Decoupling between Translational Diffusion and Viscoelasticity in Transient Networks with Controlled Network Connectivity
Abstract
1. Introduction
2. Results and Discussion
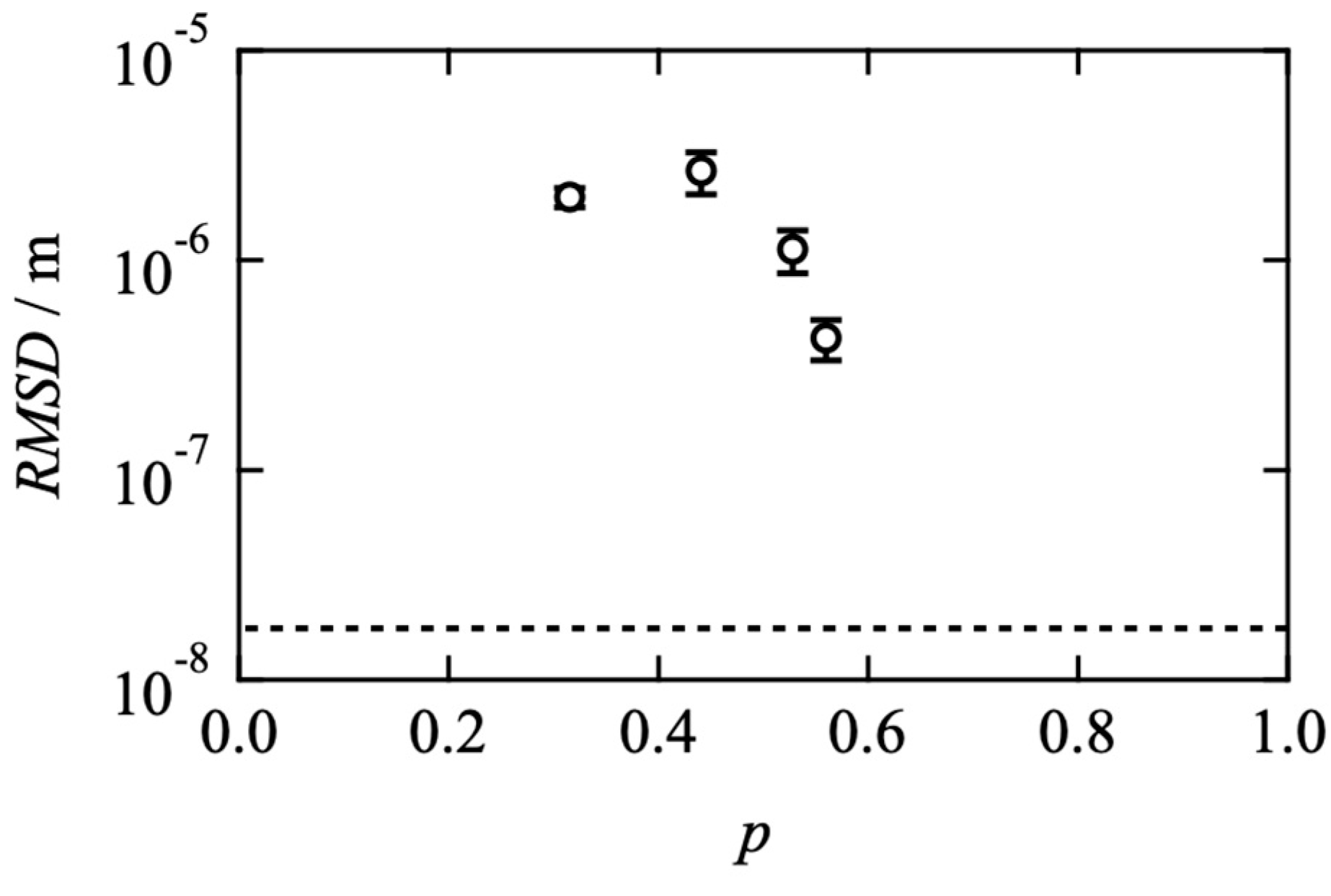
2.1. Effects of Network Connectivity on Viscoelasticity
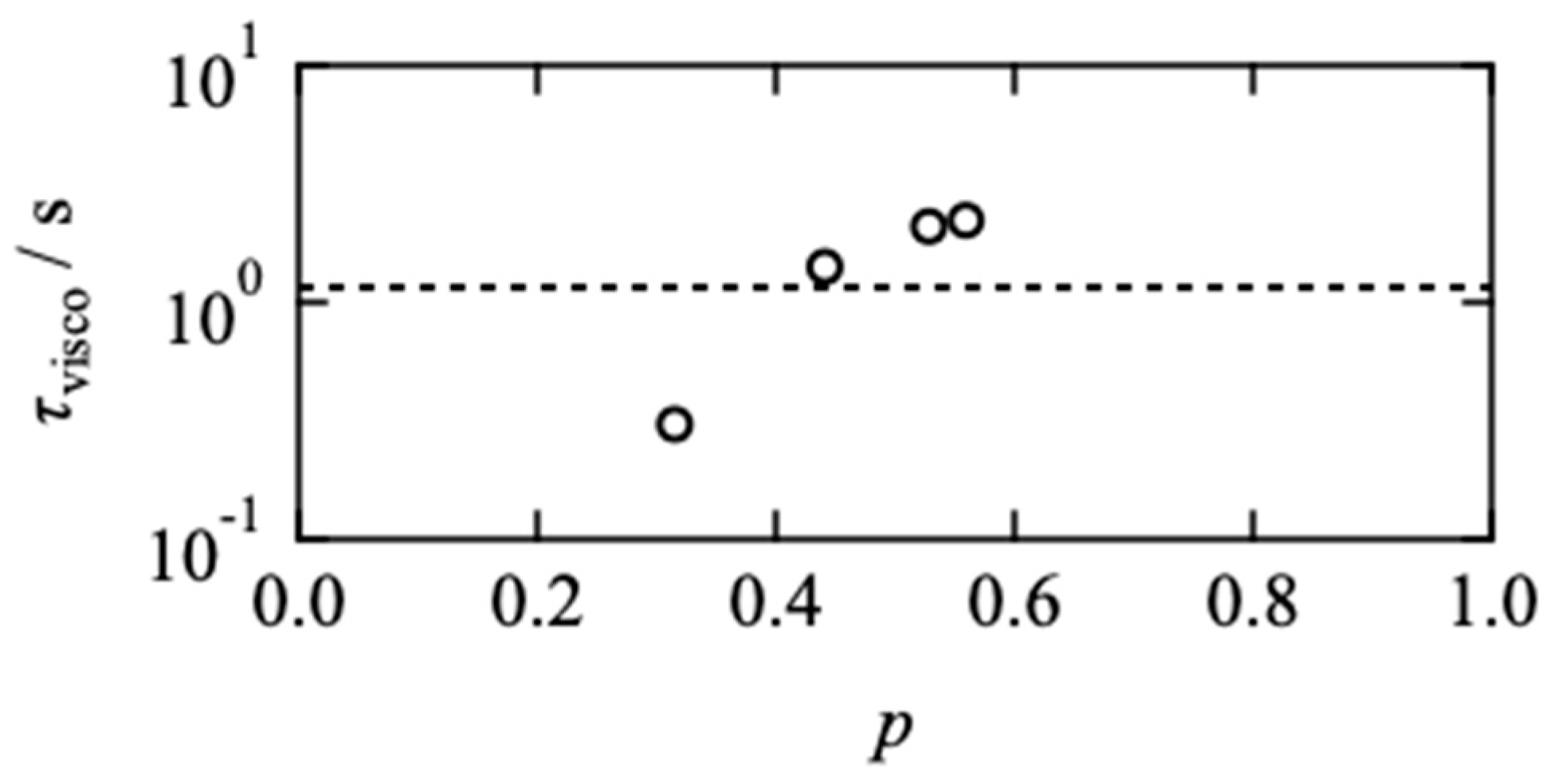
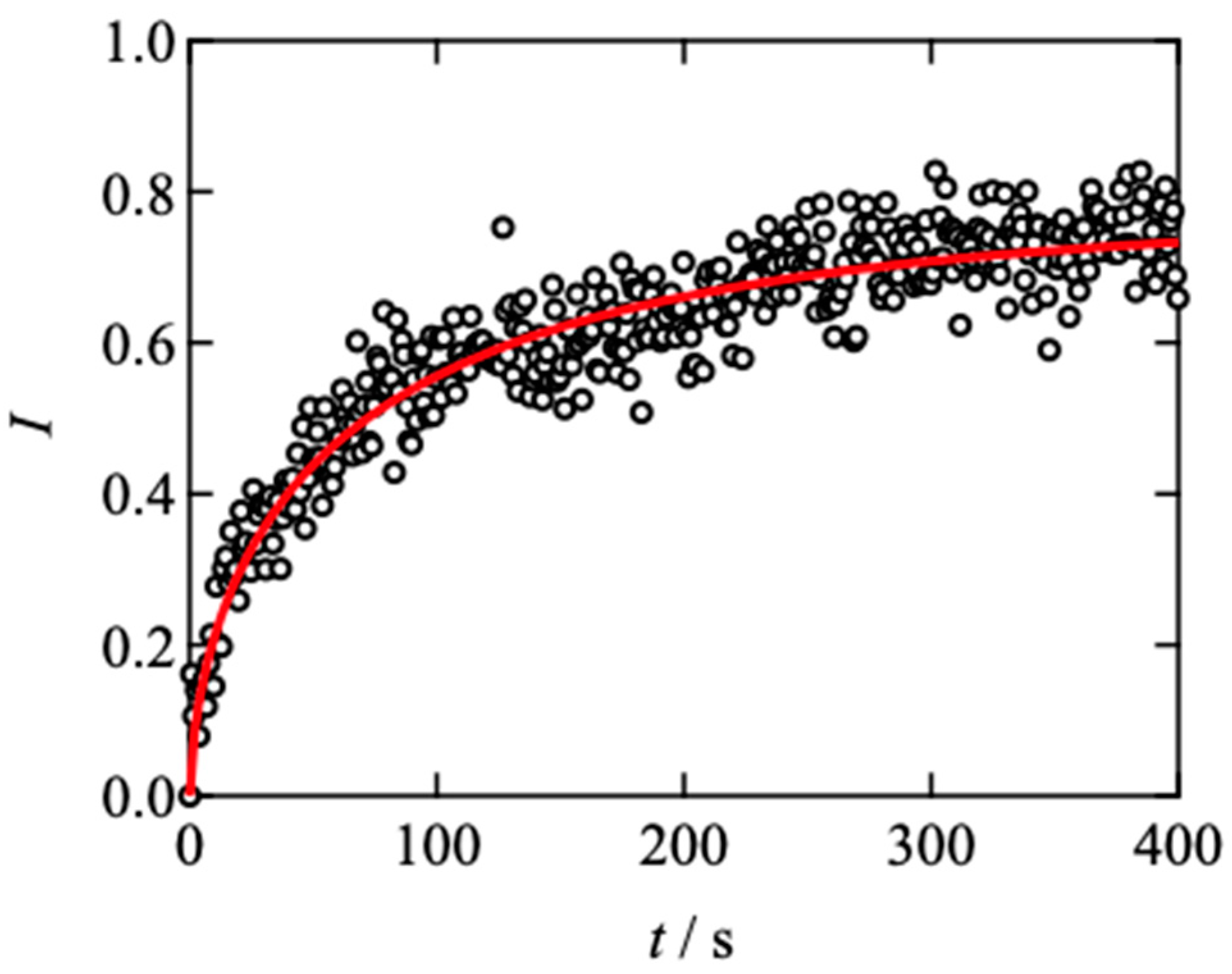
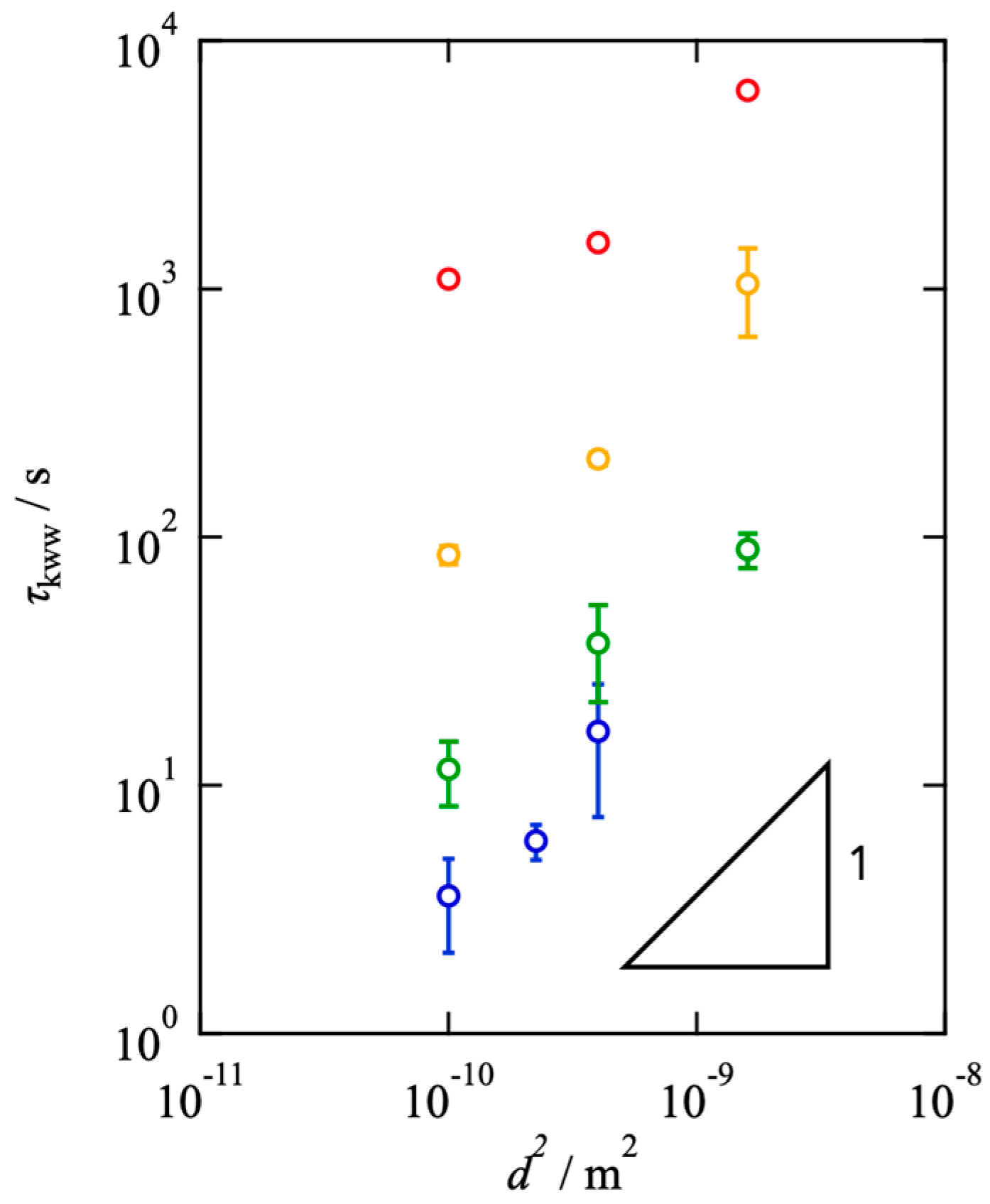
2.2. Effects of Network Connectivity on Diffusivity
2.3. Comparison between Viscoelasticity and Diffusivity
3. Conclusions
4. Materials and Methods
4.1. Synthesis of Fluorescence-Labeled Tetra-Armed Prepolymers
4.2. Sample Preparation
4.3. Dynamic Viscoelastic Measurements
4.4. Fluorescence Recovery after Photobleaching (FRAP) Measurements
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Annable, T.; Ettelaie, R. Thermodynamics of Phase Separation in Mixtures of Associating Polymers and Homopolymers in Solution. Macromolecules 1994, 27, 5616–5622. [Google Scholar] [CrossRef]
- Uneyama, T.; Suzuki, S.; Watanabe, H. Concentration Dependence of Rheological Properties of Telechelic Associative Polymer Solutions. Phys. Rev. E Stat. Nonlin. Soft Matter Phys. 2012, 86, 031802. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, M.; Katashima, T.; Nakahata, M.; Urakawa, O. Relationships between Diffusion and Viscoelasticity of Associative Polymer Networks. Nihon Reoroji Gakkaishi 2019, 47, 133–142. [Google Scholar] [CrossRef]
- Katashima, T. Rheological Studies on Polymer Networks with Static and Dynamic Crosslinks. Polym. J. 2021, 53, 1073–1082. [Google Scholar] [CrossRef]
- Mahmad Rasid, I.; Holten-Andersen, N.; Olsen, B.D. Anomalous Diffusion in Associative Networks of High-Sticker-Density Polymers. Macromolecules 2021, 54, 1354–1365. [Google Scholar] [CrossRef]
- Furó, I.; Iliopoulos, I.; Stilbs, P. Structure and Dynamics of Associative Water-Soluble Polymer Aggregates As Seen by 19F NMR Spectroscopy. J. Phys. Chem. B 2000, 104, 485–494. [Google Scholar] [CrossRef]
- Narita, T.; Mayumi, K.; Ducouret, G.; Hébraud, P. Viscoelastic Properties of Poly(Vinyl Alcohol) Hydrogels Having Permanent and Transient Cross-Links Studied by Microrheology, Classical Rheometry, and Dynamic Light Scattering. Macromolecules 2013, 46, 4174–4183. [Google Scholar] [CrossRef]
- Chiba, T.; Katashima, T.; Urakawa, O.; Inoue, T. Rheological Test for the Homogeneity of Aqueous Blends of Associative Polymer Network and Entangled Linear Polymer. Nihon Reoroji Gakkaishi 2020, 48, 49–54. [Google Scholar] [CrossRef]
- Dankers, P.Y.W.; van Luyn, M.J.A.; Huizinga-van der Vlag, A.; van Gemert, G.M.L.; Petersen, A.H.; Meijer, E.W.; Janssen, H.M.; Bosman, A.W.; Popa, E.R. Development and In-Vivo Characterization of Supramolecular Hydrogels for Intrarenal Drug Delivery. Biomaterials 2012, 33, 5144–5155. [Google Scholar] [CrossRef]
- Vintiloiu, A.; Leroux, J.-C. Organogels and Their Use in Drug Delivery-A Review. J. Control. Release 2008, 125, 179–192. [Google Scholar] [CrossRef]
- Yesilyurt, V.; Webber, M.J.; Appel, E.A.; Godwin, C.; Langer, R.; Anderson, D.G. Injectable Self-Healing Glucose-Responsive Hydrogels with PH-Regulated Mechanical Properties. Adv. Mater. 2016, 28, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Dankers, P.Y.W.; Hermans, T.M.; Baughman, T.W.; Kamikawa, Y.; Kieltyka, R.E.; Bastings, M.M.C.; Janssen, H.M.; Sommerdijk, N.A.J.M.; Larsen, A.; van Luyn, M.J.A.; et al. Hierarchical Formation of Supramolecular Transient Networks in Water: A Modular Injectable Delivery System. Adv. Mater. 2012, 24, 2703–2709. [Google Scholar] [CrossRef] [PubMed]
- Yesilyurt, V.; Ayoob, A.M.; Appel, E.A.; Borenstein, J.T.; Langer, R.; Anderson, D.G. Mixed Reversible Covalent Crosslink Kinetics Enable Precise, Hierarchical Mechanical Tuning of Hydrogel Networks. Adv. Mater. 2017, 29, 1605947. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, A.; Ishii, T.; Nishida, J.; Matsumoto, H.; Kataoka, K.; Miyahara, Y. A Synthetic Approach toward a Self-Regulated Insulin Delivery System. Angew. Chem. Weinheim Bergstr. Ger. 2012, 124, 2166–2170. [Google Scholar] [CrossRef]
- Naito, M.; Ishii, T.; Matsumoto, A.; Miyata, K.; Miyahara, Y.; Kataoka, K. A Phenylboronate-Functionalized Polyion Complex Micelle for ATP-Triggered Release of SiRNA. Angew. Chem. Int. Ed. Engl. 2012, 51, 10751–10755. [Google Scholar] [CrossRef]
- Gaskell, T. Self-Diffusion in Liquid Metals: A Generalized Stokes-Einstein Equation. J. Non-Cryst. Solids 1984, 61–62, 913–918. [Google Scholar] [CrossRef]
- Mason, T.G. Estimating the Viscoelastic Moduli of Complex Fluids Using the Generalized Stokes–Einstein Equation. Rheol. Acta 2000, 39, 371–378. [Google Scholar] [CrossRef]
- Mason, T.G.; Ganesan, K.; van Zanten, J.H.; Wirtz, D.; Kuo, S.C. Particle Tracking Microrheology of Complex Fluids. Phys. Rev. Lett. 1997, 79, 3282–3285. [Google Scholar] [CrossRef]
- Banchio, A.J.; Nägele, G.; Bergenholtz, J. Viscoelasticity and Generalized Stokes–Einstein Relations of Colloidal Dispersions. J. Chem. Phys. 1999, 111, 8721–8740. [Google Scholar] [CrossRef]
- Kholodenko, A.L.; Douglas, J.F. Generalized Stokes-Einstein Equation for Spherical Particle Suspensions. Phys. Rev. E Stat. Phys. Plasmas Fluids Relat. Interdiscip. Topics 1995, 51, 1081–1090. [Google Scholar] [CrossRef]
- Doi, M.; Edwards, S.F.; Edwards, S.F. The Theory of Polymer Dynamics; Clarendon Press: Oxford, UK, 1988; ISBN 9780198520337. [Google Scholar]
- Watanabe, H. Slow Dynamics in Homopolymer Liquids. Polym. J. 2009, 41, 929–950. [Google Scholar] [CrossRef]
- Watanabe, H. Viscoelasticity and Dynamics of Entangled Polymers. Prog. Polym. Sci. 1999, 24, 1253–1403. [Google Scholar] [CrossRef]
- Watanabe, H.; Matsumiya, Y.; Inoue, T. Dielectric and Viscoelastic Relaxation of Highly Entangled Star Polyisoprene: Quantitative Test of Tube Dilation Model. Macromolecules 2002, 35, 2339–2357. [Google Scholar] [CrossRef]
- Watanabe, H. Fundamental Findings in Softmatter Rheology on the Basis of Multiple Methods. Nihon Reoroji Gakkaishi 2013, 40, 209–222. [Google Scholar] [CrossRef][Green Version]
- Watanabe, H.; Matsumiya, Y.; Inoue, T. Revisit the Stress-Optical Rule for Entangled Flexible Chains: Overshoot of Stress, Segmental Orientation, and Chain Stretch on Start-up of Flow. Nihon Reoroji Gakkaishi 2015, 43, 105–112. [Google Scholar] [CrossRef]
- Yamamura, T.; Kusaka, T.; Takatori, E.; Inoue, T.; Nemoto, N.; Osaki, K.; Shikata, T.; Kotaka, T. Self-Diffusion of Micelles and Viscoelasticity of Aqueous Detergent Solutions. Nihon Reoroji Gakkaishi 1991, 19, 45–49. [Google Scholar] [CrossRef][Green Version]
- Katashima, T. Molecular Understanding of Viscoelasticity in Transient Polymer Networks Based on Multiple Methods. Nihon Reoroji Gakkaishi 2022, 50, 51–56. [Google Scholar] [CrossRef]
- Katashima, T.; Kudo, R.; Naito, M.; Nagatoishi, S.; Miyata, K.; Chung, U.; Tsumoto, K.; Sakai, T. Experimental Comparison of Bond Lifetime and Viscoelastic Relaxation in Transient Networks with Well-Controlled Structures. ACS Macro Lett. 2022, 11, 753–759. [Google Scholar] [CrossRef]
- Katashima, T.; Kudo, R.; Onishi, R.; Naito, M.; Nagatoishi, S.; Miyata, K.; Tsumoto, K.; Chung, U.; Sakai, T. Effects of Network Connectivity on Viscoelastic Relaxation in Transient Networks Using Experimental Approach. Front. Soft. Matter 2022, 2, 1059156. [Google Scholar] [CrossRef]
- Green, M.S.; Tobolsky, A.V. A New Approach to the Theory of Relaxing Polymeric Media. J. Chem. Phys. 1946, 14, 80–92. [Google Scholar] [CrossRef]
- Yamamoto, M. The Visco-Elastic Properties of Network Structure I. General Formalism. J. Phys. Soc. Jpn. 1956, 11, 413–421. [Google Scholar] [CrossRef]
- Tang, S.; Habicht, A.; Li, S.; Seiffert, S.; Olsen, B.D. Self-Diffusion of Associating Star-Shaped Polymers. Macromolecules 2016, 49, 5599–5608. [Google Scholar] [CrossRef]
- Ramirez, J.; Dursch, T.J.; Olsen, B.D. A Molecular Explanation for Anomalous Diffusion in Supramolecular Polymer Networks. Macromolecules 2018, 51, 2517–2525. [Google Scholar] [CrossRef]
- Kawasaki, Y.; Watanabe, H.; Uneyama, T. A Note for Kohlrausch-Williams-Watts Relaxation Function. Nihon Reoroji Gakkaishi 2011, 39, 127–131. [Google Scholar] [CrossRef]
- Baxandall, L.G. Dynamics of Reversibly Crosslinked Chains. Macromolecules 1989, 22, 1982–1988. [Google Scholar] [CrossRef]
- Kienberger, F.; Pastushenko, V.P.; Kada, G.; Gruber, H.J.; Riener, C.; Schindler, H.; Hinterdorfer, P. Static and Dynamical Properties of Single Poly(Ethylene Glycol) Molecules Investigated by Force Spectroscopy. Single Mol. 2000, 1, 123–128. [Google Scholar] [CrossRef]
- Oesterhelt, F.; Rief, M.; Gaub, H.E. Single Molecule Force Spectroscopy by AFM Indicates Helical Structure of Poly(Ethylene-Glycol) in Water. New J. Phys. 1999, 1, 6. [Google Scholar] [CrossRef]
- Zou, S.; Schönherr, H.; Vancso, G.J. Stretching and Rupturing Individual Supramolecular Polymer Chains by AFM. Angew. Chem. Int. Ed. Engl. 2005, 44, 956–959. [Google Scholar] [CrossRef]
- Akagi, Y.; Sakurai, H.; Gong, J.P.; Chung, U.; Sakai, T. Fracture Energy of Polymer Gels with Controlled Network Structures. J. Chem. Phys. 2013, 139, 144905. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katashima, T.; Kobayashi, R.; Ishikawa, S.; Naito, M.; Miyata, K.; Chung, U.-i.; Sakai, T. Decoupling between Translational Diffusion and Viscoelasticity in Transient Networks with Controlled Network Connectivity. Gels 2022, 8, 830. https://doi.org/10.3390/gels8120830
Katashima T, Kobayashi R, Ishikawa S, Naito M, Miyata K, Chung U-i, Sakai T. Decoupling between Translational Diffusion and Viscoelasticity in Transient Networks with Controlled Network Connectivity. Gels. 2022; 8(12):830. https://doi.org/10.3390/gels8120830
Chicago/Turabian StyleKatashima, Takuya, Ryunosuke Kobayashi, Shohei Ishikawa, Mitsuru Naito, Kanjiro Miyata, Ung-il Chung, and Takamasa Sakai. 2022. "Decoupling between Translational Diffusion and Viscoelasticity in Transient Networks with Controlled Network Connectivity" Gels 8, no. 12: 830. https://doi.org/10.3390/gels8120830
APA StyleKatashima, T., Kobayashi, R., Ishikawa, S., Naito, M., Miyata, K., Chung, U.-i., & Sakai, T. (2022). Decoupling between Translational Diffusion and Viscoelasticity in Transient Networks with Controlled Network Connectivity. Gels, 8(12), 830. https://doi.org/10.3390/gels8120830






