1. Introduction
Phytophthora cactorum (Lebert et Cohn) Schroet., a fungus-like organism from the family Peronosporaceae with a cosmopolitan distribution, is one of the most important plant pathogens [
1]. In 1870, Lebert and Cohn in Central Europe first discovered
P. cactorum on cacti
Carnegiea gigantea (Engelm). Britton and Rose discovered it on
Melocactus nigrotomentosus [
2]. This pathogen has a wide host range, including more than 200 plant species [
3]. This species poses a serious danger to plants and trees in ornamental horticulture, and has the potential to decrease the production of many economically important crops such as apples, pears and strawberries, causing rotting of the roots, crowns and collars, and fruit infections [
4]. In strawberries,
P. cactorum is responsible for crown rot and leather rot which is associated with major economic losses for the agricultural sector [
5]. As this pathogen is one of the major limiting factors in the commercial strawberry production system worldwide [
6], this has instigated an effort to restrict its distribution, spread and incidence in the fields.
The exact origin and natural distribution of many
Phytophthora spp. is difficult to assess because the human-caused global movement of plants, including those asymptomatically infected, has distributed these pathogen species worldwide [
7,
8]. For example,
P. nicotianae was reported for the first time in Indonesia in 1895 [
9]; in the USA it was described in 1915 [
3], but its presence in European greenhouses has been confirmed since 1927 [
10]. Goss et al. [
11] described
P. ramorum migrations between Europe, the USA and Canada, but the unclear origin and spread of this pathogen still remains under investigation. The worldwide migration and importance of
P. infestans is also notorious [
12].
The first disease incidence of leather rot caused by
P. cactorum was reported in the United States in 1924 [
13]; other cases were gradually reported in Europe and also in Asia, while the first description of crown rot was reported in 1952 in Germany [
14], in 1988 in Sweden [
15], in 1990 in Finland [
16] and in 1992 in Norway [
17]. Although the widespread distribution of this pathogen in Europe was reported by the aforementioned researchers,
P. cactorum is considered an alien species to the European continent, which is evidenced by its low genetic variability, close phylogenetic relatedness to other non-native species, and high virulence to host plant species which are indigenous to Europe [
1,
18].
The combination of cultural practices and chemical control methods used to be considered an efficient way to reduce the disease incidence rate [
19,
20]. Fungicides like mefenoxam, azoxystrobin, pyraclostrobin [
21] and dimethomorph [
22] were most effective in controlling
P. cactorum infections, but soon after their commercial release, the development of a resistance to fungicides became a serious problem [
23]. The rise of resistance depends on the frequency and dose of applications of fungicides [
24]. The resistance of
P. cactorum against metalaxyl was first reported in 1988 by Utkhede and Gupta in an in vitro environment [
25], while the resistance of
P. cactorum isolates to mefenoxam in strawberry fields in the USA was reported for the first time by Jeffers et al. in 2004 [
26]. Recently, Marin and Peres [
27] reported that the fungicides cymoxanil, fluopicolide, mandipropamid and oxathiapiprolin completely inhibited the mycelial growth of
P. cactorum isolates from Florida at a concentration of 1 µg/mL, but significant differences in the resistance to some chemical fungicides have already been found between
P. cactorum isolates or their genetic lineages [
28,
29,
30,
31,
32,
33,
34].
P. cactorum strains have been reported as host-specific by various researchers. Van Der Scheer [
32], Harris and Stickels [
33] and Hantula et al. [
4] tested the infection potential of
P. cactorum isolated from different hosts to cause crown rot in strawberries, and they concluded that crown rot can only be caused by isolates, isolated from strawberry crowns. Seemüller and Schmidle [
34] found that
P. cactorum strains isolated from strawberry crowns were less pathogenic on apple bark tissue, and vice versa. They also mentioned that
P. cactorum isolates from any other host are capable of causing leather rot in strawberry fruit, but not crown rot. Similar conclusions were reported by Belisario et al. [
35], Oudemans and Coffey [
36] and Cooke et al. [
37]. The more detailed population structure of
P. cactorum was described by Pánek et al. [
38], differentiating four genetic lineages of a complex of
P. cactorum based on isolates originating from woody plants. Recently, isolates from strawberry plants from central Europe were described as another distant genetic lineage [
39], also confirming the previous conclusions of lineage differentiation.
The current study was designed to evaluate the sensitivity pattern of
P. cactorum isolates from different hosts, distinguishing strawberry and non-strawberry isolates. In the current study, the effects of seven active ingredients of fungicides with a different mode of action, i.e., azoxystrobin, cymoxanil, dimethomorph, fluopicolide, fenamidone, metalaxyl and propamocarb, on the mycelial growth inhibition of
P. cactorum isolates, were evaluated. Four of these fungicides were evaluated as effective and have subsequently been chosen for tests of inhibition of sporangial development and the release of zoospores from sporangia. Additionally, we compared the relations between isolates based on their response to fungicides to relations based on their genetic distances published earlier [
39].
4. Discussion
The genus
Phytophthora includes the most significant plant pathogens worldwide, which endanger the supply of the most important crops such as potato, soya and many others [
46]. The suppression of their infection on various crops mainly relies on the use of fungicides, and the production of some crops is unimaginable without their use [
47]. Such a widespread use of these fungicides resulted in the rapid rise of resistance against them in various species of
Phytophthora, soon after their commercial release [
48,
49]; the populations of many
Phytophthora spp. include some number of genotypes resistant to various fungicides [
49,
50]. Our results also confirmed the presence of resistant strains of
P. cactorum in Czech strawberry fields. Resistance against fungicides is distributed unevenly, and in some measure reflects the host-specific groups of
P. cactorum; various fungicides are also unequally efficient against different life stages.
The most obvious is the variability of resistance against metalaxyl, which has been documented in the isolates we tested. The isolates are either fully sensitive or highly resistant, which is in accordance with the assumed monogenic nature of resistance to this fungicide [
51,
52]. In our analyses, only four isolates (19%) originating from strawberry plants in the Czech Republic (S) were resistant, while all of those from woody plants (W) were normally sensitive. Resistance against this fungicide is well documented in many
Phytophthora spp. [
53,
54,
55]. Two out of the four isolates resistant to metalaxyl that we found are also resistant to dimethomorph. These two multi-resistant isolates originate in the same locality, together with one isolate resistant only to dimethomorph and one isolate sensitive to both of these fungicides. The uneven distribution of resistance to metalaxyl could indicate the different origin of the isolates, as mentioned by [
56] on
P. infestans. The common presence of both resistant and sensitive isolates in one locality (
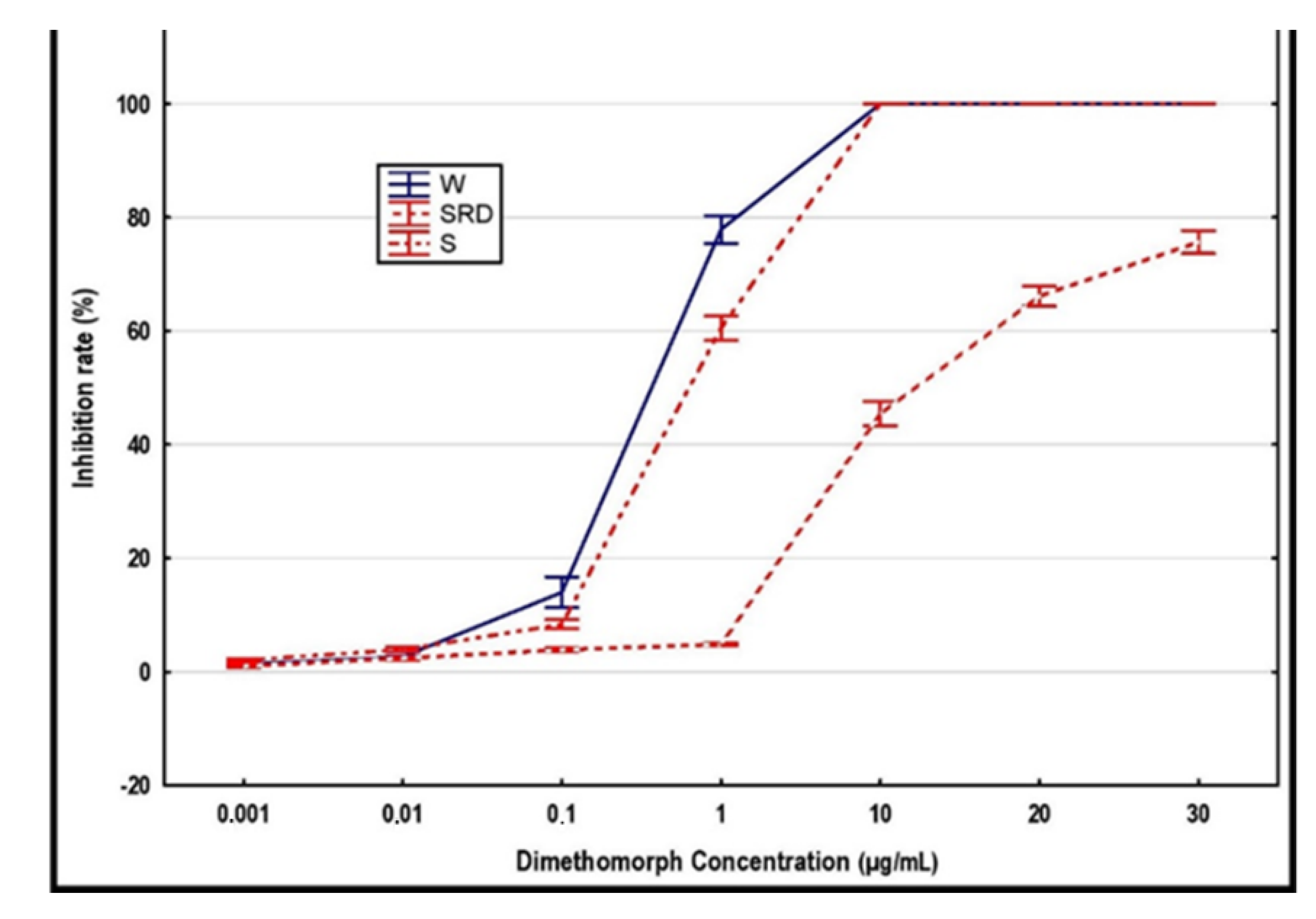
Table 3) suggests either at least two independent introductions into the locality, or the rapid development of resistance in some of the strains present. Since not all of the isolates resistant against one of these two fungicides are also resistant against the second fungicide, their multidrug resistance does not match the cross-resistance, which is also ruled out by the different mode of action (discussed below) of these fungicides. The formation of resistance against dimethomorph has been considered improbable due to the polygenic nature of such resistance [
57], although the dimethomorph-resistant mutants were acquired in laboratory conditions [
58,
59]. The results of current analyses show the resistance formation in
P. cactorum isolates in field conditions, although the number of such events is unclear, considering at least two different types of isolates resistant to this fungicide are present together in one locality (18_07_2S12 is resistant to dimethomorph + metalaxyl; 18_07_6 is resistant only to dimethomorph). Both the dimethomorph- and metalaxyl-resistant isolates that originate from
P. cactorum populations in strawberry fields in the Czech Republic, are considered to belong to the genetically homogenous lineage “S” [
39]. The variability in response to these fungicides is somewhat inconsistent with the low genetic diversity in that group.
Fluopicolide was evaluated as efficient against all of the life stages of
P. cactorum we tested. We did not find any considerably resistant isolate, represented by some exceptionally high values of EC
50, although resistance to this fungicide has already been described in various
Phytophthora spp. [
60,
61,
62]. Unlike the example of the two previous fungicides, sensitivity against fluopicolide did not differ substantially between the isolate groups S and W. However, two isolates of the W group were substantially more sensitive to this fungicide in comparison to the rest of the isolates, which evokes the question of which resistance level is common in
P. cactorum. Except for one, all of the the
P. cactorum isolates included in the study originated in Europe, where this species is assumed to be non-indigenous [
1], and the origin of the
P. cactorum species is unknown. Therefore, using an isolate from the natural population to determine the natural level of sensitivity was not possible. These natural levels of sensitivity expressed as EC
50 values determined in wild populations of
P. capsici, P. nicotianae and
P. erythroseptica [
43,
44,
45] were lower than 0.3 μg/mL, while the EC
50 values of resistant mutants were on the level of units to hundreds of μg/mL [
43]. The EC
50 values we calculated for our
P. cactorum isolates (ca. 0.7-4.0 μg/mL;
Table 1) were somewhat larger in comparison to the values published for wild populations of the other
Phytophthora spp. The resistance level of all
P. cactorum isolates against fluopicolide was also evaluated by the estimated values of the resistance factors (RF; the rate of EC
50 of a particular isolate to the EC
50 of a strain from the wild population) [
43], by comparing the calculated EC
50 of
P. cactorum to the known values of other mentioned
Phytophthora spp. These RF values were between 2.0 and 66.7, which corresponds to intermediate resistance according to Lu et al. [
43] and indicates an increase compared to the theoretical natural level. All but two
P. cactorum isolates we tested had more or less increased resistance, which is not entirely consistent with the assumed low risk of resistance development against fluopicolide for
Phytophthora spp. [
45]; however, this fungicide is still able to inhibit the growth of
P. cactorum isolates.
The rest of the tested fungicides—azoxystrobin, fenamidone, cymoxanil and propamocarb—were evaluated as inefficient in the mycelial growth assay. Their effect was indistinguishable between the S and W isolate groups except for cymoxanil, whose effect was different for each of the two groups. Cymoxanil was found inefficient in mycelial growth inhibition, where only about 25% of mycelial growth was inhibited even at the concentration of 100 µg/mL, which is congruent with the results of other studies [
63,
64]. All of the isolates we tested were resistant, although substantial variation in the resistance of the isolates was observed on an individual level. Our evidence of a
P. cactorum population highly resistant to cymoxanil is surprisingly in contrast to the sensitive behavior of the
P. cactorum in Florida (USA), as reported by Marin et al. [
27]. Such a high magnitude of sensitivity could be associated with rapid pathogen adaptation, the presence of diverse lineages in different parts of the world, or the distinct frequency of the application of this chemical [
65,
66,
67]. The mode of action of cymoxanil is unknown [
68]. The substantial differentiation between the responses of the S group isolates to cymoxanil, despite their close genetic relationship (
supplementary Table S14), indicates the probable qualitative (monogenic) nature [
69] of resistance to this fungicide, which enables such a rapid increase in resistance.
The efficiency of fungicides varies between the different life stages of a pathogen, and is probably determined by the various modes of action of the active ingredients. In the case of metalaxyl, the mycelial growth phase and sporangia formation are the stages affected by this fungicide, because metalaxyl inhibits the synthesis of ribosomal RNA by disrupting the activity of RNA polymerase [
51,
70,
71]. Since the growth of hyphae is necessarily associated with RNA synthesis, metalaxyl significantly affects such regions but does not have an impact on zoospore release, probably because such a process is not directly determined by RNA synthesis. A similar principle could also be considered for the inefficiency of dimethomorph against zoospore release, although the mode of action of dimethomorph is different. This fungicide inhibits cell wall deposition in actively growing regions of hyphae [
49,
57,
72], therefore it is efficient against mycelial growth or sporangia formation, but not against zoospore release. Azoxystrobin, which we showed to be the most effective against zoospore release, relies on the inhibition of the ATP supply by mitochondria. This is achieved by the disruption of the activity of cytochromes which determine oxidative respiration [
73]. Since respiration is necessary for energetically demanding metabolic processes, such as active movement, zoospores are affected by this fungicide. However, mitochondria are also present in growing hyphal tips in high numbers, with a high probability that they do not respire or synthesize ATP there but play other roles [
74], which could explain the inefficiency of azoxystrobin against mycelial growth. The only fungicide efficient against all three tested life stages of
P. cactorum, fluopicolide, causes the disruption of cytoskeletal proteins such as actins, integrins, tubulins and spectrins [
75]. These structures are necessary for all living cells, so their damage affects all of the life stages we tested.
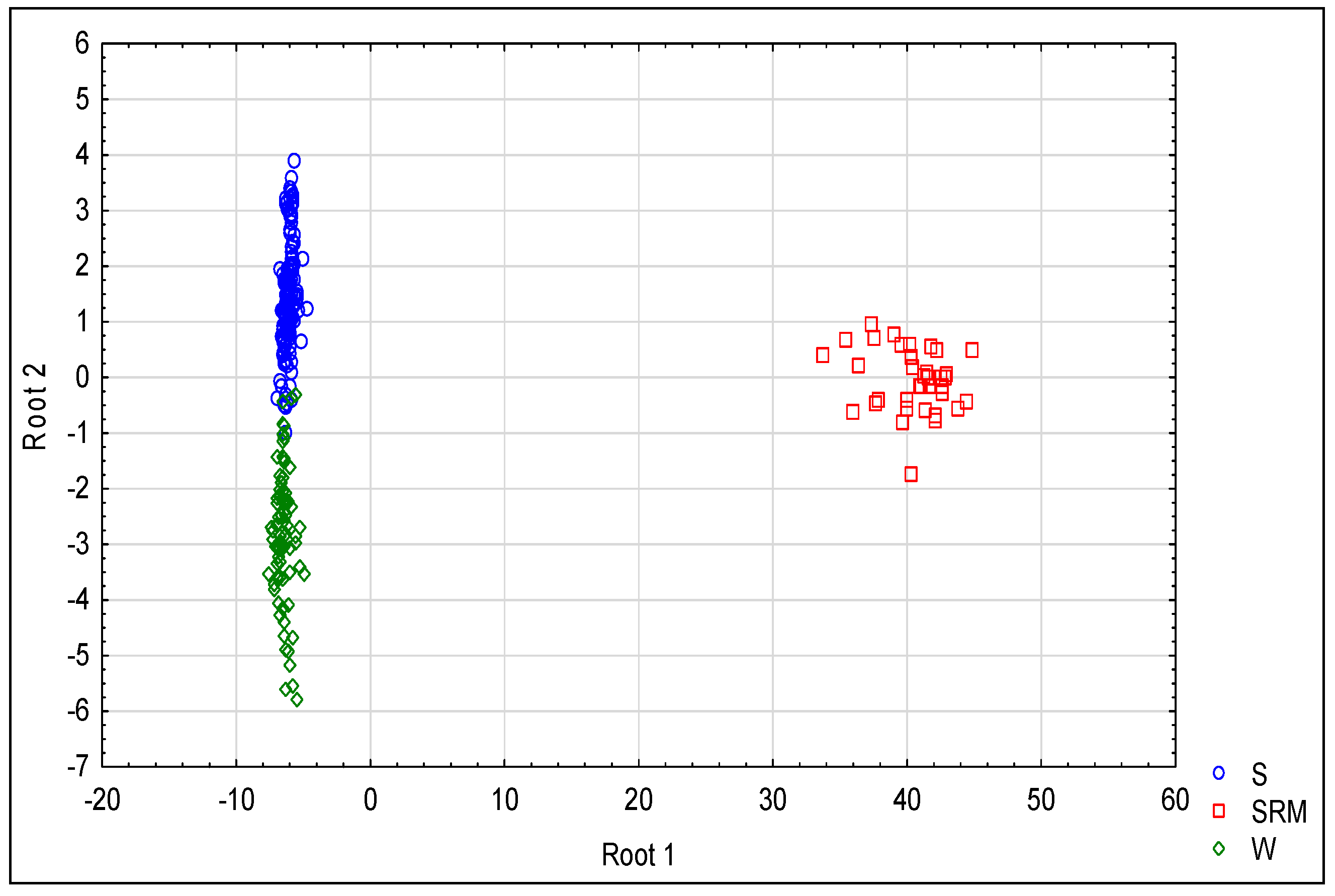
Our analyses revealed a substantial differentiation between the host-specific groups of the
P. cactorum isolates. The host specificity was revealed in this species of plant pathogen, where isolates originating from strawberry plants are significantly more virulent to this host than to others, or are even totally unable to infect other host species. Conversely, isolates from woody plants are unable to attack strawberry plants [
4,
18,
76,
77]. Finer structuring into genetic lineages was revealed, which is associated with the genetic background of this species [
39]; this division also roughly respects the host-specific grouping. Such differentiation is also obvious on the level of resistance to fungicides, which is evidenced by the low λ
W total (0.22,
p < 0.000,
Table 5) in the DA, calculated on the basis of the collective data of all current assays. For some fungicides, such differentiation is also obvious separately in individual analyses. Although we identified the isolates which are totally resistant to some fungicides, and although they are usually members of the group S, the differences between the S and W groups were also perceptible if the obviously resistant isolates were considered as a separate group in the analysis (
Table 4,
Table 5,
Table 6 and
Table 7). The reason for such a differentiation could only be hypothesized as the result of a greater use of fungicides against
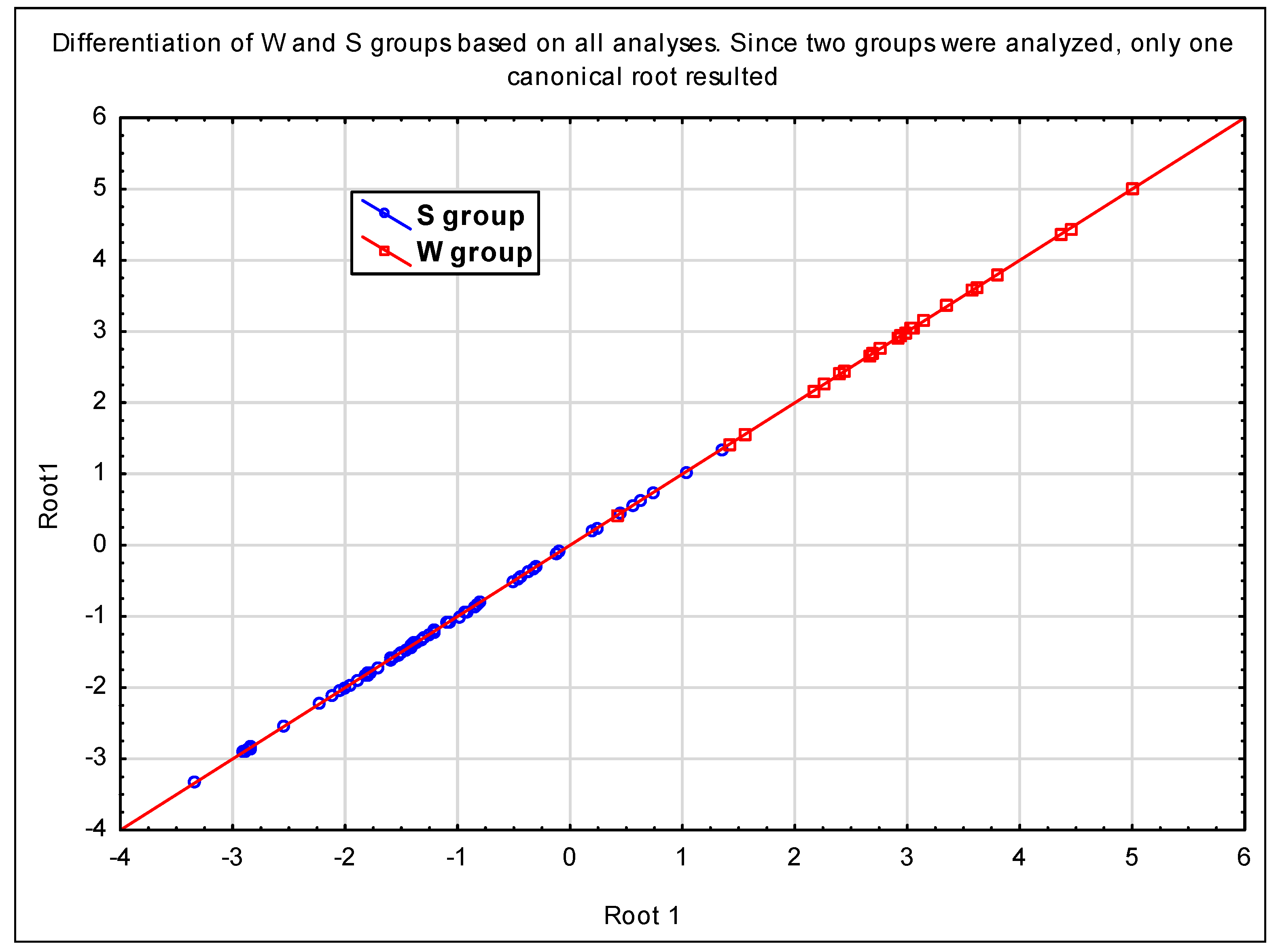
P. cactorum in strawberry fields (for S group members) than in other types of land use (for W group members). The shift in resistance is documented by our comparison of a similarity matrix based on fungicides, with one based on genetic information. This comparison shows the decreased mutual average similarity of the isolates of the S group based on fungicides (75%) compared to their genetic similarity (93%). No similar shift is visible in the W group, where the average similarities are approximately equal (83% for genetics and 84% for fungicides;
Table 8). The similarities among isolates of the S group based on fungicide resistance are lower (75%) than those among isolates of the W group (84%), although the S group members are mutually genetically closer (S—93%, W—83%) and the W group members originate from a significantly wider region. These numbers document a shift caused very probably by the frequent use of fungicides, since although some members of the W group originate from apple trees where fungicide use cannot not be excluded, their incomparably more intensive use in strawberry plantations can be assumed. However, as documented by the estimated value of the resistance factor [
43] for fluopicolide, in relation to fungicides, even isolates of the group W do not behave like wild population strains [
43,
44,
45]. Since
P. cactorum is assumed not to be indigenous to Europe [
18], the resistance of the entire European population could be influenced by the features of genotypes which were introduced in Europe.
We revealed the differences in resistance against fungicides on various levels, i.e., between host-specific groups, between diverse life stages and between isolates, and this should be carefully taken into consideration in the further use of these fungicides in plant protection. Of these generally efficient fungicides—metalaxyl, dimethomorph, azoxystrobin and fluopicolide—only the last one is useful against all stages of a pathogen. Although no isolate obviously resistant against fluopicolide was found, the entire population probably has a somewhat increased resistance against this fungicide. The differences between populations from strawberry and woody plants indicates the increased resistance against fungicides in the first mentioned group, which is most likely the result of the continual use of these fungicides. Even in the case of the careful use of fungicide rotation, some
P. cactorum strains acquire resistance against the fungicides used, because some life stages are insensitive to a particular fungicide and survive the treatment. Similar accidental contacts of a pathogen with a non-lethal fungicide application gradually increase the resistance level of the population [
78]. The use of anti-resistant strategies formulated to prevent the development of resistance [
79] should be the necessary minimum basis for the sustainability of plant protection.