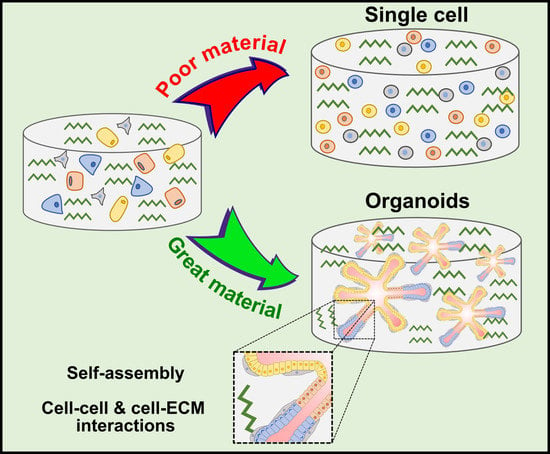
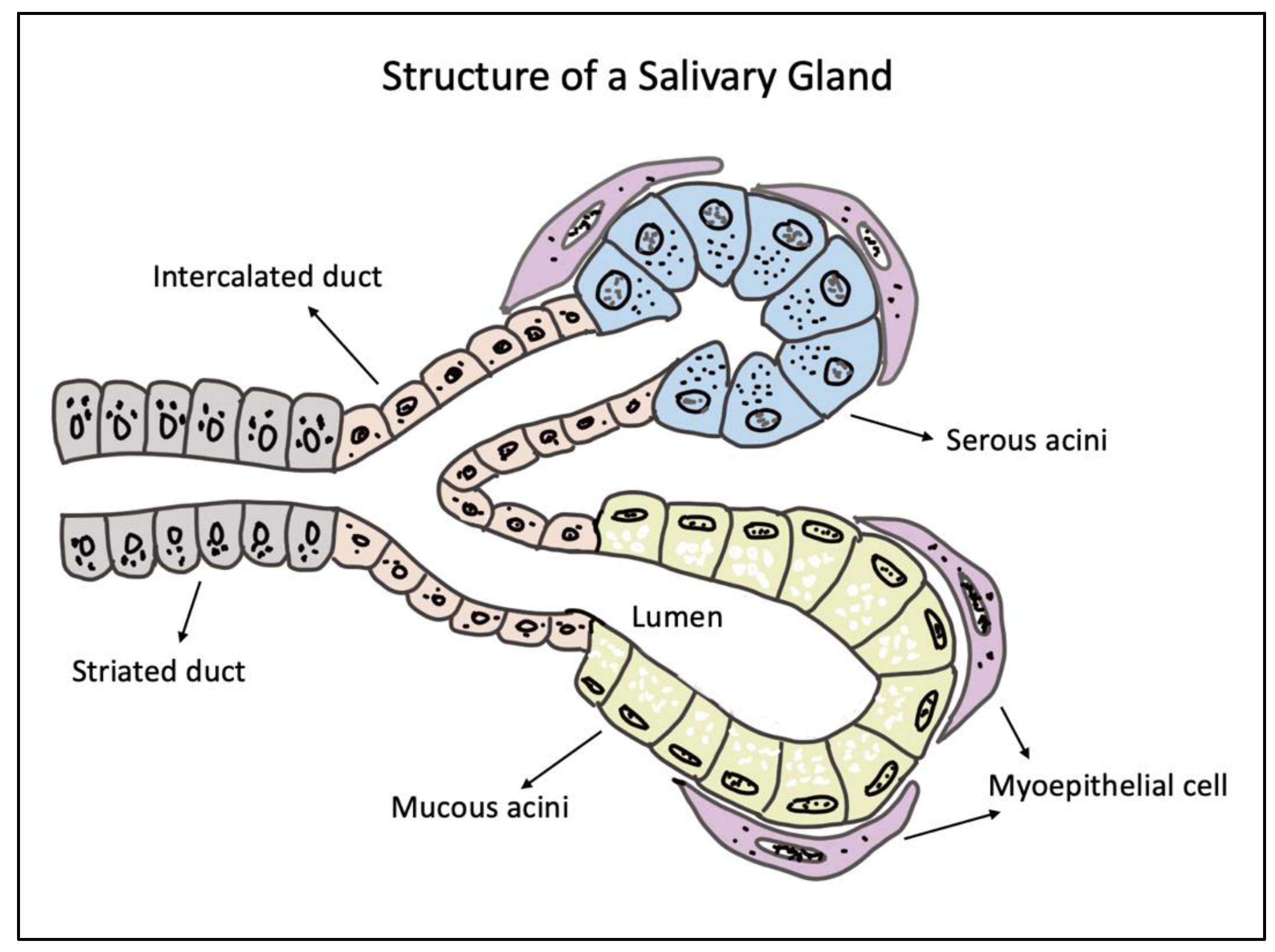
Hydrogels for Salivary Gland Tissue Engineering
Abstract
1. Introduction
2. Tuning Ideal Hydrogel Properties for SG Tissue Engineering
2.1. Hydrogel Biocompatibility
2.2. Presence of Cell Binding Motifs
2.3. Hydrogel Stiffness
2.4. Hydrogel Pore Size
2.5. Viscoelasticity and Hydrogel Architecture
3. Advances in Hydrogel Systems for Building an Artificial SG
3.1. Naturally Derived Hydrogels
3.2. Synthetic Hydrogels
4. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| 2D | Two-dimensional |
| 2.5D | Two-point-five-dimensional |
| 3D | Three-dimensional |
| Akt | Protein kinase B (PKB) |
| APC | Adenomatous polyposis coli |
| AQP-5 | Aquaporin 5 |
| BM | Basement membrane proteins |
| CD44 | Cluster of Differentiation 44 |
| DGEA | Aspartic acid-glycine-glutamic acid-alanine |
| ECM | Extracellular matrix |
| EDTA | Ethylenediaminetetraacetic acid |
| EW | Egg White |
| EYP | Egg Yolk Plasma |
| FA | Focal adhesion |
| FAKs | Focal adhesion kinases |
| GelMA | Gelatin-methacrylamide |
| GSK3β | Glycogen synthase kinase 3 beta |
| HA | Hyaluronic acid |
| Kit | Tyrosine-protein kinase (CD117) |
| Lasts 1/2 | Large tumor suppressor 1 and 2 |
| Mist-1 | Muscle, intestine and stomach expression 1 |
| MLCK | Myosin light-chain kinase |
| Mst 1/2 | Mammalian sterile 20-like kinase 1 and 2 |
| MW | Molecular weight |
| NF2 | Neurofibromatosis type 2 |
| NS-SV-AC | Normal human SG-simian virus 40-acinar cells |
| PCL | Polycaprolactone |
| PEG | Polyethelene glycol |
| PG | Parotid gland |
| PGA | Polyglycolic acid |
| PI3K | Phosphatidylinositol-3-kinase |
| PLLA | Poly-l-lactic acid |
| PLGA | Poly-l-lactic-co-glycolic acid |
| RGD | Arginine-glycine-aspartic acid |
| RhoA | Transforming protein RhoA |
| ROCK | Rho kinases |
| SGs | Salivary glands |
| SLG | Sublingual gland |
| SMG | Submandibular gland |
| SRC | Proto-oncogene tyrosine-protein kinase |
| TAZ | Transcriptional co-activator with PDZ-binding motif |
| YAP | Yes Associated Protein |
| YIGSR | Tyrosine-isoleucine-glycine-serine-arginine |
| ZO-1 | Zonula occludens-1 |
References
- Edgar, W.M. Saliva and dental health. Clinical implications of saliva: Report of a consensus meeting. Br. Dent. J. 1990, 169, 96–98. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.N.; Hoffman, M.P. Interactions between developing nerves and salivary glands. Organogenesis 2013, 9, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, S.P.; Williamson, R.T. A review of saliva: Normal composition, flow, and function. J. Prosthet. Dent. 2001, 85, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Holmberg, K.V.; Hoffman, M.P. Anatomy, biogenesis and regeneration of salivary glands. Saliva Secret. Funct. 2014, 24, 1–13. [Google Scholar]
- Roth, G.; Calmes, R. Salivary Glands and Saliva. In Oral Biology; CV Mosby: St. Louis, MO, USA, 1981; pp. 196–236. [Google Scholar]
- Nanci, A. Ten Cate’s Oral Histology-e-Book: Development, Structure, and Function; Elsevier Health Sciences: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Aure, M.; Arany, S.; Ovitt, C. Salivary glands: Stem cells, self-duplication, or both? J. Dent. Res. 2015, 94, 1502–1507. [Google Scholar] [CrossRef]
- Harrison, J.D. Salivary Gland Histology, in Surgery of the Salivary Glands; Elsevier: Amsterdam, The Netherlands, 2021; pp. 37–42. [Google Scholar]
- Brazen, B.; Dyer, J. Histology, Salivary Glands; StatPearls: Treasure Island, FL, USA, 2019. [Google Scholar]
- Rocchi, C.; Barazzuol, L.; Coppes, R.P. The evolving definition of salivary gland stem cells. NPJ Regen. Med. 2021, 6, 4. [Google Scholar] [CrossRef]
- Tran, S.; Wang, J.; Bandyopadhyay, B.; Redman, R.; Dutra, A.; Pak, E.; Swaim, W.; Gerstenhaber, J.; Bryant, J.; Zheng, C. Primary culture of polarized human salivary epithelial cells for use in developing an artificial salivary gland. Tissue Eng. 2005, 11, 172–181. [Google Scholar] [CrossRef]
- Maria, O.M.; Zeitouni, A.; Gologan, O.; Tran, S.D. Matrigel improves functional properties of primary human salivary gland cells. Tissue Eng. Part A 2011, 17, 1229–1238. [Google Scholar] [CrossRef]
- Su, X.; Fang, D.; Liu, Y.; Ramamoorthi, M.; Zeitouni, A.; Chen, W.; Tran, S. Three-dimensional organotypic culture of human salivary glands: The slice culture model. Oral Dis. 2016, 22, 639–648. [Google Scholar] [CrossRef]
- Seo, Y.J.; Lilliu, M.A.; Abu Elghanam, G.; Nguyen, T.T.; Liu, Y.; Lee, J.C.; Presley, J.F.; Zeitouni, A.; El-Hakim, M.; Tran, S.D. Cell culture of differentiated human salivary epithelial cells in a serum-free and scalable suspension system: The salivary functional units model. J. Tissue Eng. Regen. Med. 2019, 13, 1559–1570. [Google Scholar] [CrossRef]
- Song, Y.; Uchida, H.; Sharipol, A.; Piraino, L.; Mereness, J.A.; Ingalls, M.H.; Rebhahn, J.; Newlands, S.D.; DeLouise, L.A.; Ovitt, C.E. Development of a functional salivary gland tissue chip with potential for high-content drug screening. Commun. Biol. 2021, 4, 361. [Google Scholar] [CrossRef]
- Pradhan, S.; Liu, C.; Zhang, C.; Jia, X.; Farach-Carson, M.C.; Witt, R.L. Lumen formation in three-dimensional cultures of salivary acinar cells. Otolaryngol.—Head Neck Surg. 2010, 142, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Pradhan-Bhatt, S.; Harrington, D.A.; Duncan, R.L.; Jia, X.; Witt, R.L.; Farach-Carson, M.C. Implantable three-dimensional salivary spheroid assemblies demonstrate fluid and protein secretory responses to neurotransmitters. Tissue Eng. Part A 2013, 19, 1610–1620. [Google Scholar] [CrossRef] [PubMed]
- Baum, J.B.; Tran, S.D. Synergy between genetic and tissue engineering: Creating an artificial salivary gland. Periodontology 2006, 41, 218–223. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Mui, B.; Mehrabzadeh, M.; Chea, Y.; Chaudhry, Z.; Chaudhry, K.; Tran, S.D. Regeneration of tissues of the oral complex: Current. J. Can. Dent. Assoc. 2013, 79, d1. [Google Scholar]
- Aframian, J.D.; Cukierman, E.; Nikolovski, J.; Mooney, D.J.; Yamada, K.M.; Baum, B.J. The growth and morphological behavior of salivary epithelial cells on matrix protein-coated biodegradable substrata. Tissue Eng. 2000, 6, 209–216. [Google Scholar] [CrossRef]
- Mantha, S.; Pillai, S.; Khayambashi, P.; Upadhyay, A.; Zhang, Y.; Tao, O.; Pham, H.M.; Tran, S.D. Smart hydrogels in tissue engineering and regenerative medicine. Materials 2019, 12, 3323. [Google Scholar] [CrossRef]
- Maria, M.O.; Maria, O.; Liu, Y.; Komarova, S.V.; Tran, S.D. Matrigel improves functional properties of human submandibular salivary gland cell line. Int. J. Biochem. Cell Biol. 2011, 43, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Pillai, S.; Liu, Y.; Tran, S.D. Isolation, Culture, and Characterization of Primary Salivary Gland Cells. Curr. Protoc. 2022, 2, e479. [Google Scholar] [CrossRef] [PubMed]
- Miyajima, H.; Matsumoto, T.; Sakai, T.; Yamaguchi, S.; An, S.H.; Abe, M.; Wakisaka, S.; Lee, K.Y.; Egusa, H.; Imazato, S. Hydrogel-based biomimetic environment for in vitro modulation of branching morphogenesis. Biomaterials 2011, 32, 6754–6763. [Google Scholar] [CrossRef]
- Aframian, J.D.; Redman, R.; Yamano, S.; Nikolovski, J.; Cukierman, E.; Yamada, K.M.; Kriete, M.; Swaim, W.; Mooney, D.J.; Baum, B.J. Tissue compatibility of two biodegradable tubular scaffolds implanted adjacent to skin or buccal mucosa in mice. Tissue Eng. 2002, 8, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, T.; Srinivasan, P.P.; Zakheim, D.R.; Harrington, D.A.; Witt, R.L.; Farach-Carson, M.C.; Jia, X.; Pradhan-Bhatt, S. Bottom-up assembly of salivary gland microtissues for assessing myoepithelial cell function. Biomaterials 2017, 142, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Naahidi, S.; Jafari, M.; Logan, M.; Wang, Y.; Yuan, Y.; Bae, H.; Dixon, B.; Chen, P. Biocompatibility of hydrogel-based scaffolds for tissue engineering applications. Biotechnol. Adv. 2017, 35, 530–544. [Google Scholar] [CrossRef]
- Rago, P.A.; Napolitano, A.P.; Dean, D.M.; Chai, P.R.; Morgan, J.R. Miniaturization of an Anoikis assay using non-adhesive micromolded hydrogels. Cytotechnology 2008, 56, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Sisto, M.; Ribatti, D.; Lisi, S. E-Cadherin Signaling in Salivary Gland Development and Autoimmunity. J. Clin. Med. 2022, 11, 2241. [Google Scholar] [CrossRef]
- Jiang, T.; Munguia-Lopez, J.G.; Flores-Torres, S.; Kort-Mascort, J.; Kinsella, J.M. Extrusion bioprinting of soft materials: An emerging technique for biological model fabrication. Appl. Phys. Rev. 2019, 6, 011310. [Google Scholar] [CrossRef]
- Martinez, M.; Witt, R.L.; Farach-Carson, M.C.; Harrington, D.A. Functionalized biomimetic hydrogels enhance salivary stem/progenitor cell organization. bioRxiv 2021. [Google Scholar]
- Fowler, W.E.; Ravikrishnan, A.; Witt, R.L.; Pradhan-Bhatt, S.; Jia, X. RGDSP-Decorated Hyaluronate Hydrogels Facilitate Rapid 3D Expansion of Amylase-Expressing Salivary Gland Progenitor Cells. ACS Biomater. Sci. Eng. 2021, 7, 5749–5761. [Google Scholar] [CrossRef]
- Yeung, T.; Georges, P.C.; Flanagan, L.A.; Marg, B.; Ortiz, M.; Funaki, M.; Zahir, N.; Ming, W.; Weaver, V.; Janmey, P.A. Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion. Cell Motil. Cytoskelet. 2005, 60, 24–34. [Google Scholar] [CrossRef]
- d’Angelo, M.; Benedetti, E.; Tupone, M.G.; Catanesi, M.; Castelli, V.; Antonosante, A.; Cimini, A. The role of stiffness in cell reprogramming: A potential role for biomaterials in inducing tissue regeneration. Cells 2019, 8, 1036. [Google Scholar] [CrossRef]
- Žigon-Branc, S.; Markovic, M.; van Hoorick, J.; van Vlierberghe, S.; Dubruel, P.; Zerobin, E.; Baudis, S.; Ovsianikov, A. Impact of hydrogel stiffness on differentiation of human adipose-derived stem cell microspheroids. Tissue Eng. Part A 2019, 25, 1369–1380. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, Y.; Enhe, J.; Yao, B.; Wang, Y.; Zhu, D.; Li, Z.; Song, W.; Duan, X.; Yuan, X. Bioactive nanoparticle reinforced alginate/gelatin bioink for the maintenance of stem cell stemness. Mater. Sci. Eng. C 2021, 126, 112193. [Google Scholar] [CrossRef]
- Sun, Z.; Guo, S.S.; Fässler, R. Integrin-mediated mechanotransduction. J. Cell Biol. 2016, 215, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Aponte-Santamaría, C.; Sturm, S.; Bullerjahn, J.T.; Bronowska, A.; Gräter, F. Mechanism of focal adhesion kinase mechanosensing. PLoS Comput. Biol. 2015, 11, e1004593. [Google Scholar] [CrossRef] [PubMed]
- Sequeira, J.S.; Soscia, D.A.; Oztan, B.; Mosier, A.P.; Jean-Gilles, R.; Gadre, A.; Cady, N.C.; Yener, B.; Castracane, J.; Larsen, M. The regulation of focal adhesion complex formation and salivary gland epithelial cell organization by nanofibrous PLGA scaffolds. Biomaterials 2012, 33, 3175–3186. [Google Scholar] [CrossRef]
- Daley, P.W.; Gervais, E.M.; Centanni, S.W.; Gulfo, K.M.; Nelson, D.A.; Larsen, M. ROCK1-directed basement membrane positioning coordinates epithelial tissue polarity. Development 2012, 139, 411–422. [Google Scholar] [CrossRef]
- Han, C.; An, G.H.; Woo, D.-H.; Kim, J.-H.; Park, H.-K. Rho-associated kinase inhibitor enhances the culture condition of isolated mouse salivary gland cells in vitro. Tissue Cell 2018, 54, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Koslow, M.; O’Keefe, K.J.; Hosseini, Z.F.; Nelson, D.A.; Larsen, M. ROCK inhibitor increases proacinar cells in adult salivary gland organoids. Stem Cell Res. 2019, 41, 101608. [Google Scholar] [CrossRef]
- Melica, E.M.; La Regina, G.; Parri, M.; Peired, A.J.; Romagnani, P.; Lasagni, L. Substrate stiffness modulates renal progenitor cell properties via a ROCK-mediated mechanotransduction mechanism. Cells 2019, 8, 1561. [Google Scholar] [CrossRef]
- Jaalouk, E.D.; Lammerding, J. Mechanotransduction gone awry. Nat. Rev. Mol. Cell Biol. 2009, 10, 63–73. [Google Scholar] [CrossRef]
- Wang, S.; Matsumoto, K.; Lish, S.R.; Cartagena-Rivera, A.X.; Yamada, K.M. Budding epithelial morphogenesis driven by cell-matrix versus cell-cell adhesion. Cell 2021, 184, 3702–3716.e30. [Google Scholar] [CrossRef] [PubMed]
- Yui, S.; Azzolin, L.; Maimets, M.; Pedersen, M.T.; Fordham, R.P.; Hansen, S.L.; Larsen, H.L.; Guiu, J.; Alves, M.R.; Rundsten, C.F. YAP/TAZ-dependent reprogramming of colonic epithelium links ECM remodeling to tissue regeneration. Cell Stem Cell 2018, 22, 35–49.e7. [Google Scholar] [CrossRef] [PubMed]
- Panciera, T.; Azzolin, L.; Fujimura, A.; di Biagio, D.; Frasson, C.; Bresolin, S.; Soligo, S.; Basso, G.; Bicciato, S.; Rosato, A. Induction of expandable tissue-specific stem/progenitor cells through transient expression of YAP/TAZ. Cell Stem Cell 2016, 19, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Szymaniak, D.A.; Mi, R.; McCarthy, S.E.; Gower, A.C.; Reynolds, T.L.; Mingueneau, M.; Kukuruzinska, M.; Varelas, X. The Hippo pathway effector YAP is an essential regulator of ductal progenitor patterning in the mouse submandibular gland. Elife 2017, 6, e23499. [Google Scholar] [CrossRef]
- Rocchi, C.; Cinat, D.; Martinez, P.S.; Bruin, A.L.J.-d.; Baanstra, M.; Brouwer, U.; Zuivre, C.d.; Schepers, H.; van Os, R.; Barazzuol, L. The Hippo signaling pathway effector YAP promotes salivary gland regeneration after injury. Sci. Signal. 2021, 14, eabk0599. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S. Role of YAP/TAZ in mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef]
- Maimets, M.; Rocchi, C.; Bron, R.; Pringle, S.; Kuipers, J.; Giepmans, B.N.; Vries, R.G.; Clevers, H.; de Haan, G.; van Os, R. Long-term in vitro expansion of salivary gland stem cells driven by Wnt signals. Stem Cell Rep. 2016, 6, 150–162. [Google Scholar] [CrossRef]
- Du, J.; Zu, Y.; Li, J.; Du, S.; Xu, Y.; Zhang, L.; Jiang, L.; Wang, Z.; Chien, S.; Yang, C. Extracellular matrix stiffness dictates Wnt expression through integrin pathway. Sci. Rep. 2016, 6, 20395. [Google Scholar] [CrossRef]
- Hung, -C.W.; Chen, S.-H.; Paul, C.D.; Stroka, K.M.; Lo, Y.-C.; Yang, J.T.; Konstantopoulos, K. Distinct signaling mechanisms regulate migration in unconfined versus confined spaces. J. Cell Biol. 2013, 202, 807–824. [Google Scholar] [CrossRef]
- Doolin, T.M.; Stroka, K.M. P hysical confinement alters cytoskeletal contributions towards human mesenchymal stem cell migration. Cytoskeleton 2018, 75, 103–117. [Google Scholar] [CrossRef]
- Ozdemir, T.; Fowler, E.W.; Liu, S.; Harrington, D.A.; Witt, R.L.; Farach-Carson, M.C.; Pradhan-Bhatt, S.; Jia, X. Tuning hydrogel properties to promote the assembly of salivary gland spheroids in 3D. ACS Biomater. Sci. Eng. 2016, 2, 2217–2230. [Google Scholar] [CrossRef] [PubMed]
- Fraley, I.S.; Feng, Y.; Krishnamurthy, R.; Kim, D.-H.; Celedon, A.; Longmore, G.D.; Wirtz, D. A distinctive role for focal adhesion proteins in three-dimensional cell motility. Nat. Cell Biol. 2010, 12, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Elosegui-Artola, A.; Andreu, I.; Beedle, A.E.; Lezamiz, A.; Uroz, M.; Kosmalska, A.J.; Oria, R.; Kechagia, J.Z.; Rico-Lastres, P.; Le Roux, A.-L.; et al. Force triggers YAP nuclear entry by regulating transport across nuclear pores. Cell 2017, 171, 1397–1410.e14. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Kumar, A.; Makhija, E.; Shivashankar, G. The regulation of dynamic mechanical coupling between actin cytoskeleton and nucleus by matrix geometry. Biomaterials 2014, 35, 961–969. [Google Scholar] [CrossRef] [PubMed]
- Poh, -C.Y.; Shevtsov, S.P.; Chowdhury, F.; Wu, D.C.; Na, S.; Dundr, M.; Wang, N. Dynamic force-induced direct dissociation of protein complexes in a nuclear body in living cells. Nat. Commun. 2012, 3, 866. [Google Scholar] [CrossRef]
- Makhija, E.; Jokhun, D.; Shivashankar, G. Nuclear deformability and telomere dynamics are regulated by cell geometric constraints. Proc. Natl. Acad. Sci. USA 2016, 113, E32–E40. [Google Scholar] [CrossRef]
- Shubin, D.A.; Felong, T.J.; Graunke, D.; Ovitt, C.E.; Benoit, D.S. Development of poly (ethylene glycol) hydrogels for salivary gland tissue engineering applications. Tissue Eng. Part A 2015, 21, 1733–1751. [Google Scholar] [CrossRef]
- Blackburn, E.H. Switching and signaling at the telomere. Cell 2001, 106, 661–673. [Google Scholar] [CrossRef]
- Dey, K.; Agnelli, S.; Sartore, L. Dynamic freedom: Substrate stress relaxation stimulates cell responses. Biomater. Sci. 2019, 7, 836–842. [Google Scholar] [CrossRef]
- Chaudhuri, O.; Gu, L.; Klumpers, D.; Darnell, M.; Bencherif, S.A.; Weaver, J.C.; Huebsch, N.; Lee, H.-p.; Lippens, E.; Duda, G.N. Hydrogels with tunable stress relaxation regulate stem cell fate and activity. Nat. Mater. 2016, 15, 326–334. [Google Scholar] [CrossRef]
- Wisdom, M.K.; Adebowale, K.; Chang, J.; Lee, J.Y.; Nam, S.; Desai, R.; Rossen, N.S.; Rafat, M.; West, R.B.; Hodgson, L. Matrix mechanical plasticity regulates cancer cell migration through confining microenvironments. Nat. Commun. 2018, 9, 4144. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, H.S.; Yoo, H.S. Electrospun nanofibrils embedded hydrogel composites for cell cultivation in a biomimetic environment. RSC Adv. 2017, 7, 54246–54253. [Google Scholar] [CrossRef]
- Meehan, S.; Nain, A.S. Role of suspended fiber structural stiffness and curvature on single-cell migration, nucleus shape, and focal-adhesion-cluster length. Biophys. J. 2014, 107, 2604–2611. [Google Scholar] [CrossRef]
- Chaurey, V.; Block, F.; Su, Y.-H.; Chiang, P.-C.; Botchwey, E.; Chou, C.-F.; Swami, N.S. Nanofiber size-dependent sensitivity of fibroblast directionality to the methodology for scaffold alignment. Acta Biomater. 2012, 8, 3982–3990. [Google Scholar] [CrossRef] [PubMed]
- Soscia, A.D.; Sequeira, S.J.; Schramm, R.A.; Jayarathanam, K.; Cantara, S.I.; Larsen, M.; Castracane, J. Salivary gland cell differentiation and organization on micropatterned PLGA nanofiber craters. Biomaterials 2013, 34, 6773–6784. [Google Scholar] [CrossRef]
- Cao, H.; Duan, L.; Zhang, Y.; Cao, J.; Zhang, K. Current hydrogel advances in physicochemical and biological response-driven biomedical application diversity. Signal Transduct. Target. Ther. 2021, 6, 426. [Google Scholar] [CrossRef]
- Mendonsa, M.A.; Na, T.-Y.; Gumbiner, B.M. E-cadherin in contact inhibition and cancer. Oncogene 2018, 37, 4769–4780. [Google Scholar] [CrossRef]
- Kim, -G.N.; Koh, E.; Chen, X.; Gumbiner, B.M. E-cadherin mediates contact inhibition of proliferation through Hippo signaling-pathway components. Proc. Natl. Acad. Sci. USA 2011, 108, 11930–11935. [Google Scholar] [CrossRef]
- Hastings, F.J.; Skhinas, J.N.; Fey, D.; Croucher, D.R.; Cox, T.R. The extracellular matrix as a key regulator of intracellular signalling networks. Br. J. Pharmacol. 2019, 176, 82–92. [Google Scholar] [CrossRef]
- Urciuoli, E.; Peruzzi, B. Involvement of the FAK network in pathologies related to altered mechanotransduction. Int. J. Mol. Sci. 2020, 21, 9426. [Google Scholar] [CrossRef]
- Unagolla, M.J.; Jayasuriya, A.C. Hydrogel-based 3D bioprinting: A comprehensive review on cell-laden hydrogels, bioink formulations, and future perspectives. Appl. Mater. Today 2020, 18, 100479. [Google Scholar] [CrossRef] [PubMed]
- Soleymani Eil Bakhtiari, S.; Bakhsheshi-Rad, H.R.; Karbasi, S.; Razzaghi, M.; Tavakoli, M.; Ismail, A.F.; Sharif, S.; RamaKrishna, S.; Chen, X.; Berto, F. 3-Dimensional Printing of Hydrogel-Based Nanocomposites: A Comprehensive Review on the Technology Description, Properties, and Applications. Adv. Eng. Mater. 2021, 23, 2100477. [Google Scholar] [CrossRef]
- Chang, -Y.C.; Lin, C.-C. Hydrogel models with stiffness gradients for interrogating pancreatic cancer cell fate. Bioengineering 2021, 8, 37. [Google Scholar] [CrossRef]
- Moskwa, N.; Mahmood, A.; Nelson, D.A.; Altrieth, A.L.; Forni, P.E.; Larsen, M. Single-cell RNA sequencing reveals PDFGRα+ stromal cell subpopulations that promote proacinar cell differentiation in embryonic salivary gland organoids. Development 2022, 149, dev200167. [Google Scholar] [CrossRef]
- Baker, J.O.; Schulz, D.J.; Camden, J.M.; Liao, Z.; Peterson, T.S.; Seye, C.I.; Petris, M.J.; Weisman, G.A. Rat parotid gland cell differentiation in three-dimensional culture. Tissue Eng. Part C Methods 2010, 16, 1135–1144. [Google Scholar] [CrossRef]
- Aisenbrey, A.E.; Murphy, W.L. Synthetic alternatives to Matrigel. Nat. Rev. Mater. 2020, 5, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Nam, K.; Wang, C.-S.; Maruyama, C.; Lei, P.; Andreadis, S.; Baker, O. L1 peptide–conjugated fibrin hydrogels promote salivary gland regeneration. J. Dent. Res. 2017, 96, 798–806. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.D.; Munguia-Lopez, J.G.; Cho, Y.W.; Ma, X.; Song, V.; Zhu, Z.; Tran, S.D. Polymeric scaffolds for dental, oral, and craniofacial regenerative medicine. Molecules 2021, 26, 7043. [Google Scholar] [CrossRef]
- Santos, D.H.; Nam, K.; Brown, C.; Dean, S.; Lewis, S.; Pfeifer, C.; Lei, P.; Petris, M.; Andreadis, S.; Baker, O. Trimers conjugated to fibrin hydrogels promote salivary gland function. J. Dent. Res. 2021, 100, 268–275. [Google Scholar] [CrossRef] [PubMed]
- McCall, D.A.; Nelson, J.W.; Leigh, N.J.; Duffey, M.E.; Lei, P.; Andreadis, S.T.; Baker, O.J. Growth factors polymerized within fibrin hydrogel promote amylase production in parotid cells. Tissue Eng. Part A 2013, 19, 2215–2225. [Google Scholar] [CrossRef]
- Joraku, A.; Sullivan, C.A.; Yoo, J.; Atala, A. In-vitro reconstitution of three-dimensional human salivary gland tissue structures. Differentiation 2007, 75, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Charbonneau, M.A.; Tran, S.D. 3D Cell Culture of Human Salivary Glands Using Nature-Inspired Functional Biomaterials: The Egg Yolk Plasma and Egg White. Materials 2020, 13, 4807. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pham, H.M.; Munguia-Lopez, J.G.; Kinsella, J.M.; Tran, S.D. The optimization of a novel hydrogel—Egg white-alginate for 2.5 D tissue engineering of salivary spheroid-like structure. Molecules 2020, 25, 5751. [Google Scholar] [CrossRef]
- Pham, M.H.; Zhang, Y.; Munguia-Lopez, J.G.; Tran, S.D. Egg White Alginate as a Novel Scaffold Biomaterial for 3D Salivary Cell Culturing. Biomimetics 2021, 7, 5. [Google Scholar] [CrossRef] [PubMed]
- Flores-Torres, S.; Peza-Chavez, O.; Kuasne, H.; Munguia-Lopez, J.G.; Kort-Mascort, J.; Ferri, L.; Jiang, T.; Rajadurai, C.V.; Park, M.; Sangwan, V. Alginate–gelatin–Matrigel hydrogels enable the development and multigenerational passaging of patient-derived 3D bioprinted cancer spheroid models. Biofabrication 2021, 13, 025001. [Google Scholar] [CrossRef]
- Hogrebe, J.N.; Reinhardt, J.W.; Gooch, K.J. Biomaterial microarchitecture: A potent regulator of individual cell behavior and multicellular organization. J. Biomed. Mater. Res. Part A 2017, 105, 640–661. [Google Scholar] [CrossRef]
- Di Giuseppe, M.; Law, N.; Webb, B.; Macrae, R.A.; Liew, L.J.; Sercombe, T.B.; Dilley, R.J.; Doyle, B.J. Mechanical behaviour of alginate-gelatin hydrogels for 3D bioprinting. J. Mech. Behav. Biomed. Mater. 2018, 79, 150–157. [Google Scholar] [CrossRef]
- Yang, X.; Lu, Z.; Wu, H.; Li, W.; Zheng, L.; Zhao, J. Collagen-alginate as bioink for three-dimensional (3D) cell printing based cartilage tissue engineering. Mater. Sci. Eng. C 2018, 83, 195–201. [Google Scholar] [CrossRef]
- Jiang, T.; Munguia-Lopez, J.G.; Flores-Torres, S.; Grant, J.; Vijayakumar, S.; Leon-Rodriguez, A.D.; Kinsella, J.M. Directing the self-assembly of tumour spheroids by bioprinting cellular heterogeneous models within alginate/gelatin hydrogels. Sci. Rep. 2017, 7, 4575. [Google Scholar] [CrossRef]
- Garg, G.H.; Hales, C.A. Chemistry and Biology of Hyaluronan; Elsevier: Amsterdam, The Netherlands, 2004. [Google Scholar]
- Maria, O.; Maria, A.; Cai, Y.; Tran, S. Cell surface markers CD44 and CD166 localized specific populations of salivary acinar cells. Oral Dis. 2012, 18, 162–168. [Google Scholar] [CrossRef]
- Yang, C.; Cao, M.; Liu, H.; He, Y.; Xu, J.; Du, Y.; Liu, Y.; Wang, W.; Cui, L.; Hu, J. The high and low molecular weight forms of hyaluronan have distinct effects on CD44 clustering. J. Biol. Chem. 2012, 287, 43094–43107. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Ryu, J.H.; Do, M.J.; Namkoong, E.; Lee, H.; Park, K. NiCHE platform: Nature-inspired catechol-conjugated hyaluronic acid environment platform for salivary gland tissue engineering. ACS Appl. Mater. Interfaces 2020, 12, 4285–4294. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, P.P.; Patel, V.N.; Liu, S.; Harrington, D.A.; Hoffman, M.P.; Jia, X.; Witt, R.L.; Farach-Carson, M.C.; Pradhan-Bhatt, S. Primary salivary human stem/progenitor cells undergo microenvironment-driven acinar-like differentiation in hyaluronate hydrogel culture. Stem Cells Transl. Med. 2017, 6, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Shubin, D.A.; Felong, T.J.; Schutrum, B.E.; Joe, D.S.; Ovitt, C.E.; Benoit, D.S. Encapsulation of primary salivary gland cells in enzymatically degradable poly (ethylene glycol) hydrogels promotes acinar cell characteristics. Acta Biomater. 2017, 50, 437–449. [Google Scholar] [CrossRef]
- Kerimoglu, O.; Alarçin, E. Poly (lactic-co-glycolic acid) based drug delivery devices for tissue engineering and regenerative medicine. Ankem Derg 2012, 26, 86–98. [Google Scholar] [CrossRef]
- Zhang, -X.B.; Zhang, Z.-L.; Lin, A.L.; Wang, H.; Pilia, M.; Ong, J.L.; Dean, D.D.; Chen, X.-D.; Yeh, C.-K. Silk fibroin scaffolds promote formation of the ex vivo niche for salivary gland epithelial cell growth, matrix formation, and retention of differentiated function. Tissue Eng. Part A 2015, 21, 1611–1620. [Google Scholar] [CrossRef]
- Shin, -S.H.; Kook, Y.-M.; Hong, H.J.; Kim, Y.-M.; Koh, W.-G.; Lim, J.-Y. Functional spheroid organization of human salivary gland cells cultured on hydrogel-micropatterned nanofibrous microwells. Acta Biomater. 2016, 45, 121–132. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pillai, S.; Munguia-Lopez, J.G.; Tran, S.D. Hydrogels for Salivary Gland Tissue Engineering. Gels 2022, 8, 730. https://doi.org/10.3390/gels8110730
Pillai S, Munguia-Lopez JG, Tran SD. Hydrogels for Salivary Gland Tissue Engineering. Gels. 2022; 8(11):730. https://doi.org/10.3390/gels8110730
Chicago/Turabian StylePillai, Sangeeth, Jose G. Munguia-Lopez, and Simon D. Tran. 2022. "Hydrogels for Salivary Gland Tissue Engineering" Gels 8, no. 11: 730. https://doi.org/10.3390/gels8110730
APA StylePillai, S., Munguia-Lopez, J. G., & Tran, S. D. (2022). Hydrogels for Salivary Gland Tissue Engineering. Gels, 8(11), 730. https://doi.org/10.3390/gels8110730