Natural Polysaccharide-Based Hydrogels Used for Dye Removal
Abstract
1. Introduction
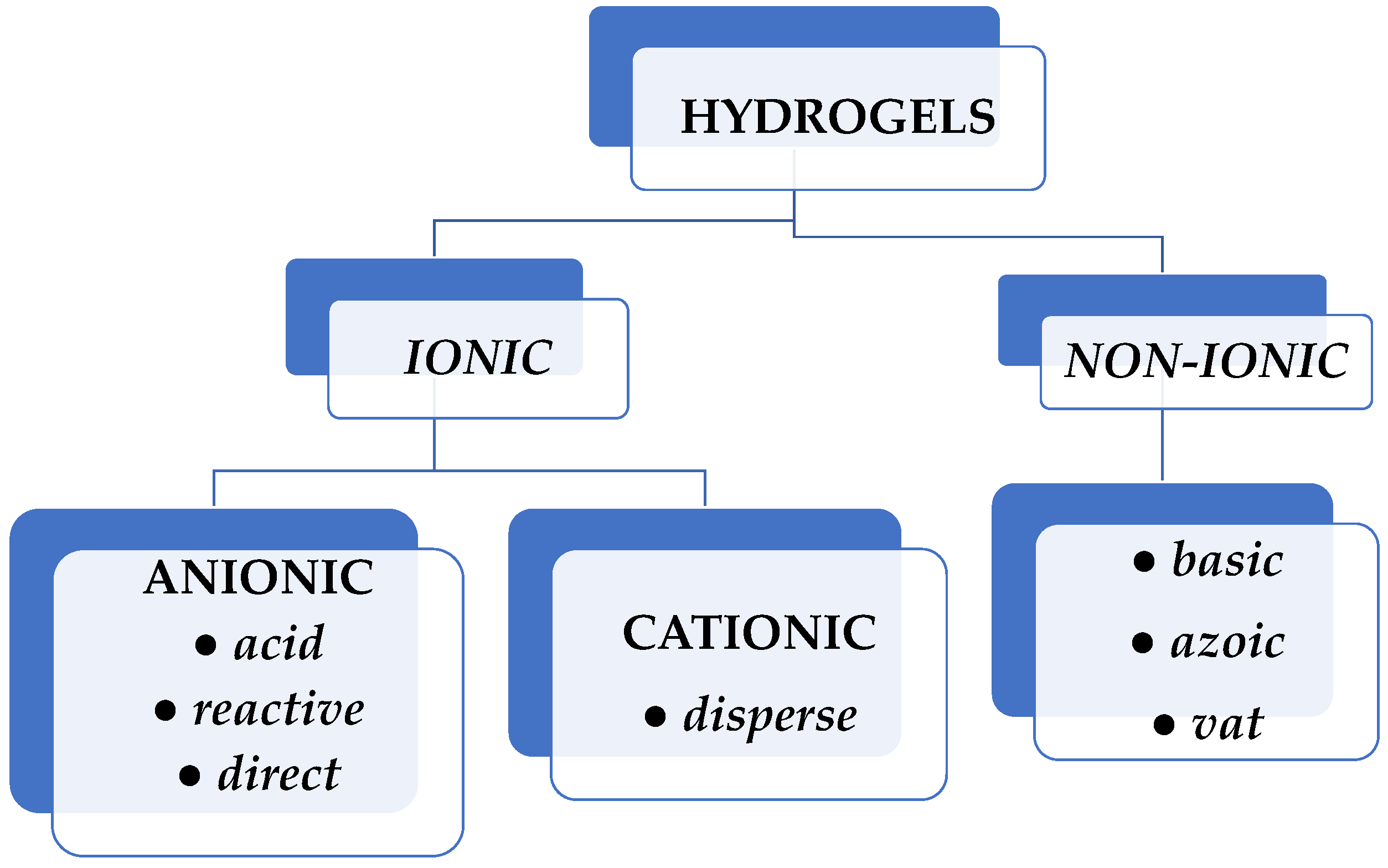
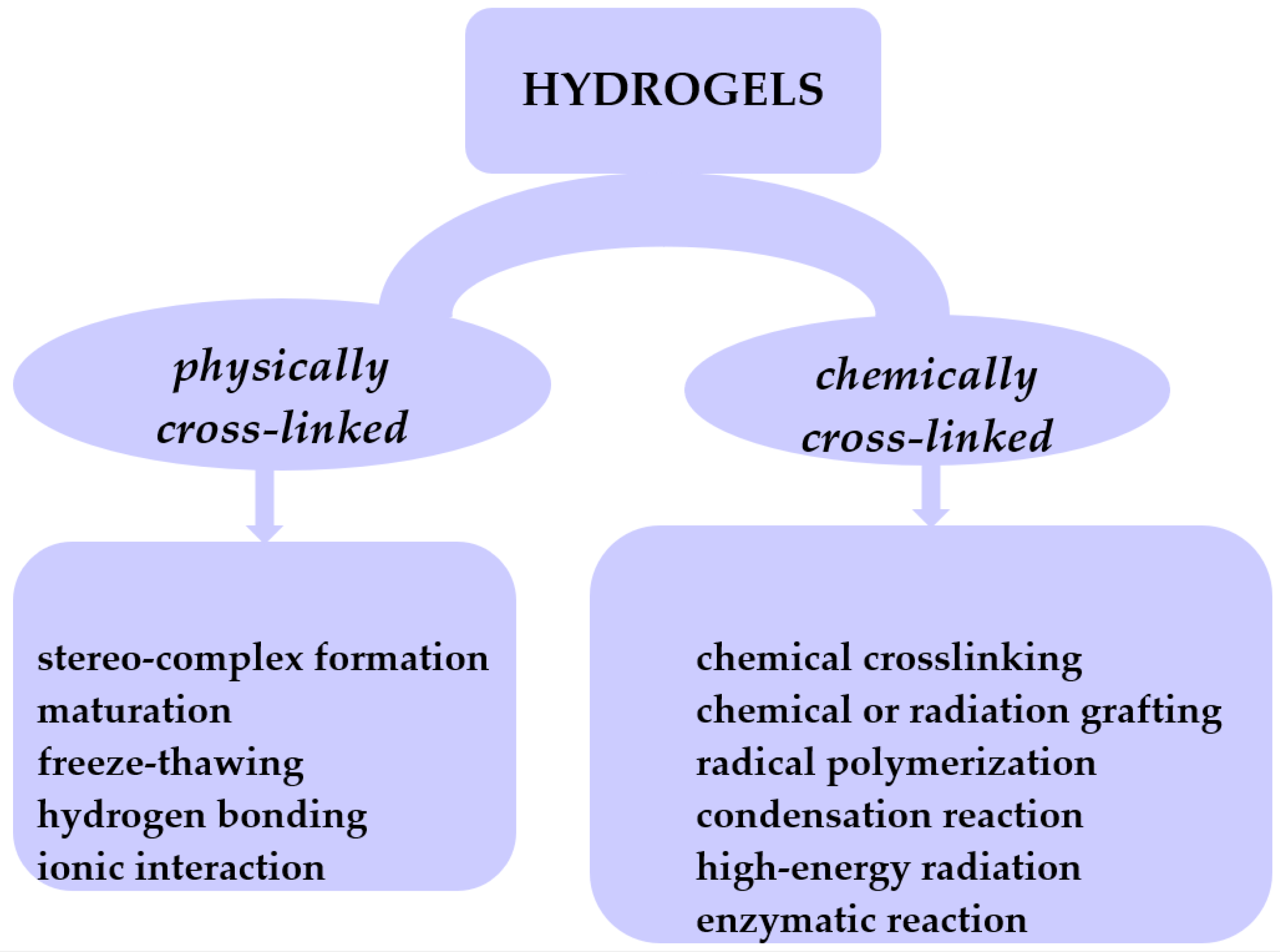
2. Hydrogels
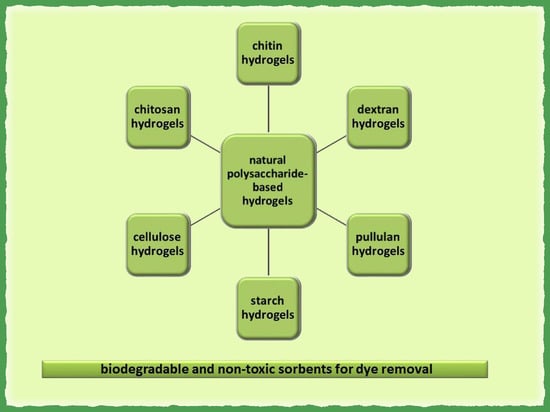
2.1. Hydrogels Based on Natural Polysaccharides Used in Dye Adsorption
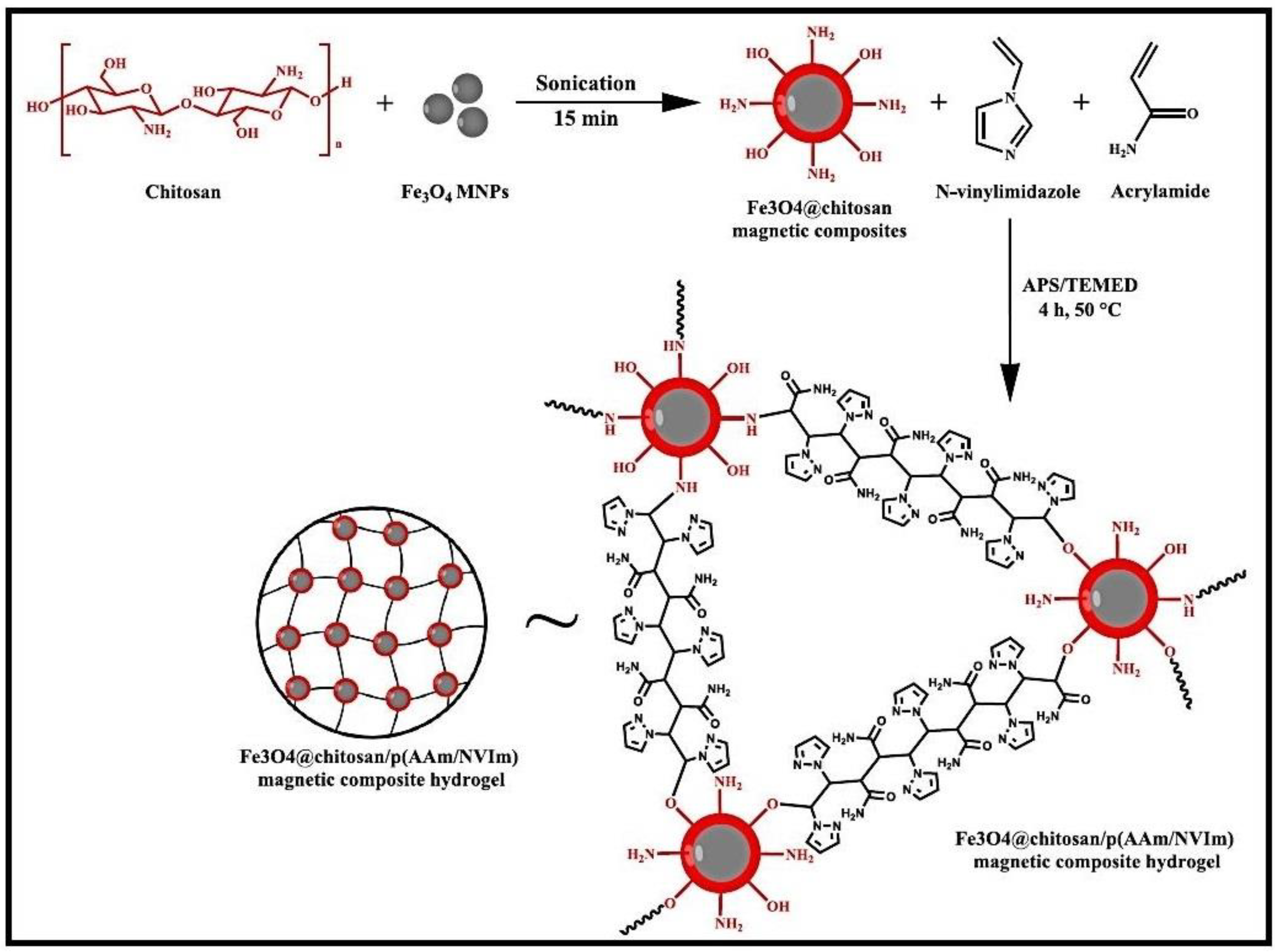
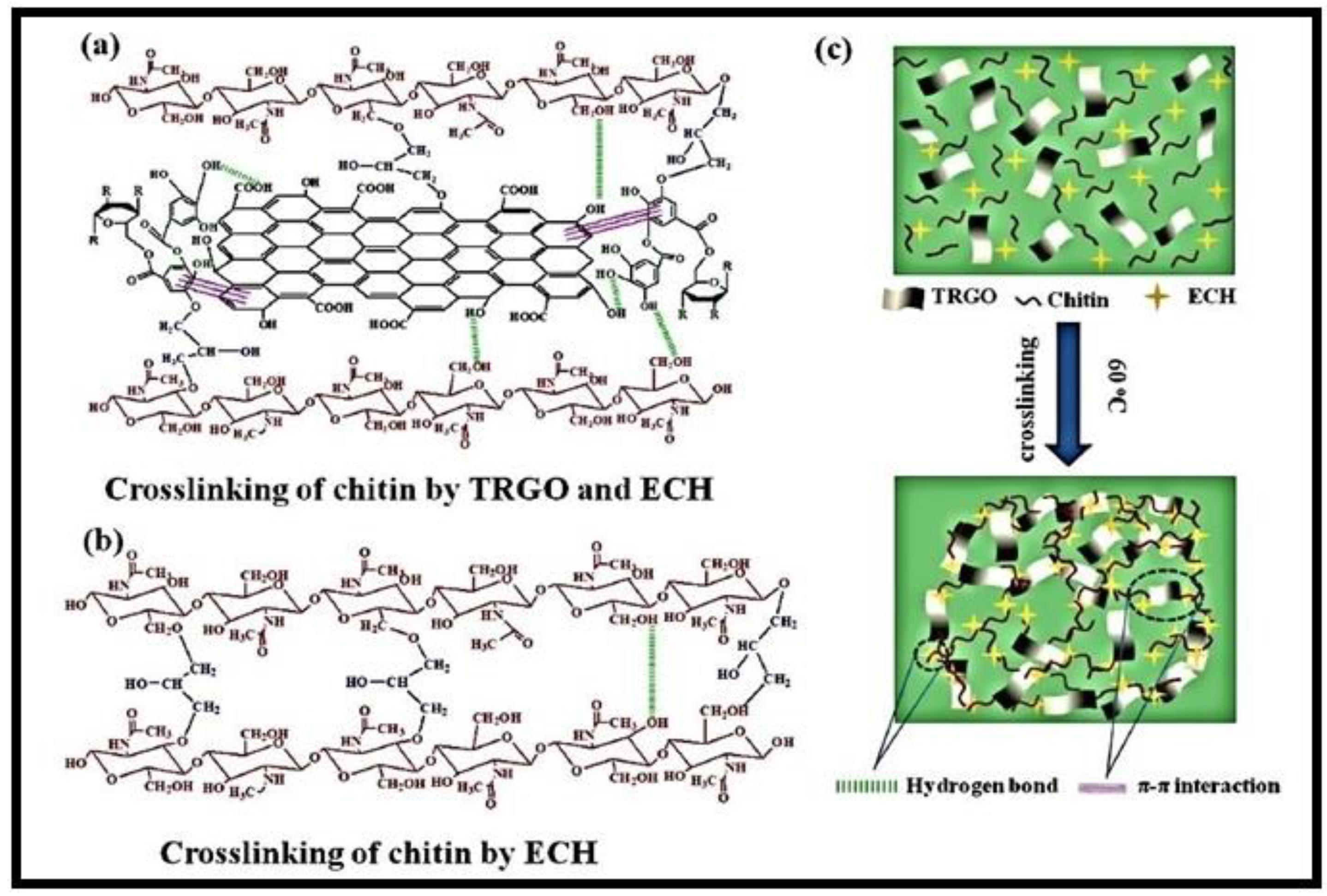
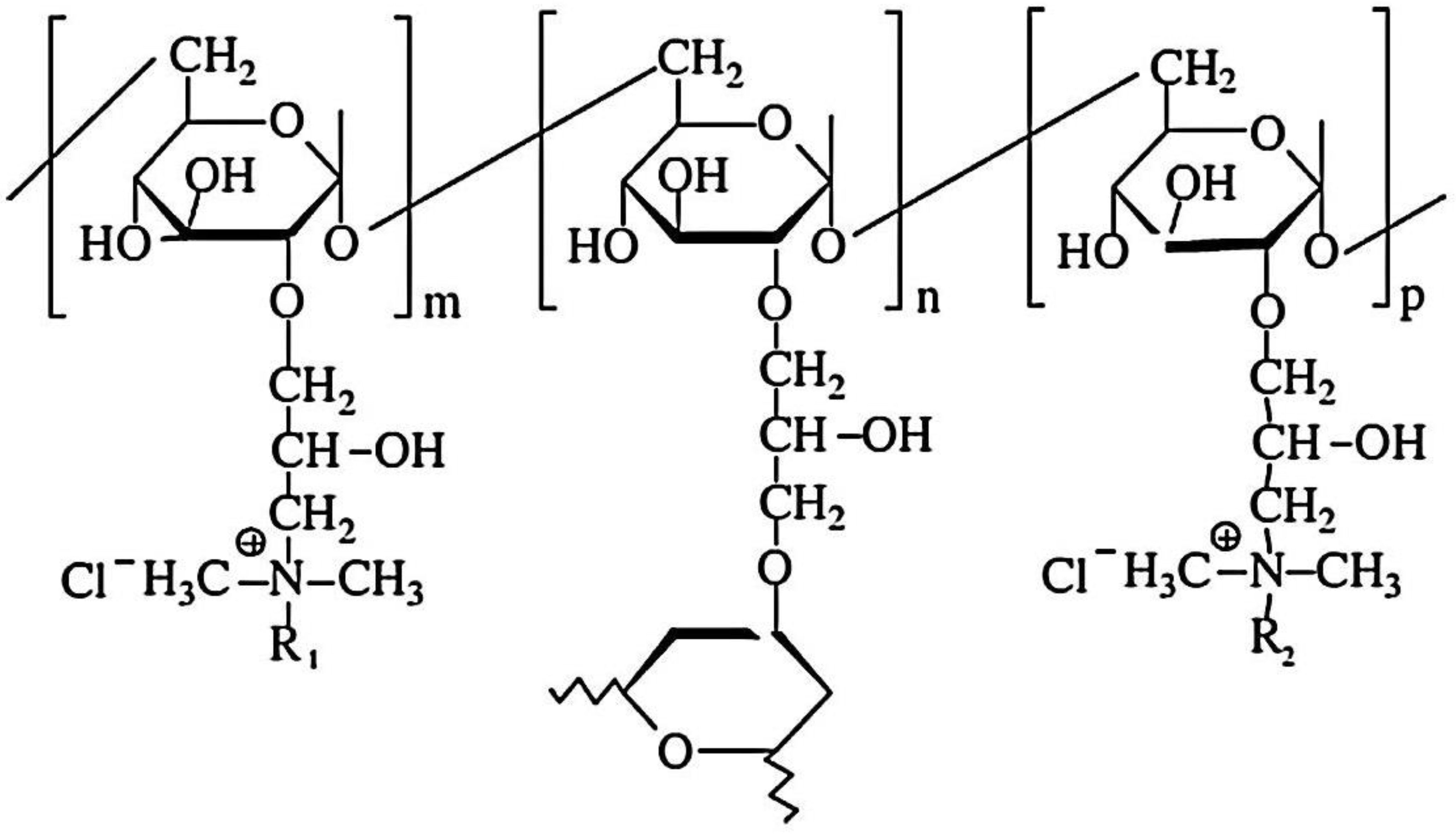
2.1.1. Chitosan and Chitin-Based Hydrogels
| Adsorbent | Dye | Dye Retention/Elimination Capacity | Reference |
|---|---|---|---|
| CS-g-P(AM-co-AA) | MO | 90% removal efficacy | [50] |
| Fe3O4, SiO2/CS–g-PAMPS | MB | 1000 mg/L | [51] |
| CS-g-EDA/CS-g-MA | CR | 1607 (mg/g) 1143 (mg/g) | [55] |
| CS-g-P(AM-co-NaMA) | Fuchsin | 97.2% removal efficacy | [56] |
| CS/MMT | RR 136 | 473 mg/g | [52] |
| Fe3O4@CS/P(AAM/NVIm) | MB | 860 mg/g | [53] |
| CS/Ag-HAp | RhB | 127.61 mg/g | [57] |
| CMC/IP6 | MO | 13.62 mg/g | [54] |
| CR | 8.49 mg/g | ||
| CS-gelatin/ZnO | CR | 90.8% photocatalytic activity | [58] |
| CS/PVA/PAA | MB | 596.14 mg/g | [60] |
| CS/iron and terephthalate ligands | CR | 590.8 mg/g | [61] |
| CS/nano-ZnO | RB 5 | 189.44 mg/g | [59] |
| CS/PA/GO | FY 3 | 296.5 ± 31.7(mg/g) | [64] |
| MB | 280.3 ± 23.9 (mg/g) | ||
| CS/TETA-GO | RB 221 | 54.2 mg/g (pH = 1) 34.8 mg/g (pH = 11) | [63] |
| chitin/GO | NR | 57 × 10−2 mmol/g | [67] |
| chitin-TRGO | CR | 230.5 mg/g | [68] |
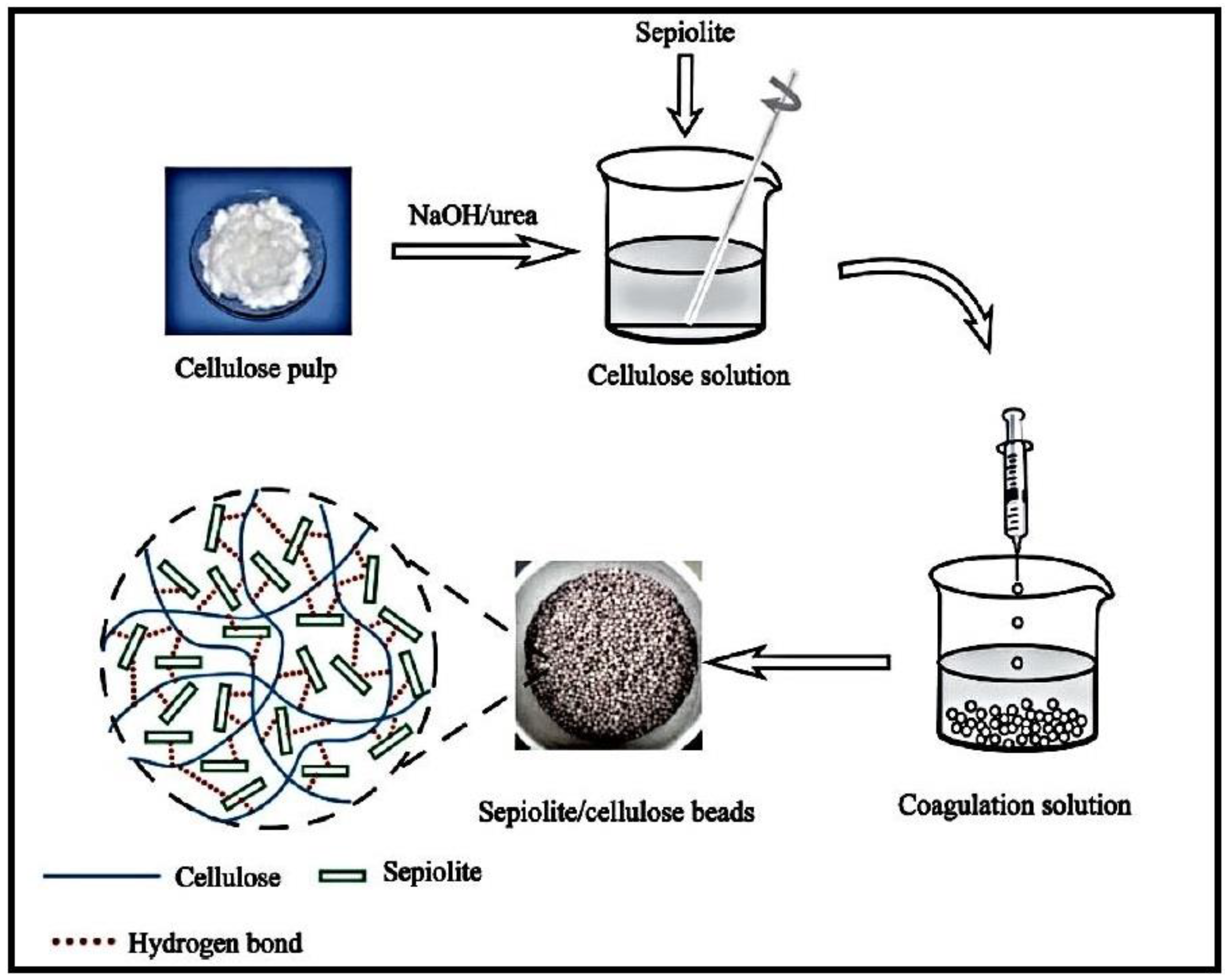
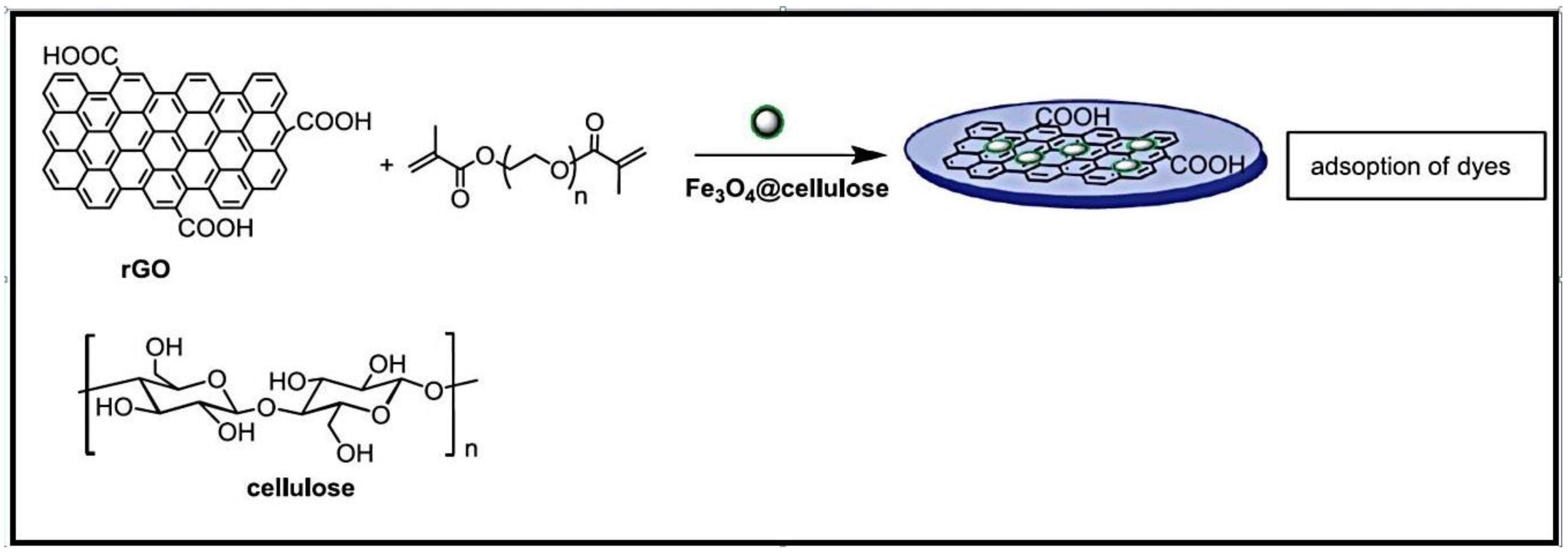
2.1.2. Cellulose-Based Hydrogels
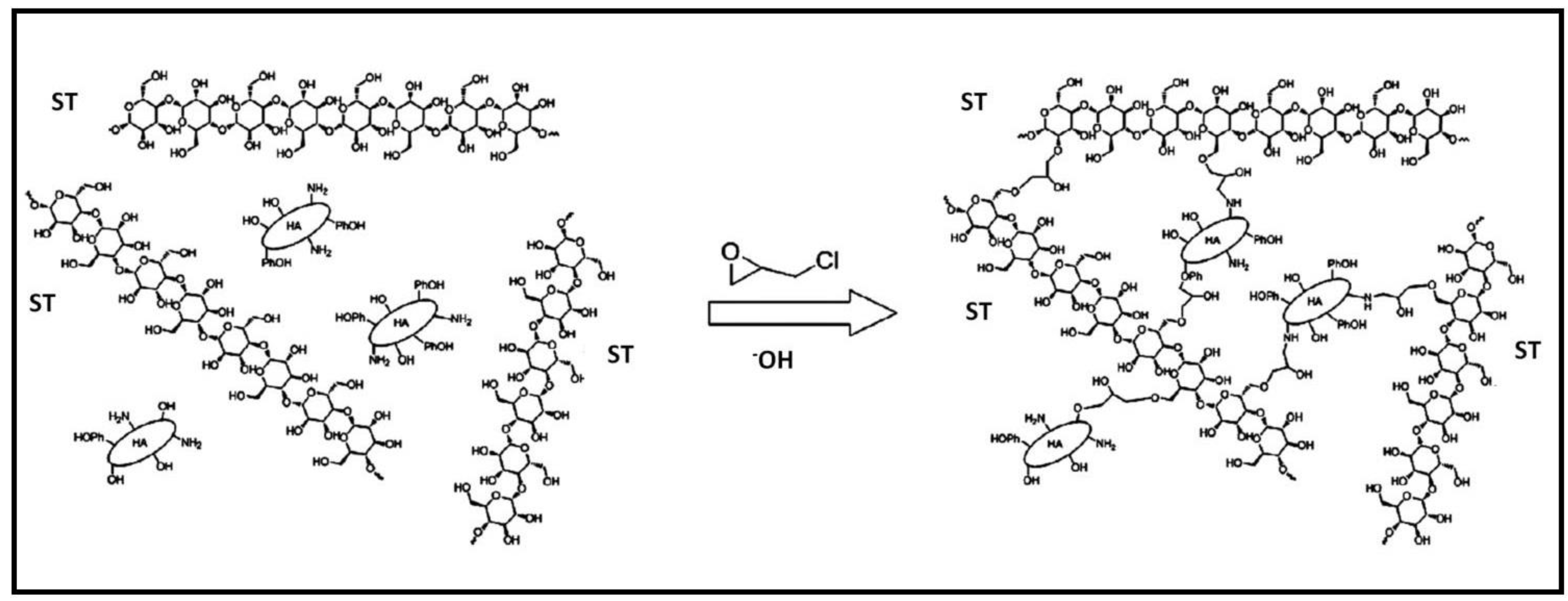
2.1.3. Starch-Based Hydrogels
| Adsorbent | Dye | Dye Retaining/Removal Capability | Reference |
|---|---|---|---|
| (CMST-g-PVIm/PVA/Fe3O4) | CV | 91.58 mg/g | [106] |
| CR | 83.66 mg/g | ||
| Fe3O4/ST-g-PAA | MV | 31.847 mg/g | [110] |
| ST phosphate | CV | 99% dye removal | [111] |
| Succinylated ST | MO | 20% dye removal | [112] |
| MB | 83% dye removal | ||
| ST@PAA | MB | 133.65 mg/g | [113] |
| CR | 64.73 mg/g | ||
| ST/HA | MB | 111.10 mg/g | [107] |
| ST-g-P(AA-co-AM)/PDA | MB | 2276 mg/g (pH 9) | [114] |
| ST-based sulfonic ion exchange resin | MB+RhB+MG | 84.04% decolorization | [115] |
| ST-g-P(AMPS-co-DMAEMA)/benzyl chloride | BV 7 | 600 mg/g | [116] |
| MST | MB | 70 mg/g | [117] |
| GST | 75 mg/g | ||
| VST | 81 mg/g | ||
| succinylated ST | MB | 84 mg/g | [118] |
| ST-g-P(AA-co-AMPS)/GO | MB | 769.23 mg/g | [109] |
| ST-g-PAM/GO/HAp | MG | 297 mg/g | [119] |
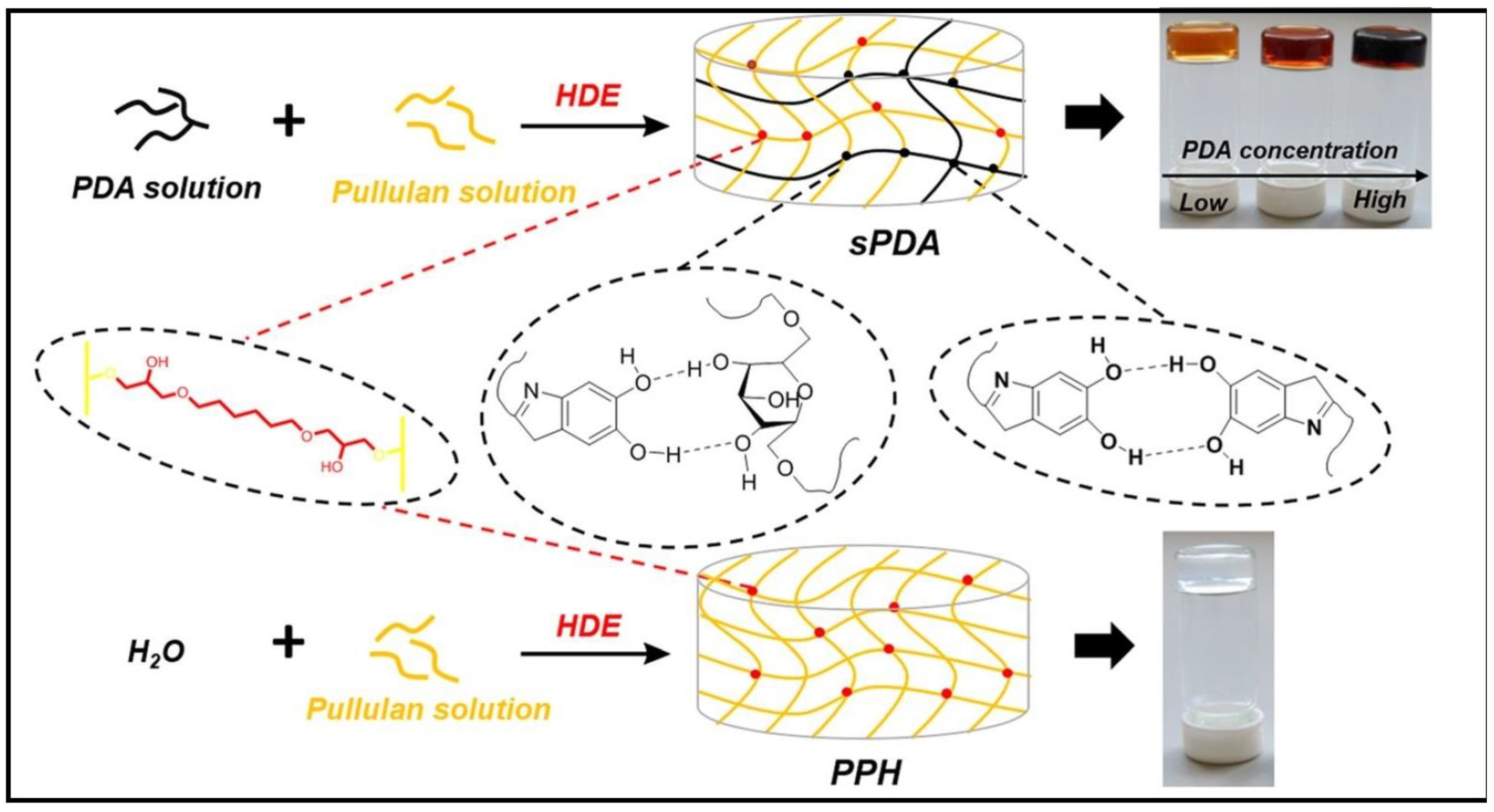
2.1.4. Pullulan-Based Hydrogels
2.1.5. Dextran-Based Hydrogels
| Adsorbent | Dye | Dye Retention/Removal Capacity | Reference |
|---|---|---|---|
| PUL/PDA/HDE | CV | 108 mg/g | [120] |
| PUL/PAM/GO | MB | 438.7 mg/g | [121] |
| PUL/PAM/AC | MB | 591.4 mg/g | [122] |
| PUL/PDA/MMT | CV | 112.45 mg/g | [123] |
| PUL/MMT | CV | 80 mg/g | [124] |
| PUL-g-PAM | MB | 386.81 mg/g | [125] |
| RB 2 | 273.24 mg/g | ||
| PUL-g-PAPTAC | Azocarmine B | 113.63 mg/g | [131] |
| Dex-Q2 | MO | 705 mg/g | [129] |
| IC | 732 mg/g | ||
| Orange II | 652 mg/g | ||
| RB | 654 mg/g | ||
| Dex-Q1 | MO | 893 mg/g | [126] |
| RB | 1718 mg/g | ||
| Dex-GMA/PAA | MB | 1994 mg/g | [130] |
| CV | 2390 mg/g |
2.2. The Effect of Various Factors on Adsorption Capacity
2.2.1. Initial Dye Concentration
2.2.2. Adsorbent Amount
2.2.3. Contact Time
2.2.4. Dye Type
2.2.5. pH
2.3. Adsorption Kinetics
2.3.1. Pseudo-First-Order Model (PFOM)
2.3.2. Pseudo-Second-Order Model (PSOM)
2.3.3. Intraparticle Diffusion Model (IDM)
2.4. Adsorption Isotherms
2.4.1. Langmuir Model (LM)
2.4.2. Freundlich Model (FM)
2.4.3. Dubinin–Radushkevich Model (D-RM)
2.4.4. Sips Model (SM)
2.4.5. Hill Model (HM)
2.5. Thermodynamic Parameters
2.6. Reusing of Adsorbents Based on Natural Polysaccharide Hydrogels
3. Conclusions and Future Prospectives
- ➢
- Following several treatment cycles, the adsorbents undergo a decline in their active sites and transform into waste, thereby causing additional pollution. Hence, it is crucial to address this problem in an environmentally-friendly manner. One potential solution is to utilize the used adsorbent for various purposes, such as catalysis, antimicrobial applications, or energy generation, in order to minimize or eliminate waste production.
- ➢
- Most adsorption studies, performed with polysaccharide-based hydrogels as adsorbents, are conducted at certain pH values, initial adsorbate amounts, adsorption rates, and isotherms. To overcome these limitations, scientists have achieved spectral and computational studies aiming to clarify the detailed adsorption mechanism on atomic or molecular scale.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AA | acrylamide | IP6 | phytic acid (inositol hexaphosphate) |
| AAM | acrylamidomethylated | KPS | potassium persulfate |
| AB 93 | Acid Blue 93 | MA | methyl acrylate |
| AB 113 | Acid blue 133 | MAM | methacrylamide |
| AMPS | 2-acrylamido-propanesulphonic acid | MB | Methylene Blue |
| AO7 | Acid Orange 7 | MBA | N, N-methylene bisacrylamide |
| APS | ammonium persulfate | METAC | 2-(methacryloyloxyethyl)trimethylammonium chloride |
| APTAC | (3-acrylamidopropyl)trimethyl ammonium chloride | MG | Malachite green |
| BEPE | 1,2-bis(2,3-epoxypropoxy)-ethane | MMT | montmorillonite |
| BNTN | bentonite | MO | Methyl Orange |
| BV 7 | Basic Violet 7 | MST | malonate starch |
| c-GO | carboxylated graphene oxide | MV | Methyl violet |
| CDNMA | acrylamidomethylated cyclodextrin | NaCMC | sodium carboxymethyl cellulose |
| CMC | carboxymethyl cellulose | NaMA | sodium methacrylate |
| CMST | carboxymethyl starch | NaMMT | sodium montmorillonite |
| CN | cellulose nanofibrillated | NR | Neutral Red |
| CS | chitosan | NVIm | N-vinylimidazole |
| CR | Congo Red | PA | polyacrylate |
| CV | Crystal violet | PAA | poly(acrylic acid) |
| β-CD | β-cyclodextrin | PAM | polyacrylamide |
| DMAEMA | dimethylaminoethyl methacrylate | PAMPS | poly-2-acrylamido-2-methylpropansulfonic acid |
| DMAM | dimethyl acrylamide | PClAETA | poly[2-(acryloyloxy)ethyl]trimethylammonium chloride |
| DB 38 | Direct Black 38 | PDA | polydopamine |
| DB BlN | Disperse Blue BLN | PEG DMA | poly(ethylene glycol) dimethacrylate |
| DR 81 | Direct Red 81 | PNVIm | poly N-vinylimidazole |
| ECH | epichlorohydrin | PVA | polyvinyl amine |
| EDA | ethylenediamine | QDs | quantum dots |
| FY 3 | Food yellow 3 | RB | Rose Bengal |
| g-C3H4 | graphitic carbon nitride | RB 2 | Reactive Blue 2 |
| GA | glutaraldehyde | RB 5 | Reactive Black 5 |
| GMA | glycidyl methacrylate | RB 221 | Reactive Blue 221 |
| GO | graphene oxide | rGO | reduced graphene oxide |
| GST | glutarate starch | RhB | Rhodamine B |
| HA | humic acid | RR 136 | Reactive red 136 |
| HAp | hydroxyapatite | SBC | sugarcane cellulose |
| HDE | 1–6-hexanediol diglycidyl ether | Sep | sepiolite |
| HEST | hydroxyethyl starch | ST | starch |
| HPC | hydroxypropyl cellulose | TA | tannic acid |
| HPMC | hydroxypropylmethyl cellulose | TETA | triethylenetetramine |
| HPSST | hydroxypropyl sulfate starch | TGDE | tetramethylene glycol diglycidyl ether |
| IA | itaconic acid | VST | valerate starch |
| IC | Indigo Carmin | r.t. | room temperature |
References
- Zhou, Y.; Lu, J.; Zhou, Y.; Liu, Y. Recent advances for dyes removal using novel adsorbents: A review. Environ. Pollut. 2019, 252, 352–365. [Google Scholar] [CrossRef]
- Ahmad, A.; Mohd-Setapar, S.H.; Chuong, C.S.; Khatoon, A.; Wani, W.A.; Kumar, R.; Rafatullah, M. Recent advances in new generation dye removal technologies: Novel search for approaches to reprocess wastewater. RSC Adv. 2015, 5, 30801–30818. [Google Scholar] [CrossRef]
- Adeyemo, A.A.; Adeoye, I.O.; Bello, O.S. Adsorption of dyes using different types of clay: A review. Appl. Water Sci. 2017, 7, 543–568. [Google Scholar] [CrossRef]
- Godiya, C.B.; Ruotolo, L.A.M.; Cai, W. Functional biobased hydrogels for the removal of aqueous hazardous pollutants: Current status, challenges, and future perspectives. J. Mater. Chem. A 2020, 8, 21585–21612. [Google Scholar] [CrossRef]
- Van Tran, V.; Park, D.; Lee, Y.C. Hydrogel applications for adsorption of contaminants in water and wastewater treatment. Environ. Sci. Pollut. Res. 2018, 25, 24569–24599. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid. Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef]
- Pavithra, K.G.; Senthil, K.P.; Jaikumar, V.; Sundar, R.P. Removal of colorants from wastewater: A review on sources and treatment strategies. J. Ind. Eng. Chem. 2019, 75, 1–19. [Google Scholar] [CrossRef]
- Ren, L.; Zhao, G.; Pan, L.; Chen, B.; Chen, Y.; Zhang, Q.; Xiao, X.; Xu, W. Efficient Removal of Dye from Wastewater without Selectivity Using Activated Carbon-Juncus effusus Porous Fibril Composites. ACS Appl. Mater. Interfaces 2021, 13, 19176–19186. [Google Scholar] [CrossRef]
- Das, A.; Maji, K.; Naskar, S.; Manna, U. Facile optimization of hierarchical topography and chemistry on magnetically active graphene oxide nanosheets. Chem. Sci. 2020, 11, 6556–6566. [Google Scholar] [CrossRef]
- Shalla, A.H.; Bhat, M.A.; Yaseen, Z. Hydrogels for removal of recalcitrant organic dyes: A conceptual overview. J. Environ. Chem. Eng. 2018, 6, 5938–5949. [Google Scholar] [CrossRef]
- Kadhom, M.; Albayati, N.; Alalwan, H.; Al-Furaiji, M. Removal of dyes by agricultural waste. Sustain. Chem. Pharm. 2020, 16, 100259. [Google Scholar] [CrossRef]
- Alves, A.; Lasmar, D.; de Andrade Miranda, I.; da Silva, C.J.; dos Santos Reis, J. The Potential of Activated Carbon in the Treatment of Water for Human Consumption, a Study of the State of the Art and Its Techniques Used for Its Development. Adv. Biosci. Biotechnol. 2021, 12, 143–153. [Google Scholar] [CrossRef]
- Varaprasad, K.; Raghavendra, G.M.; Jayaramudu, T.; Yallapu, M.M.; Sadiku, R. A mini review on hydrogels classification and recent developments in miscellaneous applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 79, 958–971. [Google Scholar] [CrossRef]
- Sharma, S.; Tiwari, S. A review on biomacromolecular hydrogel classification and its applications. Int. J. Biol. Macromol. 2020, 162, 737–747. [Google Scholar] [CrossRef]
- Enas, M.A. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- He, W.; Di, Y.; Jiang, N.; Liu, Z.; Chen, Y. Graphene-Oxide Seeds Nucleate Strong and Tough Hydrogel-Based Artificial Spider Silk. Acta Phys. Chim. Sin. 2022, 38, 2204059. [Google Scholar] [CrossRef]
- Wu, P.; Zhang, J.; Chen, Q.; Peng, W.; Wang, B. Theoretical perspective on mononuclear copper-oxygen mediated C–H and O–H activations: A comparison between biological and synthetic systems. Chin. J. Catal. 2022, 43, 913–927. [Google Scholar] [CrossRef]
- Elsayed, M.M. Hydrogel Preparation Technologies: Relevance Kinetics, Thermodynamics and Scaling up Aspects. J. Polym. Environ. 2019, 27, 871–891. [Google Scholar] [CrossRef]
- Schulze, J.; Hendrikx, S.; Schulz-Siegmund, M.; Aigner, A. Microparticulate poly(vinylalcohol) hydrogel formulations for embedding and controlled release of polyethylenimine (PEI)-based nanoparticles. Acta Biomater. 2016, 45, 210–222. [Google Scholar] [CrossRef]
- Wang, R.; Both, S.K.; Geven, M.; Calucci, L.; Forte, C.; Dijkstra, P.J.; Karperien, M. Kinetically stable metal ligand charge transfer complexes as crosslinks in nanogels/hydrogels: Physical properties and cytotoxicity. Acta Biomater. 2015, 26, 136–144. [Google Scholar] [CrossRef]
- Masruchin, N.; Park, B.-D.; Causin, V. Influence of sonication treatment on supramolecular cellulose microfibril-based hydrogels induced by ionic interaction. J. Ind. Eng. Chem. 2015, 29, 265–272. [Google Scholar] [CrossRef]
- Ishii-Mizuno, Y.; Umeki, Y.; Onuki, Y.; Watanabe, H.; Takahashi, Y.; Takakura, Y.; Nishikawa, M. Improved sustained release of antigen from immunostimulatory DNA hydrogel by electrostatic interaction with chitosan. Int. J. Pharm. 2017, 516, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Li, X.; Shen, Y.; Chang, G.; Yang, J.; Gu, Z. Self-healing pH-sensitive cytosine- and guanosine-modified hyaluronic acid hydrogels via hydrogen bonding. Polymer 2017, 108, 348–360. [Google Scholar] [CrossRef]
- Feng, W.; Zhou, W.; Dai, Z.; Yasin, A.; Yang, H. Tough polypseudorotaxane supramolecular hydrogels with dual-responsive shape memory properties. J. Mater. Chem. B 2016, 4, 1924–1931. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Moncal, K.K.; Li, J.; Peng, W.; Rivero, I.; Martin, J.A.; Ozbolat, I.T. Three-dimensional bioprinting using self-assembling scalable scaffold-free “tissue strands” as a new bioink. Sci. Rep. 2016, 6, 28714. [Google Scholar] [CrossRef] [PubMed]
- Abaee, A.; Madadlou, A.; Saboury, A.A. The formation of non-heat-treated whey protein cold-set hydrogels via non-toxic chemical cross-linking. Food Hydrocoll. 2017, 63, 43–49. [Google Scholar] [CrossRef]
- Tran, T.H.; Okabe, H.; Hidaka, Y.; Hara, K. Removal of metal ions from aqueous solutions using carboxymethyl cellulose/sodium styrene sulfonate gels prepared by radiation grafting. Carbohydr. Polym. 2017, 157, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wei, J. Hydrogel brushes grafted from stainless steel via surface-initiated atom transfer radical polymerization for marine antifouling. Appl. Surf. Sci. 2016, 382, 202–216. [Google Scholar] [CrossRef]
- Zhao, H.; Gao, J.; Liu, R.; Zhao, S. Stimulus-responsiveness and methyl violet release behaviors of poly(NIPAAm-co-AA) hydrogels chemically crosslinked with β-cyclodextrin polymer bearing methacrylates. Carbohydr. Res. 2016, 428, 79–86. [Google Scholar] [CrossRef]
- Kobe, R.; Yoshitani, K.; Teramoto, Y. Fabrication of elastic composite hydrogels using surface-modified cellulose nanofiber as a multifunctional crosslinker. J. Appl. Polym. Sci. 2016, 133, 42906. [Google Scholar] [CrossRef]
- Wei, Q.; Xu, M.; Liao, C.; Wu, Q.; Liu, M.; Zhang, Y.; Wu, C.; Cheng, L.; Wang, Q. Printable hybrid hydrogel by dual enzymatic polymerization with superactivity. Chem. Sci. 2016, 7, 2748–2752. [Google Scholar] [CrossRef] [PubMed]
- Miladinovic, Z.R.; Micic, M.; Suljovrujic, E. Temperature/pH dual responsive OPGMA based copolymeric hydrogels prepared by gamma radiation: An optimisation study. J. Polym. Res. 2016, 23, 77. [Google Scholar] [CrossRef]
- Chaykar, A.S.; Goharpey, F.; Yeganeh, J.K. Volume phase transition of electron beam cross-linked thermo-responsive PVME nanogels in the presence and absence of nanoparticles: With a view toward rheology and interactions. RSC Adv. 2016, 6, 9693–9708. [Google Scholar] [CrossRef]
- Cruz, A.; García-Uriostegui, L.; Ortega, A.; Isoshima, T.; Burillo, G. Radiation grafting of N-vinyl caprolactamonto nano and macrogels of chitosan: Synthesis and characterization. Carbohydr. Polym. 2017, 155, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Sadat Ebrahimi, M.-M.; Voss, Y.; Schönherr, H. Rapid detection of Escherichia coli via enzymatically triggered reactions in self-reporting chitosan hydrogels. ACS. Appl. Mater. Interfaces 2015, 7, 20190–20199. [Google Scholar] [CrossRef] [PubMed]
- An, Y.-M.; Liu, T.; Tian, R.; Liu, S.-X.; Han, Y.-N.; Wang, Q.-Q.; Sheng, W.-J. Synthesis of novel temperature responsive PEG-b-[PCL-g-P(MEO2MA-co-OEGMA)]-b-PEG (tBG) triblock-graft copolymers and preparation of tBG/graphene oxide composite hydrogels via click chemistry. React. Funct. Polym. 2015, 94, 1–8. [Google Scholar] [CrossRef]
- Varaprasad, K.; Sadiku, R. Development of microbial protective Kolliphor-based nanocomposite hydrogels. J. Appl. Polym. Sci. 2015, 132, 42781–42787. [Google Scholar] [CrossRef]
- Gonçalves, M.M.; Lobsinger, K.L.; Carneiro, J.; Picheth, G.F.; Pires, C.; Saul, C.K.; Maluf, D.F.; Pontarolo, R. Morphological study of electrospun chitosan/poly(vinyl alcohol)/glycerol nanofibres for skin care applications. Int. J. Biol. Macromol. 2022, 194, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Mitura, S.; Sionkowska, A.; Jaiswal, A. Biopolymers for hydrogels in cosmetics. J. Mater. Sci. Mater. Med. 2020, 31, 50. [Google Scholar] [CrossRef]
- Essawy, H.A.; Ghazy, M.B.M.; El-Hai, F.A.; Mohamed, M.F. Superabsorbent hydrogels via graft polymerization of acrylic acid fromchitosan-cellulose hybrid and their potential in controlled release of soil nutrients. Int. J. Biol. Macromol. 2016, 89, 144–151. [Google Scholar] [CrossRef]
- Majeed, F.; Razzaq, A.; Rehmat, S.; Azhar, I.; Mohyuddin, A.; Rizvi, N.B. Enhanced dye sequestration with natural polysaccharides-based hydrogels: A review. Carbohydr. Polym. 2024, 330, 121820. [Google Scholar] [CrossRef]
- Aranaz, A.S.I.; Ahmad, R.; Khan, M.S.; Kant, R.; Shahid, S.; Gautam, L.; Hasan, G.M.; Hassan, M.I. Chitin and its derivatives: Structural properties and biomedical applications. Int. J. Biol. Macromol. 2020, 164, 526–539. [Google Scholar] [CrossRef]
- Hevira, L.; Ighalo, J.O.; Sondari, D. Chitosan-based polysaccharides for effective synthetic dye adsorption. J. Mol. Liq. 2024, 393, 123604. [Google Scholar] [CrossRef]
- Huang, X.; Hadi, P.; Joshi, R.; Alhamzani, A.G.; Hsiao, B.S. A Comparative Study of Mechanism and Performance of Anionic and Cationic Dialdehyde Nanocelluloses for Dye Adsorption and Separation. ACS Omega 2023, 8, 8634–8649. [Google Scholar] [CrossRef]
- Kolya, H.; Kang, C.-W. Synthesis of starch-based smart hydrogel derived from rice-cooked wastewater for agricultural use. Int. J. Biol. Macromol. 2023, 226, 1477–1489. [Google Scholar] [CrossRef]
- Wani, S.M.; Mir, S.A.; Khanday, F.; Masoodi, F. Advances in pullulan production from agro-based wastes by Aureobasidium pullulans and its applications. Innov. Food Sci. Emerg. Technol. 2021, 74, 102846. [Google Scholar] [CrossRef]
- Ye, G.; Li, G.; Wang, C.; Ling, B.; Yang, R.; Huang, S. Extraction and characterization of dextran from Leuconostoc pseudomesenteroides YB-2 isolated from mango juice. Carbohydr. Polym. 2019, 207, 218–223. [Google Scholar] [CrossRef]
- Aranaz, I.; Alcántara, A.R.; Civera, M.C.; Arias, C.; Elorza, B.; Heras Caballero, A.; Acosta, N. Chitosan: An Overview of Its Properties and Applications. Polymers 2021, 13, 3256. [Google Scholar] [CrossRef] [PubMed]
- Kyzas, G.Z.; Bikiaris, D.N.; Mitropoulos, A.C. Chitosan adsorbents for dye removal: A review. Polym. Int. 2017, 66, 1800–1811. [Google Scholar] [CrossRef]
- Kumar, D.; Gihar, S.; Shrivash, M.K.; Kumar, P.; Kundu, P.P. A review on the synthesis of graft copolymers of chitosan and their potential applications. Int. Biol. Macromol. 2020, 163, 2097–2112. [Google Scholar] [CrossRef]
- Xu, B.; Zheng, H.; Wang, Y.; An, Y.; Luo, K.; Zhao, C.; Xiang, W. Poly (2-acrylamido-2- methylpropane sulfonic acid) grafted magnetic chitosan microspheres: Preparation, characterization and dye adsorption. Int. J. Biol. Macromol. 2018, 112, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Cai, J.; Zhong, L.; Cheng, H.; Wang, H.; Ma, Q. Adsorption of reactive red 136 onto chitosan/montmorillonite intercalated composite from aqueous solution. Appl. Clay Sci. 2019, 167, 9–22. [Google Scholar] [CrossRef]
- Hingrajiya, R.D.; Patel, M.P. Fe3O4 modified chitosan based co-polymeric magnetic composite hydrogel: Synthesis, characterization and evaluation for the removal of methylene blue from aqueous solutions. Int. J. Biol. Macromol. 2023, 244, 125251. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Zhao, H.; Gao, L.; Qin, Z.; Ma, J.; Han, Y.; Jiao, T. Preparation of carboxymethyl chitosan/phytic acid composite hydrogels for rapid dye adsorption in wastewater treatment. Colloids Surf. A Physicochem. Eng. Asp. 2021, 628, 127355. [Google Scholar] [CrossRef]
- Tahira, I.; Aslam, Z.; Abbas, A.; Monim-ul-Mehboob, M.; Ali, S.; Asghar, A. Adsorptive removal of acidic dye onto grafted chitosan: A plausible grafting and adsorption mechanism. Int. J. Biol. Macromol. 2019, 136, 1209–1218. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.G.; Sayed, A.Z.; Abd El-Wahab, H.; Sayah, M.M. Synthesis of a hydrogel by grafting of acrylamide-co-sodium methacrylate onto chitosan for effective adsorption of Fuchsin basic dye. Int. J. Biol. Macromol. 2020, 159, 422–432. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Iqbal, J.; Zhu, Y.; Zhang, P.; Chen, W.; Bhatnagar, A.; Du, Y. Chitosan/Ag-hydroxyapatite nanocomposite beads as a potential adsorbent for the efficient removal of toxic aquatic pollutants. Int. J. Biol. Macromol. 2018, 120, 1752–1759. [Google Scholar] [CrossRef] [PubMed]
- Sethi, S.; Thakur, M.S. Synthesis and characterization of nanocomposite chitosan-gelatin hydrogel loaded with ZnO and its application in photocatalytic dye degradation. Mater. Today Proc. 2023, 78, 815–824. [Google Scholar] [CrossRef]
- Çınar, S.; Kaynar, Ü.H.; Aydemir, T.; Kaynar, S.Ç.; Ayvacıklı, M. An efficient removal of RB5 from aqueous solution by adsorption onto nano-ZnO/Chitosan composite beads. Int. J. Biol. Macromol. 2017, 96, 459–465. [Google Scholar] [CrossRef]
- Wan, X.; Rong, Z.; Zhu, K.; Wu, Y. Chitosan-based dual network composite hydrogel for efficient adsorption of methylene blue dye. Int. J. Biol. Macromol. 2022, 222, 725–735. [Google Scholar] [CrossRef]
- Jin, Y.; Li, Y.; Du, Q.; Chen, B.; Chen, K.; Zhang, Y.; Wang, M.; Sun, Y.; Zhao, S.; Jing, Z.; et al. Efficient adsorption of Congo red by MIL-53(Fe)/chitosan composite hydrogel spheres. Microporous Mesoporous Mater. 2023, 348, 112404. [Google Scholar] [CrossRef]
- Razaq, A.; Bibi, F.; Zheng, X.; Papadakis, R.; Jafri, S.H.M.; Li, H. Review on Graphene-, Graphene Oxide-, Reduced Graphene Oxide-Based Flexible Composites: From Fabrication to Applications. Materials 2022, 15, 1012. [Google Scholar] [CrossRef]
- Chiu, C.-W.; Wu, M.-T.; Lin, C.-L.; Li, J.-W.; Huang, C.-Y.; Soong, Y.-C.; Lee, J.C.-M.; Lee Sanchez, W.A.; Lin, H.-Y. Adsorption Performance for Reactive Blue 221 Dye of β-Chitosan/Polyamine Functionalized Graphene Oxide Hybrid Adsorbent with High Acid–Alkali Resistance Stability in Different Acid–Alkaline Environments. Nanomaterials 2020, 10, 748. [Google Scholar] [CrossRef]
- Chang, Z.; Chen, Y.; Tang, S.; Yang, J.; Chen, Y.; Chen, S.; Li, P.; Yang, Z. Construction of chitosan/polyacrylate/graphene oxide composite physical hydrogel by semi-dissolution/acidification/sol-gel transition method and its simultaneous cationic and anionic dye adsorption properties. Carbohydr. Polym. 2020, 229, 115431. [Google Scholar] [CrossRef] [PubMed]
- Chelu, M.; Musuc, A.M.; Popa, M.; Calderon-Moreno, J. Chitosan Hydrogels for Water Purification Applications. Gels 2023, 9, 664. [Google Scholar] [CrossRef]
- Saigl, Z.; Tifouti, O.; Alkhanbashi, B. Chitosan as adsorbent for removal of some organic dyes: A review. Chem. Pap. 2023, 77, 2363–2405. [Google Scholar] [CrossRef]
- González, J.A.; Villanueva, M.E.; Piehl, L.L.; Copello, G.J. Development of a chitin/graphene oxide hybrid composite for the removal of pollutant dyes: Adsorption and desorption study. Chem. Eng. J. 2015, 280, 41–48. [Google Scholar] [CrossRef]
- Liu, C.; Liu, H.; Tang, K.; Zhang, K.; Zou, Z.; Gao, X. High-Strength Chitin Based Hydrogels Reinforced by Tannic Acid Functionalized Graphene for Congo Red Adsorption. J. Polym. Environ. 2020, 28, 984–994. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, F.; Wu, J. Physically crosslinked hydrogels from polysaccharides prepared by freeze–thaw technique. React. Funct. Polym. 2013, 73, 923–928. [Google Scholar] [CrossRef]
- Rajbhandary, A.; Nilsson, B.L. Self-Assembling Hydrogels. In GELS HANDBOOK: Fundamentals, Properties and Applications Volume 1: Fundamentals of Hydrogels; World Scientific Tuck Link: Singapore, 2016; pp. 219–250. [Google Scholar]
- Feng, Y.; Gong, J.-L.; Zeng, G.-M.; Niu, Q.-Y.; Zhang, H.-Y.; Niu, C.-G.; Deng, J.-H.; Yan, M. Adsorption of Cd (II) and Zn (II) from aqueous solutions using magnetic hydroxyapatite nanoparticles as adsorbents. Chem. Eng. J. 2010, 162, 487–494. [Google Scholar] [CrossRef]
- Wang, J.; Wei, L.; Ma, Y.; Li, K.; Li, M.; Yu, Y.; Wang, L.; Qiu, H. Collagen/cellulose hydrogel beads reconstituted from ionic liquid solution for Cu(II) adsorption. Carbohyd. Polym. 2013, 98, 736–743. [Google Scholar] [CrossRef] [PubMed]
- Elbedwehy, A.M.; Atta, A.M. Novel Superadsorbent Highly Porous Hydrogel Based on Arabic Gum and Acrylamide Grafts for Fast and Efficient Methylene Blue Removal. Polymers 2020, 12, 338. [Google Scholar] [CrossRef] [PubMed]
- Pedroso-Santana, S.; Fleitas-Salazar, N. Ionotropic gelation method in the synthesis of nanoparticles/microparticles for biomedical purposes. Polym. Int. 2020, 69, 443–447. [Google Scholar] [CrossRef]
- Zainal, S.H.; Mohd, N.H.; Suhaili, N.; Anuar, F.H.; Lazim, A.M.; Othaman, R. Preparation of Cellulose-based Hydrogel: A Review. J. Mater. Res. Technol. 2021, 10, 935–952. [Google Scholar] [CrossRef]
- Khan, M.; Lo, I.M.C. A holistic review of hydrogel applications in the adsorptive removal of aqueous pollutants: Recent progress, challenges, and perspectives. Water Res. 2016, 106, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Pakdel, P.M.; Peighambardoust, S.J. A review on acrylic based hydrogels and their applications in wastewater treatment. J. Environ. Manag. 2018, 217, 123–143. [Google Scholar] [CrossRef]
- Dai, L.; Cheng, T.; Xi, X.; Nie, S.; Ke, H.; Liu, Y.; Tong, S.; Chen, Z. A versatile TOCN/CGG self-assembling hydrogel for integrated wastewater treatment. Cellulose 2020, 27, 915–925. [Google Scholar] [CrossRef]
- Weidong, X.; Ran, S.; Sihai, H.; Chengzhen, M.; Bin, X.; Mengying, Y.; Yaoguo, W. Recent advances and future perspective on lignocellulose-based materials as adsorbents in diverse water treatment applications. Int. J. Biol. Macromol. 2023, 253, 126984. [Google Scholar] [CrossRef]
- Akter, M.; Bhattacharjee, M.; Dhar, A.K.; Rahman, F.B.A.; Haque, S.; Rashid, T.U.; Kabir, S.M.F. Cellulose-Based Hydrogels for Wastewater Treatment: A Concise Review. Gels 2021, 7, 30. [Google Scholar] [CrossRef]
- Radoor, S.; Karayil, J.; Jayakumar, A.; Kandel, D.R.; Kim, J.T.; Siengchin, S.; Lee, J. Recent advances in cellulose- and alginate-based hydrogels for water and wastewater treatment: A review. Carbohydr. Polym. 2024, 323, 121339. [Google Scholar] [CrossRef]
- Aoudi, B.; Boluk, Y.; Gamal El-Din, M. Recent advances and future perspective on nanocellulose-based materials in diverse water treatment applications. Sci. Total Environ. 2022, 843, 156903. [Google Scholar] [CrossRef] [PubMed]
- Roa, K.; Tapiero, Y.; Thotiyl, M.O.; Sánchez, J. Hydrogels Based on Poly([2-(acryloxy)ethyl] Trimethylammonium Chloride) and Nanocellulose Applied to Remove Methyl Orange Dye from Water. Polymers 2021, 13, 2265. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Chen, S.; Duan, H.; He, J.; Luo, Y. Removal of anionic and cationic dyes using porous chitosan/carboxymethyl cellulose-PEG hydrogels: Optimization, adsorption kinetics, isotherm and thermodynamics studies. Int. J. Biol. Macromol. 2023, 231, 123213. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Liu, X.; Jin, R.; Nie, W.; Zhou, Y. Dye adsorption and photo-induced recycling of hydroxypropyl cellulose/molybdenum disulfide composite hydrogels. Carbohyd. Polym. 2017, 167, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, H.; Javadi, A. Fabrication and characterization of CMC-based magnetic superabsorbent hydrogel nanocomposites for crystal violet removal. Polym. Adv. Technol. 2016, 27, 1609–1616. [Google Scholar] [CrossRef]
- Hosseinzadeh, H.; Bahador, N. Novel CdS quantum dots templated hydrogel nanocomposites: Synthesis, characterization, swelling and dye adsorption properties. J. Mol. Liq. 2017, 240, 630–641. [Google Scholar] [CrossRef]
- Chen, Z.; Pan, Y.; Cai, P. Sugarcane cellulose-based composite hydrogel enhanced by g-C3N4 nanosheet for selective removal of organic dyes from water. Int. J. Biol. Macromol. 2022, 205, 37–48. [Google Scholar] [CrossRef]
- Pakdel, P.M.; Peighambardoust, S.J.; Arsalani, N.; Aghdasinia, H. Safranin-O cationic dye removal from wastewater using carboxymethyl cellulose-grafted-poly(acrylic acid-co-itaconic acid) nanocomposite hydrogel. Environ. Res. 2022, 212, 113201. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Yi, L.; Deng, H.; Sun, P. Dyes adsorption using a synthetic carboxymethyl cellulose-acrylic acid adsorbent. J. Environ. Sci. 2014, 26, 1203–1211. [Google Scholar] [CrossRef]
- Lin, Y.; Fang, G.G.; Deng, Y.J.; Shen, K.Z.; Wu, T.; Li, M. Highly Effective Removal of Methylene Blue Using a Chemi-Mechanical Pretreated Cellulose-based Superabsorbent Hydrogel. Bioresources 2018, 13, 8709–8722. [Google Scholar] [CrossRef]
- Liu, L.; Gao, Z.Y.; Su, X.P.; Chen, X.; Jiang, L.; Yao, J.M. Adsorption Removal of Dyes from Single and Binary Solutions Using a Cellulose-based Bioadsorbent. Acs. Sustain. Chem. Eng. 2015, 3, 432–442. [Google Scholar] [CrossRef]
- Ghemati, D.; Aliouche, D. Study of the Sorption of Synthetic Dyes from Aqueous Solution onto Cellulosic Modified Polymer. J. Water Chem. Technol. 2014, 36, 265–272. [Google Scholar] [CrossRef]
- Sharma, G.; Kumar, A.; Sharma, S.; Naushad, M.; Ghfar, A.A.; Al-Muhtaseb, A.H.; Ahamad, T.; Sharma, N.; Stadler, F.J. Carboxymethyl cellulose structured nano-adsorbent for removal of methyl violet from aqueous solution: Isotherm and kinetic analyses. Cellulose 2020, 27, 3677–3691. [Google Scholar] [CrossRef]
- Mahdavinia, G.R.; Hasanpour, J.; Rahmani, Z.; Karami, S.; Etemadi, H. Nanocomposite hydrogel from grafting of acrylamide onto HPMC using sodium montmorillonite nanoclay and removal of crystal violet dye. Cellulose 2013, 20, 2591–2604. [Google Scholar] [CrossRef]
- Shi, Y.; Xue, Z.; Wang, X.; Wang, L.; Wang, A. Removal of methylene blue from aqueous solution by sorption on lignocellulose-g-poly(acrylic acid)/montmorillonite three-dimensional crosslinked polymeric network hydrogels. Polym. Bull. 2013, 70, 1163–1179. [Google Scholar] [CrossRef]
- Dai, H.; Huang, Y.; Huang, H. Eco-friendly polyvinyl alcohol/carboxymethyl cellulose hydrogels reinforced with graphene oxide and bentonite for enhanced adsorption of methylene blue. Carbohydr. Polym. 2018, 185, 1–11. [Google Scholar] [CrossRef]
- Eltaweil, A.S.; Elgarhy, G.S.; El-Subruiti, G.M.; Omer, A.M. Novel carboxymethyl cellulose/carboxylated graphene oxide composite microbeads for efficient adsorption of cationic methylene blue dye. Int. J. Biol. Macromol. 2020, 154, 307–318. [Google Scholar] [CrossRef]
- Jiang, X.; Wang, S.; Ge, L.; Lin, F.; Lu, Q.; Wang, T.; Huang, B.; Lu, B. Development of organic-inorganic hybrid beads from sepiolite and cellulose for effective adsorption of malachite green. RSC Adv. 2017, 7, 38965–38972. [Google Scholar] [CrossRef]
- Varaprasad, K.; Jayaramudu, T.; Sadiku, E.R. Removal of dye by carboxymethyl cellulose, acrylamide and graphene oxide via a free radical polymerization process. Carbohydr. Polym. 2017, 164, 186–194. [Google Scholar] [CrossRef]
- Halouane, F.; Oz, Y.; Meziane, D.; Barras, A.; Juraszek, J.; Singh, S.K.; Kurungot, S.; Shaw, P.K.; Sanyal, R.; Boukherroub, R.; et al. Magnetic reduced graphene oxide loaded hydrogels: Highly versatile and efficient adsorbents for dyes and selective Cr(VI) ions removal. J. Colloid. Interface Sci. 2017, 507, 360–369. [Google Scholar] [CrossRef]
- Visakh, P.M.; Mathew, A.P.; Oksman, K.; Thomas, S. Starch-based bionanocomposites: Processing and properties. Polysaccharide building blocks: A sustainable approach to the development of renewable biomaterials. In Polysaccharide Building Blocks: A Sustainable Approach to the Development of Renewable Biomaterials; Habibi, Y., Lucia, L.A., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 287–306. [Google Scholar] [CrossRef]
- Khoo, P.S.; Ilyas, R.A.; Uda, M.N.A.; Hassan, S.A.; Nordin, A.H.; Norfarhana, A.S.; Ab Hamid, N.H.; Rani, M.S.A.; Abral, H.; Norrrahim, M.N.F. Starch-Based Polymer Materials as Advanced Adsorbents for Sustainable Water Treatment: Current Status, Challenges, and Future Perspectives. Polymers 2023, 15, 3114. [Google Scholar] [CrossRef] [PubMed]
- El-Ghany, N.A.A.; Abu Elella, M.H.; Abdallah, H.M.; Mostafa, M.S.; Samy, M. Recent Advances in Various Starch Formulation for Wastewater Purification via Adsorption Technique: A Review. J. Polym. Environ. 2023, 31, 2792–2825. [Google Scholar] [CrossRef]
- Haq, F.; Mehmood, S.; Haroon, M.; Kiran, M.; Waseem, K.; Aziz, T.; Farid, A. Role of Starch Based Materials as a Bio-sorbents for the Removal of Dyes and Heavy Metals from Wastewater. J. Polym. Environ. 2022, 30, 1730–1748. [Google Scholar] [CrossRef]
- Pour, Z.S.; Ghaemy, M. Removal of dyes and heavy metal ions from water by magnetic hydrogel beads based on poly(vinyl alcohol)/carboxymethyl starch-g-poly(vinyl imidazole). RSC Adv. 2015, 5, 64106–64118. [Google Scholar] [CrossRef]
- Chen, R.; Zhang, Y.; Shen, L.; Wang, X.; Chen, J.; Ma, A.; Jiang, W. Lead (II) and methylene blue removal using a fully biodegradable hydrogel based on starch immobilized humic acid. Chem. Eng. J. 2015, 268, 348–355. [Google Scholar] [CrossRef]
- Hebeish, A.A.; Aly, A.A. Synthesis, Characterization and Utilization of Starch Hydroxypropyl Sulphate for Cationic Dye Removal. Int. J. Org. Chem. 2014, 4, 208–217. [Google Scholar] [CrossRef][Green Version]
- Pourjavadi, A.; Nazari, M.; Kabiri, B.; Hosseini, S.H.; Bennett, C. Preparation of Porous Graphene Oxide/Hydrogel Nanocomposites and Their Ability for Efficient Adsorption of Methylene Blue. RSC Adv. 2016, 6, 10430–10437. [Google Scholar] [CrossRef]
- Saberi, A.; Alipour, E.; Sadeghi, M. Superabsorbent magnetic Fe3O4-based starch-poly (acrylic acid) nanocomposite hydrogel for efficient removal of dyes and heavy metal ions from water. J. Polym. Res. 2019, 26, 271. [Google Scholar] [CrossRef]
- Bahrami, M.; Amiri, M.; Bagheri, F. Optimization of crystal violet adsorption by chemically modified potato starch using response surface methodology. Pollution 2020, 6, 159–170. [Google Scholar] [CrossRef]
- Soto, D.; León, O.; Muñoz-Bonilla, A.; Fernandez-García, M. Succinylated Starches for Dye Removal. Starch-Stärke 2020, 73, 2000043. [Google Scholar] [CrossRef]
- Sarmah, D.; Karak, N. Double network hydrophobic starch based amphoteric hydrogel as an effective adsorbent for both cationic and anionic dyes. Carbohydr. Polym. 2020, 242, 116320. [Google Scholar] [CrossRef] [PubMed]
- Mahmoodi-Babolan, N.; Nematollahzadeh, A.; Heydari, A.; Merikhy, A. Bioinspired catecholamine/starch composites as superadsorbent for the environmental remediation. Int. J. Biol. Macromol. 2019, 125, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, Y.; Cheng, B.; Ding, C.; Zhang, Y. Synthesis of a starch-based sulfonic ion exchange resin and adsorption of dyestuffs to the resin. Int. J. Biol. Macromol. 2020, 161, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Farag, A.M.; Sokker, H.H.; Zayed, E.M.; Eldien, F.A.; Alrahman, N.; Abd, N.M. Removal of hazardous pollutants using bifunctional hydrogel obtained from modified starch by grafting copolymerization. Int. J. Biol. Macromol. 2018, 120, 188–2199. [Google Scholar] [CrossRef] [PubMed]
- Alvarado, N.; Abarca, R.L.; Urdaneta, J.; Romero, J.; Galotto, M.J.; Guarda, A. Cassava starch: Structural modification for development of a bio-adsorber for aqueous pollutants. Characterization and adsorption studies on methylene blue. Polym. Bull. 2021, 78, 1087–1107. [Google Scholar] [CrossRef]
- Chen, Q.-J.; Zheng, X.-M.; Zhou, L.-L.; Zhang, Y.-F. Adsorption of Cu (II) and Methylene Blue by Succinylated Starch Nanocrystals. Starch-Stärke 2019, 71, 1800266. [Google Scholar] [CrossRef]
- Hosseinzadeh, H.; Ramin, S. Fabrication of starch-graft poly(acrylamide)/graphene oxide/hydroxyapatite nanocomposite hydrogel adsorbent for removal of malachite green dye from aqueous solution. Int. J. Biol. Macromol. 2018, 106, 101–115. [Google Scholar] [CrossRef] [PubMed]
- Lipeng, W.; Mingyang, S.; Ronghui, G.; Wei, D. Development of a novel pullulan/polydopamine composite hydrogel adsorbent for dye removal. Colloids Surf. A Physicochem. Eng. Asp. 2022, 652, 129632. [Google Scholar] [CrossRef]
- Chen, K.; Li, Y.; Wang, M.; Wang, M.; Cui, M.; Du, Q.; Xu, W.; Li, L.; Chen, B.; Sun, Y.; et al. Removal of Methylene Blue from Wastewater Using a Ternary Composite Hydrogel System: Pullulan Polysaccharides Grafted with Polyacrylamide and Decorated with Graphene Oxide. J. Polym. Environ. 2022, 30, 4605–4618. [Google Scholar] [CrossRef]
- Chen, K.; Li, Y.; Du, Q.; Sun, Y.; Zhang, Y.; Chen, B.; Jing, Z.; Jin, Y.; Zhao, S. Removal of Methylene Blue Dye from Aqueous Solutions by Pullulan Polysaccharide/Polyacrylamide/Activated Carbon Complex Hydrogel Adsorption. ACS Omega 2023, 8, 857–867. [Google Scholar] [CrossRef]
- Qi, X.; Zeng, Q.; Tong, X.; Su, T.; Xie, L.; Yuan, K.; Xu, J.; Shen, J. Polydopamine/montmorillonite-embedded pullulan hydrogels as efficient adsorbents for removing crystal violet. J. Hazard. Mater. 2021, 402, 123359. [Google Scholar] [CrossRef] [PubMed]
- Su, T.; Wu, L.; Pan, X.; Zhang, C.; Shi, M.; Gao, R.; Qi, X.; Dong, W. Pullulan-derived nanocomposite hydrogels for wastewater remediation: Synthesis and characterization. J. Colloid Interface Sci. 2019, 542, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Saber-Samandari, S.; Gulcan, H.O.; Saber-Samandari, S.; Gazi, M. Efficient Removal of Anionic and Cationic Dyes from an Aqueous Solution Using Pullulan-graft-Polyacrylamide Porous Hydrogel. Water Air Soil Pollut. 2014, 225, 2177. [Google Scholar] [CrossRef]
- Stanciu, M.C.; Nichifor, M. Influence of dextran hydrogel characteristics on adsorption capacity for anionic dyes. Carbohydr. Polym. 2018, 199, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, M.C.; Nichifor, M.; Ailiesei, G.L. Bile salts adsorption on dextran-based hydrogels. Int. J. Biol. Macromol. 2021, 190, 270–283. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, M.C.; Nichifor, M.; Prisacaru, A.-I. Adsorption of Sodium Cholate on Cationic Dextran Gels: Comparison of Isotherm Binding Models. Mater. Plast. 2020, 57, 181–192. [Google Scholar] [CrossRef]
- Stanciu, M.C.; Nichifor, M. Adsorption of anionic dyes on a cationic amphiphilic dextran hydrogel: Equilibrium, kinetic, and thermodynamic studies. Colloid. Polym. Sci. 2019, 297, 45–57. [Google Scholar] [CrossRef]
- Yuan, Z.; Wang, J.; Wang, Y.; Liu, Q.; Zhong, Y.; Wang, Y.; Li, L.; Lincoln, S.F.; Guo, X. Preparation of a poly(acrylic acid) based hydrogel with fast adsorption rate and high adsorption capacity for the removal of cationic dyes. RSC Adv. 2019, 9, 21075–21085. [Google Scholar] [CrossRef] [PubMed]
- Constantin, M.; Asmarandei, I.; Harabagiu, V.; Ghimici, L.; Ascenzi, P.; Fundueanu, G. Removal of anionic dyes from aqueous solutions by an ion-exchanger based on pullulan microspheres. Carbohydr. Polym. 2013, 91, 74–84. [Google Scholar] [CrossRef]
- Wang, J.; Guo, X. Adsorption kinetic models: Physical meanings, applications, and solving methods. J. Hazard. Mat. 2020, 390, 122156. [Google Scholar] [CrossRef]
- Unuabonah, E.I.; Omorogie, M.O.; Oladoja, N.A. 5-Modeling in Adsorption: Fundamentals and Applications. In Micro and Nano Technologies, Composite Nanoadsorbents; Kyzas, G.Z., Mitropoulos, A.C., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 85–118. [Google Scholar] [CrossRef]
- Simonin, J.-P. On the comparison of pseudo-first order and pseudo-second order rate laws in the modeling of adsorption kinetics. Chem. Eng. J. 2016, 300, 254–263. [Google Scholar] [CrossRef]




| Polysaccharide | Chemical Structure | Characteristics | Ref. |
|---|---|---|---|
| chitin | 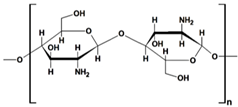 |
| [42] |
| chitosan | 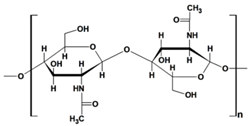 |
| [43] |
| cellulose | 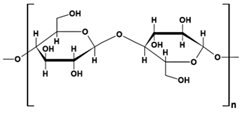 |
| [44] |
| starch |  amylose  Amylopectin |
| [45] |
| pullulan | 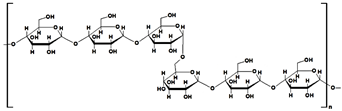 |
| [46] |
| dextran |  |
| [47] |
| Adsorbent | Dye | Dye Retaining/Elimination Ability | Reference |
|---|---|---|---|
| cellulose-g-PAM | MB | 734.816 mg/g | [82] |
| CMC-g-PAA | MO | 82.0% elimination capacity | [90] |
| DB BLN | 77.4% elimination capacity | ||
| MG | 95.3% elimination capacity | ||
| acryloyl cellulose-g-PAA | MB | 3003 mg/g | [91] |
| cellulose-g-P(AA-co-AM) | AB 93 | 82% elimination for both dyes | [92] |
| MB | |||
| cellulose-g-CDNMA | MB | 15 mg/g | [93] |
| MO | 12 mg/g | ||
| CNF-g-PClAETA | MO | 1379.0 mg/g | [83] |
| CS/CMC-PEG | CR | 1053.88 mg/g | [84] |
| MB | 331.72 mg/g | ||
| HPC-based MoS2 | MB | 6153 mg/g | [85] |
| CMC/g-C3H4/ZnO | MV | 96.43 mg/g | [94] |
| HPMC-g-PAM/NaMMT | CV | 76% removal efficiency (five cycles) | [95] |
| lignocellulose-g-PAA/MMT | MB | 1994.38 mg/g | [96] |
| CMC/PAA/Fe3+, Fe2+ | CV | 200 mg/L | [86] |
| CdS/QDs | RhB | 137 mg/g | [87] |
| g-C3N4@SBC/CMC | MB | 362.3 mg/g | [88] |
| CMC-g-P(AA-co-IA) | Safranin-O | 185,185 mg/g | [89] |
| CMC-g-P(AA-co-IA)/MMT | 191,205 mg/g | ||
| cellulose/Sep | MG | 314.47 mg/g | [99] |
| CMC/AM/GO | AB 133 | 185.45 mg/g | [100] |
| PVA/CMC/GO/BNTN | MB | 172.14 mg/g (30 °C) | [97] |
| CMC/c-GO | MB | 180.32 mg/g | [98] |
| rGO/cellulose/Fe2+, Fe3+/PEG DMA | MB | 119 mg/g | [101] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stanciu, M.-C.; Teacă, C.-A. Natural Polysaccharide-Based Hydrogels Used for Dye Removal. Gels 2024, 10, 243. https://doi.org/10.3390/gels10040243
Stanciu M-C, Teacă C-A. Natural Polysaccharide-Based Hydrogels Used for Dye Removal. Gels. 2024; 10(4):243. https://doi.org/10.3390/gels10040243
Chicago/Turabian StyleStanciu, Magdalena-Cristina, and Carmen-Alice Teacă. 2024. "Natural Polysaccharide-Based Hydrogels Used for Dye Removal" Gels 10, no. 4: 243. https://doi.org/10.3390/gels10040243
APA StyleStanciu, M.-C., & Teacă, C.-A. (2024). Natural Polysaccharide-Based Hydrogels Used for Dye Removal. Gels, 10(4), 243. https://doi.org/10.3390/gels10040243