Using Aggregation-Induced Emission to Understand Dipeptide Gels
Abstract
:1. Introduction
2. Results and Discussion
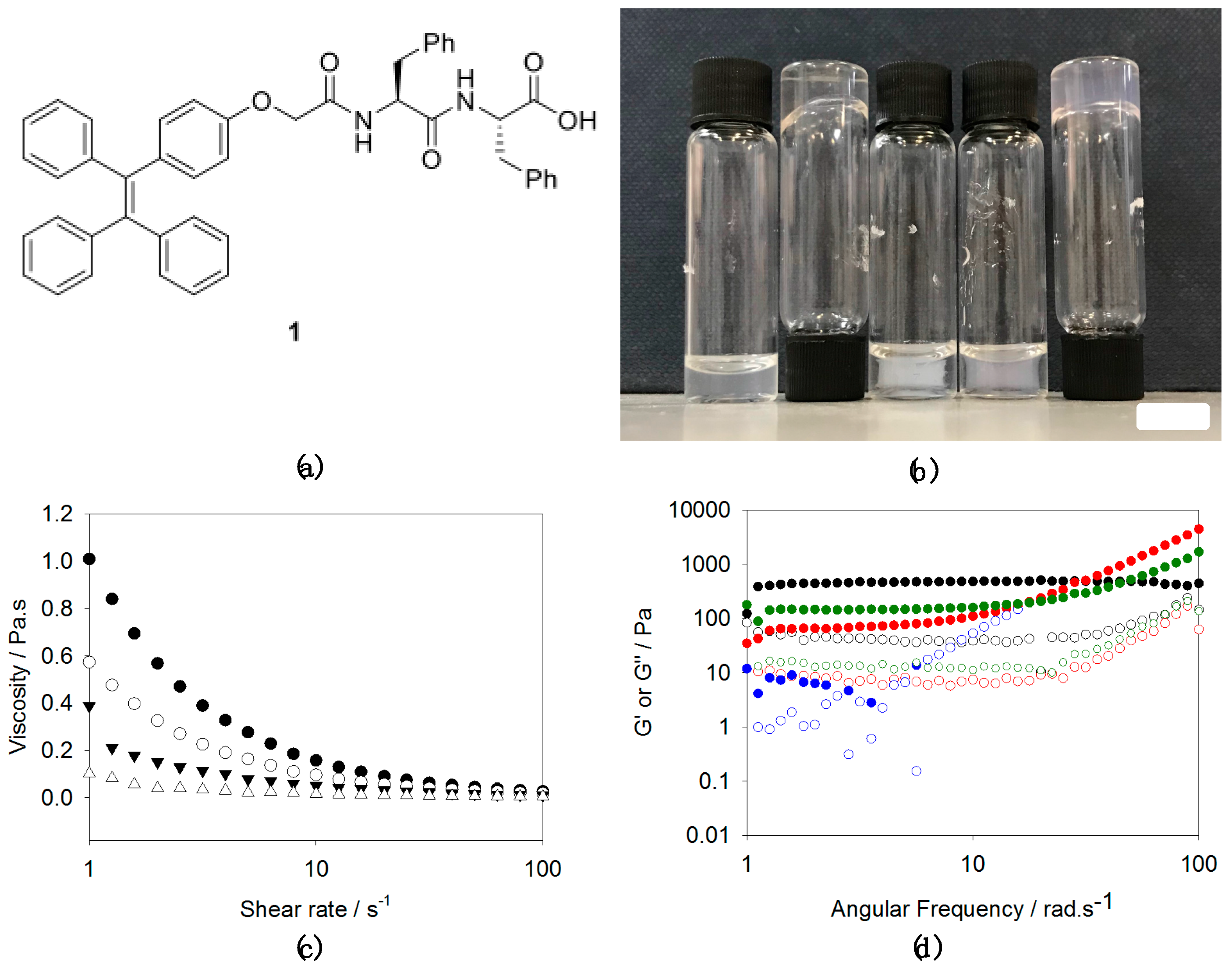
2.1. Gelation Studies
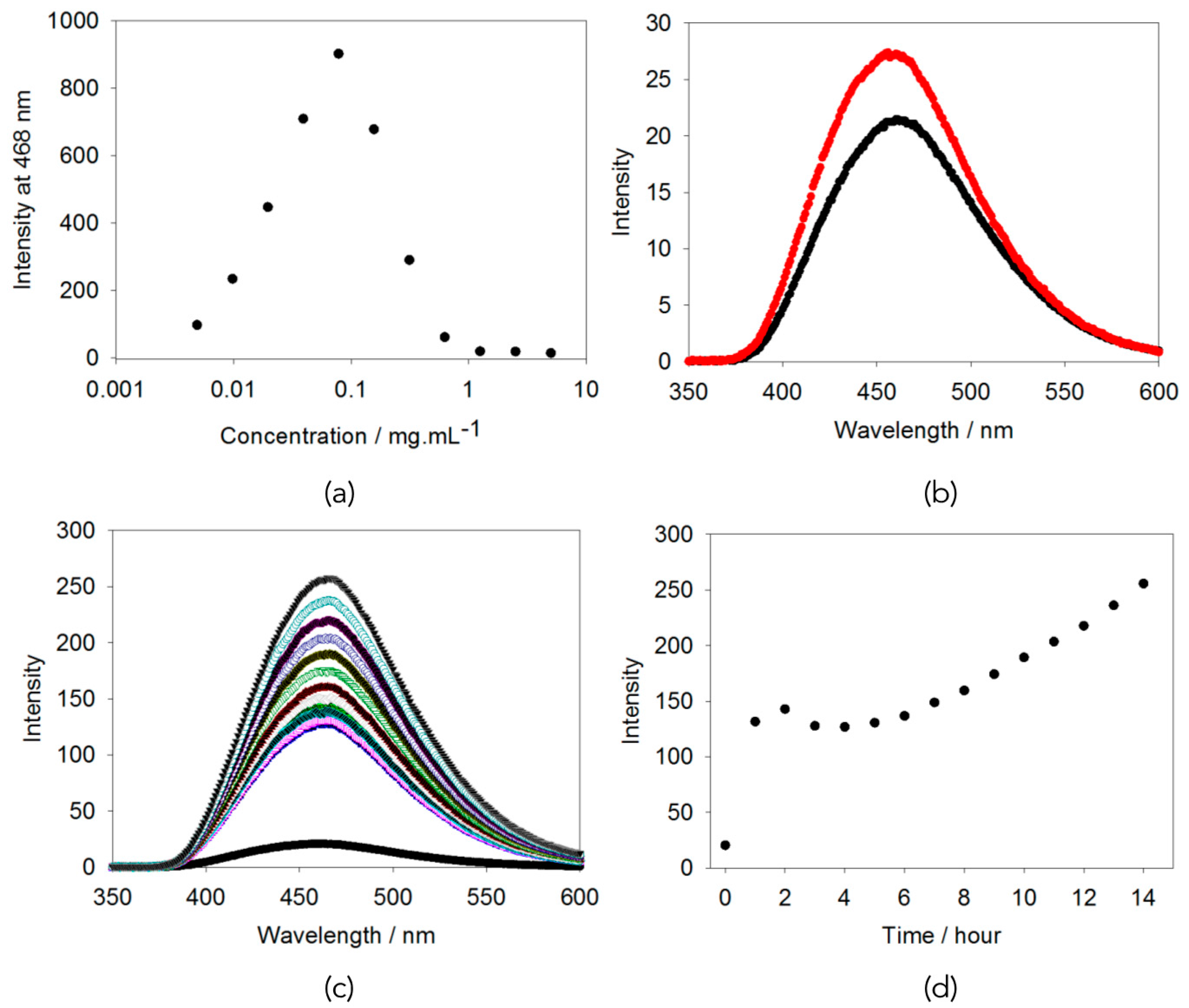
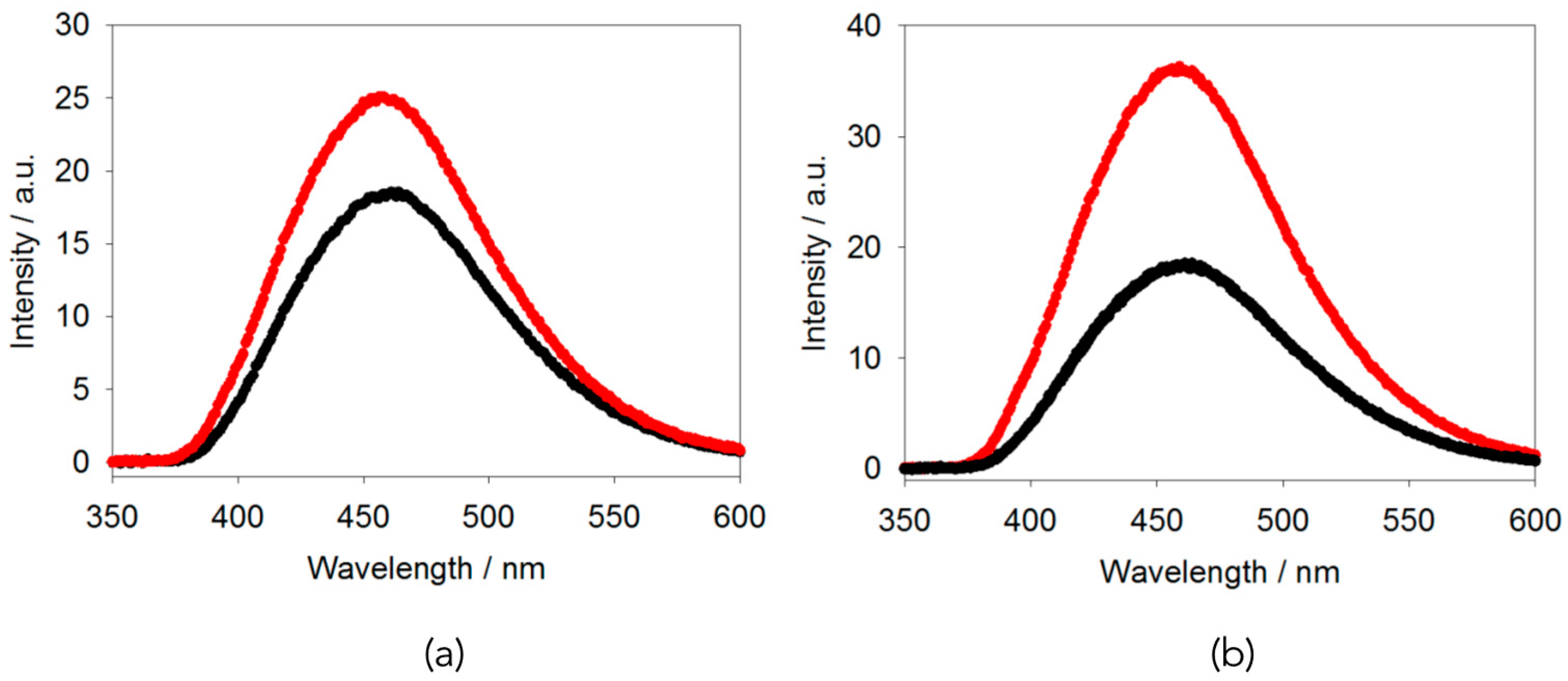
2.2. AIE Studies
3. Conclusions
4. Materials and Methods
4.1. Preparation of Solutions
4.2. Acid-Triggered Gels
4.3. Calcium-Triggered Gels
4.4. Salt-Triggered Gels
4.5. Photographs
4.6. Rheology
4.7. Fluorescence Spectroscopy
4.8. UV-Vis Spectroscopy
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Du, X.; Zhou, J.; Shi, J.; Xu, B. Supramolecular Hydrogelators and Hydrogels: From Soft Matter to Molecular Biomaterials. Chem. Rev. 2015, 115, 13165–13307. [Google Scholar] [CrossRef] [PubMed]
- Draper, E.R.; Adams, D.J. Low-Molecular-Weight Gels: The State of the Art. Chem 2017, 3, 390–410. [Google Scholar] [CrossRef]
- Weiss, R.G. The Past, Present, and Future of Molecular Gels. What Is the Status of the Field, and Where Is It Going? J. Am. Chem. Soc. 2014, 136, 7519–7530. [Google Scholar] [CrossRef] [PubMed]
- Colquhoun, C.; Draper, E.R.; Schweins, R.; Marcello, M.; Vadukul, D.; Serpell, L.C.; Adams, D.J. Controlling the network type in self-assembled dipeptide hydrogels. Soft Matter 2017, 13, 1914–1919. [Google Scholar] [CrossRef] [PubMed]
- Mahler, A.; Reches, M.; Rechter, M.; Cohen, S.; Gazit, E. Rigid, Self-Assembled Hydrogel Composed of a Modified Aromatic Dipeptide. Adv. Mater. 2006, 18, 1365–1370. [Google Scholar] [CrossRef]
- Dudukovic, N.A.; Zukoski, C.F. Gelation of Fmoc-diphenylalanine is a first order phase transition. Soft Matter 2015, 11, 7663–7673. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.M.; Williams, R.J.; Tang, C.; Coppo, P.; Collins, R.F.; Turner, M.L.; Saiani, A.; Ulijn, R.V. Fmoc-Diphenylalanine Self Assembles to a Hydrogel via a Novel Architecture Based on π–π Interlocked β-Sheets. Adv. Mater. 2008, 20, 37–41. [Google Scholar] [CrossRef]
- Eckes, K.M.; Mu, X.; Ruehle, M.A.; Ren, P.; Suggs, L.J. β Sheets Not Required: Combined Experimental and Computational Studies of Self-Assembly and Gelation of the Ester-Containing Analogue of an Fmoc-Dipeptide Hydrogelator. Langmuir 2014, 30, 5287–5296. [Google Scholar] [CrossRef] [PubMed]
- Braun, H.-G.; Cardoso, A.Z. Self-assembly of Fmoc-diphenylalanine inside liquid marbles. Coll. Surf. B 2012, 97, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Mears, L.L.E.; Draper, E.R.; Castilla, A.M.; Su, H.; Dietrich, B.Z.; Nolan, M.C.; Smith, G.N.; Doutch, J.; Rogers, S.; Akhtar, R.; et al. Drying Affects the Fiber Network in Low Molecular Weight Hydrogels. Biomacromolecules 2017, 18, 3531–3540. [Google Scholar] [CrossRef] [PubMed]
- Kwok, R.T.K.; Leung, C.W.T.; Lam, J.W.Y.; Tang, B.Z. Biosensing by luminogens with aggregation-induced emission characteristics. Chem. Soc. Rev. 2015, 44, 4228–4238. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Leung, N.L.C.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. Aggregation-Induced Emission: Together We Shine, United We Soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Zhang, G.; Zhang, D. A new gelator based on tetraphenylethylene and diphenylalanine: Gel formation and reversible fluorescence tuning. Chin. Sci. Bull. 2012, 57, 4284–4288. [Google Scholar] [CrossRef]
- Yeh, M.-Y.; Huang, C.-W.; Chang, J.-W.; Huang, Y.-T.; Lin, J.-H.; Hsu, S.-M.; Hung, S.-C.; Lin, H.-C. A novel nanostructured supramolecular hydrogel self-assembled from tetraphenylethylene-capped dipeptides. Soft Matter 2016, 12, 6347–6351. [Google Scholar] [CrossRef] [PubMed]
- Neupane, L.N.; Oh, E.-T.; Park, H.J.; Lee, K.-H. Selective and Sensitive Detection of Heavy Metal Ions in 100% Aqueous Solution and Cells with a Fluorescence Chemosensor Based on Peptide Using Aggregation-Induced Emission. Anal. Chem. 2016, 88, 3333–3340. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Liu, C.; Xue, X.; Zhang, X.; Huo, S.; Jiang, Y.; Chen, W.-Q.; Zou, G.; Liang, X.-J. Salt-Responsive Self-Assembly of Luminescent Hydrogel with Intrinsic Gelation-Enhanced Emission. ACS Appl. Mater. Interfaces 2014, 6, 757–762. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, Y.; Xue, X.; Chu, P.; Liu, C.; Yang, K.; Jiang, Y.; Chen, W.-Q.; Zou, G.; Liang, X.-J. A smart pH-switchable luminescent hydrogel. Chem. Commun. 2015, 51, 4168–4171. [Google Scholar] [CrossRef] [PubMed]
- Draper, E.R.; Su, H.; Brasnett, C.; Poole, R.J.; Rogers, S.; Cui, H.; Seddon, A.; Adams, D.J. Opening a Can of Worm(-like Micelle)s: The Effect of Temperature of Solutions of Functionalized Dipeptides. Angew. Chem. Int. Ed. 2017, 56, 10467–10470. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, A.Z.; Mears, L.L.E.; Cattoz, B.N.; Griffiths, P.C.; Schweins, R.; Adams, D.J. Linking micellar structures to hydrogelation for salt-triggered dipeptide gelators. Soft Matter 2016, 12, 3612–3621. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.J.; Butler, M.F.; Frith, W.J.; Kirkland, M.; Mullen, L.; Sanderson, P. A new method for maintaining homogeneity during liquid-hydrogel transitions using low molecular weight hydrogelators. Soft Matter 2009, 5, 1856–1862. [Google Scholar] [CrossRef]
- Pocker, Y.; Green, E. Hydrolysis of D-glucono-.delta.-lactone. I. General acid-base catalysis, solvent deuterium isotope effects, and transition state characterization. J. Am. Chem. Soc. 1973, 95, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Castilla, A.M.; Wallace, M.; Mears, L.L.E.; Draper, E.R.; Doutch, J.; Rogers, S.; Adams, D.J. On the syneresis of an OPV functionalised dipeptide hydrogel. Soft Matter 2016, 12, 7848–7854. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castilla, A.M.; Dietrich, B.; Adams, D.J. Using Aggregation-Induced Emission to Understand Dipeptide Gels. Gels 2018, 4, 17. https://doi.org/10.3390/gels4010017
Castilla AM, Dietrich B, Adams DJ. Using Aggregation-Induced Emission to Understand Dipeptide Gels. Gels. 2018; 4(1):17. https://doi.org/10.3390/gels4010017
Chicago/Turabian StyleCastilla, Ana M., Bart Dietrich, and Dave J. Adams. 2018. "Using Aggregation-Induced Emission to Understand Dipeptide Gels" Gels 4, no. 1: 17. https://doi.org/10.3390/gels4010017
APA StyleCastilla, A. M., Dietrich, B., & Adams, D. J. (2018). Using Aggregation-Induced Emission to Understand Dipeptide Gels. Gels, 4(1), 17. https://doi.org/10.3390/gels4010017






