Insight into the Systematics of Novel Entomopathogenic Fungi Associated with Armored Scale Insect, Kuwanaspis howardi (Hemiptera: Diaspididae) in China
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimen Collection and Morphological Study
2.2. DNA Extraction, Amplification and Sequencing
2.3. Phylogenetic Analyses
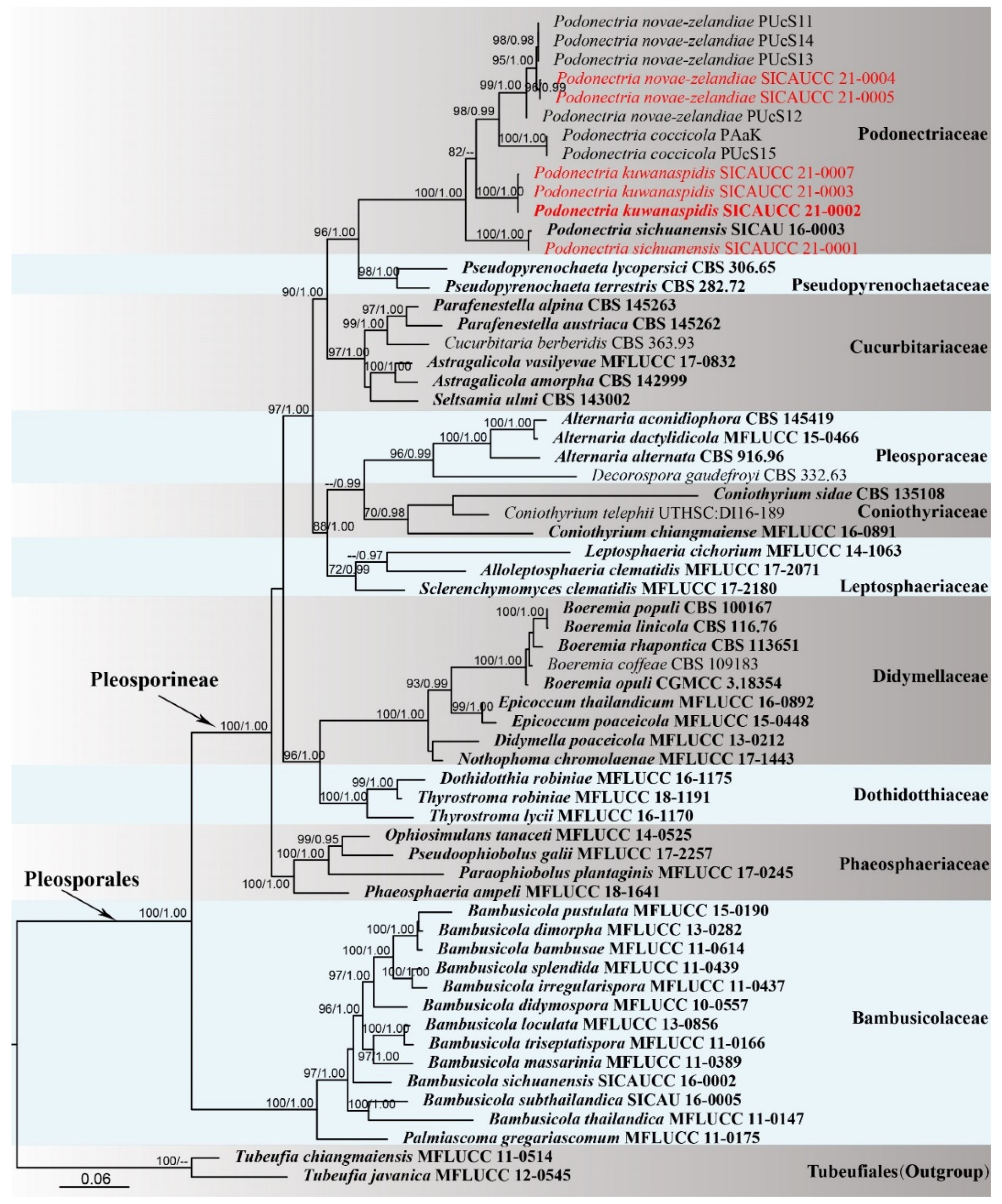
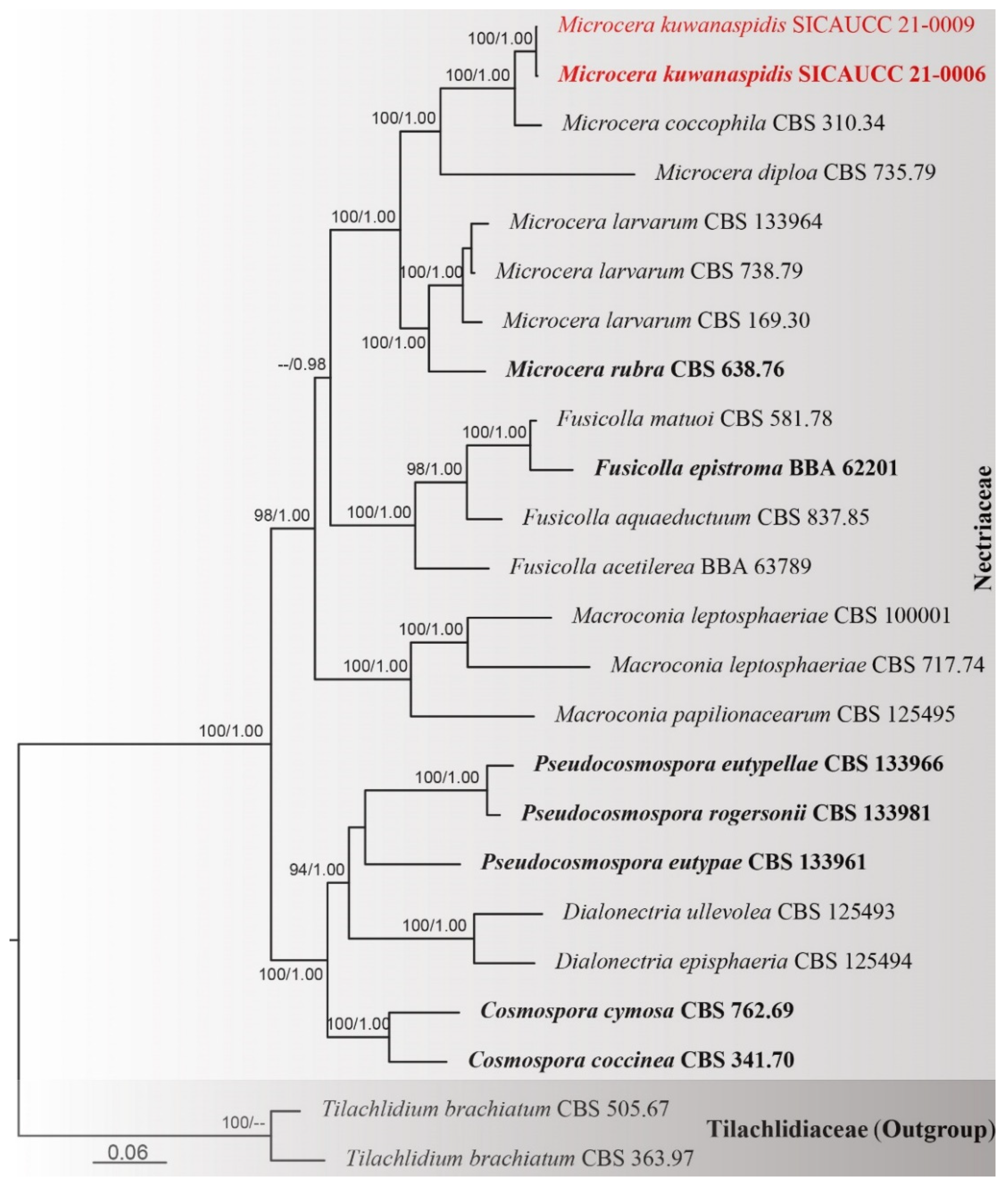
3. Results
3.1. Phylogenetic Analyses
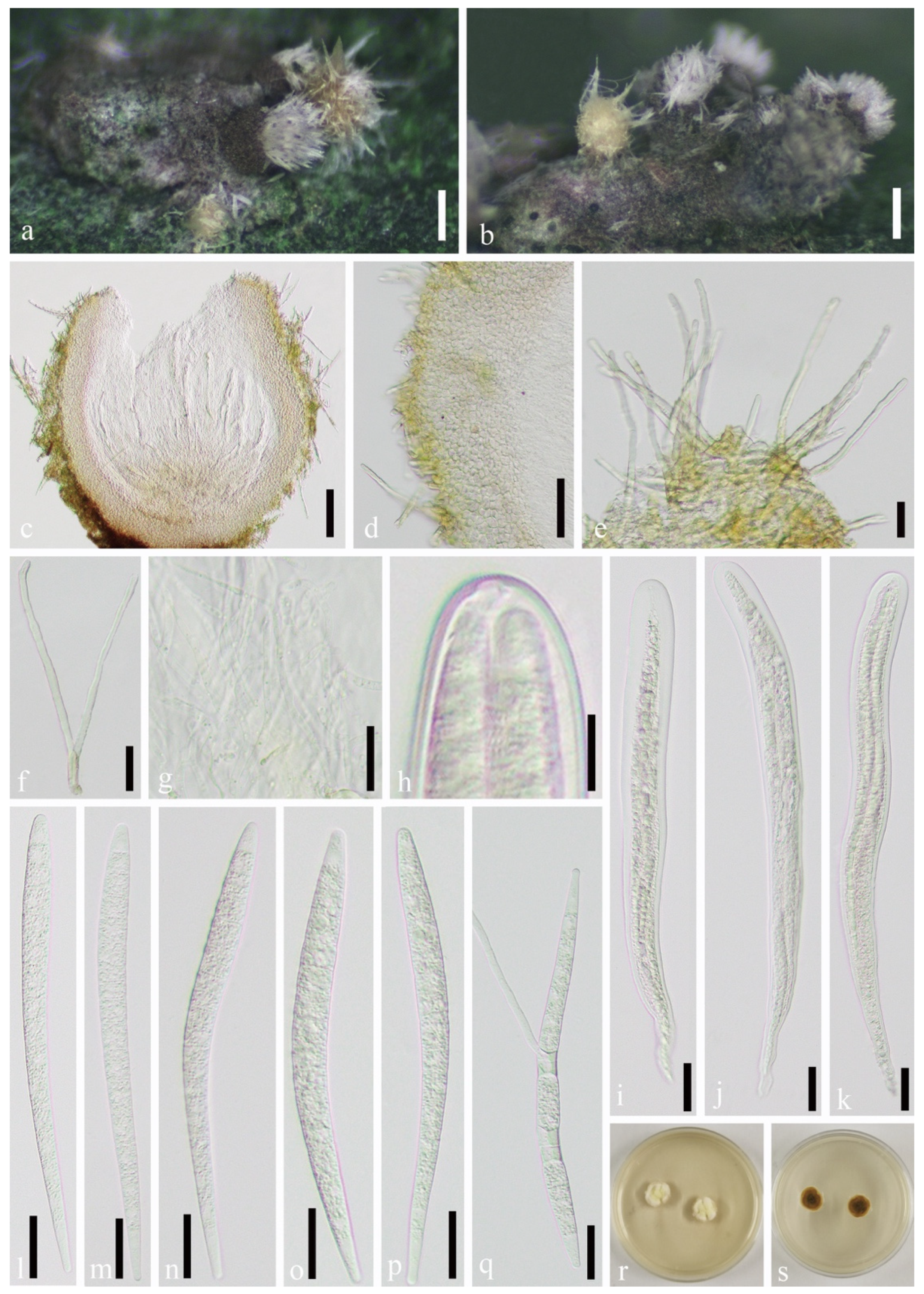
3.2. Taxonomy
4. Discussion
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Petch, T. Studies in entomogenous fungi. The Nectriae parasitic on scale insects. Trans. Brit. Myc. Socy. 1921, 7, 133–167. [Google Scholar] [CrossRef]
- Index Fungorum Database. Available online: http://www.indexfungorum.org/ (accessed on 15 April 2021).
- Dingley, J.M. The Hypocreales of new zealand VI. The genera Hypocrella, Barya, Claviceps and Podonectria. Trans. R. Soc. N. Z. 1954, 81, 489–499. [Google Scholar]
- Rossman, A.Y. Podonectria, a genus in the Pleosporales on scale insects. Mycotaxon 1978, 7, 163–182. [Google Scholar]
- Dao, H.T.; Beattie, G.A.C.; Rossman, A.Y.; Burgess, L.W.; Holford, P. Four putative entomopathogenic fungi of armoured scale insects on Citrus in Australia. Mycol. Prog. 2016, 15, 47. [Google Scholar] [CrossRef]
- Dennis, R.W.G. New British fungi. Kew Bull. 1957, 12, 399–404. [Google Scholar] [CrossRef]
- Rossman, A.Y. The genus Ophionectria (Euascomycetes, Hypocreales). Mycologia 1977, 69, 355–391. [Google Scholar] [CrossRef]
- Pirozynski, K.A. Notes on Hyperparasitic Sphaeriales, Hypocreales and ‘Hypocreoid Dothideales’. Kew Bull. 1977, 31, 595–610. [Google Scholar] [CrossRef]
- Rossman, A.Y. The Tubeufiaceae and Similar Loculoascomycetes; C.A.B. International: Farnham Royal, UK, 1987; p. 43. [Google Scholar]
- Spatafora, J.W.; Quandt, C.A.; Kepler, R.M.; Sung, G.H.; Shrestha, B.; Hywel-Jones, N.L.; Luangsa-ard, J.J. New 1F1N species Combinations in Ophiocordycipitaceae (Hypocreales). IMA Fungus 2015, 6, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Quandt, C.A.; Kepler, R.M.; Gams, W.; Araújo, J.P.M.; Ban, S.; Evans, H.C.; Hughes, D.; Humber, R.; Hywel-Jones, N.; Li, Z.Z.; et al. Phylogenetic-based nomenclatural proposals for Ophiocordycipitaceae (Hypocreales) with new combinations in Tolypocladium. IMA Fungus 2014, 5, 121–134. [Google Scholar] [CrossRef]
- Yang, C.L.; Xu, X.L.; Liu, Y.G. Podonectria sichuanensis, a potentially mycopathogenic fungus from Sichuan Province in China. Phytotaxa 2019, 402, 219–231. [Google Scholar] [CrossRef]
- Wollenweber, H.W.; Reinking, O.A. Die fusarien, ihre Beschreibung, schadwirkung und bekämpfung; Paul Parey: Berlin, Germany, 1935; pp. 1–355. [Google Scholar]
- Booth, C. The Genus Fusarium; Commonwealth Mycological Institute: Kew, UK, 1971; pp. 1–237. [Google Scholar]
- Gerlach, W.; Nirenberg, H. The genus Fusarium: A pictorial atlas. In Mitteilungen Aus der Biologischen Bundesanstalt für Land- und Forstwirtschaft; Kommissionsverlag Paul Parey: Berlin, Germany, 1982; Volume 209, pp. 1–406. [Google Scholar]
- Nelson, P.E.; Toussoun, T.A.; Marasas, W.F.O. Fusarium Species: An Illustrated Manual for Identification; Pennsylvania State University Press: University Park, PA, USA, 1983; pp. 1–193. [Google Scholar]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual, 1st ed.; Blackwell Publishing: Ames, IA, USA, 2006; pp. 1–369. [Google Scholar]
- Gräfenhan, T.; Schroers, H.J.; Nirenberg, H.I.; Seifert, K.A. An overview of the taxonomy, phylogeny, and typification of nectriaceous fungi in Cosmospora, Acremonium, Fusarium, Stilbella, and Volutella. Stud. Mycol. 2011, 68, 79–113. [Google Scholar] [CrossRef] [PubMed]
- Lombard, L.; Merwe, N.A.; Groenewald, J.Z.; Crous, P.W. Generic concepts in Nectriaceae. Stud. Mycol. 2015, 80, 189–245. [Google Scholar] [CrossRef]
- Triest, D.; Cremer, K.D.; Piérard, D.; Hendrickx, M. Unique phylogenetic lineage found in the Fusarium-like clade after re-examining BCCM/IHEM fungal culture collection material. Mycobiology 2016, 44, 121–130. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Normark, B.B.; Okusu, A.; Morse, G.E.; Peterson, D.A.; Itioka, T.; Schneider, S.A. Phylogeny and classification of armored scale insects (Hemiptera: Coccomorpha: Diaspididae). Zootaxa 2019, 4616, 1–98. [Google Scholar] [CrossRef] [PubMed]
- Fang, Z.G.; Liu, J. Biology observation and control on the Kuwanaspis howardi. J. Bamboo Res. 2000, 19, 78–80. [Google Scholar]
- Malumphy, C.; Salisbury, A. First incursion in Europe of bamboo white scale Kuwanaspis howardi (Hemiptera: Diaspididae), with a review of Kuwanaspis species detected in Britain. Br. J. Entomol. Nat. Hist. 2016, 29, 97–103. [Google Scholar]
- Senanayake, I.C.; Rathnayaka, A.R.; Marasinghe, D.S.; Calabon, M.S.; Gentekaki, E.; Lee, H.B.; Hurdeal, V.G.; Pem, D.; Dissanayake, L.S.; Wijesinghe, S.N.; et al. Morphological approaches in studying fungi: Collection, examination, isolation, sporulation and preservation. Mycosphere 2020, 11, 2678–2754. [Google Scholar] [CrossRef]
- Chomnunti, P.; Hongsanan, S.; Hudson, B.A.; Tian, Q.; Peršoh, D.; Dhami, M.K.; Alias, A.S.; Xu, J.C.; Liu, X.Z.; Stadler, M.; et al. The sooty moulds. Fungal Divers. 2014, 66, 1–36. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplifcation and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfaud, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar] [CrossRef]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef]
- Rehner, S.A.; Buckley, E. A Beauveria phylogeny inferred from nuclear ITS and EF1-α sequences: Evidence for cryptic diversification and links to Cordyceps teleomorphs. Mycologia 2005, 97, 84–98. [Google Scholar] [CrossRef]
- Liu, Y.J.; Whelen, S.; Hall, B.D. Phylogenetic relationships among Ascomycetes: Evidence from an RNA polymerse II subunit. Mol. Biol. Evol. 1999, 16, 1799–1808. [Google Scholar] [CrossRef] [PubMed]
- Dai, D.Q.; Phookamsak, R.; Wijayawardene, N.N.; Li, W.J.; Bhat, D.J.; Xu, J.C.; Taylor, J.E.; Hyde, K.D.; Chukeatirote, E. Bambusicolous fungi. Fungal Divers. 2016, 82, 1–105. [Google Scholar] [CrossRef]
- Wanasinghe, D.N.; Mortimer, P.E.; Xu, J. Insight into the systematics of microfungi colonizing dead woody twigs of Dodonaea viscosa in Honghe (China). J. Fungi 2021, 7, 180. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2019, 20, 1160–1166. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Xu, X.L.; Yang, C.L.; Jeewon, R.; Wanasinghe, D.N.; Liu, Y.G.; Xiao, Q.G. Morpho-molecular diversity of Linocarpaceae (Chaetosphaeriales): Claviformispora gen. nov. from decaying branches of Phyllostachys heteroclada. MycoKeys 2020, 70, 1–17. [Google Scholar] [CrossRef]
- Woudenberg, J.H.C.; Groenewald, J.Z.; Binder, M.; Crous, P.W. Alternaria redefined. Stud. Mycol. 2013, 75, 171–212. [Google Scholar] [CrossRef]
- Marin-Felix, Y.; Hernández-Restrepo, M.; Iturrieta-González, I.; García, D.; Gené, J.; Groenewald, J.Z.; Cai, L.; Chen, Q.; Quaedvlieg, W.; Schumacher, R.K.; et al. Genera of phytopathogenic fungi: GOPHY 3. Stud. Mycol. 2019, 94, 1–124. [Google Scholar] [CrossRef]
- Thambugala, K.M.; Wanasinghe, D.N.; Phillips, A.J.L.; Camporesi, E.; Bulgakov, T.S.; Phukhamsakda, C.; Ariyawansa, H.A.; Goonasekara, I.D.; Phookamsak, R.; Dissanayake, A.; et al. Mycosphere notes 1-50: Grass (Poaceae) inhabiting Dothideomycetes. Mycosphere 2017, 8, 697–796. [Google Scholar] [CrossRef]
- Phukhamsakda, C.; McKenzie, E.H.C.; Phillips, A.J.L.; Jones, E.B.G.; Bhat, D.J.; Marc, S.; Bhunjun, C.S.; Wanasinghe, D.N.; Thongbai, B.; Camporesi, E.; et al. Microfungi associated with Clematis (Ranunculaceae) with an integrated approach to delimiting species boundaries. Fungal Divers. 2020, 102, 1–203. [Google Scholar] [CrossRef]
- Jaklitsch, W.M.; Checa, J.; Blanco, M.N.; Olariaga, I.; Tello, S.; Voglmayr, H. A preliminary account of the Cucurbitariaceae. Stud. Mycol. 2018, 90, 71–118. [Google Scholar] [CrossRef]
- Wanasinghe, D.N.; Phukhamsakda, C.; Hyde, K.D.; Jeewon, R.; Lee, H.B.; Jones, E.B.G.; Tibpromma, S.; Tennakoon, D.S.; Dissanayake, A.J.; Jayasiri, S.C.; et al. Fungal diversity notes 709–839: Taxonomic and phylogenetic contributions to fungal taxa with an emphasis on fungi on Rosaceae. Fungal Divers. 2018, 89, 1–236. [Google Scholar] [CrossRef]
- Dai, D.Q.; Bhat, D.J.; Liu, J.K.; Chukeatirote, E.; Zhao, R.L.; Hyde, K.D. Bambusicola, a new genus from bamboo with asexual and sexual morphs. Cryptogamie Mycol. 2012, 33, 363–379. [Google Scholar] [CrossRef]
- Dai, D.Q.; Bahkali, A.H.; Li, W.J.; Bhat, D.J.; Zhao, R.L.; Hyde, K.D. Bambusicola loculata sp. nov. (Bambusicolaceae) from bamboo. Phytotaxa 2015, 213, 122–130. [Google Scholar] [CrossRef]
- Yang, C.L.; Xu, X.L.; Liu, Y.G. Two new species of Bambusicola (Bambusicolaceae, Pleosporales) on Phyllostachys heteroclada from Sichuan, China. Nova Hedwig. 2019, 108, 527–545. [Google Scholar] [CrossRef]
- Jayawardena, R.S.; Hyde, K.D.; Jeewon, R.; Ghobad-Nejhad, M.; Wanasinghe, D.N.; Liu, N.G.; Phillips, A.J.L.; Oliveira-Filho, J.R.C.; da Silva, G.A.; Gibertoni, T.B.; et al. One stop shop II: Taxonomic update with molecular phylogeny for important phytopathogenic genera: 26–50. Fungal Divers. 2019, 94, 41–129. [Google Scholar] [CrossRef]
- Valenzuela-Lopez, N.; Sutton, D.A.; Cano-Lira, J.F.; Paredes, K.; Wiederhold, N.; Guarro, J.; Stchigel, A.M. Coelomycetous fungi in the clinical setting: Morphological convergence and cryptic diversity. J. Clin. Microbiol. 2016, 55, 552–567. [Google Scholar] [CrossRef]
- Quaedvlieg, W.; Verkley, G.J.M.; Shin, H.D.; Barreto, R.W.; Alfenas, A.C.; Swart, W.J.; Groenewald, J.Z.; Crous, P.W. Sizing up Septoria. Stud. Mycol. 2013, 75, 307–390. [Google Scholar] [CrossRef]
- De Gruyter, J.; Woudenberg, J.H.C.; Aveskamp, M.M.; Verkley, G.J.M.; Groenewald, J.Z.; Crous, P.W. Redisposition of phoma-like anamorphs in Pleosporales. Stud. Mycol. 2013, 75, 1–36. [Google Scholar] [CrossRef] [PubMed]
- Inderbitzin, P.; Kohlmeyer, J.; Volkmann-Kohlmeyer, B.; Berbee, M.L. Decorospora, a new genus for the marine ascomycete Pleospora gaudefroyi. Mycologia 2002, 94, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Senwanna, C.; Wanasinghe, D.N.; Bulgakov, T.S.; Wang, Y.; Bhat, D.J.; Tang, A.M.C.; Mortimer, P.E.; Xu, J.; Hyde, K.D.; Phookamsak, R. Towards a natural classification of Dothidotthia and Thyrostroma in Dothidotthiaceae (Pleosporineae, Pleosporales). Mycosphere 2019, 10, 701–738. [Google Scholar] [CrossRef]
- Ariyawansa, H.A.; Phukhamsakda, C.; Thambugala, K.M.; Bulgakov, T.S.; Wanasinghe, D.N.; Perera, R.H.; Mapook, A.; Camporesi, E.; Kang, J.C.; Jones, E.B.G.; et al. Revision and phylogeny of Leptosphaeriaceae. Fungal Divers. 2015, 74, 19–51. [Google Scholar] [CrossRef]
- Mapook, A.; Hyde, K.D.; McKenzie, E.H.C.; Jones, E.B.G.; Bhat, D.J.; Jeewon, R.; Stadler, M.; Samarakoon, M.C.; Malaithong, M.; Tanunchai, B.; et al. Taxonomic and phylogenetic contributions to fungi associated with the invasive weed Chromolaena odorata (Siam weed). Fungal Divers. 2020, 101, 1–175. [Google Scholar] [CrossRef]
- Tibpromma, S.; Liu, J.K.; Promputtha, I.; Camporesi, E.; Bhakali, A.H.; Hyde, K.D.; Boonmee, S. Ophiosimulans tanaceti gen. et sp. nov. (Phaeosphaeriaceae) on Tanacetum sp. (Asteraceae) from Italy. Mycol. Prog. 2016, 15, 46. [Google Scholar] [CrossRef]
- Phookamsak, R.; Wanasinghe, D.N.; Hongsanan, S.; Phukhamsakda, C.; Huang, S.K.; Tennakoon, D.S.; Norphanphoun, C.; Camporesi, E.; Bulgakov, T.S.; Promputtha, I.; et al. Towards a natural classification of Ophiobolus and ophiobolus-like taxa; introducing three novel genera Ophiobolopsis, Paraophiobolus and Pseudoophiobolus in Phaeosphaeriaceae (Pleosporales). Fungal Divers. 2017, 87, 299–339. [Google Scholar] [CrossRef]
- Liu, J.K.; Hyde, K.D.; Jones, E.B.G.; Ariyawansa, H.A.; Bhat, D.J.; Boonmee, S.; Maharachchikumbura, S.S.N.; McKenzie, E.H.C.; Phookamsak, R.; Phukhamsakda, C.; et al. Fungal diversity notes 1–110: Taxonomic and phylogenetic contributions to fungal species. Fungal Divers. 2015, 72, 1–197. [Google Scholar] [CrossRef]
- Jaklitsch, W.M.; Voglmayr, H. Fenestelloid clades of the Cucurbitariaceae. Persoonia 2020, 44, 1–40. [Google Scholar] [CrossRef]
- Tennakoon, D.; Jeewon, R.; Gentekaki, E.; Kuo, C.H.; Hyde, K.D. Multi-gene phylogeny and morphotaxonomy of Phaeosphaeria ampeli sp. nov. from Ficus ampelas and a new record of P. musae from Roystonea regia. Phytotaxa 2019, 406, 111–128. [Google Scholar] [CrossRef]
- Valenzuela-Lopez, N.; Cano-Lira, J.F.; Guarro, J.; Sutton, D.A.; Wiederhold, N.; Crous, P.W.; Stchigel, A.M. Coelomycetous Dothideomycetes with emphasis on the families Cucurbitariaceae and Didymellaceae. Stud. Mycol. 2018, 90, 1–69. [Google Scholar] [CrossRef]
- Boonmee, S.; Rossman, A.Y.; Liu, J.K.; Li, W.J.; Dai, D.Q.; Bhat, J.D.; Jones, E.B.G.; McKenzie, E.H.C.; Xu, J.C.; Hyde, K.D. Tubeufiales, ord. nov., integrating sexual and asexual generic names. Fungal Divers. 2014, 68, 239–298. [Google Scholar] [CrossRef]
- O’Donnell, K.; Humber, R.A.; Geiser, D.M.; Kang, S.; Park, B.; Robert, V.A.R.G.; Crous, P.W.; Johnston, P.R.; Aoki, T.; Rooney, A.P.; et al. Phylogenetic diversity of insecticolous fusaria inferred from multilocus DNA sequence data and their molecular identification via FUSARIUM-ID and Fusarium MLST. Mycologia 2012, 104, 427–445. [Google Scholar] [CrossRef]
- Bills, G.F.; Platas, G.; Overy, D.P.; Collado, J.; Fillola, A.; Jiménez, M.R.; Martín, J.; del Val, A.G.; Vicente, F.; Tormo, J.R.; et al. Discovery of the parnafungins, antifungal metabolites that inhibit mRNA polyadenylation, from the Fusarium larvarum complex and other Hypocrealean fungi. Mycologia 2009, 101, 449–472. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.S.; Rossman, A.Y.; Samuels, G.J.; Chaverri, P. Pseudocosmospora, a new genus to accommodate Cosmospora vilior and related species. Mycologia 2013, 105, 1287–1305. [Google Scholar] [CrossRef] [PubMed]
- Rossman, A.Y.; Crous, P.W.; Hyde, K.D.; Hawksworth, D.L.; Aptroot, A.; Bezerra, J.L.; Bhat, J.D.; Boehm, E.; Braun, U.; Boonmee, S.; et al. Recommended names for pleomorphic genera in Dothideomycetes. IMA Fungus 2015, 6, 507–523. [Google Scholar] [CrossRef] [PubMed]
- Jeewon, R.; Hyde, K.D. Establishing species boundaries and new taxa among fungi: Recommendations to resolve taxonomic ambiguities. Mycosphere 2016, 7, 1669–1677. [Google Scholar] [CrossRef]
- Dao, H.T.; Beattie, G.A.C.; Rossman, A.Y.; Burgess, L.W.; Holford, P. Systematics and biology of two species of Microcera associated with armoured scales on citrus in Australia. Mycol. Prog. 2015, 14, 17. [Google Scholar] [CrossRef]
- Barr, M.E. On the family Tubeufiaceae (Pleosporales). Mycotaxon 1980, 12, 137–167. [Google Scholar]
- Barr, M.E. A classification of Loculoascomycetes. Mycologia 1979, 71, 935–957. [Google Scholar] [CrossRef]
- Kodsueb, R.; Jeewon, R.; Vijaykrishna, D.; McKenzie, E.H.C.; Lumyong, P.; Lumyong, S.; Hyde, K.D. Systematic revision of Tubeufiaceae based on morphological and molecular data. Fungal Divers. 2006, 21, 105–130. [Google Scholar]
- Lumbsch, H.T.; Huhndorf, S.M. Myconet Volume 14. Part One. Outline of Ascomycota—2009. Part Two. Notes on Ascomycete Systematics. Nos. 4751–5113. Fieldiana Life Earth Sci. 2010, 1, 1–64. [Google Scholar] [CrossRef]
- Hyde, K.D.; Jones, E.B.G.; Liu, J.K.; Ariyawansa, H.; Boehm, E.; Boonmee, S.; Braun, U.; Chomnunti, P.; Crous, P.W.; Dai, D.Q.; et al. Families of Dothideomycetes. Fungal Divers. 2013, 63, 1–313. [Google Scholar] [CrossRef]
- Boonmee, S.; Zhang, Y.; Comment, P.; Chukeatirote, E.; Tsui, C.K.; Bahkali, A.H.; Hyde, K.D. Revision of lignicolous Tubeufiaceae based on morphological reexamination and phylogenetic analysis. Fungal Divers. 2011, 51, 51–63. [Google Scholar] [CrossRef]
- Wijayawardene, N.N.; Hyde, K.D.; Lumbsch, H.T.; Liu, J.K.; Maharachchikumbura, S.S.N.; Ekanayaka, A.H.; Tian, Q.; Phookamsak, R. Outline of ascomycota: 2017. Fungal Divers. 2018, 88, 167–263. [Google Scholar] [CrossRef]
- Wijayawardene, N.N.; Hyde, K.D.; Al-Ani, L.K.T.; Tedersoo, L.; Haelewaters, D.; Rajeshkumar, K.C.; Zhao, R.L.; Aptroot, A.; Leontyev, D.V.; Saxena, R.K.; et al. Outline of fungi and fungus-like taxa. Mycosphere 2020, 11, 1060–1456. [Google Scholar] [CrossRef]
- Hongsanan, S.; Hyde, K.D.; Phookamsak, R.; Wanasinghe, D.N.; McKenzie, E.H.C.; Sarma, V.V.; Lücking, R.; Boonmee, S.; Bhat, J.D.; Liu, N.G.; et al. Refined families of Dothideomycetes: Orders and families incertae sedis in Dothideomycetes. Fungal Divers. 2020, 105, 17–318. [Google Scholar] [CrossRef]
- Punithalingam, E. Nuclei, micronuclei and appendages in tri- and tetraradiate conidia of Cornutispora and four other coelomycete genera. Mycol. Res. 2003, 107, 917–948. [Google Scholar] [CrossRef]
- Hudson, H.J.; Sutton, B.C. Trisulcosporium and Tetranacrium, two new genera of fungi imperfecti. Trans. Brit. Mycol. Soc. 1964, 47, 197–203. [Google Scholar] [CrossRef]
- Sung, G.H.; Sung, J.M.; Hywei-Jones, M.L.; Spatafora, J.W. A multi-gene phylogeny of Clavicipitaceae (Ascomycota, Fungi): Identification of localized incongruence using a combinational bootstrap approach. Mol. Phylogenet. Evol. 2007, 44, 1204–1223. [Google Scholar] [CrossRef] [PubMed]
- Lombard, L.; Crous, P.W. Phylogeny and taxonomy of the genus Gliocladiopsis. Persoonia 2012, 28, 25–33. [Google Scholar] [CrossRef]
- Lücking, R.; Aime, M.C.; Robbertse, B.; Miller, A.N.; Ariyawansa, H.A.; Aoki, T.; Cardinali, G.; Crous, P.W.; Druzhinina, I.S.; Geiser, D.M.; et al. Unambiguous identification of fungi: Where do we stand and how accurate and precise is fungal DNA barcoding. IMA Fungus 2020, 11, 14. [Google Scholar] [CrossRef]
- Wei, D.P.; Wanasinghe, D.N.; Xu, J.C.; To-anun, C.; Mortimer, P.E.; Hyde, K.D.; Elgorban, A.M.; Madawala, S.; Suwannarach, N.; Karunarathna, S.C.; et al. Three novel entomopathogenic fungi from China and Thailand. Front. Microbiol. 2021, 11, 608991. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.D.; Norphanphoun, C.; Maharachchikumbura, S.S.N.; Bhat, D.J.; Jones, E.B.G.; Bundhun, D.; Chen, Y.J.; Bao, D.F.; Boonmee, S.; Calabon, M.S.; et al. Refined families of Sordariomycetes. Mycosphere 2020, 11, 305–1059. [Google Scholar] [CrossRef]
- Fu, L.S. Pathogenic microorganisms on citrus insect scale. Hubei Plant Prot. 1994, 4, 30. [Google Scholar]
- Gao, R.X.; Ouyang, Z.A. A preliminary investigation on parasitic fungi on citrus pests. Microbiol. China 1981, 8, 57–58. [Google Scholar]
- Wang, H. Entomogenous fungi of scale insects and mealworms and their applications. J. Sichuan For. Sci. Technol. 1999, 20, 62–65. [Google Scholar] [CrossRef]
- Dong, Z.Y.; Luo, M. Isolation and identification of a parasitic fungus of citrus scale insects. Mod. Agric. Sci. Technol. 2012, 16, 163–164. [Google Scholar]
- Hely, P.C.; Pasfield, G.; Gellatley, J.G. Insect Pests of Fruit and Vegetable in NSW; Department of Agriculture, New South Wales: Sydney, Australia, 1982; pp. 1–312.
- Smith, D.; Beattie, G.A.C.; Broadley, R. Citrus Pests and Their Natural Enemies: Integrated Pest Management in Australia, 1st ed.; Department of Primary Industries: Brisbane, Australia, 1997; pp. 1–263. Available online: http://hdl.handle.net/10462/pdf/9446 (accessed on 10 April 2021).
- Petch, T. Fungi parasitic on scale insects. Trans. Br. Mycol. Soc. 1921, 7, 18–40. [Google Scholar] [CrossRef]
- Tyson, J.L.; Henderson, R.C.; Fullerton, R.A.; Jamieson, L.E.; Froud, K.J. Distribution and new host records for Cosmospora aurantiicola and Cosmospora flammea: Entomopathogens of Diaspididae in New Zealand. N. Z. Plant Prot. 2005, 58, 283–287. [Google Scholar] [CrossRef]
- Dao, H.T. Ecology of Red Scale (Aonidiella aurantii (Maskell) [Hemiptera: Sternorrhyncha: Diaspididae]) in Citrus Orchards on the Central Coast of New South Wales. Ph.D. Thesis, University of Western Sydney, Penrith, Australia, 2012. [Google Scholar]



| Species | Strain/Voucher No. | GenBank Accession Numbers | References | ||||
|---|---|---|---|---|---|---|---|
| ITS | LSU | SSU | tef1-α | rpb2 | |||
| Alternaria alternata | CBS 916.96 T | AF347031 | DQ678082 | KC584507 | KC584634 | KC584375 | [35] |
| Alternaria aconidiophora | CBS 145419 T | LR133931 | – | – | LR133968 | LR133967 | [36] |
| Alternaria dactylidicola | MFLUCC 15-0466 T | KY703616 | KY703617 | KY703618 | – | KY750720 | [37] |
| Alloleptosphaeria clematidis | MFLUCC 17-2071 T | MT310604 | MT214557 | MT226674 | MT394736 | MT394685 | [38] |
| Astragalicola amorpha | CBS 142999 T | MF795753 | MF795753 | – | MF795842 | MF795795 | [39] |
| Astragalicola vasilyevae | MFLUCC 17-0832 T | MG828870 | MG828986 | MG829098 | MG829193 | MG829248 | [40] |
| Bambusicola bambusae | MFLUCC 11-0614 T | JX442031 | JX442035 | JX442039 | KP761722 | KP761718 | [41,42] |
| Bambusicola irregularispora | MFLUCC 11-0437 T | JX442032 | JX442036 | JX442040 | KP761723 | KP761719 | [41,42] |
| Bambusicola massarinia | MFLUCC 11-0389 T | JX442033 | JX442037 | JX442041 | KP761725 | KP761716 | [41,42] |
| Bambusicola splendida | MFLUCC 11-0439 T | JX442034 | JX442038 | JX442042 | KP761726 | KP761717 | [41,42] |
| Bambusicola didymospora | MFLUCC 10-0557 T | KU940116 | KU863105 | KU872110 | KU940188 | KU940163 | [30] |
| Bambusicola pustulata | MFLUCC 15-0190 T | KU940118 | KU863107 | KU872112 | KU940190 | KU940165 | [30] |
| Bambusicola thailandica | MFLUCC 11-0147 T | KU940119 | KU863108 | KU872113 | KU940191 | KU940166 | [30] |
| Bambusicola triseptatispora | MFLUCC 11-0166 T | KU940120 | KU863109 | – | – | KU940167 | [30] |
| Bambusicola dimorpha | MFLUCC 13-0282 T | KY026582 | KY000661 | KY038354 | – | KY056663 | [37] |
| Bambusicola loculata | MFLUCC 13-0856 T | KP761732 | KP761729 | KP761735 | KP761724 | KP761715 | [42] |
| Bambusicola sichuanensis | SICAUCC 16-0002 T | MK253473 | MK253532 | MK253528 | MK262828 | MK262830 | [43] |
| Bambusicola subthailandica | SICAU 16-0005 T | MK253474 | MK253533 | MK253529 | MK262829 | MK262831 | [43] |
| Boeremia coffeae | CBS 109183 | GU237748 | GU237943 | – | KY484678 | KT389566 | [44] |
| Boeremia rhapontica | CBS 113651 T | KY484662 | – | – | KY484713 | – | [44] |
| Boeremia opuli | CGMCC 3.18354 T | KY742045 | KY742199 | – | – | KY742133 | [44] |
| Boeremia linicola | CBS 116.76 T | GU237754 | GU237938 | – | KY484705 | KT389574 | [44] |
| Boeremia populi | CBS 100167 T | GU237707 | GU237939 | – | KY484706 | – | [44] |
| Coniothyrium telephii | UTHSC DI16-189 | LT796830 | LN907332 | – | – | LT796990 | [45] |
| Coniothyrium chiangmaiense | MFLUCC 16-0891 T | KY568987 | KY550384 | KY550385 | – | KY607015 | [37] |
| Coniothyrium sidae | CBS 135108 T | KF251149 | KF251653 | – | KF253109 | KF252158 | [46] |
| Cucurbitaria berberidis | CBS 363.93 | JF740191 | GQ387606 | – | – | – | [47] |
| Decorospora gaudefroyi | CBS 332.63 | AF394541 | – | AF394542 | – | – | [48] |
| Didymella poaceicola | MFLUCC 13-0212 T | KX965726 | KX954395 | – | – | KX898364 | [37] |
| Dothidotthia robiniae | MFLUCC 16-1175 T | MK751727 | MK751817 | MK751762 | MK908017 | MK920237 | [49] |
| Epicoccum thailandicum | MFLUCC 16-0892 T | KY703619 | KY703620 | – | – | – | [37] |
| Epicoccum poaceicola | MFLUCC 15-0448 T | KX965727 | KX954396 | – | – | KX898365 | [37] |
| Leptosphaeria cichorium | MFLUCC 14-1063 T | KT454720 | KT454712 | KT454728 | – | – | [50] |
| Nothophoma chromolaenae | MFLUCC 17-1443 T | MT214364 | MT214458 | MT214410 | – | – | [51] |
| Ophiosimulans tanaceti | MFLUCC 14-0525 T | KU738890 | KU738891 | KU738892 | MG520910 | – | [52,53] |
| Palmiascoma gregariascomum | MFLUCC 11-0175 T | KP744452 | KP744495 | KP753958 | – | KP998466 | [54] |
| Parafenestella austriaca | CBS 145262 T | MK356304 | MK356304 | – | MK357576 | MK357532 | [55] |
| Parafenestella alpina | CBS 145263 T | MK356302 | MK356302 | – | MK357574 | MK357530 | [55] |
| Paraophiobolus plantaginis | MFLUCC 17-0245 T | KY797641 | KY815010 | KY815012 | MG520913 | – | [53] |
| Phaeosphaeria ampeli | MFLUCC 18-1641 T | MK503797 | MK503808 | MK503814 | MK503802 | – | [56] |
| Podonectria coccicola | DAR 81026 | KU587798 | KU519419 | – | – | – | [5] |
| Podonectria coccicola | PUcS15 | KU720533 | KU519420 | – | – | – | [5] |
| Podonectria novae-zelandiae | PUcS14 | KU720535 | KU559551 | – | – | – | [5] |
| Podonectria novae-zelandiae | PUcS13 | KU720538 | KU559548 | – | – | – | [5] |
| Podonectria novae-zelandiae | PUcS12 | KU720537 | KU529802 | – | – | – | [5] |
| Podonectria novae-zelandiae | PUcS11 | KU720536 | KU568479 | – | – | – | [5] |
| Podonectria sichuanensis | SICAU 16-0003 T | MK305903 | MK296471 | MK296467 | MK313852 | MK313855 | [12] |
| Podonectria sichuanensis | SICAUCC 21-0001 | MW484988 | MW462899 | MW462891 | MW462111 | MW462118 | This study |
| Podonectria kuwanaspidis | SICAUCC 21-0002 T | MW484989 | MW462900 | MW462892 | MW462112 | MW462119 | This study |
| Podonectria kuwanaspidis | SICAUCC 21-0003 | MW484990 | MW462901 | MW462893 | MW462113 | MW462120 | This study |
| Podonectria novae-zelandiae | SICAUCC 21-0004 | MW484991 | MW462902 | MW462894 | MW462114 | MW462121 | This study |
| Podonectria novae-zelandiae | SICAUCC 21-0005 | MW484992 | MW462903 | MW462895 | MW462115 | MW462122 | This study |
| Podonectria kuwanaspidis | SICAUCC 21-0007 | MW484994 | MW462905 | MW462897 | MW462116 | MW462123 | This study |
| Pseudoophiobolus galii | MFLUCC 17-2257 T | MG520947 | MG520967 | MG520989 | MG520926 | – | [53] |
| Pseudopyrenochaeta lycopersici | CBS 306.65 T | AY649587 | EU754205 | – | – | LT717680 | [57] |
| Pseudopyrenochaeta terrestris | CBS 282.72 T | LT623228 | LT623216 | – | – | LT623287 | [57] |
| Sclerenchymomyces clematidis | MFLUCC 17-2180 T | MT310605 | MT214558 | MT226675 | MT394737 | MT394686 | [38] |
| Seltsamia ulmi | CBS 143002 T | MF795794 | MF795794 | MF795794 | MF795882 | MF795836 | [39] |
| Thyrostroma lycii | MFLUCC 16-1170 T | MK751734 | MK751824 | MK751769 | MK908024 | MK920241 | [49] |
| Thyrostroma robiniae | MFLUCC 18-1191 T | MK751735 | MK751825 | MK751770 | MK908025 | MK920242 | [49] |
| Tubeufia javanica | MFLUCC 12-0545 T | KJ880034 | KJ880036 | KJ880035 | KJ880037 | – | [58] |
| Tubeufia chiangmaiensis | MFLUCC 11-0514 T | KF301530 | KF301538 | KF301543 | KF301557 | – | [58] |
| Species | Strain/Voucher No. | GenBank Accession No. | References | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| acl1 | act | cmdA | his3 | ITS | LSU | rpb1 | rpb2 | tef1-α | tub2 | |||
| Cosmospora coccinea | CBS 341.70 T | HQ897913 | KM231221 | KM231398 | KM231550 | HQ897827 | KM231692 | KM232242 | HQ897777 | KM231947 | KM232086 | [18,19] |
| Cosmospora cymosa | CBS 762.69 T | HQ897914 | KM231222 | KM231399 | KM231551 | HQ897828 | KM231693 | KM232243 | HQ897778 | KM231948 | KM232087 | [18,19] |
| Dialonectria episphaeria | CBS 125494 = TG 2006-11 | HQ897892 | KM231227 | KM231404 | KM231556 | HQ897811 | KM231697 | KM232248 | HQ897756 | KM231953 | KM232092 | [18,19] |
| Dialonectria ullevolea | CBS 125493 = TG 2007-56 | HQ897918 | KM231226 | KM231403 | KM231555 | KM231821 | KM231696 | KM232247 | HQ897782 | KM231952 | KM232091 | [18,19] |
| Fusicolla acetilerea | BBA 63789 T = IMI 181488 = NRRL20827 | KM231065 | – | – | – | HQ897790 | U88108 | – | HQ897701 | – | – | [18] |
| Fusicolla aquaeductuum | CBS 837.85 = BBA 64559 = NRRL 20865 | KM231067 | - | KM231406 | - | KM231823 | KM231699 | KM232250 | HQ897744 | KM231955 | KM232094 | [19] |
| Fusicolla epistroma | BBA 62201 T = IMI 85601 = NRRL 20439 | KM231069 | – | – | – | – | AF228352 | – | HQ897765 | – | – | [18] |
| Fusicolla matuoi | CBS 581.78 = ATCC 18694 = MAFF 238445 = NRRL 20427 | KM231070 | KM231228 | KM231405 | KM231557 | KM231822 | KM231698 | KM232249 | HQ897720 | KM231954 | KM232093 | [18,19] |
| Macroconia papilionacearum | CBS 125495 | HQ897912 | KM231233 | KM231411 | KM231561 | HQ897826 | KM231704 | KM232254 | HQ897776 | KM231958 | KM232096 | [18,19] |
| Macroconia leptosphaeriae | CBS 717.74 | KM231062 | KM231236 | KM231414 | KM231564 | KM231827 | KM231707 | KM232257 | KM232390 | JF735695 | KM232099 | [18,19] |
| Macroconia leptosphaeriae | CBS 100001 = CBS H-6030 | KM231063 | KM231234 | KM231412 | KM231562 | HQ897810 | KM231705 | KM232255 | HQ897755 | KM231959 | KM232097 | [18,19] |
| Microcera coccophila | CBS 310.34 = NRRL 13962 | HQ897843 | KM231232 | KM231410 | KM231560 | HQ897794 | KM231703 | – | HQ897705 | JF740692 | – | [18,19,59] |
| Microcera diploa | CBS 735.79 = BBA 62173 = NRRL 13966 | HQ897899 | – | – | – | HQ897817 | – | – | HQ897763 | – | – | [18] |
| Microcera kuwanaspidis | SICAUCC 21-0006 T | MW462125 | MW462126 | MW462127 | MW462128 | MW484993 | MW462905 | MW462129 | MW462124 | MW462117 | MW462130 | This study |
| Microcera kuwanaspidis | SICAUCC 21-0009 | MZ044037 | MZ044038 | MZ044039 | MZ044040 | MZ029437 | MZ029436 | MZ044041 | MZ044036 | MZ044035 | MZ044042 | This study |
| Microcera larvarum | CBS 169.30 | HQ897855 | – | – | EU860049 | EU860064 | EU860064 | – | HQ897717 | – | EU860025 | [18,60] |
| Microcera larvarum | CBS 738.79 = BBA 62239 = MUCL 19033 = NRRL 20473 | KM231060 | KM231230 | KM231408 | KM231559 | KM231825 | KM231701 | KM232252 | KM232387 | KM231957 | EU860026 | [19,60] |
| Microcera larvarum | A.R. 4580 = CBS 133964 | – | – | – | – | KC291751 | KC291759 | KC291894 | – | KC291832 | KC291935 | [61] |
| Microcera rubra | CBS 638.76 T = BBA 62460 = NRRL 20475 | HQ897903 | KM231231 | KM231409 | EU860050 | HQ897820 | KM231702 | KM232253 | HQ897767 | JF740696 | EU860018 | [18,19,60] |
| Pseudocosmospora rogersonii | CBS 133981 T = G.J.S. 90-56 | – | – | – | – | KC291729 | KC291780 | KC291878 | – | KC291852 | KC291915 | [61] |
| Pseudocosmospora eutypellae | CBS 133966 T = A.R. 4562 | – | – | – | – | KC291721 | KC291757 | KC291871 | – | KC291830 | KC291912 | [61] |
| Pseudocosmospora eutypae | C.H. 11-01 = CBS 133961 T | – | – | – | – | KC291735 | KC291766 | KC291884 | – | KC291837 | KC291925 | [61] |
| Tilachlidium brachiatum | CBS 505.67 | KM231076 | KM231249 | KM231436 | – | KM231839 | KM231720 | KM232272 | KM232415 | KM231976 | KM232110 | [19] |
| Tilachlidium brachiatum | CBS 363.97 | KM231077 | KM231248 | KM231435 | KM231583 | KM231838 | KM231719 | KM232271 | KM232414 | KM231975 | KM232109 | [19] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, X.-L.; Zeng, Q.; Lv, Y.-C.; Jeewon, R.; Maharachchikumbura, S.S.N.; Wanasinghe, D.N.; Hyde, K.D.; Xiao, Q.-G.; Liu, Y.-G.; Yang, C.-L. Insight into the Systematics of Novel Entomopathogenic Fungi Associated with Armored Scale Insect, Kuwanaspis howardi (Hemiptera: Diaspididae) in China. J. Fungi 2021, 7, 628. https://doi.org/10.3390/jof7080628
Xu X-L, Zeng Q, Lv Y-C, Jeewon R, Maharachchikumbura SSN, Wanasinghe DN, Hyde KD, Xiao Q-G, Liu Y-G, Yang C-L. Insight into the Systematics of Novel Entomopathogenic Fungi Associated with Armored Scale Insect, Kuwanaspis howardi (Hemiptera: Diaspididae) in China. Journal of Fungi. 2021; 7(8):628. https://doi.org/10.3390/jof7080628
Chicago/Turabian StyleXu, Xiu-Lan, Qian Zeng, Yi-Cong Lv, Rajesh Jeewon, Sajeewa S. N. Maharachchikumbura, Dhanushka N. Wanasinghe, Kevin D. Hyde, Qian-Gang Xiao, Ying-Gao Liu, and Chun-Lin Yang. 2021. "Insight into the Systematics of Novel Entomopathogenic Fungi Associated with Armored Scale Insect, Kuwanaspis howardi (Hemiptera: Diaspididae) in China" Journal of Fungi 7, no. 8: 628. https://doi.org/10.3390/jof7080628
APA StyleXu, X.-L., Zeng, Q., Lv, Y.-C., Jeewon, R., Maharachchikumbura, S. S. N., Wanasinghe, D. N., Hyde, K. D., Xiao, Q.-G., Liu, Y.-G., & Yang, C.-L. (2021). Insight into the Systematics of Novel Entomopathogenic Fungi Associated with Armored Scale Insect, Kuwanaspis howardi (Hemiptera: Diaspididae) in China. Journal of Fungi, 7(8), 628. https://doi.org/10.3390/jof7080628








