Conidium Specific Polysaccharides in Aspergillus fumigatus
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungal Strains and Growth Media
2.2. Fractionation of Cell Wall Polymers
2.3. Purification of Mannan from the ASSN Fraction of Conidia Cell Wall
2.4. Carbohydrate Analysis of Cell Wall Fractions
2.5. Nuclear Magnetic Resonance (NMR) Analysis
2.6. Statistical Analysis
3. Results
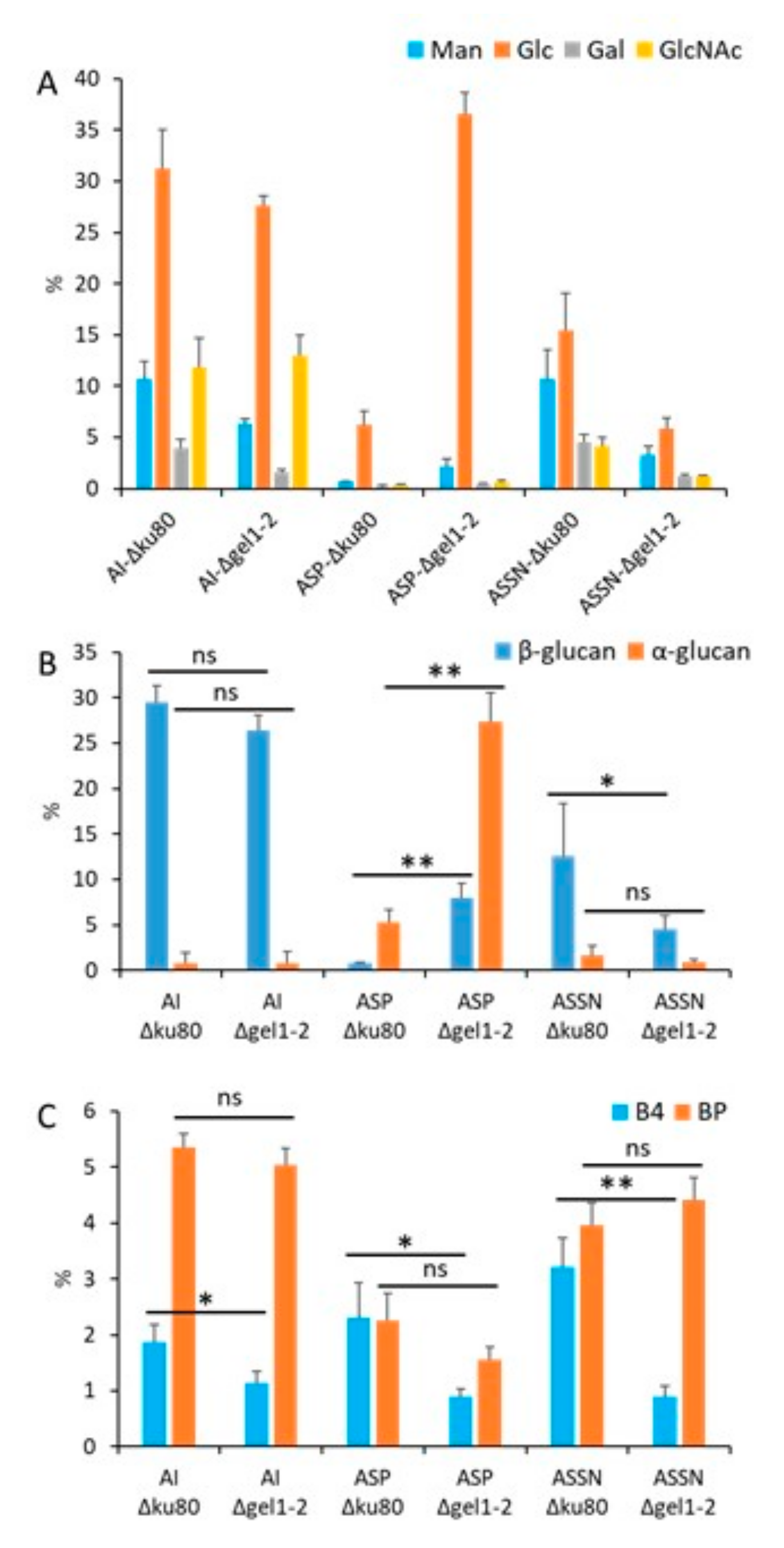
3.1. Polysaccharidome of the Conidial Cell Wall Is Unique
3.2. GH72 Family Is Involved in the β-Glucan Organization of the Conidial Cell Wall
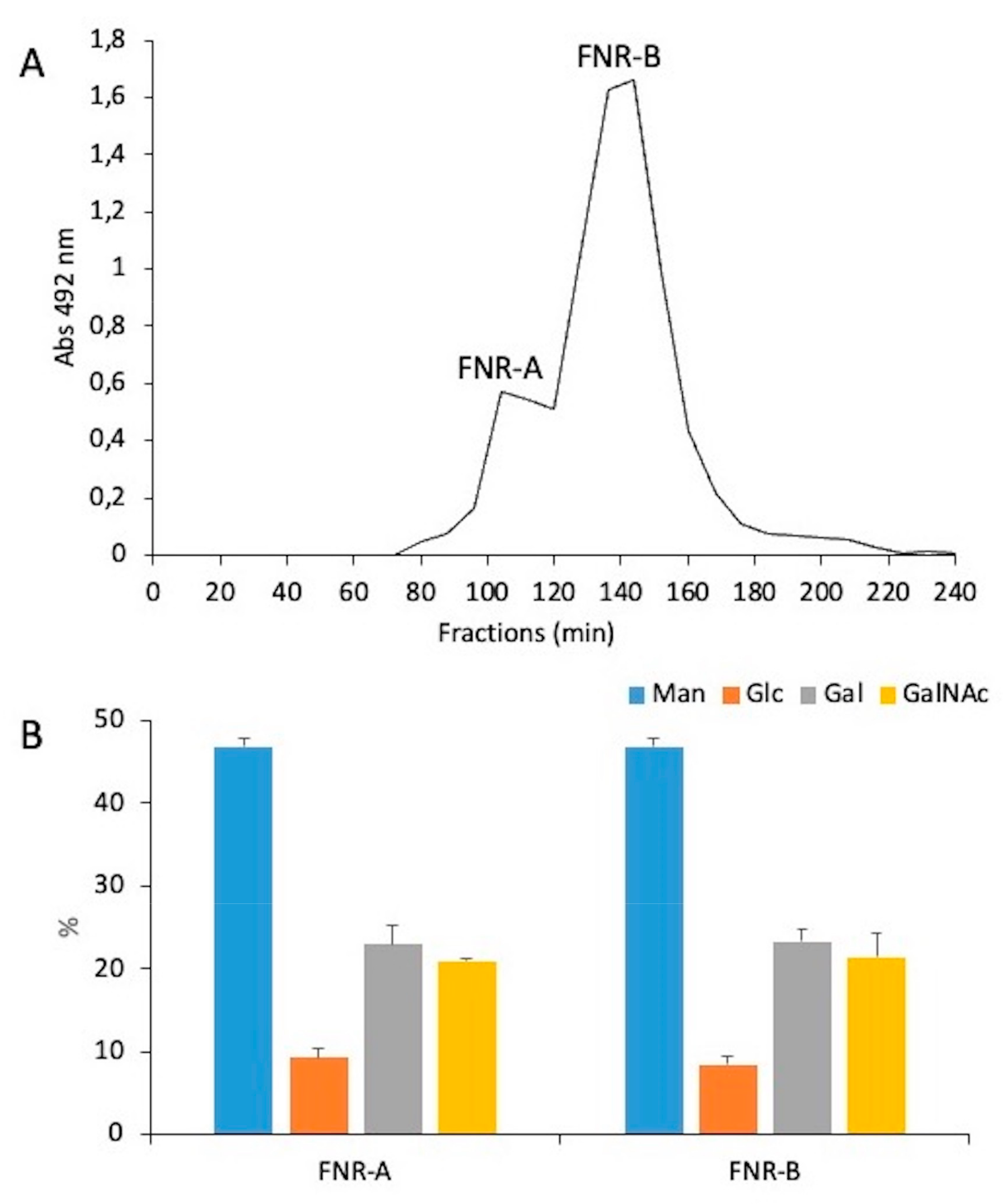
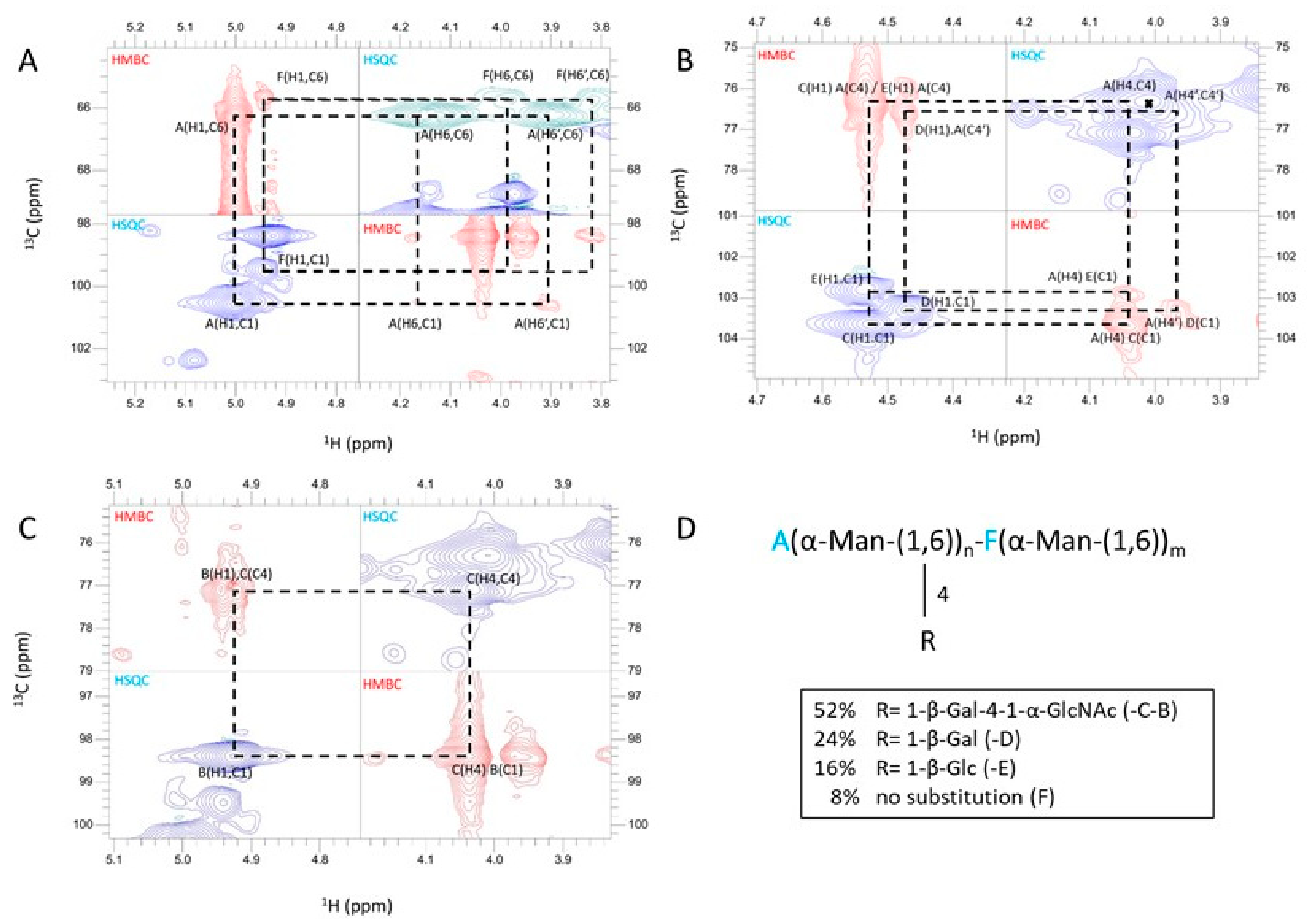
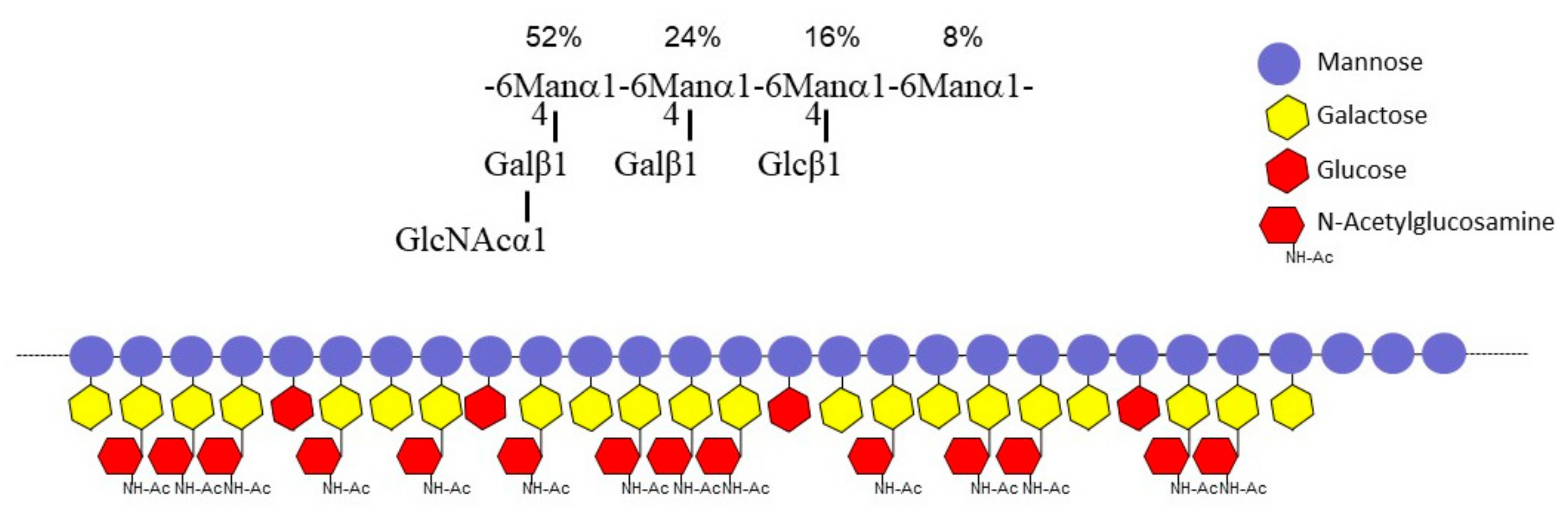
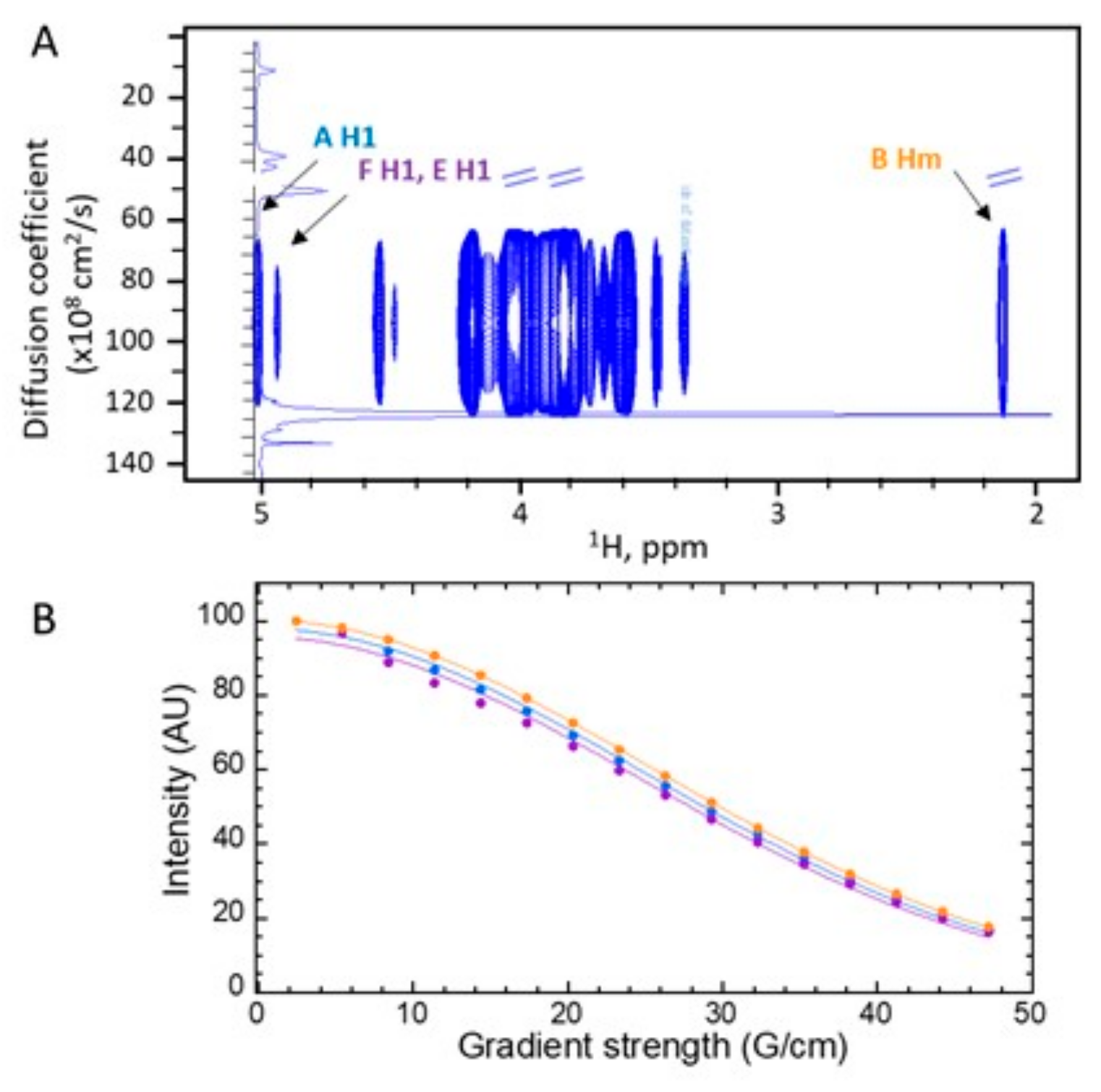
3.3. A New Mannan (G3Man) Structure Isolated from the ASSN Fraction of Conidia
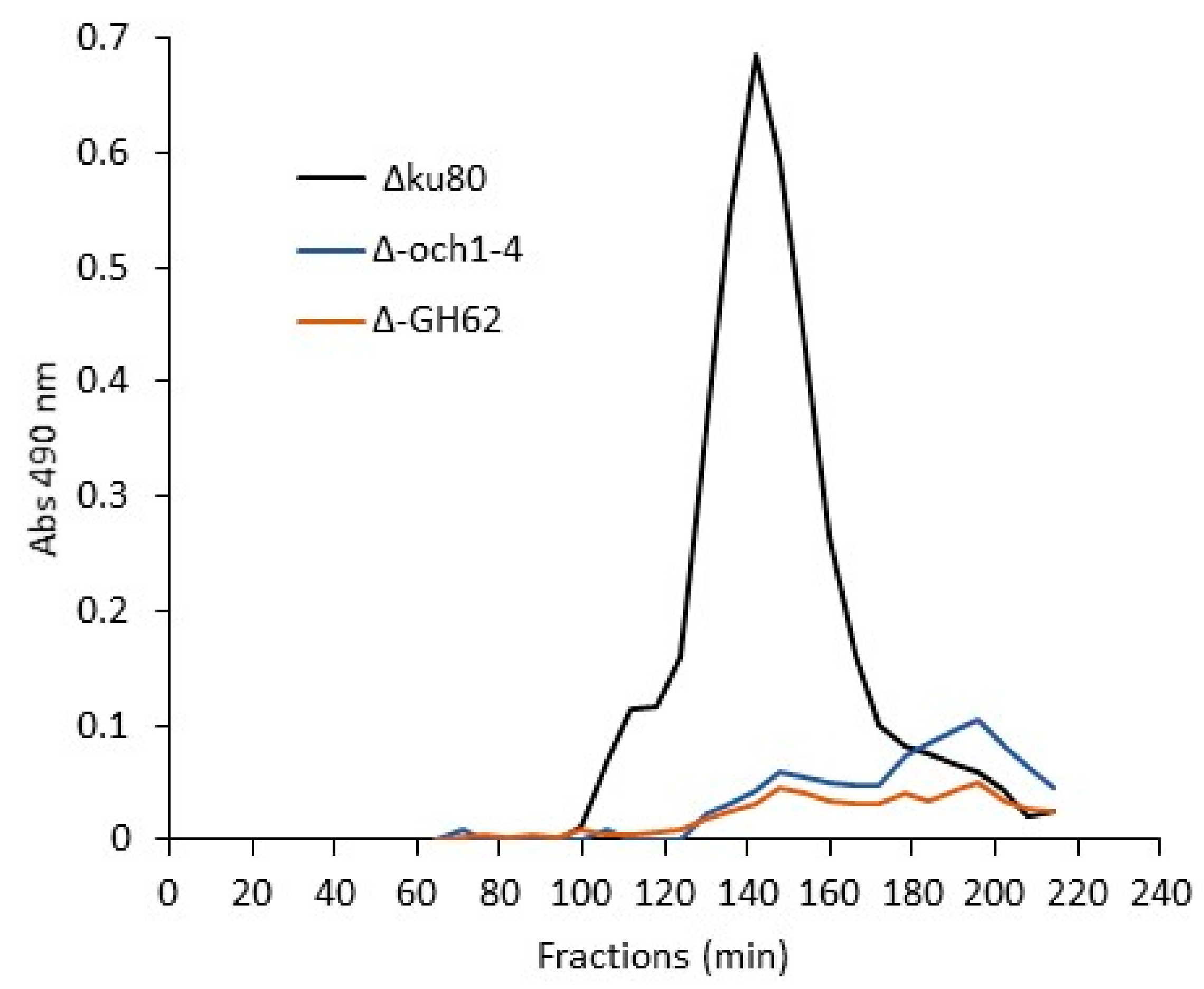
3.4. Biosynthesis of Conidial G3Man Is Dependent on OCH and MNN9/VAN1/ANP1
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Chabi, M.L.; Goracci, A.; Roche, N.; Paugam, A.; Lupo, A.; Revel, M.P. Pulmonary Aspergillosis. Diagn. Interv. Imaging 2015, 96, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Latgé, J.-P.; Chamilos, G. Aspergillus Fumigatus and Aspergillosis in 2019. Clin. Microbiol. Rev. 2019, 33, e00140-18. [Google Scholar] [CrossRef] [PubMed]
- Aimanianda, V.; Bayry, J.; Bozza, S.; Kniemeyer, O.; Perruccio, K.; Elluru, S.R.; Clavaud, C.; Paris, S.; Brakhage, A.A.; Kaveri, S.V.; et al. Surface Hydrophobin Prevents Immune Recognition of Airborne Fungal Spores. Nature 2009, 460, 1117–1121. [Google Scholar] [CrossRef] [PubMed]
- Valsecchi, I.; Dupres, V.; Michel, J.-P.; Duchateau, M.; Matondo, M.; Chamilos, G.; Saveanu, C.; Guijarro, J.I.; Aimanianda, V.; Lafont, F.; et al. The Puzzling Construction of the Conidial Outer Layer of Aspergillus fumigatus. Cell Microbiol. 2019, 21, e12994. [Google Scholar] [CrossRef]
- Bayry, J.; Beaussart, A.; Dufrêne, Y.F.; Sharma, M.; Bansal, K.; Kniemeyer, O.; Aimanianda, V.; Brakhage, A.A.; Kaveri, S.V.; Kwon-Chung, K.J.; et al. Surface Structure Characterization of Aspergillus fumigatus Conidia Mutated in the Melanin Synthesis Pathway and Their Human Cellular Immune Response. Infect. Immun. 2014, 82, 3141–3153. [Google Scholar] [CrossRef]
- Voltersen, V.; Blango, M.G.; Herrmann, S.; Schmidt, F.; Heinekamp, T.; Strassburger, M.; Krüger, T.; Bacher, P.; Lother, J.; Weiss, E.; et al. Proteome Analysis Reveals the Conidial Surface Protein CcpA Essential for Virulence of the Pathogenic Fungus Aspergillus fumigatus. mBio 2018, 9, e01557-18. [Google Scholar] [CrossRef]
- Schmidt, H.; Vlaic, S.; Krüger, T.; Schmidt, F.; Balkenhol, J.; Dandekar, T.; Guthke, R.; Kniemeyer, O.; Heinekamp, T.; Brakhage, A.A. Proteomics of Aspergillus fumigatus Conidia-Containing Phagolysosomes Identifies Processes Governing Immune Evasion. Mol. Cell Proteom. 2018, 17, 1084–1096. [Google Scholar] [CrossRef]
- Thywißen, A.; Heinekamp, T.; Dahse, H.-M.; Schmaler-Ripcke, J.; Nietzsche, S.; Zipfel, P.F.; Brakhage, A.A. Conidial Dihydroxynaphthalene Melanin of the Human Pathogenic Fungus Aspergillus fumigatus Interferes with the Host Endocytosis Pathway. Front. Microbiol. 2011, 2, 96. [Google Scholar] [CrossRef]
- Fontaine, T.; Delangle, A.; Simenel, C.; Coddeville, B.; van Vliet, S.J.; van Kooyk, Y.; Bozza, S.; Moretti, S.; Schwarz, F.; Trichot, C.; et al. Galactosaminogalactan, a New Immunosuppressive Polysaccharide of Aspergillus fumigatus. PLoS Pathog. 2011, 7, e1002372. [Google Scholar] [CrossRef]
- Mouyna, I.; Fontaine, T. Cell Wall of Aspergillus fumigatus: A Dynamic Structure. In Aspergillus fumigatus and Aspergillosis; Wiley: Hoboken, NJ, USA, 2009; pp. 169–183. [Google Scholar] [CrossRef]
- Henry, C.; Li, J.; Danion, F.; Alcazar-Fuoli, L.; Mellado, E.; Beau, R.; Jouvion, G.; Latgé, J.-P.; Fontaine, T. Two KTR Mannosyltransferases Are Responsible for the Biosynthesis of Cell Wall Mannans and Control Polarized Growth in Aspergillus fumigatus. mBio 2019, 10, e02647-18. [Google Scholar] [CrossRef]
- Henry, C.; Fontaine, T.; Heddergott, C.; Robinet, P.; Aimanianda, V.; Beau, R.; Beauvais, A.; Mouyna, I.; Prevost, M.-C.; Fekkar, A.; et al. Biosynthesis of Cell Wall Mannan in the Conidium and the Mycelium of Aspergillus fumigatus. Cell Microbiol. 2016, 18, 1881–1891. [Google Scholar] [CrossRef]
- Da Silva Ferreira, M.E.; Kress, M.R.V.Z.; Savoldi, M.; Goldman, M.H.S.; Härtl, A.; Heinekamp, T.; Brakhage, A.A.; Goldman, G.H. The AkuBKU80 Mutant Deficient for Nonhomologous End Joining Is a Powerful Tool for Analyzing Pathogenicity in Aspergillus fumigatus. Eukaryot. Cell 2006, 5, 207–211. [Google Scholar] [CrossRef]
- Cove, D.J. The Induction and Repression of Nitrate Reductase in the Fungus Aspergillus nidulans. Biochim. Biophys. Acta 1966, 113, 51–56. [Google Scholar] [CrossRef]
- Gastebois, A.; Mouyna, I.; Simenel, C.; Clavaud, C.; Coddeville, B.; Delepierre, M.; Latgé, J.-P.; Fontaine, T. Characterization of a New β(1–3)-Glucan Branching Activity of Aspergillus fumigatus. J. Biol. Chem. 2010, 285, 2386–2396. [Google Scholar] [CrossRef]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Sawardeker, J.S.; Sloneker, J.H.; Jeanes, A. Quantitative Determination of Monosaccharides as Their Alditol Acetates by Gas Liquid Chromatography. Anal. Chem. 1965, 37, 1602–1604. [Google Scholar] [CrossRef]
- Ciucanu, I.; Kerek, F. A Simple and Rapid Method for the Permethylation of Carbohydrates. Carbohydr. Res. 1984, 131, 209–217. [Google Scholar] [CrossRef]
- Fontaine, T.; Simenel, C.; Dubreucq, G.; Adam, O.; Delepierre, M.; Lemoine, J.; Vorgias, C.E.; Diaquin, M.; Latgé, J.P. Molecular Organization of the Alkali-Insoluble Fraction of Aspergillus fumigatus Cell Wall. J. Biol. Chem. 2000, 275, 27594–27607. [Google Scholar] [CrossRef]
- Fontaine, T.; Beauvais, A.; Loussert, C.; Thevenard, B.; Fulgsang, C.C.; Ohno, N.; Clavaud, C.; Prevost, M.-C.; Latgé, J.-P. Cell Wall A1-3glucans Induce the Aggregation of Germinating Conidia of Aspergillus fumigatus. Fungal Genet. Biol. 2010, 47, 707–712. [Google Scholar] [CrossRef]
- Lever, M. A New Reaction for Colorimetric Determination of Carbohydrates. Anal. Biochem. 1972, 47, 273–279. [Google Scholar] [CrossRef]
- Ancian, B.; Bourgeois, I.; Dauphin, J.-F.; Shaw, A.A. Artifact-Free Pure Absorption PFG-Enhanced DQF-COSY Spectra Including a Gradient Pulse in the Evolution Period. J. Magn. Reson. 1997, 125, 348–354. [Google Scholar] [CrossRef]
- Palmer, A.G.; Cavanagh, J.; Wright, P.E.; Rance, M. Sensitivity Improvement in Proton-Detected Two-Dimensional Heteronuclear Correlation NMR Spectroscopy. J. Magn. Reson. (1969) 1991, 93, 151–170. [Google Scholar] [CrossRef]
- Nyberg, N.T.; Duus, J.O.; Sørensen, O.W. Heteronuclear Two-Bond Correlation: Suppressing Heteronuclear Three-Bond or Higher NMR Correlations While Enhancing Two-Bond Correlations Even for Vanishing 2J(CH). J. Am. Chem. Soc. 2005, 127, 6154–6155. [Google Scholar] [CrossRef] [PubMed]
- Cicero, D.O.; Barbato, G.; Bazzo, R. Sensitivity Enhancement of a Two-Dimensional Experiment for the Measurement of Heteronuclear Long-Range Coupling Constants, by a New Scheme of Coherence Selection by Gradients. J. Magn. Reson. 2001, 148, 209–213. [Google Scholar] [CrossRef]
- Kay, L.; Keifer, P.; Saarinen, T. Pure Absorption Gradient Enhanced Heteronuclear Single Quantum Correlation Spectroscopy with Improved Sensitivity. J. Am. Chem. Soc. 1992, 114, 10663–10665. [Google Scholar] [CrossRef]
- Jerschow, A.; Müller, N. Suppression of Convection Artifacts in Stimulated-Echo Diffusion Experiments. Double-Stimulated-Echo Experiments. J. Magn. Reson. 1997, 125, 372–375. [Google Scholar] [CrossRef]
- Chakraborty, A.; Fernando, L.D.; Fang, W.; Dickwella Widanage, M.C.; Wei, P.; Jin, C.; Fontaine, T.; Latgé, J.-P.; Wang, T. A Molecular Vision of Fungal Cell Wall Organization by Functional Genomics and Solid-State NMR. Nat. Commun. 2021, 12, 6346. [Google Scholar] [CrossRef]
- Muszkieta, L.; Fontaine, T.; Beau, R.; Mouyna, I.; Vogt, M.S.; Trow, J.; Cormack, B.P.; Essen, L.-O.; Jouvion, G.; Latgé, J.-P. The Glycosylphosphatidylinositol-Anchored DFG Family Is Essential for the Insertion of Galactomannan into the β-(1,3)-Glucan–Chitin Core of the Cell Wall of Aspergillus fumigatus. mSphere 2019, 4, e00397-19. [Google Scholar] [CrossRef]
- Lamarre, C.; Beau, R.; Balloy, V.; Fontaine, T.; Hoi, J.W.S.; Guadagnini, S.; Berkova, N.; Chignard, M.; Beauvais, A.; Latgé, J.-P. Galactofuranose Attenuates Cellular Adhesion of Aspergillus fumigatus. Cell Microbiol. 2009, 11, 1612–1623. [Google Scholar] [CrossRef]
- Lambou, K.; Perkhofer, S.; Fontaine, T.; Latge, J.-P. Comparative Functional Analysis of the OCH1 Mannosyltransferase Families in Aspergillus fumigatus and Saccharomyces cerevisiae. Yeast 2010, 27, 625–636. [Google Scholar] [CrossRef]
- Leal, J.A.; Prieto, A.; Bernabé, M.; Hawksworth, D.L. An Assessment of Fungal Wall Heteromannans as a Phylogenetically Informative Character in Ascomycetes. FEMS Microbiol. Rev. 2010, 34, 986–1014. [Google Scholar] [CrossRef]
- Gemmill, T.R.; Trimble, R.B. Overview of N- and O-Linked Oligosaccharide Structures Found in Various Yeast Species. Biochim. Biophys. Acta-Gen. Subj. 1999, 1426, 227–237. [Google Scholar] [CrossRef]
- Gemmill, T.R.; Trimble, R.B. All Pyruvylated Galactose in Schizosaccharomyces Pombe N-Glycans Is Present in the Terminal Disaccharide, 4, 6-O-[(R)-(1-Carboxyethylidine)]-Galbeta1,3Galalpha1-. Glycobiology 1998, 8, 1087–1095. [Google Scholar] [CrossRef]
- Ballou, C.E. [36] Isolation, Characterization, and Properties of Saccharomyces Cerevisiae Mnn Mutants with Nonconditional Protein Glycosylation Defects. In Methods in Enzymology; Gene Expression Technology; Academic Press: Cambridge, MA, USA, 1990; Volume 185, pp. 440–470. [Google Scholar]
- Nelson, R.D.; Shibata, N.; Podzorski, R.P.; Herron, M.J. Candida Mannan: Chemistry, Suppression of Cell-Mediated Immunity, and Possible Mechanisms of Action. Clin. Microbiol. Rev. 1991, 4, 1–19. [Google Scholar] [CrossRef]
- Orlean, P. Architecture and Biosynthesis of the Saccharomyces cerevisiae Cell Wall. Genetics 2012, 192, 775–818. [Google Scholar] [CrossRef]
- Hall, R.A.; Gow, N.A.R. Mannosylation in Candida Albicans: Role in Cell Wall Function and Immune Recognition. Mol. Microbiol. 2013, 90, 1147–1161. [Google Scholar] [CrossRef]
- Mouyna, I.; Aimanianda, V.; Hartl, L.; Prevost, M.-C.; Sismeiro, O.; Dillies, M.-A.; Jagla, B.; Legendre, R.; Coppee, J.-Y.; Latgé, J.-P. GH16 and GH81 Family β-(1,3)-Glucanases in Aspergillus fumigatus Are Essential for Conidial Cell Wall Morphogenesis. Cell Microbiol. 2016, 18, 1285–1293. [Google Scholar] [CrossRef]
- Onoue, T.; Tanaka, Y.; Hagiwara, D.; Ekino, K.; Watanabe, A.; Ohta, K.; Kamei, K.; Shibata, N.; Goto, M.; Oka, T. Identification of Two Mannosyltransferases Contributing to Biosynthesis of the Fungal-Type Galactomannan α-Core-Mannan Structure in Aspergillus fumigatus. Sci. Rep. 2018, 8, 16918. [Google Scholar] [CrossRef]
- Wagener, J.; Echtenacher, B.; Rohde, M.; Kotz, A.; Krappmann, S.; Heesemann, J.; Ebel, F. The Putative α-1,2-Mannosyltransferase AfMnt1 of the Opportunistic Fungal Pathogen Aspergillus fumigatus Is Required for Cell Wall Stability and Full Virulence. Eukaryot. Cell 2008, 7, 1661–1673. [Google Scholar] [CrossRef]
- Kadooka, C.; Hira, D.; Tanaka, Y.; Chihara, Y.; Goto, M.; Oka, T. Mnt1, an α-(1 → 2)-Mannosyltransferase Responsible for the Elongation of N-Glycans and O-Glycans in Aspergillus fumigatus. Glycobiology 2022, 32, 1137–1152. [Google Scholar] [CrossRef]
- Kadooka, C.; Hira, D.; Tanaka, Y.; Miyazawa, K.; Bise, M.; Takatsuka, S.; Oka, T. Identification of an α-(1→6)-Mannosyltransferase Contributing To Biosynthesis of the Fungal-Type Galactomannan α-Core-Mannan Structure in Aspergillus fumigatus. mSphere 2022, 7, e00484-22. [Google Scholar] [CrossRef] [PubMed]
- Latgé, J.P.; Kobayashi, H.; Debeaupuis, J.P.; Diaquin, M.; Sarfati, J.; Wieruszeski, J.M.; Parra, E.; Bouchara, J.P.; Fournet, B. Chemical and Immunological Characterization of the Extracellular Galactomannan of Aspergillus fumigatus. Infect. Immun. 1994, 62, 5424–5433. [Google Scholar] [CrossRef] [PubMed]
- Costachel, C.; Coddeville, B.; Latgé, J.-P.; Fontaine, T. Glycosylphosphatidylinositol-Anchored Fungal Polysaccharide in Aspergillus fumigatus. J. Biol. Chem. 2005, 280, 39835–39842. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, T.; Latgé, J.-P. Galactomannan Produced by Aspergillus fumigatus: An Update on the Structure, Biosynthesis and Biological Functions of an Emblematic Fungal Biomarker. J. Fungi 2020, 6, 283. [Google Scholar] [CrossRef] [PubMed]
- Millet, N.; Latgé, J.-P.; Mouyna, I. Members of Glycosyl-Hydrolase Family 17 of A. fumigatus Differentially Affect Morphogenesis. J. Fungi 2018, 4, 18. [Google Scholar] [CrossRef]
- Millet, N.; Moya-Nilges, M.; Sachse, M.; Krijnse Locker, J.; Latgé, J.-P.; Mouyna, I. Aspergillus fumigatus Exoβ(1-3)Glucanases Family GH55 Are Essential for Conidial Cell Wall Morphogenesis. Cell Microbiol. 2019, 21, e13102. [Google Scholar] [CrossRef]
- Aimanianda, V.; Simenel, C.; Garnaud, C.; Clavaud, C.; Tada, R.; Barbin, L.; Mouyna, I.; Heddergott, C.; Popolo, L.; Ohya, Y.; et al. The Dual Activity Responsible for the Elongation and Branching of β-(1,3)-Glucan in the Fungal Cell Wall. MBio 2017, 8, 00619-17. [Google Scholar] [CrossRef]
- Mouyna, I.; Fontaine, T.; Vai, M.; Monod, M.; Fonzi, W.A.; Diaquin, M.; Popolo, L.; Hartland, R.P.; Latgé, J.P. Glycosylphosphatidylinositol-Anchored Glucanosyltransferases Play an Active Role in the Biosynthesis of the Fungal Cell Wall. J. Biol. Chem. 2000, 275, 14882–14889. [Google Scholar] [CrossRef]
- Mouyna, I.; Morelle, W.; Vai, M.; Monod, M.; Léchenne, B.; Fontaine, T.; Beauvais, A.; Sarfati, J.; Prévost, M.-C.; Henry, C.; et al. Deletion of GEL2 Encoding for a Beta(1-3)Glucanosyltransferase Affects Morphogenesis and Virulence in Aspergillus fumigatus. Mol. Microbiol. 2005, 56, 1675–1688. [Google Scholar] [CrossRef]
- Valiante, V.; Macheleidt, J.; Föge, M.; Brakhage, A.A. The Aspergillus fumigatus Cell Wall Integrity Signaling Pathway: Drug Target, Compensatory Pathways, and Virulence. Front. Microbiol. 2015, 6, 325. [Google Scholar] [CrossRef]
- Park, J.; Hulsman, M.; Arentshorst, M.; Breeman, M.; Alazi, E.; Lagendijk, E.L.; Rocha, M.C.; Malavazi, I.; Nitsche, B.M.; van den Hondel, C.A.M.J.J.; et al. Transcriptomic and Molecular Genetic Analysis of the Cell Wall Salvage Response of Aspergillus niger to the Absence of Galactofuranose Synthesis. Cell Microbiol. 2016, 18, 1268–1284. [Google Scholar] [CrossRef]
- Gastebois, A.; Fontaine, T.; Latgé, J.-P.; Mouyna, I. β(1-3)Glucanosyltransferase Gel4p Is Essential for Aspergillus fumigatus. Eukaryot. Cell 2010, 9, 1294–1298. [Google Scholar] [CrossRef]
- Danion, F.; van Rhijn, N.; Dufour, A.C.; Legendre, R.; Sismeiro, O.; Varet, H.; Olivo-Marin, J.-C.; Mouyna, I.; Chamilos, G.; Bromley, M.; et al. Aspergillus fumigatus, One Uninucleate Species with Disparate Offspring. J. Fungi 2021, 7, 30. [Google Scholar] [CrossRef]
- Dague, E.; Alsteens, D.; Latgé, J.-P.; Verbelen, C.; Raze, D.; Baulard, A.R.; Dufrêne, Y.F. Chemical Force Microscopy of Single Live Cells. Nano Lett. 2007, 7, 3026–3030. [Google Scholar] [CrossRef]
- Gersuk, G.M.; Underhill, D.M.; Zhu, L.; Marr, K.A. Dectin-1 and TLRs Permit Macrophages to Distinguish between Different Aspergillus fumigatus Cellular States. J. Immunol. 2006, 176, 3717–3724. [Google Scholar] [CrossRef]

| Methyl-Ethers 1 | Linkages | FNR-A (%) 2 | FNR-B (%) 2 |
|---|---|---|---|
| 2,3,4,6-Glc | t-Glc | 9.7 | 9.6 |
| 2,3,4,6-Gal | t-Gal | 8.0 | 8.9 |
| 2,3,6-Gal | 4-Gal | 22.8 | 17.5 |
| 2,3,4-Man | 6-Man | 5.8 | 7.1 |
| 2,3-Man | 4,6-Man | 47.0 | 50.6 |
| 3,4,6-GlcNAc | t-GlcNAc | 6.7 | 6.1 |
| Residue | H1, C1 3JH1/H2 | H2, C2 | H3, C3 | H4, C4 | H5, C5 | H6, H6’, C6 | Hm, Cm |
|---|---|---|---|---|---|---|---|
| A -6,4)-Man-α(1- | 5.00 100.6 1.8 Hz | 4.17 70.0 | 4.00 70.4 | 4.01 76.3 | 4.00 69.5 | 3.90 4.14 66.2 | |
| B GlcNAc-α(1- | 4.93 98.4 ~2 Hz | 3.95 54.1 | 3.84 70.6 | 3.58 69.9 | 4.18 72.2 | 3.86 3.86 60.3 | 2.12 22.0 |
| C -4)-Gal-β(1- | 4.53 103.7 8.9 Hz | 3.61 70.94 | 3.78 72.5 | 4.04 77.1 | 3.79 72.5 | 3.66 3.66 70.8 | |
| D Gal-β(1- | 4.47 103.3 8.5 Hz | 3.58 71.1 | 3.72 72.9 | 3.98 68.8 | 3.72 72.9 | 3.62 3.62 71.1 | |
| E Glc-β(1- | 4.54 102.8 9.2 Hz | 3.36 73.3 | 3.57 76.0 | 3.46 69.8 | 3.56 76.4 | 3.78 3.98 61.0 | |
| F -6)-Man-α(1- | 4.94 99.5 ~2 Hz | 4.03 70.1 | 3.87 71.0 | 3.77 66.7 | 3.88 70.9 | 3.83 3.97 65.7 |
| Conidium | Mycelium | |
|---|---|---|
| β-glucans b | 49 | 33 |
| α-(1,3)-glucans | 7 | 30 |
| chitin | 10 | 18 |
| GAG | - | 7 |
| GM | 15 | 11 |
| G3Man | 18 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.; Valsecchi, I.; Le Meur, R.A.; Simenel, C.; Guijarro, J.I.; Comte, C.; Muszkieta, L.; Mouyna, I.; Henrissat, B.; Aimanianda, V.; et al. Conidium Specific Polysaccharides in Aspergillus fumigatus. J. Fungi 2023, 9, 155. https://doi.org/10.3390/jof9020155
Liu Z, Valsecchi I, Le Meur RA, Simenel C, Guijarro JI, Comte C, Muszkieta L, Mouyna I, Henrissat B, Aimanianda V, et al. Conidium Specific Polysaccharides in Aspergillus fumigatus. Journal of Fungi. 2023; 9(2):155. https://doi.org/10.3390/jof9020155
Chicago/Turabian StyleLiu, Zhonghua, Isabel Valsecchi, Rémy A. Le Meur, Catherine Simenel, J. Iñaki Guijarro, Catherine Comte, Laetitia Muszkieta, Isabelle Mouyna, Bernard Henrissat, Vishukumar Aimanianda, and et al. 2023. "Conidium Specific Polysaccharides in Aspergillus fumigatus" Journal of Fungi 9, no. 2: 155. https://doi.org/10.3390/jof9020155
APA StyleLiu, Z., Valsecchi, I., Le Meur, R. A., Simenel, C., Guijarro, J. I., Comte, C., Muszkieta, L., Mouyna, I., Henrissat, B., Aimanianda, V., Latgé, J.-P., & Fontaine, T. (2023). Conidium Specific Polysaccharides in Aspergillus fumigatus. Journal of Fungi, 9(2), 155. https://doi.org/10.3390/jof9020155








