Evaluation of Thermography as a Diagnostic Technique in Asymptomatic or Incipient Onychomycosis
Abstract
1. Introduction
2. Materials and Methods
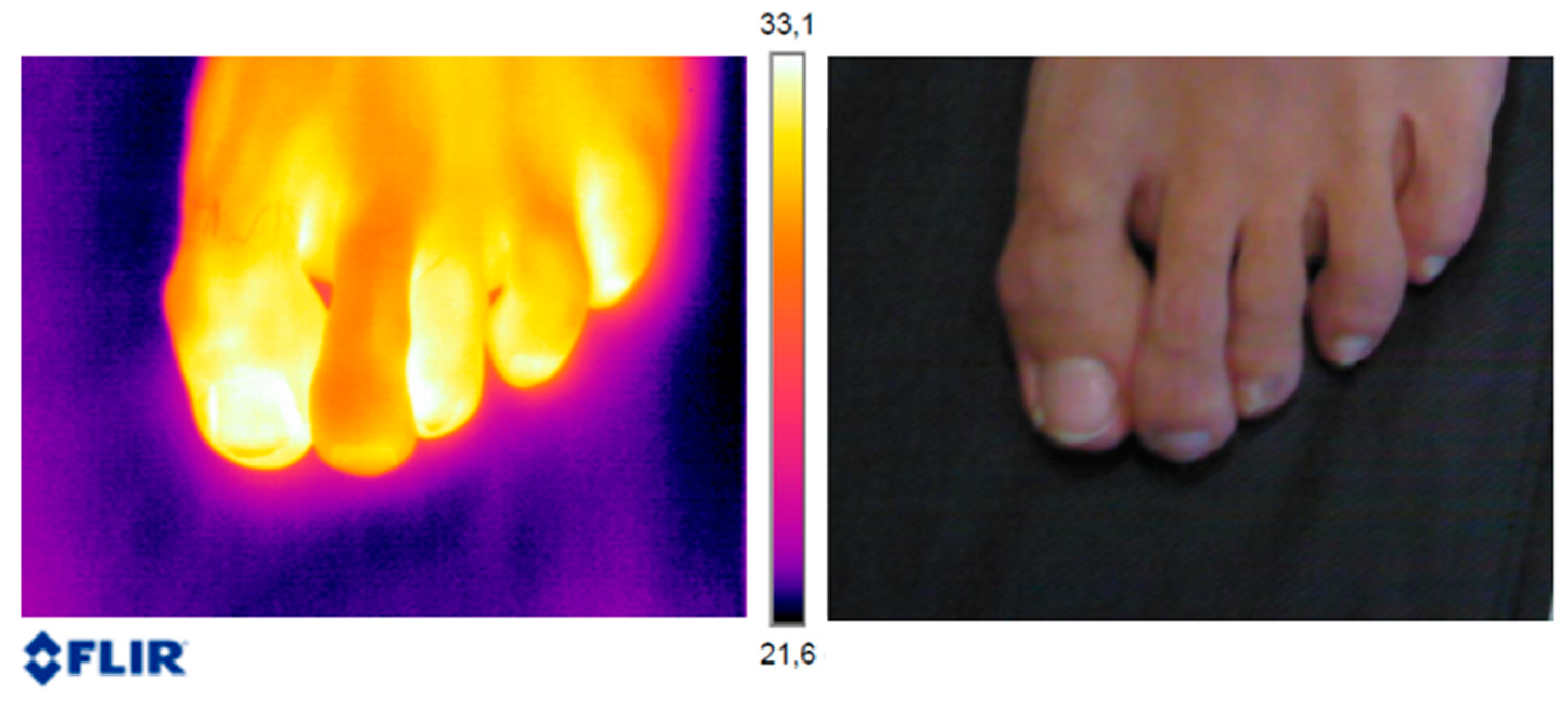
2.1. Acquisition of Thermography Images
2.2. Procedure in the Microbiological Analysis Laboratory
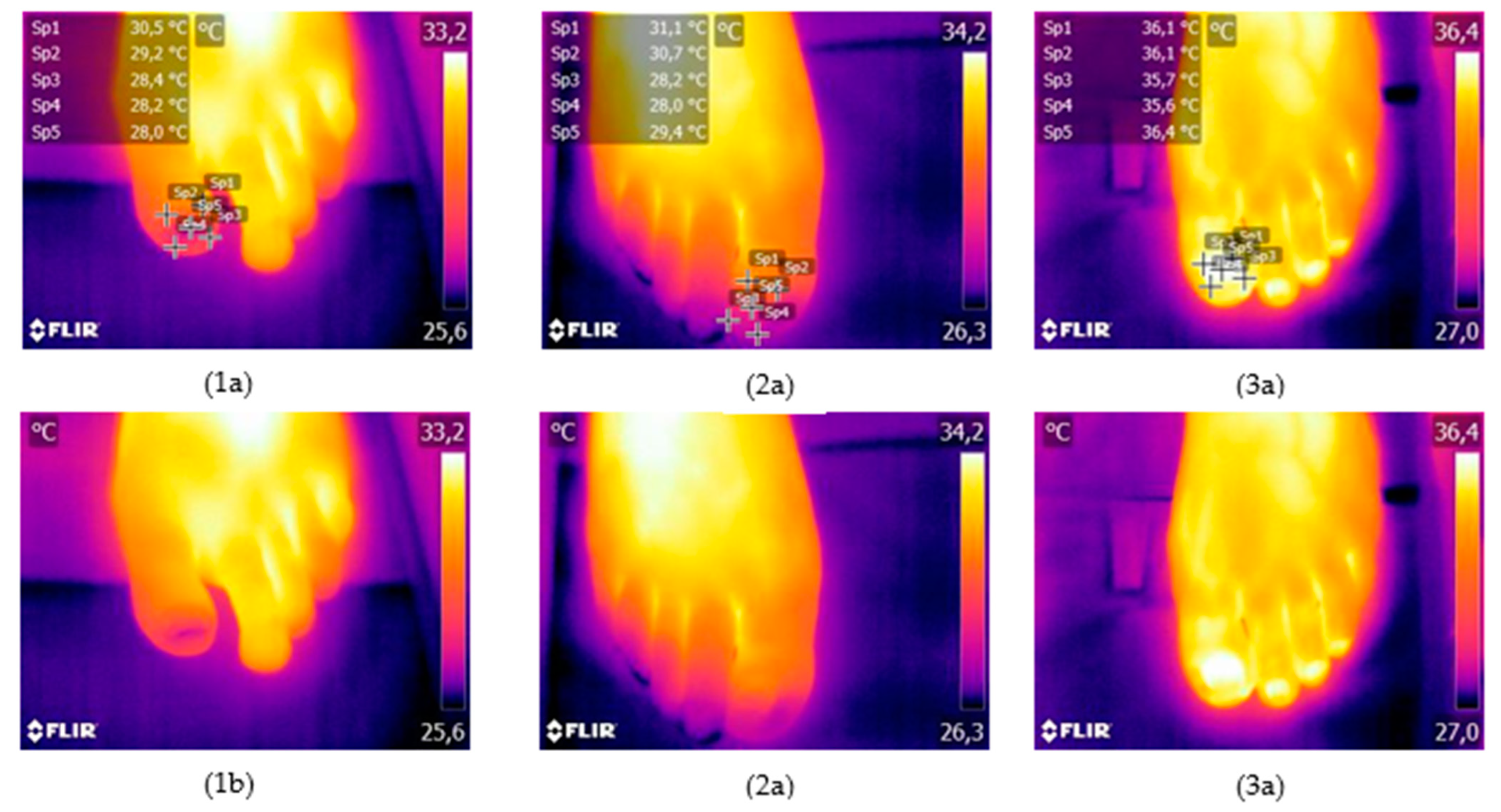
2.3. Analysis of Thermographic Images
- P1: proximal lateral upper point
- P2: proximal medial upper point
- P3: distal lateral lower point
- P4: distal medial lower point
- P5: middle point of the nail plate
2.4. Description of Variables
- Age: participant’s age in years
- Sex: participant’s sex
- Temperature: temperature taken at the reference points on the nail plate
- Infecting organism:
- ○
- Fungi: total dermatophyte and yeast infections
- ○
- Dermatophytes: dermatophyte infections
- ○
- Yeasts: yeast infections
2.5. Statistical Analysis
3. Results
3.1. Presence of Infection by Location on the Nail, Sex and Age
3.2. Thermography by Location on the Nail, Sex and Age
3.3. Relation between Thermography and Prevalence of Infection
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Papini, M.; Piraccini, B.M.; Difonzo, E.; Brunoro, A. Epidemiology of Onychomycosis in Italy: Prevalence Data and Risk Factor Identification. Mycoses 2015, 58, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Lipner, S.R.; Scher, R.K. Onychomycosis: Clinical Overview and Diagnosis. J. Am. Acad. Dermatol. 2019, 80, 835–851. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.K.C.; Lam, J.M.; Leong, K.F.; Hon, K.L.; Barankin, B.; Leung, A.A.M.; Wong, A.H.C. Onychomycosis: An Updated Review. Recent Pat. Inflamm. Allergy Drug Discov. 2019, 14, 32–45. [Google Scholar] [CrossRef]
- Hoy, N.Y.; Leung, A.K.C.; Metelitsa, A.I.; Adams, S. New Concepts in Median Nail Dystrophy, Onychomycosis, and Hand, Foot, and Mouth Disease Nail Pathology. ISRN Dermatol. 2012, 2012, 680163. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, G.; Jain, H.C.; Lynde, C.W.; Foley, K.A.; Daigle, D.; Cooper, E.A.; Summerbell, R.C. The Prevalence of Unsuspected Onychomycosis and Its Causative Organisms in a Multicentre Canadian Sample of 30 000 Patients Visiting Physicians’ Offices. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 1567–1572. [Google Scholar] [CrossRef]
- Perea, S.; Ramos, M.J.; Garau, M.; Gonzalez, A.; Noriega, A.R.; Del Palacio, A. Prevalence and Risk Factors of Tinea Unguium and Tinea Pedis in the General Population in Spain. J. Clin. Microbiol. 2000, 38, 3226–3230. [Google Scholar] [CrossRef]
- Iglesias Sánchez, M.J.; Pérez Pico, A.M.; Marcos Tejedor, F.; Iglesias Sánchez, M.J.; Mayordomo Acevedo, R. Using a Polymerase Chain Reaction as a Complementary Test to Improve the Detection of Dermatophyte Fungus in Nails. J. Am. Podiatr. Med. Assoc. 2014, 104, 233–237. [Google Scholar] [CrossRef]
- Bodman, M.A.; Krishnamurthy, K. Onychomycosis; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Gupta, A.K.; Sibbald, R.G.; Andriessen, A.; Belley, R.; Boroditsky, A.; Botros, M.; Chelin, R.; Gulliver, W.; Keast, D.; Raman, M. Toenail Onychomycosis—A Canadian Approach with a New Transungual Treatment: Development of a Clinical Pathway. J. Cutan. Med. Surg. 2015, 19, 440–449. [Google Scholar] [CrossRef]
- Joyce, A.; Gupta, A.K.; Koenig, L.; Wolcott, R.; Carviel, J. Fungal Diversity and Onychomycosis. J. Am. Podiatr. Med. Assoc. 2019, 109, 57–63. [Google Scholar] [CrossRef]
- Thomas, J.; Jacobson, G.; Narkowicz, C.; Peterson, G.; Burnet, H.; Sharpe, C. Toenail Onychomycosis: An Important Global Disease Burden. J. Clin. Pharm. Ther. 2010, 35, 497–519. [Google Scholar] [CrossRef]
- Ben Youssef, A.; Kallel, A.; Azaiz, Z.; Jemel, S.; Bada, N.; Chouchen, A.; Belhadj-Salah, N.; Fakhfakh, N.; Belhadj, S.; Kallel, K. Onychomycosis: Which Fungal Species Are Involved? Experience of the Laboratory of Parasitology-Mycology of the Rabta Hospital of Tunis. J. Mycol. Med. 2018, 28, 651–654. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Mays, R.R.; Versteeg, S.G.; Shear, N.H.; Piguet, V. Update on Current Approaches to Diagnosis and Treatment of Onychomycosis. Expert Rev. Anti. Infect. Ther. 2018, 16, 929–938. [Google Scholar] [CrossRef]
- Totri, C.R.; Feldstein, S.; Admani, S.; Friedlander, S.F.; Eichenfield, L.F. Epidemiologic Analysis of Onychomycosis in the San Diego Pediatric Population. Pediatr. Dermatol. 2017, 34, 46–49. [Google Scholar] [CrossRef]
- Ghannoum, M.A.; Hajjeh, R.A.; Scher, R.; Konnikov, N.; Gupta, A.K.; Summerbell, R.; Sullivan, S.; Daniel, R.; Krusinski, P.; Fleckman, P.; et al. A Large-Scale North American Study of Fungal Isolates from Nails: The Frequency of Onychomycosis, Fungal Distribution, and Antifungal Susceptibility Patterns. J. Am. Acad. Dermatol. 2000, 43, 641–648. [Google Scholar] [CrossRef]
- Walling, H.W.; Sniezek, P.J. Distribution of Toenail Dystrophy Predicts Histologic Diagnosis of Onychomycosis. J. Am. Acad. Dermatol. 2007, 56, 945–948. [Google Scholar] [CrossRef]
- Flores, J.M.; Castillo, V.B.; Franco, F.C.; Betanzos, A. Superficial Fungal Infections: Clinical and Epidemiological Study in Adolescents from Marginal Districts of Lima and Callao, Peru. J. Infect. Dev. Ctries. 2009, 3, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Mays, R.R.; Versteeg, S.G.; Piraccini, B.M.; Takwale, A.; Shemer, A.; Babaev, M.; Grover, C.; Di Chiacchio, N.G.; Taborda, P.R.O.; et al. Global Perspectives for the Management of Onychomycosis. Int. J. Dermatol. 2019, 58, 1118–1129. [Google Scholar] [CrossRef] [PubMed]
- Velasquez-agudelo, V.; Cardona-arias, J.A. Meta-Analysis of the Utility of Culture, Biopsy, and Direct KOH Examination for the Diagnosis of Onychomycosis. BMC Infect. Dis. 2017, 17, 166. [Google Scholar] [CrossRef]
- Levitt, J.O.; Levitt, B.H.; Akhavan, A.; Yanofsky, H. The Sensitivity and Specificity of Potassium Hydroxide Smear and Fungal Culture Relative to Clinical Assessment in the Evaluation of Tinea Pedis: A Pooled Analysis. Dermatol. Res. Pract. 2010, 2010, 764843. [Google Scholar] [CrossRef]
- Iglesias Sánchez, J.M.; Pico Pérez, A.M.; Muñoz del Rey, J.R.; Ledesma Alcázar, C.; MayordomoAcevedo, R. Métodos Moleculares: Reacción En Cadena de La Polimerasa (Pcr), Frente a Medios de Cultivo Convencionales. Análisis Comparativo En La Detección de Hongos Dermatofitos. Rev. Española Podol. 2011, 22, 146–149. [Google Scholar]
- Turner, T.A. Diagnostic Thermography. Vet. Clin. North Am. Equine Pract. 2001, 17, 95–114. [Google Scholar] [CrossRef] [PubMed]
- Usamentiaga, R.; Venegas, P.; Guerediaga, J.; Vega, L.; Molleda, J.; Bulnes, F.G. Infrared Thermography for Temperature Measurement and Non-Destructive Testing. Sensors 2014, 14, 12305–12348. [Google Scholar] [CrossRef]
- Adam, M.; Ng, E.Y.K.; Tan, J.H.; Heng, M.L.; Tong, J.W.K.; Acharya, U.R. Computer Aided Diagnosis of Diabetic Foot Using Infrared Thermography: A Review. Comput. Biol. Med. 2017, 91, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Pérez Pico, A.M.; Mingorance Álvarez, E.; Pérez Luque, C.; Mayordomo Acevedo, R. Adquisición de Competencias Para El Diagnóstico de Onicomicosis Mediante Entrenamiento Práctico Podológico Preclínico y Clínico. Educ. Med. Super. 2019, 33, 37–49. [Google Scholar]
- Meola, C. Infrared Thermography in the Architectural Field. Sci. World J. 2013, 8, 323948. [Google Scholar] [CrossRef]
- Bagavathiappan, S.; Saravanan, T.; Philip, J.; Jayakumar, T.; Raj, B.; Karunanithi, R.; Panicker, T.M.R.; Korath, P.; Jagadeesan, K. Investigation of Peripheral Vacular Disorders Using Thermal Imaging. Br. J. Diabetes Vasc. Dis. 2008, 8, 102–104. [Google Scholar] [CrossRef]
- Zaproudina, N.; Ming, Z.; Hänninen, O.O.P. Plantar Infrared Thermography Measurements and Low Back Pain Intensity. J. Manip. Physiol. Ther. 2006, 29, 219–223. [Google Scholar] [CrossRef]
- Ng, E.Y.K. A Review of Thermography as Promising Non-Invasive Detection Modality for Breast Tumor. Int. J. Therm. Sci. 2009, 48, 849–859. [Google Scholar] [CrossRef]
- Dibai Filho, A.V.; Packer, A.C.; Costa, A.C.D.S.; Berni-Schwarzenbeck, K.C.D.S.; Rodrigues-Bigaton, D. Assessment of the Upper Trapezius Muscle Temperature in Women with and without Neck Pain. J. Manip. Physiol. Ther. 2012, 35, 413–417. [Google Scholar] [CrossRef]
- Petrova, N.L.; Whittam, A.; MacDonald, A.; Ainarkar, S.; Donaldson, A.N.; Bevans, J.; Allen, J.; Plassmann, P.; Kluwe, B.; Ring, F.; et al. Reliability of a Novel Thermal Imaging System for Temperature Assessment of Healthy Feet. J. Foot Ankle Res. 2018, 11, 22. [Google Scholar] [CrossRef]
- Petrova, N.L.; Donaldson, N.K.; Tang, W.; MacDonald, A.; Allen, J.; Lomas, C.; Leech, N.; Ainarkar, S.; Bevans, J.; Plassmann, P.; et al. Infrared Thermography and Ulcer Prevention in the High-Risk Diabetic Foot: Data from a Single-Blind Multicentre Controlled Clinical Trial. Diabet. Med. 2020, 37, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Gauci, J.; Falzon, O.; Formosa, C.; Gatt, A.; Ellul, C.; Mizzi, S.; Mizzi, A.; Sturgeon Delia, C.; Cassar, K.; Chockalingam, N.; et al. Automated Region Extraction from Thermal Images for Peripheral Vascular Disease Monitoring. J. Healthc. Eng. 2018, 2018, 5092064. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Sanz, D.; Losa-Iglesias, M.E.; López-López, D.; Calvo-Lobo, C.; Palomo-López, P.; Becerro-de-Bengoa-Vallejo, R. Infrared Thermography Applied to Lower Limb Muscles in Elite Soccer Players with Functional Ankle Equinus and Non-Equinus Condition. PeerJ 2017, 2017, e3388. [Google Scholar] [CrossRef]
- Benito-De-pedro, M.; Becerro-De-bengoa-vallejo, R.; Losa-Iglesias, M.E.; Rodríguez-Sanz, D.; López-López, D.; Cosín-Matamoros, J.; Martínez-Jiménez, E.M.; Calvo-Lobo, C. Effectiveness between Dry Needling and Ischemic Compression in the Triceps Surae Latent Myofascial Trigger Points of Triathletes on Pressure Pain Threshold and Thermography: A Single Blinded Randomized Clinical Trial. J. Clin. Med. 2019, 8, 1632. [Google Scholar] [CrossRef]
- Villaseñor-mora, C.; Garay-sevilla, M.E.; Padilla-medina, J.A.; Arteaga-murillo, L.I. Procedure to Diagnose Onychomycosis through Changes in Emissivity on Infrared Images in Emissivity on Infrared Images. J. Biomed. Opt. 2013, 18, 116005. [Google Scholar] [CrossRef]
- Sakkas, H.; Kittas, C.; Kapnisi, G.; Priavali, E.; Kallinteri, A.; Bassukas, I.D.; Gartzonika, K. Onychomycosis in Northwestern Greece over a 7-Year Period. Pathogens 2020, 9, 851–858. [Google Scholar] [CrossRef]
- Gupta, A.K.; Venkataraman, M.; Talukder, M. Onychomycosis in Older Adults: Prevalence, Diagnosis, and Management. Drugs Aging 2022, 39, 191–198. [Google Scholar] [CrossRef]
- Miura, Y.; Takehara, K.; Nakagami, G.; Amemiya, A.; Kanazawa, T.; Kimura, N.; Kishi, C.; Koyano, Y.; Tamai, N.; Nakamura, T.; et al. Screening for Tinea Unguium by Thermography in Older Adults with Subungual Hyperkeratosis. Geriatr. Gerontol. Int. 2014, 15, 991–996. [Google Scholar] [CrossRef]
- Veiga, F.F.; de Castro-Hoshino, L.V.; Rezende, P.S.T.; Baesso, M.L.; Svidzinski, T.I.E. Insights on the etiopathogenesis of onychomycosis by dermatophyte, yeast and non-dermatophyte mould in ex vivo model. Exp. Dermatol. 2022, 31, 1810–1814. [Google Scholar] [CrossRef] [PubMed]

| First Toenail in Each Foot | Sexes | Age (Mean) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Infection | Total (n = 214) | Toenail | Comparison of Means | Sex | Comparison of Means | Mean Age | Comparison of Means | ||||||
| Right (n = 107) | Left (n = 107) | R2 E | P-V | Female (n = 148) | Male (n = 66) | R2 E | P-V | Positive | Negative | R2 E | P-V | ||
| Fungi (positive) | 14.0% (30) | 13.1% (14) | 15.0% (16) | 0.001 | 0.694 (ns) | 9.5% (14) | 24.2% (16) | 0.039 | 0.004 (**) | 21.7 years (±2.5) | 21.1 years (±2.9) | 0.005 | 0.303 (ns) |
| Dermatophytes (positive) | 5.1% (11) | 4.7% (5) | 5.6% (6) | 0.000 | 0.757 (ns) | 1.4% (2) | 13.6% (9) | 0.066 | 0.000 (**) | 20.9 years (±2.4) | 21.2 years (±2.8) | 0.001 | 0.745 (ns) |
| Yeast (positive) | 8.9% (19) | 8.4% (9) | 9.3% (10) | 0.000 | 0.810 (ns) | 8.1% (12) | 10.6% (7) | 0.002 | 0.553 (ns) | 22.1 years (±2.4) | 21.1 years (±2.8) | 0.011 | 0.131 (ns) |
| Thermography (°C) | Centrality | Variability | Percentile | ||||
|---|---|---|---|---|---|---|---|
| Mean | IC 95% of the Mean | Range (min/max) | Standard Deviation | P25 | Median | P75 | |
| Temperature P-1 | 30.9 | 30.5–31.4 | 21.4/36.2 | 3.4 | 27.8 | 31.5 | 33.8 |
| Temperature P-2 | 30.7 | 30.2–31.2 | 21.1/39.7 | 3.5 | 27.5 | 31.1 | 33.7 |
| Temperature P-3 | 29.6 | 29.2–30.1 | 20.9/36.1 | 3.6 | 26.6 | 29.7 | 32.7 |
| Temperature P-4 | 29.6 | 29.1–30.1 | 20.8/39.4 | 3.6 | 26.6 | 29.5 | 32.7 |
| Temperature P-5 | 30.7 | 30.2–31.2 | 21.3/36.9 | 3.6 | 27.6 | 30.9 | 33.9 |
| Mean temperature | 30.3 | 29.8–30.8 | 21.1/36.1 | 3.5 | 27.3 | 30.5 | 33.4 |
| First Toenail of Each Foot | Sex | Age | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Thermography (°C) | Mean (Standard Deviation) | Comparison of Means | Mean (Standard Deviation) | Comparison of Means | Mean (Standard Deviation) | Comparison of Means | ||||||
| Right Foot (n = 107) | Left Foot (n = 107) | ES | P-V | Female (n = 148) | Male (n = 66) | ES | P-V | <=21 years (n = 130) | =>22 years (n = 84) | ES | P-V | |
| Temperature P-1 | 31.0 (±3.4) | 30.9 (±3.4) | 0.000 | 0.836 (ns) | 30.8 (±3.5) | 31.2 (±3.3) | 0.004 | 0.368 (ns) | 30.6 (±3.4) | 31.4 (±3.2) | 0.013 | 0.103 (ns) |
| Temperature P-2 | 30.7 (±3.5) | 30.7 (±3.6) | 0.000 | 0.913 (ns) | 30.6 (±3.5) | 31.0 (±3.6) | 0.003 | 0.440 (ns) | 30.3 (±3.5) | 31.3 (±3.3) | 0.021 | 0.034 (*) |
| Temperature P-3 | 29.5 (±3.5) | 29.8 (±3.6) | 0.001 | 0.617 (ns) | 29.5 (±3.6) | 29.9 (±3.4) | 0.003 | 0.453 (ns) | 29.3 (±3.6) | 30.1 (±3.3) | 0.012 | 0.107 (ns) |
| Temperature P-4 | 29.6 (±3.6) | 29.7 (±3.7) | 0.000 | 0.829 (ns) | 29.6 (±3.8) | 29.6 (±3.3) | 0.000 | 0.958 (ns) | 29.3 (±3.7) | 30.2 (±3.4) | 0.014 | 0.084 (ns) |
| Temperature P-5 | 30.7 (±3.6) | 30.7 (±3.5) | 0.000 | 0.995 (ns) | 30.5 (±3.7) | 31.0 (±3.3) | 0.003 | 0.431 (ns) | 30.3 (±3.6) | 31.2 (±3.4) | 0.014 | 0.081 (ns) |
| Mean temperature | 30.3 (±3.5) | 30.3 (±3.5) | 0.000 | 0.932 (ns) | 30.2 (±3.6) | 30.5 (±3.3) | 0.002 | 0.510 (ns) | 30.0 (±3.5) | 30.8 (±3.3) | 0.015 | 0.073 (ns) |
| Fungi | Dermatophytes | Yeast | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Thermography (°C) | Mean (Standard Deviation) | Comparison of Means | Mean (Standard Deviation) | Comparison of Means | Mean (Standard Deviation) | Comparison of Means | ||||||
| P (n = 14) | N (n = 134) | ES | P-V | P (n = 2) | N (n = 146) | ES | P-V | P (n = 12) | N (n = 136) | ES | P-V | |
| Temperature P-1 | 31.0 (±2.8) | 30.8 (±3.5) | 0.021 | 0.398 (ns) | 26.3 (±0.3) | 30.8 (±3.4) | 0.023 | 0.033 (*) | 31.8 (±2.1) | 30.7 (±3.6) | 0.008 | 0.146 (ns) |
| Temperature P-2 | 31.1 (±2.8) | 30.5 (±3.6) | 0.046 | 0.290 (ns) | 26.2 (±0.4) | 30.6 (±3.6) | 0.021 | 0.038 (*) | 31.9 (±2.0) | 30.5 (±3.6) | 0.012 | 0.090 (ⴕ) |
| Temperature P-3 | 30.1 (±3.1) | 29.5 (±3.7) | 0.048 | 0.281 (ns) | 25.6 (±0.4) | 29.6 (±3.6) | 0.016 | 0.060 (ⴕ) | 30.8 (±2.6) | 29.4 (±3.7) | 0.011 | 0.100 (ns) |
| Temperature P-4 | 30.0 (±2.8) | 29.6 (±3.9) | 0.034 | 0.342 (ns) | 26.1 (±0.4) | 29.7 (±3.8) | 0.012 | 0.089 (ⴕ) | 30.7 (±2.4) | 29.5 (±3.9) | 0.007 | 0.157 (ns) |
| Temperature P-5 | 30.9 (±2.9) | 30.5 (±3.7) | 0.031 | 0.354 (ns) | 26.7 (±0.2) | 30.6 (±3.7) | 0.016 | 0.065 (ⴕ) | 31.6 (±2.5) | 30.5 (±3.7) | 0.007 | 0.149 (ns) |
| Mean temperature | 30.6 (±2.8) | 30.2 (±3.6) | 0.037 | 0.329 (ns) | 26.2 (±0.3) | 30.3 (±3.5) | 0.018 | 0.052 (ⴕ) | 31.4 (±2.3) | 30.1 (±3.6) | 0.009 | 0.123 (ns) |
| Fungi | Dermatophytes | Yeast | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Thermography (°C) | Mean (Standard Deviation) | Comparison of Means | Mean (Standard Deviation) | Comparison of Means | Mean (Standard Deviation) | Comparison of Means | ||||||
| P (n = 16) | N (n = 50) | ES | P-V | P (n = 9) | N (n = 57) | ES | P-V | P (n = 7) | N (n = 59) | ES | P-V | |
| Temperature P-1 | 31.0 (±4.0) | 31.3 (±3.1) | 0.002 | 0.362 (ns) | 29.7 (±4.6) | 31.5 (±3.1) | 0.036 | 0.064 (ⴕ) | 32.7 (±2.4) | 31.1 (±3.4) | 0.022 | 0.115 (ns) |
| Temperature P-2 | 30.6 (±4.2) | 31.1 (±3.4) | 0.004 | 0.305 (ns) | 29.2 (±4.9) | 31.2 (±3.3) | 0.037 | 0.060 (ⴕ) | 32.3 (±2.4) | 30.8 (±3.7) | 0.016 | 0.157 (ns) |
| Temperature P-3 | 29.7 (±3.7) | 30.0 (±3.3) | 0.002 | 0.358 (ns) | 28.1 (±3.8) | 30.2 (±3.2) | 0.045 | 0.044 (*) | 31.6 (±2.6) | 29.7 (±3.4) | 0.030 | 0.083 (ⴕ) |
| Temperature P-4 | 26.9 (±3.6) | 29.7 (±3.3) | 0.000 | 0.446 (ns) | 28.3 (±4.0) | 29.9 (±3.2) | 0.027 | 0.084 (ⴕ) | 31.2 (±2.2) | 29.5 (±3.4) | 0.025 | 0.101 (ns) |
| Temperature P-5 | 30.6 (±4.0) | 31.1 (±3.2) | 0.003 | 0.328 (ns) | 29.3 (±4.5) | 31.2 (±3.1) | 0.038 | 0.058 (ⴕ) | 32.3 (±2.6) | 30.8 (±3.4) | 0.019 | 0.132 (ns) |
| Mean temperature | 30.3 (±3.9) | 30.6 (±3.2) | 0.002 | 0.356 (ns) | 28.9 (±4.4) | 30.8 (±3.1) | 0.038 | 0.060 (ⴕ) | 32.0 (±2.4) | 30.4 (±3.4) | 0.023 | 0.112 (ns) |
| Dermatophytes | Yeast | |||||||
|---|---|---|---|---|---|---|---|---|
| Thermography (°C) | Mean (Standard Deviation) | Comparison of Means | Mean (Standard Deviation) | Comparison of Means | ||||
| Positive (n = 11) | Negative (n = 203) | ES | P-V | Positive (n = 19) | Negative (n = 195) | ES | P-V | |
| Temperature P-1 | 29.0 (±4.3) | 31.0 (±3.3) | 0.016 | 0.031 (*) | 32.1 (±2.2) | 30.8 (±3.5) | 0.012 | 0.056 (ⴕ) |
| Temperature P-2 | 28.7 (±4.5) | 30.8 (±3.4) | 0.017 | 0.027 (*) | 32.0 (±2.1) | 30.6 (±3.6) | 0.014 | 0.043 (*) |
| Temperature P-3 | 27.7 (±3.6) | 29.8 (±3.5) | 0.017 | 0.029 (*) | 31.1 (±2.6) | 29.5 (±3.6) | 0.016 | 0.031 (*) |
| Temperature P-4 | 27.9 (±3.7) | 29.7 (±3.6) | 0.013 | 0.050 (*) | 30.9 (±2.2) | 29.5 (±3.7) | 0.011 | 0.061 (ⴕ) |
| Temperature P-5 | 28.8 (±4.2) | 30.8 (±3.5) | 0.014 | 0.041 (*) | 31.9 (±2.5) | 30.6 (±3.6) | 0.011 | 0.064 (ⴕ) |
| Mean temperature | 28.4 (±4.1) | 30.4 (±3.4) | 0.016 | 0.032 (*) | 31.6 (±2.3) | 30.2 (±3.6) | 0.013 | 0.047 (*) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villar Rodríguez, J.; Pérez Pico, A.M.; García Blázquez, F.M.; Morán Cortés, J.F.; Mayordomo Acevedo, R. Evaluation of Thermography as a Diagnostic Technique in Asymptomatic or Incipient Onychomycosis. J. Fungi 2023, 9, 444. https://doi.org/10.3390/jof9040444
Villar Rodríguez J, Pérez Pico AM, García Blázquez FM, Morán Cortés JF, Mayordomo Acevedo R. Evaluation of Thermography as a Diagnostic Technique in Asymptomatic or Incipient Onychomycosis. Journal of Fungi. 2023; 9(4):444. https://doi.org/10.3390/jof9040444
Chicago/Turabian StyleVillar Rodríguez, Julia, Ana María Pérez Pico, Francisco Manuel García Blázquez, Juan Francisco Morán Cortés, and Raquel Mayordomo Acevedo. 2023. "Evaluation of Thermography as a Diagnostic Technique in Asymptomatic or Incipient Onychomycosis" Journal of Fungi 9, no. 4: 444. https://doi.org/10.3390/jof9040444
APA StyleVillar Rodríguez, J., Pérez Pico, A. M., García Blázquez, F. M., Morán Cortés, J. F., & Mayordomo Acevedo, R. (2023). Evaluation of Thermography as a Diagnostic Technique in Asymptomatic or Incipient Onychomycosis. Journal of Fungi, 9(4), 444. https://doi.org/10.3390/jof9040444








