Development of an Emulgel for the Effective Treatment of Atopic Dermatitis: Biocompatibility and Clinical Investigation
Abstract
1. Introduction
2. Results and Discussion
2.1. Emulgel Biocompatibility
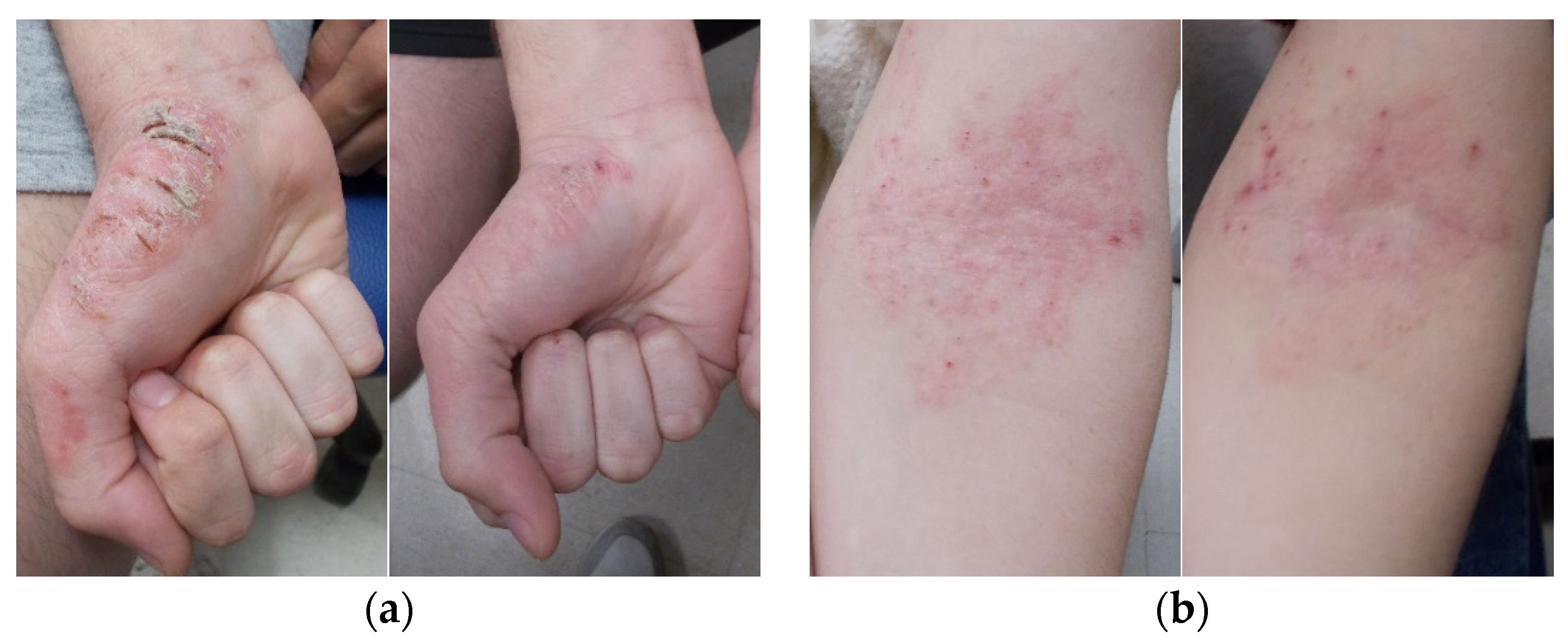
2.1.1. Skin Irritation
2.1.2. Sensitization Test
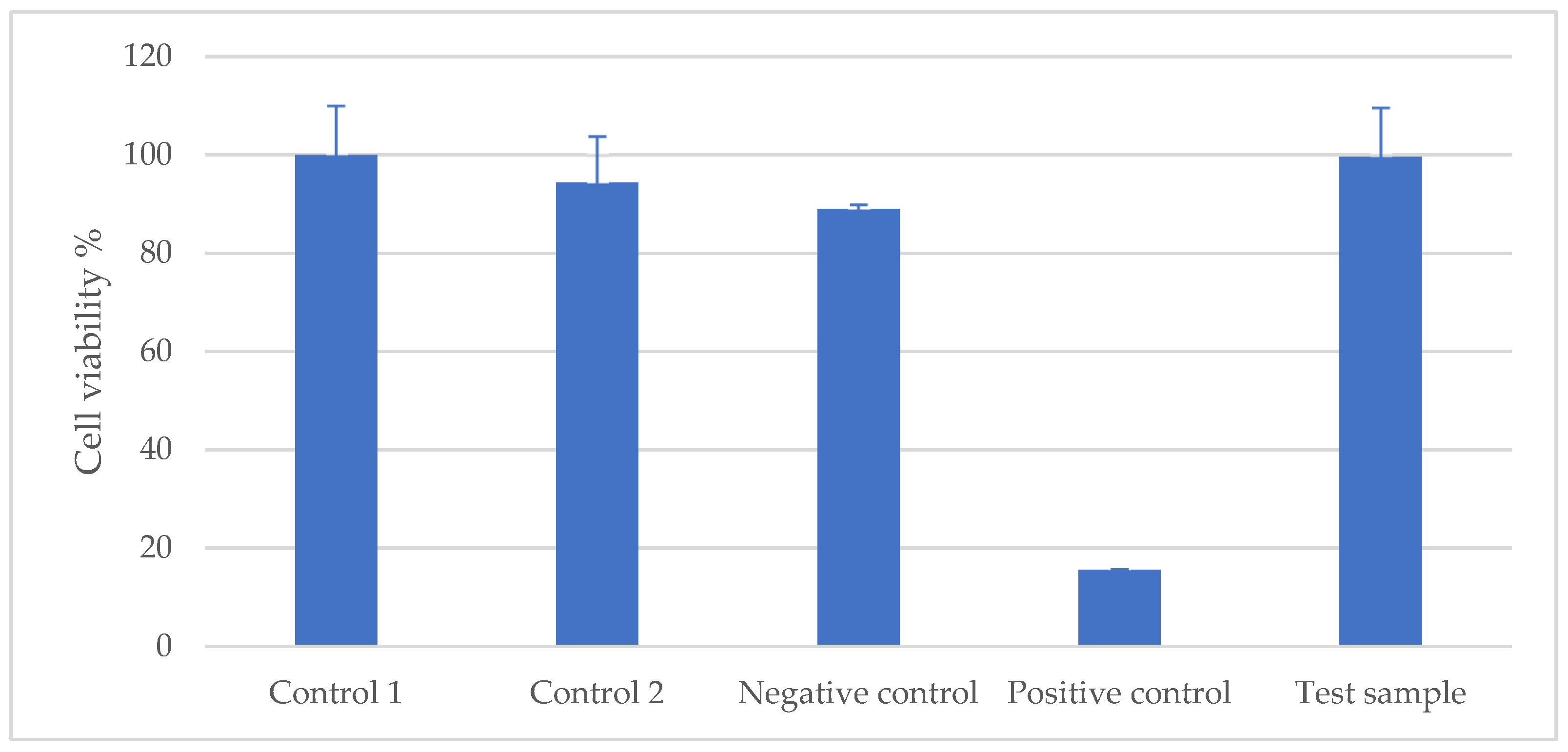
2.1.3. Cytotoxicity Test
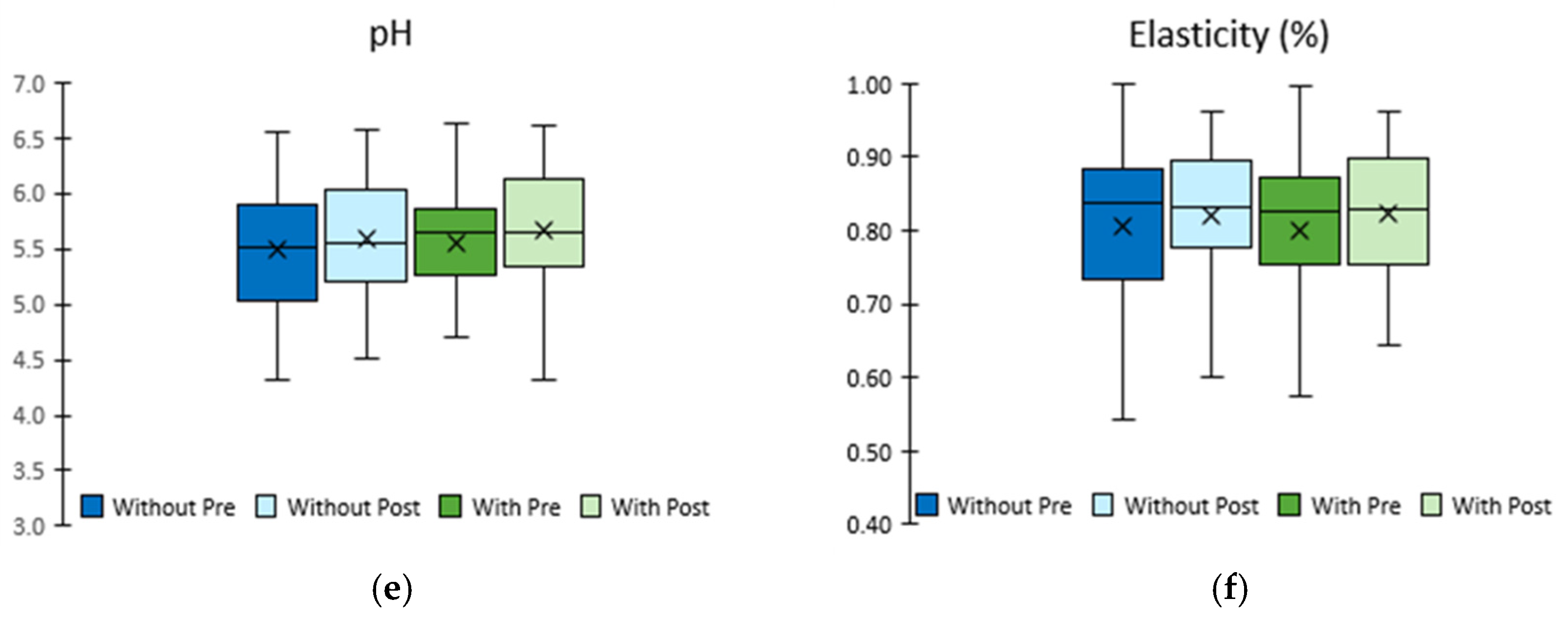
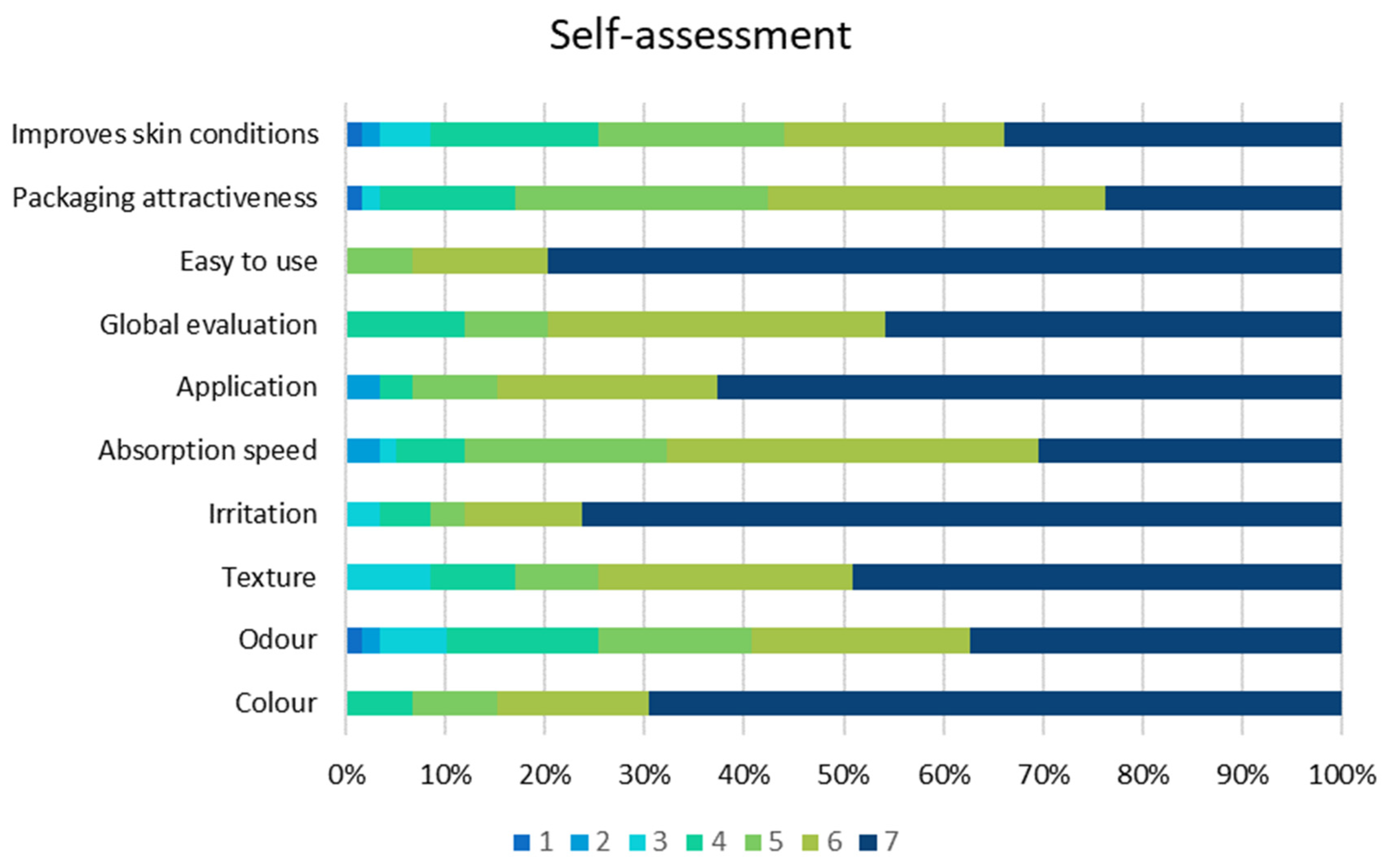
2.2. Clinical Investigation
3. Conclusions
4. Materials and Methods
4.1. Preparation of Emulgel
- Appearance: Easy to apply and absorb light green emulgel
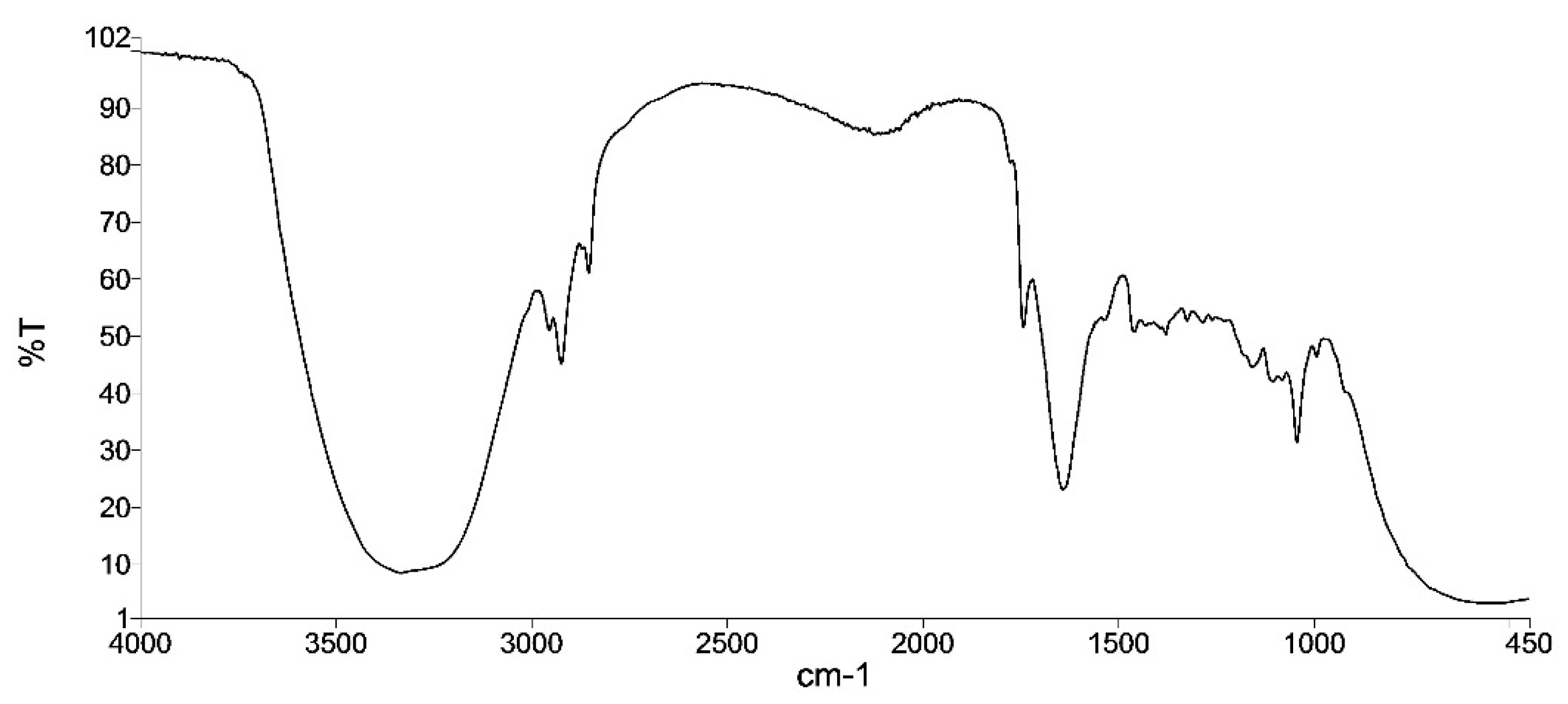
- Infrared spectrum: similar to the reference sample (the infrared spectrum correlation coefficient compares the spectrum of the sample at t = 0 with the spectrum of the corresponding sample, taking a value of 1 for the first sample and should be greater than 0.95 for the compared ones).
- pH: 5.5–6.5
- Relative density: 0.95–1.05
- Total Aerobic Microbial Count (TAMC): <1000 cfu/g or mL
- Total Yeast and Mold Count (TYMC): <100 cfu/g or mL
- Absence of pathogens: absence/g or mL
- 3.1.
- Candida albicans
- 3.2.
- Pseudomonas aeruginosa
- 3.3.
- Staphylococcus aureus
- 3.4.
- Escherichia coli
4.2. Biocompatibility Tests
4.2.1. Skin Irritation
4.2.2. Sensitization Test
4.2.3. Cytotoxicity Test
4.3. Clinical Investigation
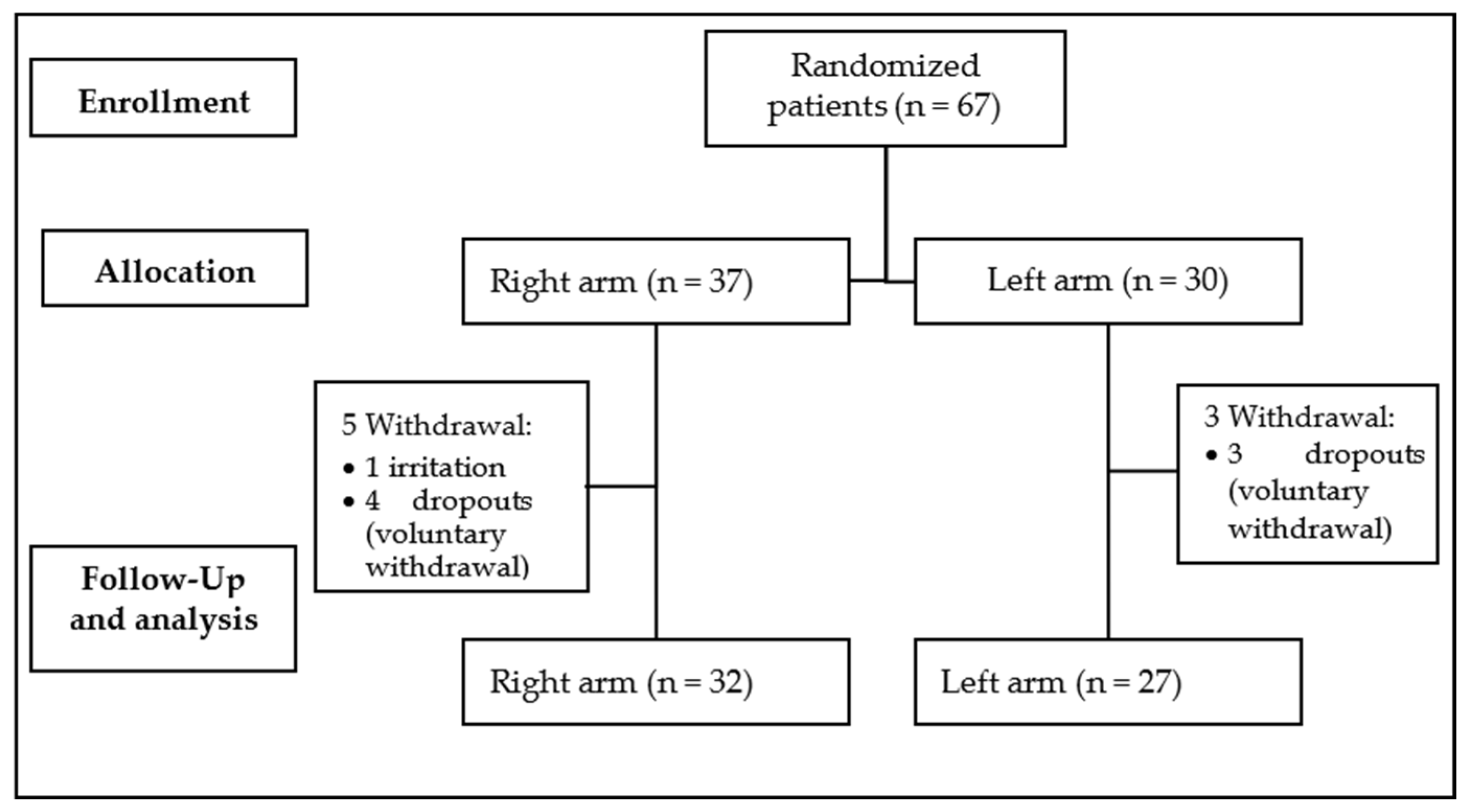
4.3.1. Study Design
4.3.2. Participants and Procedures
4.3.3. Randomization
4.3.4. Outcomes
4.3.5. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kanitakis, J. Anatomy, histology and immunohistochemistry of normal human skin. Eur. J. Dermatol. 2002, 12, 390–401. [Google Scholar] [PubMed]
- Agarwal, S.; Krishnamurthy, K. Histology, Skin. In StatPearls; StatPearls Publishing LLC.: TreasureIsland, FL, USA, 2024. [Google Scholar]
- Elias, P.M.; Hatano, Y.; Williams, M.L. Basis for the barrier abnormality in atopic dermatitis: Outside-inside-outside pathogenic mechanisms. J. Allergy Clin. Immunol. 2008, 121, 1337–1343. [Google Scholar] [CrossRef] [PubMed]
- Gatmaitan, J.G.; Lee, J.H. Challenges and Future Trends in Atopic Dermatitis. Int. J. Mol. Sci. 2023, 24, 11380. [Google Scholar] [CrossRef] [PubMed]
- Chu, A.W.L.; Wong, M.M.; Rayner, D.G.; Guyatt, G.H.; DíazMartinez, J.P.; Ceccacci, R.; Zhao, I.X.; McMullen, E.; Srivastava, A.; Wang, J.; et al. Systemic treatments for atopic dermatitis (eczema): Systematic review and network meta-analysis of randomized trials. J. Allergy Clin. Immunol. 2023, 152, 1470–1492. [Google Scholar] [CrossRef] [PubMed]
- Clebak, K.T.; Helm, L.; Uppal, P.; Davis, C.R.; Helm, M.F. Atopic Dermatitis. PrimCare 2023, 50, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Langan, S.M.; Irvine, A.D.; Weidinger, S. Atopic dermatitis. Lancet 2020, 396, 345–360. [Google Scholar] [CrossRef]
- Eichenfield, L.F.; Stripling, S.; Fung, S.; Cha, A.; O’Brien, A.; Schachner, L.A. Recent Developments and Advances in Atopic Dermatitis: A Focus on Epidemiology, Pathophysiology, and Treatment in the Pediatric Setting. Paediatr. Drugs 2022, 24, 293–305. [Google Scholar] [CrossRef] [PubMed]
- Kharwade, R.; Ali, N.; Gangane, P.; Pawar, K.; More, S.; Iqbal, M.; Bhat, A.R.; AlAsmari, A.F.; Kaleem, M. DOE-Assisted Formulation, Optimization, and Characterization of Tioconazole-Loaded Transferosomal Hydrogel for the Effective Treatment of Atopic Dermatitis: In Vitro and In Vivo Evaluation. Gels 2023, 9, 303. [Google Scholar] [CrossRef]
- Pacha, O.; Hebert, A.A. Treating atopic dermatitis: Safety, efficacy, and patient acceptability of a ceramide hyaluronic acid emollient foam. Clin. Cosmet. Investig. Dermatol. 2012, 5, 39–42. [Google Scholar] [CrossRef]
- Eichenfield, L.F.; Tom, W.L.; Chamlin, S.L.; Feldman, S.R.; Hanifin, J.M.; Simpson, E.L.; Berger, T.G.; Bergman, J.N.; Cohen, D.E.; Cooper, K.D.; et al. Guidelines of care for the management of atopic dermatitis: Section 1. Diagnosis and assessment of atopic dermatitis. J. Am. Acad. Dermatol. 2014, 70, 338–351. [Google Scholar] [CrossRef]
- Rudzki, E.; Samochocki, Z.; Rebandel, P.; Saciuk, E.; Gałecki, W.; Raczka, A.; Szmurło, A. Frequency and significance of the major and minor features of Hanifin and Rajka among patients with atopic dermatitis. Dermatology 1994, 189, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Eichenfield, L.F.; Hanifin, J.M.; Luger, T.A.; Stevens, S.R.; Pride, H.B. Consensus conference on pediatric atopic dermatitis. J. Am. Acad. Dermatol. 2003, 49, 1088–1095. [Google Scholar] [CrossRef]
- Slavkova, M.; Lazov, C.; Spassova, I.; Kovacheva, D.; Tibi, I.P.-E.; Stefanova, D.; Tzankova, V.; Petrov, P.D.; Yoncheva, K. Formulation of Budesonide-Loaded Polymeric Nanoparticles into Hydrogels for Local Therapy of Atopic Dermatitis. Gels 2024, 10, 79. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yue, Y.; Jia, R.; Liu, X.; Cheng, Z.; Cheng, Y.; Xu, Y.; Xie, Z.; Xia, H. Design and Evaluation of Paeonol-Loaded Liposomes in Thermoreversible Gels for Atopic Dermatitis. Gels 2023, 9, 198. [Google Scholar] [CrossRef] [PubMed]
- Nicol, N.H.; Rippke, F.; Weber, T.M.; Hebert, A.A. Daily Moisturization for Atopic Dermatitis: Importance, Recommendations, and Moisturizer Choices. J. Nurse Pract. 2021, 17, 920–925. [Google Scholar] [CrossRef]
- Kola-Mustapha, A.T.; Ibraheem, H.F.; Taiwo, S.; Ishola, I.O.; Usman, S.O.; Ghazali, Y.O. Formulation of Entandrophragmautileintoan Herbal Emulgel for the Management of Inflammation. Gels 2023, 9, 956. [Google Scholar] [CrossRef] [PubMed]
- Afzal, A.; Shah, S.N.H.; Javed, H.; Mumtaz, A.; Saeed, J.; Rasheed, H.M.; Arshad, R.; Ansari, S.A.; Alkahtani, H.M.; Ansari, I.A. Spilanthesacmella Extract-Based Natural Oils Loaded Emulgel for Anti-Microbial Action against Dermatitis. Gels 2023, 9, 832. [Google Scholar] [CrossRef] [PubMed]
- Khiljee, T.; Akhtar, N.; Khiljee, S.; Khiljee, B.; Rasheed, H.M.; Ansari, S.A.; Alkahtani, H.M.; Ansari, I.A. Gauging Quince Phytonutrients and Its 4% Emulgel Effecton Amplifying Facial Skin Moisturizing Potential. Gels 2023, 9, 934. [Google Scholar] [CrossRef] [PubMed]
- Masood, S.; Arshad, M.S.; Khan, H.M.S.; Begum, M.Y.; Khan, K. Encapsulation of Leptadenia pyrotechnica (Khip) Extractin Carbomer Based Emulgel for Its Enhanced Antioxidant Effects and Its In Vitro Evaluation. Gels 2023, 9, 977. [Google Scholar] [CrossRef]
- Pires, P.C.; Damiri, F.; Zare, E.N.; Hasan, A.; Neisiany, R.E.; Veiga, F.; Makvandi, P.; Paiva-Santos, A.C. A review on natural biopolymers in external drug delivery systems for wound healing and atopic dermatitis. Int. J. Biol. Macromol. 2024, 263, 130296. [Google Scholar] [CrossRef]
- Evrard, C.; Lambert de Rouvroit, C.; Poumay, Y. Epidermal Hyaluronan in Barrier Alteration-Related Disease. Cells 2021, 10, 3096. [Google Scholar] [CrossRef] [PubMed]
- Pienimaki, J.P.; Rilla, K.; Fulop, C.; Sironen, R.K.; Karvinen, S.; Pasonen, S.; Lammi, M.J.; Tammi, R.; Hascall, V.C.; Tammi, M.I. Epidermal growth factor activates hyaluronan synthase 2 in epidermal keratinocytes and increases pericellular and intracellular hyaluronan. J. Biol. Chem. 2001, 276, 20428–20435. [Google Scholar] [CrossRef]
- Zeng, F.; Harris, R.C. Epidermal growth factor, from gene organization to bedside. Semin. Cell Dev. Biol. 2014, 28, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.H.; Koh, Y.G.; Lee, W.G.; Seok, J.; Park, K.Y. The use of epidermal growth factor in dermatological practice. Int. Wound J. 2023, 20, 2414–2423. [Google Scholar] [CrossRef] [PubMed]
- Jokela, T.; Kärnä, R.; Rauhala, L.; Bart, G.; Pasonen-Seppänen, S.; Oikari, S.; Tammi, M.I.; Tammi, R.H. Human Keratinocytes Respond to Extracellular UTP by Induction of Hyaluronan Synthase 2 Expression and Increased Hyaluronan Synthesis. J. Biol. Chem. 2017, 292, 4861–4872. [Google Scholar] [CrossRef]
- Zhang, Z.; Xiao, C.; Gibson, A.M.; Bass, S.A.; Khurana Hershey, G.K. EGFR Signaling Blunts Allergen-Induced IL-6 Production and Th17 Responses in the Skin and Attenuates Development and Relapse of Atopic Dermatitis. J. Immunol. 2014, 192, 859–866. [Google Scholar] [CrossRef]
- Kim, Y.J.; Choi, M.J.; Bak, D.H.; Lee, B.C.; Ko, E.J.; Ahn, G.R.; Ahn, S.W.; Kim, M.J.; Na, J.; Kim, B.J. Topical administration of EGF suppresses immune response and protects skin barrier in DNCB-induced atopic dermatitis in NC/Nga mice. Sci. Rep. 2018, 8, 11895. [Google Scholar] [CrossRef] [PubMed]
- Ger, T.-Y.; Yang, C.-J.; Ghosh, S.; Lai, J.-Y. Biofunctionalization of nanoceria with sperminated hyaluronan enhances drug delivery performance for corneal alkali burn therapy. Chem. Eng. J. 2023, 476, 146864. [Google Scholar] [CrossRef]
- Lodén, M.; Andersson, A.C.; Andersson, C.; Frödin, T.; Oman, H.; Lindberg, M. Instrumental and dermatologist evaluation of the effect of glycerine and urea on dry skin in atopic dermatitis. Skin Res. Technol. 2001, 7, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, N.B. Selected active naturals for atopic dermatitis: Atopic Dermatitis Part 1. Clin. Dermatol. 2017, 35, 383–386. [Google Scholar] [CrossRef]
- Garavaglia, J.; Markoski, M.M.; Oliveira, A.; Marcadenti, A. Grape Seed Oil Compounds: Biological and Chemical Actions for Health. Nutr. Metab. Insights 2016, 9, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Działo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The Potential of Plant Phenolics in Prevention and Therapy of Skin Disorders. Int. J. Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.X.; Siu, W.S.; Wat, C.L.; Chan, C.L.; Koon, C.M.; Li, X.; Cheng, W.; Ma, H.; Tsang, M.S.M.; Lam, C.W.; et al. Effects of topical application of atri-herb formula on inflammatory dry-skin condition in mice with oxazolone-induced atopic dermatitis. Phytomedicine 2021, 91, 153691. [Google Scholar] [CrossRef] [PubMed]
- Finberg, M.J.; Muntingh, G.L.; van Rensburg, C.E. A comparison of the leaf gel extracts of Aloe ferox and Aloe vera in the topical treatment of atopic dermatitis in Balb/c mice. Inflammopharmacology 2015, 23, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Pal, R.R.; Parashar, P.; Singh, I.; Saraf, S.A. Tamanu oil potentiated novel sericin emulgel of levocetirizine: Repurposing for topical delivery against DNCB-induced atopic dermatitis, QbD based development and in vivo evaluation. J. Microencapsul. 2019, 36, 432–446. [Google Scholar] [CrossRef] [PubMed]
- Naeimifar, A.; Ahmad Nasrollahi, S.; Samadi, A.; Aryanian, Z.; AkbariJavar, H.; Rouini, M.; Nassiri Kashani, M.; Firooz, A. Evaluation of tolerability and efficacy of a topical emulgel containing nano liposomal ruxolitinib phosphate in the treatment of mild atopic dermatitis: A before-after single group pilot study. J. Dermatol. Treat. 2022, 33, 3160–3164. [Google Scholar] [CrossRef] [PubMed]
- Verdier-Sévrain, S.; Bonté, F. Skin hydration: A review on its molecular mechanisms. J. Cosmet. Dermatol. 2007, 6, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Danby, S.G.; Draelos, Z.D.; Gold, L.F.S.; Cha, A.; Vlahos, B.; Aikman, L.; Sanders, P.; Wu-Linhares, D.; Cork, M.J. Vehicles for atopic dermatitis therapies: More than just a placebo. J. Dermatol. Treat. 2022, 33, 685–698. [Google Scholar] [CrossRef]
- Menul Ayu Umborowati, M.D.; Sylvia Anggraeni, M.D.; Damayanti, M.D.P.; Prakoeswa, M.D.P. The beneficial effect of Aloe vera in skin barrier function improvement: A double-blind randomized trial of Madurese batik craftswomen. J. Pak. Assoc. Dermatol. 2022, 32, 142–147. [Google Scholar]
- Parente, L.M.L.; LinoJúnior, R.D.S.; Tresvenzol, L.M.F.; Vinaud, M.C.; DePaula, J.R.; Paulo, N.M. Wound healing and anti-inflammatory effect in animal models of calendula officinalis L. growing in Brazil. Evid.-Based Complement. Altern. Med. 2012, 2012, 375671. [Google Scholar] [CrossRef]
- Lee, C.M.; Jin, S.P.; Doh, E.J.; Lee, D.H.; Chung, J.H. Regional Variation of Human Skin Surface Temperature. Ann. Dermatol. 2019, 31, 349–352. [Google Scholar] [CrossRef]
- Givol, O.; Kornhaber, R.; Visentin, D.; Cleary, M.; Haik, J.; Harats, M. A systematic review of Calendula officinalis extract for wound healing. Wound Repair Regen. 2019, 27, 548–561. [Google Scholar] [CrossRef] [PubMed]
- Nicolaus, C.; Junghanns, S.; Hartmann, A.; Murillo, R.; Ganzera, M.; Merfort, I. In vitro studies to evaluate the wound healing properties of Calendula officinalis extracts. J. Ethnopharmacol. 2017, 196, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Preethi, K.C.; Kuttan, G.; Kuttan, R. Anti-inflammatory activity of flower extract of Calendula officinalis Linn. and its possible mechanism of action. Indian J. Exp. Biol. 2009, 47, 113–120. [Google Scholar] [PubMed]
- Hęś, M.; Dziedzic, K.; Górecka, D.; Jędrusek-Golińska, A.; Gujska, E. Aloe vera (L.) Webb.: Natural Sources of Antioxidants—A Review. Plant Foods Hum. Nutr. 2019, 74, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, M.; González-Burgos, E.; Iglesias, I.; Gómez-Serranillos, M.P. Pharmacological Update Properties of Aloe Vera and its Major Active Constituents. Molecules 2020, 25, 1324. [Google Scholar] [CrossRef] [PubMed]
- Algiert-Zielińska, B.; Batory, M.; Skubalski, J.; Rotsztejn, H. Evaluation of the relation between lipid coat, transepidermal water loss, and skin pH. Int. J. Dermatol. 2017, 56, 1192–1197. [Google Scholar] [CrossRef]
- Montero-Vilchez, T.; Segura-Fernández-Nogueras, M.V.; Pérez-Rodríguez, I.; Soler-Gongora, M.; Martinez-Lopez, A.; Fernández-González, A.; Molina-Leyva, A.; Arias-Santiago, S. Skin Barrier Functionin Psoriasisand Atopic Dermatitis: Transepidermal Water Lossand Temperatureas Useful Tools to Assess Disease Severity. J. Clin. Med. 2021, 10, 359. [Google Scholar] [CrossRef] [PubMed]
- Blaak, J.; Staib, P. The Relation of pH and Skin Cleansing. Curr. Probl. Dermatol. 2018, 54, 132–142. [Google Scholar] [CrossRef]
- ISO 17516:2014; Cosmetics—Microbiology—Microbiological Limits. International Organization for Standardization: Geneva, Switzerland, 2014.
- Harmonisation of ICF. Stability Testing of New Drug Substances and Drug Products—Scientific Guideline (ICH Q1A) (R2); Harmonisation of ICF: Geneva, Switzerland, 2003.
- ISO/TR 18811:2018; Cosmetics—Guidelines on the Stability Testing of Cosmetic Products. International Organization for Standardization: Geneva, Switzerland, 2018.
- ISO 10993-1:2009; Biological Evaluation of Medical Devices—Part 1: Evaluation and Testing within a Risk Management Process. International Organization for Standardization: Geneva, Switzerland, 2009.
- ISO 10993-2:2006; Biological Evaluation of Medical Devices—Part 2: Animal Welfare Requirements. International Organization for Standardization: Geneva, Switzerland, 2006.
- ISO 10993-10:2010; Biological Evaluation of Medical Devices—Part 10: Tests for Irritation and Skin Sensitization. International Organization for Standardization: Geneva, Switzerland, 2010.
- ISO 10993-5:2009; Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity. International Organization for Standardization: Geneva, Switzerland, 2009.
- ISO 114155:2020; Clinical Investigation of Medical Devices for Human Subjects—Good Clinical Practice. International Organization for Standardization: Geneva, Switzerland, 2009.
- Akdeniz, M.; Gabriel, S.; Lichterfeld-Kottner, A.; Blume-Peytavi, U.; Kottner, J. Transepidermal water loss in healthy adults: A systematic review and meta-analysis update. Br. J. Dermatol. 2018, 179, 1049–1055. [Google Scholar] [CrossRef]
- Gonzalez-Bravo, A.; Montero-Vilchez, T.; Arias-Santiago, S.; Buendia-Eisman, A. The Effect of Sunscreens on the Skin Barrier. Life 2022, 12, 2083. [Google Scholar] [CrossRef] [PubMed]
- Khosrowpour, Z.; Ahmad Nasrollahi, S.; Ayatollahi, A.; Samadi, A.; Firooz, A. Effects of four soaps on skin trans-epidermal water loss and erythema index. J. Cosmet. Dermatol. 2019, 18, 857–861. [Google Scholar] [CrossRef] [PubMed]
- Montero-Vilchez, T.; Soler-Góngora, M.; Martínez-López, A.; Ana, F.G.; Buendía-Eisman, A.; Molina-Leyva, A.; Arias-Santiago, S. Epidermal barrier changes in patients with psoriasis: The role of phototherapy. Photodermatol. Photoimmunol. Photomed. 2021, 37, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Schmid-Wendtner, M.H.; Korting, H.C. The pH of the skin surface and its impact on the barrier function. Skin Pharmacol. Physiol. 2006, 19, 296–302. [Google Scholar] [CrossRef]
- Pullar, J.M.; Carr, A.C.; Vissers, M.C.M. The Roles of Vitamin C in Skin Health. Nutrients 2017, 9, 866. [Google Scholar] [CrossRef]
- Kunz, B.; Oranje, A.P.; Labrèze, L.; Stalder, J.F.; Ring, J.; Taïeb, A. Clinical validation and guidelines for the SCORAD index: Consensus report of the European Task Force on Atopic Dermatitis. Dermatology 1997, 195, 10–19. [Google Scholar] [CrossRef]
- Oranje, A.P.; Glazenburg, E.J.; Wolkerstorfer, A.; De Waard-vander Spek, F.B. Practical issues on interpretation of scoring atopic dermatitis: The SCORAD index, objective SCORAD and the three-items everity score. Br. J. Dermatol. 2007, 157, 645–648. [Google Scholar] [CrossRef]
- Hanifin, J.M.; Baghoomian, W.; Grinich, E.; Leshem, Y.A.; Jacobson, M.; Simpson, E.L. The Eczema Area and Severity Index—A Practical Guide. Dermatitis 2022, 33, 187–192. [Google Scholar] [CrossRef]

| Samples | Primary Irritation Score | Primary Irritation Index | ||
|---|---|---|---|---|
| Rabbit ID1 | Rabbit ID2 | Rabbit ID3 | ||
| Study product | 0 | 0 | 0 | 0 |
| Positive control | 4.00 | 5.33 | 4.33 | 4.55 |
| Negative control | 0 | 0 | 0 | 0 |
| Groups | Groups Mean Topical Induction Phase—ChallengePhase | Score |
|---|---|---|
| Male test | 0.8 | 0.7 |
| Female test | 0.6 | |
| Negative control | 0.4 | 0.4 |
| Characteristics | Mean/No | SD/% |
|---|---|---|
| Women | 33 | 55.9 |
| Age (years) | 31.8 | 16.20 |
Phototype:
| 5 38 14 2 | 8.5 64.4 23.7 3.4 |
Cream application:
| 8 46 1 4 | 13.6 78.0 1.7 6.8 |
Experimental arm:
| 27 32 | 45.8 54.2 |
Treatment:
| 26 7 20 6 | 44.1 11.9 33.9 10.2 |
Comorbidities:
| 4 27 25 16 6 7 22 | 4 25 23 15 6 7 21 |
Solar exposure:
| 13 41 5 | 22 69 8 |
| Homeostasis Parameter | Healthy Zone | ||||||||
| Forearm without Emulgel | Forearm with Emulgel | p * | |||||||
| before | after | Dif. | p 1 | before | after | Dif. | p 2 | ||
| Temperature (°C) | 32.00 ± 0.13 | 31.71 ± 0.14 | −0.29 ± 0.14 | 0.050 | 31.98 ± 0.13 | 31.64 ± 0.15 | −0.34 ± 0.14 | 0.017 | 0.509 |
| Erythema (AU) | 210.81 ± 8.80 | 216.8 ± 9.58 | 6.08 ± 5.20 | 0.247 | 210.49 ± 10.17 | 215.35 ± 10.17 | 4.86 ± 6.80 | 0.478 | 0.868 |
| TEWL (g·m−2·h−1) | 11.64 ± 0.78 | 11.05 ± 0.72 | −0.59 ± 0.65 | 0.372 | 11.48 ± 0.72 | 10.81 ± 0.69 | 0.67 ± 0.65 | 0.308 | 0.894 |
| SCH (AU) | 35.79 ± 1.54 | 33.32 ± 1.62 | −2.47 ± 1.30 | 0.062 | 35.65 ± 1.59 | 44.02 ± 1.61 | 8.37 ± 1.55 | <0.000 | <0.000 |
| pH # | 5.40 ± 0.07 | 5.47 ± 0.07 | 0.07 ± 0.11 | 0.522 | 5.42 ± 0.07 | 5.51 ± 0.07 | 0.09 ± 0.09 | 0.341 | 0.748 |
| Elasticity (%) | 0.828 ± 0.011 | 0.843 ± 0.010 | 0.015 ± 0.006 | 0.024 | 0.817 ± 0.011 | 0.835 ± 0.011 | 0.019 ± 0.007 | 0.017 | 0.702 |
| Homeostasis Parameter | Eczema Zone | ||||||||
| Forearm without Emulgel | Forearm with Emulgel | p * | |||||||
| before | after | Dif. | p 1 | before | after | Dif. | p 2 | ||
| Temperature (°C) | 32.51 ± 0.12 | 32.32 ± 0.16 | −0.18 ± 0.15 | 0.229 | 31.99 ± 0.51 | 32.06 ± 0.18 | 0.07 ± 0.54 | 0.904 | 0.631 |
| Erythema (AU) | 318.00 ± 10.73 | 337.60 ± 11.37 | 19 60 ± 8.16 | 0.020 | 337.74 ± 11.78 | 320.55 ± 11.79 | −17.18 ± 12.18 | 0.164 | 0.008 |
| TEWL (g·m−2·h−1) | 17.90 ± 1.27 | 20.51 ± 1.69 | 2.61 ± 1.74 | 0.140 | 19.41 ± 1.46 | 17.63 ± 1.20 | −1.77 ± 1.50 | 0.243 | 0.006 |
| SCH (AU) | 28.67 ± 2.32 | 24.74 ± 2.02 | −3.93 ± 2.00 | 0.054 | 29.64 ± 2.29 | 37.15 ± 2.23 | 7.51 ± 2.16 | 0.001 | <0.000 |
| pH # | 5.48 ± 0.07 | 5.58 ± 0.06 | 0.10 ± 0.09 | 0.245 | 5.55 ± 0.06 | 5.66 ± 0.07 | 0.11 ± 0.09 | 0.229 | 0.862 |
| Elasticity (%) | 0.807 ± 0.013 | 0.819 ± 0.013 | 0.012 ± 0.013 | 0.373 | 0.799 ± 0.013 | 0.824 ± 0.011 | 0.025 ± 0.010 | 0.026 | 0.411 |
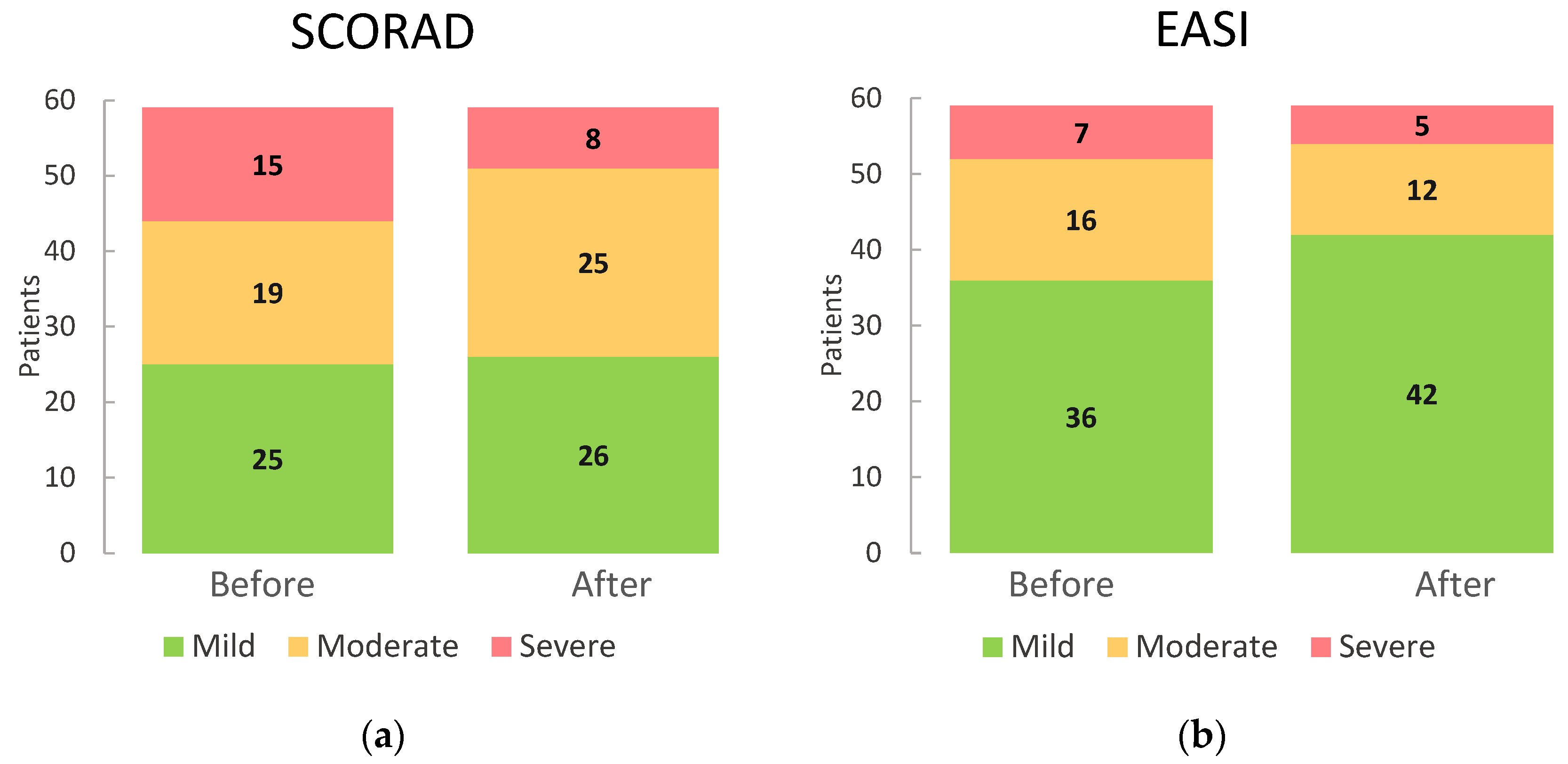
| Before Emulgel | After Emulgel | |
|---|---|---|
| SCORAD: | ||
| 25 (42.4%) | 26 (44.1%) |
| 19 (32.2%) | 25 (42.4%) |
| 15 (25.4%) | 8 (13.6%) |
| Total | 59 (100.0%) | 59 (100.0%) |
| EASI: | ||
| 36 (61.0%) | 42 (71.2%) |
| 16 (27.1%) | 12 (20.3%) |
| 7 (11.9%) | 5 (8.5%) |
| Total | 59 (100.0%) | 59 (100.0%) |
| Function | Components | Concentration (w/v) | Emulsion | Gel | |
|---|---|---|---|---|---|
| Oily Phase | Aqueous Phase | ||||
| Emollient, humectant, moisturizer | Glycerol Sodium hyaluronate Panthenol Aloe barbadensis leaf juice Calendula officinalis flower extract Propylene glycol Zinc gluconate | 5.0% 1.0% >0.1–≤1.0% 1.0% 1.8% >1.0–≤5.0% ≤0.1% | x | ||
| Grape seed oil Allantoin Caprylic/capric triglyceride C13–14isoparaffin Polyacrylamide | 3.0% >0.1–≤1.0% >1.0–≤5.0% >0.1–≤1.0% >1.0–≤5.0% | x | |||
| Antioxidant | Tocopheryl acetate Tocopherol Pentaerythrityl tetra-di-T-butyl hydroxyhydrocinnamate | ≤0.1% ≤0.1% ≤0.1% | x | ||
| Solvent | Aqua | >50.0–≤75.0% | x | ||
| Chelating | Disodium EDTA | ≤0.1% | x | ||
| Colorant | CI42090 | ≤0.1% | x | ||
| Growth Factor | Epidermal Growth Factor * | 0.00001% | x | ||
| Surfactant | PEG-18 castor oil dioleate PEG/PPG-4/12 dimethicone Laureth-7 | >0.1–≤1.0% >0.1–≤1.0% >0.1–≤1.0% | x | ||
| Preservative | Potassium sorbate Sodium benzoate | ≤0.1% ≤0.1% | x | ||
| BHT | ≤0.1% | x | |||
| Buffer | Citric acid | ≤0.1% | x | ||
| Triisopropanolamine | ≤0.1% | x | |||
| Gelling | Carbomer | >0.1–≤1.0% | x | ||
| Parameter | T = 0 | T = 1 Month | T = 3 Months | T = 6 Months |
|---|---|---|---|---|
| Appearance | ✓ | ✓ | ✓ | ✓ |
| pH | 5.7 | 5.4 | 5.3 | 5.8 |
| Infrared spectrum Correlation coefficient | 1 | 0.992 | 0.990 | 0.954 |
| TAMC | <100 cfu/mL | - | <100 cfu/mL | - |
| TYMC | <100 cfu/mL | - | <100 cfu/mL | - |
| Parameter | T = 0 | T = 3 Months | T = 6 Months | T = 9 Months | T = 12 Months | T = 18 Months | T = 24 Months | T = 36 Months | T = 48 Months | T = 60 Months |
|---|---|---|---|---|---|---|---|---|---|---|
| Appearance | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |  * * |
| pH | 5.7 | 5.3 | 5.2 | 5.3 | 5.3 | 5.3 | 5.3 | 5.3 | 5.3 | 5.2 |
| Infrared spectrum | 1 | 0.981 | 0.970 | 0.958 | 0.995 | 0.995 | 0.993 | 0.988 | 0.982 | 0.743 |
| TAMC | <100 cfu/mL | - | - | - | - | - | <100 cfu/mL | - | - | * |
| TYMC | <100 cfu/mL | - | - | - | - | - | <100 cfu/mL | - | - | * |
 : does not comply with appearance specifications * TAMC and TYMC not carried out due to the change in appearance, yellowish cream, and infrared spectrum coefficient correlation out of specifications.
: does not comply with appearance specifications * TAMC and TYMC not carried out due to the change in appearance, yellowish cream, and infrared spectrum coefficient correlation out of specifications.Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Farto, A.; Jiménez-Escobar, A.L.; Pérez-González, N.; Castán, H.; Clares, B.; Arias-Santiago, S.; Montero-Vílchez, T. Development of an Emulgel for the Effective Treatment of Atopic Dermatitis: Biocompatibility and Clinical Investigation. Gels 2024, 10, 370. https://doi.org/10.3390/gels10060370
Gómez-Farto A, Jiménez-Escobar AL, Pérez-González N, Castán H, Clares B, Arias-Santiago S, Montero-Vílchez T. Development of an Emulgel for the Effective Treatment of Atopic Dermatitis: Biocompatibility and Clinical Investigation. Gels. 2024; 10(6):370. https://doi.org/10.3390/gels10060370
Chicago/Turabian StyleGómez-Farto, Almudena, Ana Leticia Jiménez-Escobar, Noelia Pérez-González, Herminia Castán, Beatriz Clares, Salvador Arias-Santiago, and Trinidad Montero-Vílchez. 2024. "Development of an Emulgel for the Effective Treatment of Atopic Dermatitis: Biocompatibility and Clinical Investigation" Gels 10, no. 6: 370. https://doi.org/10.3390/gels10060370
APA StyleGómez-Farto, A., Jiménez-Escobar, A. L., Pérez-González, N., Castán, H., Clares, B., Arias-Santiago, S., & Montero-Vílchez, T. (2024). Development of an Emulgel for the Effective Treatment of Atopic Dermatitis: Biocompatibility and Clinical Investigation. Gels, 10(6), 370. https://doi.org/10.3390/gels10060370









