Taxonomy and Phylogeny of Cystolepiota (Agaricaceae, Agaricales): New Species, New Combinations and Notes on the C. seminuda Complex
Abstract
1. Introduction
2. Materials and Methods
2.1. Morphological Studies
2.2. DNA Extraction, PCR Amplifications and Sequencing
2.3. Phylogenetic Analyses
3. Results
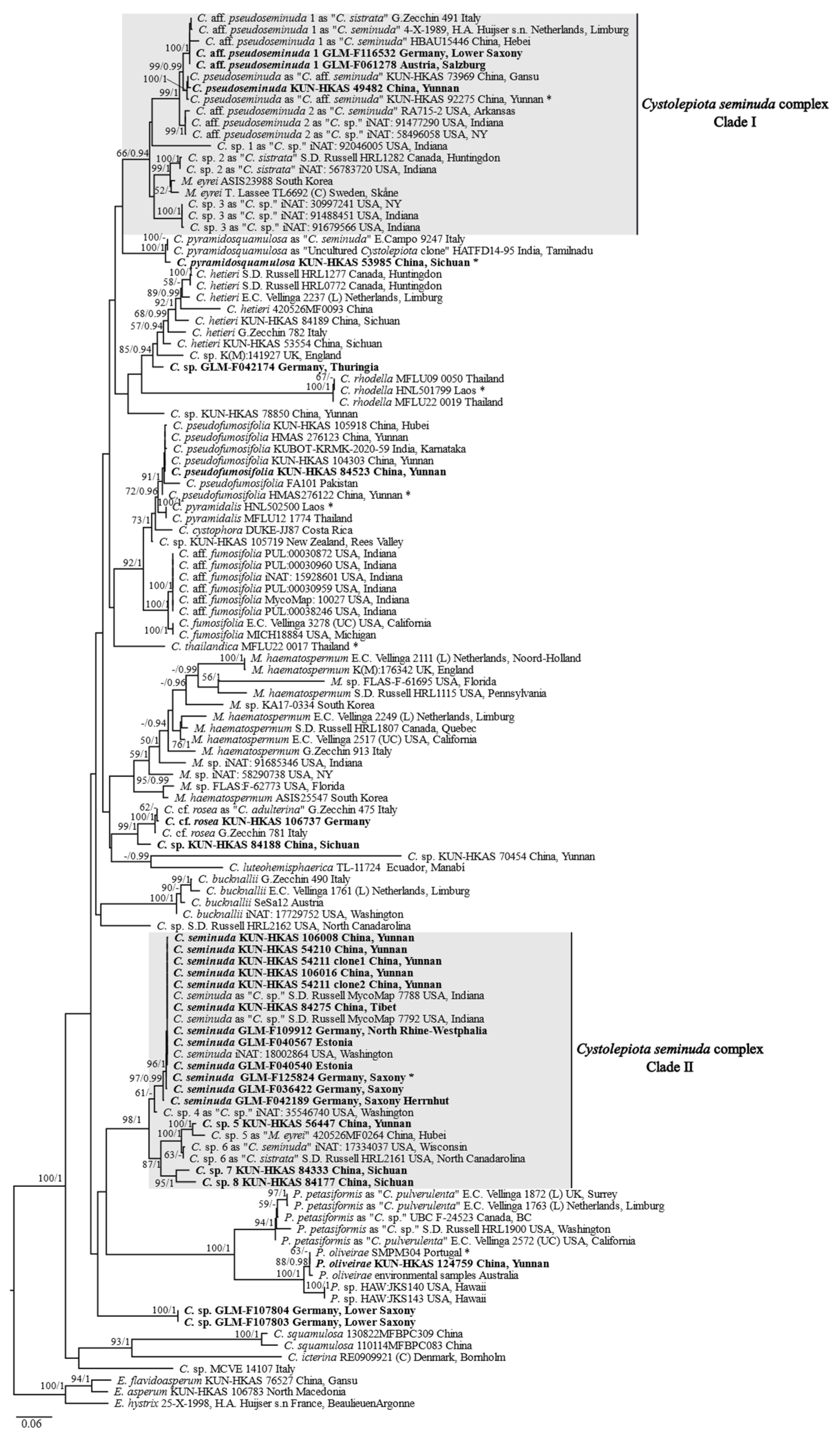
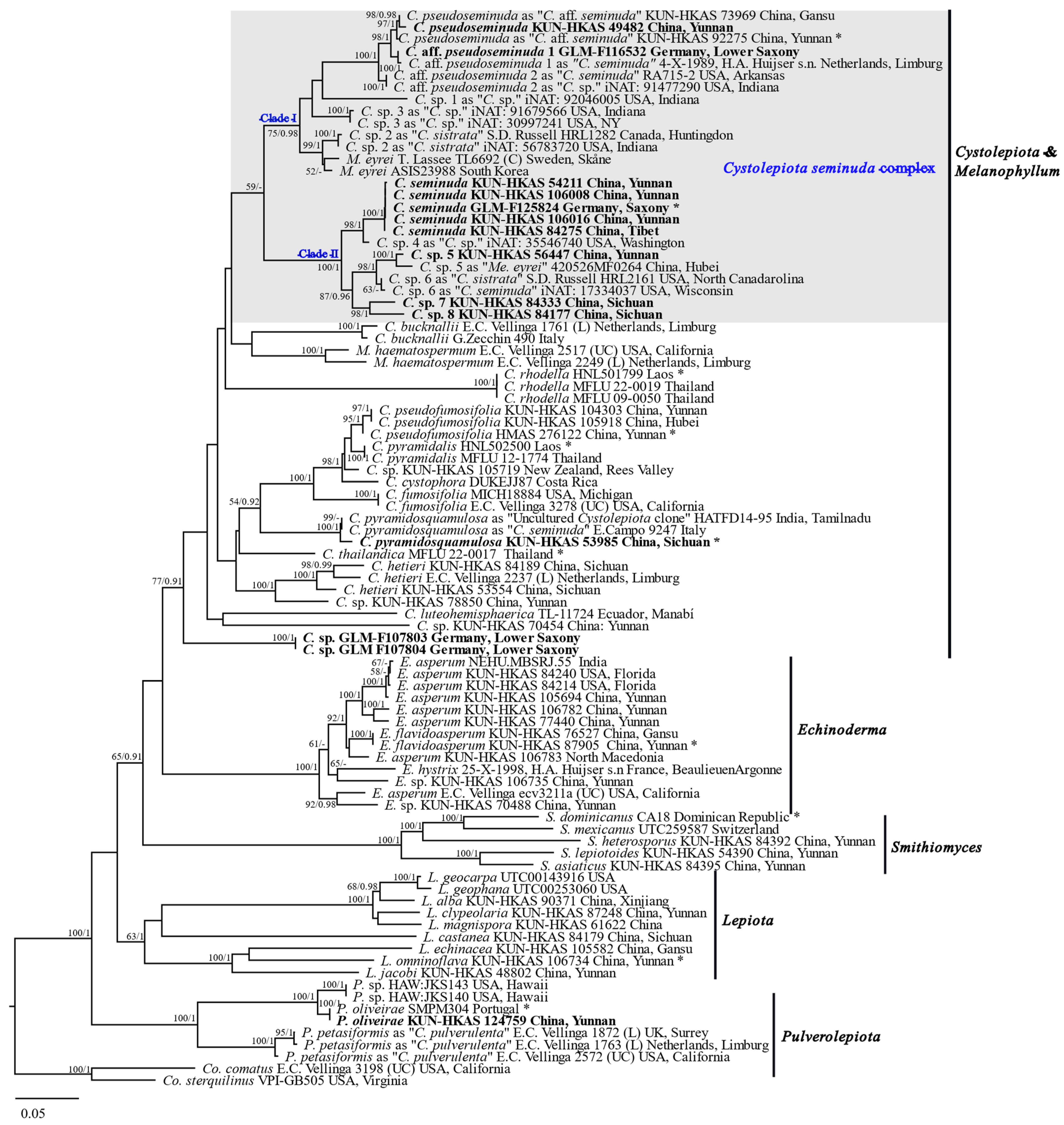
3.1. Phylogenetic Results
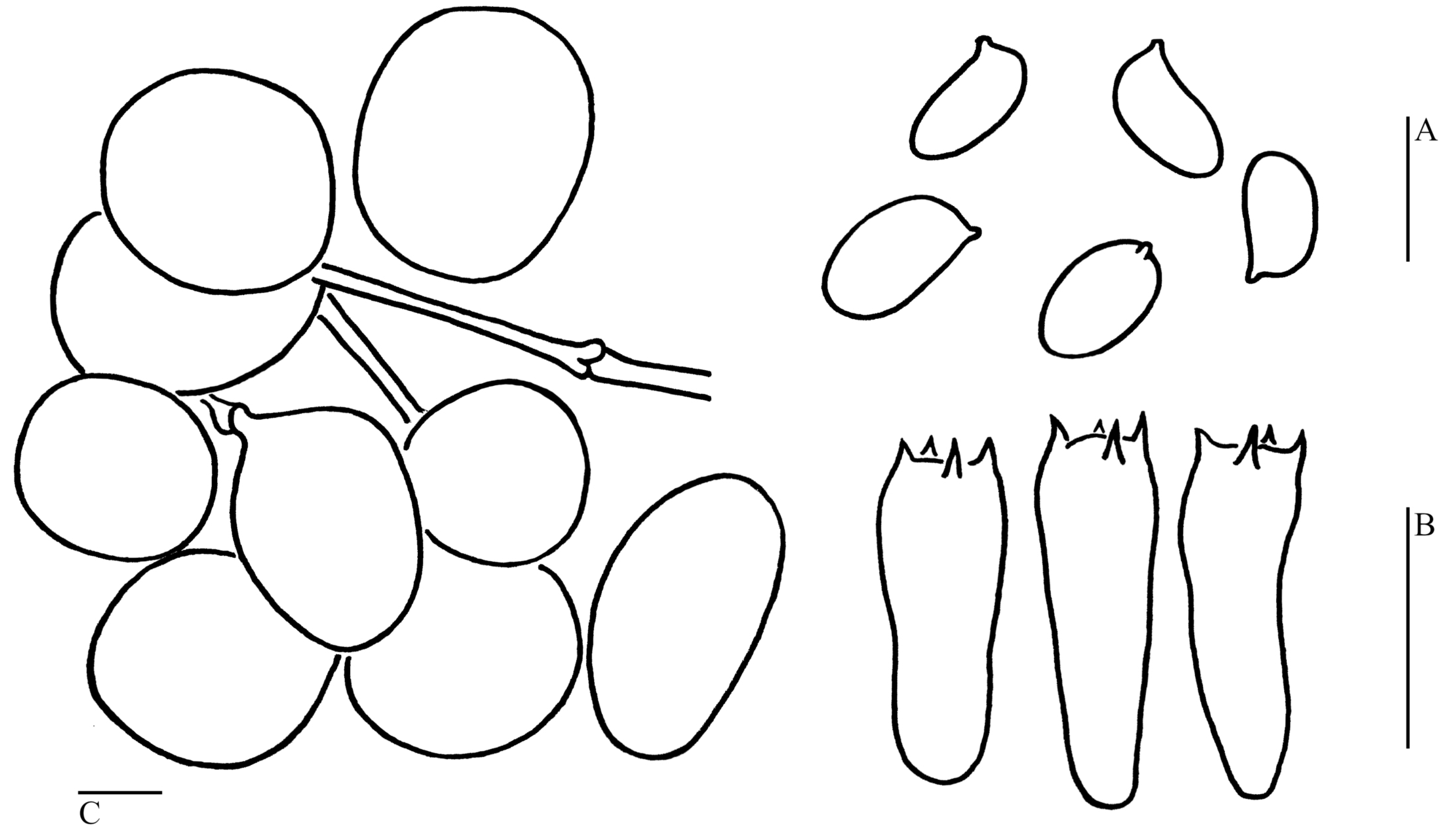
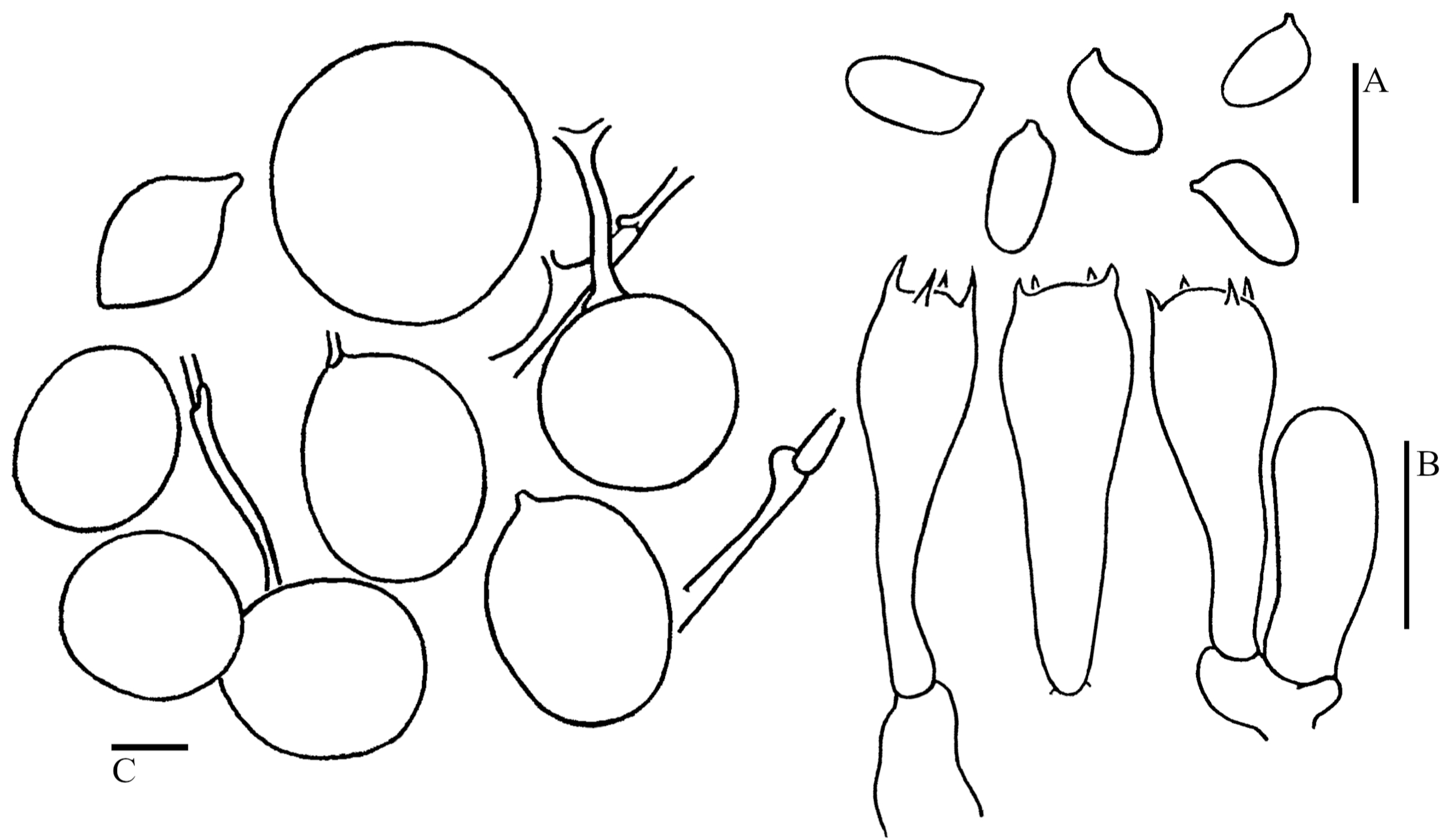
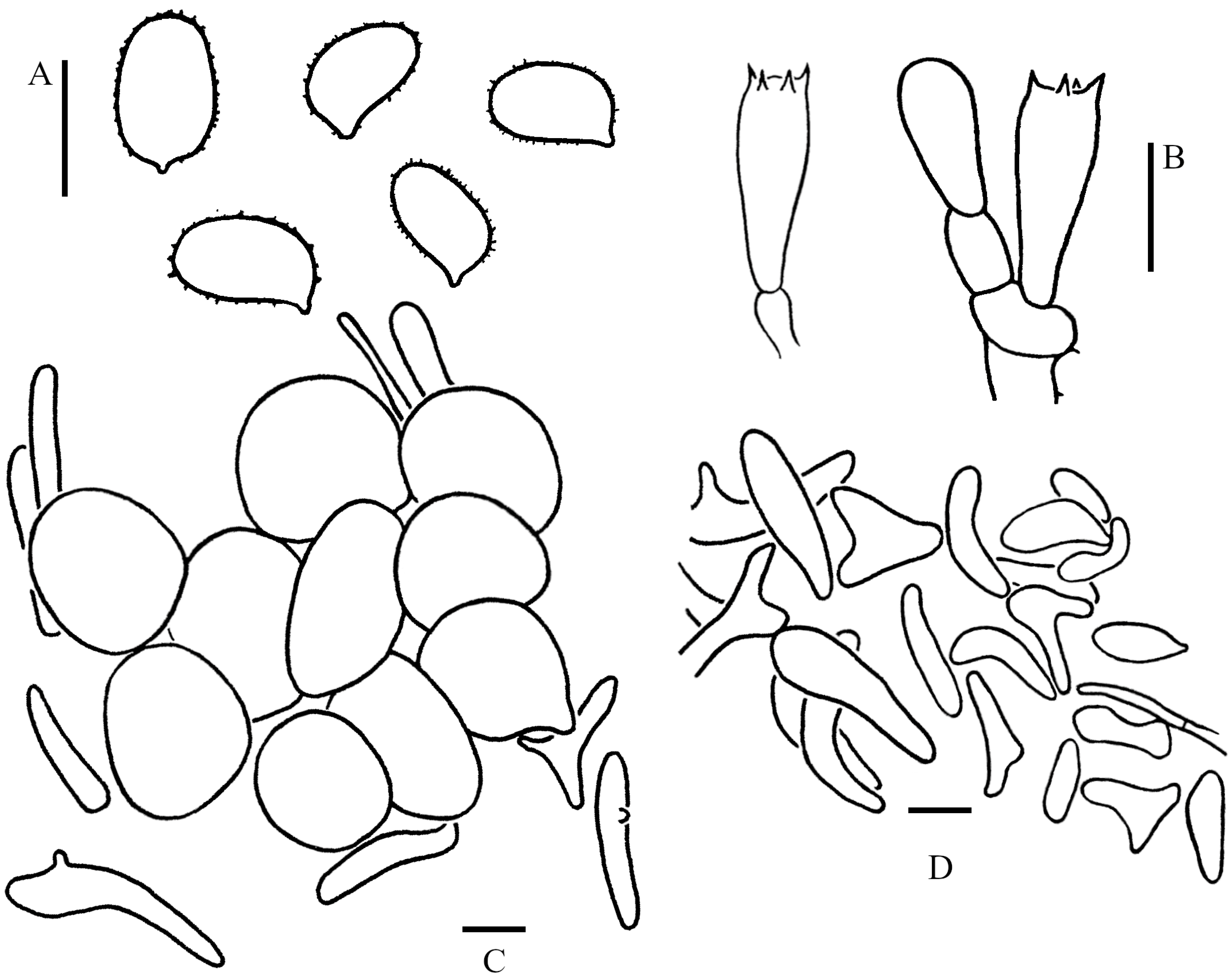
3.2. Taxonomy
4. Discussion
4.1. Circumscription and Diversity of Cystolepiota and Pulverolepiota
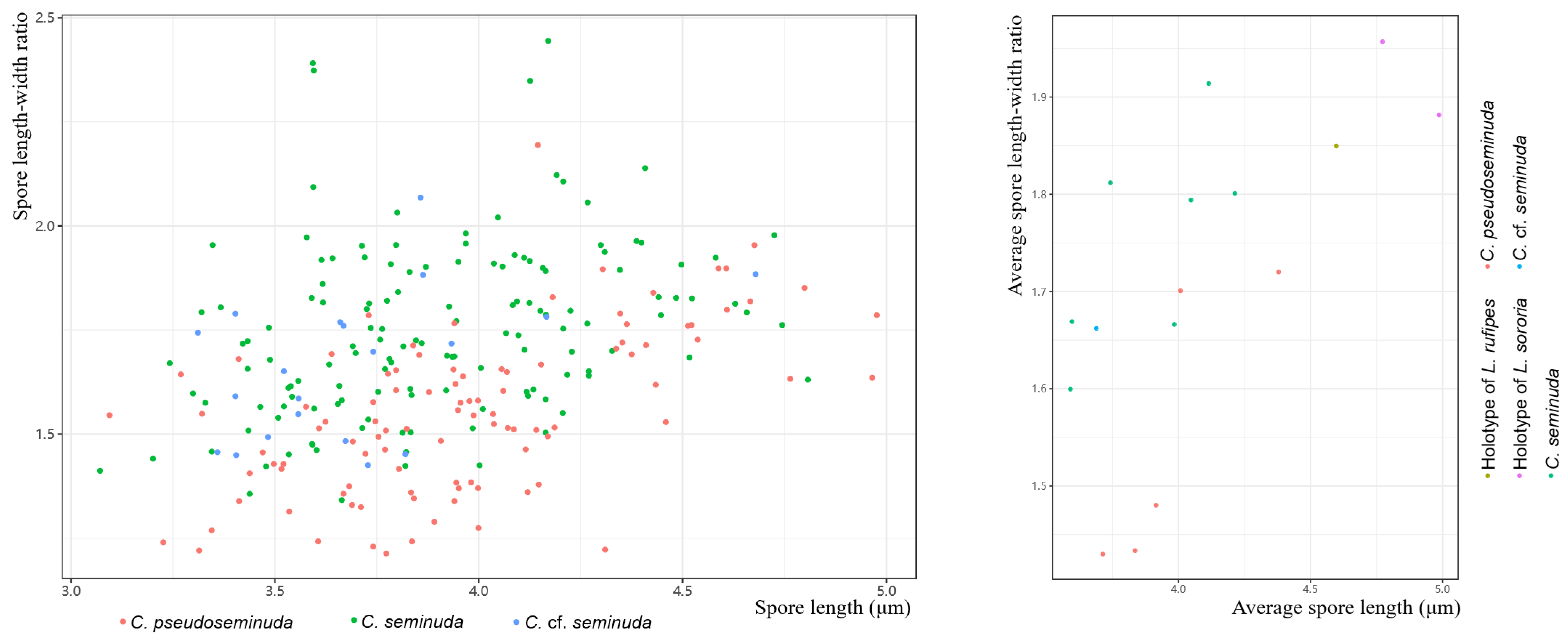
4.2. Circumscription of Species in the Cystolepiota seminuda Species Complex
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singer, R.; Digilio, A.P.L. Pródromo de la Flora Agaricina Argentina. Lilloa 1951, 25, 5–461. [Google Scholar]
- Knudsen, H. Notes on Cystolepiota Sing. and Lepiota S.F. Gray. Bot. Tidsskr. 1978, 73, 124–136. [Google Scholar]
- Singer, R.; Clemençon, H. Notes on some leucosporous and rhodosporus European Agarics. Nova Hedwig. 1972, 23, 305–351. [Google Scholar]
- Bon, M. Famille Lepiotaceae Roze ex Overeem. Doc. Mycol. 1993, 22, 27–32. [Google Scholar]
- Vellinga, E.C. Lepiotaceous fungi in California, U.S.A. 4. Type studies of Lepiota fumosifolia and L. petasiformis. Mycotaxon 2006, 98, 225–232. [Google Scholar]
- Vellinga, E.C.; Huijser, H.A. Notes on Cystolepiota: Sections Cystolepiota and Pulverolepiota. Persoonia 1998, 16, 513–526. [Google Scholar]
- Bon, M. Les lépiotes de l’herbier Boudier au Muséum National d’Histoire Naturelle de Paris. Doc. Mycol. 1977, 7, 11–22. [Google Scholar]
- Bon, M. Clé monographique des ‘Lépiotes’ d’Europe. Doc. Mycol. 1981, 11, 1–77. [Google Scholar]
- Pegler, D.N. Agaric Flora of Sri Lanka; Royal Botanic Cardens; KEW: London, UK, 1986; Volume 12, p. 519. [Google Scholar]
- Candusso, M.; Lanzoni, G. Lepiota S.L. Fungi Europaei 4; Giovanna Biella: Saronno, Italy, 1990; p. 743. [Google Scholar]
- Knudsen, H. A revision of Lepiota sect. Echinatae and Amyloideae (Agaricaceae) in Europe. Bot. Tidsskr. 1980, 75, 121–155. [Google Scholar]
- Noordeloos, M.E.; Kuyper, T.W.; Vellinga, E.C. Flora Agaricina Neerlandica: Critical Monographs on Families of Agarics and Boleti Occurring in The Netherlands; August Aimé Balkema: Amsterdam, The Netherlands, 2001; Volume 5, p. 169. [Google Scholar]
- Pegler, D.N. A revision of the genus Lepiota from Ceylon. Kew Bull. 1972, 27, 155–202. [Google Scholar] [CrossRef]
- Pegler, D.N. A Preliminary Agaric Flora of East Africa; Kew Bulletin Additional Series; KEW: London, UK, 1977; Volume 6, p. 615. [Google Scholar]
- Singer, R. The Agaricales in Modern Taxonomy; Koeltz Scientific Books: Koenigstein, Germany, 1986; p. 981. [Google Scholar]
- Bon, M. Les genres Echinoderma (Locq. ex Bon) st. nov. et Rugosomyces Raithelhuber ss. lato. Doc. Mycol. 1991, 21, 61–66. [Google Scholar]
- Vellinga, E.C. Phylogeny of Lepiota (Agaricaceae)—Evidence from nrITS and nrLSU sequences. Mycol. Prog. 2003, 2, 305–322. [Google Scholar] [CrossRef]
- Hou, Y.J.; Ge, Z.-W. New species of Echinoderma and Lepiota (Agaricaceae) from China. Phytotaxa 2020, 447, 221–236. [Google Scholar] [CrossRef]
- Ge, Z.-W.; Xu, T.X.; Qu, H.; Ma, Y.R. Three new species of Smithiomyces from tropical Asia support an amphi-Pacific disjunct distribution in the genus. Mycologia 2021, 113, 1009–1021. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, M.; Victoria, F.; Egres, C.; Putzke, J.; Pereira, A. Cystolepiota Singer (Agaricales) na Amazônia, Rio de Janeiro, Rio Grande do Sul (Brasil) e Tucumã (Argentina). Neotrop. Biol. Conserv. 2017, 12, 48–58. [Google Scholar] [CrossRef][Green Version]
- Vellinga, E.C. Notes on Cystolepiota seminuda. Persoonia 1987, 13, 321–325. [Google Scholar]
- Yang, Z.L. Fungi Lepiotoidei (Agaricaceae); Science Press: Beijing, China, 2019; Volume 52, p. 230. [Google Scholar]
- Lasch, W.G. Enumeratio Agaricorum Marchiae Brandenburgicae, nondum in Floris nostratibus nominatorum, cum observationibus in cognitos et novorum descriptionibus. Linnaea 1828, 3, 152–162. [Google Scholar]
- Bon, M. Novitates. Doc. Mycol. 1976, 6, 41–46. [Google Scholar]
- Fayod, V. Prodrome d’une histoire naturelle des Agaricinés. Ann. Sci. Naturelles. Bot. Biol. Vég. 1889, 7, 181–477. [Google Scholar]
- Kummer, P. Der Führer in Die Pilzkunde: Anleitung Zum Methodischen, Leichten und Sichern Bestimmen Der in Deutschland Vorkommenden Pilze: Mit Ausnahme Der Schimmel-und Allzu Winzigen Schleim-Und Kern-Pilzchen; Verlag Von E; Luppe’s Buchhandlung: Zerbst, Germany, 1871; p. 146. [Google Scholar]
- Kuntze, O. Revisio Generum Plantarum:Vascularium Omnium atque Cellularium Multarum Secundum Leges Nomenclaturae Internationales cum Enumeratione Plantarum Exoticarum in Itinere Mundi Collectarum. Pars II; Arthur Felix: Leipzig, Germany, 1891; p. 1011. [Google Scholar]
- Quélet, L. Mémoires de la societe d’émulation de montbeliard ser. 2. In Les Champignons du Jura et des Vosges; Baillière: Paris, France, 1872; pp. 43–332. [Google Scholar]
- Bon, M. Novitates-Validations de taxons (ouvrage général en préparation). Doc. Mycol. 1985, 15, 51–52. [Google Scholar]
- Fries, E.M. Systema Mycologicum: Sistens Fungorum Ordines, Genera et Species, Huc Usque Cognitas, Quas ad Normam Methodi Naturalis Determinavit; Ex Officina Berlingiana: Lundae, Norway, 1821; Volume 1, p. 520. [Google Scholar]
- Fries, E.M. Systema Mycologicum: Sistens Fungorum Ordines, Genera et Species, Huc Usque Cognitas, Quas ad Normam Methodi Naturalis Determinavit, Index; Ex Officina Berlingiana: Lundae, Norway, 1832; p. 202. [Google Scholar]
- Fries, E.M. Monographia Lepiotarum Sueciae; Leffler, C.A., Ed.; Reg. Acad. Typographia: Uppsala, Sweden, 1854; p. 17. [Google Scholar]
- Huijsman, H.S.C. Observations sur les Lepioteae Fayod. Persoonia 1960, 1, 325–329. [Google Scholar]
- Fries, E.M. Hymenomycetes Europaei Sive Epicriseos Systematis Mycologici; Typis Descripsit ed.; Berling: Uppsala, Sweden, 1874; p. 755. [Google Scholar]
- Fries, E.M. Epicrisis Systematis Mycologici, Seu Synopsis Hymenonycetum; Typographia Academica: Uppsala, Sweden, 1936; p. 610. [Google Scholar]
- Vellinga, E.C. Lepiota in California: Species with a hymeniform pileus covering. Mycologia 2010, 102, 664–674. [Google Scholar] [CrossRef]
- Alanbagi, R.; Alshuwaili, F.; Stephenson, S. Fungi associated with forest floor litter in northwest Arkansas. Curr. Res. Environ. Appl. Mycol. 2019, 9, 25–35. [Google Scholar] [CrossRef]
- Vellinga, E.C. Genera in the family Agaricaceae: Evidence from nrITS and nrLSU sequences. Mycol. Res. 2004, 108, 354–377. [Google Scholar] [CrossRef] [PubMed]
- Kornerup, A.; Wanscher, J.H. Methuen Handbook of Colour, 3rd ed.; Methuen Ltd.: London, UK, 1978; p. 252. [Google Scholar]
- R-Core-Team. R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 1 July 2022).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer International Publishing: New York, NY, USA, 2009; p. 205. [Google Scholar]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for Basidiomycetes—Application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef] [PubMed]
- White, T.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M., Gelfand, D., Sninsky, J., White, T., Eds.; Academic Press: New York, NY, USA, 1990; Volume 31, pp. 315–322. [Google Scholar]
- Cubeta, M.A.; Echandi, E.; Abernethy, T.; Vilgalys, R. Characterization of anastomosis groups of binucleate Rhizoctonia species using restriction analysis of an amplified ribosomal RNA gene. Phytopathology 1991, 81, 1395–1400. [Google Scholar] [CrossRef]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 18, 315–322. [Google Scholar] [CrossRef]
- Matheny, P.B. Improving phylogenetic inference of mushrooms with RPB1 and RPB2 nucleotide sequences (Inocybe; Agaricales). Mol. Phylogenet. Evol. 2005, 35, 1–20. [Google Scholar] [CrossRef]
- Rehner, S.; Buckley, E. A Beauveria phylogeny inferred from nuclear ITS and EF1-alpha sequences: Evidence for cryptic diversification and links to Cordyceps teleomorphs. Mycologia 2005, 97, 84–98. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Capella-Gutierrez, S.; Silla-Martinez, J.M.; Gabaldon, T. trimAl: A tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 2009, 25, 1972–1973. [Google Scholar] [CrossRef] [PubMed]
- Larsson, A. AliView: A fast and lightweight alignment viewer and editor for large datasets. Bioinformatics 2014, 30, 3276–3278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Gao, F.L.; Jakovlic, I.; Zou, H.; Zhang, J.; Li, W.X.; Wang, G.T. PhyloSuite: An integrated and scalable desktop platform for streamlined molecular sequence data management and evolutionary phylogenetics studies. Mol. Ecol. Resour. 2020, 20, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Lanfear, R.; Frandsen, P.B.; Wright, A.M.; Senfeld, T.; Calcott, B. PartitionFinder 2: New methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol. Biol. Evol. 2017, 34, 772–773. [Google Scholar] [CrossRef]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Hohna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef]
- Paraíso, M.; Maurice, J.-P.; Normand, A.-C.; Fouchier, F.; Roux, P. Cystolepiota oliveirae sp. nov., harvested in Portugal on trunk of dead tree ferns. Mycol. Montenegrina 2016, 19, 21–31. [Google Scholar]
- Vellinga, E.C. Notulae Ad Floram Agaricinam Neerlandicam. 18. Some Notes on Cystolepiota and Lepiota. Persoonia 1992, 14, 407–415. [Google Scholar]
- Sysouphanthong, P.; Thongklang, N.; Liu, Y.; Vellinga, E. Three new species of Cystolepiota from Laos and Thailand. Diversity 2022, 14, 449. [Google Scholar] [CrossRef]
- Xu, M.L.; Li, G.J.; Zhou, J.L.; Bai, X.M.; Zhao, R.L. New species of Cystolepiota from China. Mycology 2016, 7, 165–170. [Google Scholar] [CrossRef]
- Morgan, A.P. North American species of Lepiota. J. Mycol. 1906, 12, 154–159. [Google Scholar] [CrossRef]
- Bon, M. Novitates-Taxons nouveaux. Doc. Mycol. 1978, 8, 69–71. [Google Scholar]
- Murrill, W.A. The Agaricaceae of the Pacific coast—II. Mycologia 1912, 4, 231–262. [Google Scholar] [CrossRef]
- Vellinga, E.C. Studies in Lepiota III. Some species from California, U.S.A. Mycotaxon 2001, 80, 285–295. [Google Scholar]
- He, M.Q.; Zhao, R.L.; Hyde, K.D.; Begerow, D.; Kemler, M.; Yurkov, A.; McKenzie, E.H.C.; Raspé, O.; Kakishima, M.; Sánchez Ramírez, S.; et al. Notes, outline and divergence times of Basidiomycota. Fungal Divers. 2019, 99, 105–367. [Google Scholar] [CrossRef]
- Kalichman, J.; Kirk, P.M.; Matheny, P.B. A compendium of generic names of agarics and Agaricales. Taxon 2020, 69, 425–447. [Google Scholar] [CrossRef]
- Vellinga, E.C. Nomenclatural Overview of Lepiotaceous Fungi (Agaricaceae). Version 2010, 4, 11. [Google Scholar]
- Yang, Z.L. Flora Fungorum Sinicorum, Fungi Lepiotaoidei (Agaricaceae); Science Press: Beijing, China, 2019; Volume 52, p. 230. [Google Scholar]
- Arun Kumar, T.K.; Manimohan, P. The genus Cystolepiota (Agaricales, Basidiomycota) in Kerala State, India. Mycotaxon 2009, 107, 277–284. [Google Scholar] [CrossRef]
- Bau, T.; Li, Y. Lepiota squamulosa, a new species from China. J. Fungal Res. 2004, 2, 49–50. [Google Scholar] [CrossRef]
- Yang, Z.L.; Ge, Z.-W. Six new combinations of lepiotaceous fungi from China. Mycosystema 2017, 36, 542–551. [Google Scholar] [CrossRef]
- Gierczyk, B.; Kujawa, A.; Szczepkowski, A.; Chachua, P. Rare species of Lepiota and related genera. Acta Mycol. 2011, 46, 137–178. [Google Scholar] [CrossRef]
- Hausknecht, A.; Pidlich-Aigner, H. Lepiotaceae (Schirmlinge) in Österreich. 1. Die Gattungen Chamaemyces, Chlorophyllum, Cystolepiota, Leucoagaricus, Leucocoprinus, Macrolepiota, Melanophyllum und Sericeomyces. Osterr. Z. Für. Pilzkd. 2004, 13, 1–38. [Google Scholar]
- Lange, J.E. Flora Agaricina Danica VOLI; Recato A/S: Copenhagen, Denmark, 1935; p. 90. [Google Scholar]
- Albuquerque, M.; Pereira, A.B. A família Agaricaceae Chevall. em trechos de Mata Atlântica da Reserva Biológica do Tinguá, Nova Iguaçu, Rio de Janeiro, Brasil: Gêneros Agaricus, Cystolepiota e Lepiota. Acta Bot. Bras. 2010, 24, 497–509. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, H.; Damm, U.; Hou, Y.-J.; Ge, Z.-W. Taxonomy and Phylogeny of Cystolepiota (Agaricaceae, Agaricales): New Species, New Combinations and Notes on the C. seminuda Complex. J. Fungi 2023, 9, 537. https://doi.org/10.3390/jof9050537
Qu H, Damm U, Hou Y-J, Ge Z-W. Taxonomy and Phylogeny of Cystolepiota (Agaricaceae, Agaricales): New Species, New Combinations and Notes on the C. seminuda Complex. Journal of Fungi. 2023; 9(5):537. https://doi.org/10.3390/jof9050537
Chicago/Turabian StyleQu, Hua, Ulrike Damm, Ya-Jun Hou, and Zai-Wei Ge. 2023. "Taxonomy and Phylogeny of Cystolepiota (Agaricaceae, Agaricales): New Species, New Combinations and Notes on the C. seminuda Complex" Journal of Fungi 9, no. 5: 537. https://doi.org/10.3390/jof9050537
APA StyleQu, H., Damm, U., Hou, Y.-J., & Ge, Z.-W. (2023). Taxonomy and Phylogeny of Cystolepiota (Agaricaceae, Agaricales): New Species, New Combinations and Notes on the C. seminuda Complex. Journal of Fungi, 9(5), 537. https://doi.org/10.3390/jof9050537






