Ultraviolet and Infrared Irradiations Sensing of Gel-Orange Dye Composite-Based Flexible Electrochemical Cells
Abstract
:1. Introduction
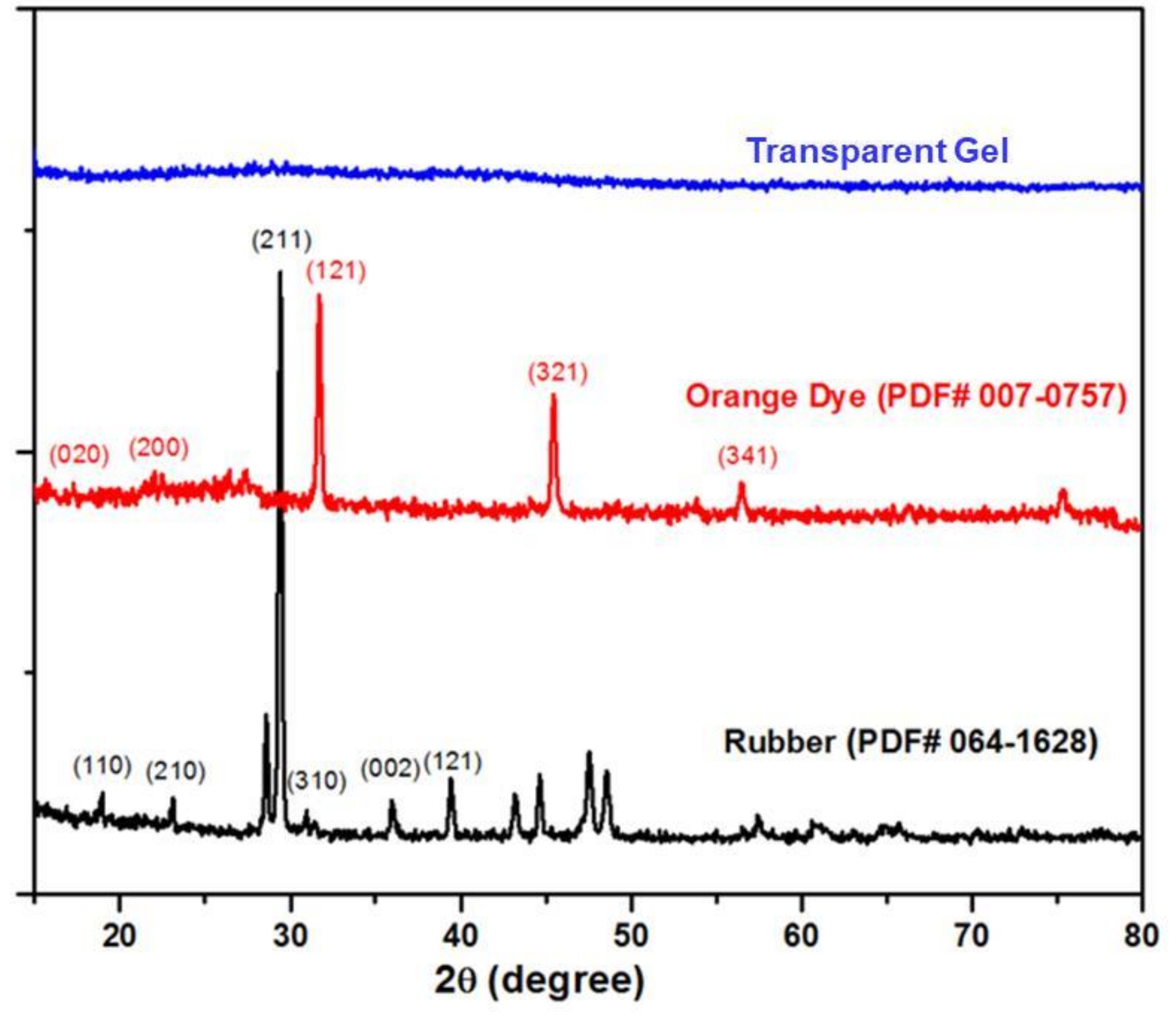
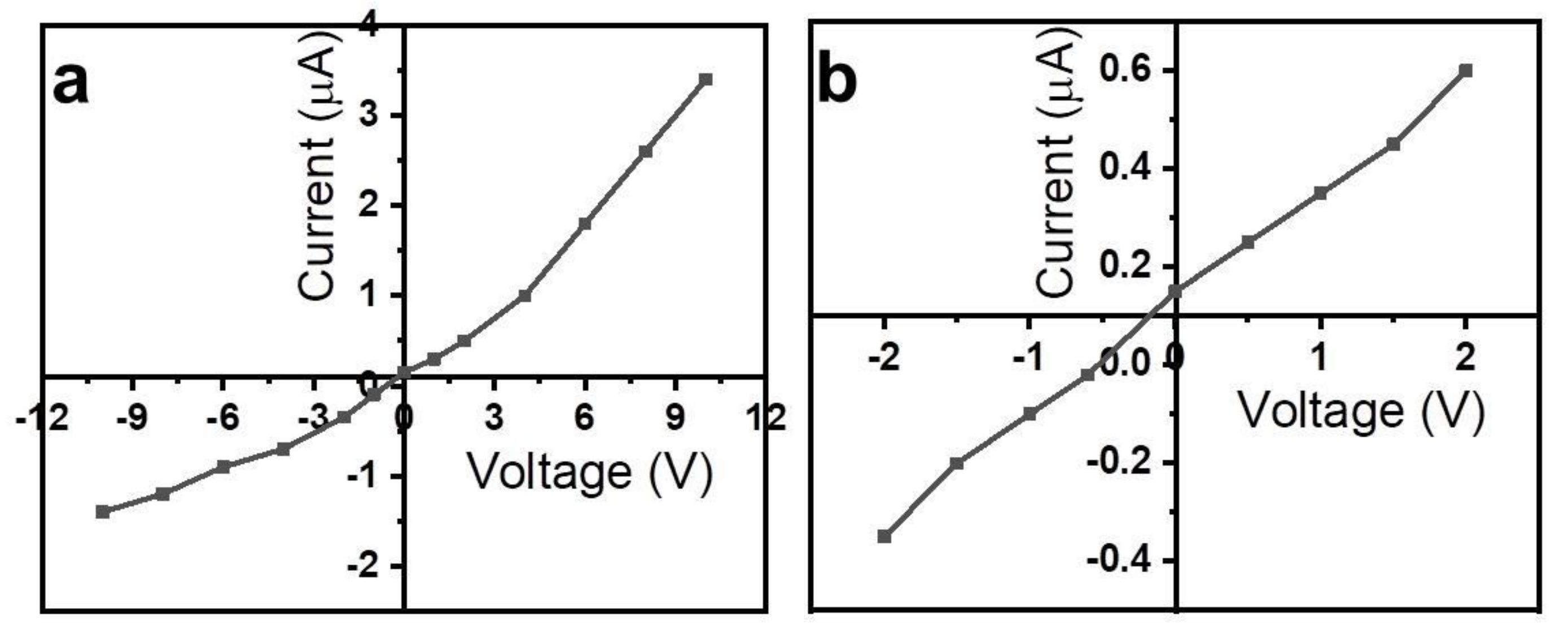
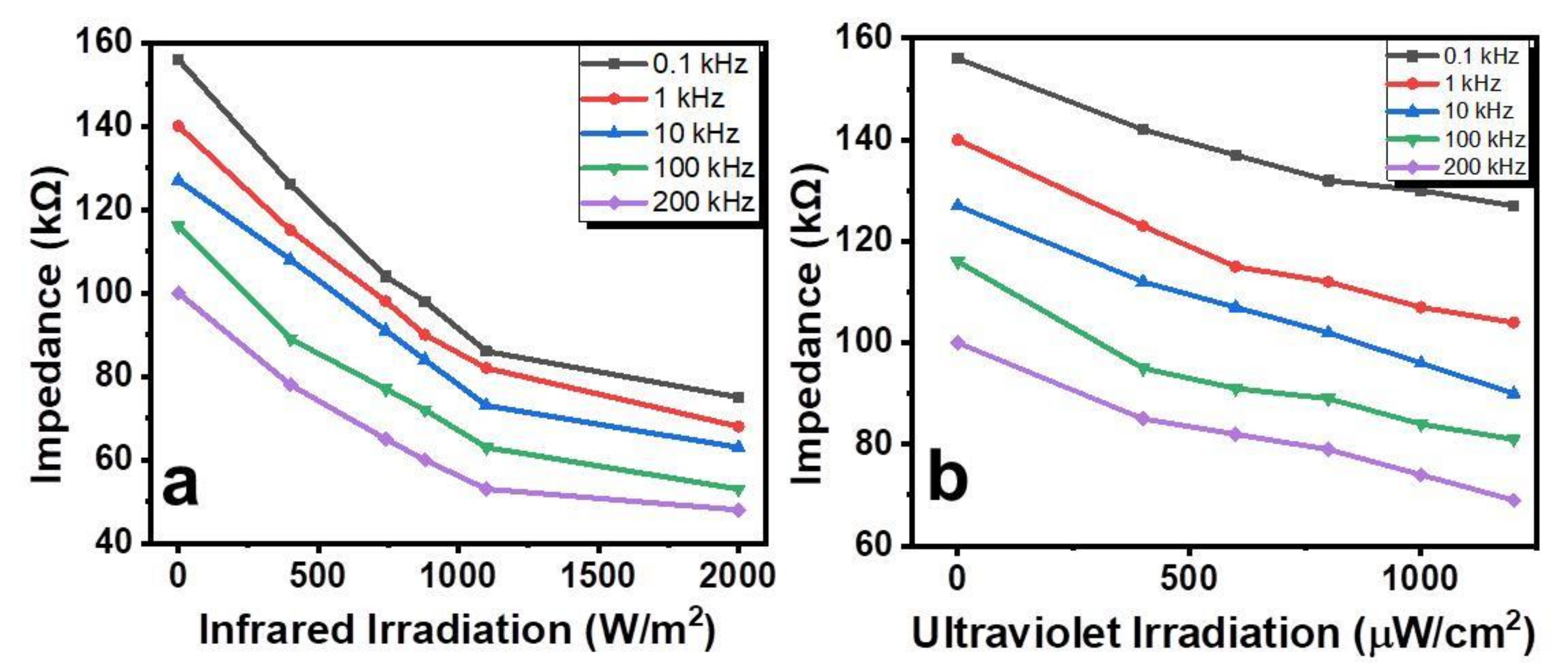
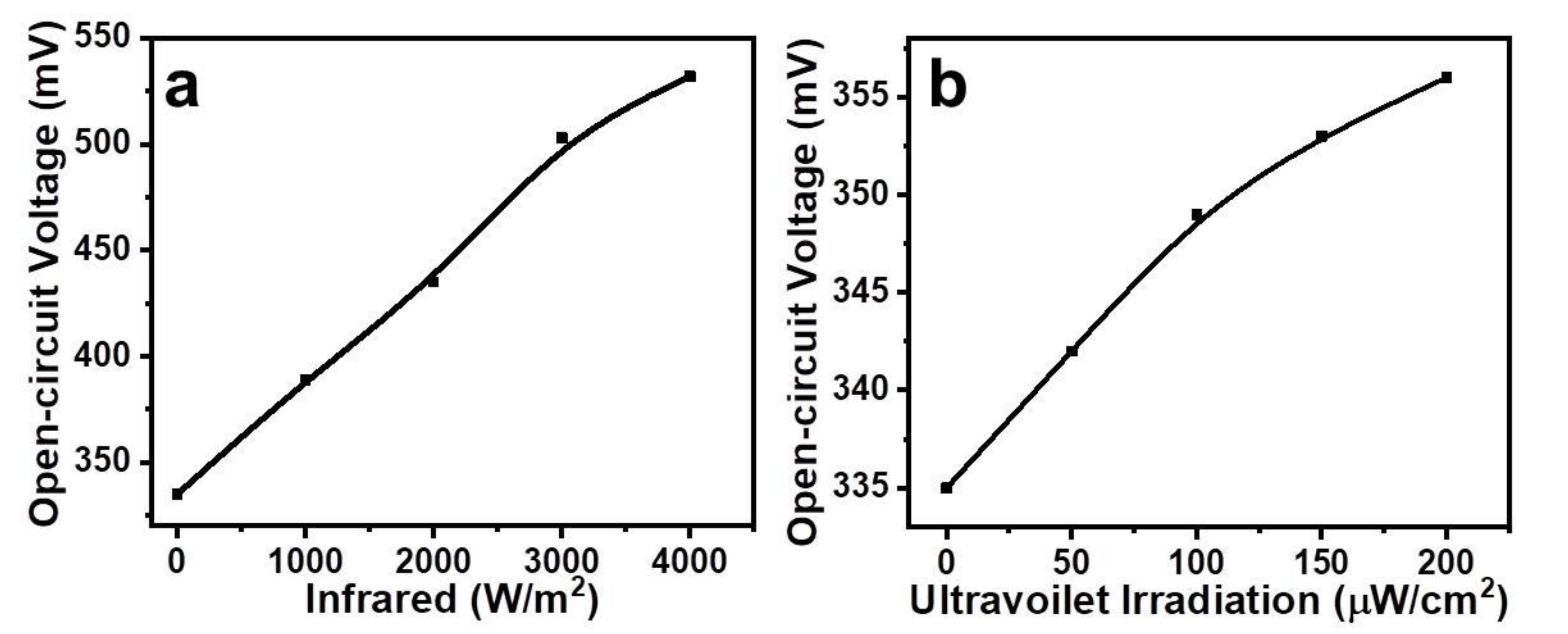
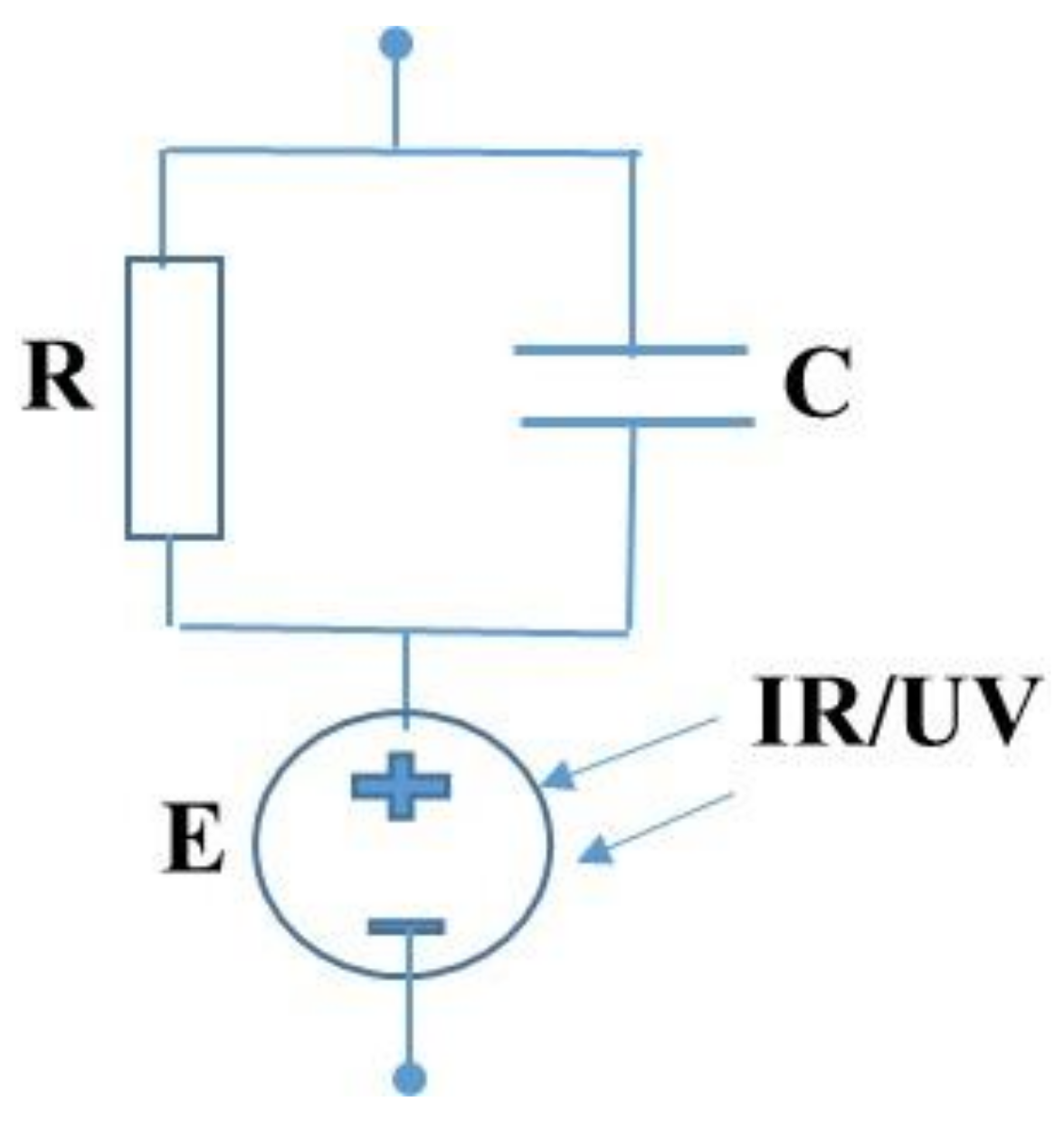
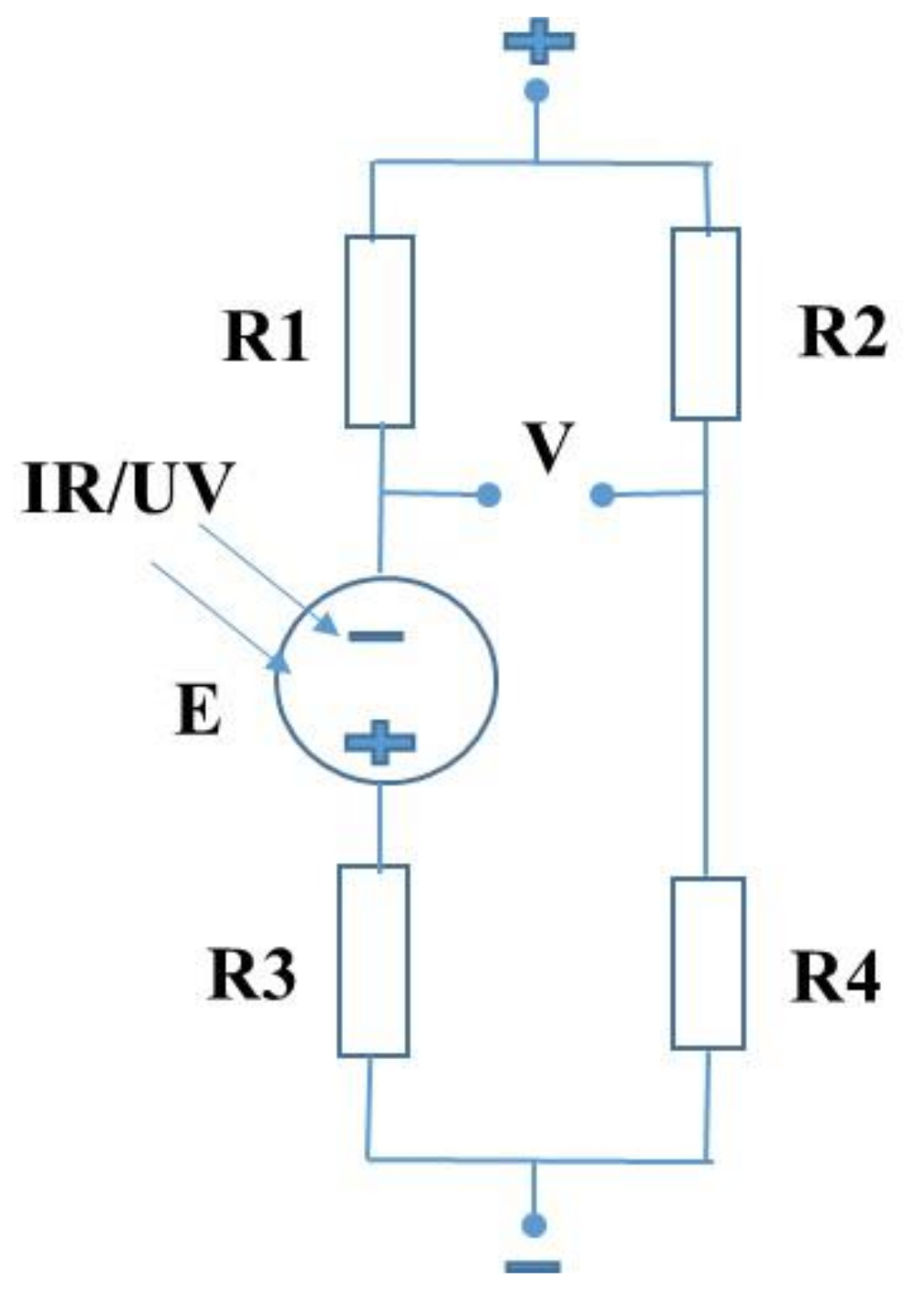
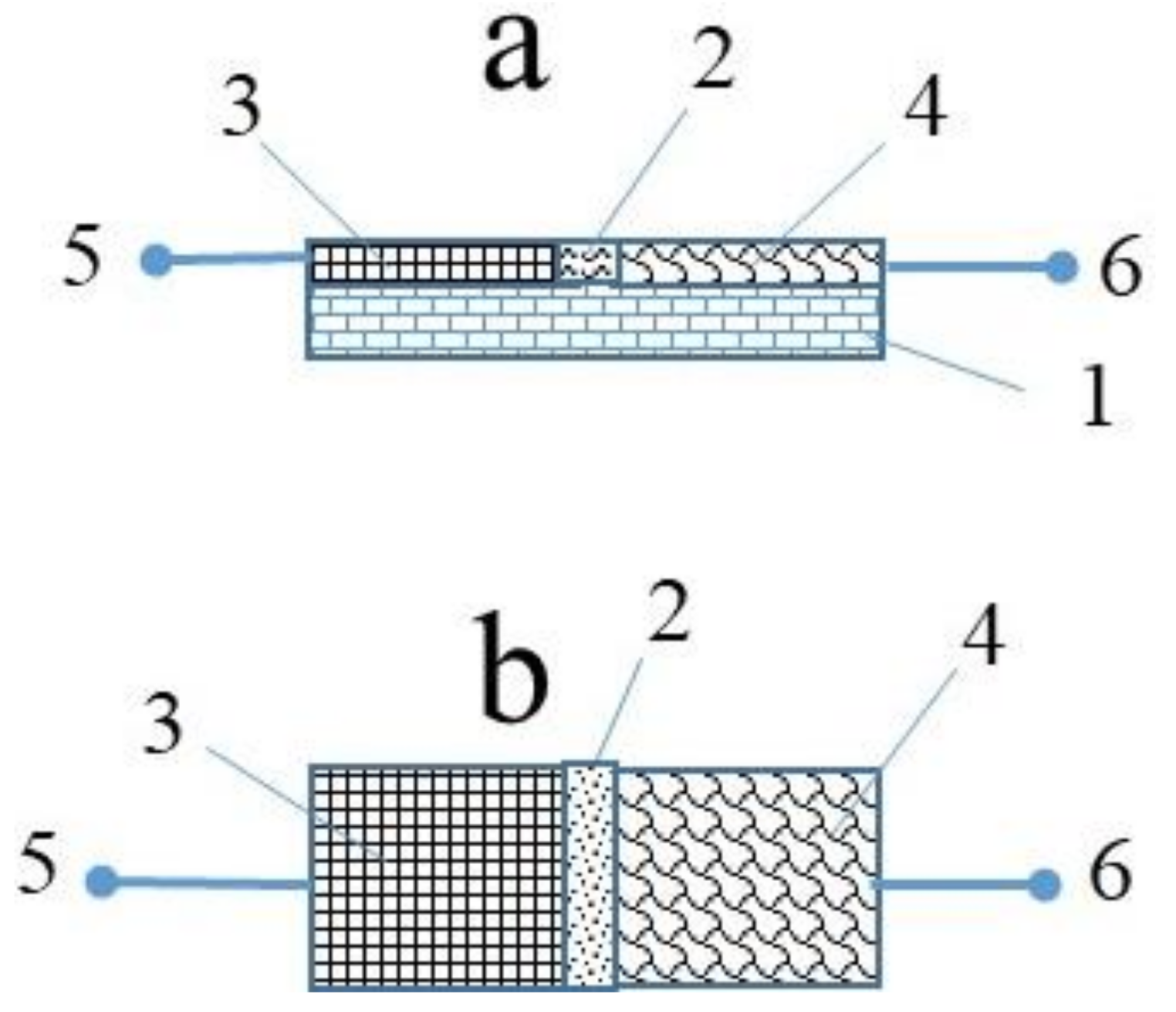
2. Results and Discussion
3. Conclusions
4. Experimental
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Karimi-Maleh, H.; Karimi, F.; Fu, L.; Sanati, A.L.; Alizadeh, M.; Karaman, C.; Orooji, Y. Cyanazine herbicide monitoring as a hazardous substance by a DNA nanostructure biosensor. J. Hazard. Mater. 2022, 423, 127058. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Maleh, H.; Khataee, A.; Karimi, F.; Baghayeri, M.; Fu, L.; Rouhi, J.; Karaman, C.; Karaman, O.; Boukherroub, R. A green and sensitive guanine-based DNA biosensor for idarubicin anticancer monitoring in biological samples: A simple and fast strategy for control of health quality in chemotherapy procedure confirmed by docking investigation. Chemosphere 2021, 132928. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.; Kumar, P.; Park, D.-S.; Shim, Y.-B. Electrochemical Sensors Based on Organic Conjugated Polymers. Sensors 2008, 8, 118–141. [Google Scholar] [CrossRef] [PubMed]
- Kus, M.; Hakli, Ö.; Zafer, C.; Varlikli, C.; Demic, S.; Özçelik, S.; Icli, S. Optical and electrochemical properties of polyether derivatives of perylenediimides adsorbed on nanocrystalline metal oxide films. Org. Electron. 2008, 9, 757–766. [Google Scholar] [CrossRef] [Green Version]
- Shao, Y.; Wang, J.; Wu, H.; Liu, J.; Aksay, I.A.; Lin, Y. Graphene Based Electrochemical Sensors and Biosensors: A Review. Electroanalysis 2010, 22, 1027–1036. [Google Scholar] [CrossRef]
- Hopkins, J.; Fidanovski, K.; Lauto, A.; Mawad, D. All-Organic Semiconductors for Electrochemical Biosensors: An Overview of Recent Progress in Material Design. Front. Bioeng. Biotechnol. 2019, 7, 237. [Google Scholar] [CrossRef] [PubMed]
- Valentine, C.J.; Takagishi, K.; Umezu, S.; Daly, R.; De Volder, M. Paper-Based Electrochemical Sensors Using Paper as a Scaffold to Create Porous Carbon Nanotube Electrodes. ACS Appl. Mater. Interfaces 2020, 12, 30680–30685. [Google Scholar] [CrossRef] [PubMed]
- White, A.M. Infrared detectors. U.S. Patents 4679063, 7 July 1987. Available online: https://patents.google.com/patent/US4679063A/en (accessed on 11 December 2021).
- Liddiard, K.C. Semiconductor Film Bolometer Thermal Infrared Detector. U.S. Patent 5,369,280, 29 November 1994. [Google Scholar]
- Mueller, M.; Cheng, J.; Pisano, A.; Cauley, T.H., III. Infrared Sensor Systems and Devices. U.S. Patent 7,547,886, 16 June 2009. [Google Scholar]
- McGill, R.A.; Kendziora, C.; Furstenberg, R.; Papantonakis, M.; Horwitz, J.S.; Hubler, G.K. Detection of Chemicals with Infrared Light. U.S. Patent 8,101,915, 24 January 2012. [Google Scholar]
- Sahatiya, P.; Puttapati, S.K.; Srikanth, V.V.; Badhulika, S. Graphene-based wearable temperature sensor and infrared photodetector on a flexible polyimide substrate. Flex. Print. Electron. 2016, 1, 025006. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, X.; Hu, W. Organic photodiodes and phototransistors toward infrared detection: Materials, devices, and applications. Chem. Soc. Rev. 2020, 49, 653–670. [Google Scholar] [CrossRef] [PubMed]
- Ranjith, K.S.; Kumar, R.T.R. Facile construction of vertically aligned ZnO nanorod/PEDOT:PSS hybrid heterojunction-based ultraviolet light sensors: Efficient performance and mechanism. Nanotechnology 2016, 27, 95304. [Google Scholar] [CrossRef] [PubMed]
- Memisoglu, G.; Varlikli, C. Highly Efficient Organic UV Photodetectors Based on Polyfluorene and Naphthalenediimide Blends: Effect of Thermal Annealing. Int. J. Photoenergy 2012, 2012, 936075. [Google Scholar] [CrossRef] [Green Version]
- Patel, J.; Tiwana, P.; Seidler, N.; Morse, G.E.; Lozman, O.R.; Johnston, M.B.; Herz, L.M. Effect of Ultraviolet Radiation on Organic Photovoltaic Materials and Devices. ACS Appl. Mater. Interfaces 2019, 11, 21543–21551. [Google Scholar] [CrossRef] [PubMed]
- Mantese, S.-C.C.V. Ferroelectric Infrared Sensors and Methods for Their Manufacturing. European Patent 1956658A3, 23 March 2011. [Google Scholar]
- Mariello, M.; Qualtieri, A.; Mele, G.; De Vittorio, M. Metal-Free Multilayer Hybrid PENG Based on Soft Electrospun/-Sprayed Membranes with Cardanol Additive for Harvesting Energy from Surgical Face Masks. ACS Appl. Mater. Interfaces 2021, 13, 20606–20621. [Google Scholar] [CrossRef] [PubMed]
- Mader, G.; Meixner, H. Pyroelectric infrared sensor arrays based on the polymer PVDF. Sens. Actuators A Phys. 1990, 22, 503–507. [Google Scholar] [CrossRef]
- Levell, J.W.; Giardini, M.E.; Samuel, I.D.W. A hybrid organic semiconductor/silicon photodiode for efficient ultraviolet photodetection. Opt. Express 2010, 18, 3219–3225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, I.J.; Ahn, I.H.; Bae, S.H. Display with Infrared Backlight Source and Multi-Touch Sensing Function. U.S. Patent 8,350,827, 1 August 2013. [Google Scholar]
- Zou, Y.; Zhang, Y.; Hu, Y.; Gu, H. Ultraviolet Detectors Based on Wide Bandgap Semiconductor Nanowire: A Review. Sensors 2018, 18, 2072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazzillo, M.C.; Sciuto, A. Semiconductor Device for Detecting Ultraviolet and Infrared Radiation and Related Manufacturing Process. U.S. Patents 9,952,094, 24 April 2018. [Google Scholar]
- Vogt, H. Bolometer Having an Organic Semiconductor Layer Arrangement. European Patent 1994384B1, 27 January 2010. [Google Scholar]
- Ginn, R.P.; Gerber, K.A.; Hampp, A.; Childs, A.C. Infrared Photodiodes and Sensor Arrays with Improved Passivation Layers and Methods of Manufacture. U.S. Patent 7,544,532, 6 September 2009. [Google Scholar]
- Kropelnicki, P.; Russ, M.; Vogt, H. Diode Bolometer and Method for Producing a Diode Bolometer. U.S. Patent 8,569,857, 29 October 2013. [Google Scholar]
- Qasuria, T.A.; Fatima, N.; Karimov, K.S.; Ibrahim, M.A. A novel and stable ultraviolet and infrared intensity sensor in impedance/capacitance modes fabricated from degraded CH3NH3PbI3-xClx perovskite materials. J. Mater. Res. Technol. 2020, 9, 12795–12803. [Google Scholar] [CrossRef]
- Cheng, J.; Poduska, K.M. Frequency-Dependent Impedance Responses of ZnO Using UV Light. ECS J. Solid State Sci. Technol. 2019, 8, N1–N4. [Google Scholar] [CrossRef]
- Li, X.; Liang, J.; Zhang, H.; Yang, X.; Zhang, H.; Pang, W.; Zhang, M. Resonant and resistive dual-mode uncooled infrared detectors toward expanded dynamic range and high linearity. Appl. Phys. Lett. 2017, 110, 263502. [Google Scholar] [CrossRef]


| Sr. No | Sensor Type | Materials Used | Fabrication Technique | Sensing Range | Impedance/Resistance Change | Ref. |
|---|---|---|---|---|---|---|
| 1 | UV | ITO/PEDOT: PSS/PFE:BNDI | Spin coating | 0–1 mW/cm2 | 61.8 to 19.5 kΩ | [15] |
| 2 | UV | CH3NH3PbI3-xClx perovskite | Spin coating | 0–200 W/m2 | 7.4 to 2.0 MΩ | [27] |
| 3 | UV | ZnO | Hand spread | 0–1.6 mW/cm2 | 2.6 to 0.7 MΩ | [28] |
| 4 | UV | gel–orange dye | Rubbing-in | 0–1200 μ W/cm2 | 127 to 90 kΩ | Present work |
| 5 | IR | CH3NH3PbI3-xClx perovskite | Spin coating | 0–6000 W/m2 | 9.0 to 5.0 kΩ | [27] |
| 6 | IR | Micromachined piezoelectric resonator | MEMS | 0–1.0 mW/mm2 | 2.7 to 1.0 GΩ | [29] |
| 7 | IR | gel–orange dye | Rubbing-in | 0–2000 W/m2 | 156 to 75 kΩ | Present work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chani, M.T.S.; Karimov, K.S.; Asiri, A.M.; Kamal, T.; Bakhsh, E.M.; Rahman, M.M. Ultraviolet and Infrared Irradiations Sensing of Gel-Orange Dye Composite-Based Flexible Electrochemical Cells. Gels 2022, 8, 83. https://doi.org/10.3390/gels8020083
Chani MTS, Karimov KS, Asiri AM, Kamal T, Bakhsh EM, Rahman MM. Ultraviolet and Infrared Irradiations Sensing of Gel-Orange Dye Composite-Based Flexible Electrochemical Cells. Gels. 2022; 8(2):83. https://doi.org/10.3390/gels8020083
Chicago/Turabian StyleChani, Muhammad Tariq Saeed, Khasan S. Karimov, Abdullah M. Asiri, Tahseen Kamal, Esraa M. Bakhsh, and Mohammed Muzibur Rahman. 2022. "Ultraviolet and Infrared Irradiations Sensing of Gel-Orange Dye Composite-Based Flexible Electrochemical Cells" Gels 8, no. 2: 83. https://doi.org/10.3390/gels8020083
APA StyleChani, M. T. S., Karimov, K. S., Asiri, A. M., Kamal, T., Bakhsh, E. M., & Rahman, M. M. (2022). Ultraviolet and Infrared Irradiations Sensing of Gel-Orange Dye Composite-Based Flexible Electrochemical Cells. Gels, 8(2), 83. https://doi.org/10.3390/gels8020083








