Bioprinting Vascularized Constructs for Clinical Relevance: Engineering Hydrogel Systems for Biological Maturity
Abstract
1. Introduction
2. Development and Maturation of Functional Vasculature in Tissue
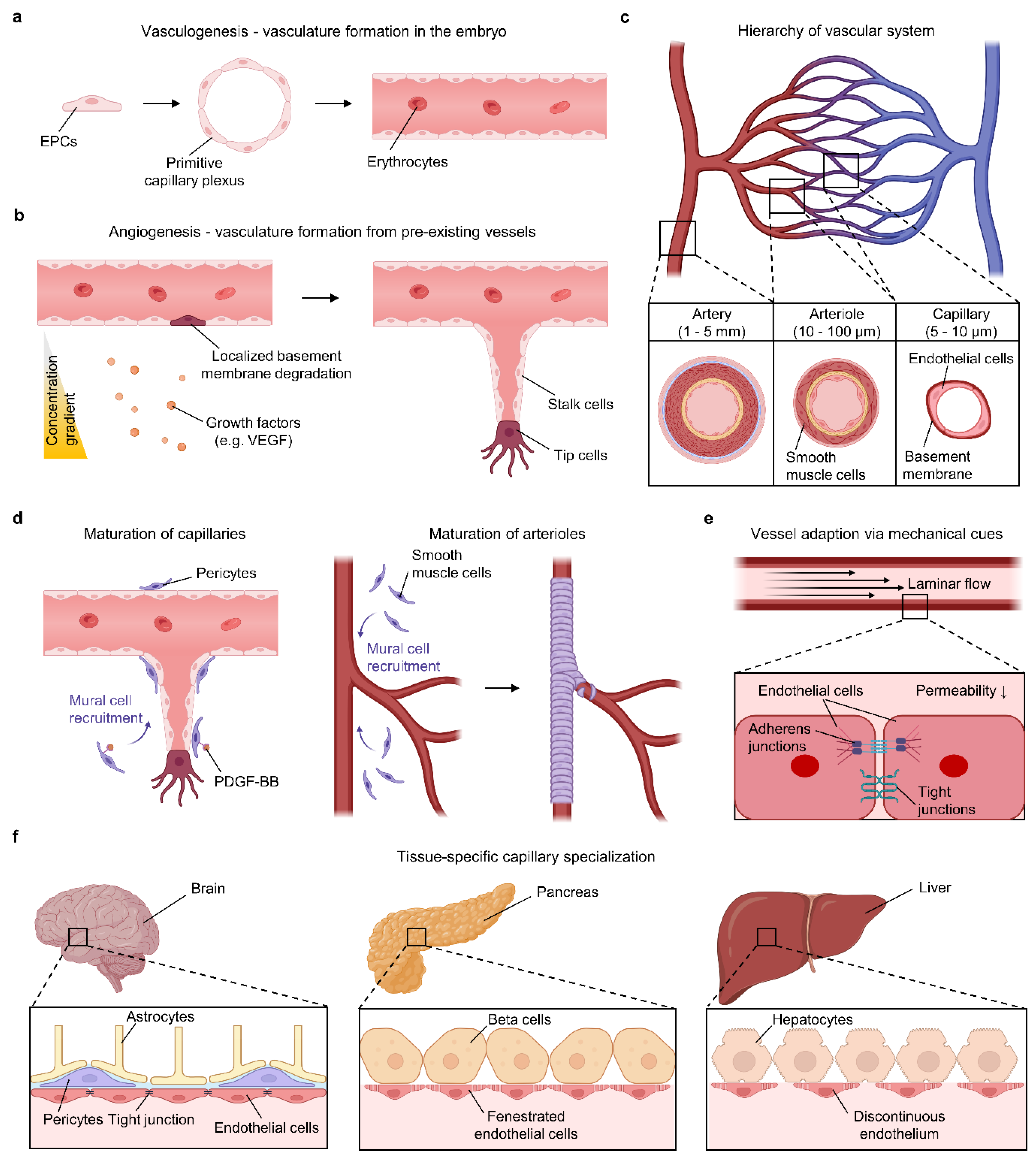
2.1. Developmental Mechanisms of Vasculature Formation
2.2. Structural Hierarchy from Capillaries to Arteries
2.3. Vascular Maturation and Stabilization
2.4. Role of Mechanical Cues in Vessel Adaptation
2.5. Tissue-Specific Vascular Requirements
3. Scale Matters: Large-Caliber vs. Microvascular Design in the Clinic
3.1. Large-Caliber Vessels: Mechanical Integrity and Thromboresistance
3.2. Microvascular Networks: Prioritizing Remodeling and Integration
3.3. Hierarchical Integration Strategies
4. Hydrogels as Dynamic Vascular Niches
4.1. Matrix Composition and Biochemical Cues
4.2. Stiffness and Mechanotransduction
4.3. Degradability and Remodeling Compatibility
5. Bioprinting Strategies for Scalable Vascularized Tissue Construction
5.1. Sacrificial Hydrogel Extrusion-Based Bioprinting

5.2. Coaxial Bioprinting: One-Step Fabrication of Multi-Layered Vessels
5.3. Embedded Bioprinting: Enabling Complex Vascular Architectures with 3D Hierarchy
5.4. Light-Based Bioprinting: High-Resolution Patterning of Capillary-Scale Vasculature
6. Future Perspectives: Hydrogel System and Bioprinting Techniques to Engineer Physiologically Relevant Vasculature
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fu, J.; Wang, D.A. In Situ Organ-Specific Vascularization in Tissue Engineering. Trends Biotechnol. 2018, 36, 834–849. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P.; Jain, R.K. Principles and mechanisms of vessel normalization for cancer and other angiogenic diseases. Nat. Rev. Drug Discov. 2011, 10, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Schulten, L.; Spillner, J.; Kanzler, S.; Teubner, A.; Jockenhoevel, S.; Apel, C. A polyurethane-based surgical adhesive for sealing blood vessel anastomoses—A feasibility study in pigs. J. Biomed. Mater. Res. Part B Appl. Biomater. 2022, 110, 1922–1931. [Google Scholar] [CrossRef]
- Lam, J.; Yu, J.; Lee, B.; Campagna, C.; Yoo, S.; Baek, K.; Jeon, N.L.; Sung, K.E. Characterizing On-Chip Angiogenesis Induction in a Microphysiological System as a Functional Measure of Mesenchymal Stromal Cell Bioactivity. Adv. Biol. 2024, 8, e2300094. [Google Scholar] [CrossRef] [PubMed]
- Nwokoye, P.N.; Abilez, O.J. Bioengineering methods for vascularizing organoids. Cell Rep. Methods 2024, 4, 100779. [Google Scholar] [CrossRef]
- Wang, D.; Maharjan, S.; Kuang, X.; Wang, Z.; Mille, L.S.; Tao, M.; Yu, P.; Cao, X.; Lian, L.; Lv, L.; et al. Microfluidic bioprinting of tough hydrogel-based vascular conduits for functional blood vessels. Sci. Adv. 2022, 8, eabq6900. [Google Scholar] [CrossRef]
- Kim, J.W.; Nam, S.A.; Yi, J.; Kim, J.Y.; Lee, J.Y.; Park, S.-Y.; Sen, T.; Choi, Y.-m.; Lee, J.Y.; Kim, H.L.; et al. Kidney Decellularized Extracellular Matrix Enhanced the Vascularization and Maturation of Human Kidney Organoids. Adv. Sci. 2022, 9, e2103526. [Google Scholar] [CrossRef]
- Kuan, C.-H.; Chang, L.; Ho, C.-Y.; Tsai, C.-H.; Liu, Y.-C.; Huang, W.-Y.; Wang, Y.-N.; Wang, W.-H.; Wang, T.-W. Immunomodulatory hydrogel orchestrates pro-regenerative response of macrophages and angiogenesis for chronic wound healing. Biomaterials 2025, 314, 122848. [Google Scholar] [CrossRef]
- Huang, W.; Guo, Q.; Wu, H.; Zheng, Y.; Xiang, T.; Zhou, S. Engineered Exosomes Loaded in Intrinsic Immunomodulatory Hydrogels with Promoting Angiogenesis for Programmed Therapy of Diabetic Wounds. ACS Nano 2025, 19, 14467–14483. [Google Scholar] [CrossRef]
- Jeong, W.; Son, J.; Choi, J.; Han, J.; Jeon, S.; Kim, M.K.; Ha, W.; Kang, H.-W. Clinically Relevant and Precisely Printable Live Adipose Tissue–Based Bio-Ink for Volumetric Soft Tissue Reconstruction. Adv. Healthc. Mater. 2025, 14, e2402680. [Google Scholar] [CrossRef] [PubMed]
- Thai, V.L.; Ramos-Rodriguez, D.H.; Mesfin, M.; Leach, J.K. Hydrogel degradation promotes angiogenic and regenerative potential of cell spheroids for wound healing. Mater. Today Bio 2023, 22, 100769. [Google Scholar] [CrossRef]
- Heltmann- Meyer, S.; Steiner, D.; Müller, C.; Schneidereit, D.; Friedrich, O.; Salehi, S.; Engel, F.B.; Arkudas, A.; Horch, R.E. Gelatin methacryloyl is a slow degrading material allowing vascularization and long-term use in vivo. Biomed. Mater. 2021, 16, 065004. [Google Scholar] [CrossRef]
- Kang, H.W.; Lee, S.J.; Ko, I.K.; Kengla, C.; Yoo, J.J.; Atala, A. A 3D bioprinting system to produce human-scale tissue constructs with structural integrity. Nat. Biotechnol. 2016, 34, 312–319. [Google Scholar] [CrossRef]
- Hinton, T.J.; Jallerat, Q.; Palchesko, R.N.; Park, J.H.; Grodzicki, M.S.; Shue, H.-J.; Ramadan, M.H.; Hudson, A.R.; Feinberg, A.W. Three-dimensional printing of complex biological structures by freeform reversible embedding of suspended hydrogels. Sci. Adv. 2015, 1, e1500758. [Google Scholar] [CrossRef]
- Skylar-Scott, M.A.; Uzel, S.G.M.; Nam, L.L.; Ahrens, J.H.; Truby, R.L.; Damaraju, S.; Lewis, J.A. Biomanufacturing of organ-specific tissues with high cellular density and embedded vascular channels. Sci. Adv. 2019, 5, eaaw2459. [Google Scholar] [CrossRef]
- Jungst, T.; Smolan, W.; Schacht, K.; Scheibel, T.; Groll, J. Strategies and Molecular Design Criteria for 3D Printable Hydrogels. Chem. Rev. 2016, 116, 1496–1539. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.S.; Davoudi, F.; Walch, P.; Manbachi, A.; Luo, X.; Dell’Erba, V.; Miri, A.K.; Albadawi, H.; Arneri, A.; Li, X.; et al. Bioprinted thrombosis-on-a-chip. Lab Chip 2016, 16, 4097–4105. [Google Scholar] [CrossRef]
- Son, J.; Hong, S.J.; Lim, J.W.; Jeong, W.; Jeong, J.H.; Kang, H.-W. Engineering Tissue-Specific, Multiscale Microvasculature with a Capillary Network for Prevascularized Tissue. Small Methods 2021, 5, e2100632. [Google Scholar] [CrossRef] [PubMed]
- Sexton, Z.A.; Rütsche, D.; Herrmann, J.E.; Hudson, A.R.; Sinha, S.; Du, J.; Shiwarski, D.J.; Masaltseva, A.; Solberg, F.S.; Pham, J.; et al. Rapid model-guided design of organ-scale synthetic vasculature for biomanufacturing. Science 2025, 388, 1198–1204. [Google Scholar] [CrossRef] [PubMed]
- Kolesky, D.B.; Homan, K.A.; Skylar-Scott, M.A.; Lewis, J.A. Three-dimensional bioprinting of thick vascularized tissues. Proc. Natl. Acad. Sci. USA 2016, 113, 3179–3184. [Google Scholar] [CrossRef]
- Coultas, L.; Chawengsaksophak, K.; Rossant, J. Endothelial cells and VEGF in vascular development. Nature 2005, 438, 937–945. [Google Scholar] [CrossRef]
- Carmeliet, P. Angiogenesis in health and disease. Nat. Med. 2003, 9, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P. Mechanisms of angiogenesis and arteriogenesis. Nat. Med. 2000, 6, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Vasculogenesis, angiogenesis, and arteriogenesis: Mechanisms of blood vessel formation and remodeling. J. Cell. Biochem. 2007, 102, 840–847. [Google Scholar] [CrossRef]
- Dejana, E. Endothelial cell–cell junctions: Happy together. Nat. Rev. Mol. Cell Biol. 2004, 5, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Bazzoni, G.; Dejana, E. Endothelial Cell-to-Cell Junctions: Molecular Organization and Role in Vascular Homeostasis. Physiol. Rev. 2004, 84, 869–901. [Google Scholar] [CrossRef]
- Pugsley, M.K.; Tabrizchi, R. The vascular system: An overview of structure and function. J. Pharmacol. Toxicol. Methods 2000, 44, 333–340. [Google Scholar] [CrossRef]
- Aird, W.C. Phenotypic heterogeneity of the endothelium: I. Structure, function, and mechanisms. Circ. Res. 2007, 100, 158–173. [Google Scholar]
- Aird, W.C. Endothelial cell heterogeneity. Cold Spring Harb. Perspect. Med. 2012, 2, a006429. [Google Scholar] [CrossRef]
- Armulik, A.; Genové, G.; Betsholtz, C. Pericytes: Developmental, Physiological, and Pathological Perspectives, Problems, and Promises. Dev. Cell 2011, 21, 193–215. [Google Scholar] [CrossRef] [PubMed]
- Basatemur, G.L.; Jørgensen, H.F.; Clarke, M.C.H.; Bennett, M.R.; Mallat, Z. Vascular smooth muscle cells in atherosclerosis. Nat. Rev. Cardiol. 2019, 16, 727–744. [Google Scholar] [CrossRef] [PubMed]
- Dessalles, C.A.; Babataheri, A.; Barakat, A.I. Pericyte mechanics and mechanobiology. J. Cell Sci. 2021, 134, jcs240226. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte–endothelial interactions at the blood–brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef]
- Hahn, C.; Schwartz, M.A. Mechanotransduction in vascular physiology and atherogenesis. Nat. Rev. Mol. Cell Biol. 2009, 10, 53–62. [Google Scholar] [CrossRef]
- Tzima, E.; Irani-Tehrani, M.; Kiosses, W.B.; Dejana, E.; Schultz, D.A.; Engelhardt, B.; Cao, G.; DeLisser, H.; Schwartz, M.A. A mechanosensory complex that mediates the endothelial cell response to fluid shear stress. Nature 2005, 437, 426–431. [Google Scholar] [CrossRef]
- Wagenseil, J.E.; Mecham, R.P. Vascular Extracellular Matrix and Arterial Mechanics. Physiol. Rev. 2009, 89, 957–989. [Google Scholar] [CrossRef]
- Baeyens, N.; Bandyopadhyay, C.; Coon, B.G.; Yun, S.; Schwartz, M.A. Endothelial fluid shear stress sensing in vascular health and disease. J. Clin. Investig. 2016, 126, 821–828. [Google Scholar] [CrossRef]
- Cooper, E.S.; Silverstein, D.C. Fluid therapy and the microcirculation in health and critical illness. Front. Vet. Sci. 2021, 8, 625708. [Google Scholar] [CrossRef]
- Augustin, H.G.; Koh, G.Y. Organotypic vasculature: From descriptive heterogeneity to functional pathophysiology. Science 2017, 357, eaal2379. [Google Scholar] [CrossRef] [PubMed]
- Stan, R.V.; Tse, D.; Deharvengt, S.J.; Smits, N.C.; Xu, Y.; Luciano, M.R.; McGarry, C.L.; Buitendijk, M.; Nemani, K.V.; Elgueta, R.; et al. The Diaphragms of Fenestrated Endothelia: Gatekeepers of Vascular Permeability and Blood Composition. Dev. Cell 2012, 23, 1203–1218. [Google Scholar] [CrossRef]
- Poisson, J.; Lemoinne, S.; Boulanger, C.; Durand, F.; Moreau, R.; Valla, D.; Rautou, P.-E. Liver sinusoidal endothelial cells: Physiology and role in liver diseases. J. Hepatol. 2017, 66, 212–227. [Google Scholar] [CrossRef]
- Chang, W.G.; Niklason, L.E. A Short Discourse on Vascular Tissue Engineering. NPJ Regen. Med. 2017, 2, 7. [Google Scholar] [CrossRef]
- Llorach-Perucho, N.; Cayetano-Paniagua, L.; Serra-Aracil, X. Importance of the diameter of the mechanical suture in rectal surgery in relation to benign anastomotic stenosis. Cross-sectional observational study. Tech. Coloproctol. 2025, 29, 137. [Google Scholar] [CrossRef] [PubMed]
- Cong, J.; Zhang, H.; Chen, C. Definition and grading of anastomotic stricture/stenosis following low anastomosis after total mesorectal excision: A single-center study. Asian J. Surg. 2023, 46, 3722–3726. [Google Scholar] [CrossRef] [PubMed]
- Shiu, Y.T.; Rotmans, J.I.; Geelhoed, W.J.; Pike, D.B.; Lee, T. Arteriovenous conduits for hemodialysis: How to better modulate the pathophysiological vascular response to optimize vascular access durability. Am. J. Physiol. Ren. Physiol. 2019, 316, F794–F806. [Google Scholar] [CrossRef]
- Andree, C.; Munder, B.I.J.; Behrendt, P.; Hellmann, S.; Audretsch, W.; Voigt, M.; Reis, C.; Beckmann, M.W.; Horch, R.E.; Bach, A.D. Improved safety of autologous breast reconstruction surgery by stabilisation of microsurgical vessel anastomoses using fibrin sealant in 349 free DIEP or fascia-muscle-sparing (fms)-TRAM flaps: A two-centre study. Breast 2008, 17, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, R.F.; Bakht, S.M.; Gomez-Florit, M.; Stievani, F.C.; Alves, A.L.G.; Reis, R.L.; Gomes, M.E.; Domingues, R.M.A. Writing 3D In Vitro Models of Human Tendon within a Biomimetic Fibrillar Support Platform. ACS Appl. Mater. Interfaces 2023, 15, 50598–50611. [Google Scholar] [CrossRef]
- Vaghela, R.; Arkudas, A.; Gage, D.; Körner, C.; von Hörsten, S.; Salehi, S.; Horch, R.E.; Hessenauer, M. Microvascular development in the rat arteriovenous loop model in vivo—A step by step intravital microscopy analysis. J. Biomed. Mater. Res. Part A 2022, 110, 1551–1563. [Google Scholar] [CrossRef]
- Soloukey, S.; Verhoef, L.; Jan van Doormaal, P.; Generowicz, B.S.; Dirven, C.M.F.; De Zeeuw, C.I.; Koekkoek, S.K.E.; Kruizinga, P.; Vincent, A.; Schouten, J.W. High-resolution micro-Doppler imaging during neurosurgical resection of an arteriovenous malformation: Illustrative case. J. Neurosurg. Case Lessons 2022, 4, case22177. [Google Scholar] [CrossRef]
- Wong, R.; Donno, R.; Leon-Valdivieso, C.Y.; Roostalu, U.; Derby, B.; Tirelli, N.; Wong, J.K. Angiogenesis and tissue formation driven by an arteriovenous loop in the mouse. Sci. Rep. 2019, 9, 10478. [Google Scholar] [CrossRef]
- Kakisis, J.D.; Antonopoulos, C.; Mantas, G.; Alexiou, E.; Katseni, K.; Sfyroeras, G.; Moulakakis, K.; Geroulakos, G. Safety and efficacy of polyurethane vascular grafts for early hemodialysis access. J. Vasc. Surg. 2017, 66, 1792–1797. [Google Scholar] [CrossRef]
- Kampf, S.; Willegger, M.; Dawoud, C.; Fülöp, G.; Lirk, P.; Willfort-Ehringer, A.; Neumayer, C.; Gollackner, B. Long-term outcome in pediatric surgical bypass grafting after traumatic injury and tumor resection: Retrospective cohort analysis. Sci. Rep. 2021, 11, 16321. [Google Scholar] [CrossRef]
- Zhang, F.J.; Xiong, Y.; Hu, Y.; Liu, J.; Wei, Y.; Yang, L.; Luo, R.; Wang, Y. Armoring Vascular Implant by Glycocalyx-Mimetic Coating with Clustered Morphology and Hydrophilic Lubrication. Adv. Funct. Mater. 2025, 35, 2414136. [Google Scholar] [CrossRef]
- Coronel-Meneses, D.; Sánchez-Trasviña, C.; Ratera, I.; Mayolo-Deloisa, K. Strategies for surface coatings of implantable cardiac medical devices. Front. Bioeng. Biotechnol. 2023, 11, 1173260. [Google Scholar] [CrossRef] [PubMed]
- Pashneh-Tala, S.; MacNeil, S.; Claeyssens, F. The Tissue-Engineered Vascular Graft-Past, Present, and Future. Tissue Eng. Part B Rev. 2016, 22, 68–100. [Google Scholar] [CrossRef] [PubMed]
- De Visscher, G.; Mesure, L.; Meuris, B.; Ivanova, A.; Flameng, W. Improved endothelialization and reduced thrombosis by coating a synthetic vascular graft with fibronectin and stem cell homing factor SDF-1α. Acta Biomater. 2012, 8, 1330–1338. [Google Scholar] [CrossRef]
- Yau, J.W.; Teoh, H.; Verma, S. Endothelial cell control of thrombosis. BMC Cardiovasc. Disord. 2015, 15, 130. [Google Scholar] [CrossRef]
- Michael, P.L.; Yang, N.; Moore, M.; Santos, M.; Lam, Y.T.; Ward, A.; Hung, J.C.; Tan, R.P.; Wise, S.G. Synthetic vascular graft with spatially distinct architecture for rapid biomimetic cell organisation in a perfusion bioreactor. Biomed. Mater. 2022, 17, 045001. [Google Scholar] [CrossRef]
- Reid, J.A.; McDonald, A.; Callanan, A. Electrospun fibre diameter and its effects on vascular smooth muscle cells. J. Mater. Sci. Mater. Med. 2021, 32, 131. [Google Scholar] [CrossRef]
- Murea, M.; Gardezi, A.I.; Goldman, M.P.; Hicks, C.W.; Lee, T.; Middleton, J.P.; Shingarev, R.; Vachharajani, T.J.; Woo, K.; Abdelnour, L.M.; et al. Study protocol of a randomized controlled trial of fistula vs. graft arteriovenous vascular access in older adults with end-stage kidney disease on hemodialysis: The AV access trial. BMC Nephrol. 2023, 24, 43. [Google Scholar] [CrossRef]
- Sawant, A.; Mills, P.K.; Dhingra, H. Increased length of stay and costs associated with inpatient management of vascular access failures. Semin. Dial. 2013, 26, 106–110. [Google Scholar] [CrossRef]
- Chang, D.K.; Louis, M.R.; Gimenez, A.; Reece, E.M. The Basics of Integra Dermal Regeneration Template and its Expanding Clinical Applications. Semin. Plast. Surg. 2019, 33, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Pepper, A.R.; Pawlick, R.; Gala-Lopez, B.; MacGillivary, A.; Mazzuca, D.M.; White, D.J.; Toleikis, P.M.; Shapiro, A.M. Diabetes Is Reversed in a Murine Model by Marginal Mass Syngeneic Islet Transplantation Using a Subcutaneous Cell Pouch Device. Transplantation 2015, 99, 2294–2300. [Google Scholar] [CrossRef] [PubMed]
- Maleckis, K.; Kamenskiy, A.; Lichter, E.Z.; Oberley-Deegan, R.; Dzenis, Y.; MacTaggart, J. Mechanically tuned vascular graft demonstrates rapid endothelialization and integration into the porcine iliac artery wall. Acta Biomater. 2021, 125, 126–137. [Google Scholar] [CrossRef]
- Baba, K.; Mikhailov, A.; Sankai, Y. Dynamic flow priming programs allow tuning up the cell layers properties for engineered vascular graft. Sci. Rep. 2021, 11, 14666. [Google Scholar] [CrossRef] [PubMed]
- Acar, Y.; Managh, A.; Hill, E.J.; Roach, P. Tuning Surface Chemistry Impacts on Cardiac Endothelial and Smooth Muscle Cell Development. J. Biomed. Mater. Res. Part A 2025, 113, e37846. [Google Scholar] [CrossRef]
- Dudley, A.C.; Griffioen, A.W. Pathological angiogenesis: Mechanisms and therapeutic strategies. Angiogenesis 2023, 26, 313–347. [Google Scholar] [CrossRef]
- Liu, Z.-L.; Chen, H.-H.; Zheng, L.-L.; Sun, L.-P.; Shi, L. Angiogenic signaling pathways and anti-angiogenic therapy for cancer. Signal Transduct. Target. Ther. 2023, 8, 198. [Google Scholar] [CrossRef]
- Chen, C.-L.; Wei, S.-Y.; Chen, W.-L.; Hsu, T.-L.; Chen, Y.-C. Reconstructing vascular networks promotes the repair of skeletal muscle following volumetric muscle loss by pre-vascularized tissue constructs. J. Tissue Eng. 2023, 14, 20417314231201231. [Google Scholar] [CrossRef]
- Jank, B.J.; Goverman, J.; Guyette, J.P.; Charest, J.M.; Randolph, M.; Gaudette, G.R.; Gershlak, J.R.; Purschke, M.; Javorsky, E.; Nazarian, R.M.; et al. Creation of a Bioengineered Skin Flap Scaffold with a Perfusable Vascular Pedicle. Tissue Eng. Part A 2017, 23, 696–707. [Google Scholar] [CrossRef]
- Stankiewicz, M.; Coyer, F.; Webster, J.; Osborne, S. Incidence and Predictors of Lower Limb Split-Skin Graft Failure and Primary Closure Dehiscence in Day-Case Surgical Patients. Dermatol. Surg. 2015, 41, 775–783. [Google Scholar] [CrossRef]
- Soares, M.A.; Ezeamuzie, O.; Ham, M.; Duckworth, A.; Rabbani, P.S.; Saadeh, P.B.; Ceradini, D.J. Targeted protection of donor graft vasculature using a phosphodiesterase inhibitor increases survival and predictability of autologous fat grafts. Plast. Reconstr. Surg. 2015, 135, 488–499. [Google Scholar] [CrossRef]
- Fischer, E.O.; Tsukerman, A.; Machour, M.; Shuhmaher, M.; Silverstein, A.; Yaakov, M.; Bar-Am, O.; Debbi, L.; Levenberg, S. Bioprinting Perfusable and Vascularized Skeletal Muscle Flaps for the Treatment of Volumetric Muscle Loss. Adv. Healthc. Mater. 2025, 14, e2404542. [Google Scholar] [CrossRef]
- Son, J.; Mohamed, H.J.; Ha, W.; Naren, A.; Choi, C.; Kwon, Y.H.; Park, S.; Joung, H.-C.; Kang, H.-W. Bioprinting of pre-vascularized constructs for enhanced in vivo neo-vascularization. Biofabrication 2023, 15, 034101. [Google Scholar] [CrossRef]
- Mohamed, H.J.; Jeong, W.; Son, J.; Kang, H.-W. Bioprinting of Adipose Tissue Graft with Enhanced Neo-Vessel Formation in Vivo. Adv. Healthc. Mater. 2025, e2500627. [Google Scholar] [CrossRef]
- Baeyens, N.; Nicoli, S.; Coon, B.G.; Ross, T.D.; Van den Dries, K.; Han, J.; Lauridsen, H.M.; Mejean, C.O.; Eichmann, A.; Thomas, J.-L.; et al. Vascular remodeling is governed by a VEGFR3-dependent fluid shear stress set point. eLife 2015, 4, e04645. [Google Scholar] [CrossRef] [PubMed]
- Honkura, N.; Richards, M.; Laviña, B.; Sáinz-Jaspeado, M.; Betsholtz, C.; Claesson-Welsh, L. Intravital imaging-based analysis tools for vessel identification and assessment of concurrent dynamic vascular events. Nat. Commun. 2018, 9, 2746. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Ruan, D.X.; Huang, M.Q.; Tian, M.; Zhu, K.S.; Gan, Z.Q.; Xiao, Z.C. Harnessing the potential of hydrogels for advanced therapeutic applications: Current achievements and future directions. Signal Transduct. Target. Ther. 2024, 9, 166. [Google Scholar] [CrossRef] [PubMed]
- Seltana, A.; Cloutier, G.; Nicolas, V.R.; Khalfaoui, T.; Teller, I.C.; Perreault, N.; Beaulieu, J.F. Fibrin(ogen) Is Constitutively Expressed by Differentiated Intestinal Epithelial Cells and Mediates Wound Healing. Front. Immunol. 2022, 13, 916187. [Google Scholar] [CrossRef] [PubMed]
- Shaik, R.; Xu, J.Z.; Wang, Y.; Hong, Y.; Zhang, G. Fibrin-Enriched Cardiac Extracellular Matrix Hydrogel Promotes. Angiogenesis. ACS Biomater. Sci. Eng. 2023, 9, 877–888. [Google Scholar] [CrossRef]
- Schweller, R.M.; West, J.L. Encoding Hydrogel Mechanics via Network Cross-Linking Structure. ACS Biomater. Sci. Eng. 2015, 1, 335–344. [Google Scholar] [CrossRef]
- Greenwald, S.E.; Berry, C.L. Improving vascular grafts: The importance of mechanical and haemodynamic properties. J. Pathol. 2000, 190, 292–299. [Google Scholar] [CrossRef]
- Park, J.S.; Chu, J.S.; Tsou, A.D.; Diop, R.; Tang, Z.; Wang, A.; Li, S. The effect of matrix stiffness on the differentiation of mesenchymal stem cells in response to TGF-beta. Biomaterials 2011, 32, 3921–3930. [Google Scholar] [CrossRef]
- Guvendiren, M.; Burdick, J.A. Stiffening hydrogels to probe short- and long-term cellular responses to dynamic mechanics. Nat. Commun. 2012, 3, 792. [Google Scholar] [CrossRef]
- Friend, N.E.; McCoy, A.J.; Stegemann, J.P.; Putnam, A.J. A combination of matrix stiffness and degradability dictate microvascular network assembly and remodeling in cell-laden poly(ethylene glycol) hydrogels. Biomaterials 2023, 295, 122050. [Google Scholar] [CrossRef]
- Arakawa, C.K.; Badeau, B.A.; Zheng, Y.; DeForest, C.A. Multicellular Vascularized Engineered Tissues through User-Programmable Biomaterial Photodegradation. Adv. Mater. 2017, 29, 1703156. [Google Scholar] [CrossRef]
- Dong, Y.W.; Ma, T.T.; Xu, T.; Feng, Z.Y.; Li, Y.G.; Song, L.L.; Yao, X.J.; Ashby, C.J.r.; Hao, G.F. Characteristic roadmap of linker governs the rational design of PROTACs. Acta Pharm. Sin. B 2024, 14, 4266–4295. [Google Scholar] [CrossRef]
- Hong, B.M.; Park, S.A.; Park, W.H. Effect of photoinitiator on chain degradation of hyaluronic acid. Biomater. Res. 2019, 23, 21. [Google Scholar] [CrossRef]
- Wan, Z.; Zhang, P.; Liu, Y.S.; Lv, L.W.; Zhou, Y.S. Four-dimensional bioprinting: Current developments and applications in bone tissue engineering. Acta Biomater. 2020, 101, 26–42. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, C.; Brady, E.; Zheng, Y.; Moore, E.; Stevens, K.R. Engineering the multiscale complexity of vascular networks. Nat. Rev. Mater. 2022, 7, 702–716. [Google Scholar] [CrossRef] [PubMed]
- Passier, M.; Bentley, K.; Loerakker, S.; Ristori, T. YAP/TAZ drives Notch and angiogenesis mechanoregulation in silico. NPJ Syst. Biol. Appl. 2024, 10, 116. [Google Scholar] [CrossRef]
- Ramot, Y.; Haim-Zada, M.; Domb, A.J.; Nyska, A. Biocompatibility and safety of PLA and its copolymers. Adv. Drug Deliv. Rev. 2016, 107, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Capuani, S.; Malgir, G.; Chua, C.Y.X.; Grattoni, A. Advanced strategies to thwart foreign body response to implantable devices. Bioeng. Transl. Med. 2022, 7, e10300. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, S.; Li, T.; Zhang, L.; Azhar, U.; Ma, J.; Zhai, C.; Zong, C.; Zhang, S. Cytocompatible and non-fouling zwitterionic hyaluronic acid-based hydrogels using thiol-ene “click” chemistry for cell encapsulation. Carbohyd. Polym. 2020, 236, 116021. [Google Scholar] [CrossRef] [PubMed]
- Suntornnond, R.; Tan, E.Y.S.; An, J.; Chua, C.K. A highly printable and biocompatible hydrogel composite for direct printing of soft and perfusable vasculature-like structures. Sci. Rep. 2017, 7, 16902. [Google Scholar] [CrossRef] [PubMed]
- Martino, M.M.; Briquez, P.S.; Ranga, A.; Lutolf, M.P.; Hubbell, J.A. Heparin-binding domain of fibrin(ogen) binds growth factors and promotes tissue repair when incorporated within a synthetic matrix. Proc. Natl. Acad. Sci. USA 2013, 110, 4563–4568. [Google Scholar] [CrossRef]
- Pal, A.; Smith, C.I.; Palade, J.; Nagaraju, S.; Alarcon-Benedetto, B.A.; Kilbourne, J.; Rawls, A.; Wilson-Rawls, J.; Vernon, B.L.; Nikkhah, M. Poly(N-isopropylacrylamide)-based dual-crosslinking biohybrid injectable hydrogels for vascularization. Acta Biomater. 2020, 107, 138–151. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.Y.; Sun, X.T.; Wang, Z.Y.; Guo, S.Y.; Yu, G.J.; Yang, H.Z. Synthesis and Properties of Gelatin Methacryloyl (GelMA) Hydrogels and Their Recent Applications in Load-Bearing Tissue. Polymers 2018, 10, 1290. [Google Scholar] [CrossRef]
- Mai, Z.; Lin, Y.; Lin, P.; Zhao, X.; Cui, L. Modulating extracellular matrix stiffness: A strategic approach to boost cancer immunotherapy. Cell Death Dis. 2024, 15, 307. [Google Scholar] [CrossRef]
- Yamamura, N.; Sudo, R.; Ikeda, M.; Tanishita, K. Effects of the mechanical properties of collagen gel on the in vitro formation of microvessel networks by endothelial cells. Tissue Eng. 2007, 13, 1443–1453. [Google Scholar] [CrossRef]
- Zanotelli, M.R.; Reinhart-King, C.A. Mechanical Forces in Tumor Angiogenesis. Adv. Exp. Med. Biol. 2018, 1092, 91–112. [Google Scholar] [PubMed]
- Grigoryan, B.; Paulsen, S.J.; Corbett, D.C.; Sazer, D.W.; Fortin, C.L.; Zaita, A.J.; Greenfield, P.T.; Calafat, N.J.; Gounley, J.P.; Ta, A.H.; et al. Multivascular networks and functional intravascular topologies within biocompatible hydrogels. Science 2019, 364, 458–464. [Google Scholar] [CrossRef]
- Han, J.; Halwachs, K.; West, T.; Larsen, B.; Sacks, M.S.; Rosales, A.M.; Zoldan, J. Matrix Stiffness Regulates Mechanotransduction and Vascular Network Formation of hiPSC-Derived Endothelial Progenitors Encapsulated in 3D Hydrogels. bioRxiv 2025. [Google Scholar] [CrossRef] [PubMed Central]
- Wu, B.; Liu, D.A.; Guan, L.; Myint, P.K.; Chin, L.; Dang, H.; Xu, Y.; Ren, J.; Li, T.; Yu, Z.; et al. Stiff matrix induces exosome secretion to promote tumour growth. Nat. Cell Biol. 2023, 25, 415–424. [Google Scholar] [CrossRef]
- Xu, Z.; Orkwis, J.A.; Harris, G.M. Cell Shape and Matrix Stiffness Impact Schwann Cell Plasticity via YAP/TAZ and Rho GTPases. Int. J. Mol. Sci. 2021, 22, 4821. [Google Scholar] [CrossRef]
- Kohn, J.C.; Zhou, D.W.; Bordeleau, F.; Zhou, A.L.; Mason, B.N.; Mitchell, M.J.; King, M.R.; Reinhart-King, C.A. Cooperative effects of matrix stiffness and fluid shear stress on endothelial cell behavior. Biophys. J. 2015, 108, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ji, L.; Wang, J.; Liu, C. Matrix stiffness regulates osteoclast fate through integrin-dependent mechanotransduction. Bioact. Mater. 2023, 27, 138–153. [Google Scholar] [CrossRef] [PubMed]
- Sant, S.; Hancock, M.J.; Donnelly, J.P.; Iyer, D.; Khademhosseini, A. Biomimetic Gradient Hydrogels for Tissue Engineering. Can. J. Chem. Eng. 2010, 88, 899–911. [Google Scholar] [CrossRef]
- Rana, D.; Padmanaban, P.; Becker, M.; Stein, F.; Leijten, J.; Koopman, B.; Rouwkema, J. Spatial control of self-organizing vascular networks with programmable aptamer-tethered growth factor photopatterning. Mater. Today Bio 2023, 19, 100551. [Google Scholar] [CrossRef]
- Wei, Z.; Schnellmann, R.; Pruitt, H.C.; Gerecht, S. Hydrogel Network Dynamics Regulate Vascular Morphogenesis. Cell Stem Cell 2020, 27, 798–812. [Google Scholar] [CrossRef]
- Sung, H.J.; Meredith, C.; Johnson, C.; Galis, Z.S. The effect of scaffold degradation rate on three-dimensional cell growth and angiogenesis. Biomaterials 2004, 25, 5735–5742. [Google Scholar] [CrossRef]
- Wang, W.Y.; Kent, R.N., 3rd; Huang, S.A.; Jarman, E.H.; Shikanov, E.H.; Davidson, C.D.; Hiraki, H.L.; Lin, D.; Wall, M.A.; Matera, D.L.; et al. Direct comparison of angiogenesis in natural and synthetic biomaterials reveals that matrix porosity regulates endothelial cell invasion speed and sprout diameter. Acta Biomater. 2021, 135, 260–273. [Google Scholar] [CrossRef]
- Barrs, R.W.; Jia, J.; Ward, M.; Richards, D.J.; Yao, H.; Yost, M.J.; Mei, Y. Engineering a Chemically Defined Hydrogel Bioink for Direct Bioprinting of Microvasculature. Biomacromolecules 2021, 22, 275–288. [Google Scholar] [CrossRef] [PubMed]
- Jongprasitkul, H.; Parihar, V.S.; Turunen, S.; Kellomäki, M. pH-Responsive Gallol-Functionalized Hyaluronic Acid-Based Tissue Adhesive Hydrogels for Injection and Three-Dimensional Bioprinting. ACS Appl. Mater. Interfaces 2023, 15, 33972–33984. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, W.; Hao, J.; Gonzales, A.; Zhao, Z.; Flores, R.S.; Kuang, X.; Mu, X.; Ching, T.; Tang, G.; et al. Molecularly cleavable bioinks facilitate high-performance digital light processing-based bioprinting of functional volumetric soft tissues. Nat. Commun. 2022, 13, 3317. [Google Scholar] [CrossRef] [PubMed]
- Chae, S.; Ha, D.-H.; Lee, H. 3D bioprinting strategy for engineering vascularized tissue models. Int. J. Bioprint. 2023, 9, 748. [Google Scholar] [CrossRef]
- Chandra, P.; Atala, A. Engineering blood vessels and vascularized tissues: Technology trends and potential clinical applications. Clin. Sci. 2019, 133, 1115–1135. [Google Scholar] [CrossRef]
- Traore, M.A.; George, S.C. Tissue engineering the vascular tree. Tissue Eng. Part B Rev. 2017, 23, 505–514. [Google Scholar] [CrossRef]
- Zhao, N.; Pessell, A.F.; Zhu, N.; Searson, P.C. Tissue-Engineered Microvessels: A Review of Current Engineering Strategies and Applications. Adv. Healthc. Mater. 2024, 13, e2303419. [Google Scholar] [CrossRef]
- Song, H.-H.G.; Rumma, R.T.; Ozaki, C.K.; Edelman, E.R.; Chen, C.S. Vascular tissue engineering: Progress, challenges, and clinical promise. Cell Stem Cell 2018, 22, 340–354. [Google Scholar] [CrossRef]
- Kolesky, D.B.; Truby, R.L.; Gladman, A.S.; Busbee, T.A.; Homan, K.A.; Lewis, J.A. 3D bioprinting of vascularized, heterogeneous cell-laden tissue constructs. Adv. Mater. 2014, 26, 3124–3130. [Google Scholar] [CrossRef]
- Miller, J.S.; Stevens, K.R.; Yang, M.T.; Baker, B.M.; Nguyen, D.-H.T.; Cohen, D.M.; Toro, E.; Chen, A.A.; Galie, P.A.; Yu, X.; et al. Rapid casting of patterned vascular networks for perfusable engineered three-dimensional tissues. Nat. Mater. 2012, 11, 768–774. [Google Scholar] [CrossRef]
- Mirabella, T.; MacArthur, J.; Cheng, D.; Ozaki, C.; Woo, Y.; Yang, M.; Chen, C. 3D-printed vascular networks direct therapeutic angiogenesis in ischaemia. Nat. Biomed. Eng. 2017, 1, 0083. [Google Scholar] [CrossRef] [PubMed]
- Homan, K.A.; Kolesky, D.B.; Skylar-Scott, M.A.; Herrmann, J.; Obuobi, H.; Moisan, A.; Lewis, J.A. Bioprinting of 3D Convoluted Renal Proximal Tubules on Perfusable Chips. Sci. Rep. 2016, 6, 34845. [Google Scholar] [CrossRef] [PubMed]
- Lin, N.Y.C.; Homan, K.A.; Robinson, S.S.; Kolesky, D.B.; Duarte, N.; Moisan, A.; Lewis, J.A. Renal reabsorption in 3D vascularized proximal tubule models. Proc. Natl. Acad. Sci. USA 2019, 116, 5399–5404. [Google Scholar] [CrossRef] [PubMed]
- Winkelman, M.A.; Kim, D.Y.; Kakarla, S.; Grath, A.; Silvia, N.; Dai, G. Interstitial flow enhances the formation, connectivity, and function of 3D brain microvascular networks generated within a microfluidic device. Lab Chip 2021, 22, 170–192. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, Y.; Zhu, J.; Wang, J.; Jiang, Z.; Zhao, C.; Yang, Q.; Huang, Y.; Yao, W.; Pang, W. Laminar flow protects vascular endothelial tight junctions and barrier function via maintaining the expression of long non-coding RNA MALAT1. Front. Bioeng. Biotechnol. 2020, 8, 647. [Google Scholar] [CrossRef]
- Park, J.Y.; White, J.B.; Walker, N.; Kuo, C.H.; Cha, W.; Meyerhoff, M.E.; Takayama, S. Responses of endothelial cells to extremely slow flows. Biomicrofluidics 2011, 5, 22211. [Google Scholar] [CrossRef]
- Cheng, K.-C.; Theato, P.; Hsu, S.-h. 3D-bioprintable endothelial cell-laden sacrificial ink for fabrication of microvessel networks. Biofabrication 2023, 15, 045026. [Google Scholar] [CrossRef]
- Cai, B.; Kilian, D.; Ghorbani, S.; Roth, J.G.; Seymour, A.J.; Brunel, L.G.; Mejia, D.R.; Rios, R.J.; Szabo, I.M.; Iranzo, S.C.; et al. One-step bioprinting of endothelialized, self-supporting arterial and venous networks. Biofabrication 2025, 17, 025012. [Google Scholar] [CrossRef]
- Nie, J.; Gao, Q.; Xie, C.; Lv, S.; Qiu, J.; Liu, Y.; Guo, M.; Guo, R.; Fu, J.; He, Y. Construction of multi-scale vascular chips and modelling of the interaction between tumours and blood vessels. Mater. Horiz. 2020, 7, 82–92. [Google Scholar] [CrossRef]
- Zheng, F.; Derby, B.; Wong, J. Fabrication of microvascular constructs using high resolution electrohydrodynamic inkjet printing. Biofabrication 2021, 13, 035006. [Google Scholar] [CrossRef]
- Gao, Q.; He, Y.; Fu, J.-z.; Liu, A.; Ma, L. Coaxial nozzle-assisted 3D bioprinting with built-in microchannels for nutrients delivery. Biomaterials 2015, 61, 203–215. [Google Scholar] [CrossRef]
- Xu, H.; Su, Y.; Liao, Z.; Liu, Z.; Huang, X.; Zhao, L.; Duan, R.; Hu, Y.; Wei, Y.; Lian, X.; et al. Coaxial bioprinting vascular constructs: A review. Eur. Polym. J. 2022, 179, 111549. [Google Scholar] [CrossRef]
- Gao, Q.; Liu, Z.; Lin, Z.; Qiu, J.; Liu, Y.; Liu, A.; Wang, Y.; Xiang, M.; Chen, B.; Fu, J.; et al. 3D Bioprinting of Vessel-like Structures with Multilevel Fluidic Channels. ACS Biomater. Sci. Eng. 2017, 3, 399–408. [Google Scholar] [CrossRef]
- Jia, W.; Gungor-Ozkerim, P.S.; Zhang, Y.S.; Yue, K.; Zhu, K.; Liu, W.; Pi, Q.; Byambaa, B.; Dokmeci, M.R.; Shin, S.R.; et al. Direct 3D bioprinting of perfusable vascular constructs using a blend bioink. Biomaterials 2016, 106, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Park, W.; Kim, B.S.; Ahn, M.; Chae, S.; Cho, W.W.; Kim, J.; Lee, J.Y.; Jang, J.; Cho, D.W. Construction of a novel in vitro atherosclerotic model from geometry-tunable artery equivalents engineered via in-bath coaxial cell printing. Adv. Funct. Mater. 2020, 31, 2008878. [Google Scholar] [CrossRef]
- Gao, G.; Lee, J.H.; Jang, J.; Lee, D.H.; Kong, J.-S.; Kim, B.S.; Choi, Y.-J.; Jang, W.B.; Hong, Y.J.; Kwon, S.-M.; et al. Tissue Engineered Bio-Blood-Vessels Constructed Using a Tissue-Specific Bioink and 3D Coaxial Cell Printing Technique: A Novel Therapy for Ischemic Disease. Adv. Funct. Mater. 2017, 27, 1700798. [Google Scholar] [CrossRef]
- Gao, G.; Kim, H.; Kim, B.S.; Kong, J.S.; Lee, J.Y.; Park, B.W.; Chae, S.; Kim, J.; Ban, K.; Jang, J.; et al. Tissue-engineering of vascular grafts containing endothelium and smooth-muscle using triple-coaxial cell printing. Appl. Phys. Rev. 2019, 6, 041402. [Google Scholar] [CrossRef]
- Shao, L.; Gao, Q.; Zhao, H.; Xie, C.; Fu, J.; Liu, Z.; Xiang, M.; He, Y. Fiber-Based Mini Tissue with Morphology-Controllable GelMA Microfibers. Small 2018, 14, e1802187. [Google Scholar] [CrossRef]
- Khattak, S.F.; Bhatia, S.R.; Roberts, S.C. Pluronic F127 as a cell encapsulation material: Utilization of membrane-stabilizing agents. Tissue Eng. 2005, 11, 974–983. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Park, J.Y.; Kim, B.S.; Jang, J.; Cho, D.-W. Coaxial Cell Printing of Freestanding, Perfusable, and Functional In Vitro Vascular Models for Recapitulation of Native Vascular Endothelium Pathophysiology. Adv. Healthc. Mater. 2018, 7, e1801102. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Liu, L.; Ouyang, J.; Li, X.; Zhang, X.; Lan, Q.; Xu, T. Coaxial 3D bioprinting of self-assembled multicellular heterogeneous tumor fibers. Sci. Rep. 2017, 7, 1457. [Google Scholar] [CrossRef]
- Singh, N.K.; Han, W.; Nam, S.A.; Kim, J.W.; Kim, J.Y.; Kim, Y.K.; Cho, D.-W. Three-dimensional cell-printing of advanced renal tubular tissue analogue. Biomaterials 2020, 232, 119734. [Google Scholar] [CrossRef]
- Singh, N.K.; Kim, J.Y.; Lee, J.Y.; Lee, H.; Gao, G.; Jang, J.; Kim, Y.K.; Cho, D.-W. Coaxial cell printing of a human glomerular model: An in vitro glomerular filtration barrier and its pathophysiology. Biofabrication 2023, 15, 024101. [Google Scholar] [CrossRef]
- Park, W.; Choi, M.-J.; Lee, J.-S.; Ahn, M.; Han, W.; Gao, G.; Cho, D.-W.; Kim, B.S. Embedded 3D-Coaxial Bioprinting of Stenotic Brain Vessels with a Mechanically Enhanced Extracellular Matrix Bioink for Investigating Hemodynamic Force-Induced Endothelial Responses. Adv. Funct. Mater. 2025, e04276. [Google Scholar] [CrossRef]
- Banigo, A.T.; Nauta, L.; Zoetebier, B.; Karperien, M. Hydrogel-Based Bioinks for Coaxial and Triaxial Bioprinting: A Review of Material Properties, Printing Techniques, and Applications. Polymers 2025, 17, 917. [Google Scholar] [CrossRef]
- Mohan, T.S.; Datta, P.; Nesaei, S.; Ozbolat, V.; Ozbolat, I.T. 3D coaxial bioprinting: Process mechanisms, bioinks and applications. Prog. Biomed. Eng. 2022, 4, 022003. [Google Scholar] [CrossRef]
- Budharaju, H.; Sundaramurthi, D.; Sethuraman, S. Embedded 3D bioprinting—An emerging strategy to fabricate biomimetic & large vascularized tissue constructs. Bioact. Mater. 2024, 32, 356–384. [Google Scholar]
- Shiwarski, D.J.; Hudson, A.R.; Tashman, J.W.; Feinberg, A.W. Emergence of FRESH 3D printing as a platform for advanced tissue biofabrication. APL Bioeng. 2021, 5, 010904. [Google Scholar] [CrossRef] [PubMed]
- Shiwarski, D.J.; Hudson, A.R.; Tashman, J.W.; Bakirci, E.; Moss, S.; Coffin, B.D.; Feinberg, A.W. 3D bioprinting of collagen-based high-resolution internally perfusable scaffolds for engineering fully biologic tissue systems. Sci. Adv. 2025, 11, eadu5905. [Google Scholar] [CrossRef]
- Bhattacharjee, T.; Zehnder, S.M.; Rowe, K.G.; Jain, S.; Nixon, R.M.; Sawyer, W.G.; Angelini, T.E. Writing in the granular gel medium. Sci. Adv. 2015, 1, e1500655. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Qi, C.; Zhang, W.; Zhou, H.; Wu, N.; Yang, M.; Meng, S.; Liu, Z.; Kong, T. In Situ Endothelialization of Free-Form 3D Network of Interconnected Tubular Channels via Interfacial Coacervation by Aqueous-in-Aqueous Embedded Bioprinting. Adv. Mater. 2023, 35, e2209263. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Guo, Y.; Wu, B.; Liu, Z.; Ye, M.; Xu, Y.; Ji, M.; Chen, L.; Lu, B.; Nie, K.; et al. Expanding Embedded 3D Bioprinting Capability for Engineering Complex Organs with Freeform Vascular Networks. Adv. Mater. 2023, 35, e2205082. [Google Scholar] [CrossRef]
- Lee, A.; Hudson, A.R.; Shiwarski, D.J.; Tashman, J.W.; Hinton, T.J.; Yerneni, S.; Bliley, J.M.; Campbell, P.G.; Feinberg, A.W. 3D bioprinting of collagen to rebuild components of the human heart. Science 2019, 365, 482–487. [Google Scholar] [CrossRef]
- Noor, N.; Shapira, A.; Edri, R.; Gal, I.; Wertheim, L.; Dvir, T. 3D Printing of Personalized Thick and Perfusable Cardiac Patches and Hearts. Adv. Sci. 2019, 6, 1900344. [Google Scholar] [CrossRef]
- Moss, S.P.; Shiwarski, D.J.; Feinberg, A.W. FRESH 3D Bioprinting of Collagen Types, I, II, and III. ACS Biomater. Sci. Eng. 2024, 11, 556–563. [Google Scholar] [CrossRef]
- Cheng, Y.-W.; Shiwarski, D.J.; Ball, R.L.; Whitehead, K.A.; Feinberg, A.W. Engineering Aligned Skeletal Muscle Tissue Using Decellularized Plant-Derived Scaffolds. ACS Biomater. Sci. Eng. 2020, 6, 3046–3054. [Google Scholar] [CrossRef]
- Stankey, P.P.; Kroll, K.T.; Ainscough, A.J.; Reynolds, D.S.; Elamine, A.; Fichtenkort, B.T.; Uzel, S.G.M.; Lewis, J.A. Embedding Biomimetic Vascular Networks via Coaxial Sacrificial Writing into Functional Tissue. Adv. Mater. 2024, 36, e2401528. [Google Scholar] [CrossRef]
- Brunel, L.G.; Hull, S.M.; Heilshorn, S.C. Engineered assistive materials for 3D bioprinting: Support baths and sacrificial inks. Biofabrication 2022, 14, 032001. [Google Scholar] [CrossRef] [PubMed]
- Brunel, L.G.; Christakopoulos, F.; Kilian, D.; Cai, B.; Hull, S.M.; Myung, D.; Heilshorn, S.C. Embedded 3D bioprinting of collagen inks into microgel baths to control hydrogel microstructure and cell spreading. Adv. Healthc. Mater. 2024, 13, e2303325. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Schimelman, J.; Wang, P.; Miller, K.L.; Ma, X.; You, S.; Guan, J.; Sun, B.; Zhu, W.; Chen, S. Photopolymerizable Biomaterials and Light-Based 3D Printing Strategies for Biomedical Applications. Chem. Rev. 2020, 120, 10695–10743. [Google Scholar] [CrossRef]
- Arakawa, C.; Gunnarsson, C.; Howard, C.; Bernabeu, M.; Phong, K.; Yang, E.; DeForest, C.A.; Smith, J.D.; Zheng, Y. Biophysical and biomolecular interactions of malaria-infected erythrocytes in engineered human capillaries. Sci. Adv. 2020, 6, eaay7243. [Google Scholar] [CrossRef]
- Rayner, S.G.; Howard, C.C.; Mandrycky, C.J.; Stamenkovic, S.; Himmelfarb, J.; Shih, A.Y.; Zheng, Y. Multiphoton-Guided Creation of Complex Organ-Specific Microvasculature. Adv. Healthc. Mater. 2021, 10, e2100031. [Google Scholar] [CrossRef] [PubMed]
- Dobos, A.; Gantner, F.; Markovic, M.; Van Hoorick, J.; Tytgat, L.; Van Vlierberghe, S.; Ovsianikov, A. On-chip high-definition bioprinting of microvascular structures. Biofabrication 2021, 13, 015016. [Google Scholar] [CrossRef] [PubMed]
- Brandenberg, N.; Lutolf, M.P. In Situ Patterning of Microfluidic Networks in 3D Cell-Laden Hydrogels. Adv. Mater. 2016, 28, 7450–7456. [Google Scholar] [CrossRef]
- Rizzo, R.; Rütsche, D.; Liu, H.; Chansoria, P.; Wang, A.; Hasenauer, A.; Zenobi-Wong, M. Multiscale Hybrid Fabrication: Volumetric Printing Meets Two-Photon Ablation. Adv. Mater. Technol. 2023, 8, 2201871. [Google Scholar] [CrossRef]
- Alparslan, C.; Bayraktar, Ş. Advances in Digital Light Processing (DLP) Bioprinting: A Review of Biomaterials and Its Applications, Innovations, Challenges, and Future Perspectives. Polymers 2025, 17, 1287. [Google Scholar] [CrossRef]
- Lai, Y.; Xiao, X.; Huang, Z.; Duan, H.; Yang, L.; Yang, Y.; Li, C.; Feng, L. Photocrosslinkable Biomaterials for 3D Bioprinting: Mechanisms, Recent Advances, and Future Prospects. Int. J. Mol. Sci. 2024, 25, 12567. [Google Scholar] [CrossRef]
- Wang, Z.; Kumar, H.; Tian, Z.; Jin, X.; Holzman, J.F.; Menard, F.; Kim, K. Visible Light Photoinitiation of Cell-Adhesive Gelatin Methacryloyl Hydrogels for Stereolithography 3D Bioprinting. ACS Appl. Mater. Inter. 2018, 10, 26859–26869. [Google Scholar] [CrossRef]
- Sharifi, S.; Sharifi, H.; Akbari, A.; Chodosh, J. Systematic optimization of visible light-induced crosslinking conditions of gelatin methacryloyl (GelMA). Sci. Rep. 2021, 11, 23276. [Google Scholar] [CrossRef]
- Noshadi, I.; Hong, S.; Sullivan, K.E.; Sani, E.S.; Portillo-Lara, R.; Tamayol, A.; Shin, S.R.; Gao, A.E.; Stoppel, W.L.; Black III, L.D. In vitro and in vivo analysis of visible light crosslinkable gelatin methacryloyl (GelMA) hydrogels. Biomater. Sci. 2017, 5, 2093–2105. [Google Scholar] [CrossRef] [PubMed]
- Madrid-Wolff, J.; Toombs, J.; Rizzo, R.; Bernal, P.N.; Porcincula, D.; Walton, R.; Wang, B.; Kotz-Helmer, F.; Yang, Y.; Kaplan, D.; et al. A review of materials used in tomographic volumetric additive manufacturing. MRS Commun. 2023, 13, 764–785. [Google Scholar] [CrossRef] [PubMed]
- Goodarzi Hosseinabadi, H.; Dogan, E.; Miri, A.K.; Ionov, L. Digital Light Processing Bioprinting Advances for Microtissue Models. ACS Biomater. Sci. Eng. 2022, 8, 1381–1395. [Google Scholar] [CrossRef]
- Mathur, V.; Dsouza, V.; Srinivasan, V.; Vasanthan, K.S. Volumetric Additive Manufacturing for Cell Printing: Bridging Industry Adaptation and Regulatory Frontiers. ACS Biomater. Sci. Eng. 2025, 11, 156–181. [Google Scholar] [CrossRef] [PubMed]
- Ryma, M.; Genç, H.; Nadernezhad, A.; Paulus, I.; Schneidereit, D.; Friedrich, O.; Andelovic, K.; Lyer, S.; Alexiou, C.; Cicha, I. A print-and-fuse strategy for sacrificial filaments enables biomimetically structured perfusable microvascular networks with functional endothelium inside 3D hydrogels. Adv. Mater. 2022, 34, e2200653. [Google Scholar] [CrossRef]
- Meng, F.; Meyer, C.M.; Joung, D.; Vallera, D.A.; McAlpine, M.C.; Panoskaltsis-Mortari, A. 3D Bioprinted In Vitro Metastatic Models via Reconstruction of Tumor Microenvironments. Adv. Mater. 2019, 31, e1806899. [Google Scholar] [CrossRef]
- Lee, V.K.; Lanzi, A.M.; Ngo, H.; Yoo, S.-S.; Vincent, P.A.; Dai, G. Generation of Multi-scale Vascular Network System Within 3D Hydrogel Using 3D Bio-printing Technology. Cell. Mol. Bioeng. 2014, 7, 460–472. [Google Scholar] [CrossRef] [PubMed]
- Park, W.; Lee, J.-S.; Gao, G.; Kim, B.S.; Cho, D.-W. 3D bioprinted multilayered cerebrovascular conduits to study cancer extravasation mechanism related with vascular geometry. Nat. Commun. 2023, 14, 7696. [Google Scholar] [CrossRef]
- Davoodi, E.; Sarikhani, E.; Montazerian, H.; Ahadian, S.; Costantini, M.; Swieszkowski, W.; Willerth, S.M.; Walus, K.; Mofidfar, M.; Toyserkani, E.; et al. Extrusion and Microfluidic-Based Bioprinting to Fabricate Biomimetic Tissues and Organs. Adv. Mater. Technol. 2020, 5, 1901044. [Google Scholar] [CrossRef]
- Teixeira, M.C.; Lameirinhas, N.S.; Carvalho, J.P.F.; Silvestre, A.J.D.; Vilela, C.; Freire, C.S.R. A Guide to Polysaccharide-Based Hydrogel Bioinks for 3D Bioprinting Applications. Int. J. Mol. Sci. 2022, 23, 6564. [Google Scholar] [CrossRef]
- Ng, W.L.; Goh, M.H.; Yeong, W.Y.; Naing, M.W. Applying macromolecular crowding to 3D bioprinting: Fabrication of 3D hierarchical porous collagen-based hydrogel constructs. Biomater. Sci. 2018, 6, 562–574. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Jeong, W.; Lee, S.M.; Kim, J.B.; Jin, S.; Kang, H.-W. Decellularized extracellular matrix-based bio-ink with enhanced 3D printability and mechanical properties. Biofabrication 2020, 12, 025003. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, H.; Lee, G.; Ryu, D.; Kim, G. Fabrication of fully aligned self-assembled cell-laden collagen filaments for tissue engineering via a hybrid bioprinting process. Bioact. Mater. 2024, 36, 14–29. [Google Scholar] [CrossRef]
- Gong, X.; Wen, Z.; Liang, Z.; Xiao, H.; Lee, S.; Rossello-Martinez, A.; Xing, Q.; Wright, T.; Nguyen, R.Y.; Mak, M. Instant assembly of collagen for tissue engineering and bioprinting. Nat. Mater. 2025, 24, 1307–1318. [Google Scholar] [CrossRef]
- Potente, M.; Mäkinen, T. Vascular heterogeneity and specialization in development and disease. Nat. Rev. Mol. Cell Biol. 2017, 18, 477–494. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wan, Z.; Pavlou, G.; Zhong, A.X.; Xu, L.; Kamm, R.D. Interstitial flow promotes the formation of functional microvascular networks in vitro through upregulation of matrix metalloproteinase-2. Adv. Funct. Mater. 2022, 32, 2206767. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Chung, M.; Ahn, J.; Lee, S.; Jeon, N.L. Interstitial flow regulates the angiogenic response and phenotype of endothelial cells in a 3D culture model. Lab Chip 2016, 16, 4189–4199. [Google Scholar] [CrossRef]
- Mote, N.; Kubik, S.; Polacheck, W.J.; Baker, B.M.; Trappmann, B. A nanoporous hydrogel-based model to study chemokine gradient-driven angiogenesis under luminal flow. Lab Chip 2024, 24, 4892–4906. [Google Scholar] [CrossRef]
- Galie, P.A.; Nguyen, D.-H.T.; Choi, C.K.; Cohen, D.M.; Janmey, P.A.; Chen, C.S. Fluid shear stress threshold regulates angiogenic sprouting. Proc. Natl. Acad. Sci. USA 2014, 111, 7968–7973. [Google Scholar] [CrossRef]
- Barrs, R.W.; Jia, J.; Silver, S.E.; Yost, M.; Mei, Y. Biomaterials for Bioprinting Microvasculature. Chem. Rev. 2020, 120, 10887–10949. [Google Scholar] [CrossRef]
- Lammers, A.; Hsu, H.-H.; Sundaram, S.; Gagnon, K.A.; Kim, S.; Lee, J.H.; Tung, Y.-C.; Eyckmans, J.; Chen, C.S. Rapid tissue perfusion using sacrificial percolation of anisotropic networks. Matter 2024, 7, 2184–2204. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, S.; Lee, J.H.; Bjørge, I.M.; Michas, C.; Kim, S.; Lammers, A.; Mano, J.F.; Eyckmans, J.; White, A.E.; Chen, C.S. Sacrificial capillary pumps to engineer multiscalar biological forms. Nature 2024, 636, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Zeenat, L.; Zolfagharian, A.; Sriya, Y.; Sasikumar, S.; Bodaghi, M.; Pati, F. 4D Printing for Vascular Tissue Engineering: Progress and Challenges. Adv. Mater. Technol. 2023, 8, 2300200. [Google Scholar] [CrossRef]


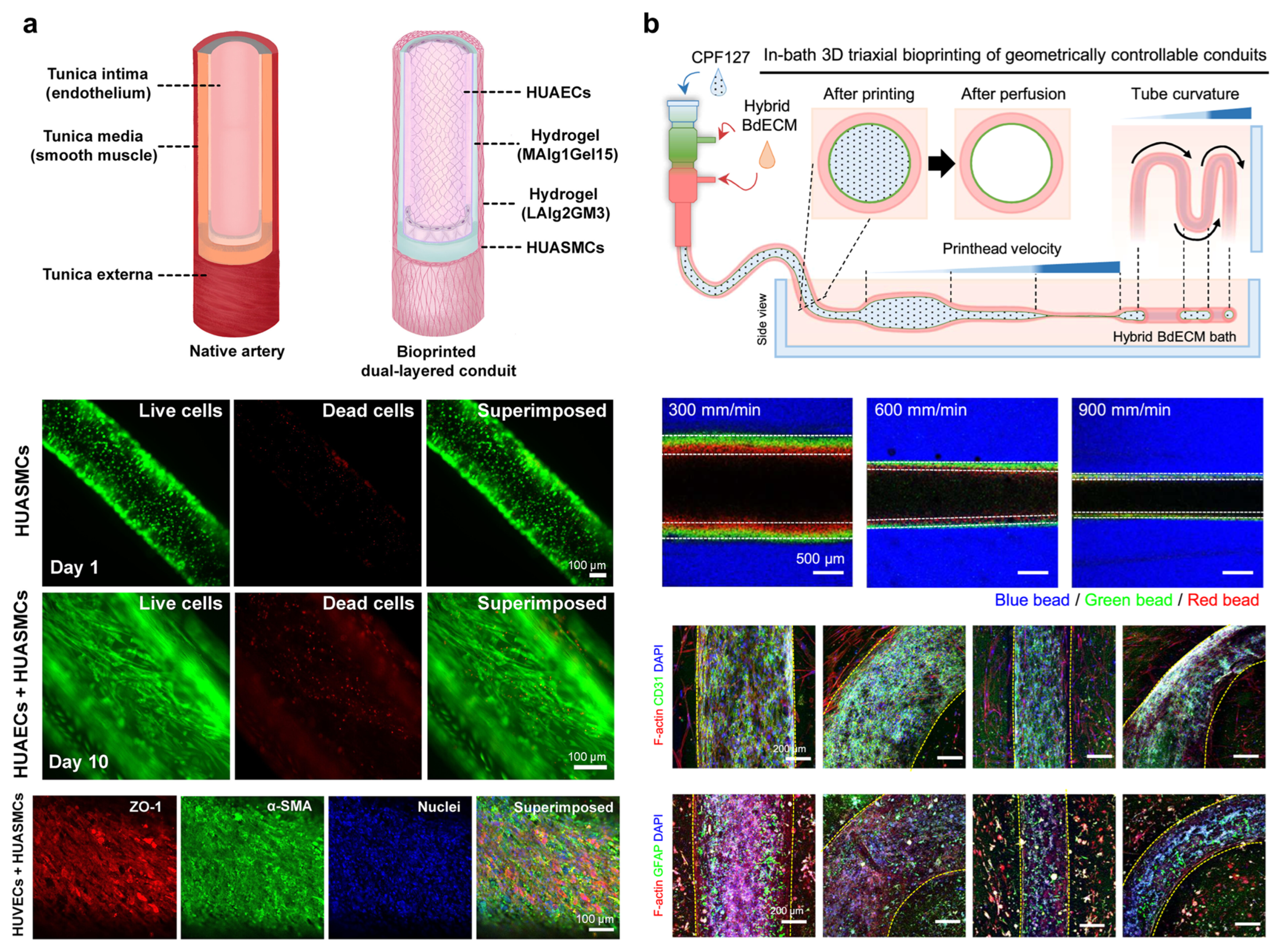


| Feature | Large-Caliber Vessels | Microvascular Networks |
|---|---|---|
| Diameter | >1 mm (≥6 mm for grafts) | <100 μm |
| Primary Role | Bulk perfusion, anastomosis | Oxygen/nutrient exchange |
| Key Functional Metrics | Patency, burst pressure | Lumen stability, remodeling |
| Clinical Risks | Clotting, intimal hyperplasia | Leakiness, perfusion failure |
| Material Needs | High strength, hemocompatible | Degradable, cell-responsive |
| Example Applications | Coronary bypass, AV grafts, tendon/flap repair | Skin grafts, islet implants |
| Functional Role | Attribution | Hydrogels |
|---|---|---|
| Endothelialization | Adhesion, spreading, lumen formation | Fibrin, collagen, GelMA, dECM [80,81] |
| Bioactivity | RGD, heparin, angiogenic factor binding | RGD-GelMA, heparin-GelMA, fibrin [78,79] |
| Mechanical Properties | Stiffness, viscoelasticity, and mechanical resistance | PEGDA blends, GelMA composites, methacrylated ECM [82,83,84,85] |
| Degradation Compatibility | MMP-cleavable, hydrolytic, photodegradable | GelMA, HA-tyramine, PEG-MMP [86,87,88] |
| ECM Mimicry | Alignment, porosity, matrix composition | Collagen, fibrin, dECM [86,89,90,91]. |
| Surface Chemistry Immune Modulation | Zwitterions, charge, macrophage polarization, reduce foreign body reaction | PVA, zwitterionic hydrogels, sericin [92,93,94] |
| Strategy | Resolution | Scalability | Material Versatility | ECM Fidelity | Compatibility with Bottom-Up Strategies | References |
|---|---|---|---|---|---|---|
| Sacrificial Printing | Moderate (~100–500 µm) | Moderate–High | High–wide ink compatibility | Moderate–High | High | [13,20,121,122,123,124,125,126,127,128,129,130,131,132] |
| Coaxial Extrusion | Low (~500–1000 µm) | High: continuous strand, rapid | Limited: mainly alginate-based materials | Low–moderate: limited ECM mimicry | Low | [6,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148] |
| Embedded Printing | High (≤20 µm) | Moderate–High: Scalable but time-intensive | Very High: compatible with soft ECM bioinks | High: native-like composition and stiffness | Moderate–High | [15,19,149,150,151,152,153,154,155,156,157,158,159,160,161] |
| Light-Based Printing | Very High (≤10 µm) | Low–Moderate: high speed but small volume | Low: constrained to photopolymerizable inks | Low–Moderate: depends on photoink tuning | Not necessary | [86,162,163,164,165,166,167,168,169,170,171,172,173,174,175] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Son, J.; Li, S.; Jeong, W. Bioprinting Vascularized Constructs for Clinical Relevance: Engineering Hydrogel Systems for Biological Maturity. Gels 2025, 11, 636. https://doi.org/10.3390/gels11080636
Son J, Li S, Jeong W. Bioprinting Vascularized Constructs for Clinical Relevance: Engineering Hydrogel Systems for Biological Maturity. Gels. 2025; 11(8):636. https://doi.org/10.3390/gels11080636
Chicago/Turabian StyleSon, Jeonghyun, Siyuan Li, and Wonwoo Jeong. 2025. "Bioprinting Vascularized Constructs for Clinical Relevance: Engineering Hydrogel Systems for Biological Maturity" Gels 11, no. 8: 636. https://doi.org/10.3390/gels11080636
APA StyleSon, J., Li, S., & Jeong, W. (2025). Bioprinting Vascularized Constructs for Clinical Relevance: Engineering Hydrogel Systems for Biological Maturity. Gels, 11(8), 636. https://doi.org/10.3390/gels11080636






