Effects of Glucosinolate-Derived Isothiocyanates on Fungi: A Comprehensive Review on Direct Effects, Mechanisms, Structure-Activity Relationship Data and Possible Agricultural Applications
Abstract
:1. Introduction
2. Direct Antifungal Effects of Glucosinolate-Derived Isothiocyanates
| Isothiocyanate | Source of ITC | Fungal Strains | Fungus Functions | Activity | Ref. |
|---|---|---|---|---|---|
| Aliphatic | |||||
| 2(R)-2OH-3BuITC | Brassica napus GSL + Sinapis alba MYR | Botrytis cinerea, Monilinia laxa, Mucor piriformis, Penicillium expansum | PP | CG | [32] |
| 2(S)-2OH-3BuITC | Crambe abyssinica GSL + S. alba MYR | Alternaria alternata, Botrytis cinerea, Colletotricum coccodes, Diaporthe phaseolorum, Fusarium culmorum, F. oxysporum, Rhizoctonia solani, Sclerotinia sclerotiorum | PP | MG | [6] |
| 3-BuITC | standard | Metarhizium anisopliae | EP | CG, MG | [33] |
| 3-BuITC | std. | Bipolaris sorokiniana, Fusarium graminearum, Gaeumannomyces graminis, Rhizoctonia solani | PP | MG | [34] |
| 3-BuITC | B. napus GSL + S. alba MYR | Monilinia laxa, Mucor piriformis | PP | CG | [32] |
| 3-BuITC | Brassica rapa GSL + S. alba MYR | Fusarium culmorum | PP | MG | [6] |
| 3-BuITC | Brassica plant GSL + S. alba MYR | Leptosphaeria maculans | PP | MG | [35] |
| 3-BuITC | std. | Alternaria brassicae, Sclerotinia sclerotiorum | PP | MG | [18] |
| 4-ITCBuA | synthesized | Candida albicans | HR | MG | [36] |
| 4-PeITC | std. | Bipolaris sorokiniana, Fusarium graminearum, Gaeumannomyces graminis, Rhizoctonia solani | PP | MG | [34] |
| 4-PeITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| 4-PeITC | std. | Alternaria brassicae, Sclerotinia sclerotiorum | PP | MG | [18] |
| AITC | std. | Rhizoctonia solani | PP | MG | [17] |
| AITC | std. | Sclerotium rolfsii, Sclerotinia sclerotiorum | PP | SCG | [38] |
| AITC | std. | Aspergillus parasiticus, Penicillium expansum | MT | MG | [39] |
| AITC | std. | Aspergillus flavus, Botrytis cinerea, Penicillium expansum | PP, MT | CG, MG | [40] |
| AITC | std. | Alternaria brassicae, A. brassicicola | PP | CG, MG | [41] |
| AITC | std. | Fusarium oxysporum | PP | CG, MG, SPG | [42] |
| AITC | std. | Aspergillus niger | PP | MG | [43] |
| AITC | std. | Metarhizium anisopliae | EP | CG, MG | [33] |
| AITC | std. | Colletotricum coccodes, Helminthosporium solani, Rhizoctonia solani | PP | MG | [44] |
| AITC | std. | Alternaria alternata, Aspergillus parasiticus, Fusarium tricinctum, F. verticilloides, Gibberella zeae | MT | mycotoxin production | [45] |
| AITC | std. | Candida albicans | HR | MG, biofilm formation | [46] |
| AITC | std. | Sclerotinia sclerotiorum | PP | MG | [47] |
| AITC | std. | Pleiochaeta setosa | PP | MG | [48] |
| AITC | std. | Geotrichum citri-aurantii | PP | CG, MG | [49] |
| AITC | std. | Fusarium oxysporum, Macrophomina phaseolina, Oidiodendron cerealis, Paraphoma radicina, Setophoma terrestris | endophyte | MG | [50] |
| AITC | std. & synt. | Alternaria tenuis, Aspergillus flavus, A. fumigatus, A. niger, A. oryzae, Cephalothecium roseum, Cladosporium herbarum, Cytospora sp., Fusarium sp., Monilia sitophila, P. brevicompactum, P. cyclopium, Rhizopus oryzae, Schizophyllum commune, Trichoderma viride, Trichophyton gypseum | PP, ENV, HR | MG | [51] |
| AITC | std. | Bipolaris sorokiniana, Fusarium graminearum, Gaeumannomyces graminis, Rhizoctonia solani | PP | MG | [34] |
| AITC | std. | Candida albicans | HR | MG | [52] |
| AITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| AITC | std. | Botrytis cinerea, Penicillium expansum | PP | CG, MG | [53] |
| AITC | std. | Penicillium notatum | ENV | MG | [54] |
| AITC | Brassica juncea GSL + S. alba MYR | Botrytis cinerea, Monilinia laxa, Mucor piriformis, Penicillium expansum, Rhizopus stolonifer | PP | CG | [32] |
| AITC | B. juncea GSL + S. alba MYR | Alternaria alternata, Botrytis cinerea, Colletotricum coccodes, Diaporthe phaseolorum, Fusarium culmorum, F. oxysporum, Rhizoctonia solani, Sclerotinia sclerotiorum | PP | MG | [6] |
| AITC | std. sinigrin + std. MYR | Saccharomyces cerevisiae | N.I. | MG | [55] |
| AITC | std. | Candida albicans | HR | MG | [56] |
| AITC | B. juncea GSL + S. alba MYR | Rhizoctonia solani | PP | soil colonisation | [28] |
| AITC | Brassica plant GSL + S. alba MYR | Leptosphaeria maculans | PP | MG | [35] |
| AITC | std. | Gaeumannomyces graminis | PP | MG | [57] |
| AITC | std. | Glomus clarum | ENV | SPG | [58] |
| AITC | std. | Rhizoctonia solani | PP | MG | [17] |
| AITC | std. | Rhizoctonia solani | PP | MG, SCG | [59] |
| AITC | std. | Sclerotium rolfsii | PP | MG | [60] |
| AITC | std. | Sclerotium rolfsii | PP | MG | [61] |
| AITC | std. sinigrin + std. MYR | Verticillium longisporum | PP | MG | [25] |
| AITC | std. | Candida albicans | HR | MG | [62] |
| AITC | std. | Fusarium oxysporum, Rhizoctonia solani | PP | MG | [63] |
| AITC | std. | Beauveria bassiana, Isaria fumosorosea | EP | CG | [64] |
| AITC | std. | Aspergillus flavus, A. niger, A. ochraceus, Botryotinia fuckeliana, Fusarium oxysporum, Geotrichum spp., Penicillium expansum, P. roqueforti, P. verrucosum, Rhizopus stolonifer | ENV | MG | [65] |
| AITC | std. | Aspergillus fumigatus, A. nomius, A. niger, Candida albicans, Cryptococcus neoformans, Eupenicillum hirayamae, Penicillium cinna-mopurpureum, P. expansum, P. viridicatum, Trichophyton rubrum | HR, ENV | MG | [66] |
| AITC | std. | Aspergillus niger, A. ochraceus, Penicillium citrinum | ENV | MG | [67] |
| AITC | std. | Candida albicans | ENV | MG | [68] |
| AITC | std. | Botrytis cinerea | PP | CG, MG | [69] |
| AITC | std. | Aspergillus flavus | MT | MG, mycotoxin production | [70] |
| AITC | std. | Alternaria brassicae, Sclerotinia sclerotiorum | PP | MG | [18] |
| AITC | std. sinigrin + transgenic MYR | Rhizoctonia solani, Sclerotium rolfsii, Sclerotinia sclerotiorum | PP | MG | [71] |
| AITC | std. | Aspergillus niger, Aureobasidium pullulans, Fomitopsis palustris, Gliocladium virens, Penicillium funiculosurn, Rhizopus stolonifer, Trametes versicolor | ENV | regrowth on wood specimens | [72] |
| AITC | std. | Aspergillus niger | PP | MG, SPG | [73] |
| AITC | std. | Phymatotrichopsis omnivora | PP | MG | [74] |
| AITC | std. | Fusarium oxysporum, Pestalotiopsis spp., Rhizoctonia solani, Verticillium dahliae | PP | MG | [75] |
| AITC | std. | Aspergillus flavus | ENV | MG | [76] |
| AITC | std. | Penicillium nordicum | ENV | MG | [77] |
| AITC | std. | Aspergillus parasiticus | MT | MG | [78] |
| AITC | std. | Alternaria alternata | PP | MG | [79] |
| BuITC | std. | Fusarium oxysporum | PP | CG, SPG | [42] |
| BuITC | std. | Metarhizium anisopliae | EP | CG, MG | [33] |
| BuITC | std. | Sclerotinia sclerotiorum | PP | MG, SCG | [47] |
| BuITC | std. | Geotrichum citri-aurantii | PP | CG, MG | [49] |
| BuITC | std. | Alternaria tenuis, Aspergillus flavus, A. fumigatus, A. niger, A. oryzae, Cladosporium herbarum, Monilia sitophila, Penicillium brevicompactum, P. cyclopium, Trichoderma viride | PP, ENV | MG | [51] |
| BuITC | synt. | Rhizoctonia solani | PP | MG | [80] |
| BuITC | std. | Phymatotrichopsis omnivora | PP | MG | [74] |
| EITC | std. | Fusarium oxysporum | PP | CG, MG, SPG | [42] |
| EITC | std. | Sclerotinia sclerotiorum | PP | MG, SCG | [47] |
| EITC | std. | Geotrichum citri-aurantii | PP | CG, MG | [49] |
| EITC | std. & synt. | Aspergillus niger | PP | MG | [51] |
| EITC | synt. | Rhizoctonia solani | PP | MG | [80] |
| EITC | std. | Botrytis cinerea, Penicillium expansum | PP | CG, MG | [53] |
| HexITC | std. & synt. | Aspergillus niger, Penicillium cyclopium, Rhizopus oryzae | PP | MG | [51] |
| HexITC | std. | Candida albicans | HR | MG | [52] |
| HexITC | synt. | Rhizoctonia solani | PP | MG | [80] |
| iBuITC | std. & synt. | Aspergillus niger, Penicillium cyclopium, Rhizopus oryzae | PP | MG | [51] |
| iPrITC | std. | Colletotricum coccodes, Helminthosporium solani, Rhizoctonia solani | PP | MG | [44] |
| iPrITC | std. & synt. | Aspergillus niger, Penicillium cyclopium, Rhizopus oryzae | PP | MG | [51] |
| MeITC | std. | Colletotricum coccodes, Helminthosporium solani | PP | MG | [44] |
| MeITC | std. | Geotrichum citri-aurantii | PP | CG, MG | [49] |
| MeITC | std. & synt. | Alternaria tenuis, Aspergillus flavus, A. fumigatus, A. niger, A. oryzae, Cladosporium herbarum, Monilia sitophila, Penicillium brevicompactum, P. cyclopium, Trichoderma viride | PP, ENV | MG | [51] |
| MeITC | std. | Bipolaris sorokiniana, Fusarium graminearum, Gaeumannomyces graminis, Rhizoctonia solani | PP | MG | [34] |
| MeITC | synt. | Rhizoctonia solani | PP | MG | [80] |
| MeITC | std. | Gaeumannomyces graminis | PP | MG | [57] |
| PeITC | std. | Metarhizium anisopliae | EP | CG, MG | [33] |
| PeITC | std. & synt. | Aspergillus niger, Penicillium cyclopium, Rhizopus oryzae | PP | MG | [51] |
| PeITC | synt. | Rhizoctonia solani | PP | MG | [80] |
| PrITC | std. | Metarhizium anisopliae | EP | CG, MG | [33] |
| PrITC | std. | Colletotricum coccodes, Helminthosporium solani, Rhizoctonia solani | PP | MG | [44] |
| PrITC | std. & synt. | Aspergillus niger, Penicillium cyclopium, Rhizopus oryzae | PP | MG | [51] |
| PrITC | synt. | Rhizoctonia solani | PP | MG | [80] |
| Aromatic | |||||
| 2-OHPEITC | Barbarea vulgaris GSL + S. alba MYR | Fusarium culmorum | PP | MG | [6] |
| 3-MeOBnITC | synt. | Aspergillus fumigatus, Candida albicans | HR, ENV | MG | [81] |
| 3-MeOBnITC | std. | Verticillium dahliae | PP | MG | [82] |
| 3-MeOBnITC | Salvadora persica | Aspergillus niger, Candida albicans | HR | MG | [83] |
| 3-OHBnITC | S. persica | Aspergillus niger | HR | MG | [83] |
| 3-PPrITC | synt. | Aspergillus brasiliensis, Candida albicans | HR, ENV | MG | [84] |
| 4-Ac-α-L-RhaBnITC | Moringa oleifera | Epidermophyton floccosum, Trichophyton rubrum | HR | MG | [21] |
| 4-MeOBnITC | std. | Aspergillus fumigatus, Candida albicans, C. crusei, C. glabrata, C. parapsilosis, Cryptococcus neoformans | HR | MG | [85] |
| 4-MeOBnITC | synt. | Aspergillus fumigatus, Candida albicans | HR, ENV | MG | [81] |
| 4-MeOBnITC | std. & synt. | Alternaria tenuis, Aspergillus flavus, A. fumigatus, A. niger, A. oryzae, Cladosporium herbarum, Monilia sitophila, Penicillium brevicompactum, P. cyclopium, Trichoderma viride | PP, ENV | MG | [51] |
| 4-OHBnITC | S. alba GSL + S. alba MYR | Botrytis cinerea, Monilinia laxa, Mucor piriformis, Penicillium expansum, Rhizopus stolonifer | PP | CG | [32] |
| 4-OHBnITC | S. alba GSL + S. alba MYR | Botrytis cinerea, Monilinia laxa, Mucor piriformis, Penicillium expansum, Rhizopus stolonifer | PP | curative activity | [86] |
| 4-OHBnITC | S. alba GSL + S. alba MYR | Fusarium culmorum | PP | MG | [6] |
| 4-OHPEITC | std. | Aspergillus fumigatus, Candida albicans | HR | MG | [85] |
| 4-PBuITC | synt. | Aspergillus brasiliensis, Candida albicans | HR, ENV | MG | [84] |
| 4-α-L-RhaBnITC | Moringa oleifera | Epidermophyton floccosum, Trichophyton rubrum | HR | MG | [21] |
| 4-α-L-RhaBnITC | Moringa oleifera | Aspergillus niger, Candida albicans | HR, ENV | MG | [87] |
| 5-PPeITC | synt. | Aspergillus brasiliensis, Candida albicans | HR, ENV | MG | [84] |
| BnITC | std. | Alternaria brassicae, A. brassicicola | PP | CG, MG | [41] |
| BnITC | std. | Candida albicans, C. glabrata, C. krusei, C. parapsilosis, C. tropicalis | HR | MG | [88] |
| BnITC | std. | Alternaria alternata | PP | MG, SPG, mycotoxin production | [89] |
| BnITC | std. | Fusarium oxysporum | PP | CG, SPG | [42] |
| BnITC | std. | Colletotricum coccodes, Helminthosporium solani | PP | MG | [44] |
| BnITC | synt. | Aspergillus fumigatus, Candida albicans | HR, ENV | MG | [81] |
| BnITC | std. | Sclerotinia sclerotiorum | PP | MG, SCG | [47] |
| BnITC | std. | Geotrichum citri-aurantii | PP | CG, MG | [49] |
| BnITC | std. & synt. | Alternaria tenuis, Aspergillus flavus, A. niger, A. oryzae, Cephalothecium roseum, Cladosporium herbarum, Cytospora sp., Fusarium sp., Monilia sitophila, Penicillium brevicompactum, P. cyclopium, Rhizopus oryzae, Schizophyllum commune, Trichoderma viride, Trichophyton gypseum | PP, ENV | MG | [51] |
| BnITC | std. | Bipolaris sorokiniana, Fusarium graminearum, Gaeumannomyces graminis, Rhizoctonia solani | PP | MG | [34] |
| BnITC | std. | Candida albicans | HR | MG | [52] |
| BnITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| BnITC | Lepidium sativum GSL + S. alba MYR | Monilinia laxa, Mucor piriformis | PP | CG | [32] |
| BnITC | L. sativum GSL + S. alba MYR | Alternaria alternata, Botrytis cinerea, Colletotricum coccodes, Diaporthe phaseolorum, Fusarium culmorum, F. oxysporum, Rhizoctonia solani, Sclerotinia sclerotiorum | PP | MG | [6] |
| BnITC | std. | Aspergillus spp., Candida spp., Cryptococcus neoformans, Fonsecaea pedrosoi, Fusarium solani, Microsporum canis, Pseudallescheria boydii, Saccharomyces cerevisiae, Sporothrix schenckii, Trichophyton rubrum | HR, ENV | MG | [23] |
| BnITC | synt. | Aspergillus brasiliensis, Candida albicans | HR, ENV | MG | [84] |
| BnITC | std. | Alternaria brassicae, Sclerotinia sclerotiorum | PP | MG | [18] |
| BnITC | Salvadora persica | Aspergillus niger | HR | MG | [83] |
| BnITC | std. | Phymatotrichopsis omnivora | PP | MG | [74] |
| BnITC | std. | Aspergillus parasiticus | MT | MG | [78] |
| BnITC | std. | Alternaria alternata | PP | MG | [79,90] |
| PEITC | std. | Fusarium oxysporum | PP | CG, SPG | [42] |
| PEITC | std. | Metarhizium anisopliae, Tolypocladium cylindrosporum | EP | MG | [91] |
| PEITC | std. | Alternaria brassicae, A. macrospora, Aspergillus niger, Bipolaris sorokiniana, Fusarium spp., Gaeumannomyces graminis, Lasiodiplodia theobromae, Pleiochaeta setosa, Rhizoctonia solani, Sclerotinia spp., Sclerotium rolfsii, Thielaviopsis basicola, Trichoderma sp. | PP, ENV | MG | [92] |
| PEITC | std. | Metarhizium anisopliae | EP | CG, MG | [33] |
| PEITC | std. | Colletotricum coccodes, Helminthosporium solani, Rhizoctonia solani | PP | MG | [44] |
| PEITC | std. | Sclerotinia sclerotiorum | PP | MG, SCG | [47] |
| PEITC | std. | Geotrichum citri-aurantii | PP | CG, MG | [49] |
| PEITC | std. | Fusarium oxysporum, Macrophomina phaseolina, Paraphoma radicina, Setophoma terrestris, Oidiodendron cerealis | endophyte | MG | [50] |
| PEITC | std. & synt. | Aspergillus niger, Penicillium cyclopium, Rhizopus oryzae | PP | MG | [51] |
| PEITC | std. | Bipolaris sorokiniana, Fusarium graminearum, Gaeumannomyces graminis, Rhizoctonia solani | PP | MG | [34] |
| PEITC | std. | Candida albicans | HR | MG | [52] |
| PEITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| PEITC | std. | Aspergillus niger, Candida albicans, Penicillium citrinum | ENV | MG | [93] |
| PEITC | std. | Candida albicans | HR | MG | [56] |
| PEITC | std. | Gaeumannomyces graminis | PP | MG | [57] |
| PEITC | std. | Rhizoctonia solani | PP | MG, SCG | [59] |
| PEITC | synt. | Aspergillus brasiliensis, Candida albicans | HR, ENV | MG | [84] |
| PEITC | std. | Alternaria brassicae, Sclerotinia sclerotiorum | PP | MG | [18] |
| PEITC | std. | Alternaria alternata | PP | MG | [79] |
| PEITC | std. | Alternaria alternata | PP | MG, SPG | [94] |
| PITC | std. | Fusarium oxysporum | PP | CG, SPG | [42] |
| PITC | std. | Phymatotrichopsis omnivora | PP | MG | [74] |
| PITC | std. | Sclerotinia sclerotiorum | PP | MG, SCG | [47] |
| PITC | std. | Candida albicans | HR | MG | [52] |
| PITC | std. | Saccharomyces cerevisiae | N.I. | MG | [55] |
| PITC | std. | Aspergillus parasiticus | MT | MG | [78] |
| PITC | std. | Alternaria alternata | PP | MG | [79] |
| Indole | |||||
| 1-MeO-3-IMeITC | Brassica plant GSL + S. alba MYR | Leptosphaeria maculans | PP | MG | [35] |
| 3-IMeITC | Brassica plant GSL + S. alba MYR | Leptosphaeria maculans | PP | MG | [35] |
| rapalexin A | synt. | Alternaria brassicicola | PP | MG | [95] |
| sinapigladioside | insect symbiont Burkholderia gladioli | Purpureocillium lilacinum | EP | MG | [96] |
| Sulfur containing | |||||
| 3-MeSOOPrITC | std. & synt. | Aspergillus niger, Penicillium cyclopium, Rhizopus oryzae | PP | MG | [51] |
| 3-MeSOOPrITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| 3-MeSOOPrITC | Cheirantus annuus GSL + S. alba MYR | Fusarium culmorum | PP | MG | [6] |
| 3-MeSOPrITC | std. | Candida albicans | HR | MG | [52] |
| 3-MeSOPrITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| 3-MeSOPrITC | Iberis amara GSL + S. alba MYR | Alternaria alternata, Botrytis cinerea, Colletotricum coccodes, Diaporthe phaseolorum, Fusarium culmorum, F. oxysporum, Rhizoctonia solani, Sclerotinia sclerotiorum | PP | MG | [6] |
| 3-MeSOPrITC | I. amara GSL + S. alba MYR | Rhizoctonia solani | PP | soil colonisation | [28] |
| 3-MeSPrITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| 4-MeSBuITC | std. | Candida albicans | HR | MG | [52] |
| 4-MeSBuITC | Eruca sativa GSL + S. alba MYR | Fusarium culmorum | PP | MG | [6] |
| 4-MeSBuITC | E. sativa GSL + S. alba MYR | Rhizoctonia solani | PP | soil colonisation | [28] |
| 4-MeSO-3-BuITC | std. | Candida albicans | HR | MG | [52] |
| 4-MeSO-3-BuITC | Raphanus sativus GSL + S. alba MYR | Botrytis cinerea, Monilinia laxa, Mucor piriformis, Penicillium expansum, Rhizopus stolonifer | PP | CG | [32] |
| 4-MeSO-3-BuITC | R. sativus GSL + S. alba MYR | Botrytis cinerea, Monilinia laxa, Mucor piriformis, Penicillium expansum, Rhizopus stolonifer | PP | curative activity | [86] |
| 4-MeSO-3-BuITC | R. sativus GSL + S. alba MYR | Fusarium culmorum | PP | MG | [6] |
| 4-MeSOBuITC | synt. | Cryptococcus neoformans | HR | MG | [97] |
| 4-MeSOBuITC | std. | Nosema ceranae | EP | SPG | [98] |
| 4-MeSOBuITC | std. | Candida albicans | HR | MG | [52] |
| 4-MeSOBuITC | std. | Alternaria brassicae, Sclerotinia sclerotiorum | PP | MG | [18] |
| 4-MeSOOBuITC | std. & synt. | Aspergillus niger, Penicillium cyclopium, Rhizopus oryzae | PP | MG | [51] |
| 5-MeSPeITC | std. & synt. | Aspergillus niger, Penicillium cyclopium, Rhizopus oryzae | PP | MG | [51] |
| 9-MeSNonITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| 9-MeSONonITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| 9-MeSOONonITC | std. | Aspergillus niger, Candida holmii, Saccharomyces cerevisiae | ENV | MG | [37] |
| Other | |||||
| AITC, BnITC, PEITC mixture | std. | Candida spp. | HR | MG | [99] |
| Source | Major ITCs Detected | Fungal Strains | Fungus Functions | Activity | Ref. |
|---|---|---|---|---|---|
| Apium graveolens | iPrITC | Fusarium oxysporum | PP | MG | [100] |
| Arabidopsis thaliana | 4-MePeITC | Lecanicillium lecanii | EP | CG | [101] |
| Arabidopsis thaliana | 3-BuITC, 3-OHPrITC, 4-MeSOBuITC, AITC | Verticillium longisporum | PP | MG | [25] |
| Armoracia rusticana | 3-BuITC, AITC, PEITC | Epidermophyton floccosum, Microsporum canis, Trichophyton mentagrophytes, T. rubrum | HR | MG | [22] |
| Armoracia rusticana | AITC, PEITC | Ascosphaera apis | EP | MG | [102] |
| Armoracia rusticana | AITC, PEITC, sBuITC | Aspergillus niger, Candida albicans, Penicillium citrinum | ENV | MG | [93] |
| Armoracia rusticana | AITC, PEITC | Aspergillus fumigatus, A. nidulans, Candida albicans, Saccharomyces cerevisiae | ENV, HR | MG | [56] |
| Armoracia rusticana | 4-PeITC, 5-MeSOPeITC, 5-MeSPeITC, AITC, PEITC | Aspergillus brasiliensis, Candida albicans | ENV, HR | MG | [84] |
| Armoracia rusticana | 3-BuITC, AITC, PEITC | Candida albicans | HR | MG | [62] |
| Aurinia leucadea | 3-BuITC, 4-PeITC, 5-MeSOPeITC, sBuITC | Candida albicans, Penicillium sp, Rhizopus stolonifer | ENV, HR | MG | [103] |
| Aurinia sinuata | 4-PeITC, 5-MeSOPeITC, 5-MeSPeITC | Aspergillus niger, Candida albicans, Penicillium sp. | ENV, HR, PP | MG | [27] |
| Brassica campestris | 4-PeITC | Rhizoctonia solani | PP | MG | [17] |
| Brassica carinata | AITC | Botrytis cinerea | PP | CG, MG | [69] |
| Brassica carinata | AITC | Fusarium sambucinum | PP | MG | [26] |
| Brassica juncea | 3-BuITC, AITC | Rhizoctonia solani | PP | MG | [104] |
| Brassica juncea | 3-BuITC, AITC, sBuITC | Rhizoctonia solani | PP | MG | [17] |
| Brassica juncea | AITC | Aspergillus parasiticus | MT | MG | [105] |
| Brassica juncea | AITC | Fusarium graminearum | PP | MG | [106] |
| Brassica juncea | AITC | Verticillium dahliae | PP | MG | [107] |
| Brassica juncea | 3-BuITC,3-MeSOPrITC,4-MeSBuITC,4-MeSOBuITC,5-MeSOPeITC, AITC | Sclerotinia sclerotiorum | PP | MG | [108] |
| Brassica juncea | AITC | Bipolaris sorokiniana, Fusarium graminearum, Gaeumannomyces graminis, Rhizoctonia solani | PP | MG | [109] |
| Brassica juncea | AITC, BnITC, PEITC | Fusarium oxysporum, Sclerotinia sclerotiorum, Sclerotium cepivorum | PP | SPG, SCG | [110] |
| Brassica juncea | AITC | Sclerotium rolfsii | PP | MG | [60] |
| Brassica juncea | AITC | Sclerotinia sclerotiorum | PP | MG | [111] |
| Brassica juncea | AITC | Sclerotinia sclerotiorum | PP | SCG | [112] |
| Brassica juncea | AITC | Colletotrichum coccodes, Fusarium sambucinum, Rhizoctonia solani, Verticillium albo-atrum, V. dahliae | PP | MG | [26] |
| Brassica juncea | AITC | Fusarium graminearum, Fusarium poae | PP | MG | [113] |
| Brassica juncea | PEITC | Gaeumannomyces graminis | PP | MG | [57] |
| Brassica juncea and Sinapis alba mixture | AITC | Hypocrea lixii, Ilyonectria destructans, Mortierella alpina, Rhizoctonia solani | PP | MG | [114] |
| Brassica napus | MeITC | Gaeumannomyces graminis | PP | MG | [57] |
| Brassica napus | PEITC | Bipolaris sorokiniana, Fusarium graminearum, Gaeumannomyces graminis, Rhizoctonia solani | PP | MG | [109] |
| Brassica napus | AITC, BnITC, PEITC | Fusarium oxysporum, Sclerotium cepivorum | PP | SPG, SCG | [110] |
| Brassica napus and Brassica rapa mixture | 3-BuITC, 4-PeITC, 5-MeSPeITC, PEITC | Rhizoctonia fragariae | PP | MG | [115] |
| Brassica nigra | AITC | Fusarium sambucinum | PP | MG | [26] |
| Brassica oleracea | AITC | Rhizoctonia solani | PP | MG | [17] |
| Brassica oleracea | 3-BuITC, AITC | Rhizoctonia solani | PP | MG | [104] |
| Brassica oleracea | 2(R)-2OH-3BuITC,4-MeSBuITC,4-MeSOBuITC, AITC | Candida albicans | HR | MG | [116] |
| Bunias orientalis | 4-MeSO-3-BuITC,4-OHBnITC, BuITC, iPrITC | Alternaria brassicae, Botrytis cinerea | PP | MG | [20] |
| Cardaria draba | 4-MeSBuITC, 4-MeSOBuITC, 4-MeSOOBuITC | Candida albicans, Penicillium sp., Rhizopus stolonifer, | ENV, HR | MG | [19] |
| Carica papaya | BnITC | Aspergillus amestelodanii, A. fumigatus, A. niger, Candida albicans, C. lipolytica, Cladosporium cladosporioides, Endomycopsis fibuliger, Gliocladium roseum, Mucor sp., Penicillium chrysogenum, P. cyclopium, P. digitatum, P. expansum, P. lilacinum, P. notatum, P. spinulosum, Saccharomyces cerevisiae, S. fragilis | ENV, HR, PP | MG | [30] |
| Degenia velebitica | 4-PeITC | Candida albicans | HR | MG | [117] |
| Diplotaxis harra | 3-BuITC, iPrITC | Aspergillus niger, Fusarium oxysporum, Kluyveromyces lactis, Saccharomyces cerevisiae | ENV, PP | MG | [118] |
| Eruca sativa | 4-MeSBuITC | Sclerotinia sclerotiorum | PP | SCG | [112] |
| Eruca sativa | 4-MeSBuITC | Malassezia furfur, Microsporum canis, Trichophyton mentagrophytes | HR | MG | [24] |
| Erucaria microcarpa | BnITC, BuITC, iPrITC | Aspergillus niger, Fusarium oxysporum, Kluyveromyces lactis, Saccharomyces cerevisiae | ENV, PP | MG | [118] |
| Erysimum corinthium | 3-MeCOPrITC, 3-MeSOOPrITC, 3-MeSOPrITC, AITC | Candida albicans | HR | MG | [119] |
| Lepidium latifolium | AITC, sBuITC | Candida albicans | ENV | MG | [68] |
| Moringa oleifera | 4-α-L-RhaBnITC | Aspergillus oryzae, Botrytis allii, Candida pseudotropicalis, C. reukaufii, Coniophora cerebella, Fusarium oxysporum, Penicillium expansum, Piricularia oryzae, Polystictus versicolor, Saccharomyces carlsbergensis, Zygorrhynchus sp. | ENV, HR, PP | MG | [29] |
| Raphanus sativus | 4-MeS-3-BuITC, 4-MeSO-3-BuITC | Candida albicans | HR | MG | [52] |
| Raphanus sativus | 4-MeSO-3-BuITC | Sclerotinia sclerotiorum | PP | SCG | [112] |
| Raphanus sativus | 3-MeSO-3-BuITC, 2-OH-4-PeITC | Candida albicans | HR | MG | [116] |
| Salvadora persica | BnITC, 3-MeOBnITC, 3-OHBnITC | Aspergillus niger, Candida albicans | HR | MG | [83] |
| Sinapis alba | 4-OHBnITC | Fusarium graminearum | PP | MG | [106] |
| Sinapis alba | 4-OHBnITC | Fusarium graminearum | PP | CG, MG, PF, SG | [120] |
| Sinapis alba | 4-OHBnITC | Sclerotinia sclerotiorum | PP | SCG | [112] |
| Sinapis alba | AITC | Candida albicans | HR | MG | [116] |
| Sinapis alba | AITC, BnITC, PEITC | Fusarium oxysporum, Sclerotium cepivorum | PP | SPG, SCG | [110] |
| Sisymbrium officinale | iPrITC, sBuITC | Aspergillus niger, Candida albicans, Penicillium sp., Saccharomyces cerevisiae, | ENV, HR, PP | MG | [121] |
| Tropaeolum pentaphyllum | BnITC | Aspergillus flavus, A. fumigatus, A. niger, Candida albicans, C. dubliniensis, C. glabrata, C. guilliermondii, C. parapsilosis, C. tropicalis, Cryptococcus neoformans, Fonsecaea pedrosoi, Fusarium solani, Microsporum canis, Pseudallescheria boydii, Saccharomyces cerevisiae, Sporothrix schenckii, Trichophyton rubrum | HR, ENV | MG | [23] |
| Wasabia japonica | AITC | Beauveria bassiana, Isaria fumosorosea | EP | CG | [64] |
3. Proposed Mechanisms of the Isothiocyanate Antifungal Activity
3.1. The Role of Isothiocyanate Reactivity in Bioactivity
3.2. Possible Targets of ITCs
3.3. Transcriptome-Level in Isothiocyanate-Exposed Fungi Reveal Defense Mechanisms
3.4. Metabolism and Detoxification of Isothiocyanates by Fungi
3.5. Efflux of Isothiocyanates
3.6. Inhibition of Aflatoxin Biosynthesis
4. Quantitative Structure—Activity Relationship (QSAR) Data
5. Synergistic Activity
6. Biofumigation, Inhibition of Plant Pathogenesis
6.1. Plant Protection Studies
6.2. Parameters Influencing Potency
6.3. Contribution of Isothiocyanates and Organic Matter to Soil Microbiome Changes during Biofumigation
6.4. Group-Level Changes in the Soil Microbial Community after Biofumigation
6.5. Functional Studies
6.6. Pattern Change Studies by Gel Separation Techniques
6.7. Pattern Change Studies by Sequencing
6.8. Combinations with Other Biocontrol Agents
6.9. Effect on Mycorrhizae and Isothiocyanate-Mediated Allelopathic Activity
6.10. Phytotoxicity
7. Preservation and Other Applications
7.1. Grains and Oil Seeds
7.2. Bread and Bakery-Products
7.3. Dairy Products
7.4. Post-Harvest Preservation of Fruits
7.5. Other Products
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Neilson, E.H.; Goodger, J.Q.D.; Woodrow, I.E.; Møller, B.L. Plant chemical defense: At what cost? Trends Plant Sci. 2013, 18, 250–258. [Google Scholar] [CrossRef]
- Pongrac, P.; Vogel-Mikuš, K.; Poschenrieder, C.; Barceló, J.; Tolrà, R.; Regvar, M. Arbuscular Mycorrhiza in Glucosinolate-Containing Plants: The Story of the Metal Hyperaccumulator Noccaea (Thlaspi) praecox (Brassicaceae). In Molecular Microbial Ecology of the Rhizosphere; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2013; pp. 1023–1032. ISBN 978-1-118-29767-4. [Google Scholar]
- Sønderby, I.E.; Geu-Flores, F.; Halkier, B. Biosynthesis of glucosinolates-gene discovery and beyond. Trends Plant Sci. 2010, 15, 283–290. [Google Scholar] [CrossRef]
- Wittstock, U.; Kurzbach, E.; Herfurth, A.M.; Stauber, E.J. Glucosinolate Breakdown. In Advances in Botanical Research; Academic Press: Cambridge, MA, USA, 2016; Volume 80, pp. 125–169. [Google Scholar]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5. [Google Scholar] [CrossRef]
- Manici, L.M.; Lazzeri, A.L.; Palmieri, S. In Vitro Fungitoxic Activity of Some Glucosinolates and Their Enzyme-Derived Products toward Plant Pathogenic Fungi. J. Agric. Food Chem. 1997, 45, 2768–2773. [Google Scholar] [CrossRef]
- Pedras, M.S.C.; Hossain, S. Interaction of cruciferous phytoanticipins with plant fungal pathogens: Indole glucosinolates are not metabolized but the corresponding desulfo-derivatives and nitriles are. Phytochemistry 2011, 72, 2308–2316. [Google Scholar] [CrossRef]
- Frerigmann, H.; Piślewska-Bednarek, M.; Vallet, A.S.; Molina, A.; Glawischnig, E.; Gigolashvili, T.; Bednarek, P. Regulation of Pathogen-Triggered Tryptophan Metabolism in Arabidopsis thaliana by MYB Transcription Factors and Indole Glucosinolate Conversion Products. Mol. Plant 2016, 9, 682–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yun, H.S.; Kang, B.G.; Kwon, C. Arabidopsis immune secretory pathways to powdery mildew fungi. Plant Signal. Behav. 2016, 11, e1226456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Y.; Xu, J.; Wang, X.; He, X.; Wang, Y.; Zhou, J.; Zhang, S.; Meng, X. The Arabidopsis Pleiotropic Drug Resistance Transporters PEN3 and PDR12 Mediate Camalexin Secretion for Resistance to Botrytis cinerea. Plant Cell 2019, 31, 2206–2222. [Google Scholar] [CrossRef]
- Bernardi, R.; Finiguerra, M.G.; Rossi, A.A.; Palmieri, S. Isolation and Biochemical Characterization of a Basic Myrosinase from Ripe Crambe abyssinica Seeds, Highly Specific forepi-Progoitrin. J. Agric. Food Chem. 2003, 51, 2737–2744. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kushad, M.M. Purification and characterization of myrosinase from horseradish (Armoracia rusticana) roots. Plant Physiol. Biochem. 2005, 43, 503–511. [Google Scholar] [CrossRef]
- Hanschen, F.S.; Lamy, E.; Schreiner, M.; Rohn, S. Reactivity and Stability of Glucosinolates and Their Breakdown Products in Foods. Angew. Chem. Int. Ed. 2014, 53, 11430–11450. [Google Scholar] [CrossRef] [PubMed]
- Zsolnai, T. The antimicrobial activity of thiocyanates and isothiocyantes. 1. Arzneimittelforschung 1966, 16, 870–876. [Google Scholar] [PubMed]
- Tang, J.; Niu, J.; Wang, W.; Huo, H.; Li, J.; Luo, L.; Cao, Y. p-Aromatic Isothiocyanates: Synthesis and Anti Plant Pathogen Activity. Russ. J. Gen. Chem. 2018, 88, 1252–1257. [Google Scholar] [CrossRef]
- Tajima, H.; Kimoto, H.; Taketo, Y.; Taketo, A. Effects of Synthetic Hydroxy Isothiocyanates on Microbial Systems. Biosci. Biotechnol. Biochem. 1998, 62, 491–495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Charron, C.S.; Sams, C.E. Inhibition of Pythium ultimum and Rhizoctonia solani by Shredded Leaves of Brassica Species. J. Am. Soc. Hortic. Sci. 1999, 124, 462–467. [Google Scholar] [CrossRef] [Green Version]
- Sotelo, T.; Lema, M.; Soengas, P.; Cartea, M.E.; Velasco, P. In Vitro Activity of Glucosinolates and Their Degradation Products against Brassica-Pathogenic Bacteria and Fungi. Appl. Environ. Microbiol. 2015, 81, 432–440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radonić, A.; Blažević, I.; Mastelić, J.; Zekić, M.; Skočibušić, M.; Maravić, A. Phytochemical Analysis and Antimicrobial Activity of Cardaria draba (L.) Desv. Volatiles. Chem. Biodivers. 2011, 8, 1170–1181. [Google Scholar] [CrossRef]
- Tewes, L.J.; Müller, C. Interactions of Bunias orientalis plant chemotypes and fungal pathogens with different host specificity In Vivo and In Vitro. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Padla, E.P.; Solis, L.T.; Levida, R.M.; Shen, C.-C.; Ragasa, C.Y. Antimicrobial Isothiocyanates from the Seeds of Moringa oleifera Lam. Z. Nat. C 2012, 67, 557–564. [Google Scholar] [CrossRef]
- Choi, K.-D.; Kim, H.-Y.; Shin, I.-S. Antifungal activity of isothiocyanates extracted from horseradish (Armoracia rusticana) root against pathogenic dermal fungi. Food Sci. Biotechnol. 2017, 26, 847–852. [Google Scholar] [CrossRef]
- Da Cruz, R.C.; Denardi, L.B.; Mossmann, N.J.; Piana, M.; Alves, S.H.; De Campos, M.M.A. Antimicrobial Activity and Chromatographic Analysis of Extracts from Tropaeolum pentaphyllum Lam. Tubers. Molecules 2016, 21, 566. [Google Scholar] [CrossRef] [Green Version]
- Taha, K.F.; El-Hawary, S.S.; El-Hefnawy, H.M.; Mabrouk, M.I.; Sanad, R.A.; El Harriry, M.Y. Formulation and Assessment of a Herbal Hair Cream against Certain Dermatophytes. Int. J. Pharm. Pharm. Sci. 2016, 8, 167–173. [Google Scholar]
- Witzel, K.; Hanschen, F.; Schreiner, M.; Krumbein, A.; Ruppel, S.; Grosch, R. Verticillium Suppression Is Associated with the Glucosinolate Composition of Arabidopsis thaliana Leaves. PLoS ONE 2013, 8, e71877. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mayton, H.S.; Olivier, C.; Vaughn, S.F.; Loria, R. Correlation of Fungicidal Activity of Brassica Species with Allyl Isothiocyanate Production in Macerated Leaf Tissue. Phytopathology 1996, 86, 267–271. [Google Scholar] [CrossRef]
- Blažević, I.; Radonić, A.; Mastelić, J.; Zekić, M.; Skočibušić, M.; Maravić, A. Glucosinolates, glycosidically bound volatiles and antimicrobial activity of Aurinia sinuata (Brassicaceae). Food Chem. 2010, 121, 1020–1028. [Google Scholar] [CrossRef]
- Manici, L.M.; Lazzeri, L.; Baruzzi, G.; Leoni, O.; Galletti, S.; Palmieri, S. Suppressive Activity of Some Glucosinolate Enzyme Degradation Products on Pythium Irregulare and Rhizoctonia Solani in Sterile Soil. Pest Manag. Sci. 2000, 56, 921–926. [Google Scholar] [CrossRef]
- Eilert, U.; Wolters, B.; Nahrstedt, A. The Antibiotic Principle of Seeds of Moringa oleifera and Moringa stenopetala. Planta Med. 1981, 42, 55–61. [Google Scholar] [CrossRef] [PubMed]
- El–Tayeb, O.; Kucera, M.; Marquis, V.O.; Kučerova, H. Contribution to the Knowledge of Nigerian Medicinal Plants-III. Study on Carica Papaya Seeds as a Source of a reliable antibiotic, the BITC. Planta Med. 1974, 26, 79–89. [Google Scholar] [CrossRef]
- Zeng, R.S.; Mallik, A.U.; Setliff, E. Growth Stimulation of Ectomycorrhizal Fungi by Root Exudates of Brassicaceae Plants: Role of Degraded Compounds of Indole Glucosinolates. J. Chem. Ecol. 2003, 29, 1337–1355. [Google Scholar] [CrossRef]
- Mari, M.; Iori, R.; Leoni, O.; Marchi, A. In Vitro activity of glucosinolate-derived isothiocyanates against postharvest fruit pathogens. Ann. Appl. Biol. 1993, 123, 155–164. [Google Scholar] [CrossRef]
- Inyang, E.N.; Butt, T.M.; Doughty, K.J.; Todd, A.D.; Archer, S. The effects of isothiocyanates on the growth of the entomopathogenic fungus Metarhizium anisopliae and its infection of the mustard beetle. Mycol. Res. 1999, 103, 974–980. [Google Scholar] [CrossRef]
- Sarwar, M.; Kirkegaard, J.A.; Wong, P.T.W.; Desmarchelier, J. Biofumigation potential of Brassicas. Plant Soil 1998, 201, 103–112. [Google Scholar] [CrossRef]
- Mithen, R.F.; Lewis, B.G.; Fenwick, G.R. In Vitro activity of glucosinolates and their products against Leptosphaeria maculans. Trans. Br. Mycol. Soc. 1986, 87, 433–440. [Google Scholar] [CrossRef]
- Radulović, N.; Dekić, M.; Stojanović-Radić, Z. A new antimicrobial glucosinolate autolysis product, 4-isothiocyanatobutanoic acid, from the diffuse wallflower (Erysimum diffusum): Methyl 4-isothiocyanatobutanoate, a long unrecognized artifact of the isolation procedure? Food Chem. 2011, 129, 125–130. [Google Scholar] [CrossRef]
- Andini, S.; Araya-Cloutier, C.; Waardenburg, L.; den Besten, H.M.W.; Vincken, J.-P. The interplay between antimicrobial activity and reactivity of isothiocyanates. LWT 2020, 134, 109843. [Google Scholar] [CrossRef]
- Dhingra, O.D.; Schurt, D.A.; Oliveira, R.D.L.; Rodrigues, F.Á. Potential of soil fumigation with mustard essential oil to substitute biofumigation by cruciferous plant species. Trop. Plant Pathol. 2013, 38, 337–342. [Google Scholar] [CrossRef] [Green Version]
- Manyes, L.; Luciano, F.B.; Mañes, J.; Meca, G. In Vitro antifungal activity of allyl isothiocyanate (AITC) against Aspergillus parasiticus and Penicillium expansum and evaluation of the AITC estimated daily intake. Food Chem. Toxicol. 2015, 83, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Delaquis, P.J.; Sholberg, P.L. Antimicrobial Activity of Gaseous Allyl Isothiocyanate. J. Food Prot. 1997, 60, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Sellam, A.; Iacomi-Vasilescu, B.; Hudhomme, P.; Simoneau, P. In Vitro antifungal activity of brassinin, camalexin and two isothiocyanates against the crucifer pathogens Alternaria brassicicola and Alternaria Brassicae. Plant Pathol. 2007, 56, 296–301. [Google Scholar] [CrossRef]
- Smolinska, U.; Morra, M.J.; Knudsen, G.R.; James, R.L. Isothiocyanates Produced by Brassicaceae Species as Inhibitors of Fusarium oxysporum. Plant Dis. 2003, 87, 407–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janatova, A.; Bernardos, A.; Smid, J.; Frankova, A.; Lhotka, M.; Kouřimská, L.; Pulkrabek, J.; Kloucek, P. Long-term antifungal activity of volatile essential oil components released from mesoporous silica materials. Ind. Crop. Prod. 2015, 67, 216–220. [Google Scholar] [CrossRef]
- Taylor, F.I.; Kenyon, D.; Rosser, S. Isothiocyanates inhibit fungal pathogens of potato in In Vitro assays: Isothiocyanates Produced by Brassica Spp. Inhibit Growth of Three Economically Important Potato Pathogens. Plant Soil 2014, 382, 281–289. [Google Scholar] [CrossRef]
- Tracz, B.L.; Bordin, K.; Nazareth, T.D.M.; Costa, L.B.; de Macedo, R.E.F.; Meca, G.; Luciano, F.B. Assessment of allyl isothiocyanate as a fumigant to avoid mycotoxin production during corn storage. LWT 2017, 75, 692–696. [Google Scholar] [CrossRef]
- Raut, J.S.; Bansode, B.S.; Jadhav, A.K.; Karuppayil, S.M. Activity of Allyl Isothiocyanate and Its Synergy with Fluconazole against Candida albicans Biofilms. J. Microbiol. Biotechnol. 2017, 27, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Kurt, Ş.; Güneş, U.; Soylu, E.M. In Vitro and In Vivo antifungal activity of synthetic pure isothiocyanates against Sclerotinia sclerotiorum. Pest Manag. Sci. 2011, 67, 869–875. [Google Scholar] [CrossRef]
- Dewitte, K.; Landschoot, S.; Carrette, J.; Audenaert, K.; DeRycke, V.; Latré, J.; Vermeir, P.; Haesaert, G. The potential of Brassicaceae biofumigant crops to manage Pleiochaeta setosa in sustainable lupin cultivation. Biol. Control 2019, 132, 161–168. [Google Scholar] [CrossRef]
- Kara, M.; Soylu, E.M. Assessment of glucosinolate-derived isothiocyanates as potential natural antifungal compounds against citrus sour rot disease agent Geotrichum citriaurantii. J. Phytopathol. 2020, 168, 279–289. [Google Scholar] [CrossRef]
- Szűcs, Z.; Plaszkó, T.; Cziáky, Z.; Kiss-Szikszai, A.; Emri, T.; Bertóti, R.; Sinka, L.T.; Vasas, G.; Gonda, S. Endophytic fungi from the roots of horseradish (Armoracia rusticana) and their interactions with the defensive metabolites of the glucosinolate-myrosinase-isothiocyanate system. BMC Plant Biol. 2018, 18, 85. [Google Scholar] [CrossRef] [Green Version]
- Drobnica, L.; Zemanova, M.; Nemec, P.; Antoš, K.; Kristian, P.; Štullerová, A.; Knoppova, V. Antifungal Activity of Isothio-cyanates and Related Compounds: I. Naturally Occurring Isothiocyanates and Their Analogues. Appl. Microbiol. 1967, 15, 701–709. [Google Scholar] [CrossRef]
- Ko, M.-O.; Kim, M.-B.; Lim, S.-B. Relationship between Chemical Structure and Antimicrobial Activities of Isothiocyanates from Cruciferous Vegetables against Oral Pathogens. J. Microbiol. Biotechnol. 2016, 26, 2036–2042. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, X.; Zhang, G.-A.; Zeng, S.-Y.; Lin, K.-C. Antifungal Vapour-phase Activity of a Combination of Allyl Isothiocyanate and Ethyl Isothiocyanate Against Botrytis cinerea and Penicillium expansum Infection on Apples. J. Phytopathol. 2011, 159, 450–455. [Google Scholar] [CrossRef]
- Tunç, S.; Chollet, E.; Chalier, P.; Preziosi-Belloy, L.; Gontard, N. Combined effect of volatile antimicrobial agents on the growth of Penicillium notatum. Int. J. Food Microbiol. 2007, 113, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Brabban, A.D.; Edwards, C. The effects of glucosinolates and their hydrolysis products on microbial growth. J. Appl. Bacteriol. 1995, 79, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Bertóti, R.; Vasas, G.; Gonda, S.; Nguyen, N.M.; Szőke, É.; Jakab, A.; Pócsi, I.; Emri, T. Glutathione protects Candida albicans against horseradish volatile oil. J. Basic Microbiol. 2016, 56, 1071–1079. [Google Scholar] [CrossRef] [PubMed]
- Angus, J.F.; Gardner, P.A.; Kirkegaard, J.A.; Desmarchelier, J.M. Biofumigation: Isothiocyanates released from Brassica roots inhibit growth of the take-all fungus. Plant Soil 1994, 162, 107–112. [Google Scholar] [CrossRef]
- Cantor, A.; Hale, A.; Aaron, J.; Traw, M.B.; Kalisz, S. Low allelochemical concentrations detected in garlic mustard-invaded forest soils inhibit fungal growth and AMF spore germination. Biol. Invasions 2011, 13, 3015–3025. [Google Scholar] [CrossRef]
- Yulianti, T.; Sivasithamparam, K.; Turner, D.W. Response of different forms of propagules of Rhizoctonia solani AG2–1 (ZG5) exposed to the volatiles produced in soil amended with green manures. Ann. Appl. Biol. 2006, 148, 105–111. [Google Scholar] [CrossRef]
- Harvey, S.G.; Hannahan, H.N.; Sams, C.E. Indian Mustard and Allyl Isothiocyanate Inhibit Sclerotium rolfsii. J. Am. Soc. Hortic. Sci. 2002, 127, 27–31. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, S.K.; Nataraj, T.; Nandini, U. Asha Biofumigation—An Effective Tool in Enhancing Yield of Capsicum by Suppressing Soil-Borne Pathogens and Augmenting Biopesticide under Protected Cultivation in India. J. Biol. Control 2020, 34, 59–65. [Google Scholar] [CrossRef]
- Park, H.-W.; Choi, K.-D.; Shin, I.-S. Antimicrobial Activity of Isothiocyanates (ITCs) Extracted from Horseradish (Armoracia rusticana) Root against Oral Microorganisms. Biocontrol Sci. 2013, 18, 163–168. [Google Scholar] [CrossRef] [Green Version]
- Hewavitharana, S.S.; Ruddell, D.; Mazzola, M. Carbon source-dependent antifungal and nematicidal volatiles derived during anaerobic soil disinfestation. Eur. J. Plant Pathol. 2014, 140, 39–52. [Google Scholar] [CrossRef]
- Atsumi, A.; Saito, T. Volatiles from wasabi inhibit entomopathogenic fungi: Implications for tritrophic interactions and biological control. J. Plant Interact. 2015, 10, 152–157. [Google Scholar] [CrossRef] [Green Version]
- Clemente, I.; Aznar, M.; Nerín, C. Synergistic properties of mustard and cinnamon essential oils for the inactivation of foodborne moulds In Vitro and on Spanish bread. Int. J. Food Microbiol. 2019, 298, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Jurado, F.; Cervantes-Rincón, T.; Bach, H.; López-Malo, A.; Palou, E. Antimicrobial activity of Mexican oregano (Lippia berlandieri), thyme (Thymus vulgaris), and mustard (Brassica nigra) essential oils in gaseous phase. Ind. Crop. Prod. 2019, 131, 90–95. [Google Scholar] [CrossRef]
- Mejía-Garibay, B.; Palou, E.; López-Malo, A. Composition, Diffusion, and Antifungal Activity of Black Mustard (Brassica nigra) Essential Oil When Applied by Direct Addition or Vapor Phase Contact. J. Food Prot. 2015, 78, 843–848. [Google Scholar] [CrossRef]
- Blažević, I.; Đulović, A.; Maravić, A.; Čulić, V.Č.; Montaut, S.; Rollin, P. Antimicrobial and Cytotoxic Activities of Lepidium latifolium L. Hydrodistillate, Extract and Its Major Sulfur Volatile Allyl Isothiocyanate. Chem. Biodivers. 2019, 16, e1800661. [Google Scholar] [CrossRef]
- Ugolini, L.; Martini, C.; Lazzeri, L.; D’Avino, L.; Mari, M. Control of postharvest grey mould (Botrytis cinerea Per.: Fr.) on strawberries by glucosinolate-derived allyl-isothiocyanate treatments. Postharvest Biol. Technol. 2014, 90, 34–39. [Google Scholar] [CrossRef]
- Nazareth, T.D.M.; Alonso-Garrido, M.; Stanciu, O.; Mañes, J.; Manyes, L.; Meca, G. Effect of allyl isothiocyanate on transcriptional profile, aflatoxin synthesis, and Aspergillus flavus growth. Food Res. Int. 2020, 128. [Google Scholar] [CrossRef]
- Chung, W.C.; Huang, H.C.; Chiang, B.T.; Huang, H.C.; Huang, J.W. Inhibition of soil-borne plant pathogens by the treatment of sinigrin and myrosinases released from reconstructed Escherichia coli and Pichia pastoris. Biocontrol Sci. Technol. 2005, 15, 455–465. [Google Scholar] [CrossRef]
- Tsunoda, K. Gaseous treatment with allyl isothiocyanate to control established microbial infestation on wood. J. Wood Sci. 2000, 46, 154–158. [Google Scholar] [CrossRef]
- Bernardos, A.; Bozik, M.; Alvarez, S.; Saskova, M.; Pérez-Esteve, É.; Kloucek, P.; Lhotka, M.; Frankova, A.; Martinez-Manez, R. The efficacy of essential oil components loaded into montmorillonite against Aspergillus niger and Staphylococcus aureus. Flavour Fragr. J. 2019, 34, 151–162. [Google Scholar] [CrossRef]
- Hu, P.; Wang, A.S.; Engledow, A.S.; Hollister, E.B.; Rothlisberger, K.L.; Matocha, J.E.; Zuberer, D.A.; Provin, T.L.; Hons, F.M.; Gentry, T.J. Inhibition of the germination and growth of Phymatotrichopsis omnivora (cotton root rot) by oilseed meals and isothiocyanates. Appl. Soil Ecol. 2011, 49, 68–75. [Google Scholar] [CrossRef]
- Ren, Z.; Li, Y.; Fang, W.; Yan, D.; Huang, B.; Zhu, J.; Wang, X.; Wang, X.; Wang, Q.; Guo, M.; et al. Evaluation of allyl isothiocyanate as a soil fumigant against soil-borne diseases in commercial tomato (Lycopersicon esculentum Mill.) production in China. Pest Manag. Sci. 2018, 74, 2146–2155. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, P.V.; Rios, R. Inhibition of fungal growth on bread by volatile components from spices and herbs, and the possible application in active packaging, with special emphasis on mustard essential oil. Int. J. Food Microbiol. 2000, 60, 219–229. [Google Scholar] [CrossRef]
- Lopes, L.F.; Meca, G.; Bocate, K.C.P.; Nazareth, T.M.; Bordin, K.; Luciano, F.B. Development of food packaging system containing allyl isothiocyanate against Penicillium nordicum in chilled pizza: Preliminary study. J. Food Process. Preserv. 2017, 42. [Google Scholar] [CrossRef]
- Saladino, F.; Quiles, J.M.; Luciano, F.B.; Mañes, J.; Fernández-Franzón, M.E.; Meca, G. Shelf life improvement of the loaf bread using allyl, phenyl and benzyl isothiocyanates against Aspergillus parasiticus. LWT 2017, 78, 208–214. [Google Scholar] [CrossRef]
- Troncoso, R.; Espinoza, C.; Sanchez-Estrada, A.; Tiznado, M.; García, H.S. Analysis of the isothiocyanates present in cabbage leaves extract and their potential application to control Alternaria rot in bell peppers. Food Res. Int. 2005, 38, 701–708. [Google Scholar] [CrossRef]
- Li, D.; Shu, Y.; Li, P.; Zhang, W.; Ni, H.; Cao, Y. Synthesis and structure–activity relationships of aliphatic isothiocyanate analogs as antibiotic agents. Med. Chem. Res. 2012, 22, 3119–3125. [Google Scholar] [CrossRef]
- Radulović, N.S.; Dekić, M.S.; Stojanović-Radić, Z.Z. Antimicrobial volatile glucosinolate autolysis products from Hornungia petraea (L.) Rchb. (Brassicaceae). Phytochem. Lett. 2012, 5, 351–357. [Google Scholar] [CrossRef]
- Zasada, I.A.; Weiland, J.E.; Reed, R.L.; Stevens, J.F. Activity of Meadowfoam (Limnanthes alba) Seed Meal Glucolimnanthin Degradation Products against Soilborne Pathogens. J. Agric. Food Chem. 2012, 60, 339–345. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Kader, M.S.; Muharram, M.M.; Foudah, A.I.; Alqarni, M.H.; Salkini, M.A. Antimicrobial Isothiocyanate Derivatives from Salvadora Persica Root “Siwak” Extract. Indo Am. J. Pharm. Sci. 2017, 4, 1224–1228. [Google Scholar] [CrossRef]
- Dekić, M.S.; Radulović, N.S.; Stojanovic, N.M.; Randjelović, P.J.; Stojanović-Radić, Z.Z.; Najman, S.; Stojanović, S. Spasmolytic, antimicrobial and cytotoxic activities of 5-phenylpentyl isothiocyanate, a new glucosinolate autolysis product from horseradish (Armoracia rusticana P. Gaertn., B. Mey. & Scherb., Brassicaceae). Food Chem. 2017, 232, 329–339. [Google Scholar] [CrossRef]
- Kurepina, N.; Kreiswirth, B.N.; Mustaev, A. Growth-inhibitory activity of natural and synthetic isothiocyanates against representative human microbial pathogens. J. Appl. Microbiol. 2013, 115, 943–954. [Google Scholar] [CrossRef] [Green Version]
- Mari, M.; Iori, R.; Leoni, O.; Marchi, A. Bioassays of glucosinolate-derived isothiocyanates against postharvest pear pathogens. Plant Pathol. 1996, 45, 753–760. [Google Scholar] [CrossRef]
- Jeon, S.R.; Lee, K.H.; Shin, D.H.; Kwon, S.S.; Hwang, J.S. Synergistic antimicrobial efficacy of mesoporous ZnO loaded with 4-(α-L-rhamnosyloxy)-benzyl isothiocyanate isolated from the Moringa oleifera seed. J. Gen. Appl. Microbiol. 2014, 60, 251–255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Ani, I.; Zimmermann, S.; Reichling, J.; Wink, M. Pharmacological synergism of bee venom and melittin with antibiotics and plant secondary metabolites against multi-drug resistant microbial pathogens. Phytomedicine 2015, 22, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Li, Y.; Bi, Y.; Zhang, M.; Zhang, T.; Zheng, X.; Dong, Y.; Huang, Y. Benzyl isothiocyanate fumigation inhibits growth, membrane integrity and mycotoxin production in Alternaria alternata. RSC Adv. 2020, 10, 1829–1837. [Google Scholar] [CrossRef] [Green Version]
- Troncoso-Rojas, R.; Sanchez-Estrada, A.; Ruelas, C.; García, H.S.; Tiznado-Hernández, M.E. Effect of benzyl isothiocyanate on tomato fruit infection development by Alternaria alternata. J. Sci. Food Agric. 2005, 85, 1427–1434. [Google Scholar] [CrossRef]
- Klingen, I.; Hajek, A.; Meadow, R.; Renwick, J.A.A. Effect of Brassicaceous plants on the survival and infectivity of insect pathogenic fungi. BioControl 2002, 47, 411–425. [Google Scholar] [CrossRef]
- Smith, B.J.; Kirkegaard, J.A. In Vitro inhibition of soil microorganisms by 2-phenylethyl isothiocyanate. Plant Pathol. 2002, 51, 585–593. [Google Scholar] [CrossRef]
- Popović, M.; Maravić, A.; Čulić Čikeš, V.; Đulović, A.; Burčul, F.; Blažević, I. Biological Effects of Glucosinolate Degradation Products from Horseradish: A Horse that Wins the Race. Biomolecules 2020, 10, 343. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.; Li, Y.; Bi, Y.; Wang, T.; Dong, Y.; Yang, Q.; Zhang, T. 2-Phenylethyl Isothiocyanate Exerts Antifungal Activity against Alternaria alternata by Affecting Membrane Integrity and Mycotoxin Production. Toxins 2020, 12, 124. [Google Scholar] [CrossRef] [Green Version]
- Pedras, M.S.C.; Abdoli, A. Biotransformation of rutabaga phytoalexins by the fungus Alternaria brassicicola: Unveiling the first hybrid metabolite derived from a phytoalexin and a fungal polyketide. Bioorgan. Med. Chem. 2017, 25, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Florez, L.V.; Scherlach, K.; Gaube, P.; Ross, C.; Sitte, E.; Hermes, C.; Rodrigues, A.; Hertweck, C.; Kaltenpoth, M. Antibiotic-producing symbionts dynamically transition between plant pathogenicity and insect-defensive mutualism. Nat. Commun. 2017, 8, 15172. [Google Scholar] [CrossRef] [PubMed]
- Johansson, N.; Pavia, C.; Chiao, J. Growth Inhibition of a Spectrum of Bacterial and Fungal Pathogens by Sulforaphane, an Isothiocyanate Product Found in Broccoli and Other Cruciferous Vegetables. Planta Med. 2008, 74, 747–750. [Google Scholar] [CrossRef]
- Borges, D.; Guzman-Novoa, E.; Goodwin, P.H. Control of the microsporidian parasite Nosema ceranae in honey bees (Apis mellifera) using nutraceutical and immuno-stimulatory compounds. PLoS ONE 2020, 15, e0227484. [Google Scholar] [CrossRef] [PubMed]
- Conrad, A.; Biehler, D.; Nobis, T.; Richter, H.; Engels, I.; Biehler, K.; Frank, U. Broad Spectrum Antibacterial Activity of a Mixture of Isothiocyanates from Nasturtium (Tropaeoli majoris herba) and Horseradish (Armoraciae rusticanae radix). Drug Res. 2013, 63, 65–68. [Google Scholar] [CrossRef]
- Gao, X.; Li, K.; Ma, Z.; Zou, H.; Jin, H.; Wang, J. Cucumber Fusarium wilt resistance induced by intercropping with celery differs from that induced by the cucumber genotype and is related to sulfur-containing allelochemicals. Sci. Hortic. 2020, 271, 109475. [Google Scholar] [CrossRef]
- Lin, Y.; Hussain, M.; Avery, P.B.; Qasim, M.; Fang, D.; Wang, L. Volatiles from Plants Induced by Multiple Aphid Attacks Promote Conidial Performance of Lecanicillium lecanii. PLoS ONE 2016, 11, e0151844. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kloucek, P.; Smid, J.; Flesar, J.; Havlik, J.; Titera, D.; Rada, V.; Drabek, O.; Kokoska, L. In Vitro Inhibitory Activity of Essential Oil Vapors against Ascosphaera apis. Nat. Prod. Commun. 2012, 7, 253–256. [Google Scholar] [CrossRef] [Green Version]
- Blažević, I.; Radonić, A.; Skočibušić, M.; De Nicola, G.R.; Montaut, S.; Iori, R.; Rollin, P.; Mastelić, J.; Zekić, M.; Maravić, A. Glucosinolate Profiling and Antimicrobial Screening of Aurinia Leucadea (Brassicaceae). Chem. Biodivers. 2011, 8, 2310–2321. [Google Scholar] [CrossRef]
- Chung, W.C.; Huang, J.W.; Huang, H.C.; Jen, J.F. Effect of ground Brassicaseed meal on control of Rhizoctonia damping-off of cabbage. Can. J. Plant Pathol. 2002, 24, 211–218. [Google Scholar] [CrossRef]
- Saladino, F.; Bordin, K.; Manyes, L.; Luciano, F.B.; Mañes, J.; Fernández-Franzón, M.; Meca, G. Reduction of the aflatoxins B1, B2, G1 and G2 in Italian piadina by isothiocyanates. LWT 2016, 70, 302–308. [Google Scholar] [CrossRef]
- Drakopoulos, D.; Meca, G.; Torrijos, R.; Marty, A.; Kägi, A.; Jenny, E.; Forrer, H.-R.; Six, J.; Vogelgsang, S. Control of Fusarium graminearum in Wheat with Mustard-Based Botanicals: From In Vitro to in planta. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.B.; Schutte, B.J.; Guzman, I.; Sanogo, S. Water availability influences the inhibitory effects of mustard seed meal on Palmer amaranth (Amaranthus palmeri) and Verticillium dahliae. Weed Technol. 2020, 34, 756–763. [Google Scholar] [CrossRef]
- Augustine, R.; Bisht, N.C. Biofortification of oilseed Brassica juncea with the anti-cancer compound glucoraphanin by suppressing GSL-ALK gene family. Sci. Rep. 2015, 5, 18005. [Google Scholar] [CrossRef] [Green Version]
- Kirkegaard, J.; Wong, P.T.W.; Desmarchelier, J.M. In Vitro suppression of fungal root pathogens of cereals by Brassica tissues. Plant Pathol. 1996, 45, 593–603. [Google Scholar] [CrossRef]
- Smolinska, U.; Horbowicz, M. Fungicidal Activity of Volatiles from Selected Cruciferous Plants against Resting Propagules of Soil-borne Fungal Pathogens. J. Phytopathol. 1999, 147, 119–124. [Google Scholar] [CrossRef]
- Rahimi, F.; Rahmanpour, S.; Rezaee, S.; Larijani, K. Effect of volatiles derived from Brassica plants on the growth of Sclerotinia sclerotiorum. Arch. Phytopathol. Plant Prot. 2013, 47, 15–28. [Google Scholar] [CrossRef]
- Warmington, R.; Clarkson, J.P. Volatiles from biofumigant plants have a direct effect on carpogenic germination of sclerotia and mycelial growth of Sclerotinia sclerotiorum. Plant Soil 2016, 401, 213–229. [Google Scholar] [CrossRef] [Green Version]
- Vandicke, J.; De Visschere, K.; Deconinck, S.; Leenknecht, D.; Vermeir, P.; Audenaert, K.; Haesaert, G. Uncovering the biofumigant capacity of allyl isothiocyanate from several Brassicaceae crops against Fusarium pathogens in maize. J. Sci. Food Agric. 2020, 100, 5476–5486. [Google Scholar] [CrossRef]
- Wang, L.; Mazzola, M. Effect of Soil Physical Conditions on Emission of Allyl Isothiocyanate and Subsequent Microbial Inhibition in Response to Brassicaceae Seed Meal Amendment. Plant Dis. 2019, 103, 846–852. [Google Scholar] [CrossRef]
- Mattner, S.W.; Porter, I.J.; Gounder, R.K.; Shanks, A.L.; Wren, D.J.; Allen, D. Factors that impact on the ability of biofumigants to suppress fungal pathogens and weeds of strawberry. Crop. Prot. 2008, 27, 1165–1173. [Google Scholar] [CrossRef]
- Goralska, K.; Dynowska, M.; Ciska, E. Fungistatic Properties of Glucosinolates—A Reconnaissance Study. Pol. J. Environ. Stud. 2009, 18, 377–382. [Google Scholar]
- Mastelić, J.; Blažević, I.; Kosalec, I. Chemical Composition and Antimicrobial Activity of Volatiles from Degenia velebitica, a European Stenoendemic Plant of the Brassicaceae Family. Chem. Biodivers. 2010, 7, 2755–2765. [Google Scholar] [CrossRef] [PubMed]
- Hashem, F.A.; Saleh, M.M. Antimicrobial Components of Some Cruciferae Plants (Diplotaxis Harra Forsk. and Erucaria Microcarpa Boiss.). Phytother. Res. 1999, 13, 329–332. [Google Scholar] [CrossRef]
- Al-Gendy, A.A.; El-Gindi, O.D.; Al Hafez, S.; Ateya, A.M. Glucosinolates, volatile constituents and biological activities of Erysimum corinthium Boiss. (Brassicaceae). Food Chem. 2010, 118, 519–524. [Google Scholar] [CrossRef]
- Drakopoulos, D.; Luz, C.; Torrijos, R.; Meca, G.; Weber, P.; Bänziger, I.; Voegele, R.T.; Six, J.; Vogelgsang, S. Use of Botanicals to Suppress Different Stages of the Life Cycle of Fusarium graminearum. Phytopathology 2019, 109, 2116–2123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blažević, I.; Radonić, A.; Mastelic, J.; Zekić, M.; Skocibusic, M.; Maravic, A. Hedge Mustard (Sisymbrium officinale): Chemical Diversity of Volatiles and Their Antimicrobial Activity. Chem. Biodivers. 2010, 7, 2023–2034. [Google Scholar] [CrossRef]
- Cejpek, K.; Valušek, J.; Velíšek, J. Reactions of Allyl Isothiocyanate with Alanine, Glycine, and Several Peptides in Model Systems. J. Agric. Food Chem. 2000, 48, 3560–3565. [Google Scholar] [CrossRef]
- Schreiner, R.P.; Koide, R.T. Mustards, mustard oils and mycorrhizas. New Phytol. 2006, 123, 107–113. [Google Scholar] [CrossRef]
- Drobnica, Ľ.; Podhradsky, D.; Gemeiner, P. Kinetics of the reactions of isothiocyanates with sulphide and thiols. Collect. Czechoslov. Chem. Commun. 1975, 40, 3688–3697. [Google Scholar] [CrossRef]
- Pócsi, I.; Prade, R.A.; Penninckx, M.J. Glutathione, Altruistic Metabolite in Fungi. In Advances in Microbial Physiology; Academic Press: Cambridge, MA, USA, 2004; Volume 49, pp. 1–76. [Google Scholar]
- Calmes, B.; N’Guyen, G.; Dumur, J.; Agustí-Brisach, C.; Campion, C.; Iacomi-Vasilescu, B.; Pignã, S.; Dias, E.; Macherel, D.; Guillemette, T.; et al. Glucosinolate-derived isothiocyanates impact mitochondrial function in fungal cells and elicit an oxidative stress response necessary for growth recovery. Front. Plant Sci. 2015, 6, 414. [Google Scholar] [CrossRef] [Green Version]
- Wilcox, A.; Murphy, M.; Tucker, D.; Laprade, D.; Roussel, B.; Chin, C.; Hallisey, V.; Kozub, N.; Brass, A.; Austriaco, N. Sulforaphane alters the acidification of the yeast vacuole. Microb. Cell 2020, 7, 129–138. [Google Scholar] [CrossRef]
- Sellam, A.; Dongo, A.; Guillemette, T.; Hudhomme, P.; Simoneau, P. Transcriptional responses to exposure to the Brassicaceous defence metabolites camalexin and allyl-isothiocyanate in the necrotrophic fungus Alternaria brassicicola. Mol. Plant Pathol. 2007, 8, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, Y.; Zhang, Z.; Cao, Y.; Li, J.; Luo, L. Allyl Isothiocyanate (AITC) Triggered Toxicity and FsYvc1 (a STRPC Family Member) Responded Sense in Fusarium solani. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Rahmanpour, S.; Backhouse, D.; Nonhebel, H. Induced tolerance of Sclerotinia sclerotiorumto isothiocyanates and toxic volatiles from Brassica species. Plant Pathol. 2009, 58, 479–486. [Google Scholar] [CrossRef]
- García-Coronado, H.; Troncoso-Rojas, R.; Tiznado-Hernández, M.E.; de la Cruz Otero, M.; Diaz-Camacho, S.P.; Báez-Flores, M.E. Analysis of a suppressive subtractive hybridization library of Alternaria alternata resistant to 2-propenyl isothiocyanate. Electron. J. Biotechnol. 2015, 18, 320–326. [Google Scholar] [CrossRef] [Green Version]
- Holland, H.L.; Brown, F.M.; Larsen, B.G. Preparation of (R)-sulforaphane by biotransformation using Helminthosporium species NRRL 4671. Tetrahedron Asymmetry 1994, 5, 1129–1130. [Google Scholar] [CrossRef]
- Holland, H.L.; Brown, F.M.; Larsen, B.G.; Zabic, M. Biotransformation of organic sulfides. Part Formation of chiral isothiocyanato sulfoxides and related compounds by microbial biotransformation. Tetrahedron Asymmetry 1995, 6, 1569–1574. [Google Scholar] [CrossRef]
- Pedras, M.S.C.; Thapa, C. Unveiling fungal detoxification pathways of the cruciferous phytoalexin rapalexin A: Sequential L-cysteine conjugation, acetylation and oxidative cyclization mediated by Colletotrichum spp. Phytochemistry 2020, 169, 112188. [Google Scholar] [CrossRef] [PubMed]
- Ishimoto, H.; Fukushi, Y.; Yoshida, T.; Tahara, S. Rhizopus and Fusarium are Selected as Dominant Fungal Genera in Rhizospheres of Brassicaceae. J. Chem. Ecol. 2000, 26, 2387–2399. [Google Scholar] [CrossRef]
- Chen, J.; Ullah, C.; Reichelt, M.; Beran, F.; Yang, Z.-L.; Gershenzon, J.; Hammerbacher, A.; Vassão, D.G. The phytopathogenic fungus Sclerotinia sclerotiorum detoxifies plant glucosinolate hydrolysis products via an isothiocyanate hydrolase. Nat. Commun. 2020, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Calmes, B.; Morel-Rouhier, M.; Bataillé-Simoneau, N.; Gelhaye, E.; Guillemette, T.; Simoneau, P. Characterization of glutathione transferases involved in the pathogenicity of Alternaria brassicicola. BMC Microbiol. 2015, 15, 123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mathieu, Y.; Prosper, P.; Buée, M.; Dumarçay, S.; Favier, F.; Gelhaye, E.; Gérardin, P.; Harvengt, L.; Jacquot, J.-P.; Lamant, T.; et al. Characterization of a Phanerochaete chrysosporium Glutathione Transferase Reveals a Novel Structural and Functional Class with Ligandin Properties. J. Biol. Chem. 2012, 287, 39001–39011. [Google Scholar] [CrossRef] [Green Version]
- Sellam, A.; Poupard, P.; Simoneau, P. Molecular cloning ofAbGst1encoding a glutathione transferase differentially expressed during exposure of Alternaria brassicicolato isothiocyanates. FEMS Microbiol. Lett. 2006, 258, 241–249. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, M.; Perrot, T.; Morel-Rouhier, M.; Mulliert, G.; Gelhaye, E.; Didierjean, C.; Favier, F. The structure of Trametes versicolor glutathione transferase Omega 3S bound to its conjugation product glutathionyl-phenethylthiocarbamate reveals plasticity of its active site. Protein Sci. 2019, 28, 1143–1150. [Google Scholar] [CrossRef]
- Perrot, T.; Schwartz, M.; Deroy, A.; Girardet, J.-M.; Kohler, A.; Morel-Rouhier, M.; Favier, F.; Gelhaye, E.; Didierjean, C. Diversity of Omega Glutathione Transferases in mushroom-forming fungi revealed by phylogenetic, transcriptomic, biochemical and structural approaches. Fungal Genet. Biol. 2021, 148, 103506. [Google Scholar] [CrossRef]
- Báez-Flores, M.E.; Troncoso-Rojas, R.; Osuna, M.A.I.; Domínguez, M.R.; Pryor, B.; Tiznado-Hernández, M.E. Differentially expressed cDNAs in Alternaria alternata treated with 2-propenyl isothiocyanate. Microbiol. Res. 2011, 166, 566–577. [Google Scholar] [CrossRef]
- Vela-Corcía, D.; Srivastava, D.A.; Dafa-Berger, A.; Rotem, N.; Barda, O.; Levy, M. MFS transporter from Botrytis cinerea provides tolerance to glucosinolate-breakdown products and is required for pathogenicity. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Saksena, N.K. Fungicidal action and structure correlation of monosubstituted phenyl isothiocyanates. Folia Microbiol. 1985, 30, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Kolm, R.H.; Danielson, U.H.; Zhang, Y.; Talalay, P.; Mannervik, B. Isothiocyanates as substrates for human glutathione transferases: Structure-activity studies. Biochem. J. 1995, 311, 453–459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drobnica, Ľ.; Augustin, J. Reaction of isothiocyanates with amino acids, peptides and proteins. III. Kinetics and mechanism of the reaction of aromatic isothiocyanates with thioglycolic acid. Collect. Czechoslov. Chem. Commun. 1965, 30, 1618–1625. [Google Scholar] [CrossRef]
- French, R.C. Stimulation of germination of teliospores of Puccinia punctiformis by nonyl, decyl, and dodecyl isothiocyanates and related volatile compounds. J. Agric. Food Chem. 1990, 38, 1604–1607. [Google Scholar] [CrossRef]
- Furuya, K.; Isshiki, K. Effect of humidity on Allyl isothiocyanate antimicrobial activity. Nippon. Shokuhin Kagaku Kogaku Kaishi 2001, 48, 738–743. [Google Scholar] [CrossRef]
- Murata, W.; Yamaguchi, Y.; Fujita, K.-I.; Yamauchi, K.; Tanaka, T.; Ogita, A. Enhancement of paraben-fungicidal activity by sulforaphane, a cruciferous vegetable-derived isothiocyanate, via membrane structural damage in Saccharomyces cerevisiae. Lett. Appl. Microbiol. 2019, 69, 403–410. [Google Scholar] [CrossRef]
- Yamada, N.; Murata, W.; Yamaguchi, Y.; Fujita, K.-I.; Ogita, A.; Tanaka, T. Enhancing the fungicidal activity of amphotericin B via vacuole disruption by benzyl isothiocyanate, a cruciferous plant constituent. Lett. Appl. Microbiol. 2021, 72, 390–398. [Google Scholar] [CrossRef]
- Lazzeri, L.; D’Avino, L.; Gies, D. Additional Benefits of the Efficacy in Containing Soilborne Pest and Pathogens with Biofumigant Plants and Materials. Acta Hortic. 2010, 883, 323–330. [Google Scholar] [CrossRef]
- Dandurand, L.-M.; Mosher, R.D.; Knudsen, G.R. Combined Effects of Brassica Napus Seed Meal and Trichoderma Harzianum on Two Soilborne Plant Pathogens. Can. J. Microbiol. 2000, 46, 1051–1057. [Google Scholar] [CrossRef]
- El-Refai, I.M. Suppression of Fusarium Oxysporum f. Sp. Vasinfectum by Volatiles Produced by Hydrolysis of Glucosinolates of Some Brassicaceae Species. Bull. Fac. Sci. Assiut Univ. D Bot. 2003, 32, 121–131. [Google Scholar]
- Motisi, N.; Montfort, F.; Doré, T.; Romillac, N.; Lucas, P. Duration of control of two soilborne pathogens following incorporation of above- and below-ground residues of Brassica juncea into soil. Plant Pathol. 2009, 58, 470–478. [Google Scholar] [CrossRef]
- Fayzalla, E.A.; El-Barougy, E.; El-Rayes, M.M. Control of Soil-Borne Pathogenic Fungi of Soybean by Biofumigation with Mustard Seed Meal. J. Appl. Sci. 2009, 9, 2272–2279. [Google Scholar] [CrossRef] [Green Version]
- Garibaldi, A.; Gilardi, G.; Clematis, F.; Gullino, M.L.; Lazzeri, L.; Malaguti, L. Effect of Green Brassica Manure and Brassica Defatted Seed Meals in Combination with Grafting and Soil Solarization against Verticillium Wilt of Eggplant and Fusarium Wilt of Lettuce and Basil. Acta Hortic. 2010, 883, 295–302. [Google Scholar] [CrossRef]
- Mazzola, M.; Zhao, X. Brassica juncea seed meal particle size influences chemistry but not soil biology-based suppression of individual agents inciting apple replant disease. Plant Soil 2010, 337, 313–324. [Google Scholar] [CrossRef]
- Yohalem, D.; Passey, T. Amendment of soils with fresh and post-extraction lavender (Lavandula angustifolia) and lavandin (Lavandula × intermedia) reduce inoculum of Verticillium dahliae and inhibit wilt in strawberry. Appl. Soil Ecol. 2011, 49, 187–196. [Google Scholar] [CrossRef]
- Handiseni, M.; Brown, J.; Zemetra, R.; Mazzola, M. Effect of Brassicaceae seed meals with different glucosinolate profiles on Rhizoctonia root rot in wheat. Crop. Prot. 2013, 48, 1–5. [Google Scholar] [CrossRef]
- Handiseni, M.; Zhou, X.-G.; Jo, Y.-K. Soil amended with Brassica juncea plant tissue reduces sclerotia formation, viability and aggressiveness of Rhizoctonia solani AG1-IA towards rice. Crop. Prot. 2017, 100, 77–80. [Google Scholar] [CrossRef]
- Morales-Rodríguez, C.; Bastianelli, G.; Aleandri, M.; Chilosi, G.; Vannini, A. Application of Trichoderma Spp. Complex and Biofumigation to Control Damping-Off of Pinus Radiata, D. Don Caused by Fusarium Circinatum Nirenberg and O’Donnell. Forests 2018, 9, 421. [Google Scholar] [CrossRef] [Green Version]
- Rongai, D.; Cerato, C.; Lazzeri, L. A natural fungicide for the control of Erysiphe betae and Erysiphe cichoracearum. Eur. J. Plant Pathol. 2009, 124, 613–619. [Google Scholar] [CrossRef]
- Rongai, D.; Basti, C.; Di Marco, C. A Natural Product for the Control of Olive Leaf Spot Caused by Fusicladium Oleagineum (Cast.) Ritschel & Braun. Phytopathol. Mediterr. 2012, 51, 276–282. [Google Scholar]
- Piccinini, E.; Ferrari, V.; Campanelli, G.; Fusari, F.; Righetti, L.; Pagnotta, E.; Lazzeri, L. Effect of two liquid formulations based on Brassica carinata co-products in containing powdery mildew on melon. Ind. Crop. Prod. 2015, 75, 48–53. [Google Scholar] [CrossRef]
- Malathrakis, N.E.; Kapetanakis, G.E.; Linardakis, D.C. Brown root rot of tomato, and its control, in Crete. Ann. Appl. Biol. 1983, 102, 251–256. [Google Scholar] [CrossRef]
- Dhingra, O.D.; Costa, M.L.N.; Silva, G.J., Jr. Potential of Allyl Isothiocyanate to Control Rhizoctonia solani Seedling Damping Off and Seedling Blight in Transplant Production. J. Phytopathol. 2004, 152, 352–357. [Google Scholar] [CrossRef]
- Ortega-Centeno, S.; Guillén-Sánchez, D.; Ramos-García, M.; Troncoso-Rojas, R.; Villanueva-Arce, R.; Bosquez-Molina, E.; Barrera-Necha, L.L. Gladiolus Rust Inoculation Methods and Evaluation of Isothiocyanates of Botanical Extracts from Plants of The Brassicaceae Family in Rust Control. Rev. Chapingo Ser. Hortic. 2010, 16, 13–22. [Google Scholar]
- Lakshman, D.K.; Chauhan, K.R.; Pandey, R.; Choudhury, B. Evaluation of Plant-Based Antifungal Chemicals and Control of Damping-off Caused by Rhizoctonia Solani. Biopestic. Int. 2017, 13, 21–34. [Google Scholar]
- Saeed, I.A.M.; Rouse, D.I.; Harkin, J.M.; Smith, K.P. Effects of Soil Water Content and Soil Temperature on Efficacy of Metham-Sodium Against Verticillium dahliae. Plant Dis. 1997, 81, 773–776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazzola, M.; Granatstein, D.M.; Elfving, D.C.; Mullinix, K. Suppression of Specific Apple Root Pathogens by Brassica napus Seed Meal Amendment Regardless of Glucosinolate Content. Phytopathology 2001, 91, 673–679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lazzeri, L.; Manici, L.M. Allelopathic Effect of Glucosinolate-containing Plant Green Manure on Pythium sp. and Total Fungal Population in Soil. HortScience 2001, 36, 1283–1289. [Google Scholar] [CrossRef]
- Pellerin, S.; Mollier, A.; Morel, C.; Plenchette, C. Effect of incorporation of Brassica napus L. residues in soils on mycorrhizal fungus colonisation of roots and phosphorus uptake by maize (Zea mays L.). Eur. J. Agron. 2007, 26, 113–120. [Google Scholar] [CrossRef]
- Prasad, P.; Kumar, J.; Pandey, S. Investigating Disease Controlling Ability of Brassica Volatiles and Their Compatibility with Trichoderma harzianum. Proc. Natl. Acad. Sci. USA India Sect. B Biol. Sci. 2018, 88, 887–896. [Google Scholar] [CrossRef]
- Lazzeri, L.; Manici, L.M. The glucosinolate-myrosinase system: A natural and practical tool for biofumigation. Acta Hortic. 2000, 89–96. [Google Scholar] [CrossRef]
- Michel, V.V.; Ançay, A.; Fleury, Y.; Camps, C. Green Manures to Control Soilborne Diseases in Greenhouse Production. Acta Hortic. 2014, 1041, 187–196. [Google Scholar] [CrossRef]
- Hoagland, L.; Carpenter-Boggs, L.; Reganold, J.P.; Mazzola, M. Role of native soil biology in Brassicaceous seed meal-induced weed suppression. Soil Biol. Biochem. 2008, 40, 1689–1697. [Google Scholar] [CrossRef]
- Van Wambeke, E.; Ceustermans, A.; De Landtsheer, A.; Coosemans, J. Combinations of soil fumigants for methyl-bromide replacement. Commun. Agric. Appl. Boil. Sci. 2009, 74, 75–84. [Google Scholar]
- Njoroge, S.M.C.; Riley, M.B.; Keinath, A.P. Effect of Incorporation of Brassica spp. Residues on Population Densities of Soilborne Microorganisms and on Damping-off and Fusarium Wilt of Watermelon. Plant Dis. 2008, 92, 287–294. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Huang, B.; Wang, Q.; Li, Y.; Fang, W.; Han, D.; Yan, D.; Guo, M.; Cao, A. Effects of fumigation with metam-sodium on soil microbial biomass, respiration, nitrogen transformation, bacterial community diversity and genes encoding key enzymes involved in nitrogen cycling. Sci. Total. Environ. 2017, 598, 1027–1036. [Google Scholar] [CrossRef]
- Yulianti, T.; Sivasithamparam, K.; Turner, D.W. Saprophytic and pathogenic behaviour of R. solani AG2–1 (ZG-5) in a soil amended with Diplotaxis tenuifolia or Brassica nigra manures and incubated at different temperatures and soil water content. Plant Soil 2007, 294, 277–289. [Google Scholar] [CrossRef]
- Yulianti, T.; Sivasithamparam, K.; Turner, D.W. Saprophytic growth of Rhizoctonia solani Kühn AG2–1 (ZG5) in soil amended with fresh green manures affects the severity of damping-off in canola. Soil Biol. Biochem. 2006, 38, 923–930. [Google Scholar] [CrossRef]
- Reardon, C.L.; Strauss, S.L.; Mazzola, M. Changes in available nitrogen and nematode abundance in response to Brassica seed meal amendment of orchard soil. Soil Biol. Biochem. 2013, 57, 22–29. [Google Scholar] [CrossRef]
- Hu, P.; Wu, L.; Hollister, E.B.; Wang, A.S.; Somenahally, A.C.; Hons, F.M.; Gentry, T.J. Fungal Community Structural and Microbial Functional Pattern Changes After Soil Amendments by Oilseed Meals of Jatropha curcas and Camelina sativa: A Microcosm Study. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Fouche, T.; Maboeta, M.; Claassens, S. Effect of Biofumigants on Soil Microbial Communities and Ecotoxicology of Earthworms (Eisenia andrei). Water Air Soil Pollut. 2016, 227, 1–11. [Google Scholar] [CrossRef]
- Wang, A.S.; Hu, P.; Hollister, E.B.; Rothlisberger, K.L.; Somenahally, A.; Provin, T.L.; Hons, F.M.; Gentry, T.J. Impact of Indian Mustard (Brassica Juncea) and Flax (Linum Usitatissimum) Seed Meal Applications on Soil Carbon, Nitrogen, and Microbial Dynamics. Appl. Environ. Soil Sci. 2012, 2012, 351609. [Google Scholar] [CrossRef] [Green Version]
- Hansen, J.C.; Schillinger, W.F.; Sullivan, T.S.; Paulitz, T.C. Rhizosphere microbial communities of canola and wheat at six paired field sites. Appl. Soil Ecol. 2018, 130, 185–193. [Google Scholar] [CrossRef]
- Ascencion, L.C.; Liang, W.-J.; Yen, T.-B. Control of Rhizoctonia solani damping-off disease after soil amendment with dry tissues of Brassica results from increase in Actinomycetes population. Biol. Control 2015, 82, 21–30. [Google Scholar] [CrossRef]
- Hansen, J.C.; Schillinger, W.F.; Sullivan, T.S.; Paulitz, T.C. Decline in Soil Microbial Abundance When Camelina Introduced into a Monoculture Wheat System. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Klose, S.; Acosta-Martínez, V.; Ajwa, H.A. Microbial community composition and enzyme activities in a sandy loam soil after fumigation with methyl bromide or alternative biocides. Soil Biol. Biochem. 2006, 38, 1243–1254. [Google Scholar] [CrossRef]
- Omirou, M.; Rousidou, C.; Bekris, F.; Papadopoulou, K.K.; Menkissoglou-Spiroudi, U.; Ehaliotis, C.; Karpouzas, D.G. The Impact of Biofumigation and Chemical Fumigation Methods on the Structure and Function of the Soil Microbial Community. Microb. Ecol. 2010, 61, 201–213. [Google Scholar] [CrossRef]
- Intanon, S.; Hulting, A.G.; Myrold, D.D.; Mallory-Smith, C.A. Short-term effects of soil amendment with meadowfoam seed meal on soil microbial composition and function. Appl. Soil Ecol. 2015, 89, 85–92. [Google Scholar] [CrossRef]
- Bressan, M.; Roncato, M.-A.; Bellvert, F.; Comte, G.; Haichar, F.E.Z.; Achouak, W.; Berge, O. Exogenous glucosinolate produced by Arabidopsis thaliana has an impact on microbes in the rhizosphere and plant roots. ISME J. 2009, 3, 1243–1257. [Google Scholar] [CrossRef]
- Mocali, S.; Landi, S.; Curto, G.; Dallavalle, E.; Infantino, A.; Colzi, C.; D’Errico, G.; Roversi, P.F.; D’Avino, L.; Lazzeri, L. Resilience of soil microbial and nematode communities after biofumigant treatment with defatted seed meals. Ind. Crop. Prod. 2015, 75, 79–90. [Google Scholar] [CrossRef]
- Yim, B.; Hanschen, F.S.; Wrede, A.; Nitt, H.; Schreiner, M.; Smalla, K.; Winkelmann, T. Effects of biofumigation using Brassica juncea and Raphanus sativus in comparison to disinfection using Basamid on apple plant growth and soil microbial communities at three field sites with replant disease. Plant Soil 2016, 406, 389–408. [Google Scholar] [CrossRef]
- Mazzola, M.; Graham, D.; Wang, L.; Leisso, R.; Hewavitharana, S.S. Application sequence modulates microbiome composition, plant growth and apple replant disease control efficiency upon integration of anaerobic soil disinfestation and mustard seed meal amendment. Crop. Prot. 2020, 132, 105125. [Google Scholar] [CrossRef]
- Hollister, E.B.; Hu, P.; Wang, A.S.; Hons, F.M.; Gentry, T.J. Differential impacts of Brassicaceous and nonBrassicaceous oilseed meals on soil bacterial and fungal communities. FEMS Microbiol. Ecol. 2013, 83, 632–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, P.; Hollister, E.B.; Somenahally, A.C.; Hons, F.M.; Gentry, T.J. Soil bacterial and fungal communities respond differently to various isothiocyanates added for biofumigation. Front. Microbiol. 2014, 5, 729. [Google Scholar] [CrossRef]
- Siebers, M.; Rohr, T.; Ventura, M.; Schütz, V.; Thies, S.; Kovacic, F.; Jaeger, K.-E.; Berg, M.; Dörmann, P.; Schulz, M. Disruption of microbial community composition and identification of plant growth promoting microorganisms after exposure of soil to rapeseed-derived glucosinolates. PLoS ONE 2018, 13, e0200160. [Google Scholar] [CrossRef] [Green Version]
- Ourry, M.; Lebreton, L.; Chaminade, V.; Guillerm-Erckelboudt, A.-Y.; Herve, M.; Linglin, J.; Marnet, N.; Ourry, A.; Paty, C.; Poinsot, D.; et al. Influence of Belowground Herbivory on the Dynamics of Root and Rhizosphere Microbial Communities. Front. Ecol. Evol. 2018, 6. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.; Gentry, T.; Hu, P.; Pierson, E.; Gu, M.; Yin, S. Impact of Brassicaceous seed meals on the composition of the soil fungal community and the incidence of Fusarium wilt on chili pepper. Appl. Soil Ecol. 2015, 90, 41–48. [Google Scholar] [CrossRef]
- Zhu, J.; Ren, Z.; Huang, B.; Cao, A.; Wang, Q.; Yan, D.; Ouyang, C.; Wu, J.; Li, Y. Effects of Fumigation with Allyl Isothiocyanate on Soil Microbial Diversity and Community Structure of Tomato. J. Agric. Food Chem. 2020, 68, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Galletti, S.; Sala, E.; Leoni, O.; Burzi, P.L.; Cerato, C. Trichoderma spp. tolerance to Brassica carinata seed meal for a combined use in biofumigation. Biol. Control 2008, 45, 319–327. [Google Scholar] [CrossRef]
- Sanchi, S.; Odorizzi, S.; Lazzeri, L.; Marciano, P. Effect of Brassica Carinata Seed Meal Treatment on The Trichoderma Harzianum T39-Sclerotinia Species Interaction. Acta Hortic. 2005, 698, 287–292. [Google Scholar] [CrossRef]
- Montfort, F.; Poggi, S.; Morlière, S.; Collin, F.; LeMarchand, E.; Bailey, D. Opportunities to Reduce Rhizoctonia Solani Expression on Carrots by Biofumigation with Indian Mustard. Acta Hortic. 2011, 149–157. [Google Scholar] [CrossRef]
- Pane, C.; Villecco, D.; Zaccardelli, M. Combined Use of Brassica Carinata Seed Meal, Thyme Oil and a Bacillus Amyloliquefaciens Strain for Controlling Three Soil-Borne Fungal Plant Diseases. J. Plant Pathol. 2017, 99, 77–84. [Google Scholar] [CrossRef]
- White, C.M.; Weil, R.R. Forage radish and cereal rye cover crop effects on mycorrhizal fungus colonization of maize roots. Plant Soil 2010, 328, 507–521. [Google Scholar] [CrossRef]
- Lankau, R.A.; Wheeler, E.; Bennett, A.E.; Strauss, S.Y. Plant-soil feedbacks contribute to an intransitive competitive network that promotes both genetic and species diversity. J. Ecol. 2010, 99, 176–185. [Google Scholar] [CrossRef]
- Cipollini, D.; Rigsby, C.M.; Barto, E.K. Microbes as Targets and Mediators of Allelopathy in Plants. J. Chem. Ecol. 2012, 38, 714–727. [Google Scholar] [CrossRef]
- Cipollini, D.; Cipollini, K. A review of garlic mustard (Alliaria petiolata, Brassicaceae) as an allelopathic plant. J. Torrey Bot. Soc. 2016, 143, 339–348. [Google Scholar] [CrossRef] [Green Version]
- Wolfe, B.E.; Rodgers, V.L.; Stinson, K.A.; Pringle, A. The invasive plant Alliaria petiolata (garlic mustard) inhibits ectomycorrhizal fungi in its introduced range. J. Ecol. 2008, 96, 777–783. [Google Scholar] [CrossRef]
- Lankau, R.A. Intraspecific variation in allelochemistry determines an invasive species’ impact on soil microbial communities. Oecologia 2011, 165, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Roche, M.D.; Pearse, I.S.; Bialic-Murphy, L.; Kivlin, S.N.; Sofaer, H.R.; Kalisz, S. Negative effects of an allelopathic invader on AM fungal plant species drive community-level responses. Ecology 2021, 102. [Google Scholar] [CrossRef]
- Hale, A.N.; Lapointe, L.; Kalisz, S. Invader disruption of belowground plant mutualisms reduces carbon acquisition and alters allocation patterns in a native forest herb. New Phytol. 2016, 209, 542–549. [Google Scholar] [CrossRef]
- Agneta, R.; Rivelli, A.R.; Ventrella, E.; Lelario, F.; Sarli, G.; Bufo, S.A. Investigation of Glucosinolate Profile and Qualitative Aspects in Sprouts and Roots of Horseradish (Armoracia rusticana) Using LC-ESI–Hybrid Linear Ion Trap with Fourier Transform Ion Cyclotron Resonance Mass Spectrometry and Infrared Multiphoton Dissociation. J. Agric. Food Chem. 2012, 60, 7474–7482. [Google Scholar] [CrossRef]
- Plaszkó, T.; Szűcs, Z.; Kállai, Z.; Csoma, H.; Vasas, G.; Gonda, S. Volatile Organic Compounds (VOCs) of Endophytic Fungi Growing on Extracts of the Host, Horseradish (Armoracia rusticana). Metabolites 2020, 10, 451. [Google Scholar] [CrossRef] [PubMed]
- Baldi, E.; Toselli, M.; Malaguti, L.; Lazzeri, L. Evaluation of the biocidal effects of Brassica seed meal on Armillaria mellea. Ann. Appl. Biol. 2015, 167, 364–372. [Google Scholar] [CrossRef]
- Dhingra, O.D.; Jham, G.N.; Rodrigues, F.A.; Silva, G.J.; Costa, M.L.N. Retardation of fungal deterioration of stored soybeans by fumigation with mustard essential oil. Australas. Plant Pathol. 2009, 38, 540–545. [Google Scholar] [CrossRef]
- Otoni, C.G.; Soares, N.D.F.F.; Da Silva, W.A.; Medeiros, E.A.A.; Junior, J.C.B. Use of Allyl Isothiocyanate-containing Sachets to Reduce Aspergillus flavus Sporulation in Peanuts. Packag. Technol. Sci. 2014, 27, 549–558. [Google Scholar] [CrossRef]
- Quiles, J.M.; Nazareth, T.M.; Luz, C.; Luciano, F.B.; Mañes, J.; Meca, G. Development of an Antifungal and Antimycotoxigenic Device Containing Allyl Isothiocyanate for Silo Fumigation. Toxins 2019, 11, 137. [Google Scholar] [CrossRef] [Green Version]
- Tracz, B.L.; Bordin, K.; Bocate, K.C.P.; Hara, R.V.; Luz, C.; Macedo, R.E.F.; Meca, G.; Luciano, F.B. Devices containing allyl isothiocyanate against the growth of spoilage and mycotoxigenic fungi in mozzarella cheese. J. Food Process. Preserv. 2018, 42. [Google Scholar] [CrossRef]
- Okano, K.; Nishioka, C.; Iida, T.; Ozu, Y.; Kaneko, M.; Watanabe, Y.; Mizukami, Y.; Ichinoe, M. Inhibition of Growth of Seed-Borne Fungi and Aflatoxin Production on Stored Peanuts by Allyl Isothiocyanate Vapor. J. Food Hyg. Soc. Jpn. 2018, 59, 45–50. [Google Scholar] [CrossRef] [Green Version]
- Lopes, L.F.; Bordin, K.; De Lara, G.H.; Saladino, F.; Quiles, J.M.; Meca, G.; Luciano, F.B. Fumigation of Brazil nuts with allyl isothiocyanate to inhibit the growth of Aspergillus parasiticus and aflatoxin production. J. Sci. Food Agric. 2017, 98, 792–798. [Google Scholar] [CrossRef] [Green Version]
- Nazareth, T.M.; Correa, J.; Pinto, A.C.S.M.; Palma, J.B.; Meca, G.; Bordin, K.; Luciano, F.B. Evaluation of gaseous allyl isothiocyanate against the growth of mycotoxigenic fungi and mycotoxin production in corn stored for 6 months. J. Sci. Food Agric. 2018, 98, 5235–5241. [Google Scholar] [CrossRef]
- Nazareth, T.D.M.; Quiles, J.M.; Torrijos, R.; Luciano, F.B.; Mañes, J.; Meca, G. Antifungal and antimycotoxigenic activity of allyl isothiocyanate on barley under different storage conditions. LWT 2019, 112, 108237. [Google Scholar] [CrossRef]
- Santos, S.B.; Martins, M.A.; Faroni, L.R.D.; Junior, V.R.; Dhingra, O.D. Quality of maize grains treated with allyl isothiocyanate stored in hermetic bags. J. Stored Prod. Res. 2010, 46, 111–117. [Google Scholar] [CrossRef]
- Okano, K.; Ose, A.; Takai, M.; Kaneko, M.; Nishioka, C.; Ohzu, Y.; Odano, M.; Sekiyama, Y.; Mizukami, Y.; Nakamura, N.; et al. Inhibition of Aflatoxin Production and Fungal Growthon Stored Corn by Allyl Isothiocyanate Vapor. J. Food Hyg. Soc. Jpn. 2015, 56, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Suhr, K.I.; Nielsen, P.V. Antifungal activity of essential oils evaluated by two different application techniques against rye bread spoilage fungi. J. Appl. Microbiol. 2003, 94, 665–674. [Google Scholar] [CrossRef]
- Torrijos, R.; Nazareth, T.M.; Pérez, J.; Mañes, J.; Meca, G. Development of a Bioactive Sauce Based on Oriental Mustard Flour with Antifungal Properties for Pita Bread Shelf Life Improvement. Molecules 2019, 24, 1019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quiles, J.M.; Manyes, L.; Luciano, F.; Mañes, J.; Meca, G. Influence of the antimicrobial compound allyl isothiocyanate against the Aspergillus parasiticus growth and its aflatoxins production in pizza crust. Food Chem. Toxicol. 2015, 83, 222–228. [Google Scholar] [CrossRef]
- Saladino, F.; Manyes, L.; Luciano, F.B.; Mañes, J.; Franzón, M.F.; Meca, G. Bioactive compounds from mustard flours for the control of patulin production in wheat tortillas. LWT 2016, 66, 101–107. [Google Scholar] [CrossRef]
- Winther, M.; Nielsen, P.V. Active Packaging of Cheese with Allyl Isothiocyanate, an Alternative to Modified Atmosphere Packaging. J. Food Prot. 2006, 69, 2430–2435. [Google Scholar] [CrossRef]
- Troncoso-Rojas, R.; Corral-Acosta, Y.; Sanchez-Estrada, A.; García-Estrada, R.; Aguilar-Valenzuela, A.; Ojeda-Contreras, J.; Tiznado-Hernández, M.E. Postharvest treatment of isothiocyanates to control Alternaria rot in netted melon. Phytoparasitica 2009, 37, 445–451. [Google Scholar] [CrossRef]
- Mari, M.; Leoni, O.; Iori, R.; Cembali, T. Antifungal vapour-phase activity of allyl-isothiocyanate against Penicillium expansum on pears. Plant Pathol. 2002, 51, 231–236. [Google Scholar] [CrossRef]
- Wang, C.Y. Maintaining postharvest quality of raspberries with natural volatile compounds. Int. J. Food Sci. Technol. 2003, 38, 869–875. [Google Scholar] [CrossRef] [Green Version]
- Gao, H.; Wu, W.; Chen, H.; Qin, Y.; Fang, X.; Jin, T.Z. Microbial inactivation and quality improvement of tomatoes treated by package film with allyl isothiocyanate vapour. Int. J. Food Sci. Technol. 2018, 53, 1983–1991. [Google Scholar] [CrossRef]
- Wu, H.; Xue, N.; Hou, C.-L.; Feng, J.-T.; Zhang, X. Microcapsule preparation of allyl isothiocyanate and its application on mature green tomato preservation. Food Chem. 2015, 175, 344–349. [Google Scholar] [CrossRef]
- Ugolini, L.; Pagnotta, E.; Matteo, R.; Malaguti, L.; Di Francesco, A.; Lazzeri, L. Brassica meal-derived allyl-isothiocyanate postharvest application: Influence on strawberry nutraceutical and biochemical parameters. J. Sci. Food Agric. 2019, 99, 4235–4241. [Google Scholar] [CrossRef]
- Kamii, E.; Isshiki, K. Antimicrobial Efficacy of Benzyl Isothiocyanate. J. Food Hyg. Soc. Jpn. 2009, 50, 311–314. [Google Scholar] [CrossRef] [Green Version]
- Franco, W.; Pérez-Díaz, I.M.; Johanningsmeier, S.D.; McFeeters, R.F. Characteristics of Spoilage-Associated Secondary Cucumber Fermentation. Appl. Environ. Microbiol. 2012, 78, 1273–1284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Papp, N.; Gonda, S.; Kiss-Szikszai, A.; Plaszkó, T.; Lőrincz, P.; Vasas, G. Ethnobotanical and ethnopharmacological data of Armoracia rusticana P. Gaertner, B. Meyer et Scherb. in Hungary and Romania: A case study. Genet. Resour. Crop. Evol. 2018, 65, 1893–1905. [Google Scholar] [CrossRef]
- Sahab, A.; Sidkey, N.; Abed, N.; Mounir, A. Application of Anise and Rocket Essential Oils in Preservation of Old Manuscripts against Fungal Deterioration. Int. J. Conserv. Sci. 2018, 9, 235–244. [Google Scholar]
- Cai, L.; Jeremic, D.; Lim, H.; Kim, Y. β-Cyclodextrins as sustained-release carriers for natural wood preservatives. Ind. Crop. Prod. 2019, 130, 42–48. [Google Scholar] [CrossRef]
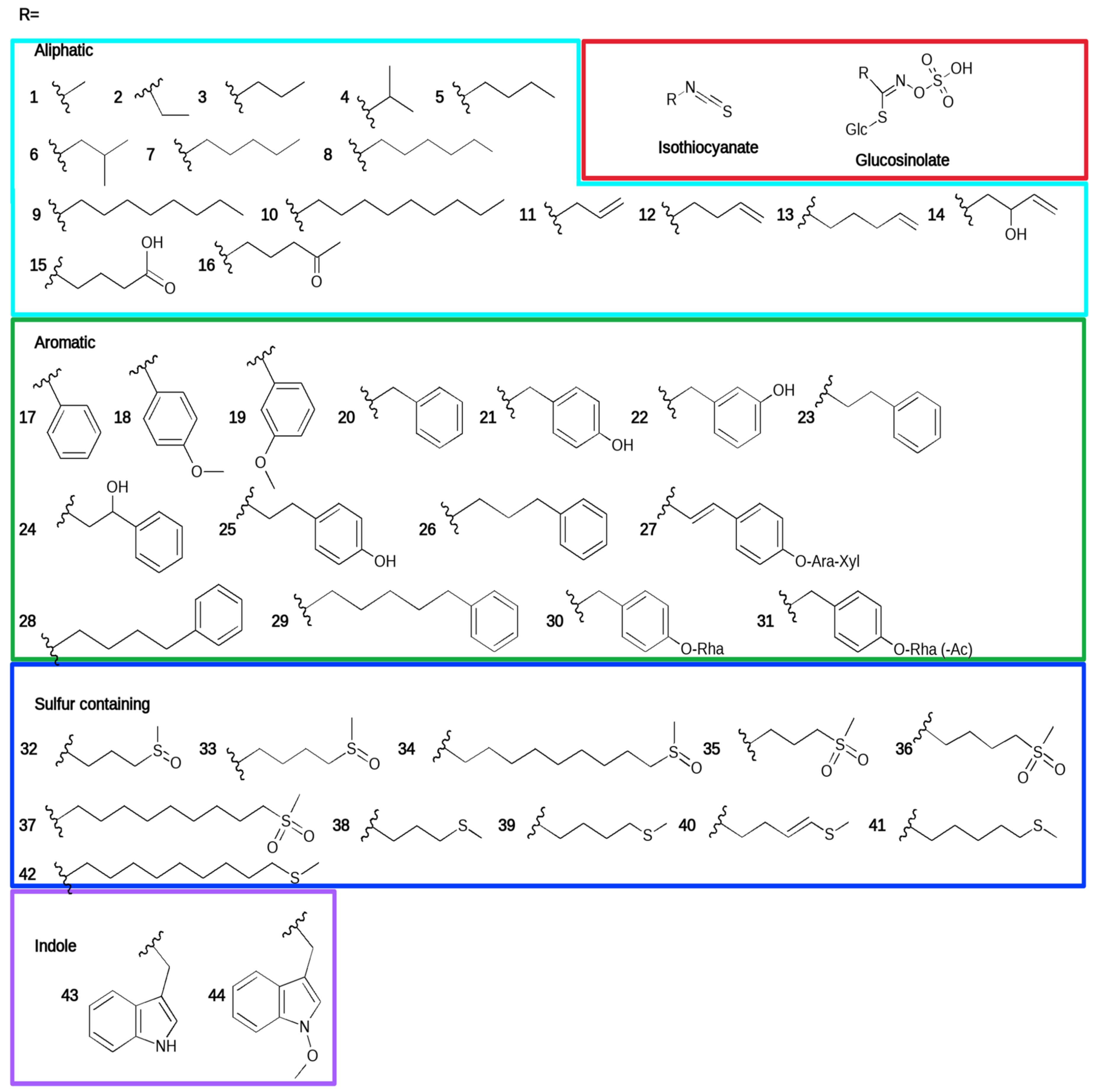


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Plaszkó, T.; Szűcs, Z.; Vasas, G.; Gonda, S. Effects of Glucosinolate-Derived Isothiocyanates on Fungi: A Comprehensive Review on Direct Effects, Mechanisms, Structure-Activity Relationship Data and Possible Agricultural Applications. J. Fungi 2021, 7, 539. https://doi.org/10.3390/jof7070539
Plaszkó T, Szűcs Z, Vasas G, Gonda S. Effects of Glucosinolate-Derived Isothiocyanates on Fungi: A Comprehensive Review on Direct Effects, Mechanisms, Structure-Activity Relationship Data and Possible Agricultural Applications. Journal of Fungi. 2021; 7(7):539. https://doi.org/10.3390/jof7070539
Chicago/Turabian StylePlaszkó, Tamás, Zsolt Szűcs, Gábor Vasas, and Sándor Gonda. 2021. "Effects of Glucosinolate-Derived Isothiocyanates on Fungi: A Comprehensive Review on Direct Effects, Mechanisms, Structure-Activity Relationship Data and Possible Agricultural Applications" Journal of Fungi 7, no. 7: 539. https://doi.org/10.3390/jof7070539
APA StylePlaszkó, T., Szűcs, Z., Vasas, G., & Gonda, S. (2021). Effects of Glucosinolate-Derived Isothiocyanates on Fungi: A Comprehensive Review on Direct Effects, Mechanisms, Structure-Activity Relationship Data and Possible Agricultural Applications. Journal of Fungi, 7(7), 539. https://doi.org/10.3390/jof7070539







