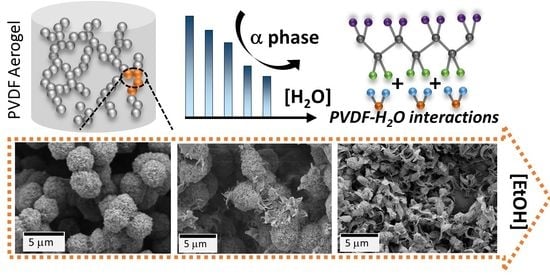
Polyvinylidene Fluoride Aerogels with Tailorable Crystalline Phase Composition
Abstract
1. Introduction
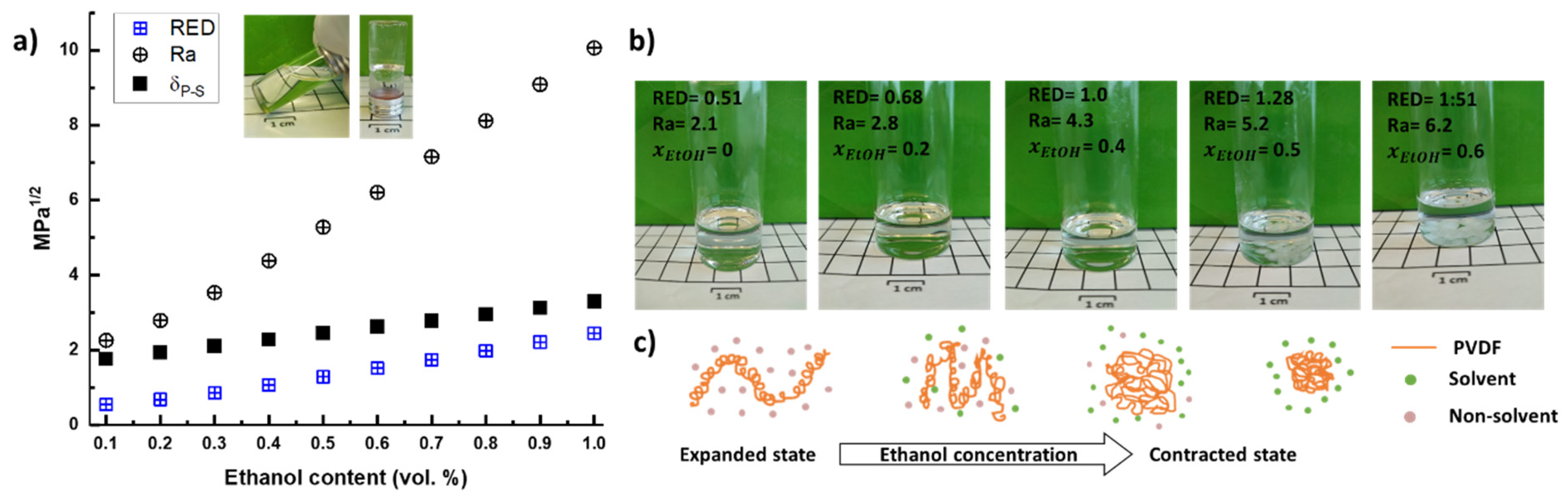
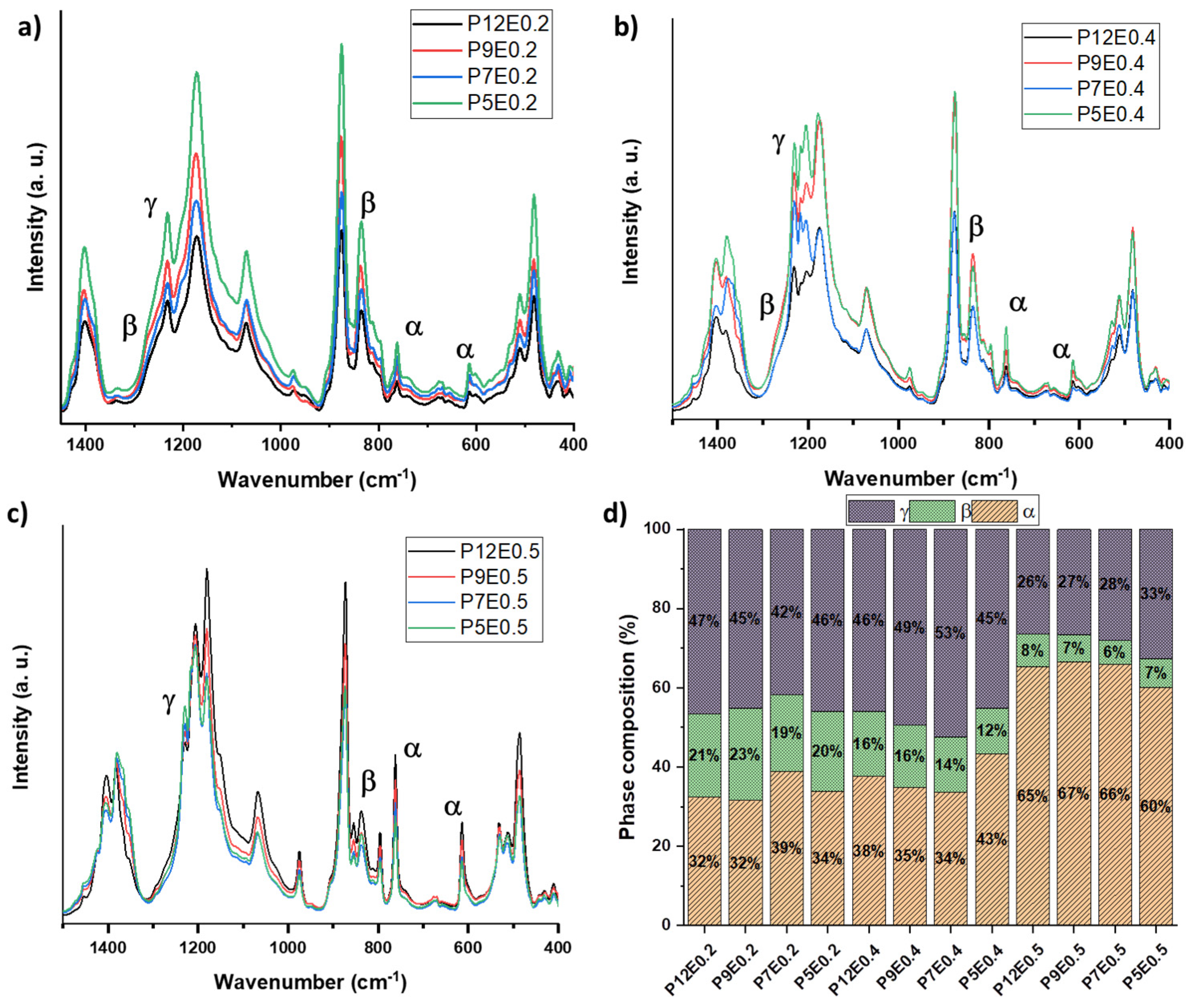
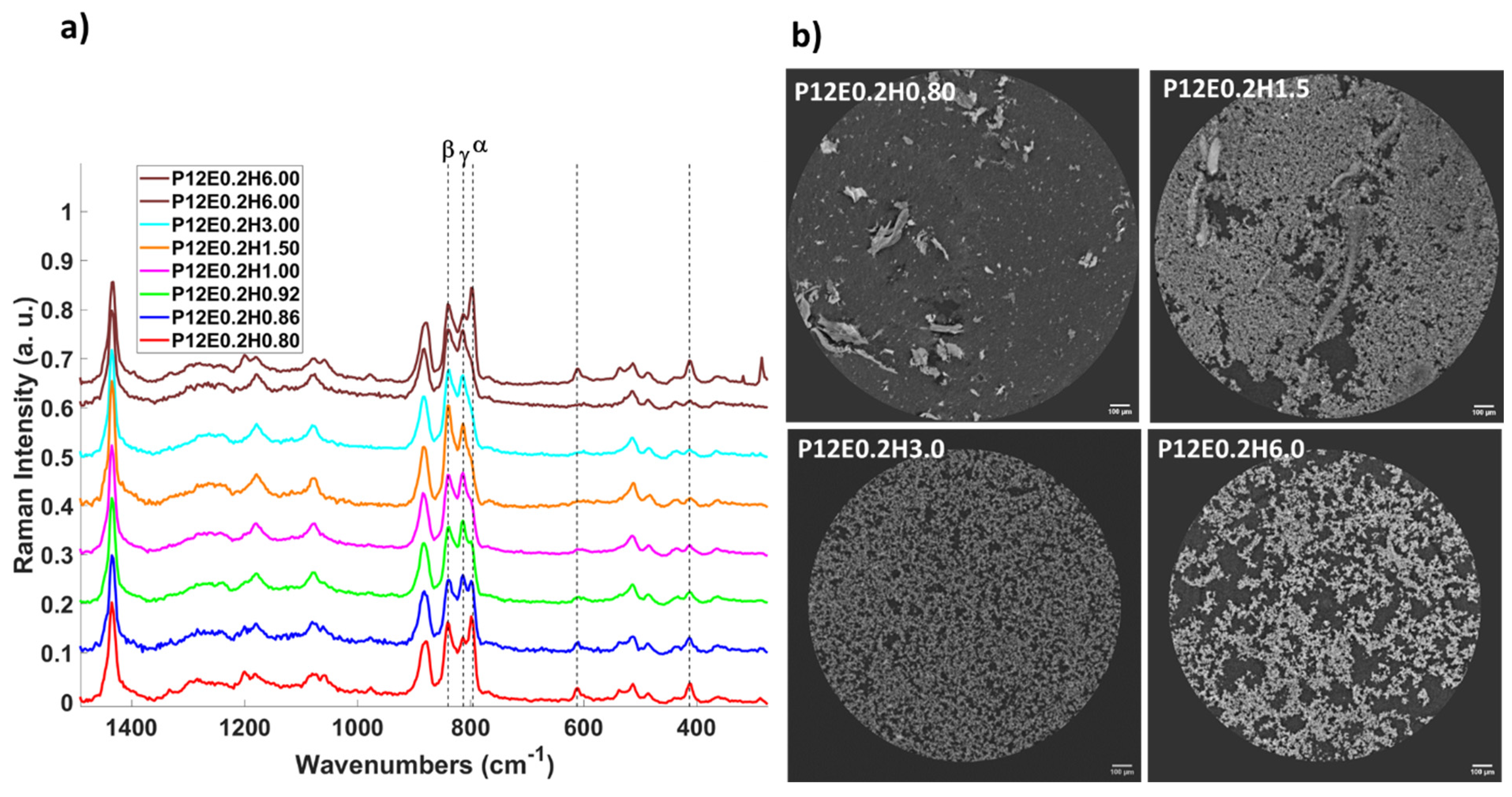
2. Results and Discussion
3. Conclusions
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gregorio, R.; Ueno, E.M. Effect of Crystalline Phase, Orientation and Temperature on the Dielectric Properties of Poly (Vinylidene Fluoride) (PVDF). J. Mater. Sci. 1999, 34, 4489–4500. [Google Scholar] [CrossRef]
- Miao, L.; Liu, G.; Wang, J. Ag-Nanoparticle-Bearing Poly(Vinylidene Fluoride) Nanofiber Mats as Janus Filters for Catalysis and Separation. ACS Appl. Mater. Interfaces 2019, 11, 7397–7404. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yao, C.; Yu, Y.; Hong, Z.; Zhi, M.; Wang, X. Mesoporous Piezoelectric Polymer Composite Films with Tunable Mechanical Modulus for Harvesting Energy from Liquid Pressure Fluctuation. Adv. Funct. Mater. 2016, 26, 6760–6765. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.; Chang, S. PVDF-Based Ferroelectric Polymers and Dielectric Elastomers for Sensor and Actuator Applications: A Review. Funct. Compos. Struct. 2019, 1, 012003. [Google Scholar] [CrossRef]
- Wan, C.; Bowen, C.R. Multiscale-Structuring of Polyvinylidene Fluoride for Energy Harvesting: The Impact of Molecular-, Micro- and Macro-Structure. J. Mater. Chem. A 2017, 5, 3091–3128. [Google Scholar] [CrossRef]
- Soulestin, T.; Ladmiral, V.; Dos Santos, F.D.; Améduri, B. Vinylidene Fluoride- and Trifluoroethylene-Containing Fluorinated Electroactive Copolymers. How Does Chemistry Impact Properties? Prog. Polym. Sci. 2017, 72, 16–60. [Google Scholar] [CrossRef]
- Salimi, A.; Yousefi, A.A. Conformational Changes and Phase Transformation Mechanisms in PVDF Solution-Cast Films. J. Polym. Sci. Part B Polym. Phys. 2004, 42, 3487–3495. [Google Scholar] [CrossRef]
- He, X.; Yao, K. Crystallization Mechanism and Piezoelectric Properties of Solution-Derived Ferroelectric Poly(Vinylidene Fluoride) Thin Films. Appl. Phys. Lett. 2006, 89, 112909. [Google Scholar] [CrossRef]
- Bansil, R.; Lal, J.; Carvalho, B.L. Effects of Gelation on Spinodal Decomposition Kinetics in Gelatin. Polymer 1992, 33, 2961–2969. [Google Scholar] [CrossRef]
- Lovinger, A.J. Ferroelectric Polymers. Science 1983, 220, 1115–1121. [Google Scholar] [CrossRef]
- Wang, H.; Yang, X.; Zhao, Y.; Yan, C.; Wang, S.; Yang, H.; Wang, X.; Schultz, J.M. Preparation of Gamma-PVDF with Controlled Orientation and Insight into Phase Transformation. Polymer 2017, 123, 282–289. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, W.; Tan, B.; Zhu, C.; Ni, Y.; Fang, L.; Lu, C.; Xu, Z. Crystallinity and β Phase Fraction of PVDF in Biaxially Stretched PVDF/PMMA Films. Polymers 2021, 13, 998. [Google Scholar] [CrossRef]
- Sajkiewicz, P.; Wasiak, A.; Goclowski, Z. Phase Transitions during Stretching of Poly(Vinylidene Fluoride). Eur. Polym. J. 1999, 35, 423–429. [Google Scholar] [CrossRef]
- Tang, C.W.; Li, B.; Sun, L.; Lively, B.; Zhong, W.H. The Effects of Nanofillers, Stretching and Recrystallization on Microstructure, Phase Transformation and Dielectric Properties in PVDF Nanocomposites. Eur. Polym. J. 2012, 48, 1062–1072. [Google Scholar] [CrossRef]
- El Mohajir, B.E.; Heymans, N. Changes in Structural and Mechanical Behaviour of PVDF with Processing and Thermomechanical Treatments. 1. Change in Structure. Polymer 2001, 42, 5661–5667. [Google Scholar] [CrossRef]
- Qiao, S.; Zhang, H.; Kang, S.; Quan, J.; Hu, Z.; Yu, J.; Wang, Y.; Zhu, J. Hydrophobic, Pore-Tunable Polyimide/Polyvinylidene Fluoride Composite Aerogels for Effective Airborne Particle Filtration. Macromol. Mater. Eng. 2020, 305, 2000129. [Google Scholar] [CrossRef]
- Pickford, T.; Gu, X.; Heeley, E.L.; Wan, C. Effects of an Ionic Liquid and Processing Conditions on the β-Polymorph Crystal Formation in Poly(Vinylidene Fluoride). CrystEngComm 2019, 21, 5418–5428. [Google Scholar] [CrossRef]
- Ma, W.; Zhang, J.; Chen, S.; Wang, X. Crystalline Phase Formation of Poly(Vinylidene Fluoride) from Tetrahydrofuran/N,N-Dimethylformamide Mixed Solutions. J. Macromol. Sci. Part B Phys. 2008, 47, 434–449. [Google Scholar] [CrossRef]
- Gheorghiu, F.; Stanculescu, R.; Curecheriu, L.; Brunengo, E.; Stagnaro, P.; Tiron, V.; Postolache, P.; Buscaglia, M.T.; Mitoseriu, L. PVDF–Ferrite Composites with Dual Magneto-Piezoelectric Response for Flexible Electronics Applications: Synthesis and Functional Properties. J. Mater. Sci. 2020, 55, 3926–3939. [Google Scholar] [CrossRef]
- Bodkhe, S.; Rajesh, P.S.M.; Kamle, S.; Verma, V. Beta-Phase Enhancement in Polyvinylidene Fluoride through Filler Addition: Comparing Cellulose with Carbon Nanotubes and Clay. J. Polym. Res. 2014, 21, 434. [Google Scholar] [CrossRef]
- Santos, J.P.F.; da Silva, A.B.; Arjmand, M.; Sundararaj, U.; Bretas, R.E.S. Nanofibers of Poly(Vinylidene Fluoride)/Copper Nanowire: Microstructural Analysis and Dielectric Behavior. Eur. Polym. J. 2018, 101, 46–55. [Google Scholar] [CrossRef]
- Zhou, H.; Wang, H.; Liu, Z.; Yang, H.; Yuan, C.; Wang, Y. Facilitated Phase Transformation of PVDF in Its Composite with an Ionic Liquid. Polymer 2021, 220, 123564. [Google Scholar] [CrossRef]
- Zhao, X.; Cheng, J.; Zhang, J.; Chen, S.; Wang, X. Crystallization Behavior of PVDF/PMMA Blends Prepared by in Situ Polymerization from DMF and Ethanol. J. Mater. Sci. 2012, 47, 3720–3728. [Google Scholar] [CrossRef]
- Zhao, X.; Chen, S.; Zhang, J.; Zhang, W.; Wang, X. Crystallization of PVDF in the PVDF/PMMA Blends Precipitated from Their Non-Solvents: Special Orientation Behavior, Morphology, and Thermal Properties. J. Cryst. Growth 2011, 328, 74–80. [Google Scholar] [CrossRef]
- Wei, D.; Zhou, S.; Li, M.; Xue, A.; Zhang, Y.; Zhao, Y.; Zhong, J.; Yang, D. PVDF/Palygorskite Composite Ultrafiltration Membranes: Effects of Nano-Clay Particles on Membrane Structure and Properties. Appl. Clay Sci. 2019, 181, 105171. [Google Scholar] [CrossRef]
- Zhou, Z.; Du, X.; Luo, J.; Yao, L.; Zhang, Z.; Yang, H.; Zhang, Q. Coupling of Interface Effects and Porous Microstructures in Translucent Piezoelectric Composites for Enhanced Energy Harvesting and Sensing. Nano Energy 2021, 84, 105895. [Google Scholar] [CrossRef]
- Cardea, S.; Gugliuzza, A.; Sessa, M.; Aceto, M.C.; Drioli, E.; Reverchon, E. Supercritical Gel Drying: A Powerful Tool for Tailoring Symmetric Porous PVDF-HFP Membranes. ACS Appl. Mater. Interfaces 2009, 1, 171–180. [Google Scholar] [CrossRef]
- Cardea, S.; Sessa, M.; Reverchon, E. Processing of Co-Crystalline and Nanoporous-Crystalline Polymers: Supercritical Co2 Processing of Drug Loaded Membranes Based on Nanoporous PVDF-HFP Aerogels. Soft Mater. 2011, 9, 264–279. [Google Scholar] [CrossRef]
- Cardea, S.; Sessa, M.; Reverchon, E. Supercritical CO2 Assisted Formation of Poly(Vinylidenefluoride) Aerogels Containing Amoxicillin, Used as Controlled Release Device. J. Supercrit. Fluids 2011, 59, 149–156. [Google Scholar] [CrossRef]
- Li, J.; Tenjimbayashi, M.; Zacharia, N.S.; Shiratori, S. One-Step Dipping Fabrication of Fe3O4/PVDF-HFP Composite 3D Porous Sponge for Magnetically Controllable Oil-Water Separation. ACS Sustain. Chem. Eng. 2018, 6, 10706–10713. [Google Scholar] [CrossRef]
- Chen, F.; Lu, Y.; Liu, X.; Song, J.; He, G.; Tiwari, M.K.; Carmalt, C.J.; Parkin, I.P. Table Salt as a Template to Prepare Reusable Porous PVDF–MWCNT Foam for Separation of Immiscible Oils/Organic Solvents and Corrosive Aqueous Solutions. Adv. Funct. Mater. 2017, 27, 1702926. [Google Scholar] [CrossRef]
- Chen, X.; Liang, Y.N.; Tang, X.Z.; Shen, W.; Hu, X. Additive-Free Poly (Vinylidene Fluoride) Aerogel for Oil/Water Separation and Rapid Oil Absorption. Chem. Eng. J. 2017, 308, 18–26. [Google Scholar] [CrossRef]
- Seraji, S.M.; Jin, X.; Yi, Z.; Feng, C.; Salim, N.V. Ultralight Porous Poly (Vinylidene Fluoride)-Graphene Nanocomposites with Compressive Sensing Properties. Nano Res. 2021, 14, 2620–2629. [Google Scholar] [CrossRef]
- Cheraghi Bidsorkhi, H.; D’Aloia, A.G.; Tamburrano, A.; De Bellis, G.; Delfini, A.; Ballirano, P.; Sarto, M.S. 3D Porous Graphene Based Aerogel for Electromagnetic Applications. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Zhang, Y.; Yu, Z.; Zheng, J.; Wang, Y.; Zhou, H. PANI/PVDF-TrFE Porous Aerogel Bulk Piezoelectric and Triboelectric Hybrid Nanogenerator Based on in-Situ Doping and Liquid Nitrogen Quenching. Nano Energy 2021, 80, 105519. [Google Scholar] [CrossRef]
- Zhang, J.; Kong, Y.; Shen, X. Polyvinylidene Fluoride Aerogel with High Thermal Stability and Low Thermal Conductivity. Mater. Lett. 2020, 259, 126890. [Google Scholar] [CrossRef]
- Kim, S.J.; Raut, P.; Jana, S.C.; Chase, G. Electrostatically Active Polymer Hybrid Aerogels for Airborne Nanoparticle Filtration. ACS Appl. Mater. Interfaces 2017, 9, 6401–6410. [Google Scholar] [CrossRef]
- Ewa Piorkowska, G.C.R. Handbook of Polymer Crystallization; Piorkowska, E., Rutledge, G.C., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; ISBN 9781118541838. [Google Scholar]
- Cheng, L.P.; Young, T.H.; Fang, L.; Gau, J.J. Formation of Particulate Microporous Poly(Vinylidene Fluoride) Membranes by Isothermal Immersion Precipitation from the 1-Octanol/Dimethylformamide/Poly(Vinylidene Fluoride) System. Polymer 1999, 40, 2395–2403. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of Gases, with Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Cai, X.; Lei, T.; Sun, D.; Lin, L. A Critical Analysis of the α, β and γ Phases in Poly(Vinylidene Fluoride) Using FTIR. RSC Adv. 2017, 7, 15382–15389. [Google Scholar] [CrossRef]
- Tanigami, T.; Suzuki, H.; Yamaura, K.; Matsuzawa, S. Gelation and Crystallization of Poly (4-Methyl-1-Pentene) in Cyclohexane Solution. Macromolecules 1985, 18, 2595–2600. [Google Scholar] [CrossRef]
- Coniglio, A.; Stanley, H.E.; Klein, W. Site-Bond Correlated-Percolation Problem: A Statistical Mechanical Model of Polymer Gelation. Phys. Rev. Lett. 1979, 42, 518–522. [Google Scholar] [CrossRef]
- Díaz de los Ríos, M.; Hernández Ramos, E. Determination of the Hansen Solubility Parameters and the Hansen Sphere Radius with the Aid of the Solver Add-in of Microsoft Excel. SN Appl. Sci. 2020, 2, 676. [Google Scholar] [CrossRef]
- Charles, H. Hansen Solubility Parameters: A User´s Handbook, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Bottino, A.; Capannelli, G.; Munari, S.; Turturro, A. Solubility Parameters of Poly(Vinylidene Fluoride). J. Polym. Sci. Part B Polym. Phys. 1988, 26, 785–794. [Google Scholar] [CrossRef]
- Novo, L.P.; Curvelo, A.A.S. Hansen Solubility Parameters: A Tool for Solvent Selection for Organosolv Delignification. Ind. Eng. Chem. Res. 2019, 58, 14520–14527. [Google Scholar] [CrossRef]
- Zhao, X.; Cheng, J.; Chen, S.; Zhang, J.; Wang, X. Controlled Crystallization of Poly(Vinylidene Fluoride) Chains from Mixed Solvents Composed of Its Good Solvent and Nonsolvent. J. Polym. Sci. Part B Polym. Phys. 2010, 48, 575–581. [Google Scholar] [CrossRef]
- Bower, D. An Introduction to Polymer Physics; Cambridge University Press: Cambrigde, UK, 2002. [Google Scholar]
- Wu, Y.; Hsu, S.L.; Honeker, C.; Bravet, D.J.; Williams, D.S. The Role of Surface Charge of Nucleation Agents on the Crystallization Behavior of Poly(Vinylidene Fluoride). J. Phys. Chem. B 2012, 116, 7379–7388. [Google Scholar] [CrossRef]
- Guillen, G.R.; Pan, Y.; Li, M.; Hoek, E.M.V. Preparation and Characterization of Membranes Formed by Nonsolvent Induced Phase Separation: A Review. Ind. Eng. Chem. Res. 2011, 50, 3798–3817. [Google Scholar] [CrossRef]
- Li, R.; Chen, C.; Li, J.; Xu, L.; Xiao, G.; Yan, D. A Facile Approach to Superhydrophobic and Superoleophilic Graphene/Polymer Aerogels. J. Mater. Chem. A 2014, 2, 3057–3064. [Google Scholar] [CrossRef]
- Jungnickel, J.B. Polymeric Materials Handbook; CRC Press Inc: New York, NY, USA, 1996; Volume 9. [Google Scholar]
- Shepelin, N.A.; Glushenkov, A.M.; Lussini, V.C.; Fox, P.J.; Dicinoski, G.W.; Shapter, J.G.; Ellis, A.V. New Developments in Composites, Copolymer Technologies and Processing Techniques for Flexible Fluoropolymer Piezoelectric Generators for Efficient Energy Harvesting. Energy Environ. Sci. 2019, 12, 1143–1176. [Google Scholar] [CrossRef]
- Pradhan, S.K.; Kumar, A.; Sinha, A.N.; Kour, P.; Pandey, R.; Kumar, P.; Kar, M. Study of Ferroelectric Properties on PVDF-PZT Nanocomposite. Ferroelectrics 2017, 516, 18–27. [Google Scholar] [CrossRef]
- Xu, F.; Zhang, K.; Zhou, Y.; Qu, Z.; Wang, H.; Zhang, Y.; Zhou, H.; Yan, C. Facile Preparation of Highly Oriented Poly(Vinylidene Fluoride) Uniform Films and Their Ferro- and Piezoelectric Properties. RSC Adv. 2017, 7, 17038–17043. [Google Scholar] [CrossRef]
- Boryczka, S.; Jastrzebska, M.; Bębenek, E.; Kusz, J.; Zubko, M.; Kadela, M.; Michalik, E. X-Ray Diffraction and Infrared Spectroscopy of N,N- Dimethylformamide and Dimethyl Sulfoxide Solvatomorphs of Betulonic Acid. J. Pharm. Sci. 2012, 101, 4458–4471. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Tashiro, K.; Tadokoro, H. Molecular Vibrations of Three Crystal Forms of Poly(Vinylidene Fluoride). Macromolecules 1975, 8, 158–171. [Google Scholar] [CrossRef]
- Tashiro, K.; Kobayashi, M.; Tadokoro, H. Vibrational Spectra and Disorder-Order Transition of Poly(Vinylidene Fluoride) Form III. Macromolecules 1981, 14, 1757–1764. [Google Scholar] [CrossRef]
- Tashiro, K.; Kobayashi, M. Structural Phase Transition in Ferroelectric Fluorine Polymers: X-Ray Diffraction and Infrared/Raman Spectroscopic Study. Phase Transit. 1989, 18, 213–246. [Google Scholar] [CrossRef]
- Dhevi, D.M.; Prabu, A.A.; Kim, K.J. Infrared Spectroscopic Studies on Crystalline Phase Transition of PVDF and PVDF/Hyperbranched Polyester Blend Ultrathin Films. Vib. Spectrosc. 2018, 94, 74–82. [Google Scholar] [CrossRef]
- Liu, Y.; Aziguli, H.; Zhang, B.; Xu, W.; Lu, W.; Bernholc, J.; Wang, Q. Ferroelectric Polymers Exhibiting Behaviour Reminiscent of a Morphotropic Phase Boundary. Nature 2018, 562, 96–100. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Tromba, G.; Longo, R.; Abrami, A.; Arfelli, F.; Astolfo, A.; Bregant, P.; Brun, F.; Casarin, K.; Chenda, V.; Dreossi, D.; et al. The SYRMEP Beamline of Elettra: Clinical Mammography and Bio-Medical Applications. In AIP Conference Proceedings; American institute of Physics: College Park, MD, USA, 2010; pp. 18–23. [Google Scholar]
- Brun, F.; Massimi, L.; Fratini, M.; Dreossi, D.; Billé, F.; Accardo, A.; Pugliese, R.; Cedola, A. SYRMEP Tomo Project: A Graphical User Interface for Customizing CT Reconstruction Workflows. Adv. Struct. Chem. Imaging 2017, 3, 4. [Google Scholar] [CrossRef]
- Paganin, D.; Mayo, S.C.; Gureyev, T.E.; Miller, P.R.; Wilkins, S.W. Simultaneous Phase and Amplitude Extraction from a Single Defocused Image of a Homogeneous Object. J. Microsc. 2002, 206, 33–40. [Google Scholar] [CrossRef]
- D’Amico, F.; Saito, M.; Bencivenga, F.; Marsi, M.; Gessini, A.; Camisasca, G.; Principi, E.; Cucini, R.; Di Fonzo, S.; Battistoni, A.; et al. UV Resonant Raman Scattering Facility at Elettra. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrometers Detect. Assoc. Equip. 2013, 703, 33–37. [Google Scholar] [CrossRef]
- Birarda, G.; Bedolla, D.; Piccirilli, F.; Stani, C.; Vondracek, H.; Vaccari, L. Chemical Analyses at Micro and Nano Scale at SISSI-Bio Beamline at Elettra-Sincrotrone Trieste. In Proceedings of the Biomedical Vibrational Spectroscopy 2022: Advances in Research and Industry, San Francisco, CA, USA, 30 March 2022; Huang, Z., Ed.; SPIE: Bellingham, WA, USA, 2022; p. 31. [Google Scholar]
- Gregorio, R., Jr.; Cestari, M. Effect of Crystallization Temperature on the Crystalline Phase Content and Morphology of Poly(Vinylidene Fluoride). J. Polym. Sci. Part B Polym. Phys. 1994, 32, 859–870. [Google Scholar] [CrossRef]



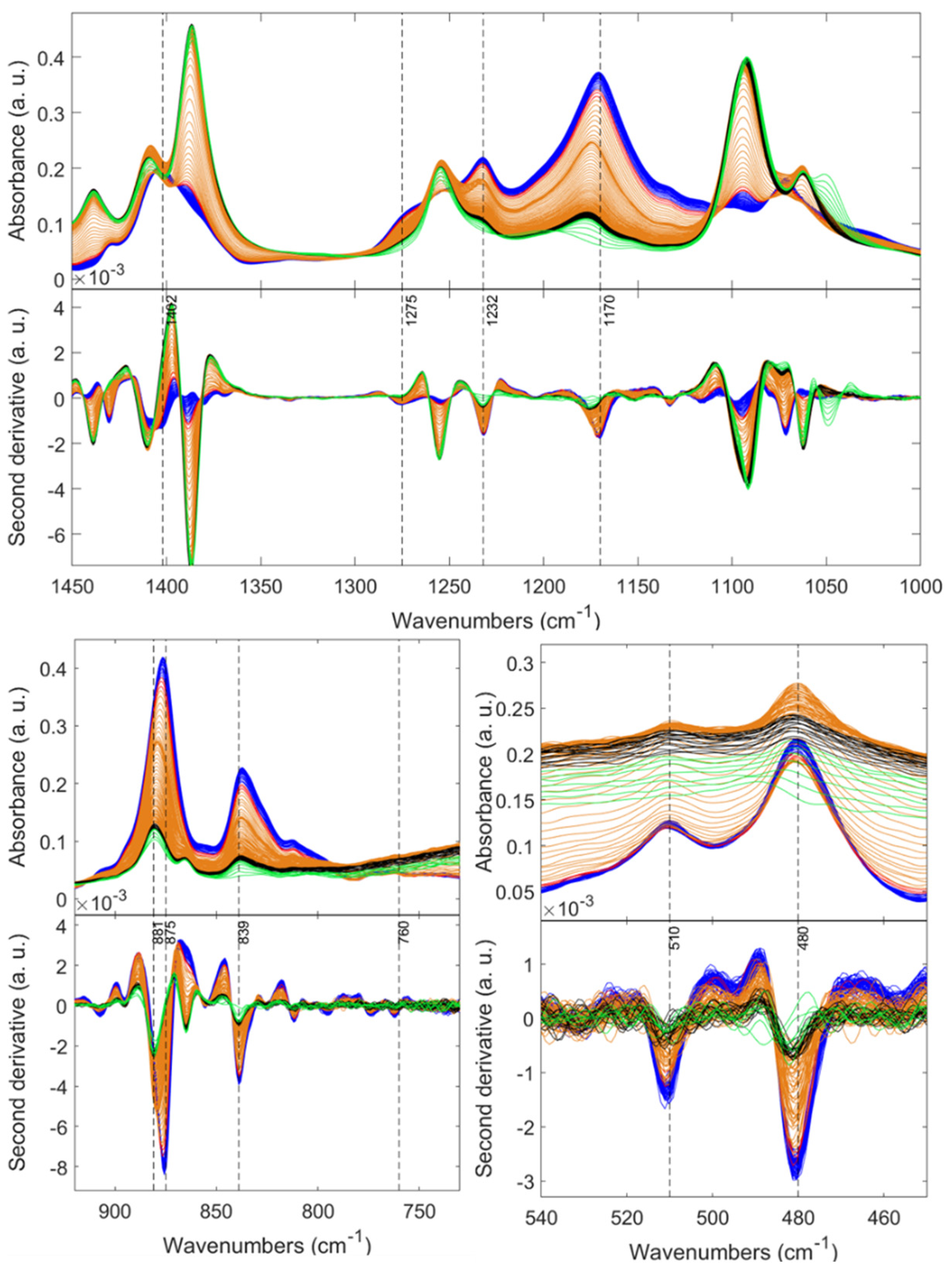
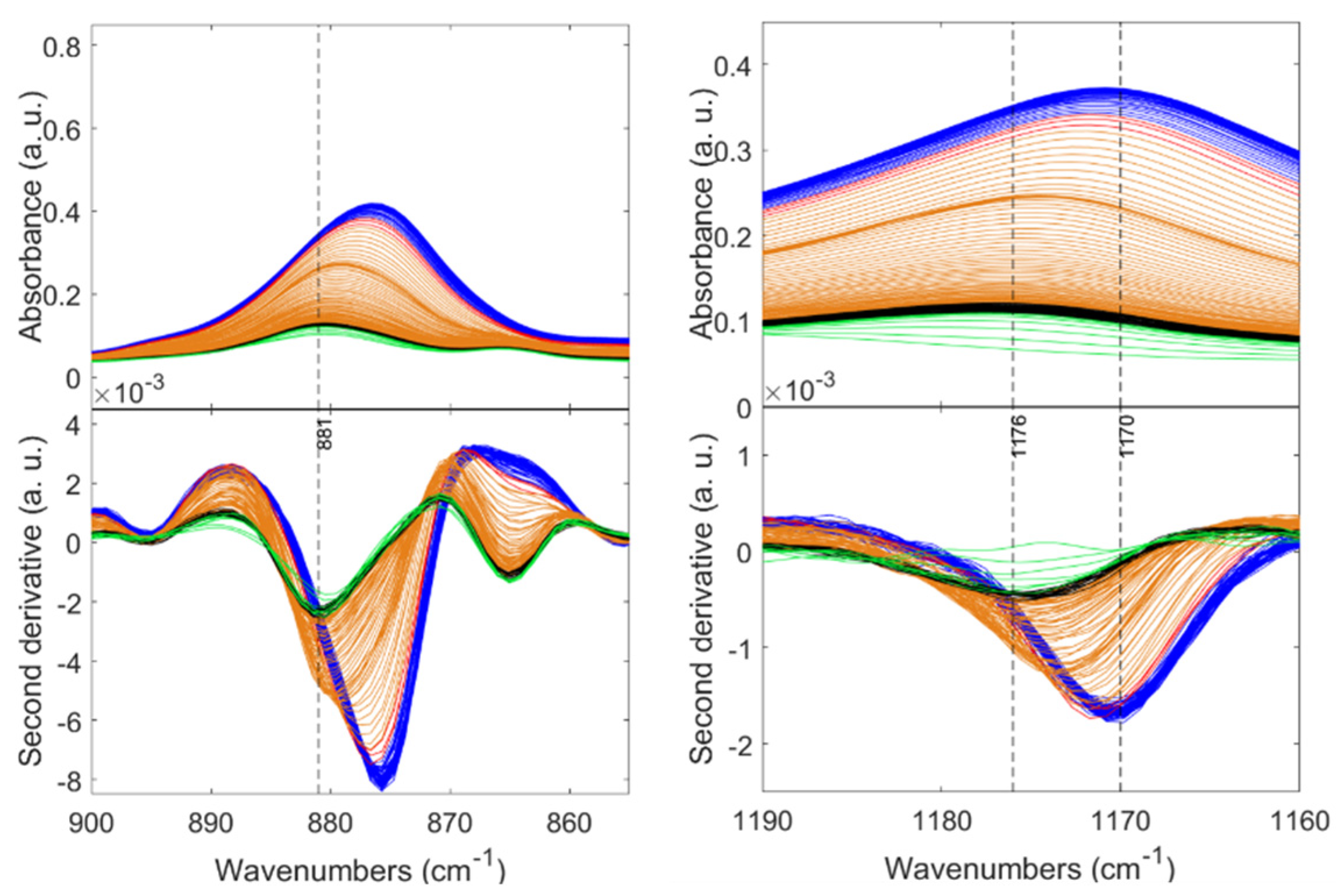
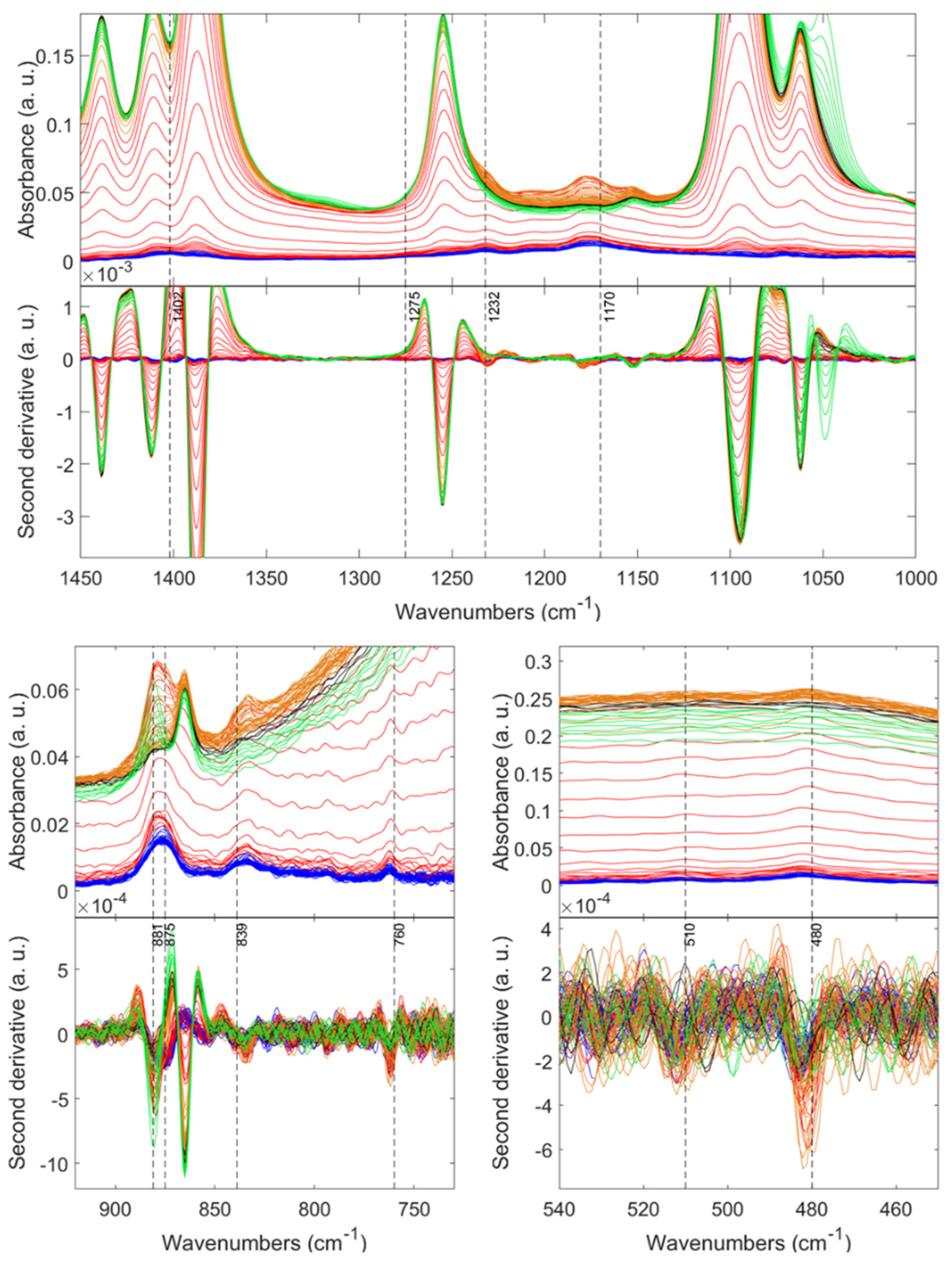
| Sample | SBET (m2 g−1) a | Pore Diameter (nm) b | Pore Volume (cm3 g−1) c | tgel min d | (g cm−3) e | (g cm−3) f | Porosity (%) |
|---|---|---|---|---|---|---|---|
| P12 | 145 | 23 | 0.80 | 30 | 0.123 | 1.47 | 92 |
| P9 | 168 | 23 | 0.99 | 45 | 0.093 | 1.39 | 93 |
| P7 | - | - | - | No gel | - | - | - |
| P5 | - | - | - | No gel | - | - | - |
| P3 | - | - | - | No gel | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres-Rodriguez, J.; E. Bedolla, D.; D’Amico, F.; Koopmann, A.-K.; Vaccari, L.; Saccomano, G.; Kohns, R.; Huesing, N. Polyvinylidene Fluoride Aerogels with Tailorable Crystalline Phase Composition. Gels 2022, 8, 727. https://doi.org/10.3390/gels8110727
Torres-Rodriguez J, E. Bedolla D, D’Amico F, Koopmann A-K, Vaccari L, Saccomano G, Kohns R, Huesing N. Polyvinylidene Fluoride Aerogels with Tailorable Crystalline Phase Composition. Gels. 2022; 8(11):727. https://doi.org/10.3390/gels8110727
Chicago/Turabian StyleTorres-Rodriguez, Jorge, Diana E. Bedolla, Francesco D’Amico, Ann-Kathrin Koopmann, Lisa Vaccari, Giulia Saccomano, Richard Kohns, and Nicola Huesing. 2022. "Polyvinylidene Fluoride Aerogels with Tailorable Crystalline Phase Composition" Gels 8, no. 11: 727. https://doi.org/10.3390/gels8110727
APA StyleTorres-Rodriguez, J., E. Bedolla, D., D’Amico, F., Koopmann, A.-K., Vaccari, L., Saccomano, G., Kohns, R., & Huesing, N. (2022). Polyvinylidene Fluoride Aerogels with Tailorable Crystalline Phase Composition. Gels, 8(11), 727. https://doi.org/10.3390/gels8110727