Biogeography of Fungal Communities Associated with Pinus sylvestris L. and Picea abies (L.) H. Karst. along the Latitudinal Gradient in Europe
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites and Sampling
2.2. Molecular Analyses
2.3. Bioinformatics
2.4. Statistical Analyses
2.4.1. Fungal Community Richness, Diversity, and Structure
2.4.2. Climate Variables and Selection of Model for Future Predictions
3. Results
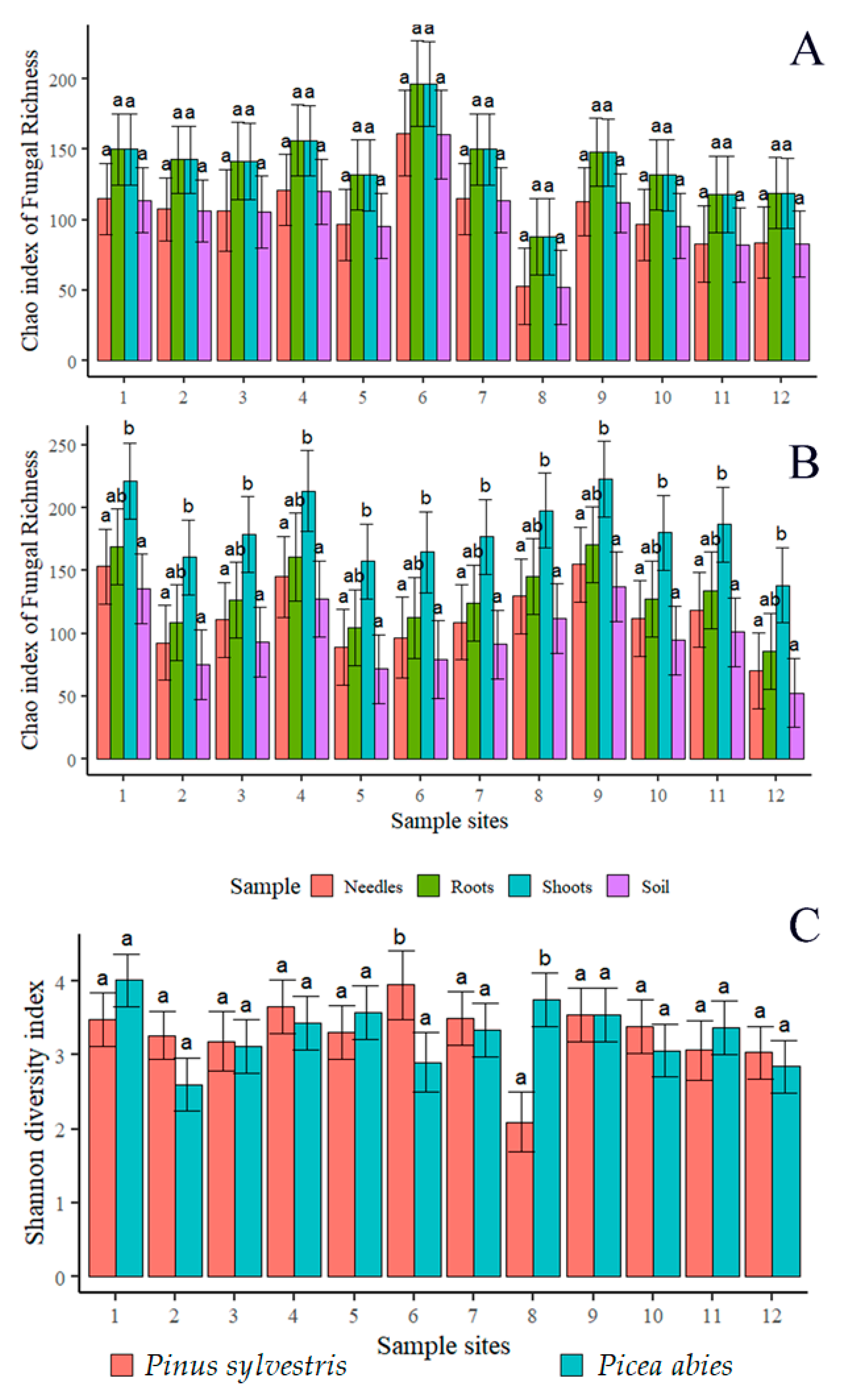
3.1. Sequence Quality and Fungal Diversity
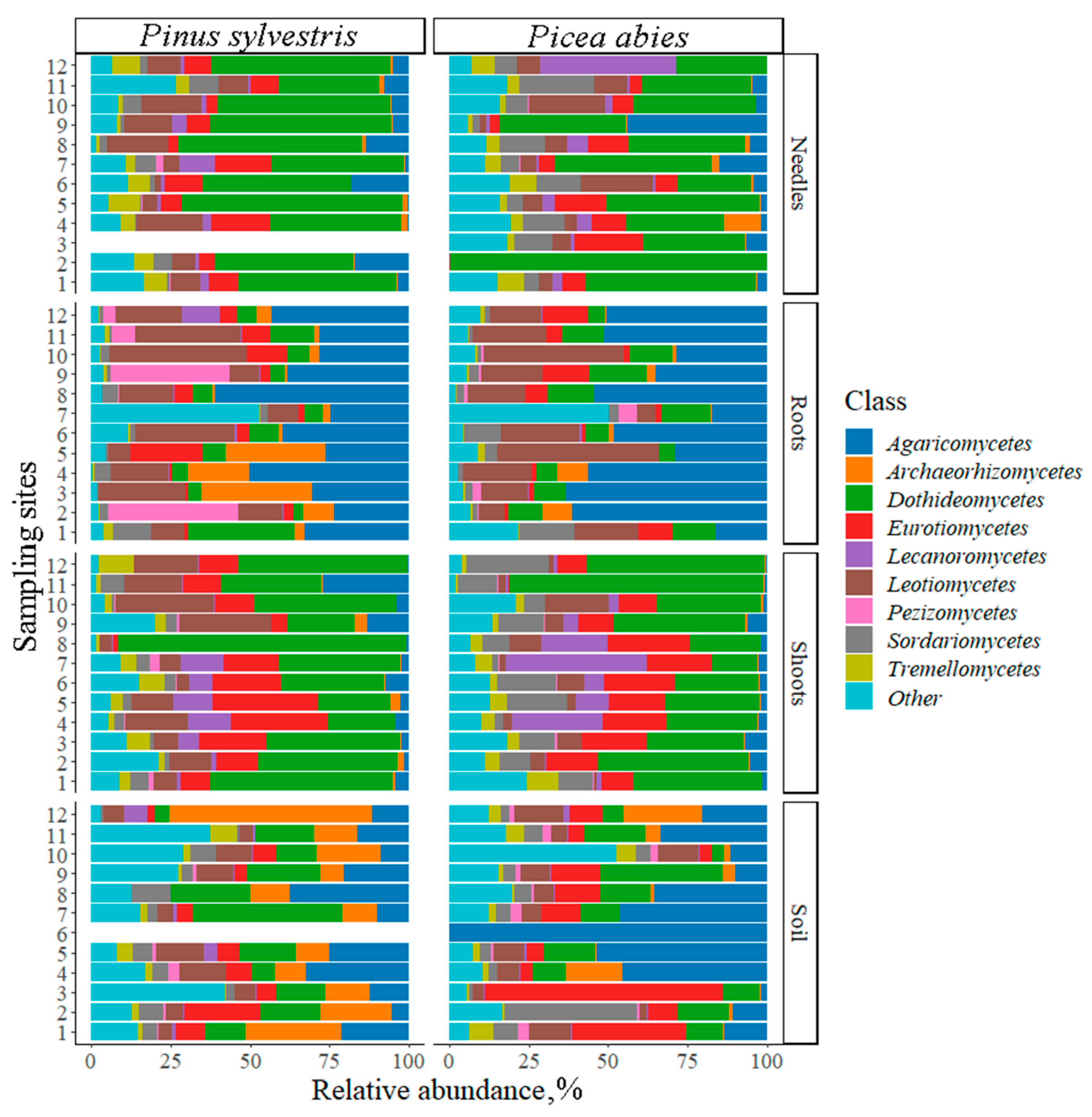
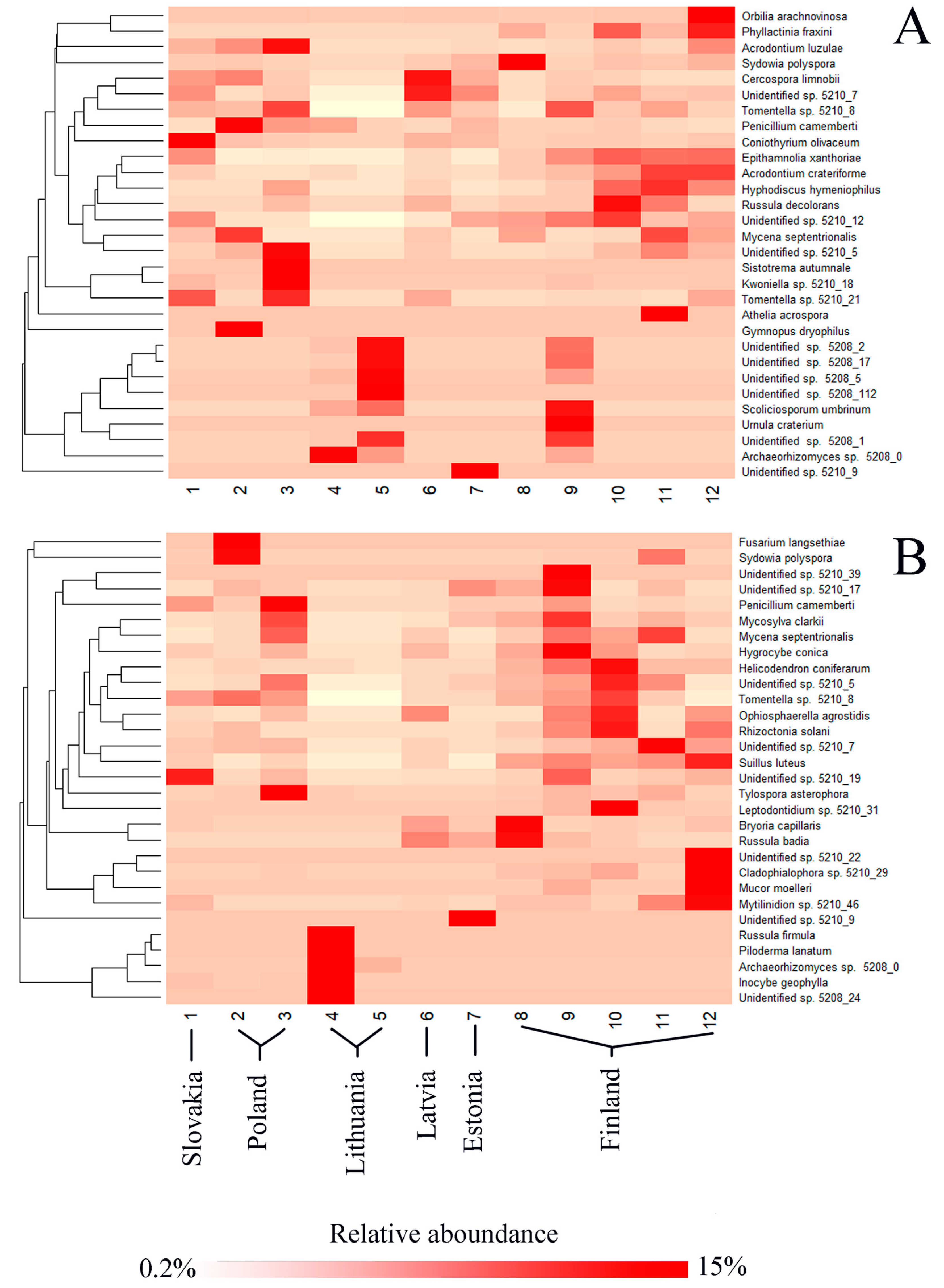
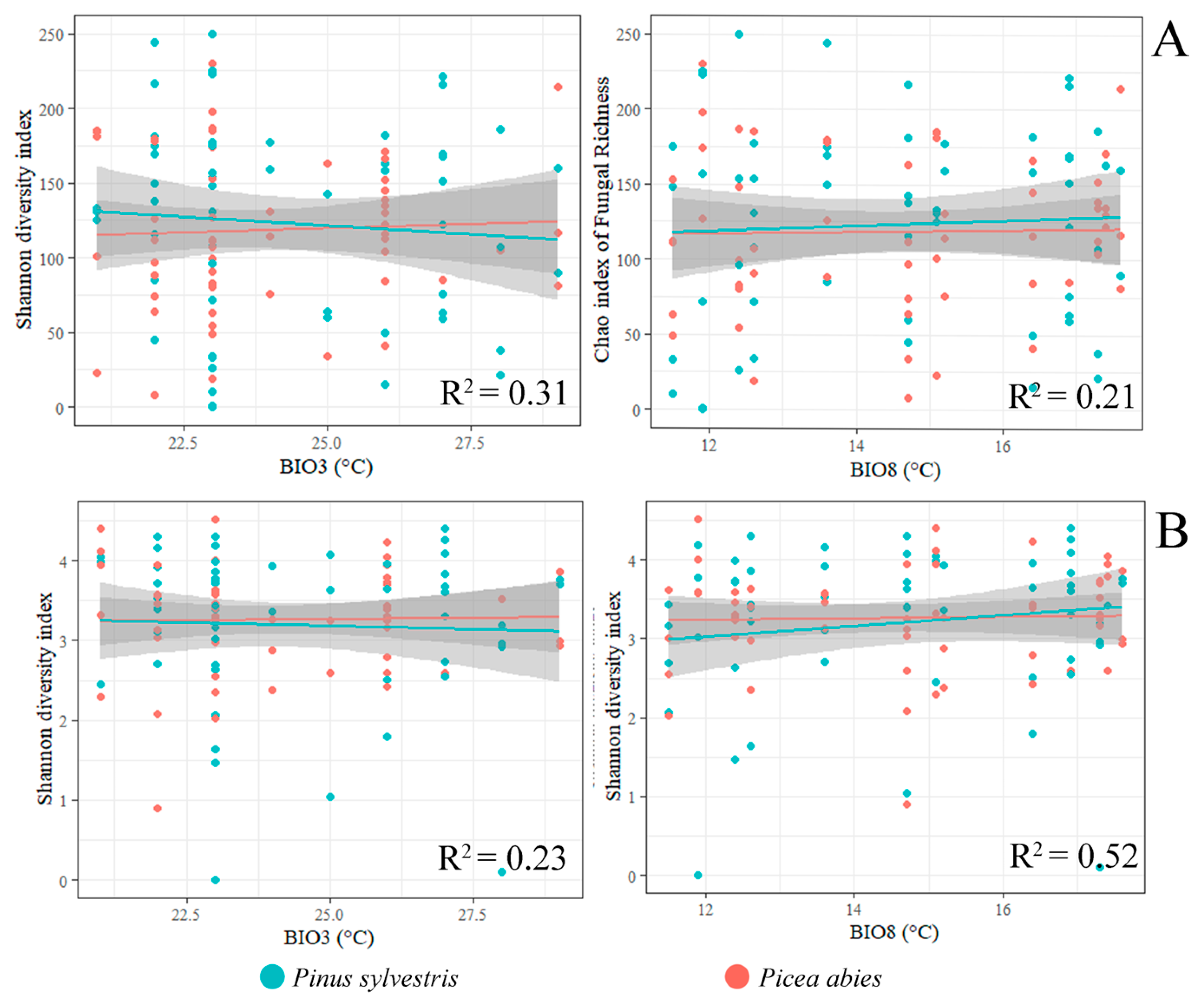
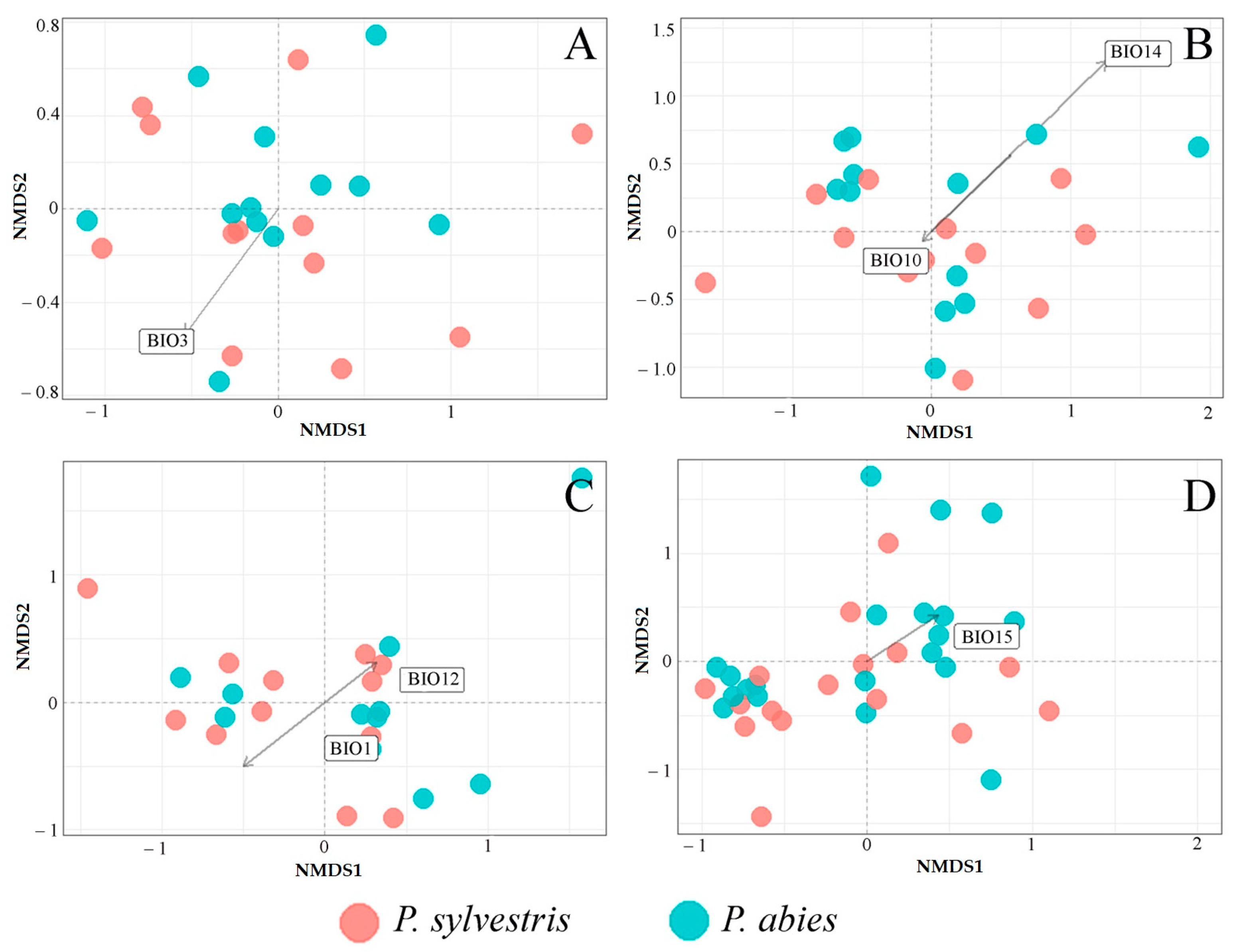
3.2. Fungal Communities and Bioclimatic Factors Explaining the Community Structure
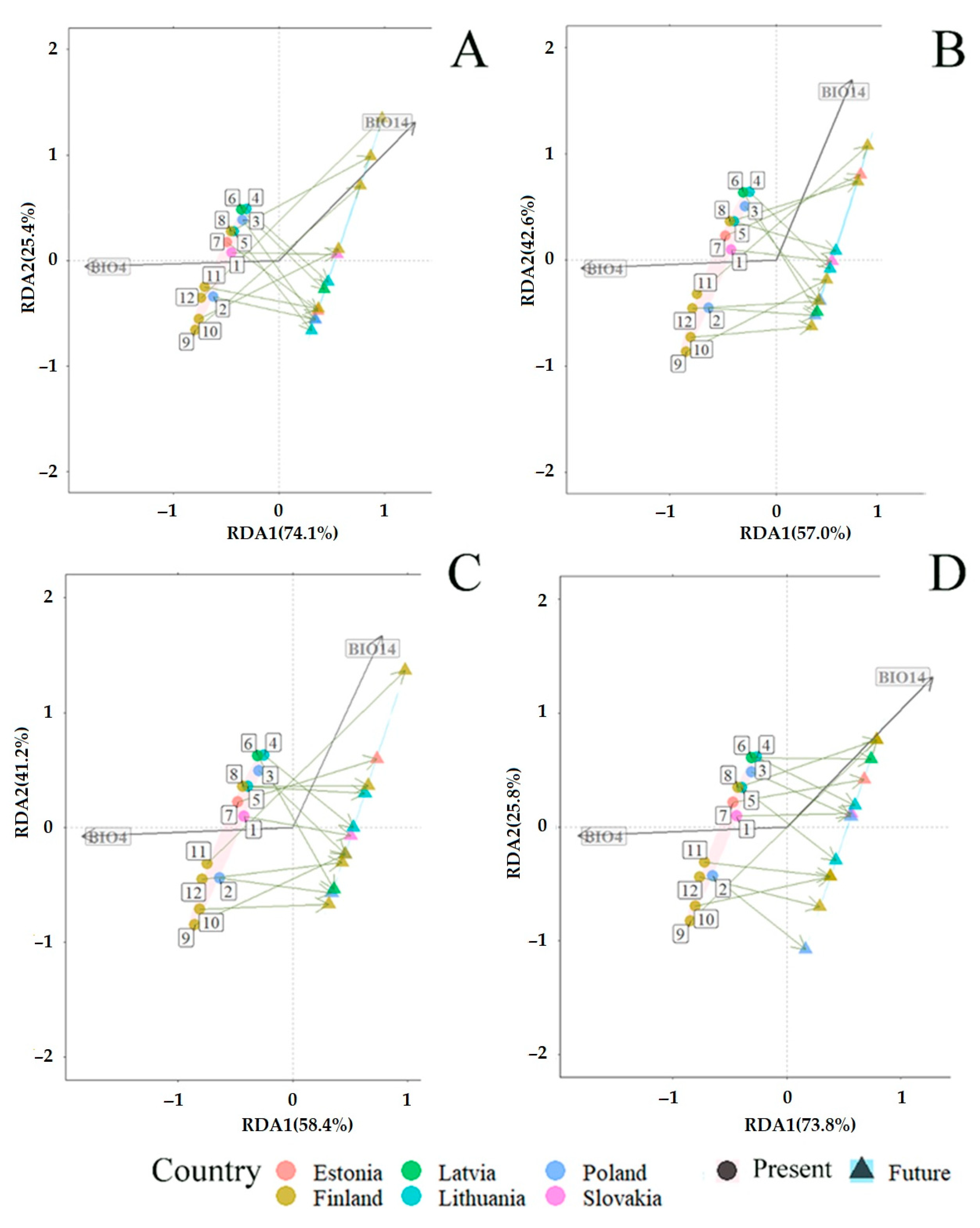
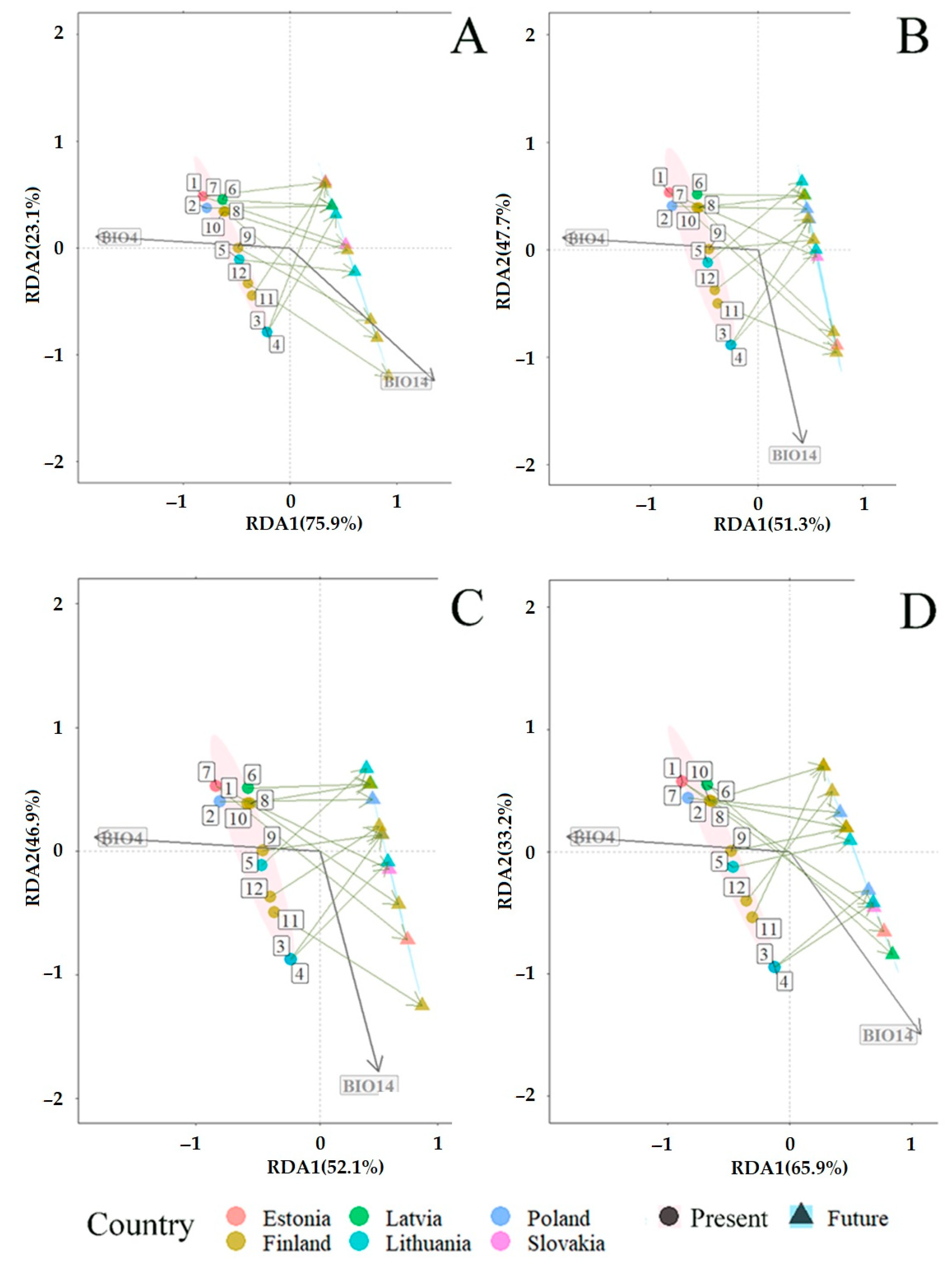
3.3. Projected Shifts in Fungal Communities under the CMIP6 Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mauri, A.; Girardello, M.; Strona, G.; Beck, P.S.A.; Forzieri, G.; Caudullo, G.; Manca, F.; Cescatti, A. EU-Trees4F, a Dataset on the Future Distribution of European Tree Species. Sci. Data 2022, 9, 37. [Google Scholar] [CrossRef] [PubMed]
- De Rigo, D.; San-Miguel-Ayanz, J.; Caudullo, G.; Durrant, T.H.; Mauri, A. (Eds.) European Atlas of Forest Tree Species; Publications Office of the European Union: Luxembourg, 2016; ISBN 978-92-79-36740-3. [Google Scholar]
- Bottero, A.; Forrester, D.I.; Cailleret, M.; Kohnle, U.; Gessler, A.; Michel, D.; Bose, A.K.; Bauhus, J.; Bugmann, H.; Cuntz, M.; et al. Growth Resistance and Resilience of Mixed Silver Fir and Norway Spruce Forests in Central Europe: Contrasting Responses to Mild and Severe Droughts. Glob. Chang. Biol. 2021, 27, 4403–4419. [Google Scholar] [CrossRef]
- Durrant, T.H.; De Rigo, D.; Caudullo, G. Pinus sylvestris in Europe: Distribution, habitat, usage and threats. Eur. Atlas For. Tree Species 2016, 14, 845–846. [Google Scholar]
- Dering, M.; Baranowska, M.; Beridze, B.; Chybicki, I.J.; Danelia, I.; Iszkuło, G.; Kvartskhava, G.; Kosiński, P.; Rączka, G.; Thomas, P.A.; et al. The evolutionary heritage and ecological uniqueness of Scots pine in the Caucasus ecoregion is at risk of climate changes. Sci. Rep. 2011, 11, 22845. [Google Scholar] [CrossRef] [PubMed]
- Kalbarczyk, R.; Ziemiańska, M.; Nieróbca, A.; Dobrzańska, J. The Impact of Climate Change and Strong Anthropopressure on the Annual Growth of Scots Pine (Pinus sylvestris L.) Wood Growing in Eastern Poland. Forests 2018, 9, 661. [Google Scholar] [CrossRef]
- Tabakova, M.A.; Arzac, A.; Martínez, E.; Kirdyanov, A.V. Climatic Factors Controlling Pinus Sylvestris Radial Growth along a Transect of Increasing Continentality in Southern Siberia. Dendrochronologia 2020, 62, 125709. [Google Scholar] [CrossRef]
- Illés, G.; Móricz, N. Climate envelope analyses suggests significant rearrangements in the distribution ranges of Central European tree species. Ann. For. Sci. 2022, 79, 35. [Google Scholar] [CrossRef]
- Sladonja, B.; Poljuha, D.; Uzelac, M. Non-Native Invasive Species as Ecosystem Service Providers. In Ecosystem Services and Global Ecology; Hufnagel, L., Ed.; IntechOpen: London, UK, 2018; pp. 39–59. [Google Scholar] [CrossRef]
- Tyrmi, J.S.; Vuosku, J.; Acosta, J.J.; Li, Z.; Sterck, L.; Cervera, M.T.; Savolainen, O.; Pyhäjärvi, T. Genomics of Clinal Local Adaptation in Pinus Sylvestris under Continuous Environmental and Spatial Genetic Setting. Genomics 2019, 10, 2683–2696. [Google Scholar] [CrossRef]
- Jamnická, G.; Fleischer, P.; Konôpková, A.; Pšidová, E.; Kučerová, J.; Kurjak, D.; Živčák, M.; Ditmarová, Ľ. Norway Spruce (Picea abies L.) Provenances Use Different Physiological Strategies to Cope with Water Deficit. Forests 2019, 10, 651. [Google Scholar] [CrossRef]
- Spiecker, H. Silvicultural Management in Maintaining Biodiversity and Resistance of Forests in Europe—Temperate Zone. J. Environ. Manag. 2003, 67, 55–65. [Google Scholar] [CrossRef]
- Netherer, S.; Kandasamy, D.; Jirosová, A.; Kalinová, B.; Schebeck, M.; Schlyter, F. Interactions among Norway Spruce, the Bark Beetle Ips Typographus and Its Fungal Symbionts in Times of Drought. J. Pest. Sci. 2021, 94, 591–614. [Google Scholar] [CrossRef]
- Lindner, M.; Maroschek, M.; Netherer, S.; Kremer, A.; Barbati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolström, M.; et al. Climate Change Impacts, Adaptive Capacity, and Vulnerability of European Forest Ecosystems. For. Ecol. Manag. 2010, 259, 698–709. [Google Scholar] [CrossRef]
- Vimal, S.R.; Singh, J.S.; Arora, N.K.; Singh, S. Soil-Plant-Microbe Interactions in Stressed Agriculture Management: A Review. Pedosphere 2017, 27, 177–192. [Google Scholar] [CrossRef]
- Andreote, F.D.; e Silva, M.D.C.P. Microbial Communities Associated with Plants: Learning from Nature to Apply It in Agriculture. Curr. Opin. Microbiol. 2017, 37, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S.; Lata, C. Heavy Metal Stress, Signaling, and Tolerance Due to Plant-Associated Microbes: An Overview. Front. Plant Sci. 2018, 9, 452. [Google Scholar] [CrossRef] [PubMed]
- Compant, S.; Samad, A.; Faist, H.; Sessitsch, A. A Review on the Plant Microbiome: Ecology, Functions, and Emerging Trends in Microbial Application. J. Adv. Res. 2019, 19, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Qu, M.; Pu, X.; Lin, J.; Shu, B. Distinct microbial communities among different tissues of citrus tree Citrus reticulata cv. Chachiensis. Sci. Rep. 2020, 10, 6068. [Google Scholar] [CrossRef] [PubMed]
- Turner, T.R.; James, E.K.; Poole, P.S. The plant microbiome. Genome Biol. 2013, 14, 209. [Google Scholar] [CrossRef]
- Vandenkoornhuyse, P.; Quaiser, A.; Duhamel, M.; Le Van, A.; Dufresne, A. The Importance of the Microbiome of the Plant Holobiont. New Phytol. 2015, 206, 1196–1206. [Google Scholar] [CrossRef]
- Gehring, C.; Sthultz, C.; Flores-Rentería, L.; Whipple, A.; Whitham, T. Tree genetics defines fungal partner communities that may confer drought tolerance. Proc. Natl. Acad. Sci. USA 2017, 114, 11169–11174. [Google Scholar] [CrossRef]
- Allsup, C.M.; George, I.; Lankau, R.A. Shifting microbial communities can enhance tree tolerance to changing climates. Science 2023, 380, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Adair, K.L.; Lindgreen, S.; Poole, A.M.; Young, L.M.; Bernard-Verdier, M.; Wardle, D.A.; Tylianakis, J.M. Above and belowground community strategies respond to different global change drivers. Sci. Rep. 2019, 9, 2540. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhu, K.; Wurzburger, N.; Zhang, J. Relationships between plant diversity and soil microbial diversity vary across taxonomic groups and spatial scales. Ecosphere 2020, 11, e02999. [Google Scholar] [CrossRef]
- Kaul, S.; Sharma, T.K.; Dhar, M. “Omics” Tools for Better Understanding the Plant–Endophyte Interactions. Front. Plant Sci. 2016, 7, 955. [Google Scholar] [CrossRef]
- Busby, P.E.; Soman, C.; Wagner, M.R.; Friesen, M.L.; Kremer, J.; Bennett, A.; Morsy, M.; Eisen, J.A.; Leach, J.E.; Dangl, J.L. Research Priorities for Harnessing Plant Microbiomes in Sustainable Agriculture. PLoS Biol. 2017, 15, e2001793. [Google Scholar] [CrossRef] [PubMed]
- Terhonen, E.; Blumenstein, K.; Kovalchuk, A.; Asiegbu, F.O. Forest Tree Microbiomes and Associated Fungal Endophytes: Functional Roles and Impact on Forest Health. Forests 2019, 10, 42. [Google Scholar] [CrossRef]
- Contreras-Cornejo, H.A.; Macías-Rodríguez, L.; del-Val, E.; Larsen, J. The Root Endophytic Fungus Trichoderma Atroviride Induces Foliar Herbivory Resistance in Maize Plants. Appl. Soil Ecol. 2018, 124, 45–53. [Google Scholar] [CrossRef]
- Bano, A.; Hussain, J.; Akbar, A.; Mehmood, K.; Anwar, M.; Hasni, M.S.; Ullah, S.; Sajid, S.; Ali, I. Biosorption of Heavy Metals by Obligate Halophilic Fungi. Chemosphere 2018, 199, 218–222. [Google Scholar] [CrossRef]
- Rajamani, T.; Suryanarayanan, T.S.; Murali, T.S.; Thirunavukkarasu, N. Distribution and Diversity of Foliar Endophytic Fungi in the Mangroves of Andaman Islands, India. Fungal Ecol. 2018, 36, 109–116. [Google Scholar] [CrossRef]
- Carroll, G. Fungal Endophytes in Stems and Leaves: From Latent Pathogen to Mutualistic Symbiont. Ecology 1988, 69, 2–9. [Google Scholar] [CrossRef]
- Migliorini, D.; Ghelardini, L.; Tondini, E.; Luchi, N.; Santini, A. The Potential of Symptomless Potted Plants for Carrying Invasive Soilborne Plant Pathogens. Divers. Distrib. 2015, 21, 1218–1229. [Google Scholar] [CrossRef]
- Fang, K.; Miao, Y.-F.; Chen, L.; Zhou, J.; Yang, Z.-P.; Dong, X.-F.; Zhang, H.-B. Tissue-Specific and Geographical Variation in Endophytic Fungi of Ageratina Adenophora and Fungal Associations with the Environment. Front. Microbiol. 2019, 10, 2919. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Wang, X.; Cao, P.; Gao, Z.; Chen, A.J.; Han, J. Microbial Community Changes in the Rhizosphere Soil of Healthy and Rusty Panax Ginseng and Discovery of Pivotal Fungal Genera Associated with Rusty Roots. BioMed Res. Int. 2020, 2020, 8018525. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.H.; Song, Z.; Bates, S.T.; Branco, S.; Tedersoo, L.; Menke, J.; Schilling, J.S.; Kennedy, P.G. FUNGuild: An Open Annotation Tool for Parsing Fungal Community Datasets by Ecological Guild. Fungal Ecol. 2016, 20, 241–248. [Google Scholar] [CrossRef]
- U’Ren, J.M.; Lutzoni, F.; Miadlikowska, J.; Zimmerman, N.B.; Carbone, I.; May, G.; Arnold, A.E. Host Availability Drives Distributions of Fungal Endophytes in the Imperilled Boreal Realm. Nat. Ecol. Evol. 2019, 3, 1430–1437. [Google Scholar] [CrossRef]
- Oita, S.; Ibáñez, A.; Lutzoni, F.; Miadlikowska, J.; Geml, J.; Lewis, L.A.; Hom, E.F.Y.; Carbone, I.; U’Ren, J.M.; Arnold, A.E. Climate and Seasonality Drive the Richness and Composition of Tropical Fungal Endophytes at a Landscape Scale. Commun. Biol. 2021, 4, 313. [Google Scholar] [CrossRef]
- Suryanarayanan, T.S.; Shaanker, R.U. Can Fungal Endophytes Fast-Track Plant Adaptations to Climate Change? Fungal Ecol. 2021, 50, 101039. [Google Scholar] [CrossRef]
- Naylor, D.; Sadler, N.; Bhattacharjee, A.; Graham, E.B.; Anderton, C.R.; McClure, R.; Lipton, M.; Hofmockel, K.S.; Jansson, J.K. Soil Microbiomes Under Climate Change and Implications for Carbon Cycling. Annu. Rev. Environ. Resour. 2020, 45, 29–59. [Google Scholar] [CrossRef]
- Chanda, A.; Maghrawy, H.; Sayour, H.; Gummadidala, P.M.; Gomaa, O.M. Impact of Climate Change on Plant-Associated Fungi. In Climate Change Impacts on Agriculture and Food Security in Egypt; Ewis Omran, E.-S., Negm, A.M., Eds.; Springer Water; Springer International Publishing: Cham, Switzerland, 2020; pp. 83–96. ISBN 978-3-030-41628-7. [Google Scholar]
- von Tiedemann, A.; Weigel, H.J.; Jäger, H.J. Effects of Open-Top Chamber Fumigations with Ozone on Three Fungal Leaf Diseases of Wheat and the Mycoflora of the Phyllosphere. Environ. Pollut. 1991, 72, 205–224. [Google Scholar] [CrossRef]
- Manning, W.J.; V Tiedemann, A. Climate Change: Potential Effects of Increased Atmospheric Carbon Dioxide (CO2), Ozone (O3), and Ultraviolet-B (UV-B) Radiation on Plant Diseases. Environ. Pollut. 1995, 88, 219–245. [Google Scholar] [CrossRef]
- Sandermann, H. Ozone/Biotic Disease Interactions: Molecular Biomarkers as a New Experimental Tool. Environ. Pollut. 2000, 108, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Nickstadt, A.; Thomma, B.P.H.J.; Feussner, I.; Kangasjarvi, J.; Zeier, J.; Loeffler, C.; Scheel, D.; Berger, S. The Jasmonate-Insensitive Mutant Jin1 Shows Increased Resistance to Biotrophic as Well as Necrotrophic Pathogens. Mol. Plant. Pathol. 2004, 5, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Andrade-Linares, D.R.; Veresoglou, S.D.; Rillig, M.C. Temperature Priming and Memory in Soil Filamentous Fungi. Fungal Ecol. 2016, 21, 10–15. [Google Scholar] [CrossRef]
- Kinlock, N.L.; Prowant, L.; Herstoff, E.M.; Foley, C.M.; Akin-Fajiye, M.; Bender, N.; Umarani, M.; Ryu, H.Y.; Şen, B.; Gurevitch, J. Explaining Global Variation in the Latitudinal Diversity Gradient: Meta-Analysis Confirms Known Patterns and Uncovers New Ones. Glob. Ecol. Biogeogr. 2018, 27, 125–141. [Google Scholar] [CrossRef]
- Duffy, K.J.; Waud, M.; Schatz, B.; Petanidou, T.; Jacquemyn, H. Latitudinal Variation in Mycorrhizal Diversity Associated with a European Orchid. J. Biogeogr. 2019, 46, 968–980. [Google Scholar] [CrossRef]
- Gaytán, Á.; Abdelfattah, A.; Faticov, M.; Moreira, X.; Castagneyrol, B.; Van Halder, I.; De Frenne, P.; Meeussen, C.; Timmermans, B.G.H.; Ten Hoopen, J.P.J.G.; et al. Changes in the Foliar Fungal Community between Oak Leaf Flushes along a Latitudinal Gradient in Europe. J. Biogeogr. 2022, 49, 2269–2280. [Google Scholar] [CrossRef]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km Spatial Resolution Climate Surfaces for Global Land Areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Buivydaitė, V.; Vaičys, M. Kn. 32: Lietuvos dirvožemiai. In Naujoji Lietuvos Dirvožemių Klasifikacija; Lietuvos mokslas: Tęstinis, serijinis mokslo darbų leidinys Lietuvai ir pasauliui lietuvių ir anglų kalbomis; Lietuvos mokslas: Vilnius, Lithuania, 2001. (In Lithuanian) [Google Scholar]
- Karazija, S. Eglės Pomiškio Atsiradimo Dėsningumai. Miškininkystė 1998, 41, 26–33. [Google Scholar]
- Rosling, A.; Landeweert, R.; Lindahl, B.D.; Larsson, K.-H.; Kuyper, T.W.; Taylor, A.F.S.; Finlay, R.D. Vertical Distribution of Ectomycorrhizal Fungal Taxa in a Podzol Soil Profile. New Phytol. 2003, 159, 775–783. [Google Scholar] [CrossRef]
- Ihrmark, K.; Bödeker, I.T.M.; Cruz-Martinez, K.; Friberg, H.; Kubartova, A.; Schenck, J.; Strid, Y.; Stenlid, J.; Brandström-Durling, M.; Clemmensen, K.E.; et al. New Primers to Amplify the Fungal ITS2 Region—Evaluation by 454-Sequencing of Artificial and Natural Communities. FEMS Microbiol. Ecol. 2012, 82, 666–677. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols; Elsevier: Amsterdam, The Netherlands, 1990; pp. 315–322. ISBN 978-0-12-372180-8. [Google Scholar]
- Kõljalg, U.; Nilsson, H.R.; Schigel, D.; Tedersoo, L.; Larsson, K.-H.; May, T.W.; Taylor, A.F.S.; Jeppesen, T.S.; Frøslev, T.G.; Lindahl, B.D.; et al. The Taxon Hypothesis Paradigm—On the Unambiguous Detection and Communication of Taxa. Microorganisms 2020, 8, 1910. [Google Scholar] [CrossRef]
- R: A Language and Environment for Statistical Computing. In R Foundation for Statistical Computing; R Core Team: Vienna, Austria, 2020.
- Hsieh, T.C.; Ma, K.H.; Chao, A. INEXT: An R Package for Rarefaction and Extrapolation of Species Diversity (Hill Numbers). Methods Ecol. Evol. 2016, 7, 1451–1456. [Google Scholar] [CrossRef]
- Cortez, P.; Cerdeira, A.; Almeida, F.; Matos, T.; Reis, J. Modeling Wine Preferences by Data Mining from Physicochemical Properties. Decis. Support Syst. 2009, 47, 547–553. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, B.; Simpson, G.; Solymos, P.; Stevens, H.; Wagner, H. Vegan: Community Ecology Package, R Package Version 2.5–3. 2018. Available online: https://cran.r-project.org (accessed on 28 November 2021).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis, 2nd ed.; Springer International Publishing: New York, NY, USA, 2016; ISBN 978-3-319-24275-0. [Google Scholar]
- Kindt, R. AlleleShift: An R Package to Predict and Visualize Population-Level Changes in Allele Frequencies in Response to Climate Change. PeerJ 2021, 9, e11534. [Google Scholar] [CrossRef]
- Martin-Benito, D.; Pederson, N.; Köse, N.; Doğan, M.; Bugmann, H.; Mosulishvili, M.; Bigler, C. Pervasive Effects of Drought on Tree Growth across a Wide Climatic Gradient in the Temperate Forests of the Caucasus. Glob. Ecol. Biogeogr. 2018, 27, 1314–1325. [Google Scholar] [CrossRef]
- Wang, S.; Bao, X.; Feng, K.; Deng, Y.; Zhou, W.; Shao, P.; Zheng, T.; Yao, F.; Yang, S.; Liu, S.; et al. Warming-Driven Migration of Core Microbiota Indicates Soil Property Changes at Continental Scale. Sci. Bull. 2021, 66, 2025–2035. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Chen, L.; Ji, N.; Wang, Y.; Gao, C.; Jin, S.; Hu, H.; Huang, Z.; He, J.; Guo, L.; et al. Assembly Processes Lead to Divergent Soil Fungal Communities within and among 12 Forest Ecosystems along a Latitudinal Gradient. New Phytol. 2021, 231, 1183–1194. [Google Scholar] [CrossRef] [PubMed]
- Tedersoo, L.; Bahram, M.; Cajthaml, T.; Põlme, S.; Hiiesalu, I.; Anslan, S.; Harend, H.; Buegger, F.; Pritsch, K.; Koricheva, J.; et al. Tree Diversity and Species Identity Effects on Soil Fungi, Protists and Animals Are Context Dependent. ISME J. 2016, 10, 346–362. [Google Scholar] [CrossRef]
- Liu, J.; Sui, Y.; Yu, Z.; Shi, Y.; Chu, H.; Jin, J.; Liu, X.; Wang, G. Soil Carbon Content Drives the Biogeographical Distribution of Fungal Communities in the Black Soil Zone of Northeast China. Soil Biol. Biochem. 2015, 83, 29–39. [Google Scholar] [CrossRef]
- Shi, L.-L.; Mortimer, P.E.; Ferry Slik, J.W.; Zou, X.-M.; Xu, J.; Feng, W.-T.; Qiao, L. Variation in Forest Soil Fungal Diversity along a Latitudinal Gradient. Fungal Divers. 2014, 64, 305–315. [Google Scholar] [CrossRef]
- Sokolski, M.; Zymliński, R.; Biegus, J.; Siwołowski, P.; Nawrocka-Millward, S.; Todd, J.; Yerramilli, M.R.; Estis, J.; Jankowska, E.A.; Banasiak, W.; et al. Urinary Levels of Novel Kidney Biomarkers and Risk of True Worsening Renal Function and Mortality in Patients with Acute Heart Failure. Eur. J. Heart Fail. 2017, 19, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Millberg, H.; Boberg, J.; Stenlid, J. Changes in Fungal Community of Scots Pine (Pinus sylvestris) Needles along a Latitudinal Gradient in Sweden. Fungal Ecol. 2015, 17, 126–139. [Google Scholar] [CrossRef]
- Allen, W.J.; DeVries, A.E.; Bologna, N.J.; Bickford, W.A.; Kowalski, K.P.; Meyerson, L.A.; Cronin, J.T. Intraspecific and Biogeographical Variation in Foliar Fungal Communities and Pathogen Damage of Native and Invasive Phragmites Australis. Glob. Ecol. Biogeogr. 2020, 29, 1199–1211. [Google Scholar] [CrossRef]
- Moler, E.R.V.; Aho, K. Whitebark Pine Foliar Fungal Endophyte Communities in the Southern Cascade Range, USA: Host Mycobiomes and White Pine Blister Rust. Fungal Ecol. 2018, 33, 104–114. [Google Scholar] [CrossRef]
- Bailes, G.; Thomas, D.; Bridgham, S.D.; Roy, B.A. Drivers of Grass Endophyte Communities in Prairies of the Pacific Northwest, USA. bioRxiv 2020. [Google Scholar] [CrossRef]
- Marčiulynienė, D.; Marčiulynas, A.; Mishcherikova, V.; Lynikienė, J.; Gedminas, A.; Franic, I.; Menkis, A. Principal Drivers of Fungal Communities Associated with Needles, Shoots, Roots and Adjacent Soil of Pinus Sylvestris. JoF 2022, 8, 1112. [Google Scholar] [CrossRef]
- O’Donnell, M.; Ignizio, D. Bioclimatic Predictors for Supporting Ecological Applications in the Conterminous United States. US Geol. Surv. Data Ser. 2012, 691, 4–9. [Google Scholar] [CrossRef]
- Talley, S.M.; Coley, P.D.; Kursar, T.A. Antifungal Leaf-Surface Metabolites Correlate with Fungal Abundance in Sagebrush Populations. J. Chem. Ecol. 2002, 28, 2141–2168. [Google Scholar] [CrossRef]
- Hawkins, B.A.; Field, R.; Cornell, H.V.; Currie, D.J.; Guégan, J.F.; Kaufman, D.M.; Kerr, J.T.; Mittelbach, G.G.; Oberdorff, T.; O’Brien, E.M.; et al. Turner Energy, water, and broad-scale geographic patterns of species richness. Ecology 2003, 84, 3105–3117. [Google Scholar] [CrossRef]
- Větrovský, T.; Kohout, P.; Kopecký, M.; Machac, A.; Man, M.; Bahnmann, B.D.; Brabcová, V.; Choi, J.; Meszárošová, L.; Human, Z.R.; et al. A Meta-Analysis of Global Fungal Distribution Reveals Climate-Driven Patterns. Nat. Commun. 2019, 10, 5142. [Google Scholar] [CrossRef]
- Hawkes, C.V.; Kivlin, S.N.; Rocca, J.D.; Huguet, V.; Thomsen, M.A.; Suttle, K.B. Fungal community responses to precipitation. Glob. Chang. Biol. 2010, 17, 1637–1645. [Google Scholar] [CrossRef]
- Chen, K.; Horton, R.M.; Bader, D.A.; Lesk, C.; Jiang, L.; Jones, B.; Zhou, L.; Chen, X.; Bi, J.; Kinney, P.L. Impact of Climate Change on Heat-Related Mortality in Jiangsu Province, China. Environ. Pollut. 2017, 224, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Praeg, N.; Schwinghammer, L.; Illmer, P. Larix decidua and Additional Light Affect the Methane Balance of Forest Soil and the Abundance of Methanogenic and Methanotrophic Microorganisms. FEMS Microbiol. Lett. 2019, 366, fnz259. [Google Scholar] [CrossRef] [PubMed]
- McMahon, S.M.; Harrison, S.P.; Armbruster, W.S.; Bartlein, P.J.; Beale, C.M.; Edwards, M.E.; Kattge, J.; Midgley, G.; Morin, X.; Prentice, I.C. Improving Assessment and Modelling of Climate Change Impacts on Global Terrestrial Biodiversity. Trends Ecol. Evol. 2011, 26, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Burrows, M.T.; Schoeman, D.S.; Buckley, L.B.; Moore, P.; Poloczanska, E.S.; Brander, K.M.; Brown, C.; Bruno, J.F.; Duarte, C.M.; Halpern, B.S.; et al. The Pace of Shifting Climate in Marine and Terrestrial Ecosystems. Science 2011, 334, 652–655. [Google Scholar] [CrossRef]
- Alkhalifah, D.H.M.; Damra, E.; Melhem, M.B.; Hozzein, W.N. Fungus under a Changing Climate: Modeling the Current and Future Global Distribution of Fusarium Oxysporum Using Geographical Information System Data. Microorganisms 2023, 11, 468. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Marotzke, J.; Bala, G.; Cao, L.; Corti, S.; Dunne, J.P.; Engelbrecht, F.; Fischer, E.; Fyfe, J.C.; Jones, C.; et al. Future Global Climate: Scenario-Based Projections and Near-Term Information; Masson-Delmotte, V., Zhai, P., Pirani, A., Connors, S.L., Pean, C., Berger, S., Caud, N., Chen, Y., Goldfarb, L., Gomis, M.I., et al., Eds.; IPCC: Genf, Switzerland, 2021; pp. 1–195. [Google Scholar] [CrossRef]
- Held, I.M.; Soden, B.J. Robust Responses of the Hydrological Cycle to Global Warming. J. Clim. 2006, 19, 5686–5699. [Google Scholar] [CrossRef]
- Silén, M.; Svantesson, M.; Kjellström, S.; Sidenvall, B.; Christensson, L. Moral Distress and Ethical Climate in a Swedish Nursing Context: Perceptions and Instrument Usability. J. Clin. Nurs. 2011, 20, 3483–3493. [Google Scholar] [CrossRef]
- Witt, M.J.; Hawkes, L.A.; Godfrey, M.H.; Godley, B.J.; Broderick, A.C. Predicting the Impacts of Climate Change on a Globally Distributed Species: The Case of the Loggerhead Turtle. J. Exp. Biol. 2010, 213, 901–911. [Google Scholar] [CrossRef]
- Abram, N.J.; Henley, B.J.; Sen Gupta, A.; Lippmann, T.J.R.; Clarke, H.; Dowdy, A.J.; Sharples, J.J.; Nolan, R.H.; Zhang, T.; Wooster, M.J.; et al. Connections of Climate Change and Variability to Large and Extreme Forest Fires in Southeast Australia. Commun. Earth Environ. 2021, 2, 8. [Google Scholar] [CrossRef]
- Du, Y.; Zhao, Q.; Chen, L.; Yao, X.; Zhang, W.; Zhang, B.; Xie, F. Effect of Drought Stress on Sugar Metabolism in Leaves and Roots of Soybean Seedlings. Plant Physiol. Biochem. 2020, 146, 1–12. [Google Scholar] [CrossRef]
- Agan, A.; Solheim, H.; Adamson, K.; Hietala, A.M.; Tedersoo, L.; Drenkhan, R. Seasonal Dynamics of Fungi Associated with Healthy and Diseased Pinus Sylvestris Needles in Northern Europe. Microorganisms 2021, 9, 1757. [Google Scholar] [CrossRef] [PubMed]
- Sanz-Ros, A.V.; Müller, M.M.; San Martín, R.; Diez, J.J. Fungal Endophytic Communities on Twigs of Fast and Slow Growing Scots Pine (Pinus sylvestris L.) in Northern Spain. Fungal Biol. 2015, 119, 870–883. [Google Scholar] [CrossRef] [PubMed]
- Zamora, P.; Martínez-Ruiz, C.; Diez, J. Fungi in Needles and Twigs of Pine Plantations from Northern Spain. Fungal Divers. 2008, 30, 171–184. [Google Scholar]
- Botella, L.; Santamaría, O.; Diez, J.J. Fungi Associated with the Decline of Pinus Halepensis in Spain. Fungal Divers. 2010, 40, 1–11. [Google Scholar] [CrossRef]
- Hyde, K.D.; Jones, E.B.G.; Liu, J.-K.; Ariyawansa, H.; Boehm, E.; Boonmee, S.; Braun, U.; Chomnunti, P.; Crous, P.W.; Dai, D.-Q.; et al. Families of Dothideomycetes: In Loving Memory of Majorie Phyllis Hyde (Affectionately Known as Mum or Marj), 29 August 1921–18 January 2013—Without Mum’s Determination, a Character Passed on to Children, This Treatise Would Never Have Been Completed—K.D. Hyde. Fungal Divers. 2013, 63, 1–313. [Google Scholar] [CrossRef]
- Taylor, J.W.; Hann-Soden, C.; Branco, S.; Sylvain, I.; Ellison, C.E. Clonal Reproduction in Fungi. Proc. Natl. Acad. Sci. USA 2015, 112, 8901–8908. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhao, F.; Wu, Y. Increased Response of Vegetation to Soil Moisture in the Northern Hemisphere Drylands. Front. Earth Sci. 2023, 11, 1144410. [Google Scholar] [CrossRef]
- Murgia, M.; Fiamma, M.; Barac, A.; Deligios, M.; Mazzarello, V.; Paglietti, B.; Cappuccinelli, P.; Al-Qahtani, A.; Squartini, A.; Rubino, S.; et al. Biodiversity of Fungi in Hot Desert Sands. MicrobiologyOpen 2019, 8, e00595. [Google Scholar] [CrossRef]
- Ruibal, C.; Gueidan, C.; Selbmann, L.; Gorbushina, A.A.; Crous, P.W.; Groenewald, J.Z.; Muggia, L.; Grube, M.; Isola, D.; Schoch, C.L.; et al. Phylogeny of Rock-Inhabiting Fungi Related to Dothideomycetes. Stud. Mycol. 2009, 64, 123–133. [Google Scholar] [CrossRef]
- Selbmann, L.; Egidi, E.; Isola, D.; Onofri, S.; Zucconi, L.; de Hoog, G.S.; Chinaglia, S.; Testa, L.; Tosi, S.; Balestrazzi, A.; et al. Biodiversity, Evolution and Adaptation of Fungi in Extreme Environments. Plant Biosyst.-An. Int. J. Deal. All Asp. Plant Biol. 2013, 147, 237–246. [Google Scholar] [CrossRef]
- Blaschke, H. Multiple Mycorrhizal Associations of Individual Calcicole Host Plants in the Alpine Grass-Heath Zone. Mycorrhiza 1991, 1, 31–34. [Google Scholar] [CrossRef]
- Hibbett, D.S.; Bauer, R.; Binder, M.; Giachini, A.J.; Hosaka, K.; Justo, A.; Larsson, E.; Larsson, K.H.; Lawrey, J.D.; Miettinen, O.; et al. 14 Agaricomycetes. In Systematics and Evolution: Part. A.; McLaughlin, D.J., Spatafora, J.W., Eds.; The Mycota; Springer: Berlin/Heidelberg, Germany, 2014; pp. 373–429. ISBN 978-3-642-55318-9. [Google Scholar]
- Clemmensen, K.E.; Bahr, A.; Ovaskainen, O.; Dahlberg, A.; Ekblad, A.; Wallander, H.; Stenlid, J.; Finlay, R.D.; Wardle, D.A.; Lindahl, B.D. Roots and Associated Fungi Drive Long-Term Carbon Sequestration in Boreal Forest. Science 2013, 339, 1615–1618. [Google Scholar] [CrossRef]
- Peay, K.G.; Baraloto, C.; Fine, P.V. Strong Coupling of Plant and Fungal Community Structure across Western Amazonian Rainforests. ISME J. 2013, 7, 1852–1861. [Google Scholar] [CrossRef]
- Rasmann, S.; Pellissier, L.; Defossez, E.; Jactel, H.; Kunstler, G. Climate-Driven Change in Plant–Insect Interactions along Elevation Gradients. Funct. Ecol. 2014, 28, 46–54. [Google Scholar] [CrossRef]
- Chen, C.; Wang, J.; Qiu, F.; Zhao, D. Resilient Distribution System by Microgrids Formation After Natural Disasters. IEEE Trans. Smart Grid 2016, 7, 958–966. [Google Scholar] [CrossRef]
- Johnston, S.R.; Hiscox, J.; Savoury, M.; Boddy, L.; Weightman, A.J. Highly Competitive Fungi Manipulate Bacterial Communities in Decomposing Beech Wood (Fagus sylvatica). FEMS Microbiol. Ecol. 2019, 95, fiy225. [Google Scholar] [CrossRef]
- Lazarević, J.; Menkis, A. Cytospora friesii and Sydowia polyspora are associated with the sudden dieback of Abies concolor in Southern Europe. Plant Prot. Sci. 2022, 58, 258–263. [Google Scholar] [CrossRef]
- Pan, Y.; Ye, H.; Lu, J.; Chen, P.; Zhou, X.D.; Qiao, M.; Yu, Z.F. Isolation and identification of Sydowia polyspora and its pathogenicity on Pinus yunnanensis in Southwestern China. J. Phytopathol. 2018, 166, 386–395. [Google Scholar] [CrossRef]
- Lynikienė, J.; Marčiulynienė, D.; Marčiulynas, A.; Gedminas, A.; Vaičiukynė, M.; Menkis, A. Managed and Unmanaged Pinus sylvestris Forest Stands Harbour Similar Diversity and Composition of the Phyllosphere and Soil Fungi. Microorganisms 2020, 8, 259. [Google Scholar] [CrossRef]
- Cleary, M.; Oskay, F.; Doğmuş, H.T.; Lehtijärvi, A.; Woodward, S.; Vettraino, A.M. Cryptic risks to forest biosecurity associated with the global movement of commercial seed. Forests 2019, 10, 459. [Google Scholar] [CrossRef]
- Ridout, M.; Newcombe, G. Sydowia polyspora is both a Foliar endophyte and a preemergent seed pathogen in Pinus ponderosa. Plant Dis. 2018, 102, 640–644. [Google Scholar] [CrossRef]
- Talgø, V.; Chastagner, G.; Thomsen, I.M.; Cech, T.; Riley, K.; Lange, K.; Klemsdal, S.S.; Stensvand, A. Sydowia polyspora associated with current season needle necrosis (CSNN) on true fir (Abies spp.). Fungal Biol. 2010, 114, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Tinivella, F.; Dani, E.; Minuto, G.; Minuto, A. First report of Sydowia polyspora on Aleppo Pine (Pinus halepensis) in Italy. Plant Dis. 2014, 98, 281. [Google Scholar] [CrossRef]
- Sa’diyah, W.; Hashimoto, A.; Okada, G.; Ohkuma, M. Notes on Some Interesting Sporocarp-Inhabiting Fungi Isolated from Xylarialean Fungi in Japan. Diversity 2021, 13, 574. [Google Scholar] [CrossRef]
- Videira, S.I.R.; Groenewald, J.Z.; Braun, U.; Shin, H.D.; Crous, P.W. All that glitters is not Ramularia. Stud. Mycol. 2016, 83, 49–163. [Google Scholar] [CrossRef]
- Bakhshi, M.; Zare, R.; Jafary, H. Identification of Acrodontium luzulae and Ramularia coleosporii, two fungicolous fungi on Melampsora hypericorum, the causal agent of Hypericum androsaemum rust. Nova Hedwig. 2021, 113, 323–337. [Google Scholar] [CrossRef]
- Abbas, A.; Dobson, A.D.W. Yeasts and Molds|Penicillium camemberti. In Encyclopedia of Dairy Sciences, 2nd ed.; Fuquay, J.W., Ed.; Academic Press: San Diego, CA, USA, 2011; pp. 776–779. [Google Scholar] [CrossRef]
- Leclercq-Perlat, M.-N. Cheese|Camembert, Brie, and Related Varieties. In Encyclopedia of Dairy Sciences, 2nd ed.; Fuquay, J.W., Ed.; Academic Press: San Diego, CA, USA, 2011; pp. 773–782. [Google Scholar] [CrossRef]
- de Melo, M.; V Araujo, A.C.; N Chogi, M.A.; Duarte, I.C. Cellulolytic and lipolytic fungi isolated from soil and leaf litter samples from the Cerrado (Brazilian Savanna). Rev. Biol. Trop 2018, 66, 237–245. [Google Scholar] [CrossRef]
- Kolanlarli, T.K.; Ahmet, A.S.A.N.; Burhan, Ş.E.N.; ÖKTEN, S. Biodiversity of Penicillium species isolated from Edirne Söğütlük Forest soil (Turkey). J. Fungus Nisan 2019, 10, 26–39. [Google Scholar] [CrossRef]
- Povilaitienė, A.; Gedminas, A.; Varnagirytė-Kabašinskienė, I.; Marčiulynienė, D.; Marčiulynas, A.; Lynikienė, J.; Mishcherikova, V.; Menkis, A. Changes in Chemical Properties and Fungal Communities of Mineral Soil after Clear-Cutting and Reforestation of Scots Pine (Pinus sylvestris L.) Sites. Forests 2022, 13, 1780. [Google Scholar] [CrossRef]
- Szymańska, K.; Strumińska-Parulska, D. Atmospheric fallout impact on 210Po and 210Pb content in wild growing mushrooms. Environ. Sci. Pollut. Res. 2020, 27, 20800–20806. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.J.; Yu, P.Y.; Chen, J.Z.; Li, S.M.; Jiang, L.J. Effects of slippery jack (Suillus luteus) on the heavy metal accumulation and soil properties of masson’s pine (pinus massoniana lamb) in a mining area of china. Ecol. Environ. Res. 2020, 18, 3741–3755. [Google Scholar] [CrossRef]
- Krznaric, E.; Verbruggen, N.; Wevers, J.H.L.; Carleer, R.; Vangronsveld, J.; Colpaert, J.V. Cd-tolerant Suillus luteus: A fungal insurance for pines exposed to Cd. Environ. Pollut. 2009, 157, 1581–1588. [Google Scholar] [CrossRef]
- Luo, Z.; Wu, C.; Zhang, C.; Li, H.; Lipka, U.; Polle, A. The role of ectomycorrhizas in heavy metal stress tolerance of host plants. Environ. Exp. Bot. 2014, 108, 47–62. [Google Scholar] [CrossRef]
- Nartey, L.K.; Pu, Q.; Zhu, W.; Zhang, S.; Li, J.; Yao, Y.; Hu, X. Antagonistic and plant growth promotion effects of Mucor moelleri, a potential biocontrol agent. Microbiol. Res. 2022, 255, 126922. [Google Scholar] [CrossRef] [PubMed]
- Ruano-Rosa, D.; Mercado-Blanco, J. Combining Biocontrol Agents and Organics Amendments to Manage Soil-Borne Phytopathogens. In Organic Amendments and Soil Suppressiveness in Plant Disease Management; Meghvansi, M., Varma, A., Eds.; Soil Biology; Springer: Cham, Switzerland, 2015; Volume 46, pp. 457–478. [Google Scholar] [CrossRef]
- Walther, G.; Pawłowska, J.; Alastruey-Izquierdo, A.; Wrzosek, M.; Rodriguez-Tudela, J.L.; Dolatabadi, A.; Chakrabarti, S.; de Hoog, G.S. DNA barcoding in Mucorales: An inventory of biodiversity Persoonia. Pers.-Mol. Phylogeny Evol. Fungi 2013, 30, 11–47. [Google Scholar] [CrossRef] [PubMed]
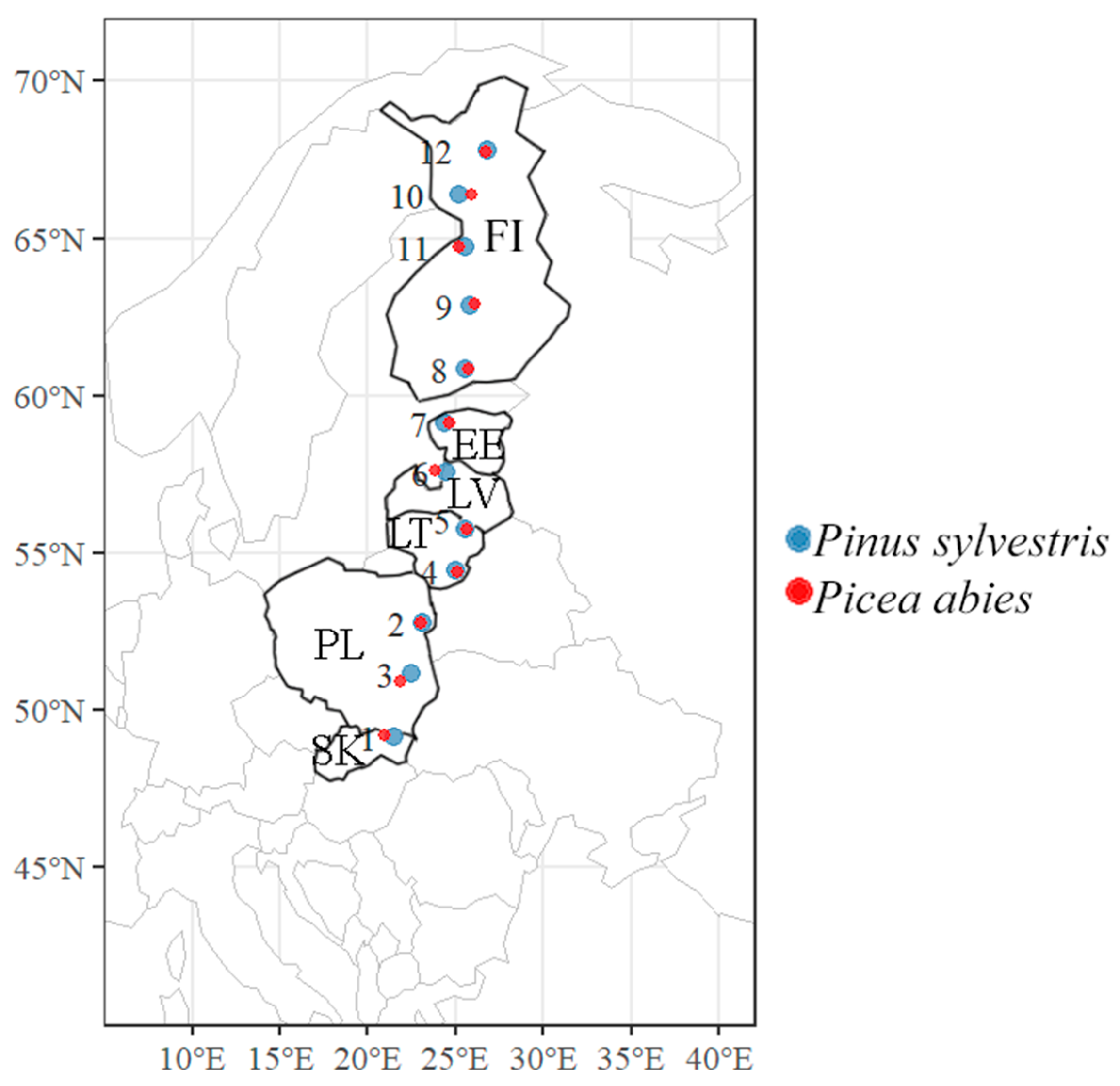

| Site no. | Country | Latitude | Longitude | Habitat/ Vegetation Type * | Tree Age, y | Sample Size, Units | |||
|---|---|---|---|---|---|---|---|---|---|
| Needles | Shoots | Roots | Soil | ||||||
| Pinus sylvestris | |||||||||
| 1 | Slovakia | 49.12828 | 21.52911 | Nb/vm | 80 | 10 | 10 | 5 | 10 |
| 2 | Poland | 51.14678 | 22.51703 | Nc/ox | 70 | 10 | 10 | 5 | 10 |
| 3 | Poland | 52.77492 | 23.08233 | Nc/ox | 50 | 10 | 10 | 5 | 10 |
| 4 | Lithuania | 54.42183 | 24.95956 | Lbl/m | 65 | 10 | 10 | 5 | 10 |
| 5 | Lithuania | 56.05589 | 25.70758 | Nbl/vm | 70 | 10 | 10 | 5 | 10 |
| 6 | Latvia | 57.57453 | 24.43836 | Nb/vm | 90 | 10 | 10 | 5 | 10 |
| 7 | Estonia | 59.12831 | 24.42283 | Nb/vm | 70 | 10 | 10 | 5 | 10 |
| 8 | Finland | 60.86994 | 25.53239 | Nb/vm | 60 | 10 | 10 | 5 | 10 |
| 9 | Finland | 62.88594 | 25.82100 | Nc/hox | 70 | 10 | 10 | 5 | 10 |
| 10 | Finland | 64.71364 | 25.57986 | Nb/vm | 60 | 10 | 10 | 5 | 10 |
| 11 | Finland | 66.37572 | 25.21342 | Na/v | 80 | 10 | 10 | 5 | 10 |
| 12 | Finland | 67.80269 | 26.78244 | Lb/m | 60 | 10 | 10 | 5 | 10 |
| Total P. sylvestris | 120 | 120 | 60 | 120 | |||||
| Picea abies | |||||||||
| 1 | Slovakia | 49.22103 | 21.57144 | Nc/ox | 30 | 10 | 10 | 5 | 10 |
| 2 | Poland | 50.93917 | 22.30369 | Nc/hox | 60 | 10 | 10 | 5 | 10 |
| 3 | Poland | 52.76811 | 23.07808 | Lc/mox | 50 | 10 | 10 | 5 | 10 |
| 4 | Lithuania | 54.42106 | 24.95953 | Lcp/mox | 60 | 10 | 10 | 5 | 10 |
| 5 | Lithuania | 56.05342 | 23.67956 | Ncl/ox | 68 | 10 | 10 | 5 | 10 |
| 6 | Latvia | 57.60172 | 24.40689 | Lc/mox | 70 | 10 | 10 | 5 | 10 |
| 7 | Estonia | 59.12831 | 24.42283 | Nc/hox | 60 | 10 | 10 | 5 | 10 |
| 8 | Finland | 60.86961 | 25.53197 | Nc/ox | 50 | 10 | 10 | 5 | 10 |
| 9 | Finland | 62.90411 | 25.80914 | Lc/mox | 60 | 10 | 10 | 5 | 10 |
| 10 | Finland | 64.71394 | 25.57769 | Nb/vm | 50 | 10 | 10 | 5 | 10 |
| 11 | Finland | 66.37719 | 25.27164 | Lc/mox | 60 | 10 | 10 | 5 | 10 |
| 12 | Finland | 67.76231 | 26.76128 | Pc/fils | 70 | 10 | 10 | 5 | 10 |
| Total P. abies | 120 | 120 | 60 | 120 | |||||
| Code | Bioclimatic Variable |
|---|---|
| BIO1 | Annual mean temperature |
| BIO2 | Mean diurnal range (mean of monthly, maximum temperature—minimum temperature) |
| BIO3 | Isothermality (BIO2/NIO7) (×100) |
| BIO4 | Temperature seasonality (standard deviation ×100) |
| BIO5 | Maximum temperature of warmest month |
| BIO6 | Minimum temperature of coldest month |
| BIO7 | Temperature annual range (BIO5-BIO6) |
| BIO8 | Mean temperature of wettest quarter |
| BIO9 | Mean temperature of driest quarter |
| BIO10 | Mean temperature of warmest quarter |
| BIO11 | Mean temperature of coldest quarter |
| BIO12 | Annual precipitation |
| BIO13 | Precipitation of wettest month |
| BIO14 | Precipitation of driest month |
| BIO15 | Precipitation seasonality (coefficient of variation) |
| BIO16 | Precipitation of wettest quarter |
| BIO17 | Precipitation of driest quarter |
| BIO18 | Precipitation of warmest quarter |
| BIO19 | Precipitation of coldest quarter |
| Phylum | Reference Sequence | Species Name | Similarity, % | Needles, % | Shoots, % | Roots, % | Soil, % | All, % |
|---|---|---|---|---|---|---|---|---|
| Ascomycota | MK762617 | Sydowia polyspora | 100 | 3.86 | 15.58 | 0.62 | 2.35 | 5.84 |
| Ascomycota | KX287273 | Acrodontium luzulae | 100 | 0.31 | 0.22 | 7.00 | 11.87 | 5.04 |
| Ascomycota | MT236531 | Unidentified sp. 5210_4 | 99 | - | 0.003 | 7.07 | 0.04 | 2.65 |
| Ascomycota | MG828280 | Unidentified sp. 5210_5 | 83 | 0.08 | 0.09 | 6.41 | 0.15 | 2.46 |
| Ascomycota | KP891398 | Unidentified sp. 5208_1 | 100 | 0.58 | 6.17 | - | 0.11 | 1.93 |
| Basidiomycota | MG597398 | Mycena septentrionalis | 100 | 0.03 | 0.00 | 5.05 | 0.04 | 1.90 |
| Ascomycota | KP897305 | Unidentified sp. 5210_12 | 100 | 7.00 | 2.99 | 0.08 | 0.12 | 1.88 |
| Ascomycota | JX536159 | Gymnopus dryophilus | 100 | - | 0.01 | 4.72 | 0.01 | 1.77 |
| Zygomycota | MT242128 | Unidentified sp. 5210_2 | 100 | 0.23 | 0.01 | 4.38 | 0.02 | 1.67 |
| Ascomycota | MG827987 | Unidentified sp. 5210_9 | 99 | - | - | 3.39 | 0.41 | 1.35 |
| Ascomycota | KY660851 | Phyllactinia fraxini | 100 | 0.51 | 3.93 | 0.01 | 0.05 | 1.25 |
| Ascomycota | UDB0754226 | Unidentified sp. 5208_17 | 100 | 0.66 | 3.13 | - | 0.09 | 1.04 |
| Ascomycota | LR876918 | Unidentified sp. 5210_7 | 100 | 3.06 | 0.95 | 0.43 | 0.75 | 1.00 |
| Ascomycota | MN902367 | Unidentified sp. 5208_2 | 100 | 1.46 | 2.56 | 0.003 | 0.20 | 1.00 |
| Ascomycota | UDB0754118 | Unidentified sp. 5210_30 | 91 | - | - | 2.56 | - | 0.95 |
| Ascomycota | MH248043 | Archaeorhizomyces sp. 5208_0 | 100 | 0.30 | 0.44 | 0.35 | 3.16 | 0.92 |
| Basidiomycota | KT275603 | Tomentella sp. 5210_8 | 99 | 1.40 | 0.75 | 0.47 | 1.47 | 0.87 |
| Ascomycota | UDB028437 | Coniothyrium olivaceum | 100 | 0.80 | 0.57 | 1.34 | 0.07 | 0.79 |
| Basidiomycota | UDB018458 | Tomentella sp. 5210_21 | 100 | - | 0.003 | 2.08 | 0.01 | 0.78 |
| Zygomycota | MN395040 | Kwoniella sp. 5210_18 | 99 | - | - | 0.02 | 3.87 | 0.77 |
| All | 20.26 | 37.39 | 45.97 | 24.82 | 35.84 |
| Phylum | Reference Number | Species Name | Similarity, % | Needles, % | Shoots, % | Roots, % | Soil, % | All, % |
|---|---|---|---|---|---|---|---|---|
| Ascomycota | MK762617 | Sydowia polyspora | 100 | 49.66 | 12.26 | 0.22 | 0.38 | 11.48 |
| Ascomycota | MT355566 | Penicillium camemberti | 100 | 0.06 | 0.03 | 1.93 | 12.86 | 4.56 |
| Zygomycota | MT242128 | Unidentified sp. 5210_2 | 100 | - | 0.08 | 15.86 | 0.24 | 3.18 |
| Ascomycota | KU059580 | Suillus luteus | 99 | 0.88 | 6.71 | 0.03 | 0.05 | 2.42 |
| Basidiomycota | MN902821 | Unidentified sp. 5208_24 | 100 | - | - | - | 6.28 | 2.03 |
| Ascomycota | LR876918 | Unidentified sp. 5210_7 | 100 | 1.79 | 3.52 | 0.57 | 0.38 | 1.68 |
| Basidiomycota | MG597398 | Mycena septentrionalis | 100 | 0.03 | 0.01 | 7.06 | 0.60 | 1.57 |
| Ascomycota | MK390491 | Mucor moelleri | 100 | 0.01 | 3.93 | 0.02 | 0.01 | 1.34 |
| Ascomycota | UDB035461 | Mycosylva clarkii | 99 | 0.03 | 0.00 | 1.88 | 2.62 | 1.22 |
| Ascomycota | MG828280 | Unidentified sp. 5210_5 | 83 | 0.11 | 0.06 | 5.03 | 0.34 | 1.12 |
| Ascomycota | MN902647 | Unidentified sp. 5210_17 | 96 | 0.01 | 0.00 | 3.09 | 1.46 | 1.07 |
| Ascomycota | MH248043 | Archaeorhizomyces sp. 5208_0 | 100 | 0.26 | 0.05 | 0.31 | 2.60 | 0.96 |
| Ascomycota | MT595563 | Cladophialophora sp. 5210_29 | 99 | 0.30 | 2.63 | 0.02 | 0.01 | 0.94 |
| Ascomycota | MG679813 | Russula badia | 100 | 0.49 | 2.38 | 0.02 | 0.07 | 0.90 |
| Ascomycota | MT236513 | Unidentified sp. 5210_39 | 98 | - | - | - | 2.65 | 0.86 |
| Ascomycota | LS450480 | Unidentified sp. 5210_19 | 98 | 0.01 | 0.01 | 1.49 | 1.71 | 0.85 |
| Ascomycota | ON963481 | Leptodontidium sp. 5210_31 | 99 | 1.26 | 1.80 | 0.01 | 0.14 | 0.84 |
| Ascomycota | MG827987 | Unidentified sp. 5210_9 | 99 | - | - | 4.29 | 0.01 | 0.83 |
| Basidiomycota | LR874260 | Piloderma lanatum | 100 | - | - | - | 2.52 | 0.81 |
| Basidiomycota | KT275603 | Tomentella sp. 5210_8 | 99 | 1.32 | 0.45 | 0.42 | 1.10 | 0.78 |
| All | 56.21 | 33.92 | 42.24 | 36.04 | 39.43 |
| Site No. | Tree Species | Needles | Roots | Shoots | Soil | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of Fungal OTUs | No. of Fungal Sequences | Shannon Diversity Index | No. of Fungal OTUs | No. of Fungal Sequences | Shannon Diversity Index | No. of Fungal OTUs | No. of Fungal Sequences | Shannon Diversity Index | No. of Fungal OTUs | No. of Fungal Sequences | Shannon Diversity Index | ||
| 1 | P. abies | 177 | 956 | 4.30 | 63 | 173 | 3.76 | 235 | 2039 | 4.52 | 304 | 4079 | 3.73 |
| 2 | 21 | 8819 | 0.09 | 115 | 1529 | 2.96 | 178 | 2053 | 3.83 | 203 | 2206 | 3.36 | |
| 3 | 149 | 872 | 415 | 185 | 5271 | 2.71 | 167 | 1144 | 4.01 | 114 | 4336 | 1.27 | |
| 4 | 100 | 423 | 3.87 | 55 | 1205 | 2.56 | 178 | 2043 | 4.12 | 213 | 6816 | 2.98 | |
| 5 | 155 | 1660 | 4.15 | 17 | 88 | 2.00 | 217 | 4355 | 4.15 | 349 | 11,519 | 4.31 | |
| 6 | 169 | 1542 | 3.69 | 77 | 1032 | 2.90 | 245 | 3468 | 4.35 | - | - | 0.69 | |
| 7 | 85 | 378 | 3.81 | 132 | 2416 | 3.23 | 186 | 1586 | 3.77 | 192 | 1743 | 3.66 | |
| 8 | 197 | 1231 | 3.60 | 120 | 1913 | 2.40 | 236 | 3531 | 4.07 | 157 | 1752 | 3.22 | |
| 9 | 91 | 873 | 3.05 | 160 | 2525 | 2.67 | 264 | 4446 | 3.30 | 291 | 5864 | 3.80 | |
| 10 | 166 | 1089 | 2.33 | 102 | 2019 | 3.44 | 267 | 5526 | 3.90 | 104 | 685 | 4.47 | |
| 11 | 78 | 257 | 1.89 | 162 | 2657 | 4.03 | 140 | 6810 | 3.79 | 245 | 1460 | 3.86 | |
| 12 | 12 | 16 | 3.67 | 185 | 3528 | 3.44 | 157 | 5548 | 4.65 | 62 | 193 | 3.69 | |
| Total P. abies | 752 | 18,116 | 3.38 | 753 | 24,356 | 4.42 | 1094 | 42,549 | 4.97 | 1408 | 40,653 | 5.07 | |
| 1 | P. sylvestris | 161 | 1812 | 3.80 | 149 | 4211 | 3.20 | 119 | 651 | 3.73 | 208 | 1426 | 3.61 |
| 2 | 92 | 833 | 3.56 | 175 | 5087 | 2.70 | 36 | 63 | 3.30 | 279 | 2443 | 3.85 | |
| 3 | 113 | 800 | 3.60 | 122 | 5764 | 2.40 | 184 | 1833 | 4.07 | 181 | 2276 | 3.21 | |
| 4 | 92 | 775 | 3.56 | 45 | 1050 | 2.57 | 176 | 5462 | 3.51 | 288 | 4534 | 461 | |
| 5 | 131 | 2187 | 3.08 | 25 | 258 | 2.08 | 196 | 4829 | 3.72 | 215 | 2734 | 4.42 | |
| 6 | 208 | 2695 | 3.64 | 137 | 1472 | 3.57 | 250 | 1689 | 4.60 | - | - | - | |
| 7 | 145 | 673 | 4.12 | 139 | 3971 | 2.84 | 183 | 1664 | 3.84 | 181 | 1191 | 3.54 | |
| 8 | 70 | 554 | 4.31 | 121 | 2654 | 3.74 | 103 | 4336 | 4.52 | 171 | 1610 | 3.98 | |
| 9 | 136 | 1329 | 4.15 | 191 | 2877 | 2.69 | 107 | 850 | 4.02 | 84 | 198 | 4.05 | |
| 10 | 88 | 545 | 0.09 | 153 | 2538 | 2.91 | 201 | 2776 | 3.81 | 120 | 738 | 3.36 | |
| 11 | 95 | 520 | 2.35 | 194 | 5331 | 1.01 | 110 | 2451 | 3.01 | 21 | 120 | 3.71 | |
| 12 | 117 | 944 | 2.21 | 160 | 3012 | 4.13 | 117 | 3863 | 4.27 | 98 | 2806 | 4.49 | |
| Total P. sylvestris | 731 | 13,667 | 4.94 | 863 | 38,225 | 4.52 | 855 | 30,467 | 4.63 | 1142 | 20,078 | 5.29 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mishcherikova, V.; Lynikienė, J.; Marčiulynas, A.; Gedminas, A.; Prylutskyi, O.; Marčiulynienė, D.; Menkis, A. Biogeography of Fungal Communities Associated with Pinus sylvestris L. and Picea abies (L.) H. Karst. along the Latitudinal Gradient in Europe. J. Fungi 2023, 9, 829. https://doi.org/10.3390/jof9080829
Mishcherikova V, Lynikienė J, Marčiulynas A, Gedminas A, Prylutskyi O, Marčiulynienė D, Menkis A. Biogeography of Fungal Communities Associated with Pinus sylvestris L. and Picea abies (L.) H. Karst. along the Latitudinal Gradient in Europe. Journal of Fungi. 2023; 9(8):829. https://doi.org/10.3390/jof9080829
Chicago/Turabian StyleMishcherikova, Valeriia, Jūratė Lynikienė, Adas Marčiulynas, Artūras Gedminas, Oleh Prylutskyi, Diana Marčiulynienė, and Audrius Menkis. 2023. "Biogeography of Fungal Communities Associated with Pinus sylvestris L. and Picea abies (L.) H. Karst. along the Latitudinal Gradient in Europe" Journal of Fungi 9, no. 8: 829. https://doi.org/10.3390/jof9080829
APA StyleMishcherikova, V., Lynikienė, J., Marčiulynas, A., Gedminas, A., Prylutskyi, O., Marčiulynienė, D., & Menkis, A. (2023). Biogeography of Fungal Communities Associated with Pinus sylvestris L. and Picea abies (L.) H. Karst. along the Latitudinal Gradient in Europe. Journal of Fungi, 9(8), 829. https://doi.org/10.3390/jof9080829






