Preparation of Bismuth Vanadates with Rich Oxygen Vacancies Using Different Sol pH and Their Photocatalytic Behavior in Degradation of Methylene Blue
Abstract
1. Introduction
2. Results and Discussion
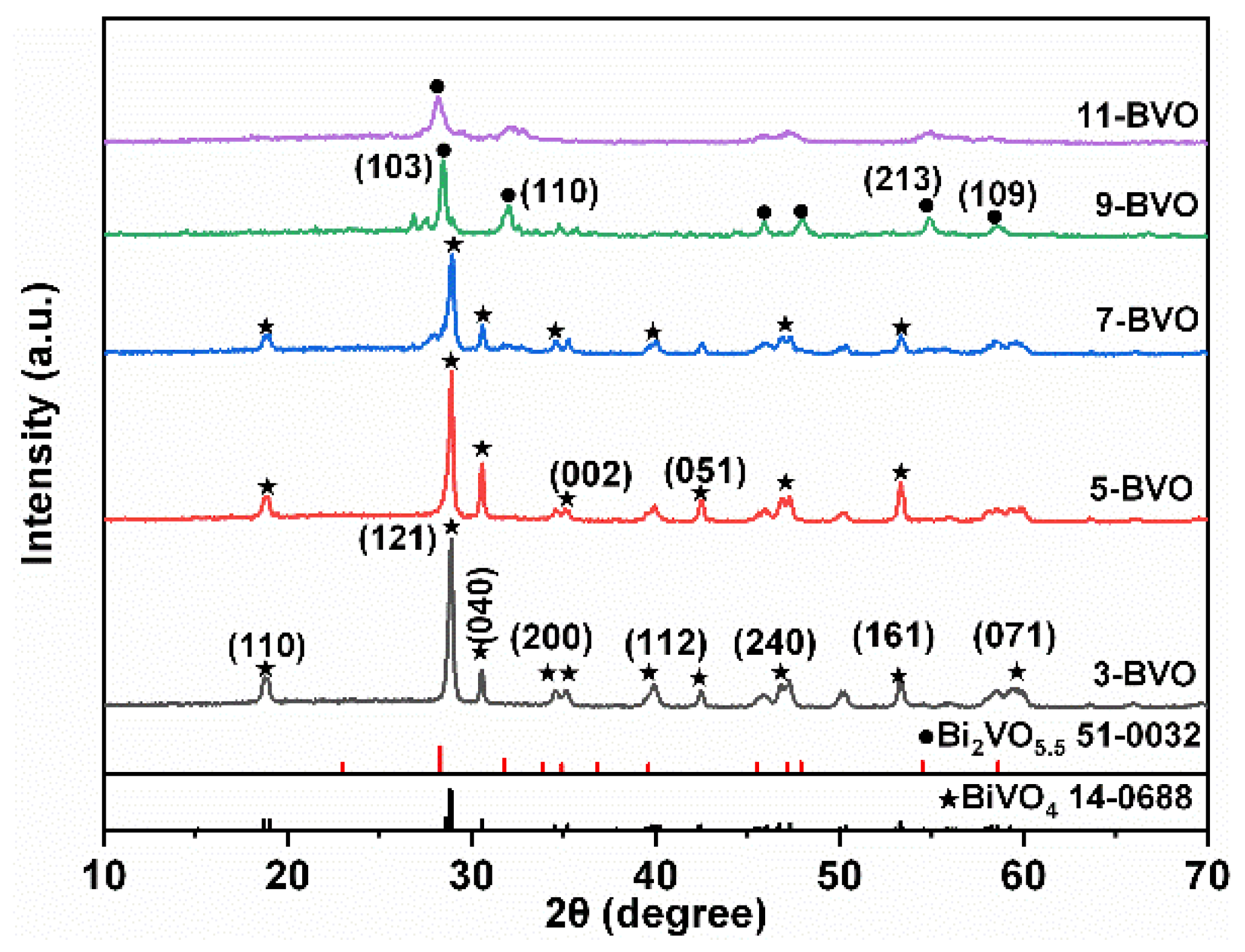
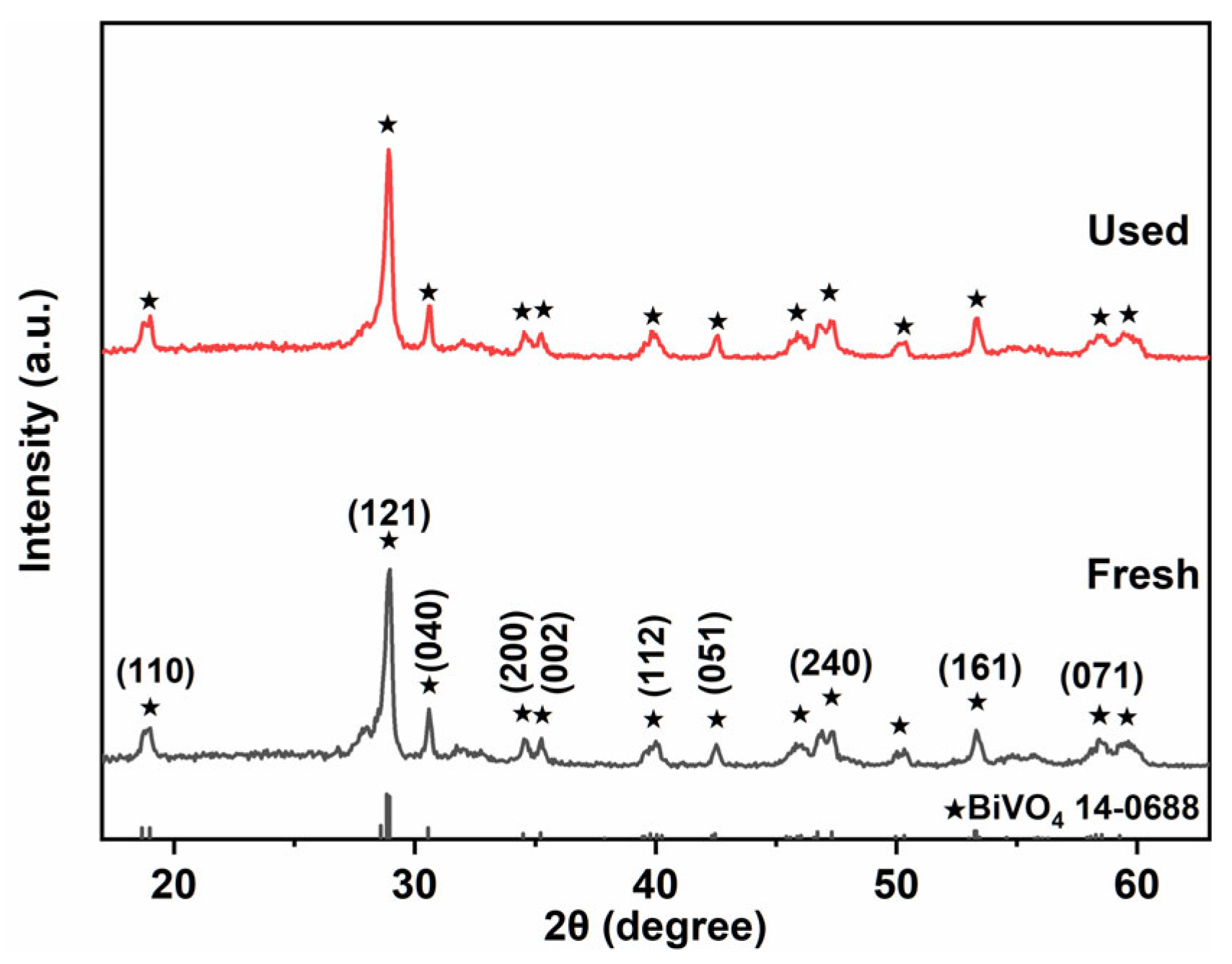
2.1. Crystal Structure and Chemical Bond
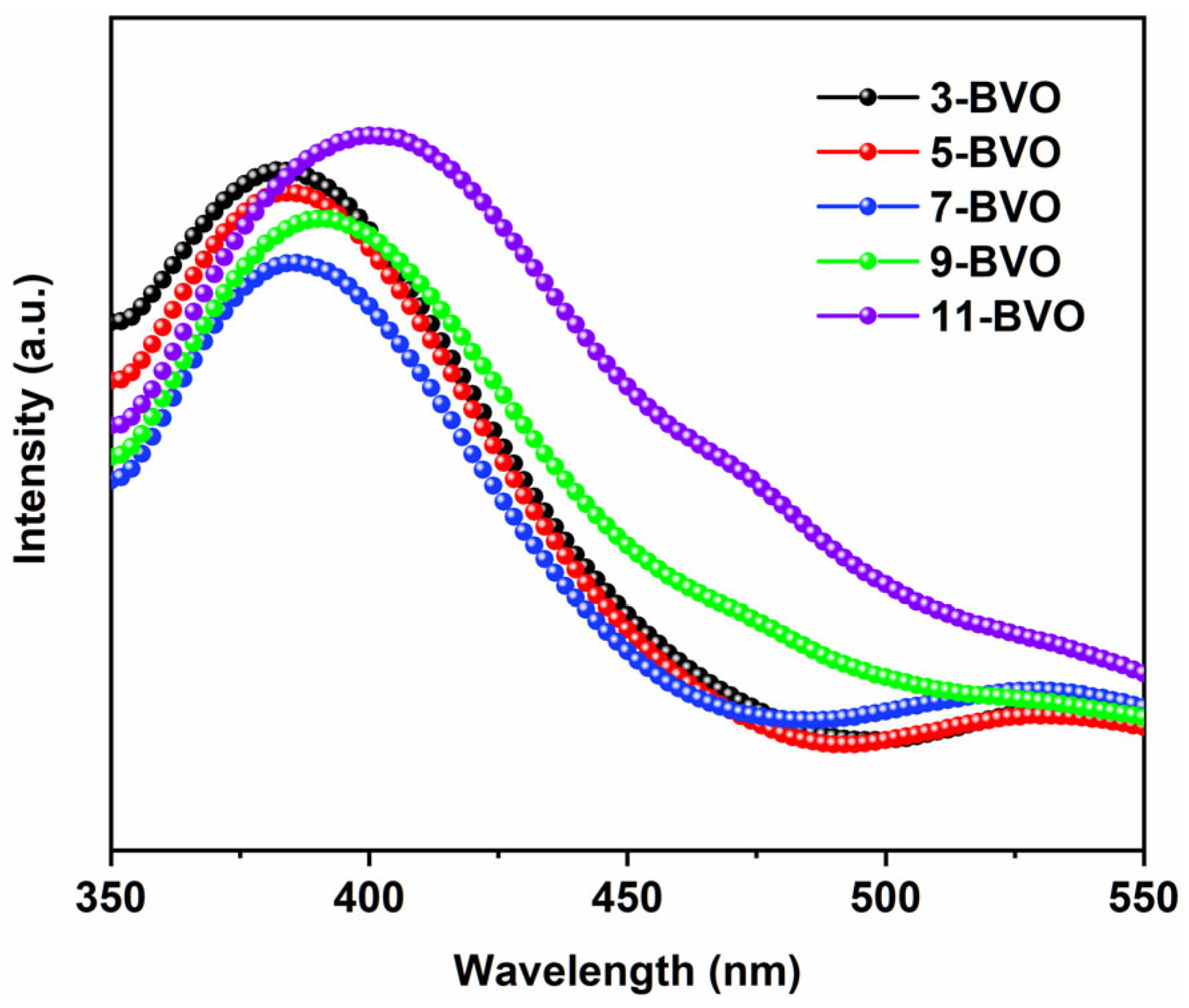
2.2. Chemical Valence States Analysis
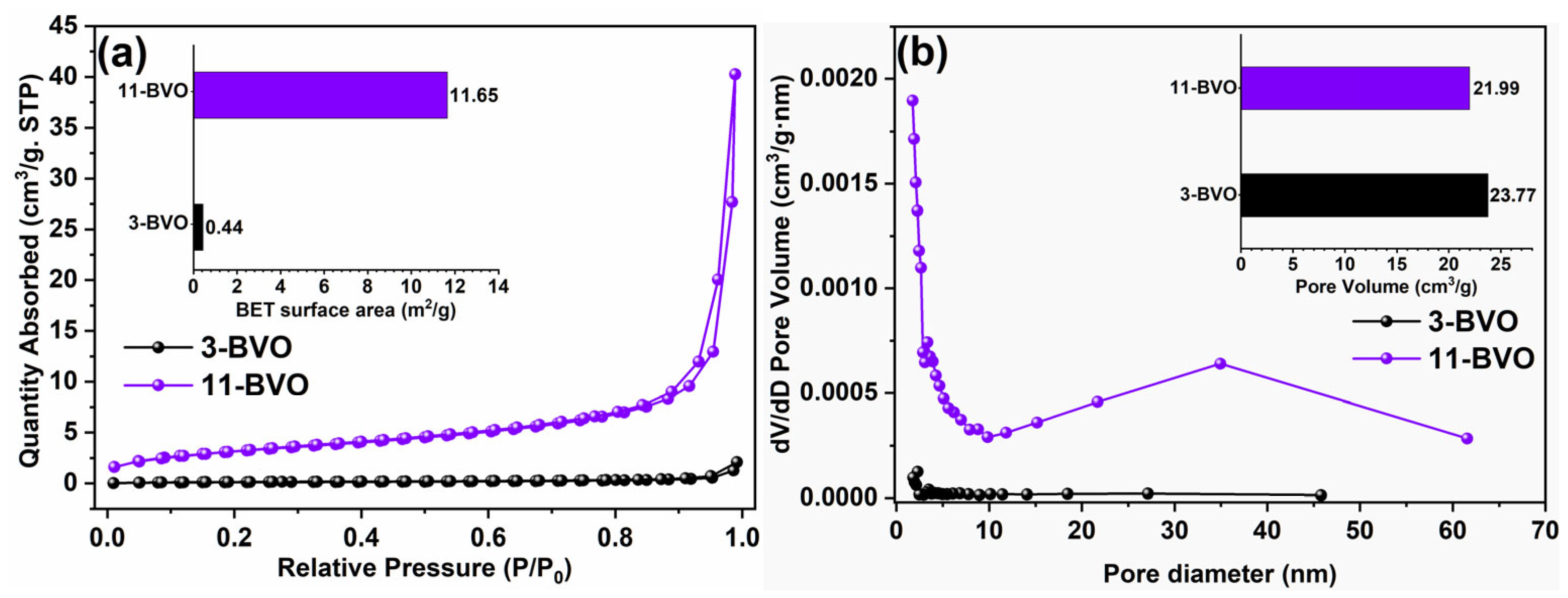
2.3. Morphology and Specific Surface Area Analysis
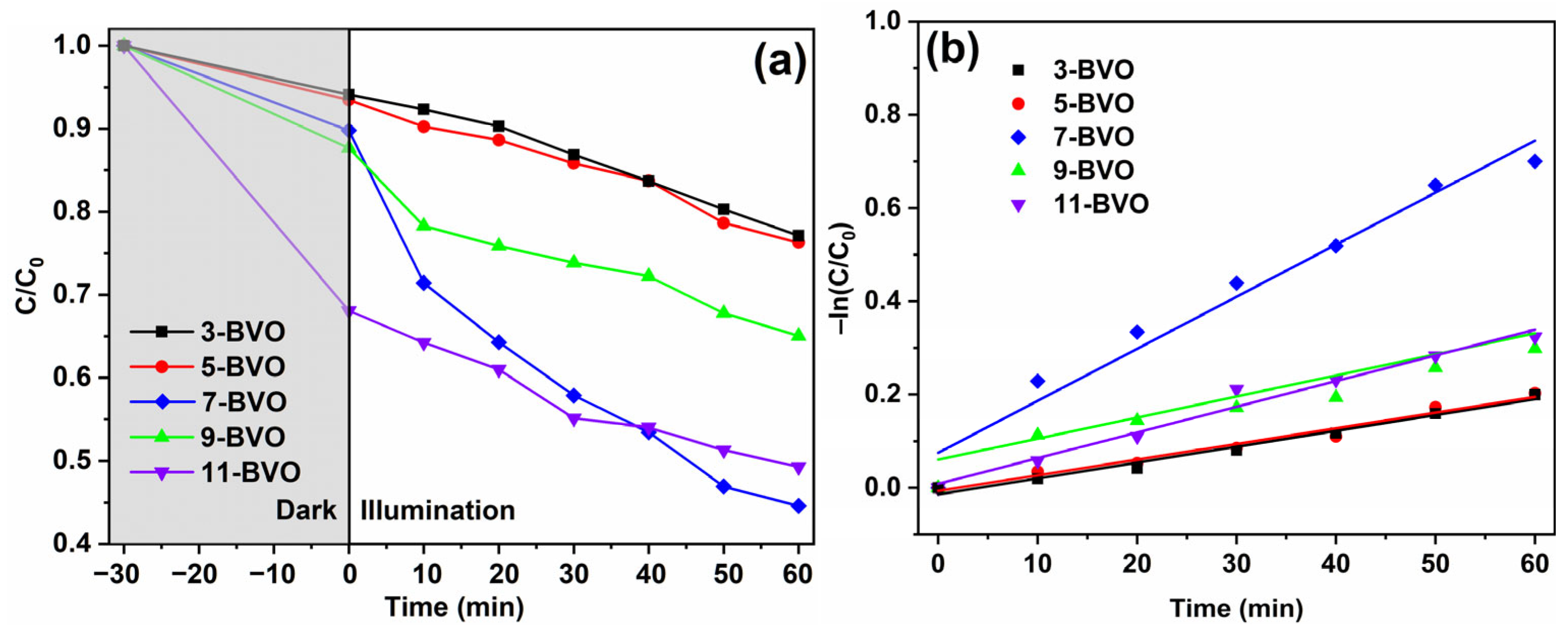
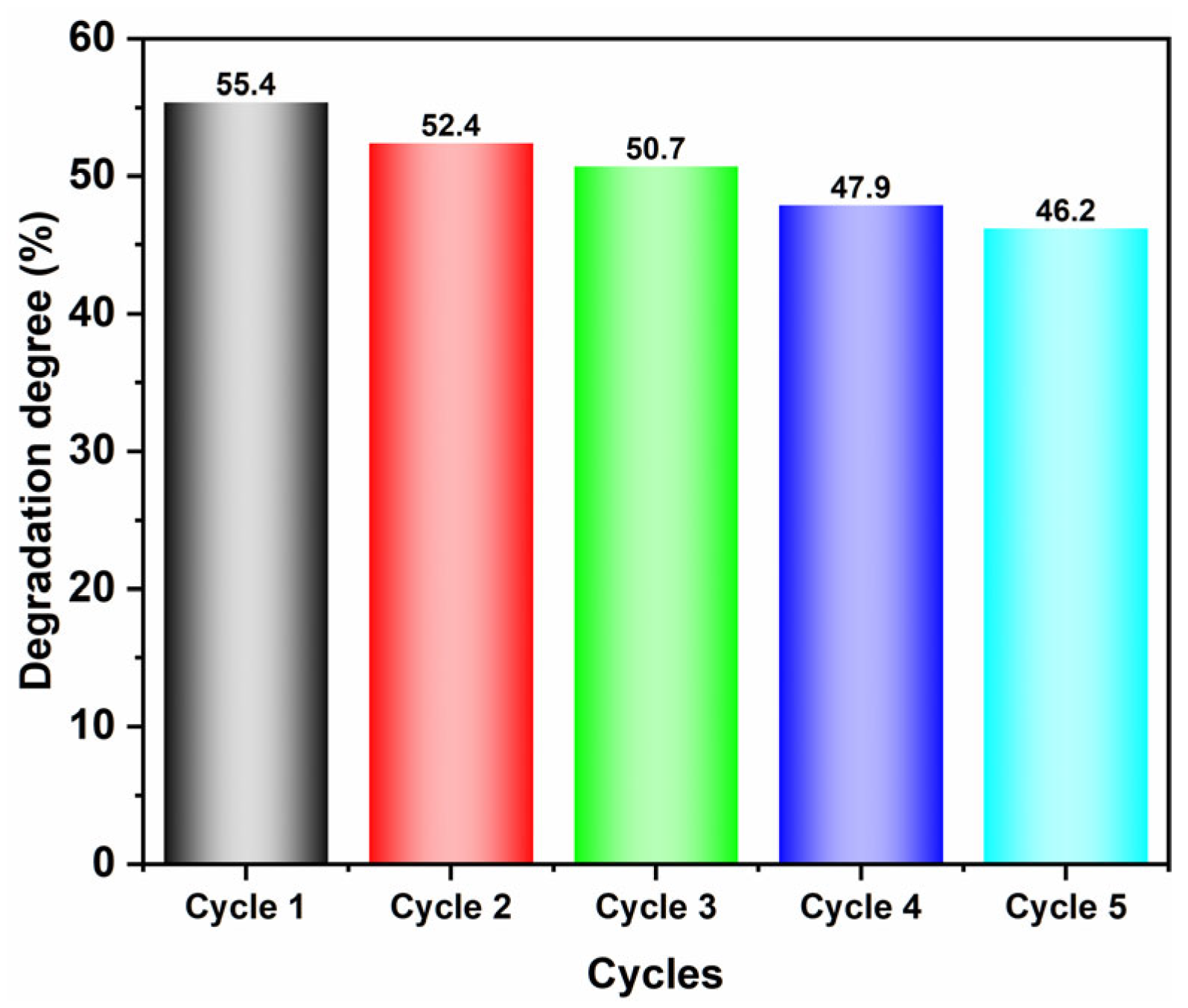
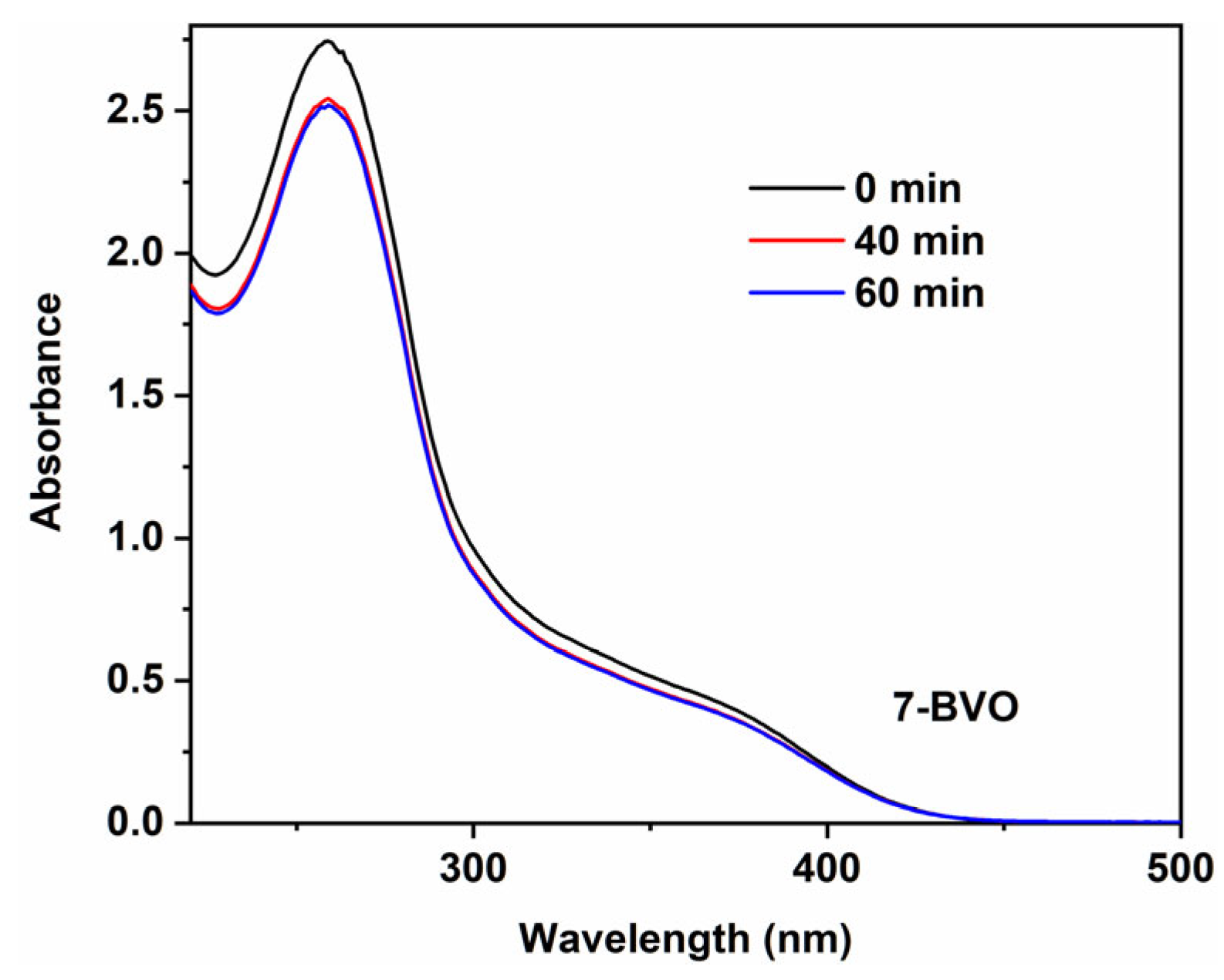
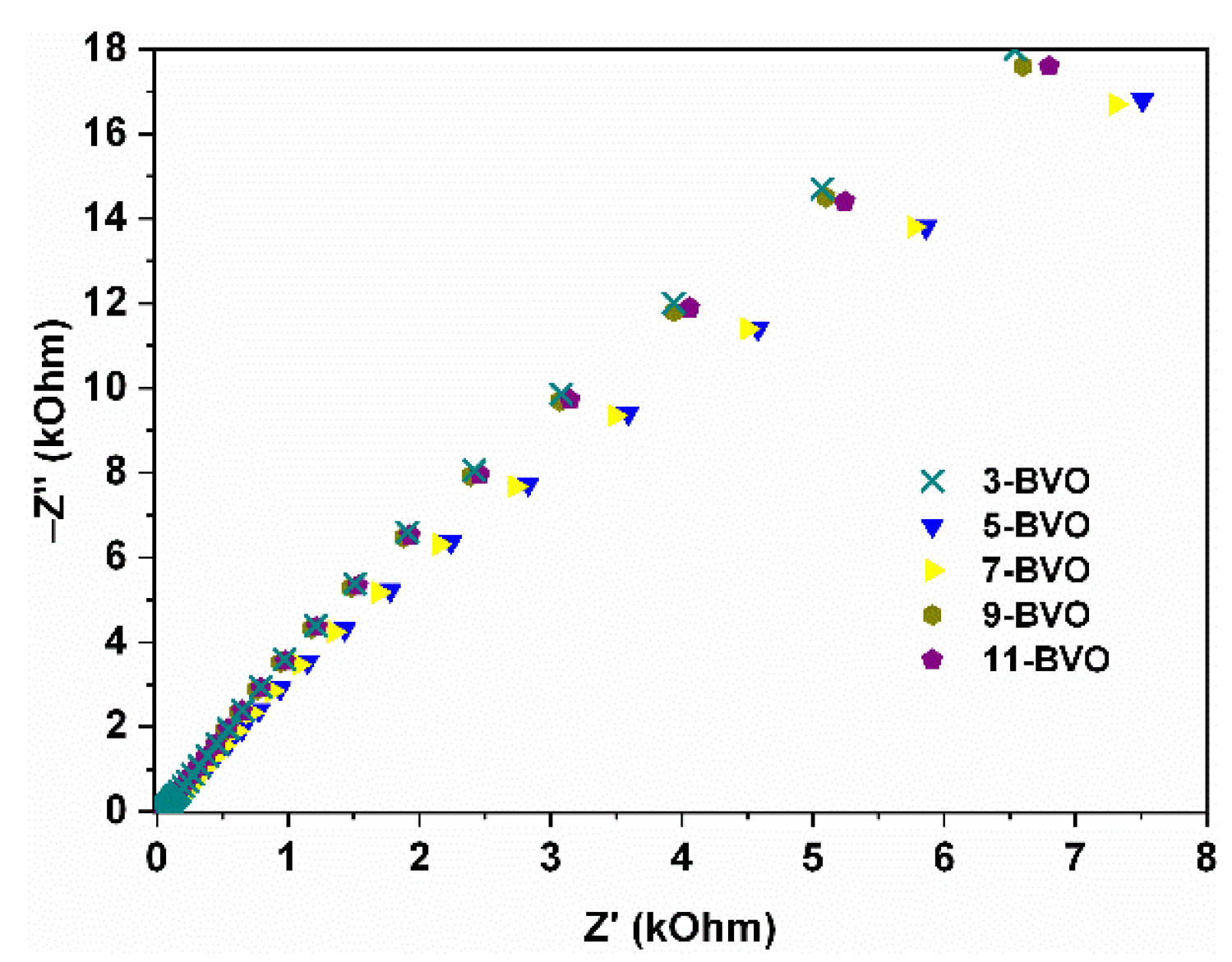
2.4. Photocatalytic Performance Analysis
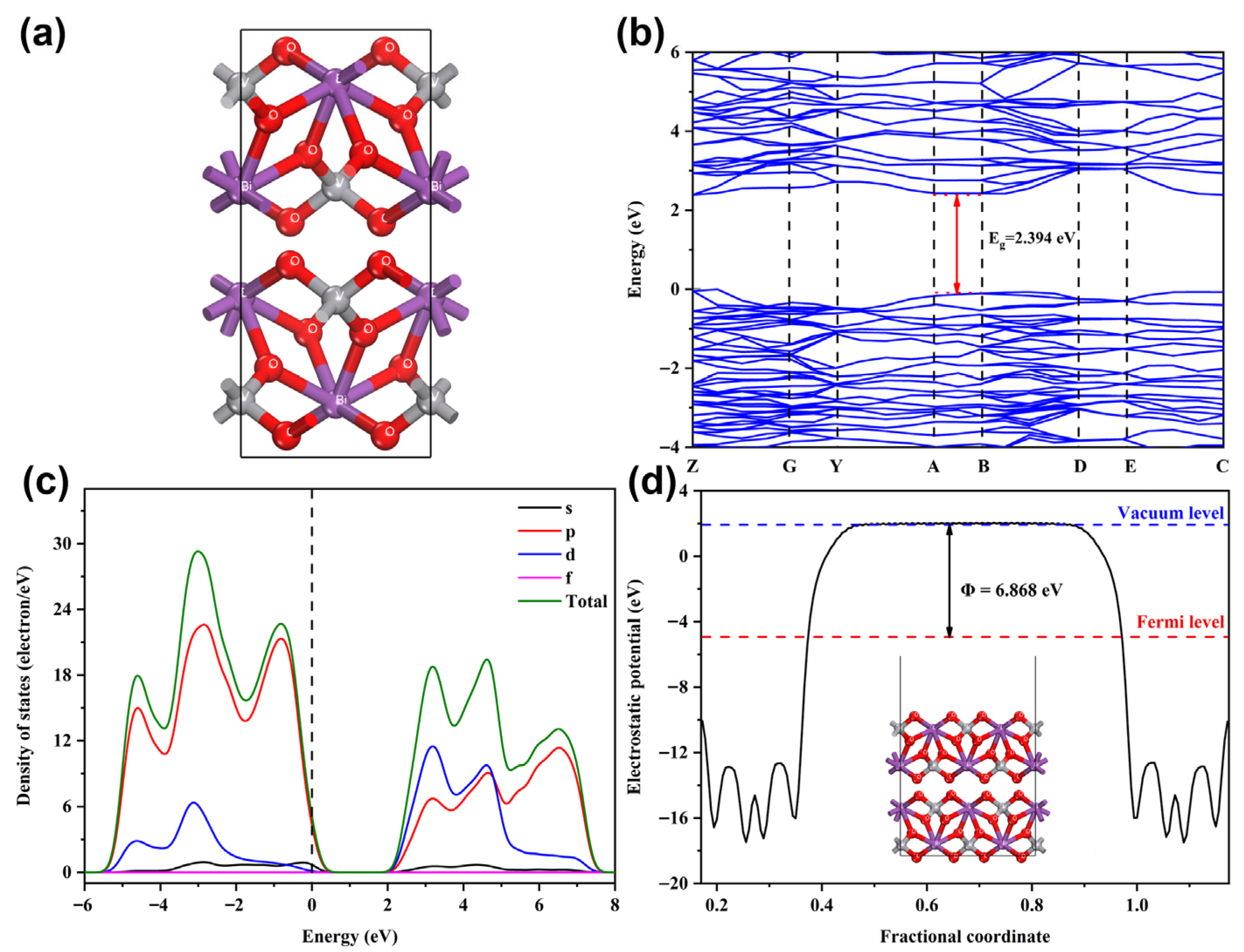
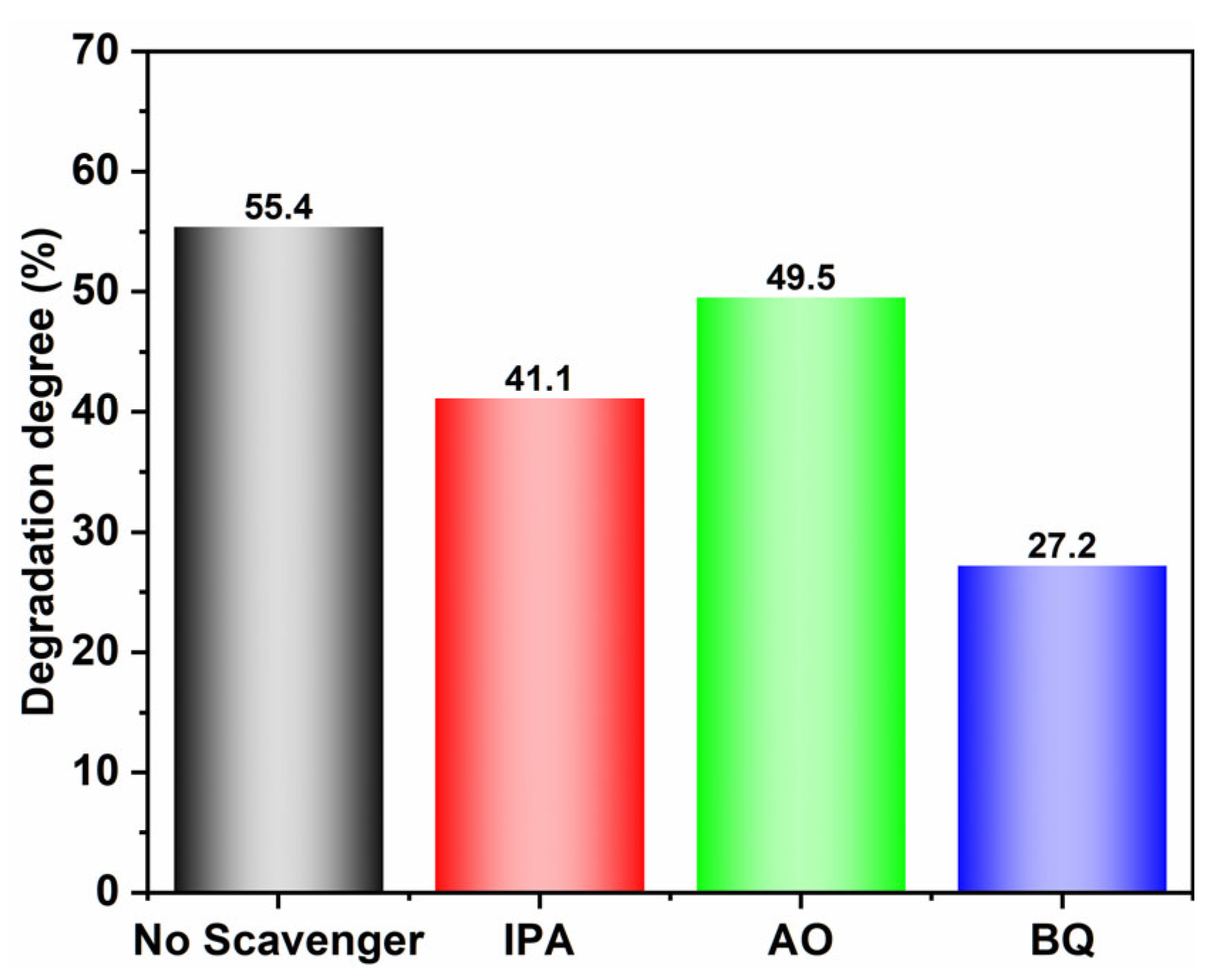
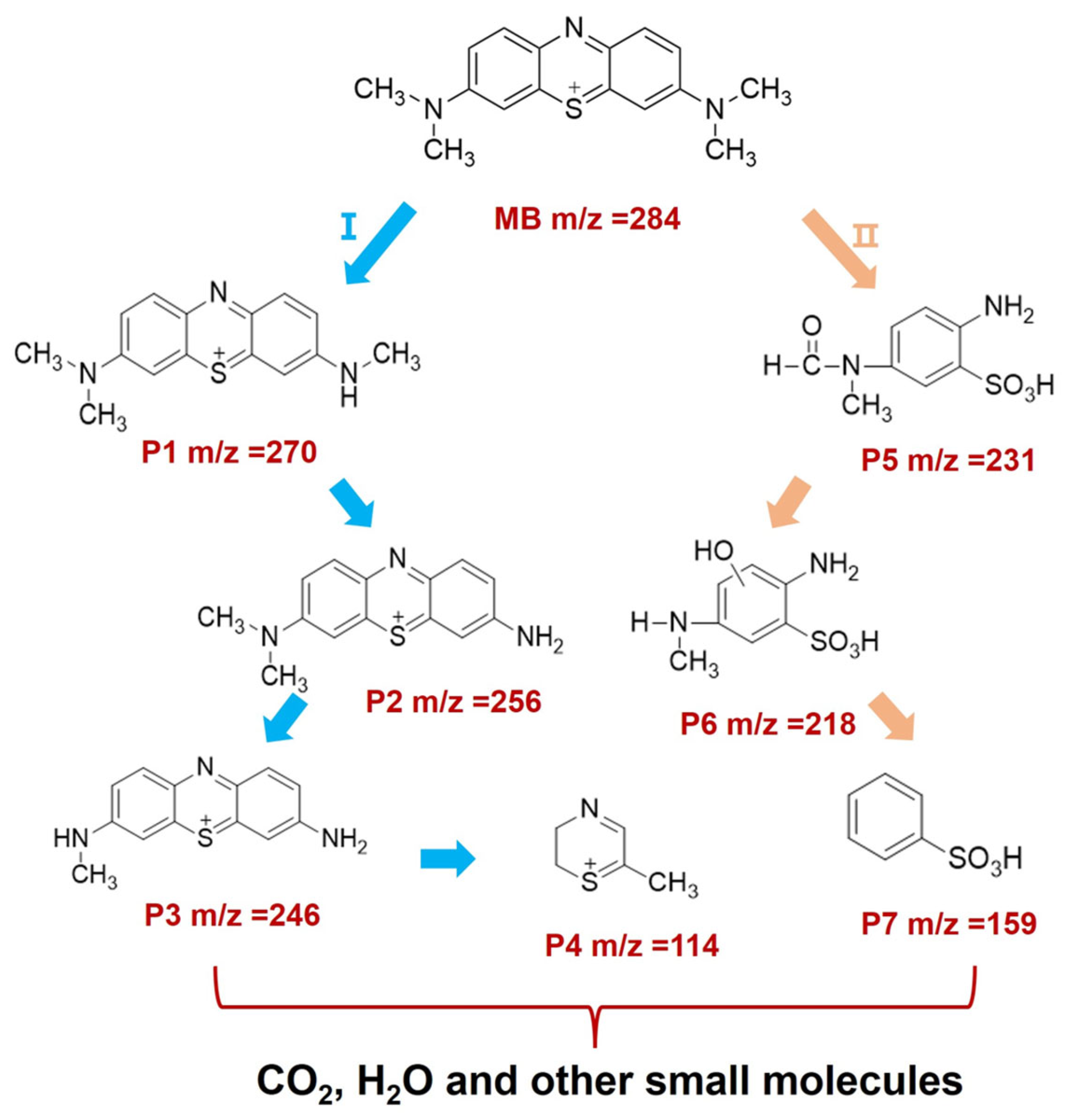
2.5. Photocatalytic Mechanism
3. Conclusions
4. Materials and Methods
4.1. Reagents
4.2. Synthesis of Photocatalytic Materials
4.3. Characterization
4.4. Photocatalytic Activity Test
4.5. Active Species Trapping Experiment
4.6. NBT Reduction Test
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dou, L.; Jin, X.; Chen, J.; Zhong, J.; Li, J.; Zeng, Y.; Duan, R. One-pot solvothermal fabrication of S-scheme OVs-Bi2O3/Bi2SiO5 microsphere heterojunctions with enhanced photocatalytic performance toward decontamination of organic pollutants. Appl. Surf. Sci. 2020, 527, 146775. [Google Scholar] [CrossRef]
- Baral, S.C.; Maneesha, P.; Datta, S.; Dukiya, K.; Sasmal, D.; Samantaray, K.S.; Br, V.K.; Dasgupta, A.; Sen, S. Enhanced photocatalytic degradation of organic pollutants in water using copper oxide (CuO) nanosheets for environmental application. JCIS Open 2024, 13, 100102. [Google Scholar] [CrossRef]
- Madkhali, N.; Prasad, C.; Malkappa, K.; Choi, H.Y.; Govinda, V.; Bahadur, I.; Abumousa, R.A. Recent update on photocatalytic degradation of pollutants in waste water using TiO2-based heterostructured materials. Results Eng. 2023, 17, 100920. [Google Scholar] [CrossRef]
- Aljawrneh, B.; Ocak, Y.S.; Albiss, B.A.; Dwiri, A.; Tawalbeh, M.; Al-Othman, A. ZrO2 nanoparticles for effective dye degradation in wastewater: Synthesis, characterization, and photocatalytic performance under sunlight. J. Alloys Compd. 2024, 1008, 176522. [Google Scholar] [CrossRef]
- Sendão, R.M.S.; Esteves Da Silva, J.C.G.; Pinto Da Silva, L. Photocatalytic removal of pharmaceutical water pollutants by TiO2–carbon dots nanocomposites: A review. Chemosphere 2022, 301, 134731. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Sakai, Y.; Guan, G.; Kansha, Y. Illumination conditions of photocatalyst-containing mist on the degradation of organic compounds. em. Eng. Chem. Eng. Process. 2025, 208, 110106. [Google Scholar] [CrossRef]
- Yifira, M.T.; Mersha, A.K.; Gebreslassie, G.; Mekonnen, K.N. Cellulose-based photocatalytic membranes for dye degradation: A review. Carbohydr. Polym. Technol. Appl. 2024, 8, 100589. [Google Scholar] [CrossRef]
- Mishra, S.; Sundaram, B. A review of the photocatalysis process used for wastewater treatment. Mater. Today Proc. 2024, 102, 393–409. [Google Scholar] [CrossRef]
- Zhu, X.; Qin, F.; Luo, Y.; Zhang, L.; Yang, D.; Feng, W.; Chen, S. Enhanced photocatalytic activity of p (BaSnO3)-n (anatase/rutile/brookite TiO2) heterojunction composites by efficient interfacial charge transfer. J. Mol. Struct. 2023, 1294, 136440. [Google Scholar] [CrossRef]
- Qin, F.; Luo, Y.; Yu, Q.; Cheng, J.; Qin, Q.; Zhu, X.; Feng, W. Enhanced charge transfer and photocatalytic activity of BiOBr/Bi2WO6 p-n heterojunctions. J. Mol. Struct. 2024, 1304, 137719. [Google Scholar] [CrossRef]
- Dou, L.; Li, J.; Long, N.; Lai, C.; Zhong, J.; Li, J.; Huang, S. Fabrication of 3D flower-like OVs-Bi2SiO5 hierarchical microstructures for visible light-driven removal of tetracycline. Surf. Interfaces 2022, 29, 101787. [Google Scholar] [CrossRef]
- Xu, N.; Li, C.; Lin, X.; Lin, X.; Zhao, X.; Nan, J.; Xiao, X. Design of atomically dispersed N-Bi(3+x)+–OV sites in ultrathin Bi2O2CO3 nanosheets for efficient and durable visible-light-driven CO2 reduction. Appl. Catal. Gen. 2024, 680, 119776. [Google Scholar] [CrossRef]
- Qin, F.; Xia, Y.; Yang, D.; Xiao, T.; Zhu, X.; Feng, W.; Qi, Z. Enhanced photocatalytic activity of g-C3N4/Bi2WO6 heterojunction via Z-scheme charge-transfer mechanism. J. Mol. Struct. 2024, 1316, 139023. [Google Scholar] [CrossRef]
- Hou, W.; Xu, H.; Cai, Y.; Zou, Z.; Li, D.; Xia, D. Precisely control interface OVs concentration for enhance 0D/2D Bi2O2CO3/BiOCl photocatalytic performance. Appl. Surf. Sci. 2020, 530, 147218. [Google Scholar] [CrossRef]
- Nguyen, T.D.; Hong, S.S. Facile solvothermal synthesis of monoclinic-tetragonal heterostructured BiVO4 for photodegradation of Rhodamine B. Catal. Commun. 2020, 136, 105920. [Google Scholar] [CrossRef]
- Dhakal, D.R.; Kshetri, Y.K.; Chaudhary, B.; Choi, J.; Murali, G.; Kim, T.H. Enhancement of upconversion luminescence in Yb3+/Er3+-doped BiVO4 through calcination. Mater. Today Commun. 2023, 37, 107258. [Google Scholar] [CrossRef]
- Zhang, S.; Ou, X.; Yang, X.; Wang, D.; Zhang, C. Preparation and properties of Al3+-doped BiVO4 semiconductor photocatalyst. Chem. Phys. Lett. 2021, 778, 138747. [Google Scholar] [CrossRef]
- Zhong, Y.; Chen, S.; Xiao, T.; Zhu, X.; Feng, W.; Qi, Z. Interfacial modification of BiVO4 photocatalyst: Construction of heterojunction with AgI. J. Phys. Chem. Solids 2025, 202, 112691. [Google Scholar] [CrossRef]
- Zhang, H.; Cao, Y.; Han, J.; Wang, S.; Zhang, Z.; Wei, Z.; Wu, Z.; Zhu, Y.; Guo, Q. An investigation of Z-scheme homojunction BiVO4 photocatalysts for efficient green removal of ammonia nitrogen. J. Alloys Compd. 2024, 1008, 176640. [Google Scholar] [CrossRef]
- Yan, S.; Liang, X.; Liu, S.; Zhang, Y.; Zeng, J.; Bai, J.; Zhu, X.; Li, J. Synthesis of PANI@α-Fe2O3/Al2O3 photo-fenton composite for the enhanced efficient methylene blue removal. J. Sol-Gel Sci. Technol. 2024, 109, 137–149. [Google Scholar] [CrossRef]
- Nguyen, H.P.; Gyawali, G.; Jo, Y.H.; Kim, T.H.; Lee, S.W. Effect of heat treatment on ultrasonic synthesized bismuth ferrites: An effective visible light-driven photocatalyst. Res. Chem. Intermed. 2017, 43, 5113–5122. [Google Scholar] [CrossRef]
- Baral, B.; Reddy, K.H.; Parida, K.M. Construction of m-BiVO4/t-BiVO4 isotype heterojunction for enhanced photocatalytic degradation of norfloxacine and oxygen evolution reaction. J. Colloid Interface Sci. 2019, 554, 278–295. [Google Scholar] [CrossRef]
- Abo El-Yazeed, W.S.; El-Hakam, S.A.; Salah, A.A.; Ibrahim, A.A. Fabrication and characterization of reduced graphene-BiVO4 nanocomposites for enhancing visible light photocatalytic and antibacterial activity. J. Photochem. Photobiol. Chem. 2021, 417, 113362. [Google Scholar] [CrossRef]
- Sayan, M.; Akhan, S.C. Antiviral drug-associated potential vaccine-escape hepatitis B virus mutants in turkish patients with chronic hepatitis B. Int. J. Infect. Dis. 2011, 15, 722–726. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lu, L.; Zhou, M.; Yin, L.; Zhou, G.; Jiang, T.; Wan, X.; Shi, H. Tuning the physicochemical property of BiOBr via pH adjustment: Towards an efficient photocatalyst for degradation of bisphenol A. J. Mol. Catal. Chem. 2016, 423, 379–385. [Google Scholar] [CrossRef]
- Ma, Q.; Ke, J.; Ma, W.; Chen, X.; Yi, Z.; Yang, H. Influence of pH adjustment and surfactant addition on the particle morphology and photocatalytic performance of hydrothermally synthesized BiVO4. Opt. Mater. 2023, 146, 114532. [Google Scholar] [CrossRef]
- Yang, Z.; Ma, X.; Liu, J.; Wang, Y.; Li, H.; Zhang, X.; Li, R. Facile fabrication of visible-light-responsive BiVO4 oxygen evolution photocatalysts with tunable band gap structure. Mater. Lett. 2024, 370, 136786. [Google Scholar] [CrossRef]
- Cao, D.; Wang, Y.; Qiao, M.; Zhao, X. Enhanced photoelectrocatalytic degradation of norfloxacin by an Ag3PO4/BiVO4 electrode with low bias. J. Catal. 2018, 360, 240–249. [Google Scholar] [CrossRef]
- Kohri, H.; Yagasaki, T. Effect of element substitution at V site on thermoelectric properties of aurivillius phase Bi2VO5.5. J. Electron. Mater. 2016, 45, 4928–4934. [Google Scholar] [CrossRef]
- Xie, W.; Zhong, L.; Wang, Z.; Liang, F.; Tang, X.; Zou, C.; Liu, G. Photocatalytic performance of Bi2VO5.5/Bi2O3 laminated composite films under simulated sunlight irradiation. Solid State Sci. 2019, 94, 1–7. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, J.; Tao, F.; Dong, Y.; Wang, L.; Hong, T. Facile construction of Z-scheme g-C3N4/BiVO4 heterojunctions for boosting visible-light photocatalytic activity. Mater. Sci. Eng. B 2022, 279, 115676. [Google Scholar] [CrossRef]
- Zhao, X.; Li, K.; Shao, X.; Chen, D.; Ji, X.; Sun, M.; Lu, H.; Liu, Y. Oxygen vacancies enhanced WO3/H-BiVO4 photoanode with conformal BiVO4-layer for promoting photoelectrochemical activity. Catal. Lett. 2025, 155, 126. [Google Scholar] [CrossRef]
- Clament Sagaya Selvam, N.; Kim, Y.G.; Kim, D.J.; Hong, W.H.; Kim, W.; Park, S.H.; Jo, W.K. Reduced graphene oxide-mediated Z-scheme BiVO4/CdS nanocomposites for boosted photocatalytic decomposition of harmful organic pollutants. Sci. Total Environ. 2018, 635, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Aguirre-Cortes, J.M.; Moral-Rodríguez, A.I.; Bailón-García, E.; Carrasco-Marín, F.; Pérez-Cadenas, A.F. BiVO4-based photocatalysts for the degradation of antibiotics in wastewater: Calcination role after solvothermal synthesis. Catalysts 2024, 14, 474. [Google Scholar] [CrossRef]
- Bao, J.; Wang, S.; Wu, J.; Fan, F.; Wang, Y.; Luo, D.; Jiang, G.; Yao, W. Enhanced photocatalytic activity of Z-scheme BiVO4 heterophase junction via giant built-in electric field. Surf. Interfaces 2024, 54, 105116. [Google Scholar] [CrossRef]
- Kukreti, K.; Roy, A.; Biswas, R.; Adhikari, S.D.; Himanshu, M.; Singh, R.P.; Dutta, A.; Sharma, M.P.; Yadav, K.L.; Yadav, K. Role of oxygen vacancy enriched m-BiVO4/t-BiVO4 isotype heterojunction for enhanced photocatalytic degradation of rhodamine B dye and in ferroelastic to paraelastic phase transition. Surf. Interfaces 2024, 52, 104932. [Google Scholar] [CrossRef]
- Chen, J.; Gao, J.; Liu, X.; Wang, P.; Yu, X.; Zhao, F.; Sun, Y.; Feng, W.; Wang, Q. Controllable phase transformation and enhanced photocatalytic performance of nano-TiO2 by using oxalic acid. Nanomaterials 2022, 12, 3019. [Google Scholar] [CrossRef]
- Zhu, X.; Qin, F.; Xia, Y.; Zhong, Y.; Zhang, X.; Feng, W.; Jiao, Y. Synthesis of Ag@AgCl modified anatase/rutile/brookite mixed phase TiO2 and their photocatalytic property. Nanotechnol. Rev. 2022, 11, 2916–2927. [Google Scholar] [CrossRef]
- Liu, Q.; Shen, J.; Yang, X.; Zhang, T.; Tang, H. 3D Reduced graphene oxide aerogel-mediated Z-scheme photocatalytic system for highly efficient solar-driven water oxidation and removal of antibiotics. Appl. Catal. B Environ. 2018, 232, 562–573. [Google Scholar] [CrossRef]
- Shan, L.; Ding, J.; Sun, W.; Han, Z.; Jin, L. Core–shell heterostructured BiVO4/BiVO4:Eu3+ with improved photocatalytic activity. J. Inorg. Organomet. Polym. Mater. 2017, 27, 1750–1759. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, C.; Zhu, Z.; Zhang, Y.; Tu, S.; Zhang, Y.; Ma, T.; Chen, F.; Huang, H. Efficient piezo-photocatalysis of 0D/2D α-Fe2O3/Bi2WO6: Synergy of weak force-driven piezoelectric polarization and Z-scheme junction. J. Colloid Interface Sci. 2023, 650, 1536–1549. [Google Scholar] [CrossRef]
- Xu, F.; Meng, K.; Cheng, B.; Wang, S.; Xu, J.; Yu, J. Unique S-scheme heterojunctions in self-assembled TiO2/CsPbBr3 hybrids for CO2 photoreduction. Nat. Commun. 2020, 11, 4613. [Google Scholar] [CrossRef]
- Du, C.; Feng, W.; Nie, S.; Zhang, J.; Liang, Y.; Han, X.; Wu, Y.; Feng, J.; Dong, S.; Liu, H.; et al. Harnessing efficient in-situ H2O2 production via a KPF6/BiOBr photocatalyst for the degradation of polyethylene. Sep. Purif. Technol. 2021, 279, 119734. [Google Scholar] [CrossRef]
- Singh Shagolsem, B.; Mohondas Singh, N. An environmentally friendly photocatalyst for the breakdown of RhB, MB, 2,4-DCP, and TCAA: Reusable Fe3O4/Ag doped g-C3N4. J. Mol. Liq. 2024, 413, 125913. [Google Scholar] [CrossRef]
- Kumaravel, S.; Lee, D.S.; Niyitanga, T.; Kaliyamoorthy, S.; Hasan, I.; Balu, K. Development of 2D/2D h-MoO3/Ag/g-C3N4 heterojunction photocatalyst with enhanced photocatalytic performance for the detoxification of methylene blue and tetracycline under solar light illumination. J. Alloys Compd. 2024, 994, 174666. [Google Scholar] [CrossRef]
- Latifian, P.; Hosseini, S.F.; Seyed Dorraji, M.S.; Rasoulifard, M.H. Fabrication of Ti-doped Bi2S3/NiO p-n heterojunction with enhanced visible-light–driven photocatalytic activity. J. Mol. Liq. 2023, 376, 121445. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.; Zhong, Y.; Yang, J.; Yang, D.; Liu, D.; Zhu, X.; Huang, L. Preparation of Bismuth Vanadates with Rich Oxygen Vacancies Using Different Sol pH and Their Photocatalytic Behavior in Degradation of Methylene Blue. Gels 2025, 11, 625. https://doi.org/10.3390/gels11080625
Chen S, Zhong Y, Yang J, Yang D, Liu D, Zhu X, Huang L. Preparation of Bismuth Vanadates with Rich Oxygen Vacancies Using Different Sol pH and Their Photocatalytic Behavior in Degradation of Methylene Blue. Gels. 2025; 11(8):625. https://doi.org/10.3390/gels11080625
Chicago/Turabian StyleChen, Shengli, Yuanyuan Zhong, Jie Yang, Daixiong Yang, Dong Liu, Xiaodong Zhu, and Lin Huang. 2025. "Preparation of Bismuth Vanadates with Rich Oxygen Vacancies Using Different Sol pH and Their Photocatalytic Behavior in Degradation of Methylene Blue" Gels 11, no. 8: 625. https://doi.org/10.3390/gels11080625
APA StyleChen, S., Zhong, Y., Yang, J., Yang, D., Liu, D., Zhu, X., & Huang, L. (2025). Preparation of Bismuth Vanadates with Rich Oxygen Vacancies Using Different Sol pH and Their Photocatalytic Behavior in Degradation of Methylene Blue. Gels, 11(8), 625. https://doi.org/10.3390/gels11080625





