New Formulations of Polysaccharide-Based Hydrogels for Drug Release and Tissue Engineering
Abstract
:1. Introduction
2. Polysaccharide-Based Hydrogels
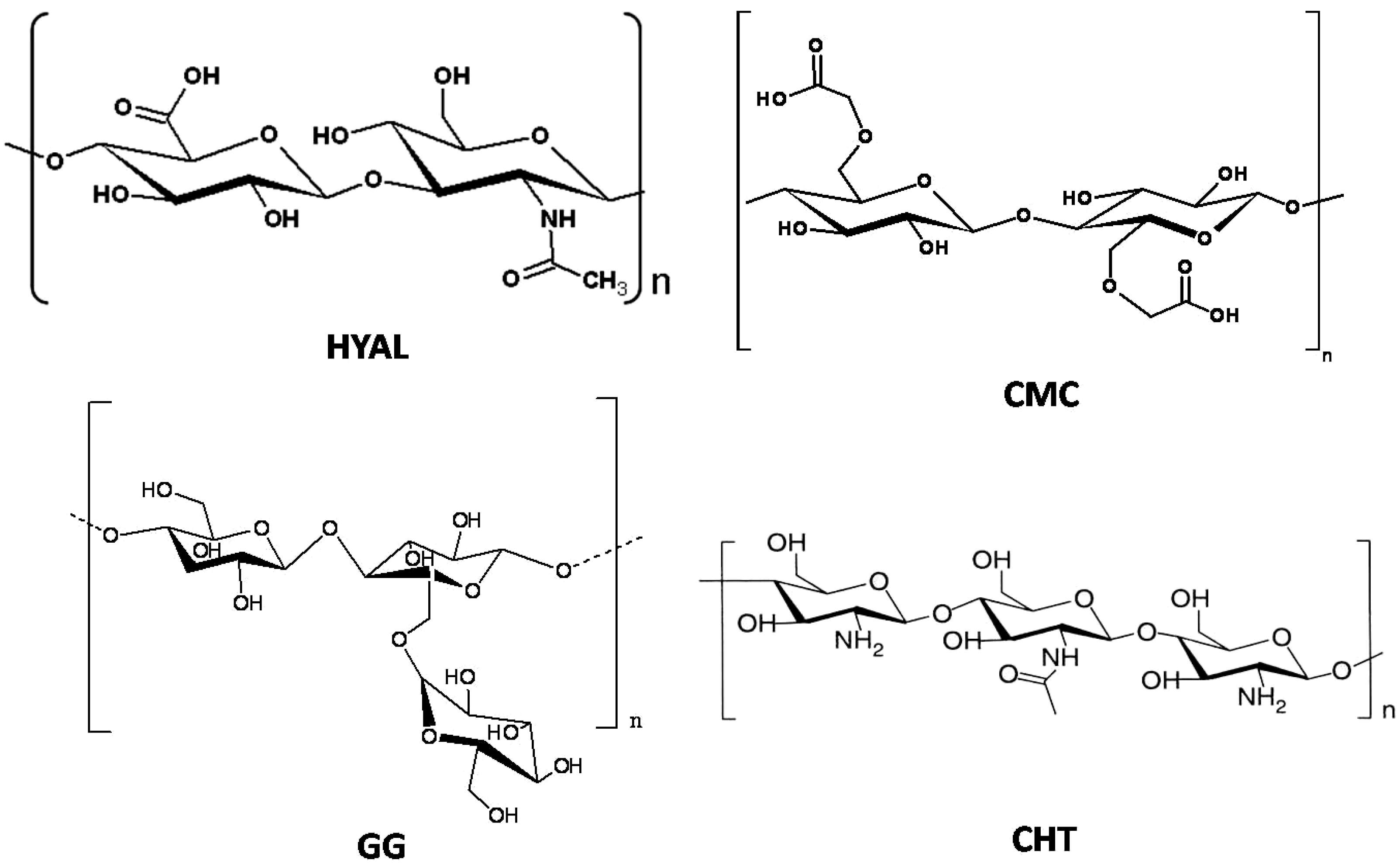
2.1. Cross-Linking Density
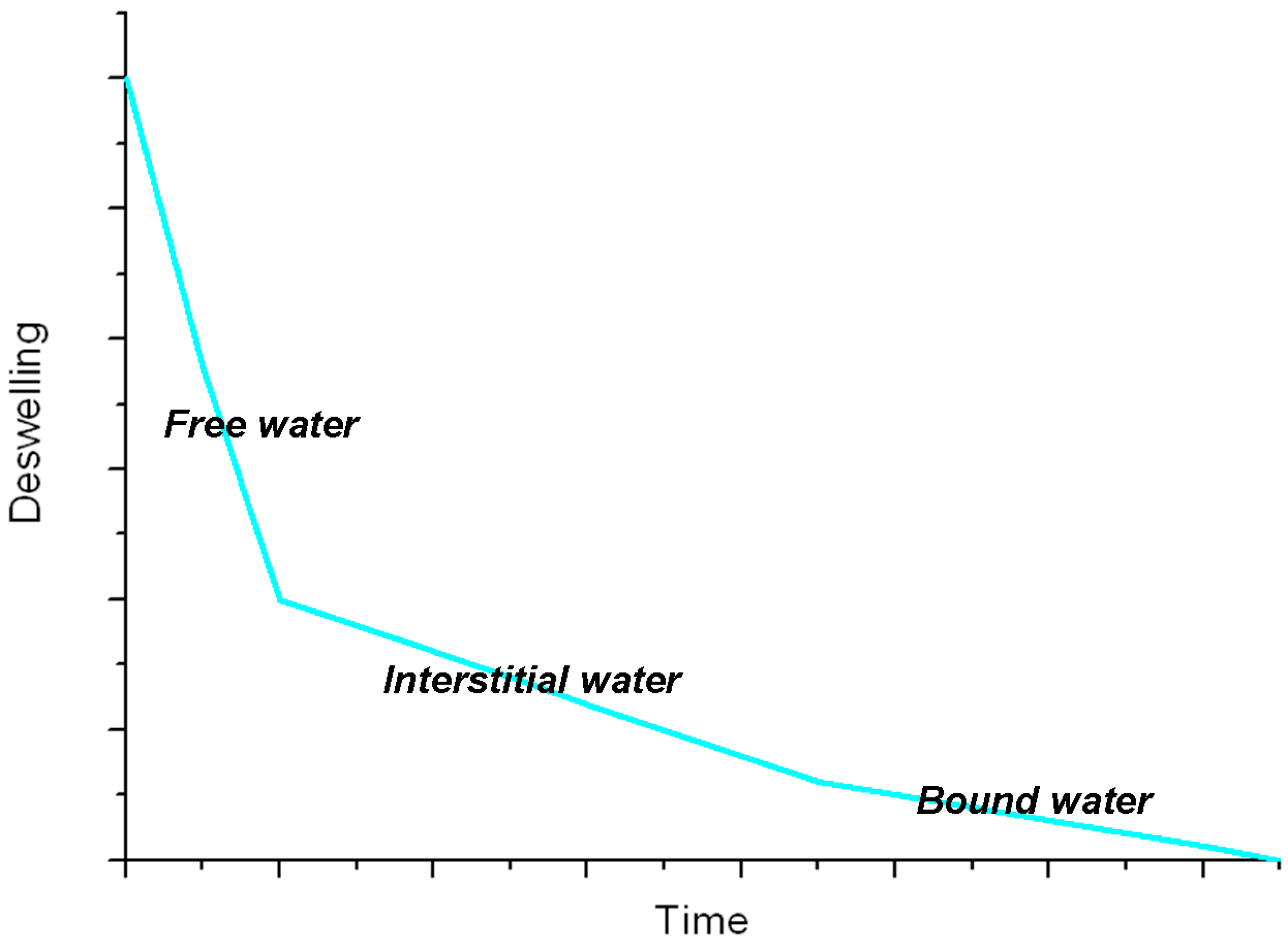
2.2. Swelling Behavior
2.3. Thixotropy
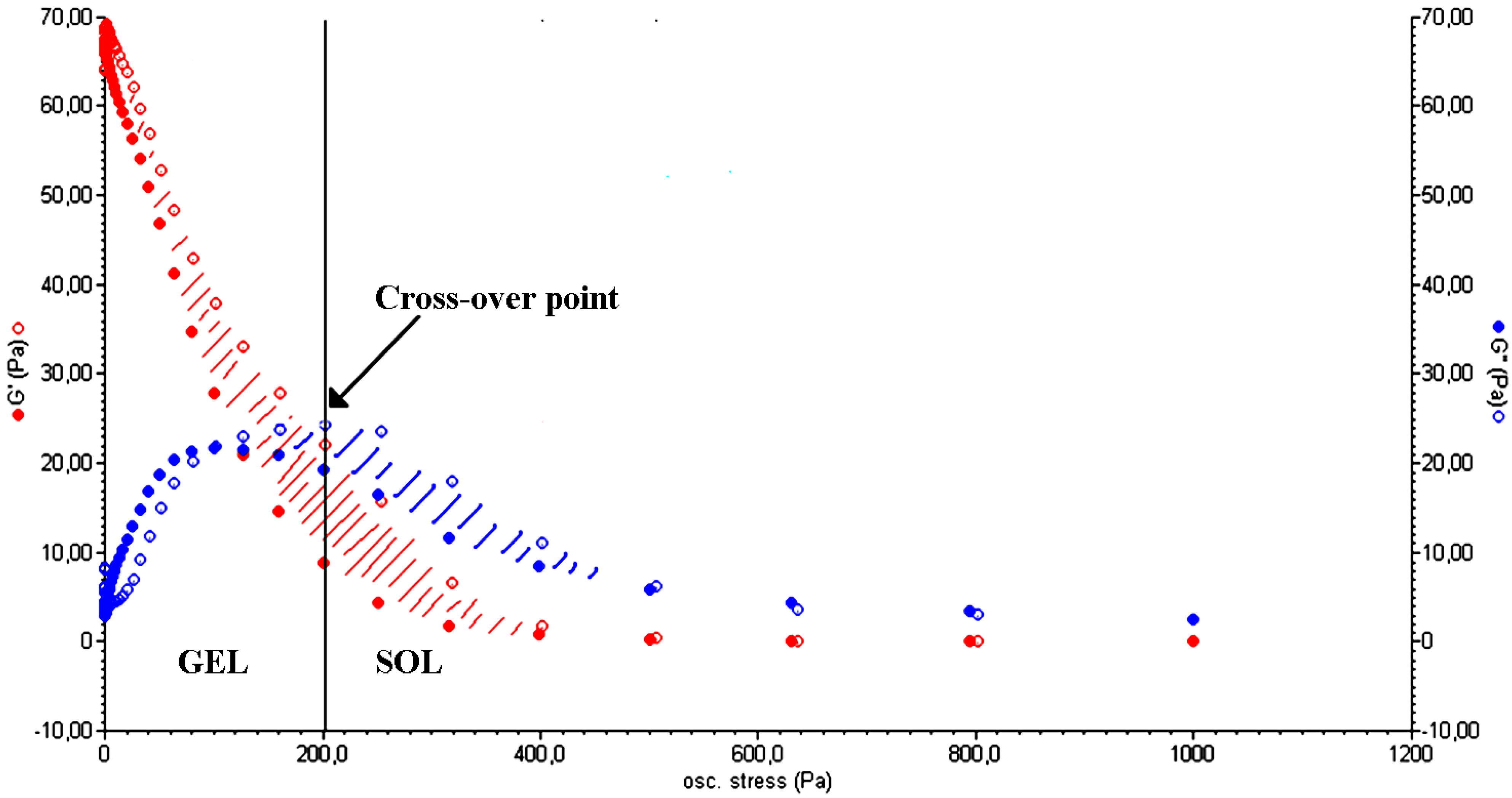
| Hydrogel | State | G' (Pa) | G" (Pa) | ||
|---|---|---|---|---|---|
| Carboxymethylcellulose | Native | 550 | ±30 | 25 | ±1 |
| Squeezed | 240 | ±20 | 20 | ±2 | |
| Hyaluronic acid | Native | 970 | ±25 | 65 | ±10 |
| Squeezed | 340 | ±20 | 45 | ±5 | |
| Chitosan | Native | 4350 | ±650 | 165 | ±45 |
| Squeezed | 3460 | ±150 | 230 | ±15 | |
3. Medical Applications of Injectable Polysaccharide Hydrogels
- regenerative medicine;
- local therapy through drug delivery.
3.1. Minimally Invasive Surgery for Regenerative Medicine

3.2. Interpenetrating Hydrogels as Three-Dimensional Cell Scaffolds

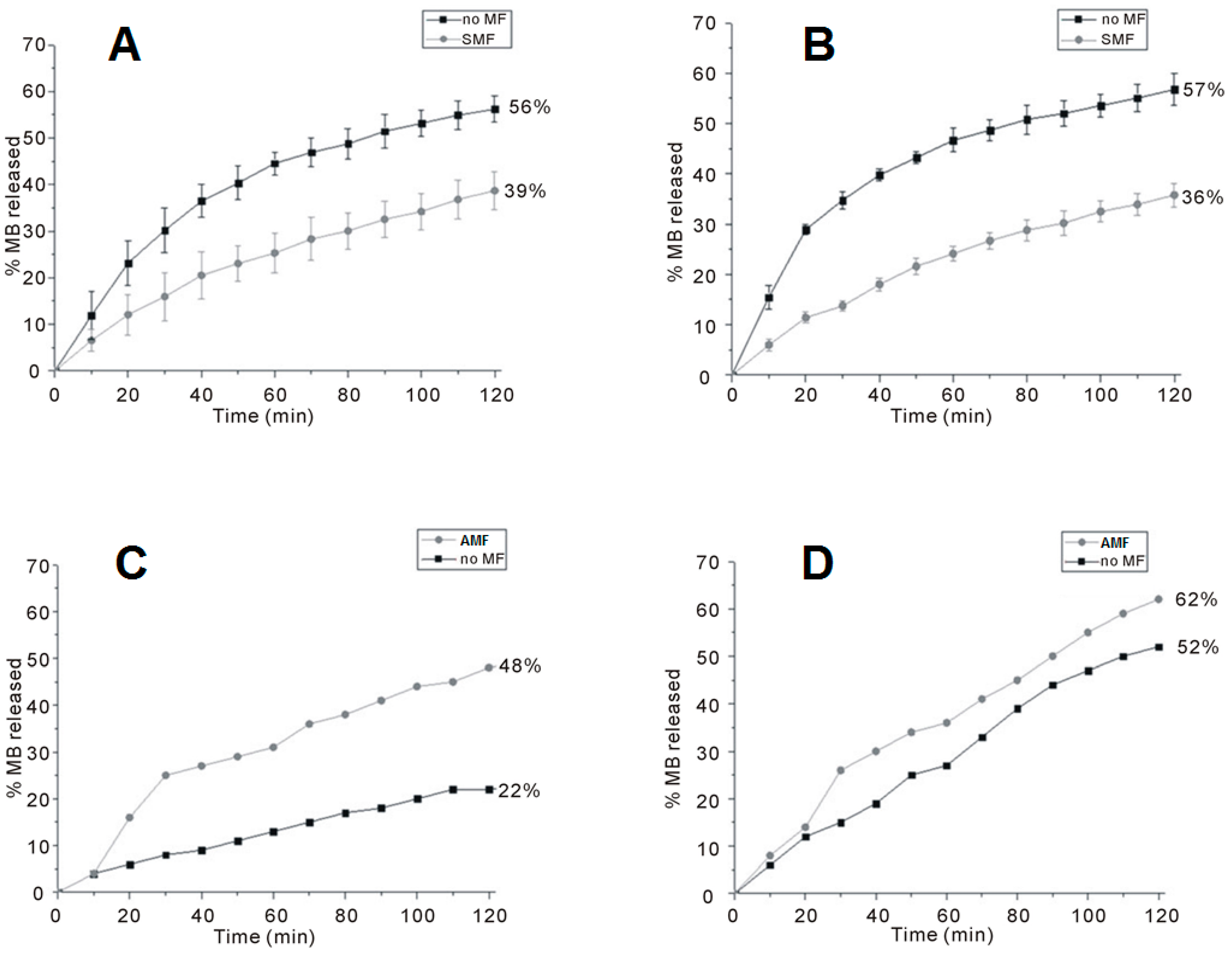
3.3. Drug Delivery for Local Therapy
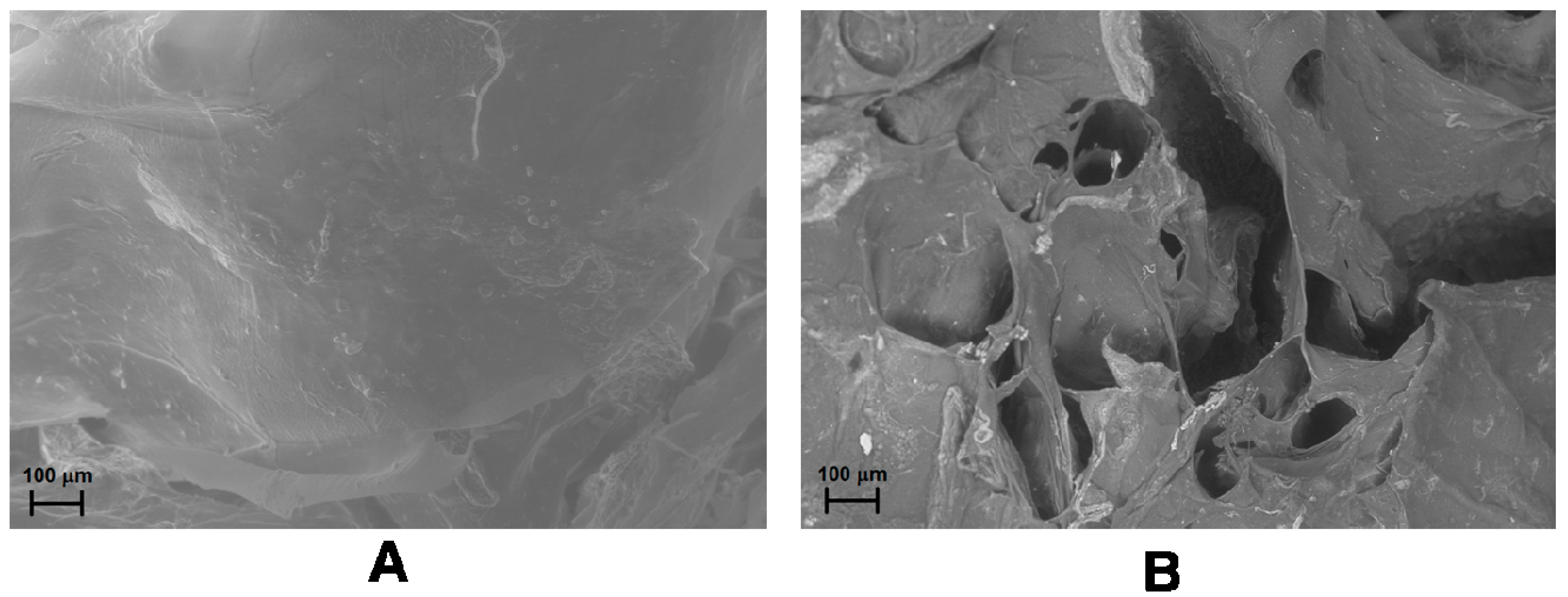
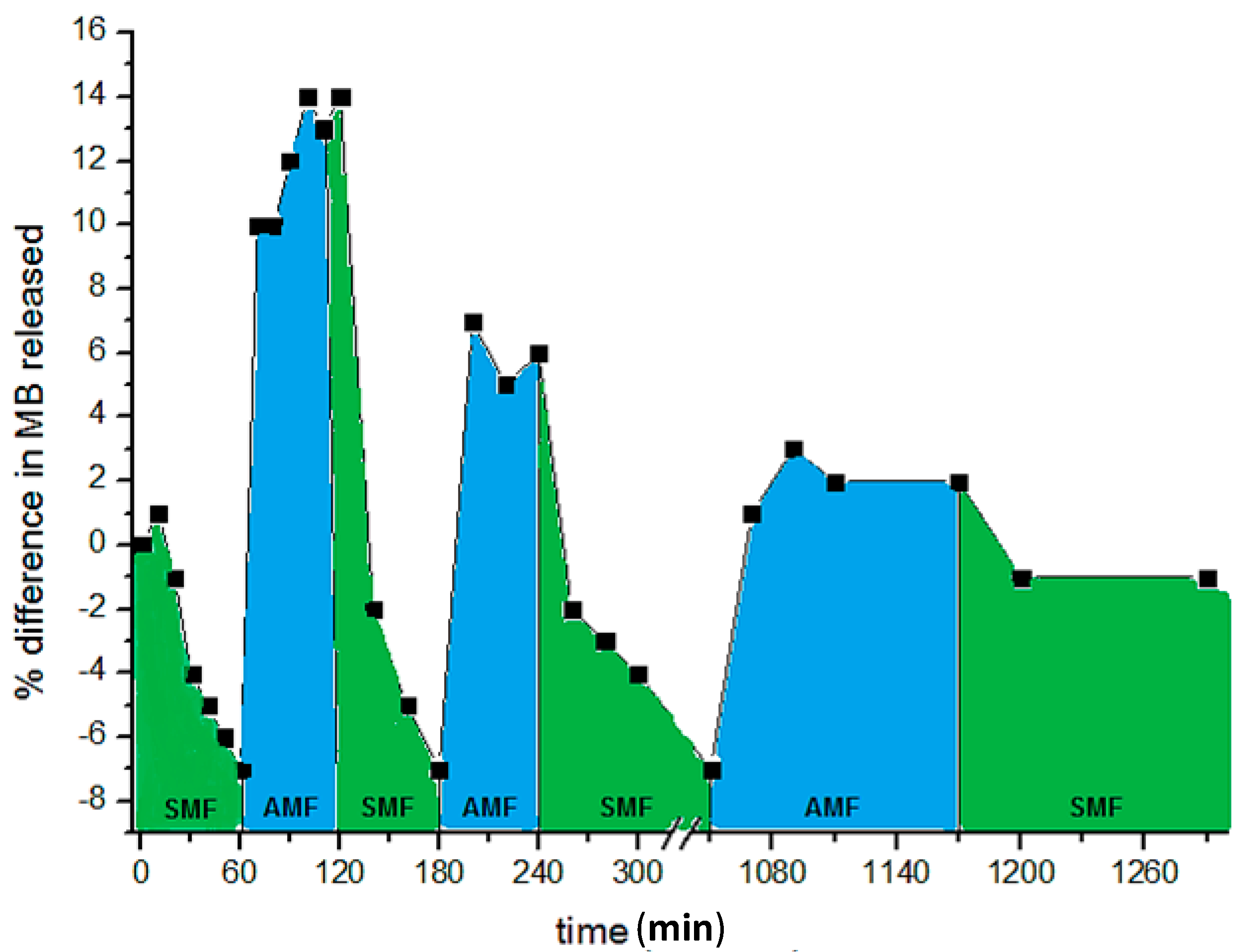
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hoffman, A.S. Hydrogels for biomedical applications. Ann. N. Y. Acad. Sci. 2001, 944, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Slaughter, B.V.; Kanzelberger, M.A. Hydrogels in Polymer Science: A Comprehensive Reference; Matyjaszewski, K., Möller, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2012; Volume 9, pp. 385–395. [Google Scholar]
- Guenet, J.M. Thermoreversible Gelation of Polymers and Biopolymers; Academic Press: New York, NY, USA, 1992. [Google Scholar]
- Percec, V.; Bera, T.K.; Butera, R.J. A new strategy for the preparation of supramolecular neutral hydrogels. Biomacromolecules 2002, 3, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.K.; Ma, P.X. Maintaining dimensions and mechanical properties of ionically crosslinked alginate hydrogel scaffolds in vitro. Biomaterials 2001, 22, 511–521. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Barr-Howell, B.D. Hydrogels in Medicine and Pharmacy; Peppas, N.A., Ed.; CRC Press: Boca Raton, FL, USA, 1986; Chapter 2. [Google Scholar]
- Schacht, E.H. Polymer chemistry and hydrogel systems. J. Phys. 2004, 3, 22–28. [Google Scholar]
- Trabbic-Carlson, K.; Setton, L.A.; Chilkoti, A. Swelling and mechanical behaviors of chemically cross-linked hydrogels of elastin-like polypeptides. Biomacromolecules 2003, 4, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Ratner, B.D.; Hoffman, A.S. Hydrogels for Medical and Related Applications; Andrade, J.D., Ed.; American Chemical Society: New York, NY, USA, 1976; Volume 31. [Google Scholar]
- Omidian, H.; Park, K. Introduction to hydrogels. In Biomedical Applications of Hydrogels Handbook; Ottenbrite, R.M., Ed.; Springer: London, UK, 2010; pp. 1–16. [Google Scholar]
- Philippova, O.E.; Khokhlov, A.R. Polymer gels. In Polymer Science: A Comprehensive Reference; Matyjaszewski, K., Möller, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2012; Volume 1, pp. 339–366. [Google Scholar]
- Liechty, W.B.; Caldorera-Moore, M.; Phillips, M.A.; Schoener, C.; Peppas, N.A. Advanced molecular design of biopolymers for transmucosal and intracellular delivery of chemotherapeutic agents and biological therapeutics. J. Control. Release 2011, 155, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Seok Gil, E.; Hudson, S.M. Stimuli-reponsive polymers and their bioconjugates. Prog. Polym. Sci. 2004, 29, 1173–1222. [Google Scholar] [CrossRef]
- Pasqui, D.; de Cagna, M.; Barbucci, R. Polysaccharide-based hydrogels: The key role of water in affecting mechanical properties. Polymers 2012, 4, 1517–1534. [Google Scholar] [CrossRef]
- Ilić-Stojanović, S.S.; Nikolić, L.; Nikolić, V.; Stanković, M.; Stamenković, J.; Mladenović-Ranisavljević, I.; Petrović, S. Influence of monomer and crosslinker molar ratio on the swelling behaviour of thermosensitive hydrogels. Chem. Ind. Chem. Eng. Q. 2012, 18, 1–9. [Google Scholar] [CrossRef]
- Li, H. Smart Hydrogel Modelling; Springer: London, UK, 2009. [Google Scholar]
- Barbucci, R.; Giardino, R.; de Cagna, M.; Golini, L.; Pasqui, D. Inter-penetrating hydrogels (IPHs) as a new class of injectable polysaccharide hydrogels with thixotropic nature and interesting mechanical and biological properties. Soft Matter 2010, 6, 3524–3532. [Google Scholar] [CrossRef]
- Lyons, J.G.; Geever, L.M.; Nugent, M.J.D.; Kennedy, J.E.; Higginbotham, C.L. Development and characterisation of an agar-polyvinyl alcohol blend hydrogel. J. Mech. Behav. Biomed. Mater. 2009, 5, 485–493. [Google Scholar] [CrossRef]
- Barbucci, R.; Leone, G.; Chiumiento, A.; di Cocco, M.E.; D’Orazio, G.; Gianferri, R.; Delfini, M. Low- and high-resolution nuclear magnetic resonance (NMR) characterisation of hyaluronan-based native and sulfated hydrogels. Carbohydr. Res. 2006, 341, 1848–1858. [Google Scholar] [CrossRef] [PubMed]
- Okano, T. Biorelated Polymers and Gels: Controlled Release and Applications in Biomedical Engineering (Polymers Interfaces and Biomaterials); Academic Press: Waltham, MA, USA, 1998. [Google Scholar]
- Barbucci, R.; Pasqui, D. Hydrogels: Characteristics and properties. In Scaffolds for Tissue Engineering: Biological Design, Materials and Fabrication; Migliaresi, C., Motta, A., Eds.; Pan Stanford Publishing: Singapore, 2013. [Google Scholar]
- Popa, L.; Ghica, M.V.; Moisescu, S. Thixotropy of NaCMC hydrogels with indomethacin. Ovidius Univ. Ann. Med. Sci.-Pharm. 2003, 1, 222–226. [Google Scholar]
- Frendlich, H.; Juliiusberg, F. Thixotropy, influenced by the orientation of anisometric particles in sols and suspensions. Trans. Faraday Soc. 1935, 31, 920–921. [Google Scholar] [CrossRef]
- Sakai, S.; Hirose, K.; Moriyama, K. Control of cellular adhesiveness in an alginate-based hydrogel by varying peroxidase and H2O2 concentrations during gelation. Acta Biomater. 2010, 6, 1446–1452. [Google Scholar] [CrossRef] [PubMed]
- Ker, R.F. Dynamic tensile properties of the plantaris tendon of sheep (Ovis aries). J. Exp. Biol. 1996, 199, 1501–1508. [Google Scholar] [PubMed]
- Trevors, J.T.; Pollack, G. Hypothesis: The origin of life in a hydrogel environment. Prog. Biophys. Mol. Biol. 2005, 89, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Barbucci, R.; Pasqui, D.; Giani, G.; de Cagna, M.; Fini, M.; Giardino, R.; Atrei, A. A novel strategy for engineering ferromagnetic nanoparticles as crosslinkers of polymer chains for potential applications as a targeted drug delivery system. Soft Matter 2011, 7, 5558–5565. [Google Scholar] [CrossRef]
- Li, W.; Sun, B.; Wu, P. Study on hydrogen bonds of carboxymethyl cellulose sodium film with two-dimensional correlation infrared spectroscopy. Carbohydr. Polym. 2009, 78, 454–461. [Google Scholar] [CrossRef]
- Ko, H.F.; Sfeir, C.; Kumta, P.N. Novel synthesis strategies for natural polymer and composite biomaterials as potential scaffolds for tissue engineering. Philos. Trans. A Math. Phys. Eng. Sci. 2010, 368, 1981–1997. [Google Scholar] [CrossRef] [PubMed]
- Vacanti, J.P.; Langer, R. Tissue engineering: The design and fabrication of living replacement devices for surgical reconstruction and transplantation. Lancet 1999, 354, SI32–SI34. [Google Scholar] [CrossRef] [PubMed]
- Hollister, S.J.; Maddox, R.D.; Taboas, J.M. Optimal design and fabrication of scaffolds to mimic tissue properties and satisfy biological constraints. Biomaterials 2002, 23, 4095–4103. [Google Scholar] [CrossRef] [PubMed]
- Leonard, M.; de Boisson, M.R.; Hubert, P.; Dalencon, F.; Dellacherie, E. Hydrophobically modified alginate hydrogels as protein carriers with specific controlled release properties. J. Control. Release 2004, 98, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Dahl, L.B.; Dahl, I.M.; Enstrom-Laurent, A.; Granath, K. Concentration and molecular weight of sodium hyaluronate in synovial fluid from patients with rheumatoid arthritis and other arthropathies. Ann. Rheum. Dis. 1985, 44, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Barbucci, R.; Lamponi, S.; Borzacchiello, A.; Ambrosio, L.; Fini, M.; Torricelli, P.; Giardino, R. Hyaluronic acid hydrogel in the treatment of osteoarthritis. Biomaterials 2002, 23, 4503–4513. [Google Scholar] [CrossRef] [PubMed]
- Leone, G.; Fini, M.; Torricelli, P.; Giardino, R.; Barbucci, R. An amidated carboxymethylcellulose hydrogel for cartilage regeneration. J. Mater. Sci. Mater. Med. 2008, 19, 2873–2880. [Google Scholar] [CrossRef] [PubMed]
- Tautzenberger, A.; Kovtun, A.; Ignatius, A. Nanoparticles and their potential for application in bone. Int. J. Nanomed. 2012, 7, 4545–4557. [Google Scholar] [CrossRef]
- Ruppel, M.E.; Miller, L.M.; Burr, D.B. The effect of the microscopic and nanoscale structure on bone fragility. Osteoporos. Int. 2008, 19, 1251–1265. [Google Scholar] [CrossRef] [PubMed]
- Saiz, E.; Zimmermann, E.A.; Lee, J.S.; Wegst, U.G.; Tomsia, A.P. Perspectives on the role of nanotechnology in bone tissue engineering. Dent. Mater. 2013, 29, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Kunzler, T.P.; Drobek, T.; Schuler, M.; Spencer, N.D. Systematic study of osteoblast and fibroblast response to roughness by means of surface-morphology gradients. Biomaterials 2007, 28, 2175–2182. [Google Scholar] [CrossRef] [PubMed]
- Hunter, K.T.; Ma, T. In vitro evaluation of hydroxyapatite-chitosan-gelatin composite membrane in guided tissue regeneration. J. Biomed. Mater. Res. A 2013, 101, 1016–1025. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.K.; Ferreira, J.C.K.; Luo, T.J.; Geng, H.; Lin, F.C.; Ko, C.C. Direct scaffolding of biomimetic hydroxyapatite-gelatin nanocomposites using aminosilane cross-linker for bone regeneration. J. Mater. Sci. Mater. Med. 2012, 22, 2115–2126. [Google Scholar] [CrossRef]
- Lin, J.; Lai, J.; Lin, W.; Wu, C.; Xu, S. Preparation and biocompatibility of a novel biomimetic osteochondral scaffold: Collagen-chitosan/nano-hydroxyapatite-collagen-polylactic acid. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2012, 26, 1001–1006. [Google Scholar] [PubMed]
- Kweon, H.; Lee, K.G.; Chae, C.H.; Balázsi, C.; Min, S.K.; Kim, J.Y.; Choi, J.Y.; Kim, S.G. Development of nano-hydroxyapatite graft with silk fibroin scaffold as a new bone substitute. J. Oral Maxillofac. Surg. 2011, 69, 1578–1586. [Google Scholar] [CrossRef] [PubMed]
- Ramli, N.A.; Wong, T.W. Sodium carboxymethylcellulose scaffolds and their physicochemical effects on partial thickness wound healing. Int. J. Pharm. 2011, 403, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Okajima, K. Role of molecular characteristics on some physiological properties of cellulose derivatives. In Cellulose: Structural and Functional Aspects; Kennedy, J.F., Phillips, G.O., Williams, P.A., Eds.; Ellis Horwood: Chichester, UK, 1989; pp. 439–446. [Google Scholar]
- Krylova, E.; Ivanov, A.; Orlovski, V.; el-Registan, G.; Barinov, S. Hydroxypatite-polysaccharide granules for drug delivery. J. Mater. Sci. Mater. Med. 2002, 13, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Zakharov, N.A.; Ezhova, A.; Koval, E.M.; Kalinnikov, V.T.; Chalykh, A.E. Hydroxyapatite-carboxymethyl cellulose nanocomposite biomaterial. Inorg. Mater. 2005, 41, 509–515. [Google Scholar] [CrossRef]
- Pasqui, D.; Torricelli, P.; de Cagna, M.; Fini, M.; Barbucci, R. Carboxymethyl cellulose-hydroxyapatite hybrid hydrogel as a composite material for bone tissue engineering applications. J. Biomed. Mater. Res. A 2014, 102, 1568–1579. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Kim, S.S.; Lee, Y.M. Interpenetrating polymer network hydrogels based on poly(ethylene glycol) macromer and chitosan. Carbohydr. Polym. 2000, 41, 197–205. [Google Scholar] [CrossRef]
- Yin, L.; Fei, L.; Cui, F.; Tang, C.; Yin, C. Superporous hydrogels containing poly(acrylic acid-co-acrylamide)/O-carboxymethyl chitosan interpenetrating polymer networks. Biomaterials 2007, 28, 1258–1266. [Google Scholar] [CrossRef] [PubMed]
- Barbucci, R.; Leone, G.; Lamponi, S. Thixotropy property of hydrogels to evaluate the cell growing on the inside of the material bulk (Amber effect). J. Biomed. Mater. Res. B Appl. Biomater. 2006, 76, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Barbucci, R.; Fini, M.; Giavaresi, G.; Torricelli, P.; Giardino, R.; Lamponi, S.; Leone, G. Hyaluronic acid hydrogel added with ibuprofen-lysine for the local treatment of chondral lesions in the knee: In vitro and in vivo investigations. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 75B, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.B.; Kim, S.G.; Hwang, S.G.; Chang, S.; Kang, S.S.; Chun, J.S. Non-steroidal anti-inflammatory drugs inhibit nitric oxide-induced apoptosis and dedifferentiation of articular chondrocytes independent of cyclooxygenase activity. J. Biol. Chem. 2003, 278, 15319–15325. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.; Rachita, C.; Aisen, P.S.; Pasinetti, G.M. Non-steroidal anti-inflammatory drugs protect against chondrocyte apoptotic death. Clin. Exp. Rheumatol. 2001, 19, S7–S11. [Google Scholar] [PubMed]
- Hinton, R.H.; Moody, R.L.; Davis, A.W.; Thomas, S.F. Osteoarthritis: Diagnosis and therapeutic considerations. Am. Fam. Physician 2002, 65, 841–848. [Google Scholar] [PubMed]
- Oddis, C.V. New perspectives on osteoarthritis. Am. J. Med. 1996, 100, 10S–15S. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.D.; Sakata, T.; Halloran, B.P. The impact of skeletal unloading on bone formation. Gravit. Space Biol. Bull. 2003, 16, 45–54. [Google Scholar] [PubMed]
- Giangregorio, L.; Blimki, C.J. Skeletal adaptations to alterations in weight-bearing activity: A comparison of models of disuse osteoporosis. Sports Med. 2002, 32, 459–476. [Google Scholar] [CrossRef] [PubMed]
- Daskalov, I.; Tsoneva, I. Electrochemotherapy of Mycosis fungoides by interferon-α. Bioelectrochemistry 2007, 70, 283–286. [Google Scholar] [CrossRef] [PubMed]
- Zupanic, A.; Ribaric, S.; Miklavcic, D. Increasing the repetition frequency of electric pulse delivery reduces unpleasant sensations that occur in electrochemotherapy. Neoplasma 2007, 54, 246–250. [Google Scholar] [PubMed]
- Marty, M.; Serša, G.; Garbay, J.R.; Gehl, J.; Collins, C.G.; Snoj, M.; Billard, V.; Geertsen, P.F.; Larkin, J.O.; Miklavcic, D.; et al. Electrochemotherapy—An easy, highly effective and safe treatment of cutaneous and subcutaneous metastases: Results of ESOPE (European Standard Operating Procedures of Electrochemotherapy) study. Eur. J. Cancer 2006, 4, 3–13. [Google Scholar] [CrossRef]
- Byrne, C.M.; Thompson, J.F.; Johnston, H.; Hersey, P.; Quinn, M.J.; Michael Hughes, T.; McCarthy, W.H. Treatment of metastatic melanoma using electroporation therapy with bleomycin (electrochemotherapy). Melanoma Res. 2005, 15, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Mir, M.; Orlowski, S.; Poddevin, B.; Paoletti, C. Electrochemotherapy of spontaneous mammary tumours in mice. Eur. J. Cancer 1991, 27, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Mir, M.; Orlowski, S. The basis of electrochemotherapy. Adv. Drug Deliv. Rev. 1999, 35, 107–108. [Google Scholar] [CrossRef] [PubMed]
- Colombo, G.L.; di Matteo, S.; Mir, L.M. Cost-effectiveness analysis of electrochemotherapy with the Cliniporator® vs. other methods for the control and treatment of cutaneous and subcutaneous tumors. Ther. Clin. Risk Manag. 2008, 4, 541–548. [Google Scholar] [PubMed]
- Morris, S.M.; Domon, O.E.; McGarriti, L.J.; Chen, J.J.; Manjanatha, M.G.; Andrews, A.M.; Aidoo, A.; Casciano, D.A. A role for apoptosis in the toxicity and mutagenicity of bleomycin in AHH-1tk+/− human lymphoblastoid cells. Mutat. Res. 1996, 357, 143–165. [Google Scholar] [CrossRef] [PubMed]
- Fantozzi, F.; Arturoni, E.; Barbucci, R. The effects of the electric fields on hydrogels to achieve antitumoral drug release. Bioelectrochemistry 2010, 78, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Šatkauskas, S.; Batiuškaitė, D.; Šalomskaitė-Davalgienė, S.; Venslauskas, M.S. Effectiveness of tumor electrochemotherapy as a function of electric pulse strength and duration. Bioelectrochemistry 2005, 65, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Murali Mohan, Y.; Vimala, K.; Thomas, V.; Varaprasad, K.; Sridhar, B.; Bajpai, S.K.; Mohana Raju, K. Controlling of silver nanoparticles structure by hydrogel networks. J. Colloid Interface Sci. 2010, 342, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Webster, T. Nanotechnology and nanomaterials: Promises for improved tissue regeneration. Nano Today 2009, 4, 66–80. [Google Scholar] [CrossRef]
- Kumar, A.; Jena, P.K.; Behera, S.; Lockey, R.F.; Mohapatra, S.; Mohapatra, S. Multifunctional magnetic nanoparticles for targeted delivery. Nanomedicine 2010, 6, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Jurgons, R.; Seliger, C.; Hilpert, A.; Trahms, L.; Odenbach, S.; Alexiou, C. Drug loaded magnetic nanoparticles for cancer therapy. J. Phys. Condens. Matter 2006, 18, 2893–2902. [Google Scholar] [CrossRef]
- Neuberger, T.; Schopf, B.; Hofmann, H.; Hofmann, M.; Rechenberg, B. Superparamagnetic nanoparticles for biomedical applications: Possibilities and limitations of a new drug delivery system. J. Magn. Magn. Mater. 2005, 293, 483–496. [Google Scholar] [CrossRef]
- Satarkar, S.; Hilt, Z.J. Hydrogel nanocomposites as remote-controlled biomaterials. Acta Biomater. 2008, 4, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Satarkar, S.; Hilt, Z.J. Magnetic hydrogel nanocomposites for remote controlled pulsatile drug release. Control. Release 2008, 130, 246–251. [Google Scholar] [CrossRef]
- Hu, S.H.; Liu, T.Y.; Liu, D.M.; Chen, Y. Controlled pulsatile drug release from a ferrogel by a high frequency magnetic field. Macromolecules 2007, 40, 6786–6788. [Google Scholar] [CrossRef]
- Brulé, S.; Levy, M.; Wilhelm, C.; Letourneur, D.; Gazeau, F.; Ménager, C.; le Visage, C. Doxorubicin release triggered by alginate embedded magnetic nanoheaters: A combined therapy. Adv. Mater. 2011, 23, 787–790. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, K.; Takehisa, T.; Fan, S. Effects of clay content on the properties of nanocomposite hydrogels composed of poly(nisopropylacrylamide) and clay. Macromolecules 2002, 35, 10162–10171. [Google Scholar] [CrossRef]
- Sinha, A.; Guha, A. Biomimetic patterning of polymer hydrogels with hydroxyapatite nanopartcles. Mater. Sci. Eng. C 2009, 29, 1330–1333. [Google Scholar] [CrossRef]
- Liu, T.Y.; Hu, S.H.; Liu, T.Y.; Liu, D.M.; Chen, S.Y. Biomedical nanoparticle carriers with combined thermal and magnetic responses. Langmuir 2006, 22, 5974–5978. [Google Scholar] [CrossRef] [PubMed]
- Hamming, L.M.; Qiao, R.; Messersmith, P.B.; Brinson, L.C. Effects of dispersion and interfacial modification on the macroscale properties of TiO2 polymer matrix nanocomposites. Comps. Sci. Technol. 2009, 69, 1880–1886. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Lee, J.H.; Jang, H.J.; Nah, C.W. Preparing PP/clay nanocomposites using a swelling agent. Comps. B Eng. 2004, 35, 133–138. [Google Scholar] [CrossRef]
- Hackenberg, S.; Friehs, G.; Froelich, K.; Ginzkey, C.; Koehler, C.; Scherzed, A.; Burghartz, M.; Hagen, R.; Kleinsasser, N. Intracellular distribution, geno- and cytotoxic effects of nanosized titanium dioxide particles in the anatase crystal phase on human nasal mucosa cells. Toxicol. Lett. 2010, 195, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.J.; Kim, B.M.; Lee, Y.J.; Chung, H.W. Titanium dioxide nanoparticles trigger p53-mediated damage response in peripheral blood lymphocytes. Environ. Mol. Mutagen. 2010, 49, 399–405. [Google Scholar] [CrossRef]
- Pasqui, D.; Atrei, A.; Giani, G.; de Cagna, M.; Barbucci, R. Metal oxide nanoparticles as cross-linkers in polymeric hybrid hydrogels. Mater. Lett. 2011, 65, 392–395. [Google Scholar] [CrossRef]
- Giani, G.; Fedi, S.; Barbucci, R. Hybrid magnetic hydrogel: A potential system for controlled drug delivery by means of alternating magnetic fields. Polymers 2012, 4, 1157–1169. [Google Scholar] [CrossRef]
- Barbucci, R.; Giani, G.; Fedi, S.; Bottari, S.; Casolaro, M. Biohydrogels with magnetic nanoparticles as cross-linkers: Characteristics and potential use for controlled antitumor drug-delivery. Acta Biomater. 2012, 8, 4244–4252. [Google Scholar] [CrossRef] [PubMed]
- Uva, M.; Pasqui, D.; Mencuccini, L.; Fedi, S.; Barbucci, R. Influence of alternating and static magnetic fields on drug release from hybrid hydrogels containing magnetic nanoparticles. J. Biomater. Nanobiotechnol. 2014, 5, 116–127. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camponeschi, F.; Atrei, A.; Rocchigiani, G.; Mencuccini, L.; Uva, M.; Barbucci, R. New Formulations of Polysaccharide-Based Hydrogels for Drug Release and Tissue Engineering. Gels 2015, 1, 3-23. https://doi.org/10.3390/gels1010003
Camponeschi F, Atrei A, Rocchigiani G, Mencuccini L, Uva M, Barbucci R. New Formulations of Polysaccharide-Based Hydrogels for Drug Release and Tissue Engineering. Gels. 2015; 1(1):3-23. https://doi.org/10.3390/gels1010003
Chicago/Turabian StyleCamponeschi, Francesca, Andrea Atrei, Giulia Rocchigiani, Lorenzo Mencuccini, Marianna Uva, and Rolando Barbucci. 2015. "New Formulations of Polysaccharide-Based Hydrogels for Drug Release and Tissue Engineering" Gels 1, no. 1: 3-23. https://doi.org/10.3390/gels1010003
APA StyleCamponeschi, F., Atrei, A., Rocchigiani, G., Mencuccini, L., Uva, M., & Barbucci, R. (2015). New Formulations of Polysaccharide-Based Hydrogels for Drug Release and Tissue Engineering. Gels, 1(1), 3-23. https://doi.org/10.3390/gels1010003






