The Use of Thioflavin T for the Estimation and Measurement of the Plasma Membrane Electric Potential Difference in Different Yeast Strains
Abstract
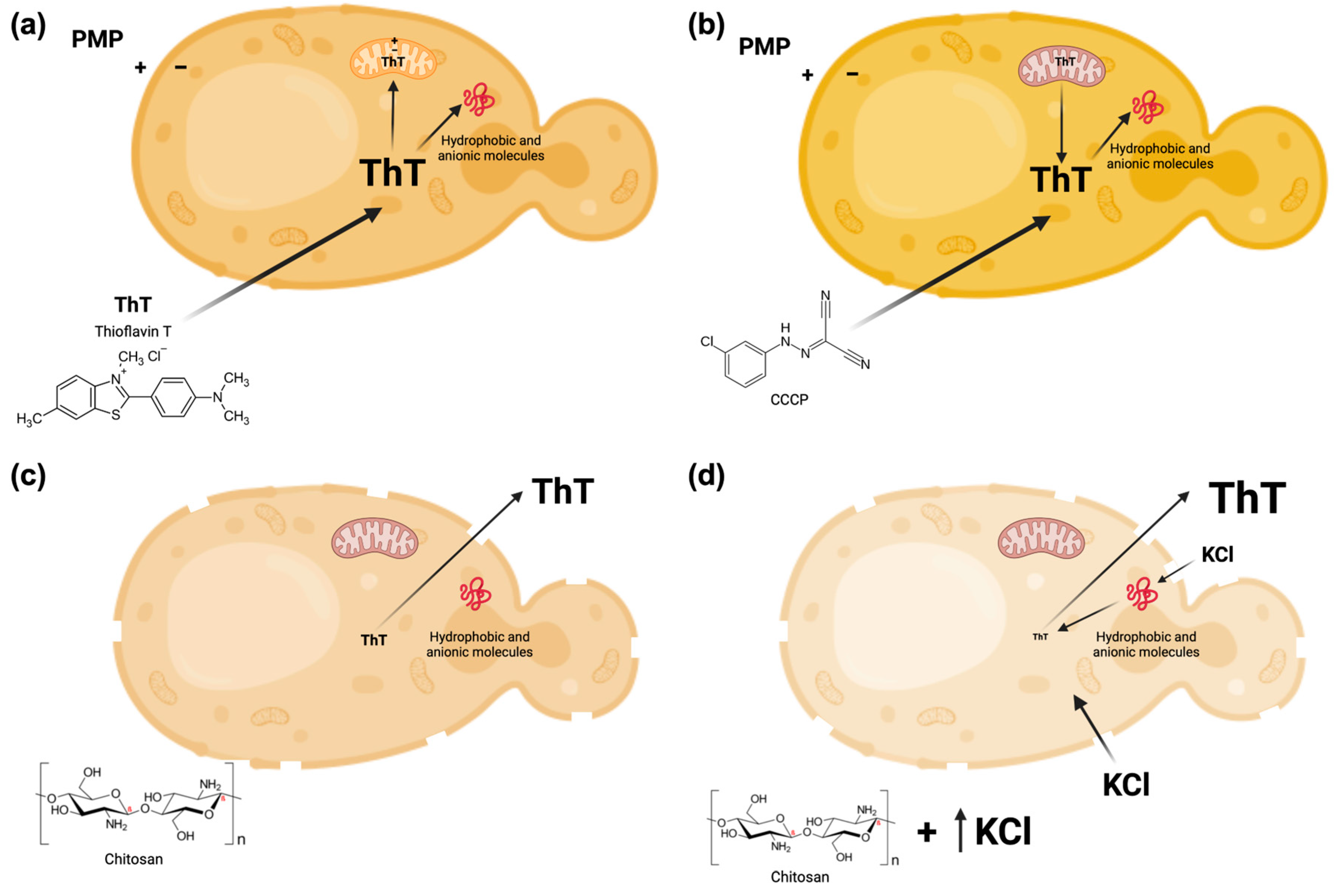
:1. Introduction
2. Materials and Methods
2.1. Strains and Growth Conditions
2.2. Membrane Potential Estimations, Fluorescence of ThT
2.3. Fluorescence Measured with a Multi-Well Plate Reader
2.4. Microscopy
2.5. Membrane Potential Measurements by the Accumulation of ThT
- Δψ is the plasma membrane potential difference, measured in volts (V);
- R is the universal gas constant, (8.314 J·K−1·mol−1);
- T is the temperature in Kelvin degrees (30 °C = 303 K);
- z is the number of elementary charges of thioflavin T (ThT) (=1);
- F is the Faraday constant, equal to 96,485 J·V−1·mol−1;
- [int] is the intracellular concentration of ThT;
- [ext] is the extracellular concentration of ThT;
- 2.3026 RT/F = 60.12 mV (considering our experiments at 30 °C).
2.6. Flow Cytometry
2.7. K+ Uptake
2.8. Oxygen Consumption
2.9. Statistical Analysis
3. Results
3.1. Estimation of the PMP by Changes in the Fluorescence of ThT with Different Yeast Strains
3.2. Results Obtained by Fluorescence in a Multi-Well Plate Reader
3.3. Fluorescence Images of the Cells
3.4. Values of PMP Obtained from the Accumulation of ThT
3.5. ThT Fluorescence as a Monitor of PMP, Can also Be Measured by Flow Cytometry
3.6. K+ Uptake and Oxygen Consumption
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peña, A.; Puyou, E.; Cinco, G.; Tuena, R.I.; Gómez-Puyou, A.; Tuena, M. Effect of the pH of the incubation medium on glycolysis and respiration in Saccharomyces cerevisiae. Arch. Biochem. Biophys. 1972, 153, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Cyert, M.S.; Philpott, C.C. Regulation of cation balance in Saccharomyces cerevisiae. Genetics 2013, 193, 677–713. [Google Scholar] [CrossRef] [PubMed]
- Goffeau, A.; Amory, A.; Villalobo, A.; Dufour, J.P. The H+-ATPase of the yeast plasma membrane. Ann. N. Y. Acad. Sci. 1982, 402, 91–98. [Google Scholar] [CrossRef]
- Zhao, P.; Zhao, C.; Chen, D.; Yun, C.; Li, H.; Bai, L. Structure and activation mechanism of the hexameric plasma membrane H+-ATPase. Nat. Commun. 2021, 12, 6439. [Google Scholar] [CrossRef]
- Consortium, T.U. UniProt: The universal protein knowledgebase in 2023. Nucleic Acids Res. 2022, 51, D523–D531. [Google Scholar] [CrossRef]
- Varadi, M.; Anyango, S.; Deshpande, M.; Nair, S.; Natassia, C.; Yordanova, G.; Yuan, D.; Stroe, O.; Wood, G.; Laydon, A.; et al. AlphaFold protein structure database: Massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. 2022, 50, D439–D444. [Google Scholar] [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Slayman, C.L.; Long, W.S.; Lu, C.Y.-H. The relationship between ATP and an electrogenic pump in the plasma membrane of Neurospora crassa. J. Membr. Biol. 1973, 14, 305–338. [Google Scholar] [CrossRef]
- Goffeau, A.; Slayman, C.W. The proton-translocating ATPase of the fungal plasma membrane. Biochim. Biophys. Acta-Bioenerg. 1981, 639, 197–223. [Google Scholar] [CrossRef]
- Sze, H. Proton-pumping adenosine triphosphatase in membrane vesicles of Tobacco callus: Sensitivity to vanadate and K+. Biochim. Biophys. Acta-Biomembr. 1983, 732, 586–594. [Google Scholar] [CrossRef]
- Peña, A. Studies on the mechanism of K+ transport in yeast. Arch. Biochem. Biophys. 1975, 167, 397–409. [Google Scholar] [CrossRef] [PubMed]
- Borst-Pauwels, G.W.F.H. Ion transport in yeast. Biochim. Biophys. Acta-Biomembr. 1981, 650, 88–127. [Google Scholar] [CrossRef] [PubMed]
- Vacata, V.; Kotyk, A.; Sigler, K. Membrane potential in yeast cells measured by direct and indirect methods. Biochim. Biophys. Acta-Biomembr. 1981, 643, 265–268. [Google Scholar] [CrossRef]
- de la Peña, P.; Barros, F.; Gascón, S.; Ramos, S.; Lazo, P.S. The electrochemical proton gradient of Saccharomyces. The role of potassium. Eur. J. Biochem. 1982, 123, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Peña, A.; Ramírez, G. Interaction of ethidium bromide with the transport system for monovalent cations in yeast. J. Membr. Biol. 1975, 22, 369–384. [Google Scholar] [CrossRef] [PubMed]
- Peña, A.; Uribe, S.; Pardo, J.P.; Borbolla, M. The use of a cyanine dye in measuring membrane potential in yeast. Arch. Biochem. Biophys. 1984, 231, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Peña, A.; Sánchez, N.S.; Calahorra, M. Estimation of the electric plasma membrane potential difference in yeast with fluorescent dyes: Comparative study of methods. J. Bioenerg. Biomembr. 2010, 42, 419–432. [Google Scholar] [CrossRef]
- Plášek, J.; Gášková, D.; Lichtenberg-Fraté, H.; Ludwig, J.; Höfer, M. Monitoring of real changes of plasma membrane potential by DiS-C3(3) fluorescence in yeast cell suspensions. J. Bioenerg. Biomembr. 2012, 44, 559–569. [Google Scholar] [CrossRef]
- Plášek, J.; Gášková, D. Complementary methods of processing DiS-C3(3) fluorescence spectra used for monitoring the plasma membrane potential of yeast: Their pros and cons. J. Fluoresc. 2014, 24, 541–547. [Google Scholar] [CrossRef]
- Peña, A.; Sánchez, N.S.; Calahorra, M. The plasma membrane electric potential in yeast: Probes, results, problems, and solutions: A new application of an old dye? In Old Yeasts—New Questions; Lucas, C., Pais, C., Eds.; IntechOpen: Rijeka, Croatia, 2017. [Google Scholar] [CrossRef]
- Xue, C.; Lin, T.Y.; Chang, D.; Guo, Z. Thioflavin T as an amyloid dye: Fibril quantification, optimal concentration and effect on aggregation. R. Soc. Open Sci. 2017, 4, 160696. [Google Scholar] [CrossRef]
- Calahorra, M.; Sánchez, N.S.; Peña, A. Retraction Note to: Acridine yellow. a novel use to estimate and measure the plasma membrane potential in Saccharomyces cerevisiae. J. Bioenerg. Biomembr. 2019, 51, 313. [Google Scholar] [CrossRef] [PubMed]
- Amdursky, N.; Erez, Y.; Huppert, D. Molecular rotors: What lies behind the high sensitivity of the Thioflavin T fluorescent marker. Acc. Chem. Res. 2012, 45, 1548–1557. [Google Scholar] [CrossRef] [PubMed]
- Voropai, E.S.; Samtsov, M.P.; Kaplevskii, K.N.; Maskevich, A.A.; Stepuro, V.I.; Povarova, O.I.; Kuznetsova, I.M.; Turoverov, K.K.; Fink, A.L.; Uverskii, V.N. Spectral properties of Thioflavin T and its complexes with amyloid fibrils. J. Appl. Spectrosc. 2003, 70, 868–874. [Google Scholar] [CrossRef]
- Stsiapura, V.I.; Maskevich, A.A.; Kuzmitsky, V.A.; Turoverov, K.K.; Kuznetsova, I.M. Computational study of Thioflavin T torsional relaxation in the excited state. J. Phys. Chem. A. 2007, 111, 4829–4835. [Google Scholar] [CrossRef]
- Khurana, R.; Coleman, C.; Ionescu-Zanetti, C.; Carter, S.A.; Krishna, V.; Grover, R.K.; Roy, R.; Singh, S. Mechanism of Thioflavin T binding to amyloid fibrils. J. Struct. Biol. 2005, 151, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Biancalana, M.; Makabe, K.; Koide, A.; Koide, S. Molecular mechanism of Thioflavin T binding to the surface of β-rich peptide self-assemblies. J. Mol. Biol. 2009, 385, 1052–1063. [Google Scholar] [CrossRef] [PubMed]
- Biancardi, A.; Biver, T.; Burgalassi, A.; Mattonai, M.; Secco, F.; Venturini, M. Mechanistic aspects of Thioflavin T self-aggregation and DNA binding: Evidence for dimer attack on DNA grooves. Phys. Chem. 2014, 16, 20061–20072. [Google Scholar] [CrossRef]
- Xu, S.; Li, Q.; Xiang, J.; Yang, Q.; Sun, H.; Guan, A.; Wang, L.; Liu, Y.; Yu, L.; Shi, Y.; et al. Thioflavin T as an efficient fluorescence sensor for selective recognition of RNA G-quadruplexes. Sci. Rep. 2016, 6, 24793. [Google Scholar] [CrossRef]
- Peña, A.; Sánchez, N.S.; Calahorra, M. Estimations and actual measurements of the plasma membrane electric potential difference in yeast. In Membrane Potential: An Overview; Marušić, M., Ed.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2020; Volume 1, pp. 81–104. ISBN 978-1-53616-743-6. [Google Scholar]
- Padilla-Garfias, F.; Ríos-Cifuentes, L.; Sánchez, N.S.; Calahorra, M.; Peña, A. Study of the mechanism of ε-poly-L-lysine as an antifungal on Candida albicans and Saccharomyces cerevisiae. Biochim. Biophys. Acta-Gen. Subj. 2022, 1866, 130197. [Google Scholar] [CrossRef]
- García-Navarrete, M.; Avdovic, M.; Pérez-Garcia, S.; Ruiz Sanchis, D.; Wabnik, K. Macroscopic control of cell electrophysiology through ion channel expression. Elife. 2022, 11, e78075. [Google Scholar] [CrossRef]
- Estrada-Ávila, A.K.; González-Hernández, J.C.; Calahorra, M.; Sánchez, N.S.; Peña, A. Xylose and yeasts: A story beyond xylitol production. Biochim. Biophys. Acta-Gen. Subj. 2022, 1866, 130154. [Google Scholar] [CrossRef] [PubMed]
- Masuda, C.A.; Ramírez, J.; Peña, A.; Montero-Lomelí, M. Regulation of monovalent ion homeostasis and pH by the Ser-Thr protein phosphatase SIT4 in Saccharomyces cerevisiae. J. Biol. Chem. 2000, 275, 30957–30961. [Google Scholar] [CrossRef] [PubMed]
- Peña, A.; Calahorra, M.; Michel, B.; Ramírez, J.; Sánchez, N.S. Effects of amiodarone on K+, internal pH and Ca2+ homeostasis in Saccharomyces cerevisiae. FEMS Yeast Res. 2009, 9, 832–848. [Google Scholar] [CrossRef] [PubMed]
- Michel, B.; Lozano, C.; Rodríguez, M.; Coria, R.; Ramírez, J.; Peña, A. The yeast potassium transporter TRK2 is able to substitute for TRK1 in its biological function under low K+ and low pH conditions. Yeast 2006, 23, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Wallis, J.W.; Chrebet, G.; Brodsky, G.; Rolfe, M.; Rothstein, R. A hyper-recombination mutation in S. cerevisiae identifies a novel eukaryotic topoisomerase. Cell 1989, 58, 409–419. [Google Scholar] [CrossRef]
- Maresova, L.; Urbankova, E.; Gaskova, D.; Sychrova, H. Measurements of plasma membrane potential changes in Saccharomyces cerevisiae cells reveal the importance of the Tok1 channel in membrane potential maintenance. FEMS Yeast Res. 2006, 6, 1039–1046. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An Open-Source Platform for Biological-Image Analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Peña, A.; Sánchez, N.S.; Calahorra, M. Effects of chitosan on Candida albicans: Conditions for its antifungal activity. Biomed. Res. Int. 2013, 2013, 527549. [Google Scholar] [CrossRef]
- Sánchez, N.S.; Arreguín, R.; Calahorra, M.; Peña, A. Effects of salts on aerobic metabolism of Debaryomyces hansenii. FEMS Yeast Res. 2008, 8, 1303–1312. [Google Scholar] [CrossRef]
- Stratford, J.P.; Edwards, C.L.A.; Ghanshyam, M.J.; Malyshev, D.; Delise, M.A.; Hayashi, Y.; Asally, M. Electrically induced bacterial membrane-potential dynamics correspond to cellular proliferation capacity. Proc. Natl. Acad. Sci. USA 2019, 116, 9552–9557. [Google Scholar] [CrossRef]
- Nicholls, D.G. Fluorescence measurement of mitochondrial membrane potential changes in cultured cells. In Mitochondrial Bioenergetics; Palmeira, C.M., Moreno, A.J., Eds.; Springer: New York, NY, USA, 2012; Volume 810, pp. 119–133. [Google Scholar] [CrossRef]
- Skates, E.; Delattre, H.; Schofield, Z.; Asally, M.; Soyer, O.S. Thioflavin T indicates membrane potential in mammalian cells and can affect it in a blue light dependent manner. bioRxiv 2021. [Google Scholar] [CrossRef]
- Farkas, D.L.; Wei, M.D.; Febbroriello, P.; Carson, J.H.; Loew, L.M. Simultaneous imaging of cell and mitochondrial membrane potentials. Biophys. J. 1989, 56, 1053–1069. [Google Scholar] [CrossRef] [PubMed]
- Darghal, N.; Garnier-Suillerot, A.; Salerno, M. Mechanism of Thioflavin T accumulation inside cells overexpressing P-glycoprotein or multidrug resistance-associated protein: Role of lipophilicity and positive charge. Biochem. Biophys. Res. Commun. 2006, 343, 623–629. [Google Scholar] [CrossRef]
- Latonen, L. Phase-to-phase with nucleoli—Stress responses, protein aggregation and novel roles of RNA. Front. Cell. Neurosci. 2019, 13, 151. [Google Scholar] [CrossRef]
- Madrid, R.; Gómez, M.J.; Ramos, J.; Rodríguez-Navarro, A. Ectopic potassium uptake in trk1 trk2 mutants of Saccharomyces cerevisiae correlates with a highly hyperpolarized membrane potential. J. Biol. Chem. 1998, 273, 14838–14844. [Google Scholar] [CrossRef]
- Arad, E.; Green, H.; Jelinek, R.; Rapaport, H. Revisiting Thioflavin T (ThT) fluorescence as a marker of protein fibrillation—The prominent role of electrostatic interactions. J. Colloid Interface Sci. 2020, 573, 87–95. [Google Scholar] [CrossRef]
- Bertl, A.; Ramos, J.; Ludwig, J.; Lichtenberg-Fraté, H.; Reid, J.; Bihler, H.; Calero, F.; Martínez, P.; Ljungdahl, P.O. Characterization of potassium transport in wild-type and isogenic yeast strains carrying all combinations of trk1, trk2 and tok1 null mutations. Mol. Microbiol. 2003, 47, 767–780. [Google Scholar] [CrossRef]
- Bihler, H.; Slayman, C.L.; Bertl, A. NSC1: A novel high-current inward rectifier for cations in the plasma membrane of Saccharomyces cerevisiae. FEBS Lett. 1998, 432, 59–64. [Google Scholar] [CrossRef]
- Bihler, H.; Slayman, C.L.; Bertl, A. Low-affinity potassium uptake by Saccharomyces cerevisiae is mediated by NSC1, a calcium-blocked non-specific cation channel. Biochim. Biophys. Acta Biomembr. 2002, 1558, 109–118. [Google Scholar] [CrossRef]
- Ko, C.H.; Buckley, A.M.; Gaber, R.F. TRK2 is required for low affinity K+ transport in Saccharomyces cerevisiae. Genetics 1990, 125, 305–312. [Google Scholar] [CrossRef]
- Ramos, J.; Alijo, R.; Haro, R.; Rodriguez-Navarro, A. TRK2 is not a low-affinity potassium transporter in Saccharomyces cerevisiae. J. Bacteriol. 1994, 176, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Vidal, M.; Buckley, A.M.; Yohn, C.; Hoeppner, D.J.; Gaber, R.F. Identification of essential nucleotides in an upstream repressing sequence of Saccharomyces cerevisiae by selection for increased expression of TRK2. Proc. Natl. Acad. Sci. USA 1995, 92, 2370–2374. [Google Scholar] [CrossRef] [PubMed]
- Ramos, J.; Contreras, P.; Rodríguez-Navarro, A. A potassium transport mutant of Saccharomyces cerevisiae. Arch. Microbiol. 1985, 143, 88–93. [Google Scholar] [CrossRef]
- Gaber, R.F.; Styles, C.A.; Fink, G.R. TRK1 encodes a plasma membrane protein required for high-affinity potassium transport in Saccharomyces cerevisiae. Mol. Cell Biol. 1988, 8, 2848–2859. [Google Scholar] [CrossRef]
- Ketchum, K.A.; Joiner, W.J.; Sellers, A.J.; Kaczmarek, L.K.; Goldstein, S.A.N. A new family of outwardly rectifying potassium channel proteins with two pore domains in tandem. Nature 1995, 376, 690–695. [Google Scholar] [CrossRef]
- Zhou, X.-L.; Vaillant, B.; Loukin, S.H.; Kung, C.; Saimi, Y. YKC1 encodes the depolarization-activated K+ channel in the plasma membrane of yeast. FEBS Lett. 1995, 373, 170–176. [Google Scholar] [CrossRef]
- Reid, J.D.; Lukas, W.; Shafaatian, R.; Bertl, A.; Scheurmann-Kettner, C.; Guy, H.R.; North, R.A. The S.cerevisiae outwardly-rectifying potassium channel (DUK1) identifies a new family of channels with duplicated pore domains. Recept. Channels. 1996, 4, 51–62. [Google Scholar]
- Breuer, U.; Harms, H. Debaryomyces hansenii—An extremophilic yeast with biotechnological potential. Yeast 2006, 23, 415–437. [Google Scholar] [CrossRef]
- Ramírez-Castrillón, M.; Jaramillo-Garcia, V.P.; Rosa, P.D.; Landell, M.F.; Vu, D.; Fabricio, M.F.; Ayub, M.A.Z.; Robert, V.; Henriques, J.A.P.; Valente, P. The oleaginous yeast Meyerozyma guilliermondii BI281A as a new potential biodiesel feedstock: Selection and lipid production optimization. Front. Microbiol. 2017, 8, 1776. [Google Scholar] [CrossRef]
- Rodríguez-Navarro, A. Potassium transport in fungi and plants. Biochim. Biophys. Acta Biomembr. 2000, 1469, 1–30. [Google Scholar] [CrossRef]
- McKinnon, K.M. Flow cytometry: An overview. Curr. Protoc. Immunol. 2018, 120, 5.1.1–5.1.11. [Google Scholar] [CrossRef] [PubMed]
- Nobre, A.; Lucas, C.; Leão, C. Transport and utilization of hexoses and pentoses in the halotolerant yeast Debaryomyces hansenii. Appl. Environ. Microbiol. 1999, 65, 3594–3598. [Google Scholar] [CrossRef]
- Papon, N.; Savini, V.; Lanoue, A.; Simkin, A.J.; Crèche, J.; Giglioli-Guivarc’h, N.; Clastre, M.; Courdavault, V.; Sibirny, A.A. Candida guilliermondii: Biotechnological applications, perspectives for biological control, emerging clinical importance and recent advances in genetics. Curr. Genet. 2013, 59, 73–90. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Ge, M.; Zhang, Z.; Wang, W.; Wu, G. Spectroscopic studies on the interaction between riboflavin and albumins. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2006, 65, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Zanetti-Polzi, L.; Aschi, M.; Daidone, I.; Amadei, A. Theoretical modeling of the absorption spectrum of aqueous riboflavin. Chem. Phys. Lett. 2017, 669, 119–124. [Google Scholar] [CrossRef]
- Mannazzu, I.; Landolfo, S.; da Silva, T.L.; Buzzini, P. Red yeasts and carotenoid production: Outlining a future for non-conventional yeasts of biotechnological interest. World J. Microbiol. Biotechnol. 2015, 31, 1665–1673. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Ghoshal, G. Characterization and cytotoxic activity of pigment extracted from Rhodotorula mucilaginosa to assess its potential as bio-functional additive in confectionary products. J. Food Sci. Technol. 2021, 58, 2688–2698. [Google Scholar] [CrossRef] [PubMed]








| Strain | Genotype | Source |
|---|---|---|
| Candida albicans | ATCC 10231 | |
| Debaryomyces hansenii Y7426 | US Department of Agriculture, Peoria, IL, | |
| Meyerozyma guilliermondii | [33] | |
| Rhodotorula mucilaginosa | ATCC 66034 | |
| Saccharomyces cerevisiae Azteca | Commercial strain | |
| Saccharomyces cerevisiae FY833 | MATa (his3Δ200 leu2Δ1 lys2Δ202 trp1Δ63 ura3-52) | [34] |
| trk1Δ | MATa (his3Δ200 leu2Δ1 lys2Δ202 trp1Δ63 ura3-52) trk1Δ::kanMX | [35] |
| trk2Δ | MATa (his3Δ200 leu2Δ1 lys2Δ202 trp1Δ63 ura3-52) trk2Δ::HIS3 | [35] |
| trk1Δ trk2Δ | MATa (his3Δ200 leu2Δ1 lys2Δ202 trp1Δ63 ura3-52) trk1Δ::TRP1-trk2Δ::kanMX | [36] |
| Saccharomyces cerevisiae W303-1A | MATa (ade2-1 can1-100 his3-11,15 leu2-3,112 trp1-1 ura3-1 mal10) | [37] |
| TOW (tok1Δ) | MATa (ade2-1 can1-100 his3-11,15 leu2-3,112 trp1-1 ura3-1 mal 10) tok1Δ::kanMX | [38] |
| % Dry Weight | Internal Cell Volume mL/g (w. w.) | Vacuole/Cell Volume Ratio | Total Vacuole Water/g Cells (mL) | Cytoplasm Volume/g Cells (mL) | Cytoplasm Volume/150 mg Cells (mL) | |
|---|---|---|---|---|---|---|
| S. cerevisiae Azteca | 22.2 | 0.430 | 0.117 | 0.050 | 0.380 | 0.0569 |
| S. cerevisiae FY833 | 18.0 | 0.424 | 0.276 | 0.117 | 0.307 | 0.0460 |
| S. cerevisiae W303-1A | 22.4 | 0.440 | 0.252 | 0.111 | 0.329 | 0.0494 |
| C. albicans | 27.0 | 0.410 | 0.150 | 0.061 | 0.349 | 0.0523 |
| D. hansenii | 21.0 | 0.440 | 0.091 | 0.040 | 0.400 | 0.0600 |
| M. guilliermondii | 28.2 | 0.390 | 0.099 | 0.039 | 0.351 | 0.0527 |
| R. mucilaginosa | 14.5 | 0.420 | 0.071 | 0.030 | 0.390 | 0.0585 |
| trk1Δ | 19.9 | 0.460 | 0.124 | 0.057 | 0.403 | 0.0605 |
| trk2Δ | 18.3 | 0.478 | 0.265 | 0.127 | 0.352 | 0.0528 |
| trk1Δ trk2Δ | 18.4 | 0.463 | 0.142 | 0.066 | 0.397 | 0.0595 |
| TOW | 15.4 | 0.529 | 0.264 | 0.140 | 0.390 | 0.0585 |
| Non-Corrected | Corrected | |
|---|---|---|
| Δψ (mV) ± SD | Δψ (mV) ± SD | |
| S.c. Azteca | ||
| Glucose | −201.6 ± 3.4 | −192.8 ± 22.3 |
| CCCP | −166.2 ± 9.9 | −148.3 ± 13.9 |
| CCCP + KCl | −100.5 ± 6.1 | −72.5 ± 8.0 |
| S. c. FY833 | ||
| Glucose | −164.3 ± 12.6 b | −153.51 ± 12.3 b |
| CCCP | −130.4 ± 11.1 | −115.46 ± 12.8 |
| CCCP + KCl | −36.9 ± 15.2 | −71.66 * |
| S.c. W303-1A | ||
| Glucose | −179.3 ± 15.6 b | −160.7 ± 16.5 b |
| CCCP | −154.3 ± 16.4 | −133.1 ± 15.8 |
| CCCP + KCl | −112.1 ± 24.1 | −96.2 ± 27.7 |
| C. albicans | ||
| Glucose | −248.8 ± 14.9 | −228.2 ± 19.0 |
| CCCP | −185.7 ± 16.5 | −153.2 ± 15.5 |
| CCCP + KCl | −142.0 ± 18.1 | −140.4 ± 19.9 |
| D. hansenii | ||
| Glucose | −154.4 ± 12.1 a,b | −116.9 ± 17.1 a,b |
| CCCP | −120.7 ± 12.2 | −56.7 ± 9.7 |
| CCCP + KCl | −101.5 ± 10.6 | −73.3 ± 18.4 |
| M. guilliermondii | ||
| Glucose | −167.1 ± 13.1 b | −135.7 ± 8.6 a,b |
| CCCP | −135.2 ± 10.9 | −96.9 ± 12.6 |
| CCCP + KCl | −111.8 ± 13.5 | −115.5 ± 19.0 |
| R. mucilaginosa | ||
| Glucose | −148.3 ± 20.3 a,b | −139.0 ± 13.8 a,b |
| CCCP | −70.5 ± 10.8 | −32.4 ± 2.6 |
| CCCP + KCl | −75.1 ± 8.3 | −72.7 ± 8.8 |
| Non-Corrected | Corrected | |
|---|---|---|
| Δψ (mV) ± SD | Δψ (mV) ± SD | |
| FY833 | ||
| Glucose | −164.3 ± 12.6 | −153.51 ± 12.3 |
| CCCP | −130.4 ± 11.1 | −115.46 ± 12.8 |
| CCCP + KCl | −36.9 ± 15.2 | −71.66 * |
| trk1Δ | ||
| Glucose | −186.4 ± 5.7 a | −174.9 ± 4.8 a |
| CCCP | −143.1 ± 7.3 | −128.1 ± 8.3 |
| CCCP + KCl | −134.9 ± 4.8 | −126.6 ± 8.3 |
| trk2Δ | ||
| Glucose | −157.0 ± 8.6 ns | −147.9 ± 7.3 ns |
| CCCP | −129.9 ± 4.2 | −118.8 ± 5.5 |
| CCCP + KCl | −54.4 ± 4.9 | −21.17 ** |
| trk1Δ trk2Δ | ||
| Glucose | −193.8 ± 12.7 a | −179.7 ± 13.9 a |
| CCCP | −158.9 ± 10.6 | −143.2 ± 11.8 |
| CCCP + KCl | −163.8 ± 9.7 | −155.9 ± 9.2 |
| W303-1A | ||
| Glucose | −186.8 ± 7.0 | −167.7 ± 11.8 |
| CCCP | −162.9 ± 4.4 | −143.8 ± 6.4 |
| CCCP + KCl | −117.9 ± 21.7 | −121.2 ± 3.5 |
| TOW | ||
| Glucose | −172.8 ± 9.4 ns | −159.4 ± 6.7 ns |
| CCCP | −141.1 ± 7.3 | −125.1 ± 4.5 |
| CCCP + KCl | −96.6 ± 9.3 | −82.1 ± 9.5 |
| Glucose | CCCP | CCCP + KCl | |
|---|---|---|---|
| 10 µM CCCP | −167.1 | −135.2 | −111.8 |
| 5 µM CCCP | −121.81 | −113.9 | −97.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peña, A.; Sánchez, N.S.; Padilla-Garfias, F.; Ramiro-Cortés, Y.; Araiza-Villanueva, M.; Calahorra, M. The Use of Thioflavin T for the Estimation and Measurement of the Plasma Membrane Electric Potential Difference in Different Yeast Strains. J. Fungi 2023, 9, 948. https://doi.org/10.3390/jof9090948
Peña A, Sánchez NS, Padilla-Garfias F, Ramiro-Cortés Y, Araiza-Villanueva M, Calahorra M. The Use of Thioflavin T for the Estimation and Measurement of the Plasma Membrane Electric Potential Difference in Different Yeast Strains. Journal of Fungi. 2023; 9(9):948. https://doi.org/10.3390/jof9090948
Chicago/Turabian StylePeña, Antonio, Norma Silvia Sánchez, Francisco Padilla-Garfias, Yazmín Ramiro-Cortés, Minerva Araiza-Villanueva, and Martha Calahorra. 2023. "The Use of Thioflavin T for the Estimation and Measurement of the Plasma Membrane Electric Potential Difference in Different Yeast Strains" Journal of Fungi 9, no. 9: 948. https://doi.org/10.3390/jof9090948







