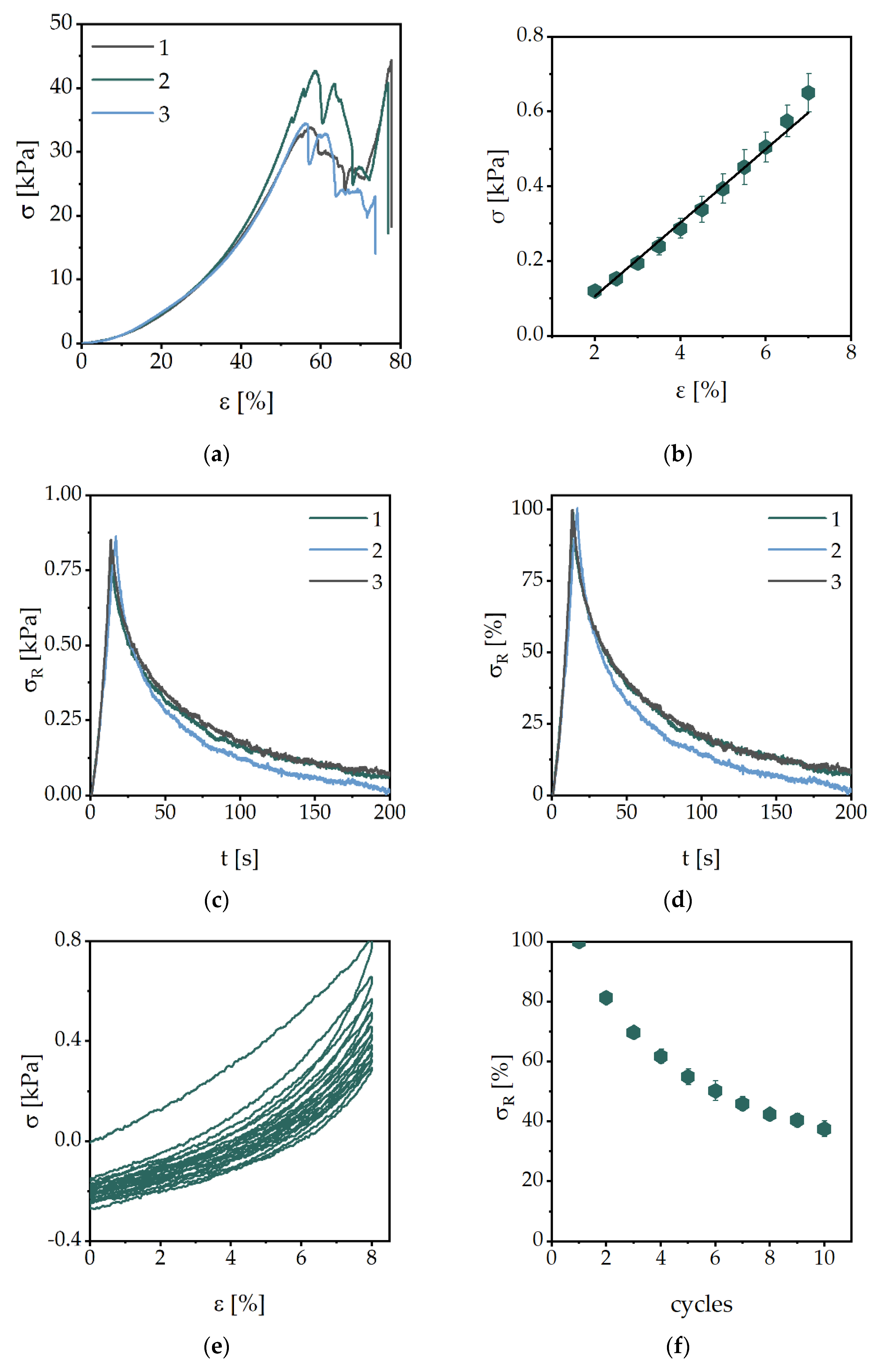
2.2. Mechanical Characterization
2.3. Comparison of a Biomaterial Used for Treatment of Cartilage Lesions
3. Conclusions
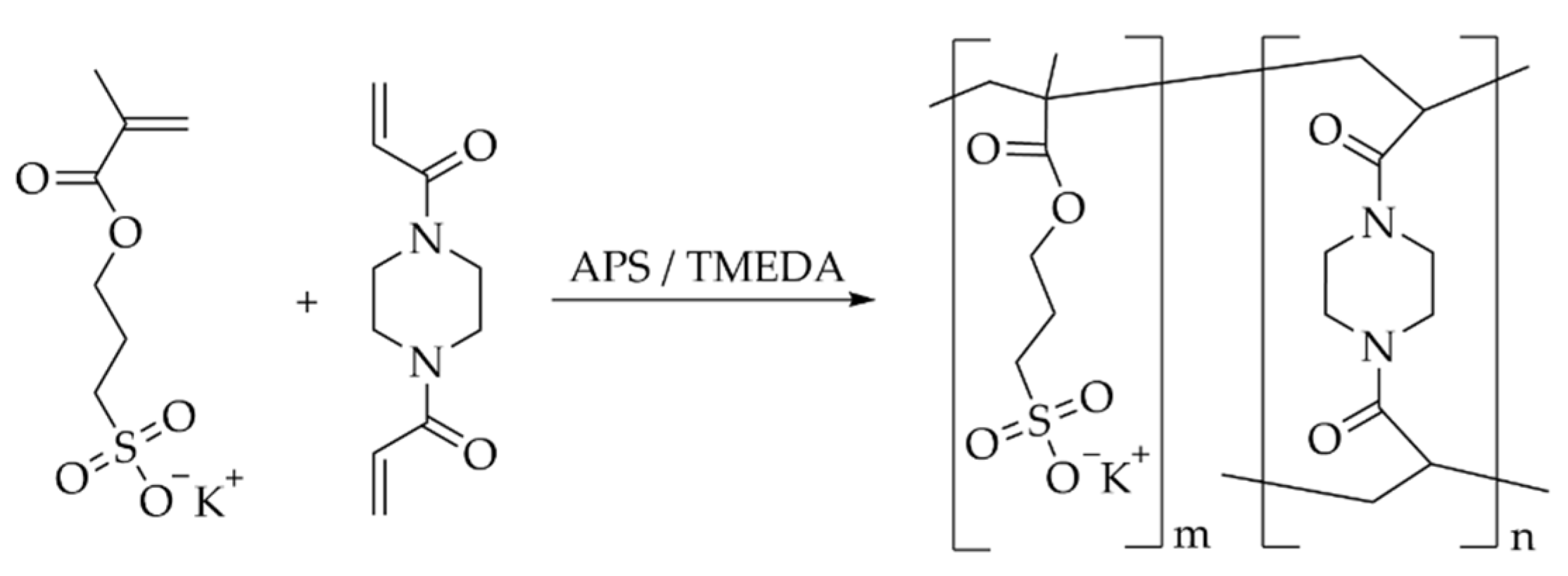
4. Materials and Methods
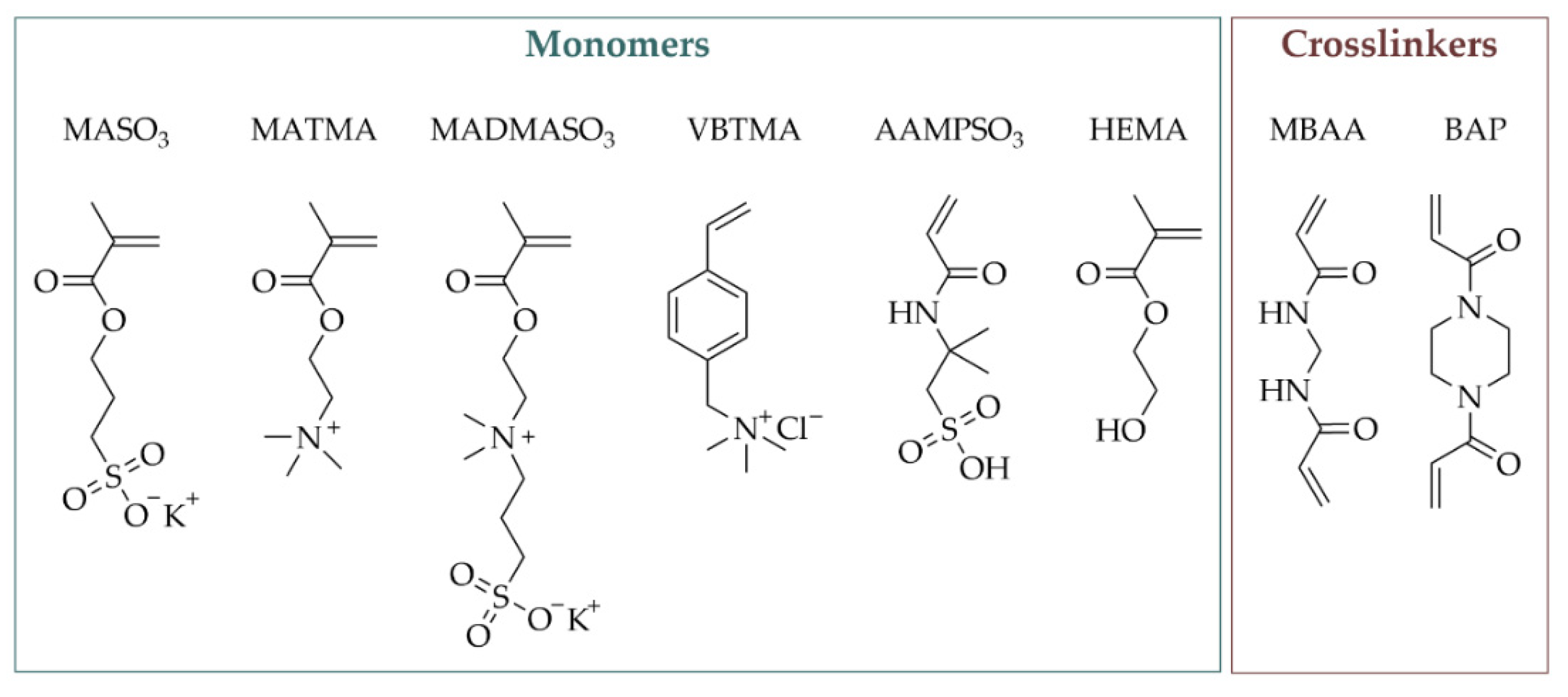
4.1. Chemicals
4.2. General Procedure for the Hydrogel Synthesis
4.3. ChondroFiller® Liquid
4.4. Gravimetric and Volumetric Swelling Experiments
4.5. Mechanical Characterization
Author Contributions
Funding
Institutional Review Board Statement
Informed Con sent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, I.L.; Mauck, R.L.; Burdick, J.A. Hydrogel Design for Cartilage Tissue Engineering: A Case Study with Hyaluronic Acid. Biomaterials 2011, 32, 8771–8782. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bobick, B.E.; Chen, F.H.; Le, A.M.; Tuan, R.S. Regulation of the Chondrogenic Phenotype in Culture. Birth Defects Res. Part C Embryo Today Rev. 2009, 87, 351–371. [Google Scholar] [CrossRef] [PubMed]
- Becerra, J.; Andrades, J.A.; Guerado, E.; Zamora-Navas, P.; López-Puertas, J.M.; Reddi, A.H. Articular Cartilage: Structure and Regeneration. Tissue Eng. Part B Rev. 2010, 16, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Söntjens, S.H.M.; Nettles, D.L.; Carnahan, M.A.; Setton, L.A.; Grinstaff, M.W. Biodendrimer-Based Hydrogel Scaffolds for Cartilage Tissue Repair. Biomacromolecules 2006, 7, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Jin, X.; Cong, Y.; Liu, Y.; Fu, J. Degradable Natural Polymer Hydrogels for Articular Cartilage Tissue Engineering. J. Chem. Technol. Biotechnol. 2013, 88, 327–339. [Google Scholar] [CrossRef]
- Liu, M.; Zeng, X.; Ma, C.; Yi, H.; Ali, Z.; Mou, X.; Li, S.; Deng, Y.; He, N. Injectable Hydrogels for Cartilage and Bone Tissue Engineering. Bone Res. 2017, 5, 17014. [Google Scholar] [CrossRef] [PubMed]
- Risbud, M.V.; Sittinger, M. Tissue Engineering: Advances in in Vitro Cartilage Generation. Trends Biotechnol. 2002, 20, 351–356. [Google Scholar] [CrossRef]
- Hjelle, K.; Solheim, E.; Strand, T.; Muri, R.; Brittberg, M. Articular Cartilage Defects in 1000 Knee Arthroscopies. Arthroscopy 2002, 18, 730–734. [Google Scholar] [CrossRef]
- Balakrishnan, B.; Banerjee, R. Biopolymer-Based Hydrogels for Cartilage Tissue Engineering. Chem. Rev. 2011, 111, 4453–4474. [Google Scholar] [CrossRef]
- Hoffmann, M.; Reuter, T.; Lange, M.; Meuche, F. Elastisches Und Hyperelastisches Modell von Gelenkknorpel-Ein Vergleich von Simulation Und Experiment. Biomed. Eng. 2010, 55, 6–9. [Google Scholar] [CrossRef]
- Pisko, E.J.; Turner, R.A.; Soderstrom, L.P.; Panetti, M.; Foster, S.L.; Treadway, W.J. Inhibition of Neutrophil Phagocytosis and Enzyme Release by Hyaluronic Acid. Clin. Exp. Rheumatol. 1983, 1, 41–44. [Google Scholar] [PubMed]
- Forrester, J.V.; Balazs, E.A. Inhibition of Phagocytosis by High Molecular Weight Hyaluronate. Immunology 1980, 40, 435–446. [Google Scholar] [PubMed]
- Nguyen, Q.T.; Hwang, Y.; Chen, A.C.; Varghese, S.; Sah, R.L. Cartilage-like Mechanical Properties of Poly (Ethylene Glycol)-Diacrylate Hydrogels. Biomaterials 2012, 33, 6682–6690. [Google Scholar] [CrossRef] [Green Version]
- Roberts, J.J.; Bryant, S.J. Comparison of Photopolymerizable Thiol-Ene PEG and Acrylate-Based PEG Hydrogels for Cartilage Development. Biomaterials 2013, 34, 9969–9979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonakdar, S.; Emami, S.H.; Shokrgozar, M.A.; Farhadi, A.; Ahmadi, S.A.H.; Amanzadeh, A. Preparation and Characterization of Polyvinyl Alcohol Hydrogels Crosslinked by Biodegradable Polyurethane for Tissue Engineering of Cartilage. Mater. Sci. Eng. C 2010, 30, 636–643. [Google Scholar] [CrossRef]
- Yang, F.; Zhao, J.; Koshut, W.J.; Watt, J.; Riboh, J.C.; Gall, K.; Wiley, B.J. A Synthetic Hydrogel Composite with the Mechanical Behavior and Durability of Cartilage. Adv. Funct. Mater. 2020, 30, 2003451. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Wu, C.; Pei, X.; Cong, Y.; Zhang, R.; Fu, J. Antibacterial Zwitterionic Polyelectrolyte Hydrogel Adhesives with Adhesion Strength Mediated by Electrostatic Mismatch. ACS Appl. Mater. Interfaces 2020, 12, 46816–46826. [Google Scholar] [CrossRef]
- Leong, M.F.; Toh, J.K.C.; Du, C.; Narayanan, K.; Lu, H.F.; Lim, T.C.; Wan, A.C.A.; Ying, J.Y. Patterned Prevascularised Tissue Constructs by Assembly of Polyelectrolyte Hydrogel Fibres. Nat. Commun. 2013, 4, 2353. [Google Scholar] [CrossRef] [Green Version]
- Claus, J.; Jastram, A.; Piktel, E.; Bucki, R.; Janmey, P.A.; Kragl, U. Polymerized Ionic Liquids-Based Hydrogels with Intrinsic Antibacterial Activity: Modern Weapons against Antibiotic-Resistant Infections. J. Appl. Polym. Sci. 2021, 138, 50222. [Google Scholar] [CrossRef]
- Jastram, A.; Lindner, T.; Luebbert, C.; Sadowski, G.; Kragl, U. Swelling and Diffusion in Polymerized Ionic Liquids-Based Hydrogels. Polymers 2021, 13, 1834. [Google Scholar] [CrossRef]
- Jastram, A.; Claus, J.; Janmey, P.A.; Kragl, U. Rheological Properties of Hydrogels Based on Ionic Liquids. Polym. Test. 2021, 93, 106943. [Google Scholar] [CrossRef]
- Weizel, A.; Distler, T.; Schneidereit, D.; Friedrich, O.; Bräuer, L.; Paulsen, F.; Detsch, R.; Boccaccini, A.R.; Budday, S.; Seitz, H. Complex Mechanical Behavior of Human Articular Cartilage and Hydrogels for Cartilage Repair. Acta Biomater. 2020, 118, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Korhonen, R.K.; Laasanen, M.S.; As, J.T.O.; Rieppo, J.; Hirvonen, J.; Helminen, H.J.; Jurvelin, J.S. Comparison of the Equilibrium Response of Articular Cartilage in Unconfined Compression, Confined Compression and Indentation. J. Biomech. 2002, 35, 903–909. [Google Scholar] [CrossRef]
- Robinson, D.L.; Kersh, M.E.; Walsh, N.C.; Ackland, D.C.; de Steiger, R.N.; Pandy, M.G. Mechanical Properties of Normal and Osteoarthritic Human Articular Cartilage. J. Mech. Behav. Biomed. Mater. 2016, 61, 96–109. [Google Scholar] [CrossRef] [Green Version]
- Kiviranta, P.; Lammentausta, E.; Töyräs, J.; Kiviranta, I.; Jurvelin, J.S. Indentation Diagnostics of Cartilage Degeneration. Osteoarthr. Cartil. 2008, 16, 796–804. [Google Scholar] [CrossRef] [Green Version]
- Li, S.N.; Li, B.; Gong, L.X.; Yu, Z.R.; Feng, Y.; Jia, D.; Zhou, Y.; Tang, L.C. Enhanced Mechanical Properties of Polyacrylamide/Chitosan Hydrogels by Tuning the Molecular Structure of Hyperbranched Polysiloxane. Mater. Des. 2019, 162, 162–170. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Tavakolizadeh, M.; Hosseini, S.H.; Rabiee, N.; Bagherzadeh, M. Highly Stretchable, Self-Adhesive, and Self-Healable Double Network Hydrogel Based on Alginate/Polyacrylamide with Tunable Mechanical Properties. J. Polym. Sci. 2020, 58, 2062–2073. [Google Scholar] [CrossRef]
- Voronova, M.I.; Surov, O.V.; Afineevskii, A.V.; Zakharov, A.G. Properties of Polyacrylamide Composites Reinforced by Cellulose Nanocrystals. Heliyon 2020, 6, e05529. [Google Scholar] [CrossRef]
- Kanca, Y.; Avcioglu, E.; Hopkins, L.; Bilge, E. Mechanical and Tribological Analysis of Polyacrylamide/Alginate Hybrid Hydrogels for Potential Cartilage Treatment. Afyon Kocatepe Univ. J. Sci. Eng. 2022, 22, 252–261. [Google Scholar] [CrossRef]
- Claus, J.; Eickner, T.; Grabow, N.; Kragl, U.; Oschatz, S. Ion Exchange Controlled Drug Release from Polymerized Ionic Liquids. Macromol. Biosci. 2020, 20, 2000152. [Google Scholar] [CrossRef]
- Claus, J.; Brietzke, A.; Lehnert, C.; Oschatz, S.; Grabow, N.; Kragl, U. Swelling Characteristics and Biocompatibility of Ionic Liquid Based Hydrogels for Biomedical Applications. PLoS ONE 2020, 15, e0231421. [Google Scholar] [CrossRef] [PubMed]
- Waletzko-Hellwig, J.; Saemann, M.; Schulze, M.; Frerich, B.; Bader, R.; Dau, M. Mechanical Characterization of Human Trabecular and Formed Granulate Bone Cylinders Processed by High Hydrostatic Pressure. Materials 2021, 14, 1069. [Google Scholar] [CrossRef] [PubMed]

| Monomer | Crosslinker | cM (mol/L) | cCL (mol%) | qm,∞ | Initial Swelling Rate (min−1) | kS (10−2) | EWC |
|---|---|---|---|---|---|---|---|
| MASO3 | BAP | 2 | 2 | 7.7 ± 0.3 | 0.167 | 1.23 | 0.89 ± 0.01 |
| 2.5 | 4.3 ± 0.1 | 0.082 | 2.48 | 0.81 ± 0.01 | |||
| 3 | 4.9 ± 0.2 | 0.150 | 3.43 | 0.83 ± 0.01 | |||
| MBAA | 2 | 2 | 7.7 ± 0.3 | 0.177 | 1.05 | 0.89 ± 0.01 | |
| 2.5 | 4.4 ± 1.3 | 0.113 | 3.17 | 0.81 ± 0.05 | |||
| 3 | 4.0 ± 0.2 | 0.090 | 4.24 | 0.80 ± 0.01 | |||
| MATMA | BAP | 3 | 2 | 4.9 ± 0.1 | 0.102 | 3.38 | 0.45 ± 0.02 |
| MBAA | 3 | 2 | 5.4 ± 0.4 | 0.065 | 1.24 | 0.84 ± 0.01 | |
| HEMA | BAP | 3 | 2 | 1.0 ± 0.1 | * | 1.28 | 0.50 ± 0.03 |
| MBAA | 3 | 2 | 0.9 ± 0.1 | * | 1.51 | 0.51 ± 0.03 |
| Monomer | Crosslinker | cM (mol/L) | cCL (mol%) | qm,∞ | qv,∞ |
|---|---|---|---|---|---|
| MASO3 | BAP | 2 | 2 | 7.7 ± 0.3 | 10.7 ± 0.8 |
| 2.5 | 5.0 ± 0.9 | 4.1 ± 0.2 | |||
| 3 | 5.5 ± 0.2 | 6.8 ± 0.4 | |||
| 2.5 | 2 | 4.3 ± 0.1 | * | ||
| 2.5 | 5.1 ± 0.4 | 6.1 ± 0.9 | |||
| 3 | * | * | |||
| 3 | 2 | 4.9 ± 0.2 | * | ||
| 2.5 | 4.6 ± 0.3 | 5.7 ± 0.4 | |||
| 3 | 4.7 ± 0.9 | 5.2 ± 0.9 | |||
| MBAA | 2 | 2 | 7.7 ± 0.3 | 10.7 ± 0.8 | |
| 2.5 | 4.4 ± 1.3 | * | |||
| 3 | 4.0 ± 0.2 | * | |||
| MATMA | BAP | 3 | 2 | 4.9 ± 0.1 | 5.7 ± 0.2 |
| MBAA | 3 | 2 | 5.4 ± 0.4 | 6.2 ± 0.2 | |
| HEMA | BAP | 3 | 2 | 1.0 ± 0.1 | 0.6 ± 0.1 |
| 3 | 0.9 ± 0.1 | 0.9 ± 0.1 | |||
| 4 | 3 | 0.1 ± 0.0 | 0.1 ± 0.0 | ||
| 4 | 0.1 ± 0.0 | * | |||
| MBAA | 3 | 2 | 1.0 ± 0.1 | 1.0 ± 0.3 | |
| MADMASO3 | BAP | 2 | 2 | 2.8 ± 0.1 | 3.2 ± 0.2 |
| AAMPSO3 | BAP | 2 | 2 | 2.1 ± 0.1 | 2.4 ± 0.4 |
| 2.5 | 1.8 ± 0.6 | 1.8 ± 0.5 | |||
| 3 | 1.7 ± 0.1 | 1.5 ± 0.2 | |||
| 2.5 | 2 | 2.5 ± 0.1 | 3.0 ± 0.2 | ||
| 2.5 | 1.8 ± 0.2 | 2.2 ± 0.2 | |||
| 3 | 1.7 ± 0.1 | * | |||
| 3 | 2 | * | * | ||
| 2.5 | 2.2 ± 0.1 | * | |||
| 3 | * | * | |||
| VBTMA | BAP | 2 | 2 | 7.2 ± 0.3 | 7.3 ± 1.5 |
| 2.5 | 2.9 ± 0.4 | 2.3 ± 0.3 | |||
| 3 | 4.2 ± 0.3 | 2.5 ± 0.2 | |||
| 2.5 | 2 | 2.7 ± 0.2 | 2.5 ± 0.4 | ||
| 2.5 | 4.8 ± 0.9 | 2.7 ± 0.1 | |||
| 3 | 7.3 ± 0.7 | 5.9 ± 0.2 | |||
| 3 | 2 | 5.5 ± 0.4 | 6.7 ± 0.4 | ||
| 2.5 | 4.2 ± 0.3 | 4.3 ± 0.1 | |||
| 3 | 5.6 ± 0.7 | 4.2 ± 0.1 |
| cM (mol/L) | cCL (mol%) | Compressive Modulus [kPa] | ||
|---|---|---|---|---|
| MASO3 | AAMPSO3 | VBTMA | ||
| 2 | 2 | 146.3 ± 9.4 | 537.2 ± 27.1 | 2.5 ± 0.1 |
| 2.5 | * | 1230.2 ± 48.3 | 98.1 ± 13.7 | |
| 3 | * | 995.4 ± 83.0 | 129.5 ± 14.5 | |
| 2.5 | 2 | 430.6 ± 20.1 | 569.2 ± 15.6 | 84.9 ± 6.0 |
| 2.5 | * | 1108.6 ± 80.1 | 26.5 ± 1.5 | |
| 3 | * | 1637.4 ± 164.7 | 22.7 ± 1.6 | |
| 3 | 2 | 353.9 ± 26.2 | 841.3 ± 78.8 | 92.3 ± 3.3 |
| 2.5 | * | 1708.7 ± 67.7 | 107.2 ± 7.3 | |
| 3 | * | * | 126.5 ± 8.1 | |
| Monomer | cM (mol/L) | cCL (mol%) | Stress-Relaxation Residual Stress σR [%] | Residual Stress σR of Each Cycle (%) | ||
|---|---|---|---|---|---|---|
| 2 | 5 | 10 | ||||
| AAMPSO3 | 2 | 2 | 85 ± 2 | 95 ± 1 | 90 ± 2 | 86 ± 3 |
| 2.5 | 89 ± 1 | 98 ± 1 | 95 ± 1 | 93 ± 1 | ||
| 3 | 87 ± 1 | 99 ± 1 | 97 ± 2 | 94 ± 2 | ||
| 2.5 | 2 | 91 ± 1 | 98 ± 1 | 95 ± 2 | 92 ± 2 | |
| 2.5 | 89 ± 1 | 98 ± 1 | 95 ± 1 | 91 ± 2 | ||
| 3 | 87 ± 1 | 97 ± 1 | 94 ± 2 | 92 ± 2 | ||
| 3 | 2 | 85 ± 7 | 97 ± 1 | 93 ± 3 | 89 ± 3 | |
| 2.5 | 81 ± 6 | 99 ± 1 | 94 ± 3 | 90 ± 3 | ||
| MASO3 | 3 | 2 | 88 ± 5 | 98 ± 2 | 96 ± 3 | 94 ± 4 |
| VBTMA | 3 | 2 | 90 ± 1 | 99 ± 1 | 98 ± 2 | 97 ± 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romischke, J.; Scherkus, A.; Saemann, M.; Krueger, S.; Bader, R.; Kragl, U.; Meyer, J. Swelling and Mechanical Characterization of Polyelectrolyte Hydrogels as Potential Synthetic Cartilage Substitute Materials. Gels 2022, 8, 296. https://doi.org/10.3390/gels8050296
Romischke J, Scherkus A, Saemann M, Krueger S, Bader R, Kragl U, Meyer J. Swelling and Mechanical Characterization of Polyelectrolyte Hydrogels as Potential Synthetic Cartilage Substitute Materials. Gels. 2022; 8(5):296. https://doi.org/10.3390/gels8050296
Chicago/Turabian StyleRomischke, Johanna, Anton Scherkus, Michael Saemann, Simone Krueger, Rainer Bader, Udo Kragl, and Johanna Meyer. 2022. "Swelling and Mechanical Characterization of Polyelectrolyte Hydrogels as Potential Synthetic Cartilage Substitute Materials" Gels 8, no. 5: 296. https://doi.org/10.3390/gels8050296
APA StyleRomischke, J., Scherkus, A., Saemann, M., Krueger, S., Bader, R., Kragl, U., & Meyer, J. (2022). Swelling and Mechanical Characterization of Polyelectrolyte Hydrogels as Potential Synthetic Cartilage Substitute Materials. Gels, 8(5), 296. https://doi.org/10.3390/gels8050296