Selection of Salinity-Adapted Endorhizal Fungal Consortia from Two Inoculum Sources and Six Halophyte Plants
Abstract
:1. Introduction
2. Materials and Methods
2.1. Root and Rhizospheric Soil Sampling of Halophyte Plants
2.2. Physical and Chemical Analysis of Rhizospheric Soil
2.3. Propagation of AMF and DSF Consortia
2.4. Establishment of Fungal Trap Cultures
2.5. Evaluation and Selection of Endorhizal Fungi Consortia
2.5.1. Fungal Infectivity
2.5.2. Taxonomical Morphotypes Identification of Spores of AMF
2.5.3. Fungal Effectiveness
2.5.4. Elemental Analysis in AMF Spores and Hyphae
2.6. Surface Disinfection of Roots, DNA Extraction, and PCR Amplification
2.7. Experimental Design and Statistical Analysis
3. Results
3.1. Physical and Chemical Analysis of the Rhizospheric Soil of Each Composite Sample
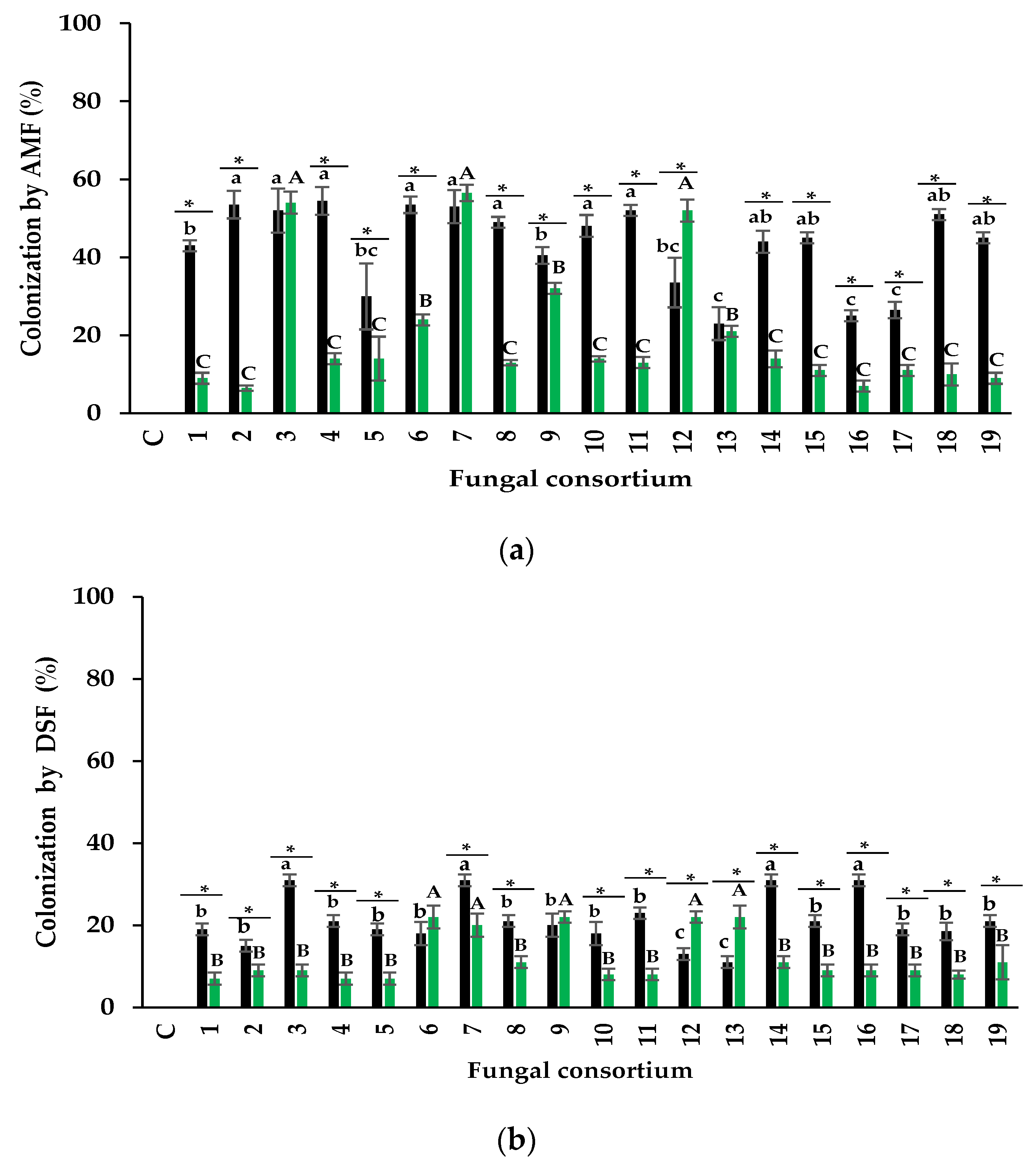
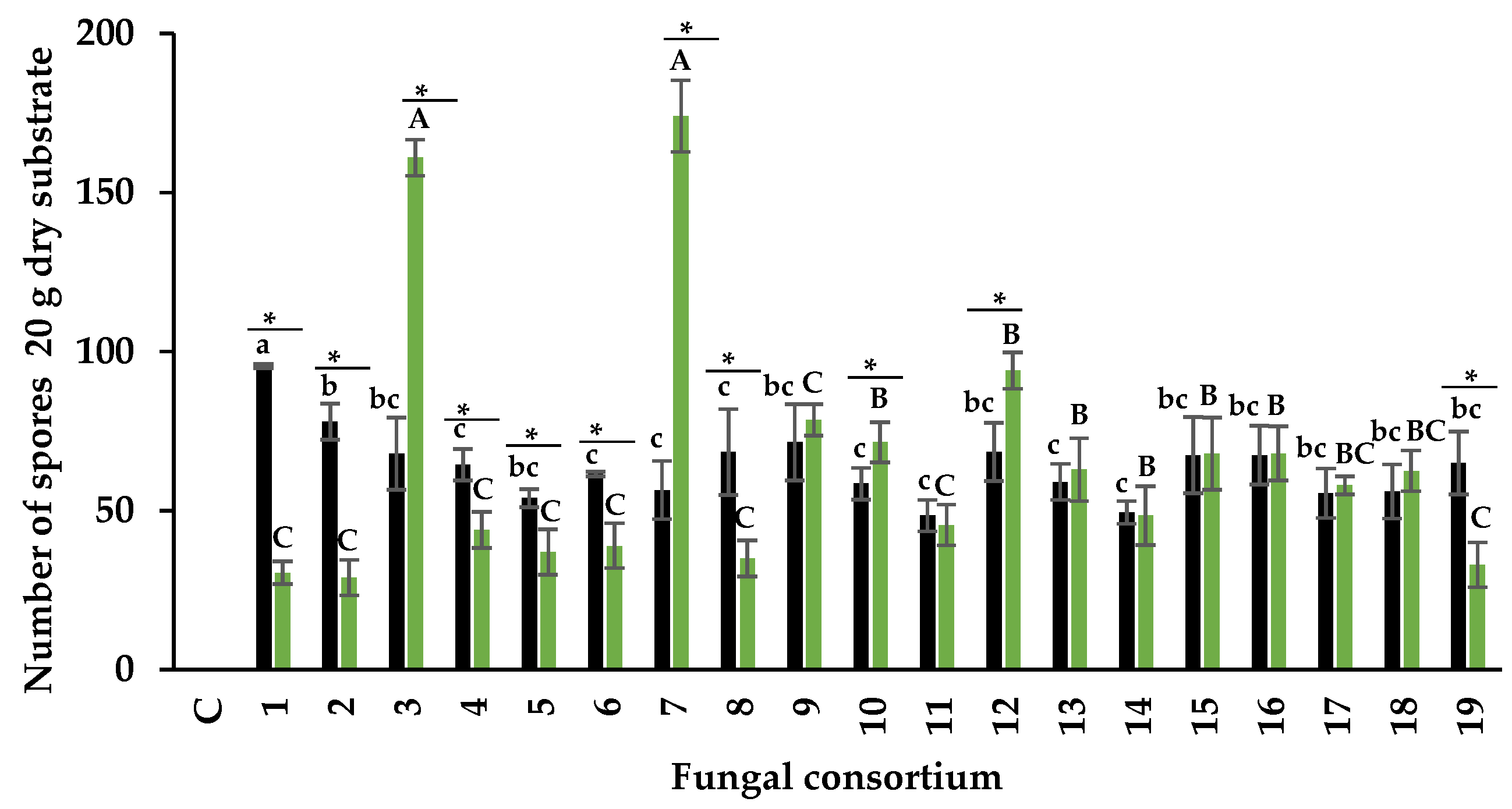
3.2. Infectivity of Fungal Consortia
3.2.1. Arbuscular Mycorrhizal Fungi
Mycorrhizal Colonization
Number of Spores of AMF
3.2.2. Dark Septate Fungi
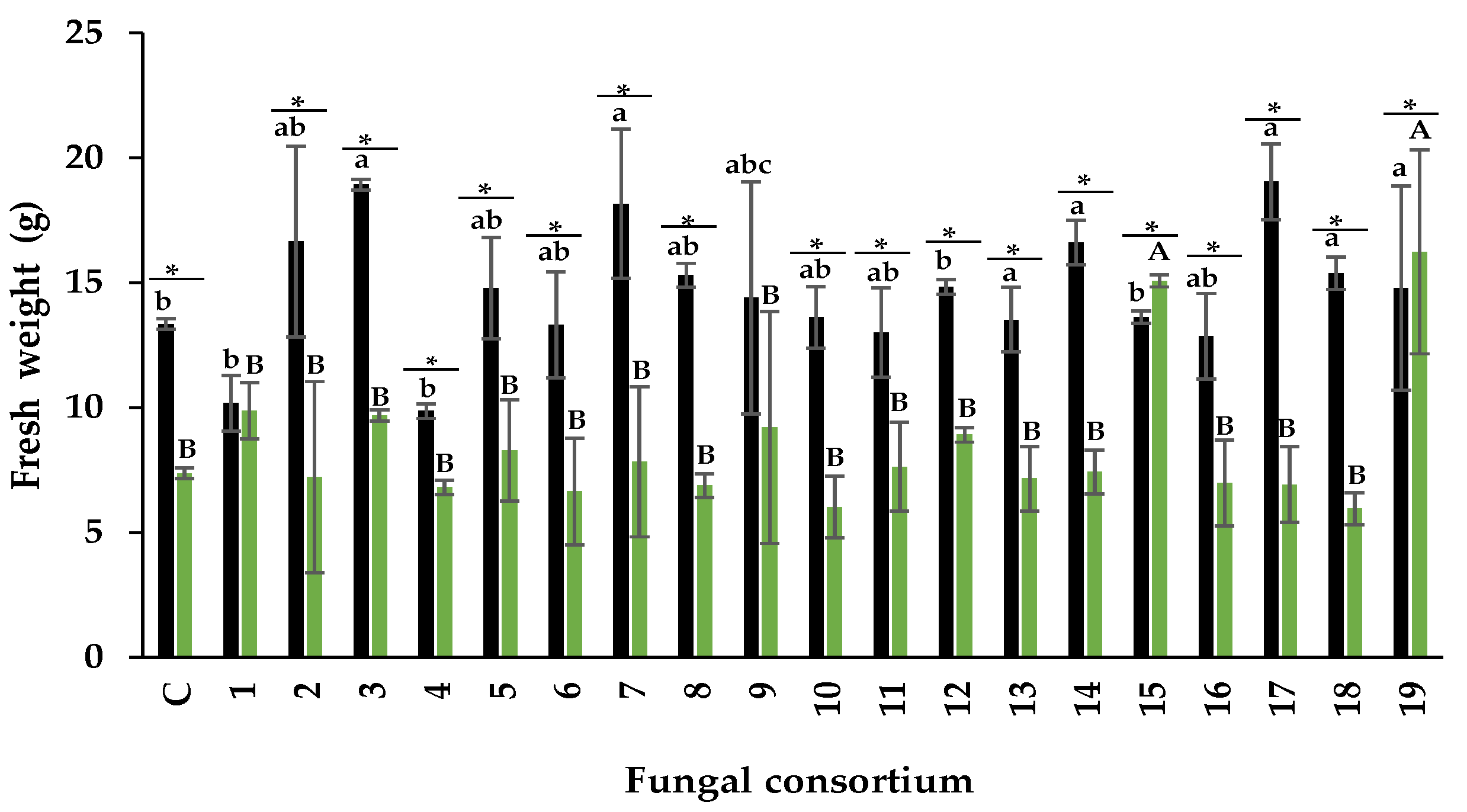
3.3. Mitigation of Salt Stress in Alfalfa Plants
3.3.1. Effectiveness of Fungal Consortia Propagated on Alfalfa
Fresh Weight
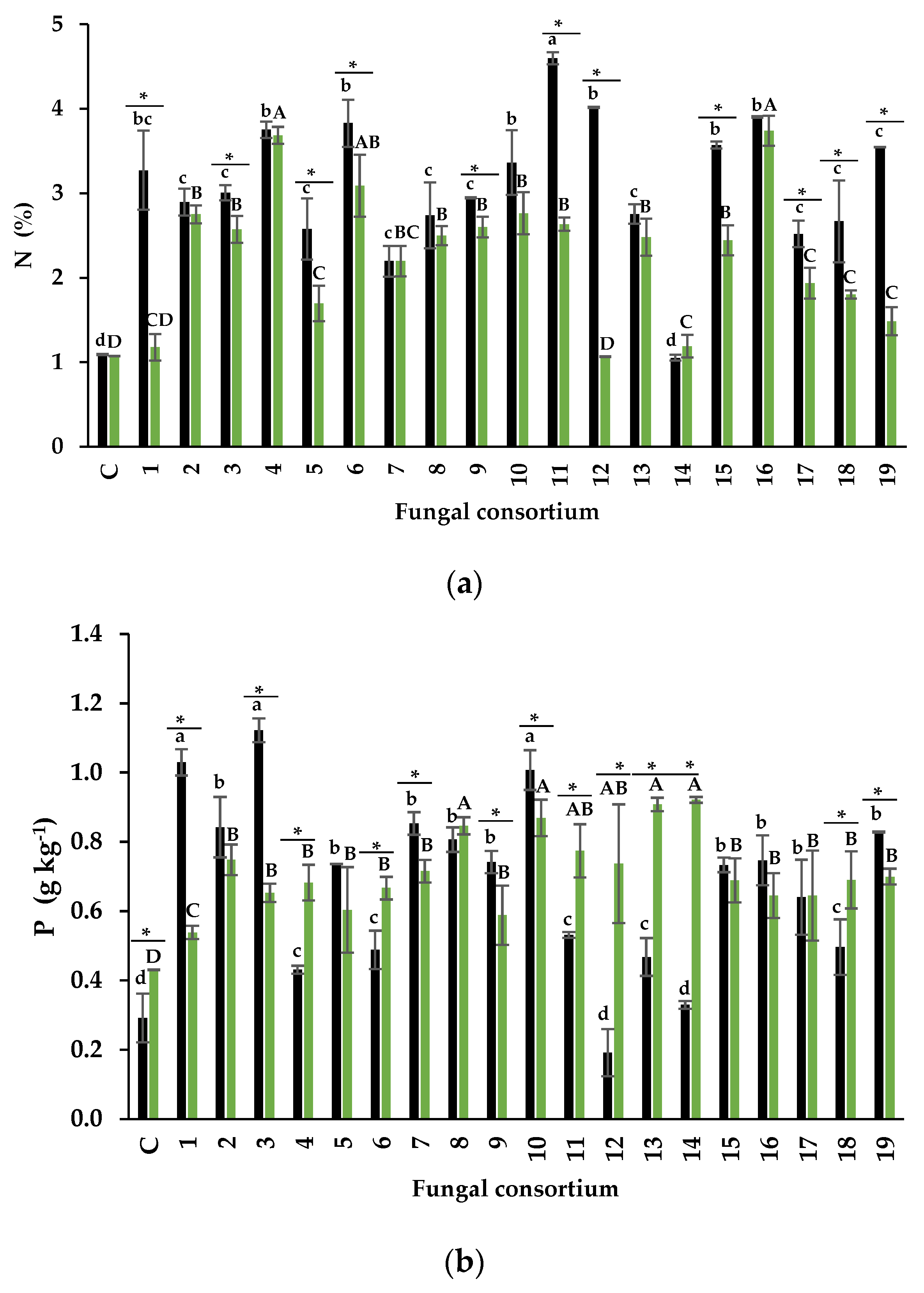
The Foliar Concentration of N and P
The Foliar Concentration of Na and Protective Osmolytes
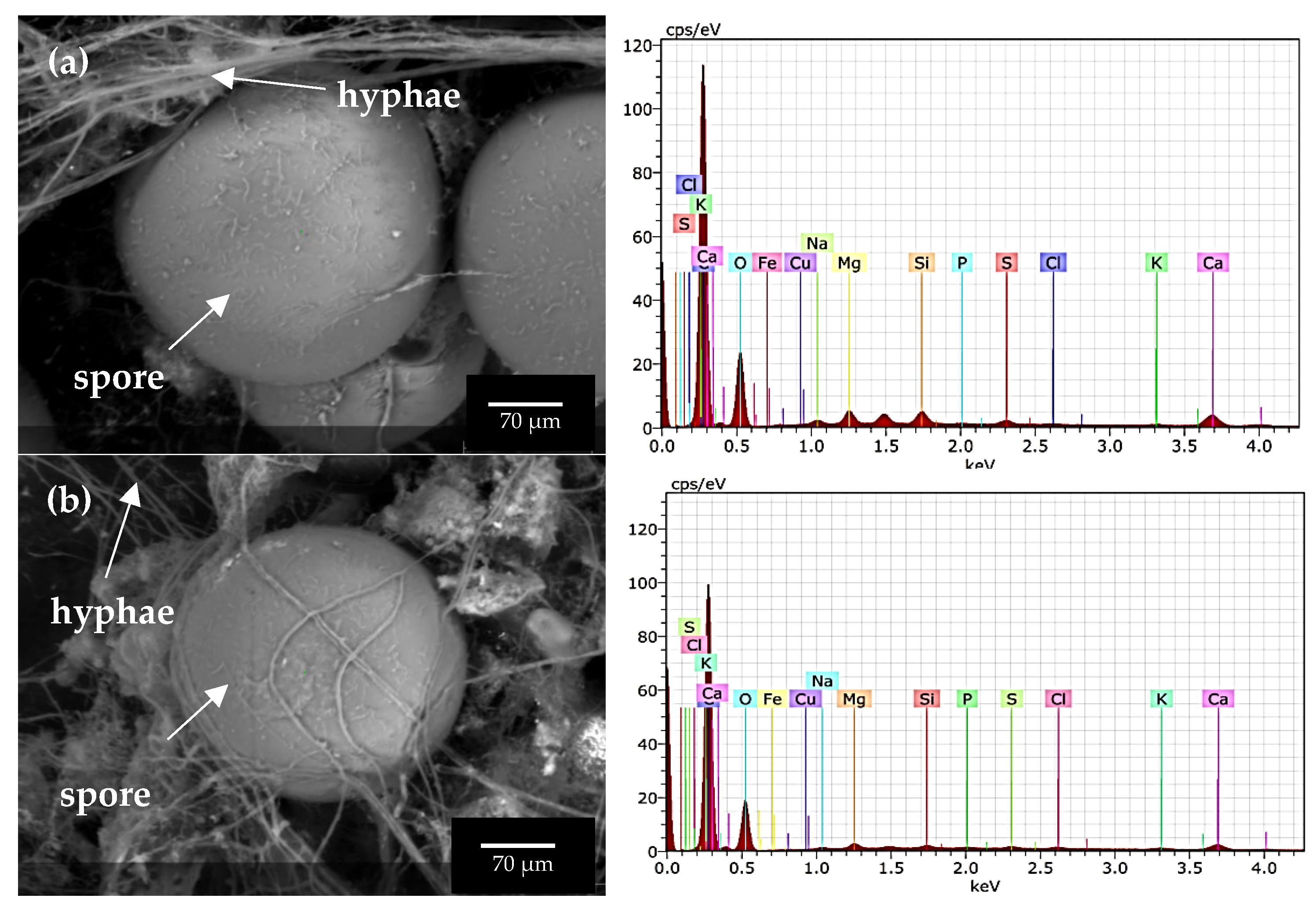
3.4. Scanning Electron Microscopy (SEM) and Elemental Analysis in AMF Fungal Structures
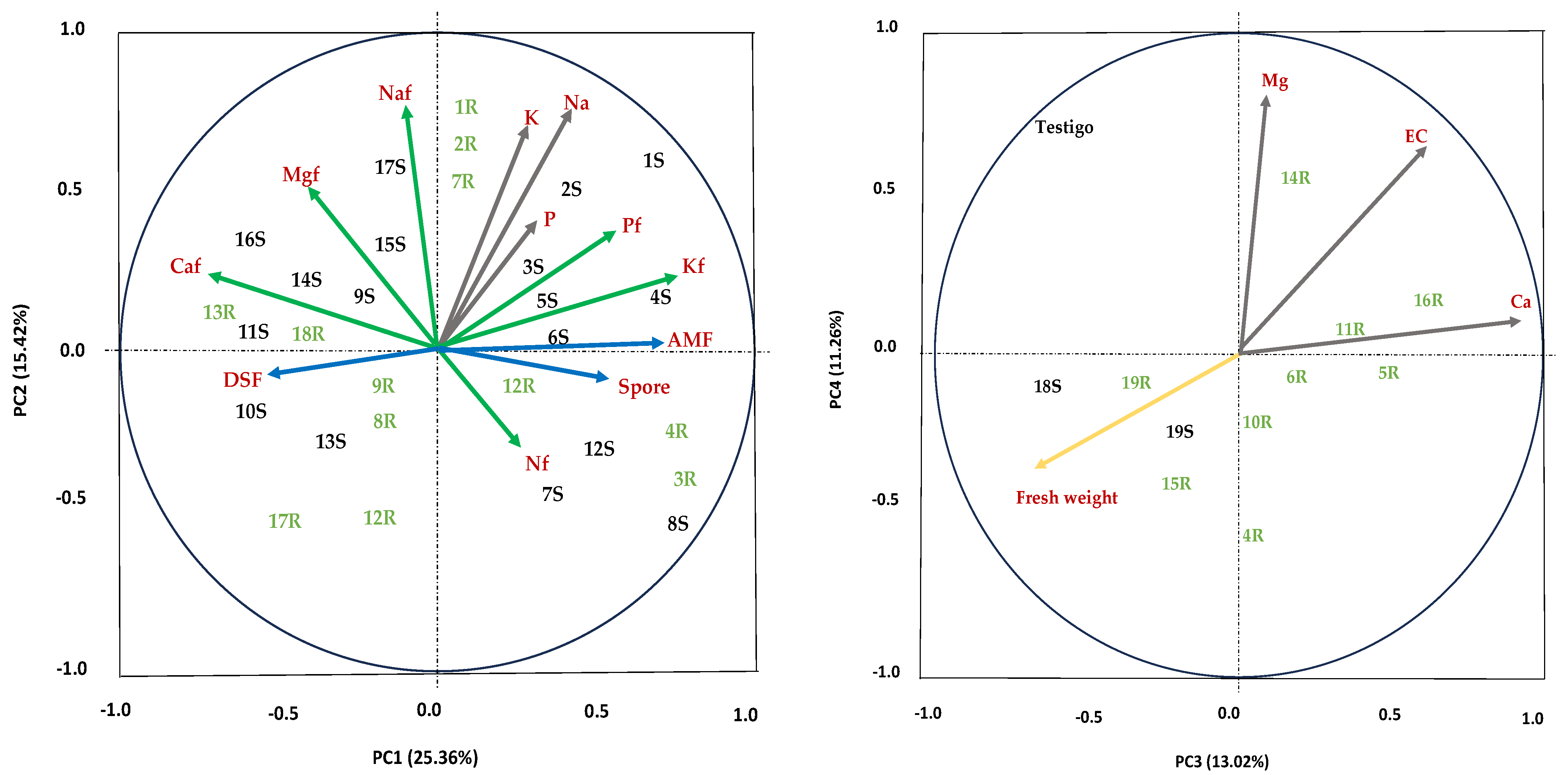
3.5. Principal Component Analysis (PCA)
3.6. Selection of Fungal Inoculants
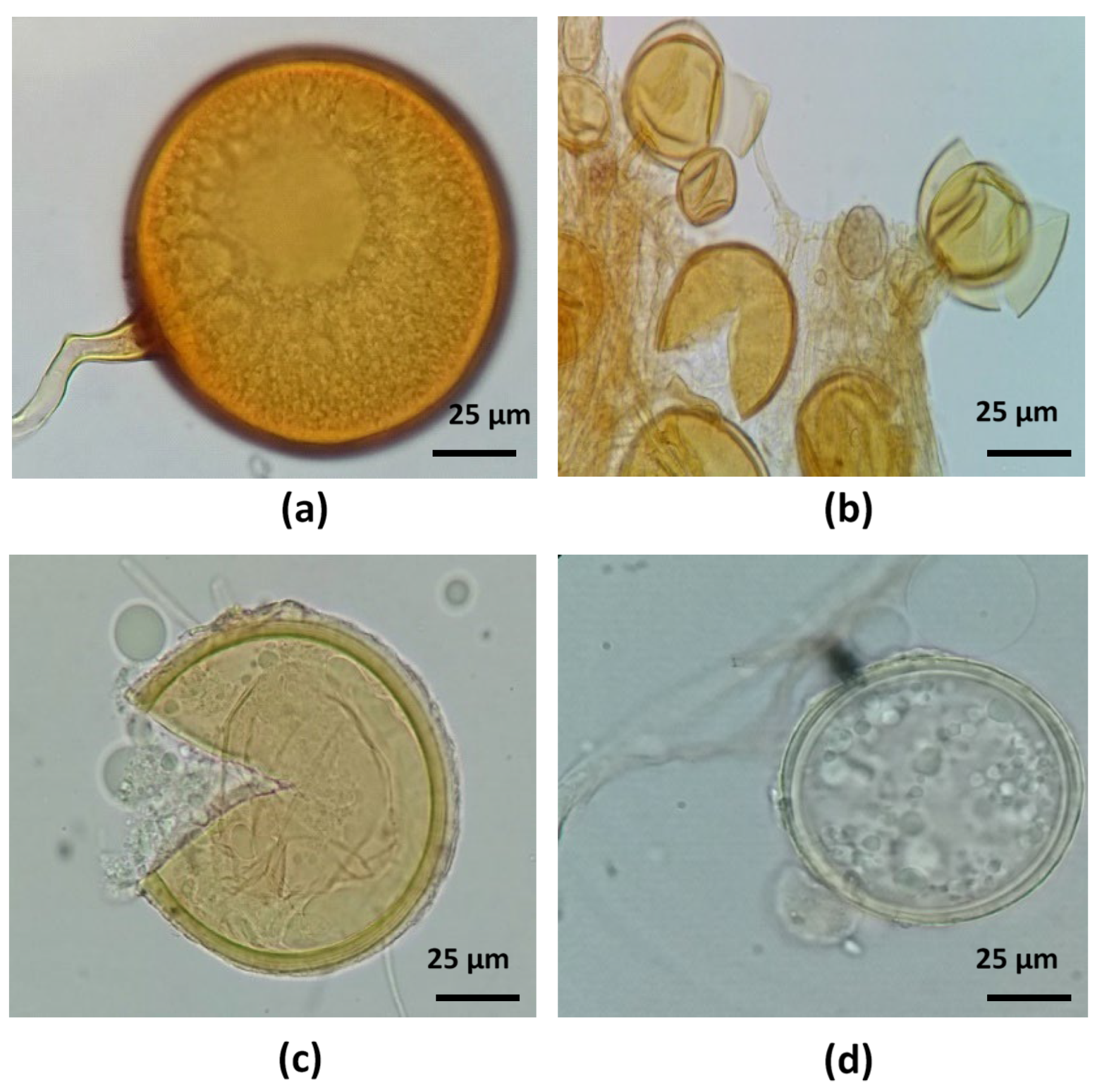
3.7. Identification of AMF Spore Morphospecies in the Selected Consortium
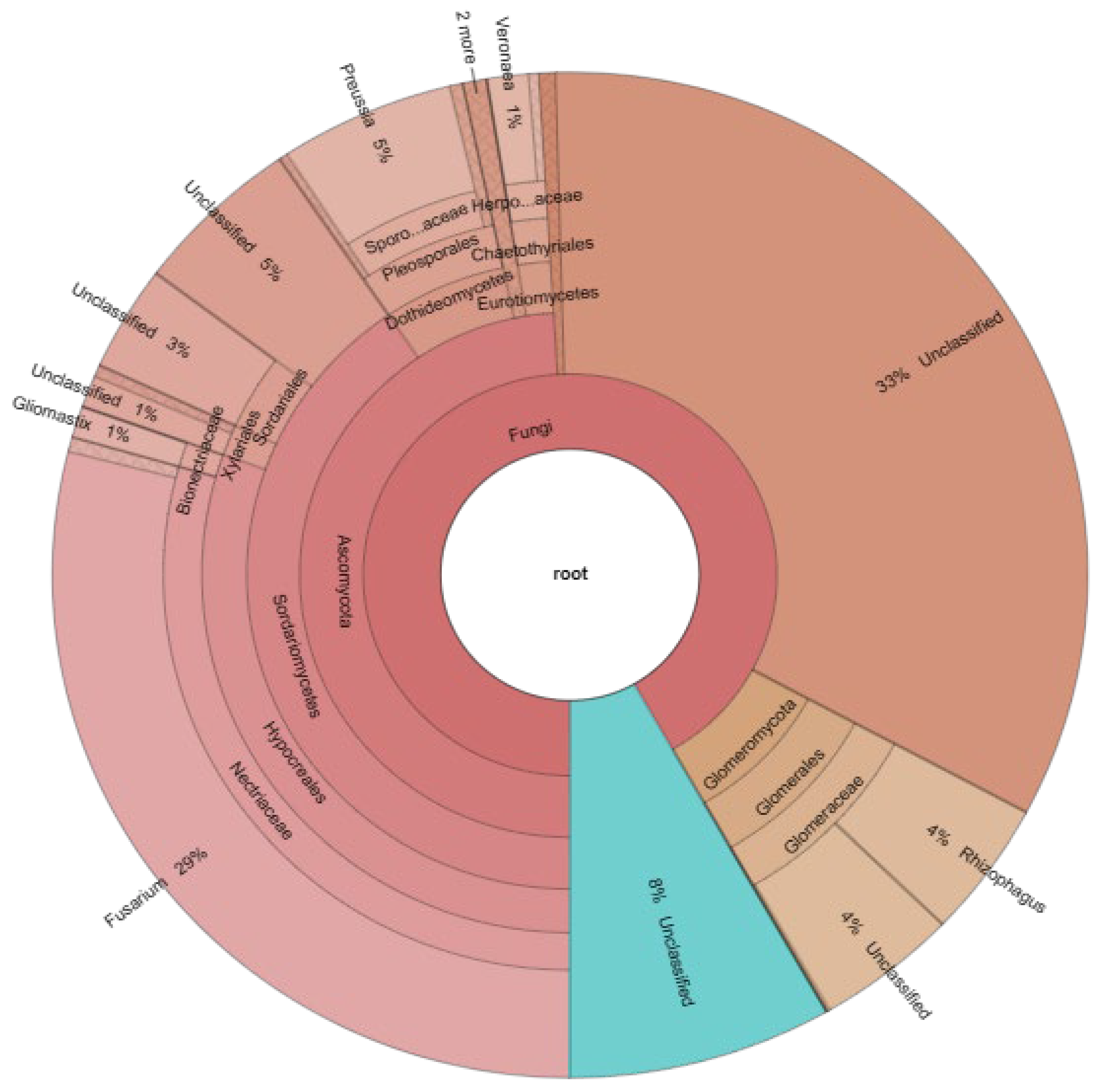
3.8. Endorhizal Community of the Selected Consortium
4. Discussion of Results
4.1. Physical and Chemical Analysis of the Rhizospheric Soil of Each Composite Sample
4.2. Infectivity of Fungal Consortia
4.2.1. Arbuscular Mycorrhizal Fungi
Mycorrhizal Colonization
Number of Spores
4.2.2. Dark Septate Fungi
4.3. Mitigation of Salt Stress in Alfalfa Plants
Effectiveness of Fungal Consortia Propagated on Alfalfa
4.4. Scanning Electron Microscopy (SEM) and Elemental Analysis in AMF Fungal Structures
4.5. Principal Component Analysis (PCA)
4.6. Selection of Fungal Inoculants
4.7. Identification of AMF Spore Morphospecies in the Selected Consortium
4.8. Endorhizal Community of the Selected Consortium
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shrivastava, P.; Kumar, R. Soil salinity: A serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J. Biol. Sci. 2015, 22, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Read, D. Mycorrhizal Symbiosis; Academic Press Ltd.: Cambridge, UK, 2008. [Google Scholar]
- Evelin, H.; Kapoor, R.; Giri, B. Arbuscular mycorrhizal fungi in alleviation of salt stress: A review. Ann. Bot. 2009, 104, 1263–1280. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Lozano, J.M.; Porcel, R.; Azcon, C.; Aroca, R. Regulation by arbuscular mycorrhizae of the integrated physiological response to salinity in plants: New challenges in physiological and molecular studies. J. Exp. Bot. 2012, 63, 4033–4044. [Google Scholar] [CrossRef] [PubMed]
- Hammer, E.C.; Nasr, H.; Pallon, J.; Olsson, P.A.; Wallander, H. Elemental composition of arbuscular mycorrhizal fungi at high salinity. Mycorrhiza 2011, 21, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Ruotsalainen, A.L.; Kauppinen, M.; Wäli, P.R.; Siekkinen, K.; Helander, M.; Tuomi, J. Dark septate endophytes: Mutualism from by-products? Trends Plant Sci. 2022, 27, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Jumpponen, A.R.I.; Trappe, J.M. Dark septate endophytes: A review of facultative biotrophic root-colonizing fungi. New Phytol. 1998, 140, 295–310. [Google Scholar] [CrossRef] [PubMed]
- Jumpponen, A. Dark septate endophytes–are they mycorrhizal? Mycorrhiza 2001, 11, 207–211. [Google Scholar] [CrossRef]
- Piercey, M.M.; Graham, S.W.; Currah, R.S. Patterns of genetic variation in Phialocephala fortinii across a broad latitudinal transect in Canada. Mycol. Res. 2004, 108, 955–964. [Google Scholar] [CrossRef]
- Knapp, D.G.; Kovács, G.M.; Zajta, E.; Groenewald, J.Z.; Crous, P.W. Dark septate endophytic Pleosporalean genera from semiarid areas. Pers.-Mol. Phylogeny Evol. Fungi 2015, 35, 87–100. [Google Scholar] [CrossRef]
- Yuan, Z.; Druzhinina, I.S.; Gibbons, J.G.; Zhong, Z.; van de Peer, Y.; Rodriguez, R.J.; Martin, F.M. Divergence of a genomic island leads to the evolution of melanization in a halophyte root fungus. ISME J. 2021, 15, 3468–3479. [Google Scholar] [CrossRef]
- Vergara, C.; Araujo, K.E.C.; Sperandio, M.V.L.; Santos, L.A.; Urquiaga, S.; Zilli, J.É. Dark septate endophytic fungi increase the activity of proton pumps, efficiency of 15N recovery from ammonium sulphate, N content, and micronutrient levels in rice plants. Braz. J. Microbiol. 2019, 50, 825–838. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Bai, N.; Wang, P.; Su, J.; Chang, Q.; Zhang, Q. Co-Inoculation with Arbuscular mycorrhizal fungi and dark septate Endophytes under Drought Stress: Synergistic or Competitive Effects on Maize Growth, Photosynthesis, Root Hydraulic properties and aquaporins? Plants 2023, 12, 2596. [Google Scholar] [CrossRef] [PubMed]
- Friede, M.; Unger, S.; Hellmann, C.; Beyschlag, W. Conditions promoting mycorrhizal parasitism are of minor importance for competitive interactions in two differentially mycotrophic species. Front. Plant Sci. 2016, 7, 1465. [Google Scholar] [CrossRef] [PubMed]
- Ruotsalainen, A.L.; Väre, H.; Vestberg, M. Seasonality of root fungal colonization in low-alpine herbs. Mycorrhiza 2002, 12, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, B.; Haselwandter, K. Red list plants: Colonization by arbuscular mycorrhizal fungi and dark septate endophytes. Mycorrhiza 2004, 14, 277–281. [Google Scholar] [CrossRef]
- Lingfei, L.; Anna, Y.; Zhiwei, Z. Seasonality of arbuscular mycorrhizal symbiosis and dark septate endophytes in a grassland site in southwest China. FEMS Microbiol. Ecol. 2005, 54, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Muthukuma, T.; Senthilkumar, M.; Rajangam, M.; Udaiyan, K. Arbuscular mycorrhizal morphology and dark septate fungal associations in medicinal and aromatic plants of Western Ghats, Southern India. Mycorrhiza 2006, 17, 11–24. [Google Scholar] [CrossRef]
- Postma, J.W.; Olsson, P.A.; Falkemgren-Grerup, U. Root colonization by arbuscular mycorrhizal, fine endophytic and dark septate fungi across a pH gradient in acid beech forests. Soil Biol. Biochem. 2007, 39, 400–408. [Google Scholar] [CrossRef]
- Marins, J.F.; Carrenho, R. Arbuscular mycorrhizal fungi and dark septate fungi in plants associated with aquatic environments. Acta Bot. Bras. 2017, 31, 295–308. [Google Scholar] [CrossRef]
- Xie, L.; Bi, Y.; Ma, S.; Shang, J.; Hu, Q.; Christie, P. Combined inoculation with dark septate endophytes and arbuscular mycorrhizal fungi: Synergistic or competitive growth effects on maize? BMC Plant Biol. 2021, 21, 498. [Google Scholar] [CrossRef]
- He, Y.M.; Fan, X.M.; Zhang, G.Q.; Li, B.; Li, T.G.; Zu, Y.Q.; Zhan, F.D. Effects of arbuscular mycorrhizal fungi and dark septate endophytes on maize performance and root traits under a high cadmium stress. S. Afr. J. Bot. 2020, 134, 415–423. [Google Scholar] [CrossRef]
- Cofré, M.N.; Becerra, A.G.; Nouhra, E.R.; Soteras, M.F. Arbuscular mycorrhizae and dark-septate endophytes on Atriplex cordobensis in saline sites from Argentina. J. Agric. Technol. 2012, 8, 2201–2214. [Google Scholar]
- Trejo-Aguilar, D.; Banuelos, J. Isolation and culture of arbuscular mycorrhizal fungi from field samples. In Arbuscular Mycorrhizal Fungi: Methods and Protocols; Methods in Molecular Biology; Ferrol, N., Lanfranco, L., Eds.; Humana Press: New York, NY, USA, 2020; pp. 1–18. [Google Scholar] [CrossRef]
- Douds, D.D., Jr.; Nagahashi, G.; Pfeffer, P.E.; Kayser, W.M.; Reider, C. On-farm production and utilization of arbuscular mycorrhizal fungus inoculum. Can. J. Plant Sci. 2005, 85, 15–21. [Google Scholar] [CrossRef]
- Bencherif, K.; Laruelle, F.; Tisserant, B.; Dalpé, Y.; Lounés-Hadj Sahraoui, A. Engineering approach for production of arbuscular mycorrhizal inoculum adapted to saline soil management. Stresses 2023, 3, 404–423. [Google Scholar] [CrossRef]
- Valenzuela-Encinas, C.; Neria-González, I.; Alcántara-Hernández, R.J.; Enríquez-Aragón, J.A.; Estrada-Alvarado, I.; Hernández-Rodríguez, C.; Marsch, R. Phylogenetic analysis of the archaeal community in an alkaline-saline soil of the former lake Texcoco (Mexico). Extremophiles 2008, 12, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.R. Estimation of Available Phosphorus in Soils by Extraction with Sodium Bicarbonate; No. 939; US Department of Agriculture: Washington, DC, USA, 1954.
- Rodrı́guez-Navarro, A. Potassium transport in fungi and plants. Biochim. Biophys. Acta BBA-Rev. Biomembr. 2000, 1469, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Borax. Effects of Sodium in Borax on Soils and Crops; U.S Borax: Boron, CA, USA, 2002. [Google Scholar]
- Wood, C.W.; Adams, J.F.; Wood, B.H. Macronutrients. In Encyclopedia of Soils in the Environment; Elsevier: Amsterdam, The Netherlands, 2005; pp. 387–393. [Google Scholar] [CrossRef]
- Marschner, H. Marschner’s Mineral Nutrition of Higher Plants; Academic Press: San Diego, CA, USA, 2012; Volume 89. [Google Scholar]
- Millner, P.D.; Kitt, D.G. The Beltsville method for soilless production of vesicular-arbuscular mycorrhizal fungi. Mycorrhiza 1992, 2, 9–15. [Google Scholar] [CrossRef]
- INVAM. Results of Infectivity (MIP) Assays of Commercial Inoculants. Available online: http://invam.caf.wvu.edu/otherinfo/commercial/commercial_MIPs.pdf (accessed on 29 December 2011).
- Phillips, J.M.; Hayman, D.S. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc. 1970, 55, 158-IN18. [Google Scholar] [CrossRef]
- Koske, R.E.; Gemma, J.N. A modified procedure for staining roots to detect VA mycorrhizas. Mycol. Res. 1989, 92, 486. [Google Scholar] [CrossRef]
- Gerdemann, J.W.; Nicolson, T.H. Spores of mycorrhizal Endogone species extracted from soil by wet sieving and decanting. Trans. Br. Mycol. Soc. 1963, 46, 235–244. [Google Scholar] [CrossRef]
- David, D.J. Determination of calcium in plant material by atomic-absorption spectrophotometry. Analyst 1959, 84, 536–545. [Google Scholar] [CrossRef]
- Havre, G.N. The flame photometric determination of sodium, potassium and calcium in plant extracts with special reference to interference effects. Anal. Chim. Acta 1961, 25, 557–566. [Google Scholar] [CrossRef]
- Kitson, R.E.; Mellon, M.G. Colorimetric determination of germanium as molybdigermanic acid. Ind. Eng. Chem. Anal. Ed. 1944, 16, 128–130. [Google Scholar] [CrossRef]
- Kendall, C. Tracing nitrogen sources and cycling in catchments. In Isotope Tracers in Catchment Hydrology; Elsevier: Amsterdam, The Netherlands, 1998; pp. 519–576. [Google Scholar] [CrossRef]
- Kassambara, A.; Mundt, F.; Package ‘factoextra’. Extract and Visualize the Results of Multivariate Data Analyses. 2017. Available online: http://www.sthda.com/english/rpkgs/factoextra (accessed on 17 August 2023).
- Silva, C.C. Análisis del Cambio en las Comunidades Microbianas en un Suelo Salino-Alcalino del Exlago de Texcoco en Presencia de Antraceno. Ph.D. Dissertation, Cinvestav, Mexico City, Mexico, 2013. Available online: https://repositorio.cinvestav.mx/handle/cinvestav/1336 (accessed on 17 August 2023).
- Beltrán-Hernández, R.I.; Luna-Guido, M.L.; Dendooven, L. Emission of carbon dioxide and dynamics of inorganic N in a gradient of alkaline saline soils of the former lake Texcoco. Appl. Soil Ecol. 2007, 35, 390–403. [Google Scholar] [CrossRef]
- Santoyo de la Cruz, M.F.; Flores-Magdaleno, H.; Khalil-Gardezi, A.; Mancilla-Villa, Ó.R.; Rubiños-Panta, J.E. Composición iónica y comparación de índices de salinidad de suelo agrícola de Texcoco, México. Nova Sci. 2021, 13, 1–20. [Google Scholar] [CrossRef]
- Jones, J.B., Jr.; Wolf, B. Manual Soil Testing Procedure Using Modified (Wolf) Morgan Extracting Reagent; No. 631.42 J62; Benton Laboratories: Athens, GA, USA, 1984. [Google Scholar]
- Xie, W.; Yang, J.; Gao, S.; Yao, R.; Wang, X. The effect and influence mechanism of soil salinity on phosphorus availability in coastal salt-affected soils. Water 2022, 14, 2804. [Google Scholar] [CrossRef]
- Mahmood, I.A.; Arshad, A.; Aslam, M.; Shahzad, A.; Sultan, T.; Hussain, F. Phosphorus availability in different salt-affected soils as influenced by crop residue incorporation. Int. J. Agric. Biol. 2013, 15, 472–478. [Google Scholar]
- Hou, L.; Yu, J.; Zhao, L.; He, X. Dark septate endophytes improve the growth and the tolerance of Medicago sativa and Ammopiptanthus mongolicus under cadmium stress. Front. Microbiol. 2020, 10, 3061. [Google Scholar] [CrossRef]
- Ban, Y.; Tang, M.; Chen, H.; Xu, Z.; Zhang, H.; Yang, Y. The response of dark septate endophytes (DSE) to heavy metals in pure culture. PLoS ONE 2012, 7, 47968. [Google Scholar] [CrossRef]
- Abbott, L.K.; Gazey, C. An ecological view of the formation of VA mycorrhizas. Plant Soil 1994, 159, 69–78. [Google Scholar] [CrossRef]
- Biermann, B.; Linderman, R.G. Use of vesicular-arbuscular mycorrhizal roots, intra-radical vesicles and extra-radical vesicles as inoculum. New Phytol. 1983, 95, 97–105. [Google Scholar] [CrossRef]
- Aliasgharzadeh, N.; Rastin, S.N.; Towfighi, H.; Alizadeh, A. Occurrence of arbuscular mycorrhizal fungi in saline soils of the Tabriz Plain of Iran in relation to some physical and chemical properties of soil. Mycorrhiza 2001, 11, 119–122. [Google Scholar] [CrossRef]
- Barrow, J.R.; Havstad, K.M.; McCaslin, B.D. Fungal root endophytes in fourwing saltbush, Atriplex canescens, on arid rangelands of southwestern USA. Arid. Land Res. Manag. 1997, 11, 177–185. [Google Scholar] [CrossRef]
- Hirrel, M.C. The effect of sodium and chloride salts on the germination of Gigaspora margarita. Mycologia 1981, 73, 610–617. [Google Scholar] [CrossRef]
- Kim, C.K.; Weber, D.J. Distribution of VA mycorrhiza on halophytes on inland salt playas. Plant Soil 1985, 83, 207–214. [Google Scholar] [CrossRef]
- Hayman, D.S. Endogone spore numbers in soil and vesicular-arbuscular mycorrhiza in wheat as influenced by season and soil treatment. Trans. Br. Mycol. Soc. 1970, 54, 53-IN12. [Google Scholar] [CrossRef]
- Daft, M.J.; Nicolson, T.H. Effect of Endogone mycorrhiza on plant growth. IV. Quantitative relationships between the growth of the host and the development of the endophyte in tomato and maize. New Phytol. 1972, 71, 287–295. [Google Scholar] [CrossRef]
- Currah, R.S.; Tsuneda, A.; Murakami, S. Morphology and ecology of Phialocephala fortinii in roots of Rhododendron brachycarpum. Can. J. Bot. 1993, 71, 1639–1644. [Google Scholar] [CrossRef]
- Gonzalez Mateu, M.; Baldwin, A.H.; Maul, J.E.; Yarwood, S.A. Dark septate endophyte improves salt tolerance of native and invasive lineages of Phragmites australis. ISME J. 2020, 14, 1943–1954. [Google Scholar] [CrossRef]
- Farias, G.C.; Nunes, K.G.; Soares, M.A.; de Siqueira, K.A.; Lima, W.C.; Neves, A.L.R.; de Lacerda, C.F. Dark septate endophytic fungi mitigate the effects of salt stress on cowpea plants. Braz. J. Microbiol. 2020, 51, 243–253. [Google Scholar] [CrossRef]
- Hawksworth, D.L.; Rossman, A.Y. Where are all the undescribed fungi? Phytopathology 1997, 87, 888–891. [Google Scholar] [CrossRef] [PubMed]
- Sonjak, S.; Udovič, M.; Wraber, T.; Likar, M.; Regvar, M. Diversity of halophytes and identification of arbuscular mycorrhizal fungi colonising their roots in an abandoned and sustained part of Sečovlje salterns. Soil Biol. Biochem. 2009, 41, 1847–1856. [Google Scholar] [CrossRef]
- Anand, A.; Baig, M.J.; Mandal, P.K. Response of alfalfa genotypes to saline water irrigation. Biol. Plant. 2000, 43, 455–457. [Google Scholar] [CrossRef]
- Maggio, A.; Chiaranda, F.Q.; Cefariello, R.; Fagnano, M. Responses to ozone pollution of alfalfa exposed to increasing salinity levels. Environ. Pollut. 2009, 157, 1445–1452. [Google Scholar] [CrossRef] [PubMed]
- Bertram, N.A.; Alfonso, C.; Grande, S.; Chiacchiera, S.; Ohanian, A.; Bonvillani, J.; Angeletti, F.R. Efecto de la concentración salina y el régimen hídrico sobre la germinación, emergencia y establecimiento de alfalfa (Medicago sativa L.). RIA Rev. Investig. Agropecu. 2021, 47, 267–272. [Google Scholar]
- Campanelli, A.; Ruta, C.; de Mastro, G.; Morone-Fortunato, I. The role of arbuscular mycorrhizal fungi in alleviating salt stress in Medicago sativa L. var. icon. Symbiosis 2013, 59, 65–76. [Google Scholar] [CrossRef]
- Evelin, H.; Giri, B.; Kapoor, R. Contribution of Glomus intraradices inoculation to nutrient acquisition and mitigation of ionic imbalance in NaCl-stressed Trigonella foenumgraecum. Mycorrhiza 2012, 22, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Porcel, R.; Aroca, R.; Azcon, R.; Ruiz-Lozano, J.M. Regulation of cation transporter genes by the arbuscular mycorrhizal symbiosis in rice plants subjected to salinity suggests improved salt tolerance due to reduced Na+ root-to-shoot distribution. Mycorrhiza 2016, 26, 673–684. [Google Scholar] [CrossRef]
- Mattson, W.J., Jr. Herbivory in relation to plant nitrogen content. Annu. Rev. Ecol. Syst. 1980, 11, 119–161. [Google Scholar] [CrossRef]
- Grattan, S.R.; Grieve, C.M. Salinity mineral nutrient relations in horticultural crops. Sci. Hortic. 1998, 78, 127–157. [Google Scholar] [CrossRef]
- Martínez, C.M.; Marcos, M.F.; Rodriguez, E.A. Factors influencing phosphorus adsorption in mine soils in Galicia, Spain. Sci. Total Environ. 1996, 180, 137–145. [Google Scholar] [CrossRef]
- Martinez, V.; Läuchli, A. Salt-induced inhibition of phosphate uptake in plants of cotton (Gossypium hirsutum L.). New Phytol. 1994, 126, 609–614. [Google Scholar] [CrossRef]
- Malhotra, H.; Sharma, S.; Pandey, R. Phosphorus nutrition: Plant growth in response to deficiency and excess. In Plant Nutrients and Abiotic Stress Tolerance; Hasanuzzaman, M., Fujita, M., Oku, H., Nahar, K., Hawrylak-Nowak, B., Eds.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 171–190. [Google Scholar] [CrossRef]
- Chandra, P.; Singh, A.; Prajapat, K.; Rai, A.K.; Yadav, R.K. Native arbuscular mycorrhizal fungi improve growth, biomass yield, and phosphorus nutrition of sorghum in saline and sodic soils of the semi–arid region. Environ. Exp. Bot. 2022, 201, 104982. [Google Scholar] [CrossRef]
- Della Mónica, I.F.; Saparrat, M.C.; Godeas, A.M.; Scervino, J.M. The co-existence between DSE and AMF symbionts affects plant P pools through P mineralization and solubilization processes. Fungal Ecol. 2015, 17, 10–17. [Google Scholar] [CrossRef]
- Latef, A.A.H.A.; Chaoxing, H. Effect of arbuscular mycorrhizal fungi on growth, mineral nutrition, antioxidant enzymes activity and fruit yield of tomato grown under salinity stress. Sci. Hortic. 2011, 127, 228–233. [Google Scholar] [CrossRef]
- Gaber, D.A.; Berthelot, C.; Camehl, I.; Kovacs, G.M.; Blaudez, D.; Franken, P. Salt stress tolerance of dark septate endophytes is independent of melanin accumulation. Front. Microbiol. 2020, 11, 3158. [Google Scholar] [CrossRef] [PubMed]
- Laouane, R.B.; Baslam, M.; Ait-El-Mokhtar, M.; Anli, M.; Boutasknit, A.; Ait-Rahou, Y.; Meddich, A. Potential of native arbuscular mycorrhizal fungi, rhizobia, and/or green compost as alfalfa (Medicago sativa) enhancers under salinity. Microorganisms 2020, 8, 1695. [Google Scholar] [CrossRef]
- Estrada, B.; Aroca, R.; Maathuis, F.J.; Barea, J.M.; Ruiz-Lozano, J.M. Arbuscular mycorrhizal fungi native from a Mediterranean saline area enhance maize tolerance to salinity through improved ion homeostasis. Plant Cell Environ. 2013, 36, 1771–1782. [Google Scholar] [CrossRef]
- Zuccarini, P.; Okurowska, P. Effects of mycorrhizal colonization and fertilization on growth and photosynthesis of sweet basil under salt stress. J. Plant Nutr. 2008, 31, 497–513. [Google Scholar] [CrossRef]
- Yun, P.; Xu, L.; Wang, S.-S.; Shabala, L.; Shabala, S.; Zhang, W.Y. Piriformospora indica improves salinity stress tolerance in Zea mays L. plants by regulating Na+ and K+ loading in root and allocating K+ in shoots. Plant Growth Regul. 2018, 86, 323–331. [Google Scholar] [CrossRef]
- Song, M.; Chai, Q.; Li, X.; Yao, X.; Li, C.; Christensen, M.J.; Nan, Z. An asexual Epichloë endophyte modifies the nutrient stoichiometry of wild barley (Hordeum brevisubulatum) under salt stress. Plant Soil 2015, 387, 153–165. [Google Scholar] [CrossRef]
- Ghabooli, M. Effect of Piriformospora indica inoculation on some physiological traits of barley (Hordeum vulgare) under salt stress. Chem. Nat. Compd. 2014, 50, 1082–1087. [Google Scholar] [CrossRef]
- LaHaye, P.; Epstein, E. Salt toleration by plants: Enhancement with calcium. Science 1969, 166, 395–396. [Google Scholar] [CrossRef] [PubMed]
- Marschner, H.; Cakmak, I. High light intensity enhances chlorosis and necrosis in leaves of zinc, potassium, and magnesium deficient bean (Phaseolus vulgaris) plants. J. Plant Physiol. 1989, 134, 308–315. [Google Scholar] [CrossRef]
- Yin, L.; Ren, A.; Wei, M.; Wu, L.; Zhou, Y.; Li, X.; Gao, Y. Neotyphodium coenophialum-infected tall fescue and its potential application in the phytoremediation of saline soils. Int. J. Phytoremed. 2014, 16, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Giri, B.; Mukerji, K.G. Mycorrhizal inoculant alleviates salt stress in Sesbania aegyptiaca and Sesbania grandiflora under field conditions: Evidence for reduced sodium and improved magnesium uptake. Mycorrhiza 2004, 14, 307–312. [Google Scholar] [CrossRef]
- Zare-Maivan, H.; Khanpour-Ardestani, N.; Ghanati, F. Influence of mycorrhizal fungi on growth, chlorophyll content, and potassium and magnesium uptake in maize. J. Plant Nutr. 2017, 40, 2026–2032. [Google Scholar] [CrossRef]
- Vergara, C.; Araujo, K.E.; Urquiaga, S.; Schultz, N.; Balieiro, F.D.C.; Medeiros, P.S.; Zilli, J.E. Dark septate endophytic fungi help tomato to acquire nutrients from ground plant material. Front. Microbiol. 2017, 8, 2437. [Google Scholar] [CrossRef]
- Porras-Alfaro, A.; Bayman, P. Hidden fungi, emergent properties: Endophytes and microbiomes. Annu. Rev. Phytopathol. 2011, 49, 291–315. [Google Scholar] [CrossRef]
- Choudhury, M.R.; Christopher, J.; Das, S.; Apan, A.; Menzies, N.W.; Chapman, S.; Dang, Y.P. Detection of calcium, magnesium, and chlorophyll variations of wheat genotypes on sodic soils using hyperspectral red edge parameters. Environ. Technol. Innov. 2022, 27, 102469. [Google Scholar] [CrossRef]
- Dhiman, P.; Rajora, N.; Bhardwaj, S.; Sudhakaran, S.S.; Kumar, A.; Raturi, G.; Chakraborty, K.; Gupta, O.P.; Devanna, B.N.; Tripathi, D.K.; et al. Fascinating role of silicon to combat salinity stress in plants: An updated overview. Plant Physiol. Biochem. 2021, 162, 110–123. [Google Scholar] [CrossRef] [PubMed]
- Saqib, M.; Zörb, C.; Schubert, S. Silicon-mediated improvement in the salt resistance of wheat (Triticum aestivum) results from increased sodium exclusion and resistance to oxidative stress. Funct. Plant Biol. 2008, 35, 633–639. [Google Scholar] [CrossRef]
- Ashraf, M.; McNeilly, T.; Bradshaw, A.D. The potential for evolution of salt (NaCl) tolerance in seven grass species. New Phytol. 1986, 103, 299–309. [Google Scholar] [CrossRef]
- Nagy, R.; Drissner, D.; Amrhein, N.; Jakobsen, I.; Bucher, M. Mycorrhizal phosphate uptake pathway in tomato is phosphorus-repressible and transcriptionally regulated. New Phytol. 2009, 181, 950–959. [Google Scholar] [CrossRef]
- Abbott, L.K.; Robson, A.D.; Gazey, C. Selection of Inoculant Vesicular-arbuscular Mycorrhizal Fungi. In Methods in Microbiology; Academic Press: Cambridge, MA, USA, 1992; Volume 24, pp. 1–21. [Google Scholar] [CrossRef]
- Wang, F.Y.; Liu, R.J.; Lin, X.G.; Zhou, J.M. Arbuscular mycorrhizal status of wild plants in saline-alkaline soils of the Yellow River Delta. Mycorrhiza 2004, 14, 133–137. [Google Scholar] [CrossRef]
- Berthelot, C.; Chalot, M.; Leyval, C.; Blaudez, D. From darkness to light: Emergence of the mysterious dark septate endophytes in plant growth promotion and stress alleviation. In Endophytes for a Growing World; Cambridge University Press: Cambridge, UK, 2019; pp. 143–164. [Google Scholar] [CrossRef]
- Mapperson, R.R.; Kotiw, M.; Davis, R.A.; Dearnaley, J.D. The diversity and antimicrobial activity of Preussia sp. endophytes isolated from Australian dry rainforests. Curr. Microbiol. 2014, 68, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Redman, R.S.; Anderson, J.A.; Biaggi, T.M.; Malmberg, K.E.; Rienstra, M.N.; Weaver, J.L.; Rodriguez, R.J. Symbiotic modulation as a driver of niche expansion of coastal plants in the San Juan Archipelago of Washington State. Front. Microbiol. 2022, 13, 868081. [Google Scholar] [CrossRef]
- Maciá-Vicente, J.G.; Ferraro, V.; Burruano, S.; Lopez-Llorca, L.V. Fungal assemblages associated with roots of halophytic and non-halophytic plant species vary differentially along a salinity gradient. Microb. Ecol. 2012, 64, 668–679. [Google Scholar] [CrossRef]
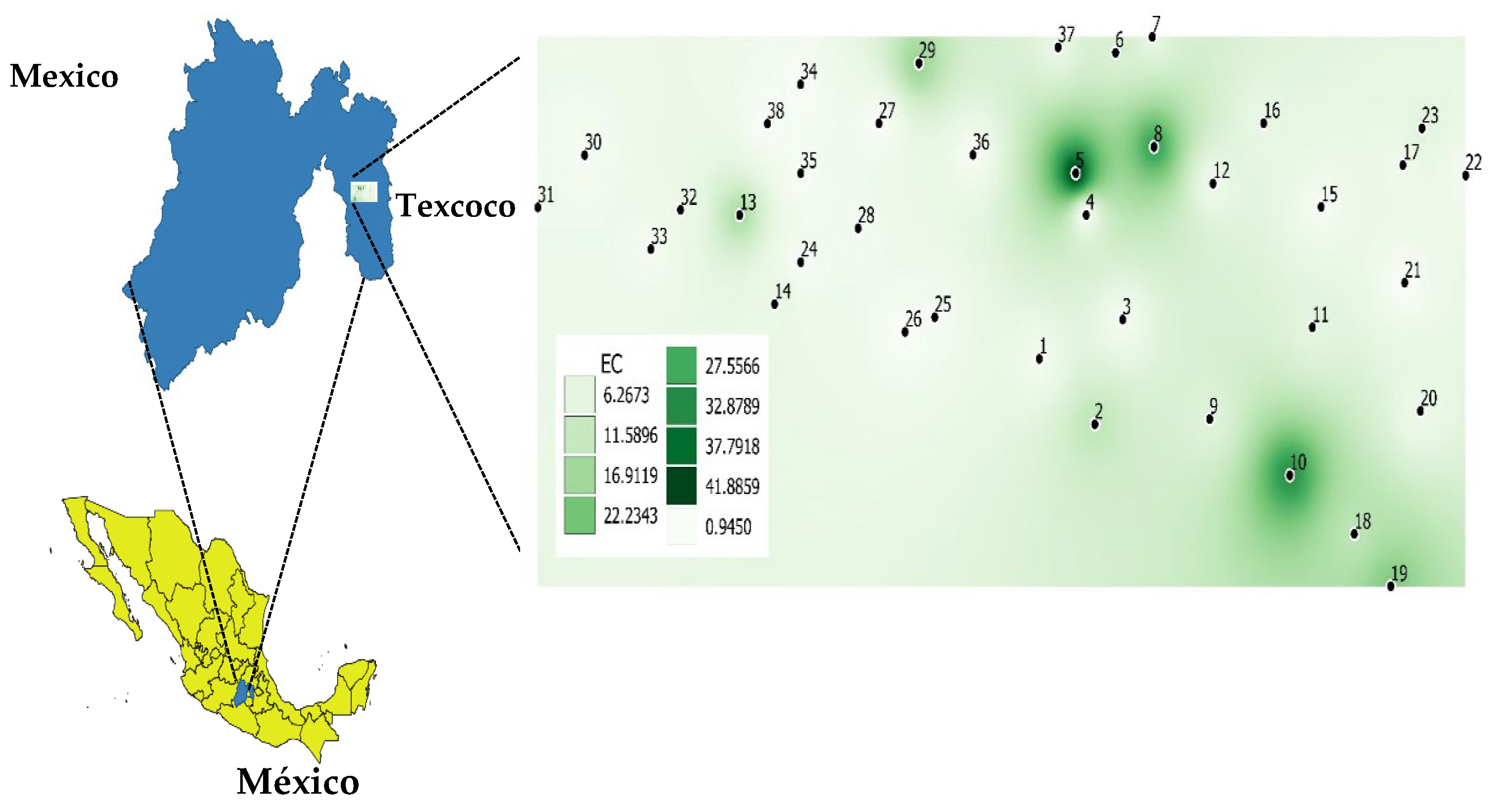


| Consortium | # of Individual Soil Samples † | Cations Concentration (mM) †† | EC (dS m−1) †† | pH †† | SAR (mM) †† | Soil Type | PO4−2 Olsen (mg kg−1) †† | Vegetal Species | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Na | K | Ca | Mg | S | SS | |||||||
| 1 | 5 | 655.9 | 60.2 | 2.0 | 0.7 | 42.5 | 7.8 | 580.4 | + | 11.0 | Distichlis spicata | |
| 2 | 8, 10 | 585.3 | 70.8 | 5.1 | 1.4 | 36.3 | 8.1 | 325.1 | + | 15.5 | Distichlis spicata | |
| 3 | 19 | 322.9 | 39.3 | 3.3 | 0.8 | 20.3 | 8.1 | 225.1 | + | 12.2 | Kochia scoparia | |
| 4 | 29 | 272.5 | 15.2 | 3.0 | 0.5 | 17.5 | 9.2 | 206.4 | + | 15.1 | Eragrostis obtusiflora | |
| 5 | 13 | 222.0 | 18.3 | 3.3 | 0.8 | 15.4 | 8.3 | 154.0 | + | 13.9 | Distichlis spicata | |
| 6 | 2, 18 | 212.0 | 24.7 | 3.3 | 2.4 | 14.8 | 8.5 | 213.6 | + | 15.7 | Suaeda torreyana | |
| 7 | 6 | 94.9 | 12.6 | 2.3 | 1.5 | 8.5 | 8.1 | 69.2 | + | 13.3 | Distichlis spicata | |
| 8 | 9, 11 | 131.2 | 18.6 | 4.4 | 3.1 | 9.1 | 8.4 | 67.2 | + | 16.5 | Baccharis salicifolia | |
| 9 | 20, 23 | 52.4 | 16.8 | 8.2 | 2.1 | 8.7 | 8.6 | 23.0 | + | 13.0 | Distichlis spicata | |
| 10 | 7 | 80.8 | 9.1 | 3.9 | 1.6 | 6.0 | 9.4 | 49.0 | + | 17.4 | Kochia scoparia | |
| 11 | 12, 14, 16 | 181.6 | 12.3 | 5.7 | 3.2 | 6.4 | 9.6 | 86.5 | + | 15.0 | Eragrostis obtusiflora | |
| 12 | 32, 37 | 212.0 | 12.6 | 3.1 | 1.7 | 6.6 | 8.2 | 135.2 | + | 6.5 | Eragrostis obtusiflora | |
| 13 | 17 | 50.5 | 9.1 | 3.9 | 1.0 | 3.8 | 8.5 | 32.5 | + | 8.1 | Cynodon dactylon | |
| 14 | 4 | 52.5 | 8.1 | 4.9 | 1.4 | 3.1 | 8.7 | 29.6 | + | 10.9 | Baccharis salicifolia | |
| 15 | 3 | 131.1 | 8.1 | 6.0 | 1.4 | 2.6 | 8.7 | 68.3 | + | 16.1 | Kochia scoparia | |
| 16 | 21, 22 | 38.3 | 7.3 | 3.9 | 3.0 | 2.2 | 8.4 | 20.6 | + | 10.8 | Distichlis spicata | |
| 17 | 1, 27, 28, 30, 31, 33 | 141.3 | 3.4 | 3.1 | 1.2 | 3.8 | 7.9 | 96.8 | + | 7.2 | Eragrostis obtusiflora | |
| 18 | 15, 34, 35, 36, 38 | 31.3 | 6.3 | 6.3 | 3.3 | 2.0 | 8.0 | 14.3 | + | 8.8 | Baccharis salicifolia | |
| 19 | 24, 25 | 31.3 | 6.0 | 5.2 | 1.6 | 0.9 | 8.4 | 17.0 | + | 12.1 | Kochia scoparia | |
| Normal in the soil solution | 0.2 ††† | 20 †††† | 0.1 ‡ | 0.12–8.5 ‡‡ | ||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barajas González, J.A.; Carrillo-González, R.; González-Chávez, M.d.C.A.; Chimal Sánchez, E.; Tapia Maruri, D. Selection of Salinity-Adapted Endorhizal Fungal Consortia from Two Inoculum Sources and Six Halophyte Plants. J. Fungi 2023, 9, 893. https://doi.org/10.3390/jof9090893
Barajas González JA, Carrillo-González R, González-Chávez MdCA, Chimal Sánchez E, Tapia Maruri D. Selection of Salinity-Adapted Endorhizal Fungal Consortia from Two Inoculum Sources and Six Halophyte Plants. Journal of Fungi. 2023; 9(9):893. https://doi.org/10.3390/jof9090893
Chicago/Turabian StyleBarajas González, Jesús Adrián, Rogelio Carrillo-González, Ma. del Carmen Angeles González-Chávez, Eduardo Chimal Sánchez, and Daniel Tapia Maruri. 2023. "Selection of Salinity-Adapted Endorhizal Fungal Consortia from Two Inoculum Sources and Six Halophyte Plants" Journal of Fungi 9, no. 9: 893. https://doi.org/10.3390/jof9090893
APA StyleBarajas González, J. A., Carrillo-González, R., González-Chávez, M. d. C. A., Chimal Sánchez, E., & Tapia Maruri, D. (2023). Selection of Salinity-Adapted Endorhizal Fungal Consortia from Two Inoculum Sources and Six Halophyte Plants. Journal of Fungi, 9(9), 893. https://doi.org/10.3390/jof9090893






