Enhancing Rheology and Wettability of Drilling Fluids at Ultra-Low Temperatures Using a Novel Amide Material
Abstract
1. Introduction
2. Results and Discussion
2.1. Structural Characterization
2.1.1. Infrared Spectrum
2.1.2. Nuclear Magnetic Hydrogen Spectrum
2.2. Performance Evaluation
2.2.1. Rheological Properties of HAS
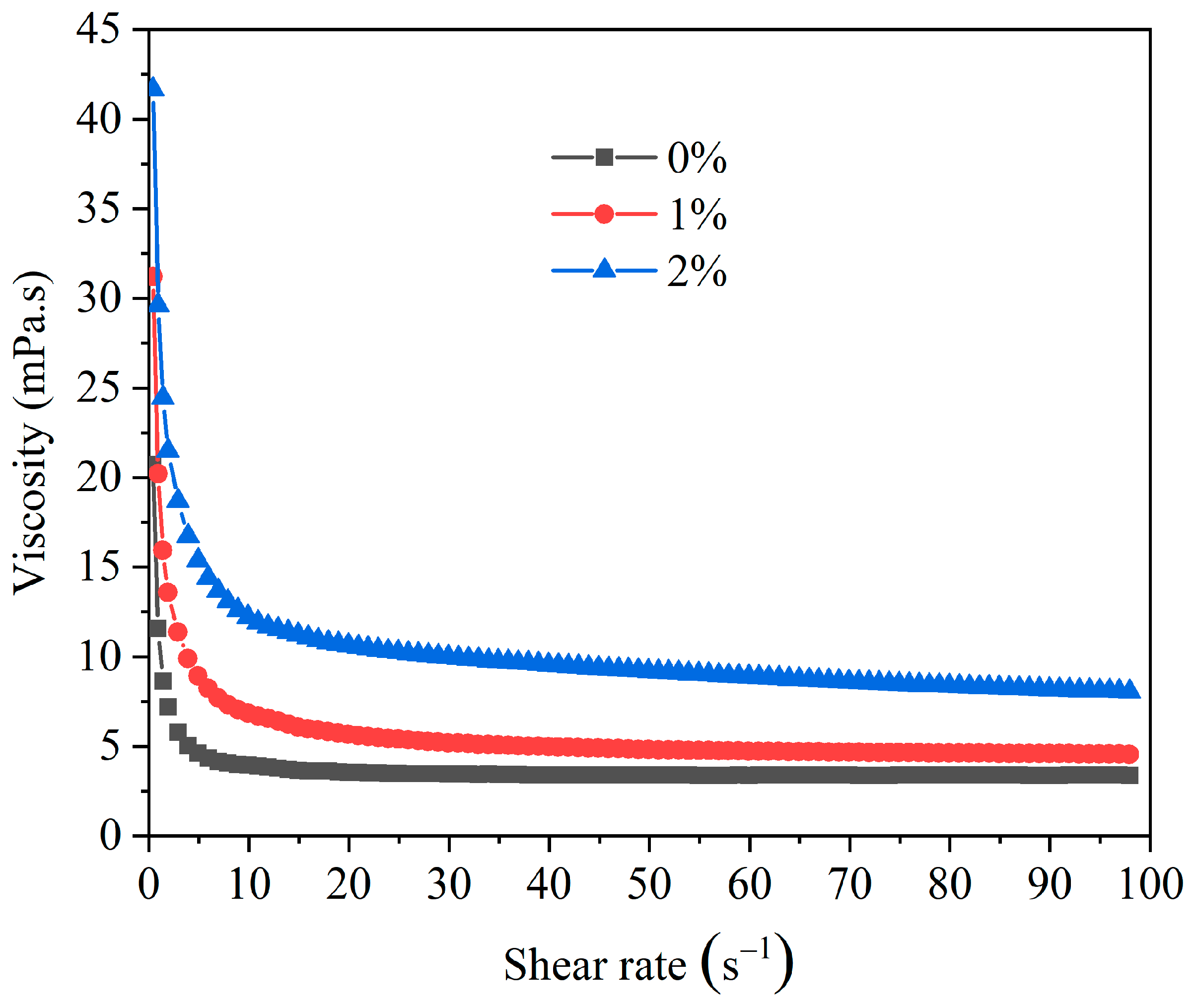
The Impact of HAS on the Viscosity and Yield Point for Drilling Fluid
The Impact of HAS on the Shear Dilution of Drilling Fluid
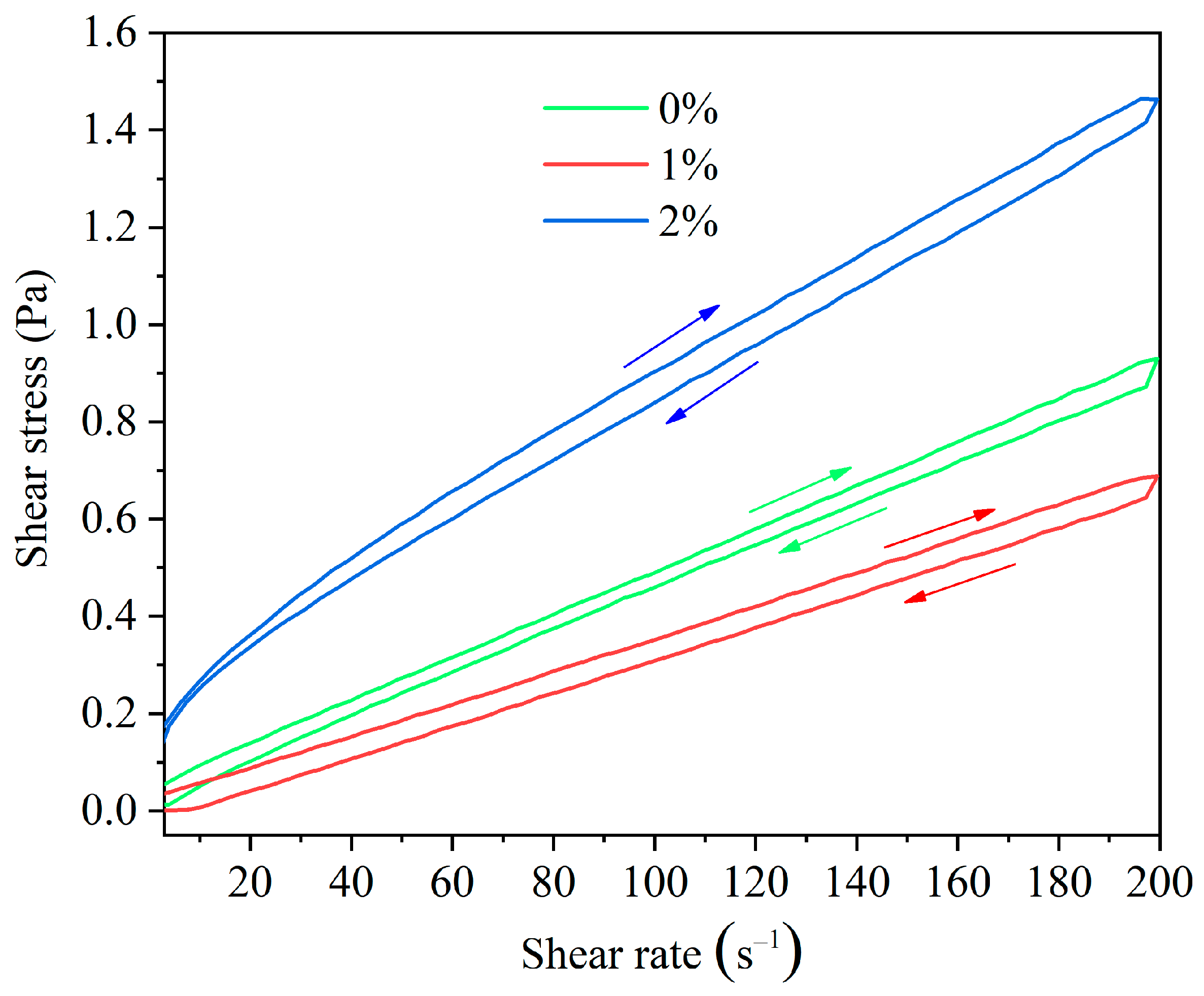
The Impact of HAS on the Thixotropy
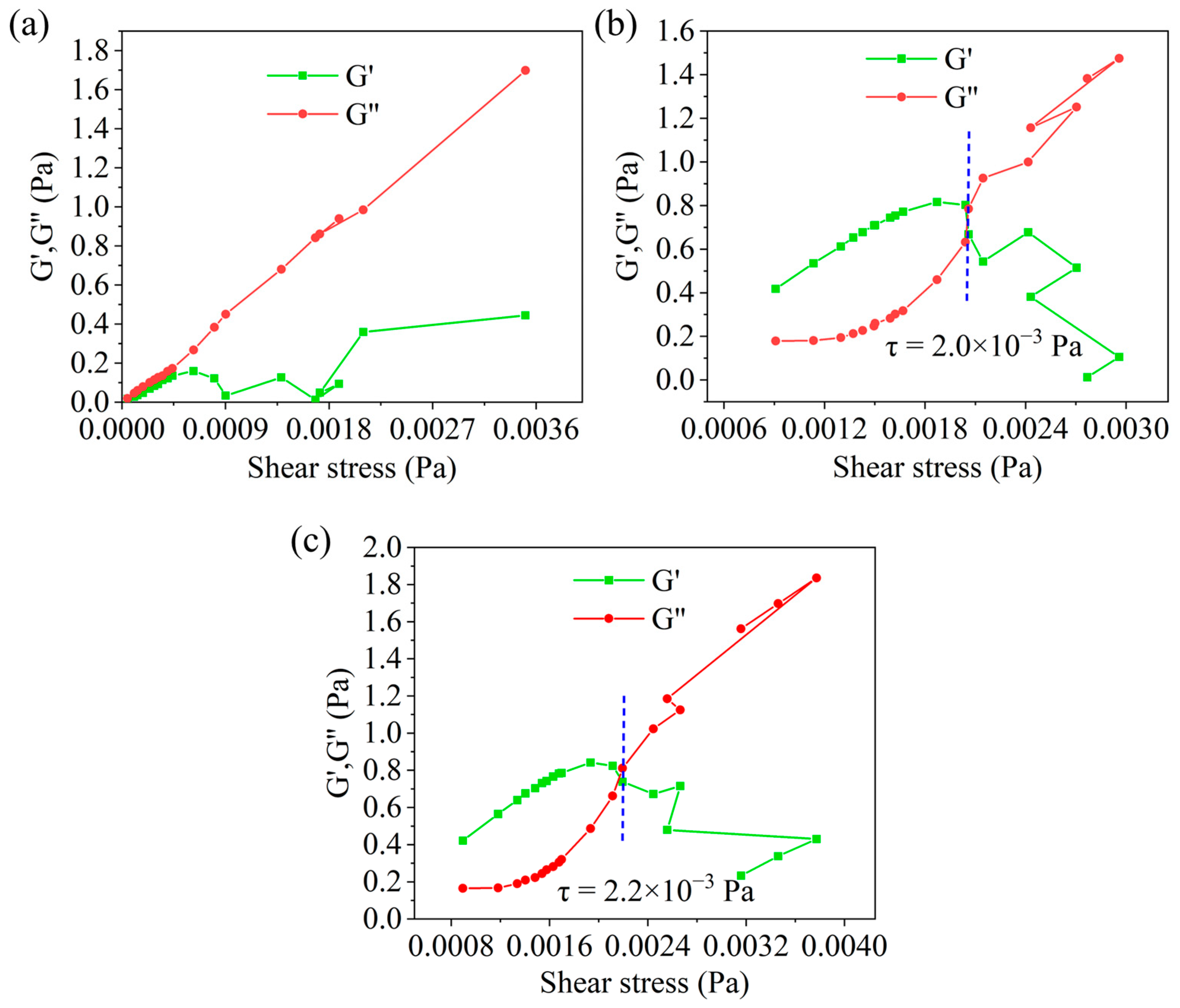
The Effect of HAS on the Modulus
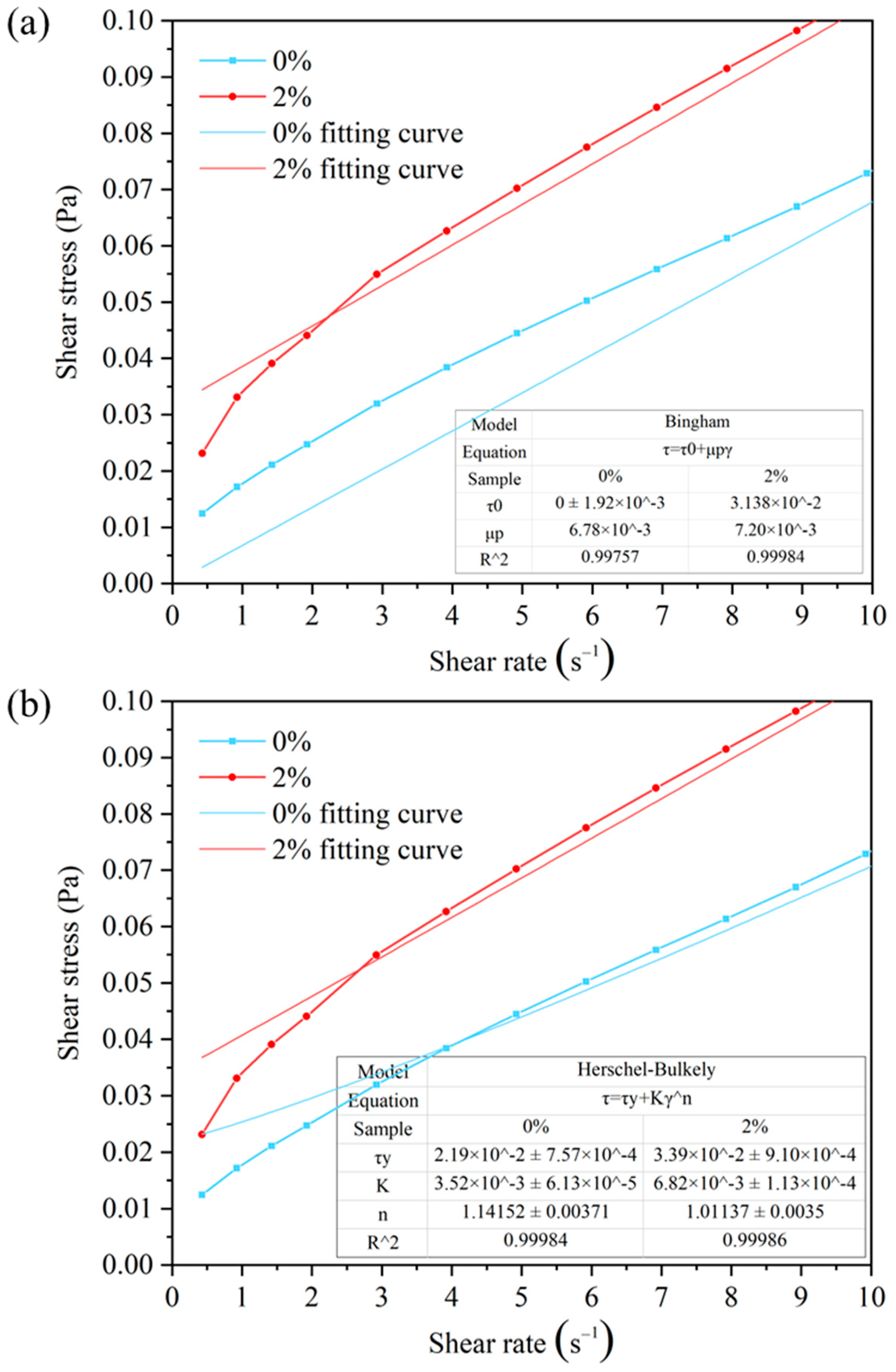
The Analysis of Rheological Model
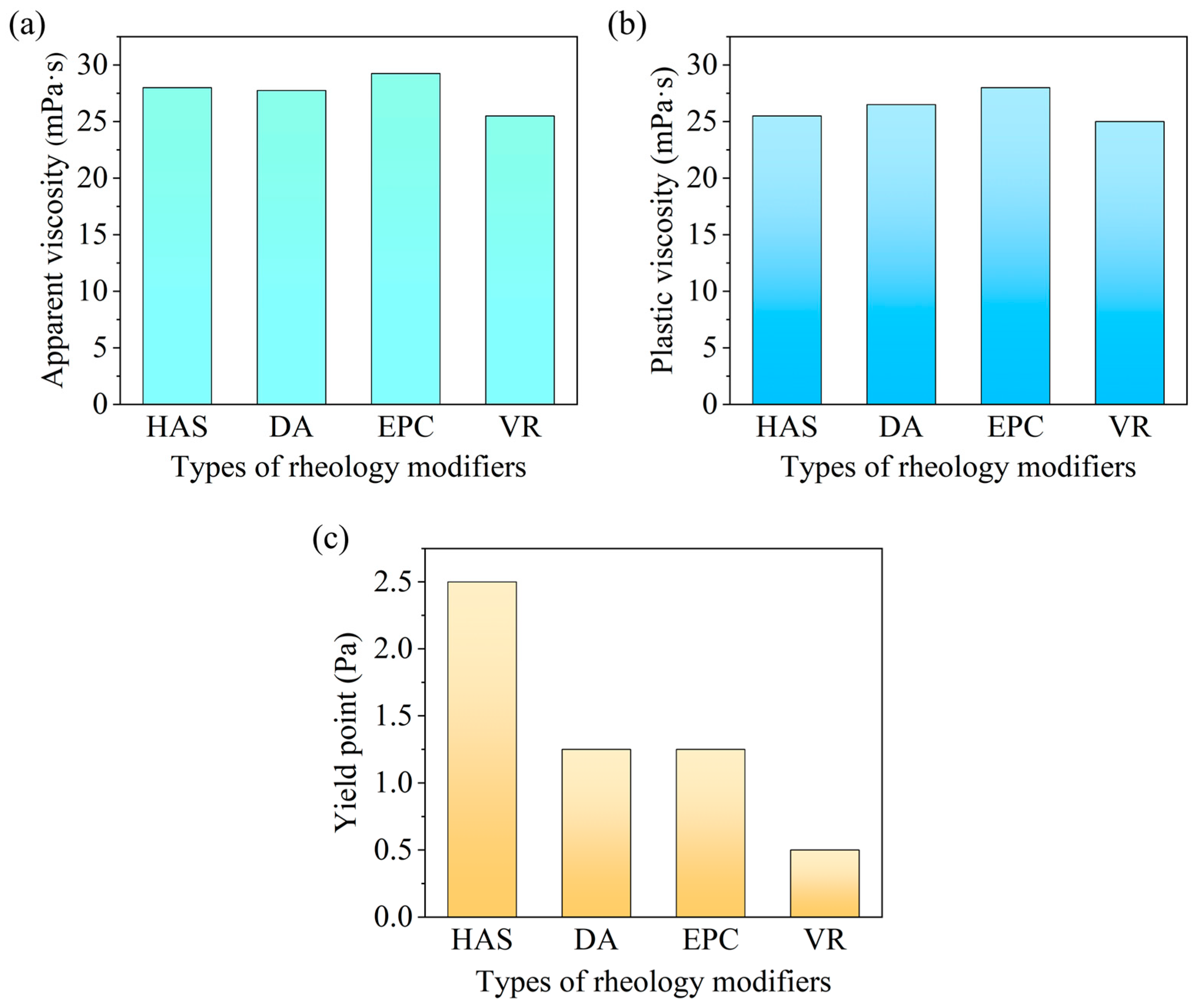
Performance Comparison Between HAS and Traditional Rheology Modifiers
2.2.2. Wetting Performance of HAS
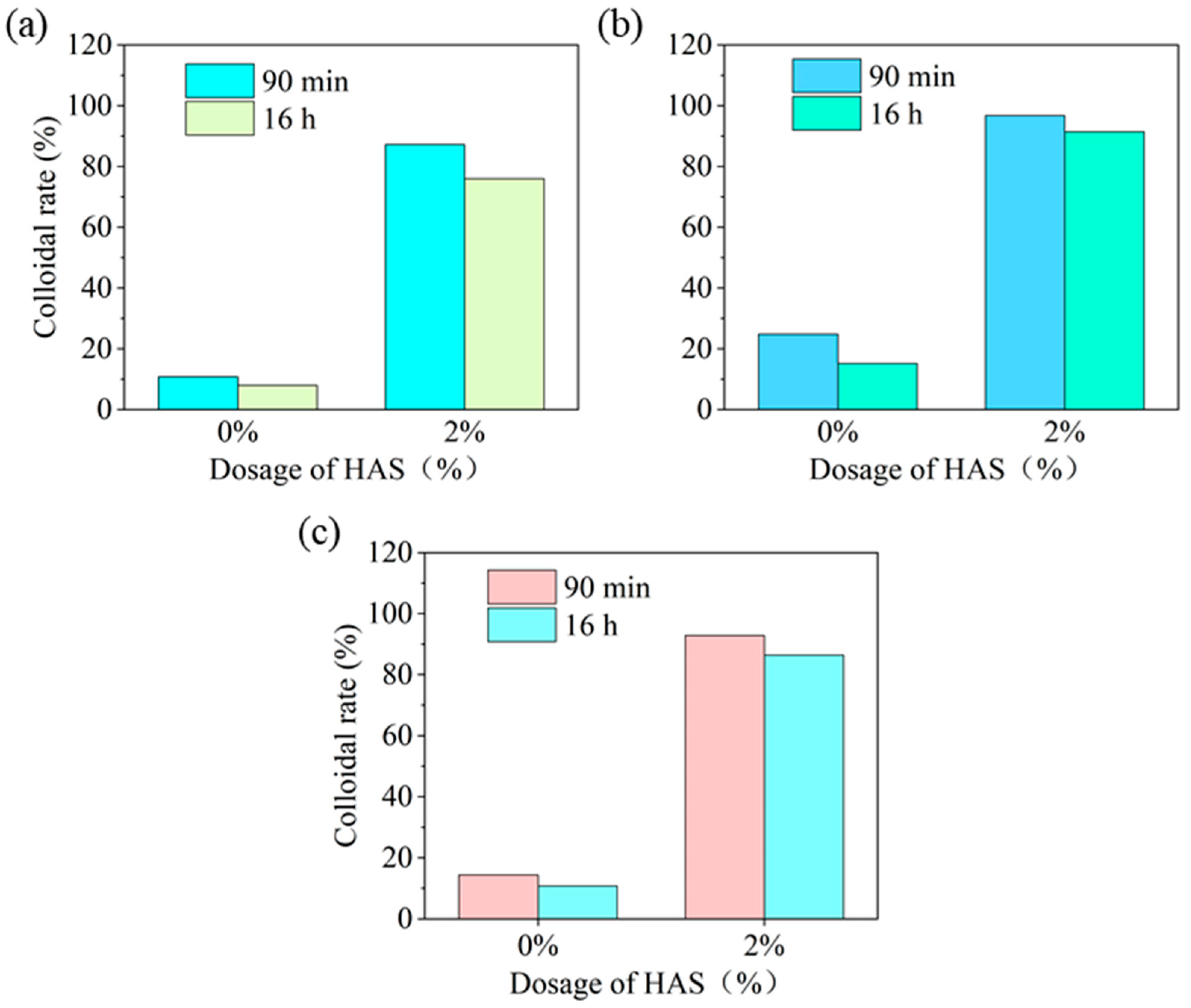
Wettability of HAS in Drilling Fluids with Different Base Oil
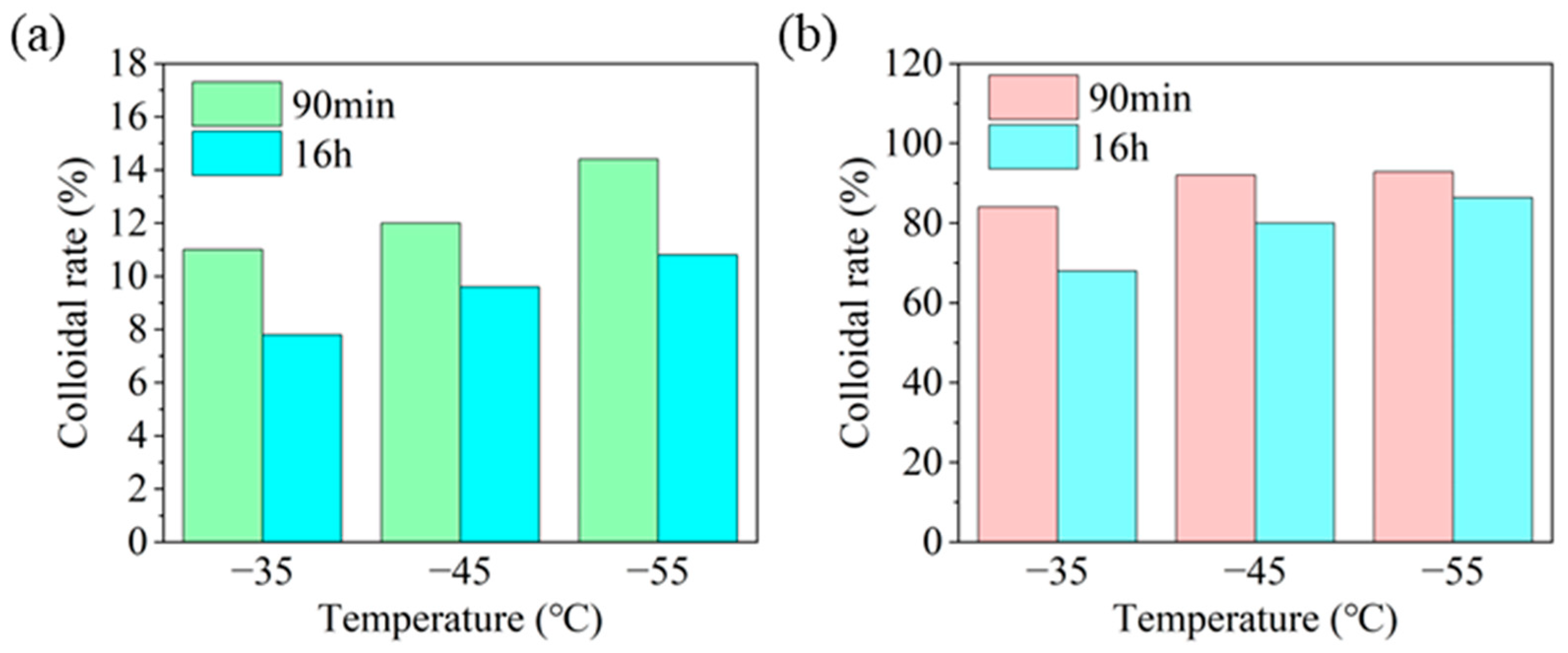
Wettability of HAS in Drilling Fluid at Different Temperatures
Wettability of HAS in Drilling Fluid at Different Densities
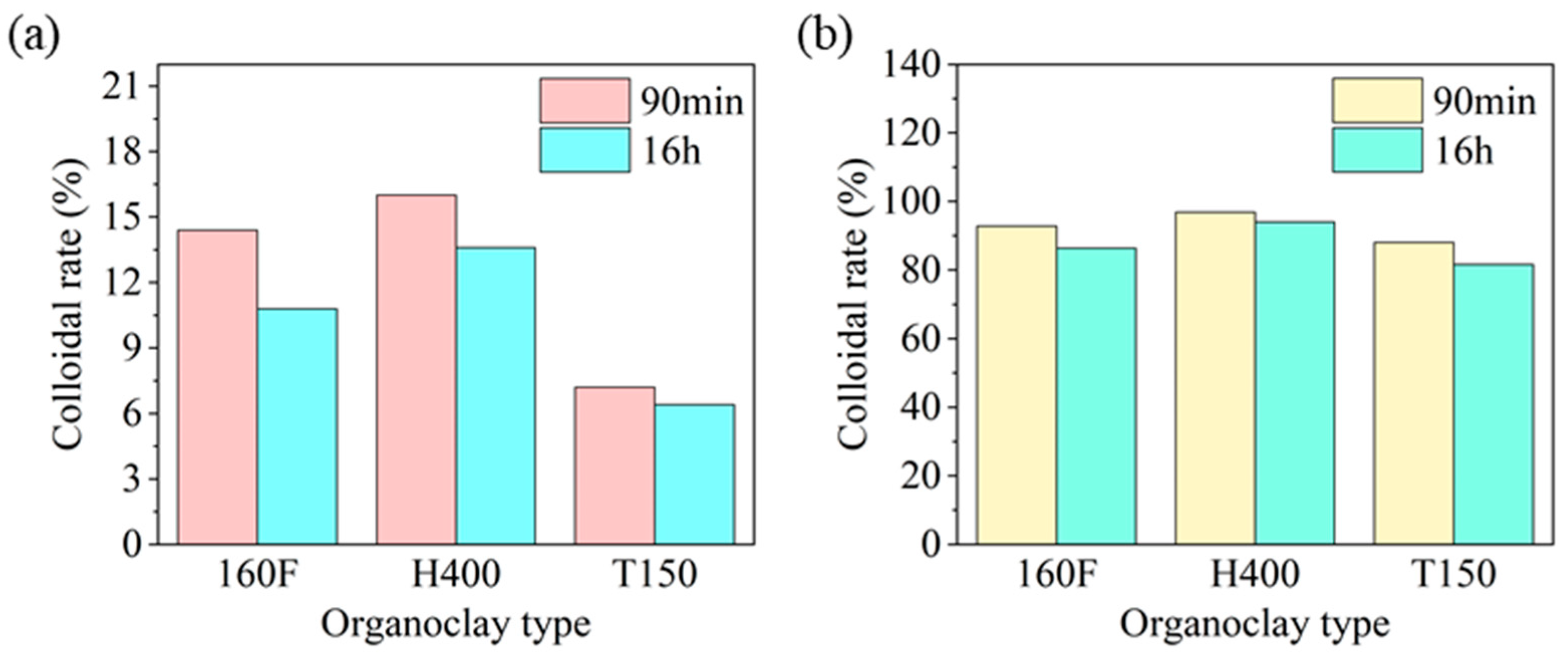
Wettability of HAS in Drilling Fluids with Different Organoclays
2.2.3. Assessment of Environmental Compatibility for HAS
2.2.4. Filtration Performance for Drilling Fluid
2.3. Mechanism Research
2.3.1. Particles Size and Number
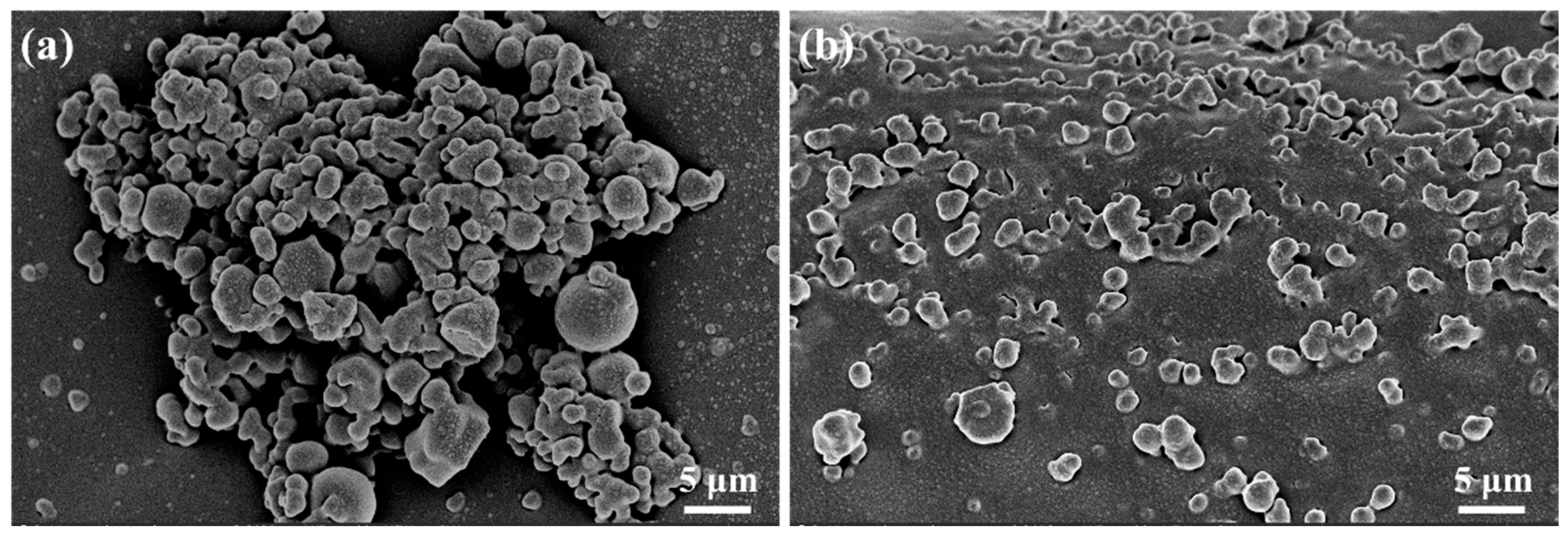
2.3.2. Microscopic Morphology
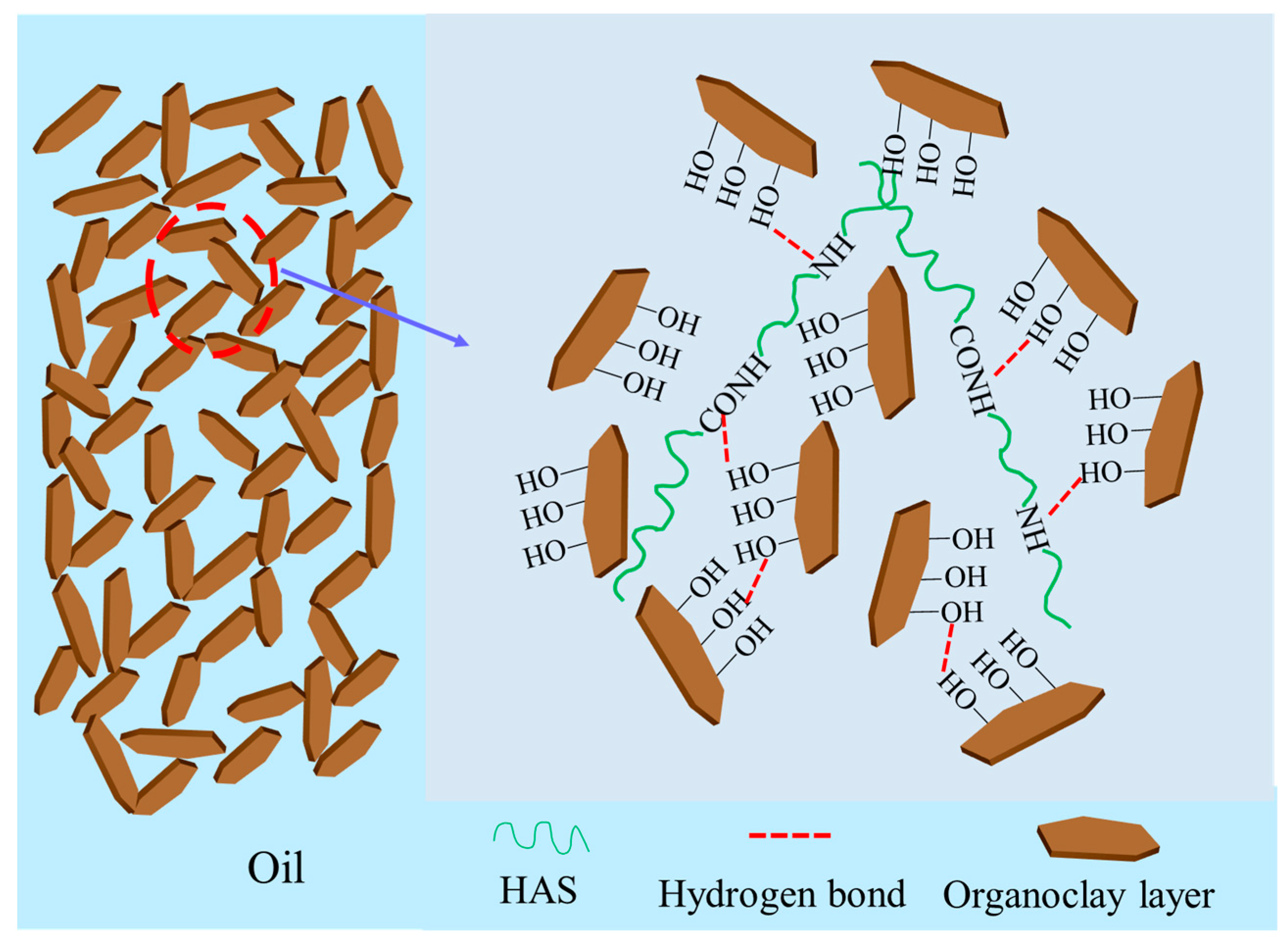
2.3.3. Mechanism Analysis
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. HAS Preparation
4.3. Characterization of HAS
4.3.1. Infrared Spectral Analysis
4.3.2. The Analysis of Nuclear Magnetic Hydrogen Spectrum
4.4. Preparation of Base Fluid and Antarctic Drilling Fluid
4.5. Performance Evaluation
4.5.1. Rheological Performance of HAS
4.5.2. Wettability of HAS
4.5.3. Environmental Compatibility Assessment
4.5.4. Evaluation of Filtration Performance
4.6. Studies of Action Mechanism
4.6.1. Particle Size and Number
4.6.2. Cryo-SEM
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fretwell, P.; Pritchard, H.D.; Vaughan, D.G.; Bamber, J.L.; Barrand, N.E.; Bell, R.; Bianchi, C.; Bingham, R.G.; Blankenship, D.D.; Casassa, G.; et al. Bedmap2: Improved ice bed, surface and thickness datasets for Antarctica. Cryosphere 2013, 7, 375–393. [Google Scholar] [CrossRef]
- Goodge, J.W.; Severinghaus, J.P. Rapid Access Ice Drill: A new tool for exploration of the deep Antarctic ice sheets and subglacial geology. J. Glaciol. 2016, 62, 1049–1064. [Google Scholar] [CrossRef]
- Talalay, P.G. Deep drilling in Antarctic ice: Methods and perspectives. Earth-Sci. Rev. 2023, 243, 104471. [Google Scholar] [CrossRef]
- Deng, S.; Cao, L.; Yan, X.; Peng, M.; Ke, K.; Wang, L.; Hu, Z.; Huo, B.; Wei, R.; Zhang, Y.; et al. A review of progress and challenges in research on the environmentally sound treatment of polar drilling fluids. J. Clean. Prod. 2024, 479, 144035. [Google Scholar] [CrossRef]
- Talalay, P.G.; Zhang, N. Antarctic mineral resources: Looking to the future of the Environmental Protocol. Earth-Sci. Rev. 2022, 232, 104142. [Google Scholar] [CrossRef]
- Alekhina, I.A.; Marie, D.; Petit, J.R.; Lukin, V.V.; Zubkov, V.M.; Bulat, S.A. Molecular analysis of bacterial diversity in kerosene-based drilling fluid from the deep ice borehole at Vostok, East Antarctica. FEMS Microbiol. Ecol. 2007, 59, 289–299. [Google Scholar] [CrossRef]
- Wang, T.; Sun, M.; Pan, Y.; Fu, L.; Yang, S. Development a new type of oil based drilling fluid with good temperature resistant. Energy Sources Part A 2021, 47, 10764–10778. [Google Scholar] [CrossRef]
- Shi, H.; Ni, X.; Wang, X.; Yang, Z.; Wang, J.; Cheng, R.; Yan, L.; Zhang, J.; Yang, H.; Cui, X.; et al. Research and application of low oil-water ratio organoclay-free oil-based drilling fluid technology. E3S Web Conf. 2024, 560, 01016. [Google Scholar] [CrossRef]
- Ni, X.; Shi, H.; Zhang, J.; Liu, R.; Wang, J.; Cheng, R. Modified Laponite synthesized with special wettability as a multifunctional additive in oil-based drilling fluids. J. Pet. Sci. Eng. 2023, 220, 111211. [Google Scholar] [CrossRef]
- Murtaza, M.; Alarifi, S.A.; Kamal, M.S.; Onaizi, S.A.; Al-Ajmi, M.; Mahmoud, M. Experimental investigation of the rheological behavior of an oil-based drilling fluid with rheology modifier and oil wetter additives. Molecules 2021, 26, 4877. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ma, Y.; Sun, J.; Li, X.; Lu, W.; Bi, J. Using aged oil to produce drilling-fluid lubricants. Geoenergy Sci. Eng. 2024, 234, 212635. [Google Scholar] [CrossRef]
- Sui, D.; Sun, Y.; Zhao, J.; Sui, D.; Zhao, F.; Xu, J. Research on a new oil based drilling fluid system. IOP Conf. Ser. Earth Environ. Sci. 2018, 170, 022044. [Google Scholar] [CrossRef]
- Paswan, B.K.; Mahto, V. Development of environment-friendly oil-in-water emulsion based drilling fluid for shale gas formation using sunflower oil. J. Pet. Sci. Eng. 2020, 191, 107129. [Google Scholar] [CrossRef]
- Ma, C.; Li, L.; Yang, Y.P.; Hao, W.W.; Zhang, Q.; Lv, J. Study on the effect of polymeric rheology modifier on the rheological properties of oil-based drilling fluids. IOP Conf. Ser. Mater. Sci. Eng. 2018, 292, 012106. [Google Scholar] [CrossRef]
- Mahmoud, A.; Gajbhiye, R.; Elkatatny, S. Investigating the efficacy of novel organoclay as a rheological additive for enhancing the performance of oil-based drilling fluids. Sci. Rep. 2024, 14, 5323. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Du, M.; He, J.; Liu, H.; Lv, Y.; Guo, L.; Zhang, P.; Bai, Y. An amphiphilic multiblock polymer as a High-Temperature gelling agent for Oil-Based drilling fluids and its mechanism of action. Gels 2023, 9, 966. [Google Scholar] [CrossRef] [PubMed]
- Minakov, A.; Lysakova, E.; Skorobogatova, A.; Pryazhnikov, M.; Ivleva, Y.; Voronin, A.; Simunin, M. Experimental study of the effect of crystalline aluminum oxide nanofibers on the properties of oil-based drilling fluids. J. Mol. Liq. 2023, 388, 122676. [Google Scholar] [CrossRef]
- da Silva, R.; de Castro Dantas, T.; Barillas, J.; Santanna, V.C. The use of organopalygorskite as rheological additive in non-aqueous drilling fluids: Colloidal stability, contact angle, and cutting’s transport ratio. Geoenergy Sci. Eng. 2023, 223, 211499. [Google Scholar] [CrossRef]
- Liu, K.; Pei, H.; Liang, X.; Zhao, W.; Dai, G. Characteristics and modification mechanism of recycled waste silty clay slurry as the shield slag conditioner. Constr. Build. Mater. 2024, 444, 137792. [Google Scholar] [CrossRef]
- Martin, C.; Babaie, M.; Nourian, A.; Nasr, G. Designing Smart drilling fluids using modified nano silica to improve drilling operations in Geothermal wells. Geothermics 2024, 107, 102600. [Google Scholar] [CrossRef]
- Ahmad, H.; Murtaza, M.; Gbadamosi, A.; Kamal, M.S.; Hussain, S.M.S.; Mahmoud, M.; Patil, S. Application of Novel Magnetic Surfactant-Based Drilling Fluids for Clay Swelling Inhibition. Energy Fuels 2023, 37, 8212–8223. [Google Scholar] [CrossRef]
- Guo, D.; Yuan, T.; Sun, Q.; Yan, Z.; Kong, Z.; Zhong, L.; Zhou, Y.; Sha, L. Cellulose nanofibrils as rheology modifier and fluid loss additive in water-based drilling fluids: Rheological properties, rheological modeling, and filtration mechanisms. Ind. Crops Prod. 2023, 193, 116253. [Google Scholar] [CrossRef]
- Ahmad, H.M.; Kamal, M.S.; Al-Harthi, M.A. High molecular weight copolymers as rheology modifier and fluid loss additive for water-based drilling fluids. J. Mol. Liq. 2018, 252, 133–143. [Google Scholar] [CrossRef]
- Deng, C.; Du, G.; Li, X.; Xi, J.; Lin, C. Effect of high temperature and high pressure on the biodegradability and biotoxicity of typical additives in drilling fluid. J. Pet. Sci. Eng. 2022, 208, 109773. [Google Scholar] [CrossRef]
- Yue, W.; Wang, C.; Feng, Q.; Zhao, F.; Chen, Z.; Wu, Z. Nano-enhancing polymer as a fluid loss additive for ultra-high temperature cementing. Constr. Build. Mater. 2024, 449, 138248. [Google Scholar] [CrossRef]





| Name | Measured Value | Standard Value | Classification |
|---|---|---|---|
| BOD5: COD, % | 31.1 | >25 | Environmentally friendly |
| Drilling Fluid Formulation | Test Conditions | FLAPI (mL) | Mud Cake Morphology |
|---|---|---|---|
| 70% aviation kerosene + 30% white oil + 3% organoclay + 2% HAS | Cooling at −55 °C for 16 h | 14 mL | thin and dense |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, N.; Sun, J.; Liu, J.; Lv, K.; Deng, X.; Zhang, T.; Sun, Y.; Yan, H.; Hou, D. Enhancing Rheology and Wettability of Drilling Fluids at Ultra-Low Temperatures Using a Novel Amide Material. Gels 2025, 11, 687. https://doi.org/10.3390/gels11090687
Huang N, Sun J, Liu J, Lv K, Deng X, Zhang T, Sun Y, Yan H, Hou D. Enhancing Rheology and Wettability of Drilling Fluids at Ultra-Low Temperatures Using a Novel Amide Material. Gels. 2025; 11(9):687. https://doi.org/10.3390/gels11090687
Chicago/Turabian StyleHuang, Ning, Jinsheng Sun, Jingping Liu, Kaihe Lv, Xuefei Deng, Taifeng Zhang, Yuanwei Sun, Han Yan, and Delin Hou. 2025. "Enhancing Rheology and Wettability of Drilling Fluids at Ultra-Low Temperatures Using a Novel Amide Material" Gels 11, no. 9: 687. https://doi.org/10.3390/gels11090687
APA StyleHuang, N., Sun, J., Liu, J., Lv, K., Deng, X., Zhang, T., Sun, Y., Yan, H., & Hou, D. (2025). Enhancing Rheology and Wettability of Drilling Fluids at Ultra-Low Temperatures Using a Novel Amide Material. Gels, 11(9), 687. https://doi.org/10.3390/gels11090687






