Green Leaf Volatile Confers Management of Late Blight Disease: A Green Vaccination in Potato
Abstract
1. Introduction
2. Materials and Methods
2.1. Phytophthora Infestans Genotypes
2.2. Plant Material
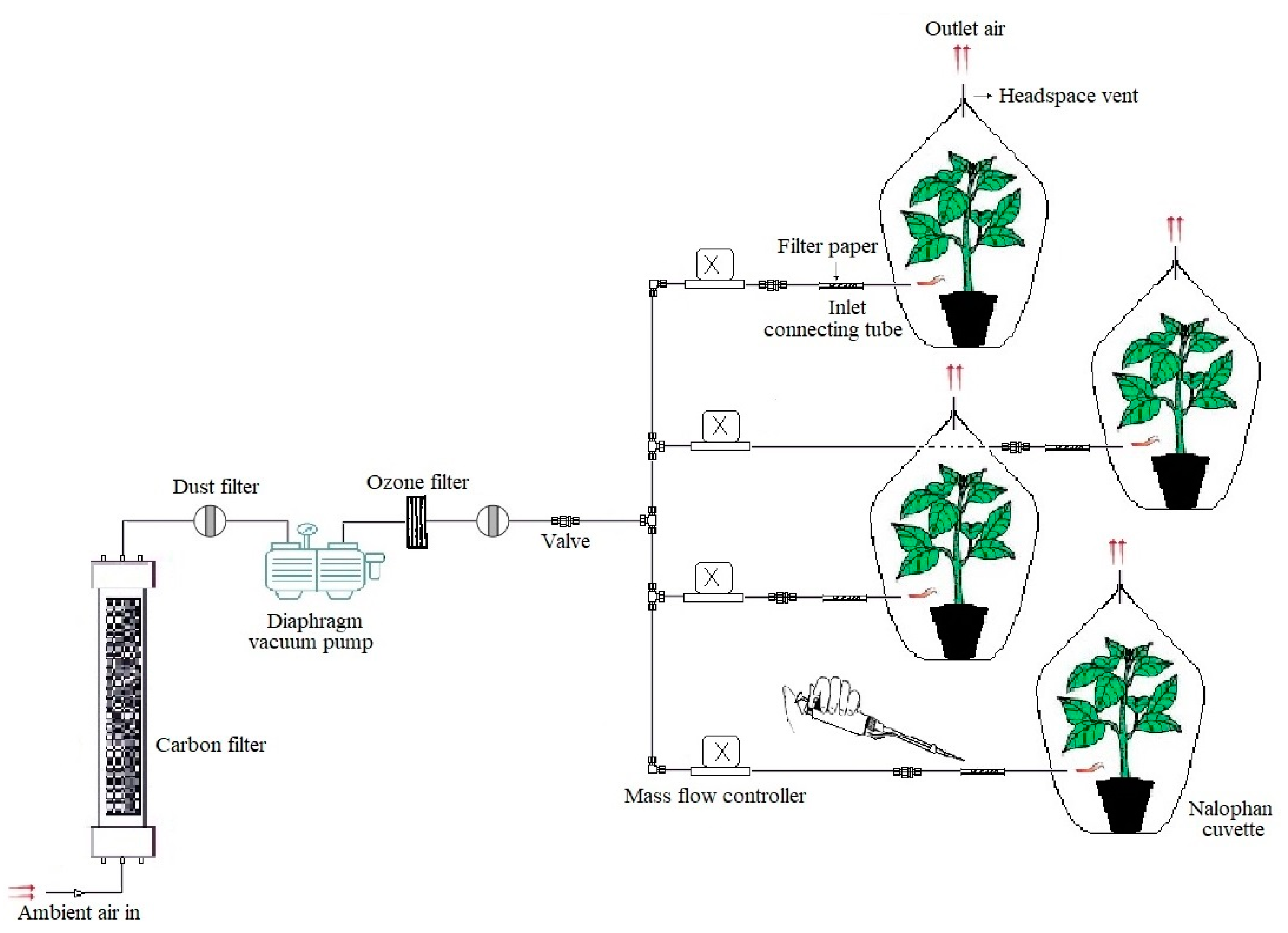
2.3. Application of Z-3-HAC to Potato Plants
2.4. Biocidal Activity of Z-3-HAC In Vivo
2.5. Visualization of Cell Death
2.6. RNA Extraction and cDNA Synthesis
2.7. Gene Expression Analysis
2.8. Statistical Analysis
3. Results
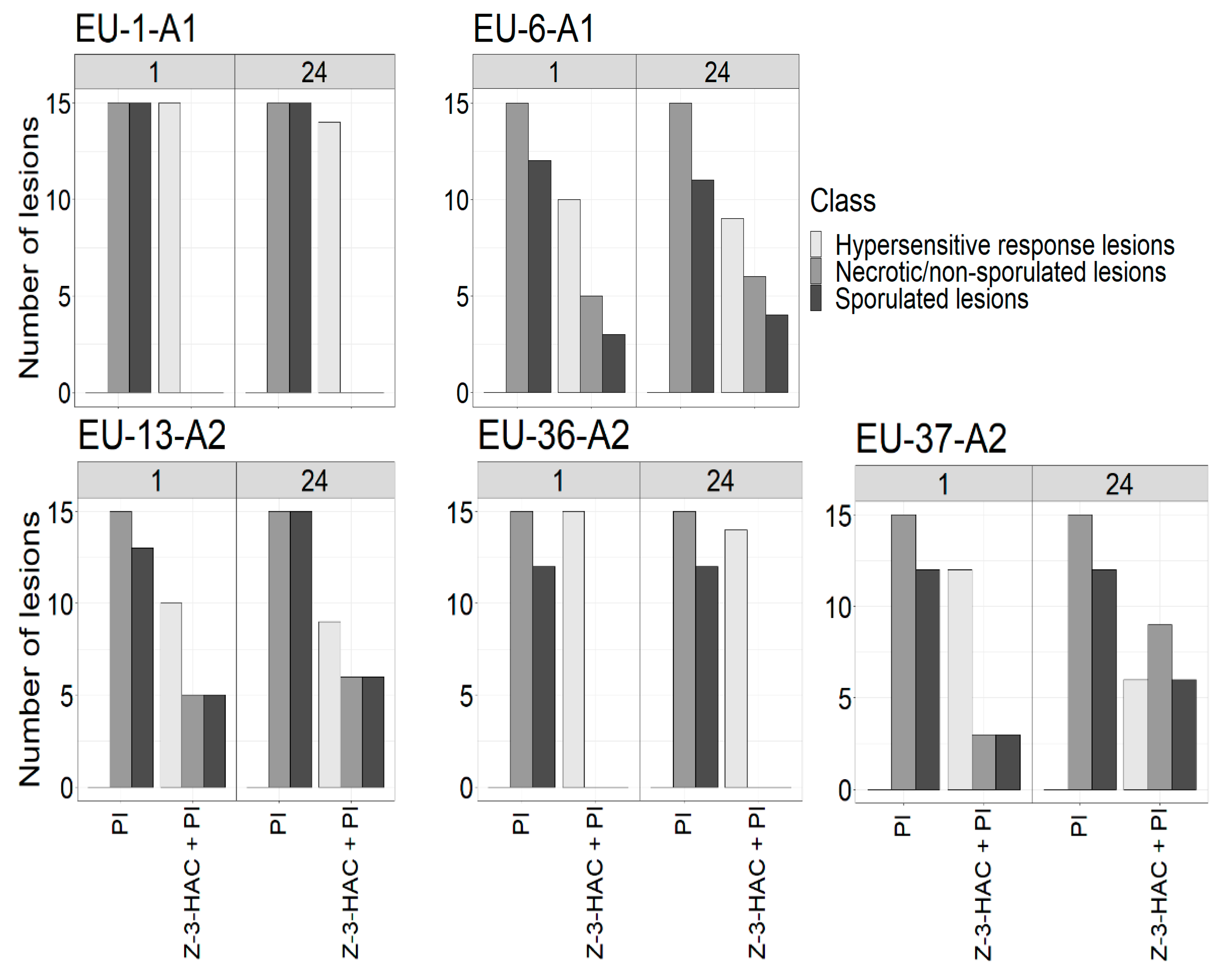
3.1. Effect of Pre-Exposure Z-3-HAC on Late Blight Symptoms
3.2. Effect of Pre-Exposure to the Z-3-HAC on Intensity of Sporulation
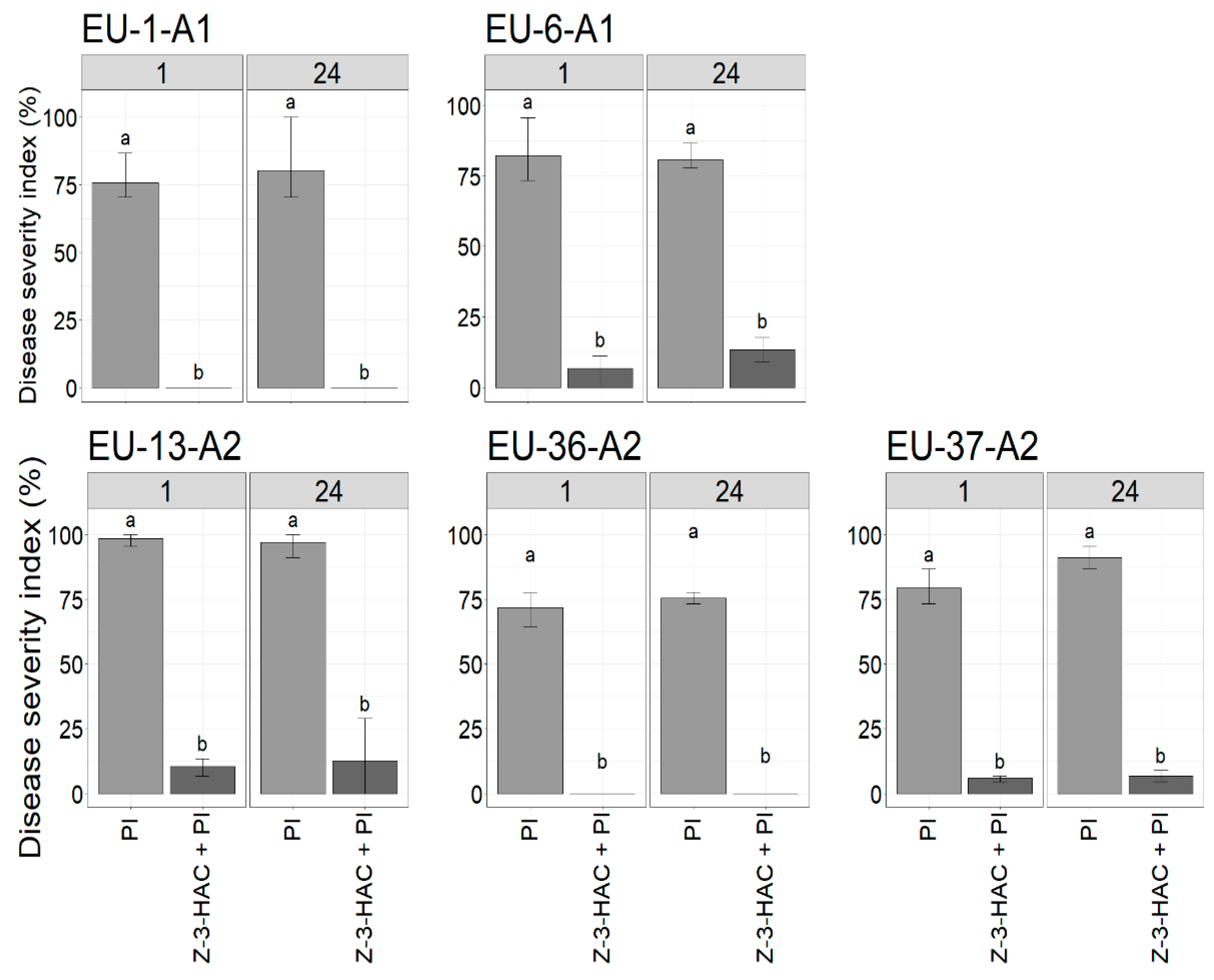
3.3. Effect of Z-3-HAC Pre-Exposure on the Severity of Late Blight Disease
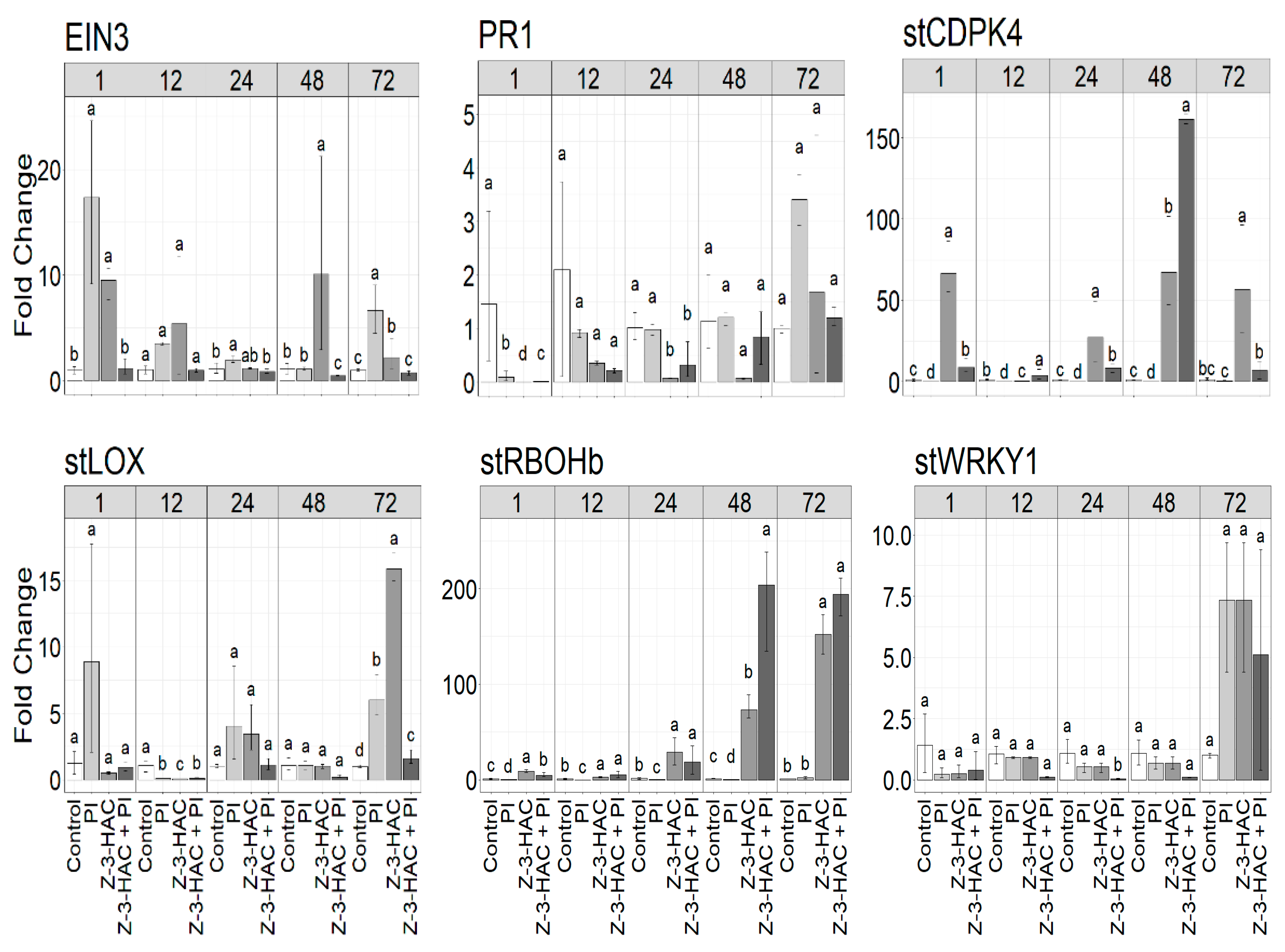
3.4. Expression Analysis of Z-3-HAC-Regulated Genes in Potato Plant
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Agrios, G.N. Plant Pathology; Academic press: Cambridge, MA, USA, 2005. [Google Scholar]
- Chinchilla, D.; Bruisson, S.; Meyer, S.; Zühlke, D.; Hirschfeld, C.; Joller, C.; L’Haridon, F.; Mène-Saffrané, L.; Riedel, K.; Weisskopf, L. A sulfur-containing volatile emitted by potato-associated bacteria confers protection against late blight through direct anti-oomycete activity. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef]
- Fry, W. Phytophthora infestans: The plant (and R gene) destroyer. Mol. Plant Pathol. 2008, 9, 385–402. [Google Scholar] [CrossRef]
- Haesaert, G.; Vossen, J.H.; Custers, R.; De Loose, M.; Haverkort, A.; Heremans, B.; Hutten, R.; Kessel, G.; Landschoot, S.; Van Droogenbroeck, B. Transformation of the potato variety Desiree with single or multiple resistance genes increases resistance to late blight under field conditions. Crop Prot. 2015, 77, 163–175. [Google Scholar] [CrossRef]
- Nowicki, M.; Foolad, M.R.; Nowakowska, M.; Kozik, E.U. Potato and tomato late blight caused by Phytophthora infestans: An overview of pathology and resistance breeding. Plant Dis. 2012, 96, 4–17. [Google Scholar] [CrossRef]
- Haverkort, A.; Boonekamp, P.; Hutten, R.; Jacobsen, E.; Lotz, L.; Kessel, G.; Visser, R.; Van der Vossen, E. Societal costs of late blight in potato and prospects of durable resistance through cisgenic modification. Potato Res. 2008, 51, 47–57. [Google Scholar] [CrossRef]
- Foolad, M.R.; Sullenberger, M.T.; Ashrafi, H. Detached-leaflet evaluation of tomato germplasm for late blight resistance and its correspondence to field and greenhouse screenings. Plant Dis. 2015, 99, 718–722. [Google Scholar] [CrossRef] [PubMed]
- Guenthner, J.; Michael, K.; Nolte, P. The economic impact of potato late blight on US growers. Potato Res. 2001, 44, 121–125. [Google Scholar] [CrossRef]
- Olle, M.; Tsahkna, A.; Tähtjärv, T.; Williams, I.H. Plant protection for organically grown potatoes–a review. Biol. Agric. Hortic. 2015, 31, 147–157. [Google Scholar] [CrossRef]
- Cha, J.-S.; Cooksey, D.A. Copper resistance in Pseudomonas syringae mediated by periplasmic and outer membrane proteins. Proc. Natl. Acad. Sci. USA 1991, 88, 8915–8919. [Google Scholar] [CrossRef] [PubMed]
- Lamichhane, J.R.; Osdaghi, E.; Behlau, F.; Köhl, J.; Jones, J.B.; Aubertot, J.-N. Thirteen decades of antimicrobial copper compounds applied in agriculture. A review. Agron. Sustain. Dev. 2018, 38, 28. [Google Scholar] [CrossRef]
- Wang, X.; Guo, M.; Min, F.; Gao, Y.; Xu, F.; Yang, S.; Lu, D. Virulence complexity and high levels of fungicide resistance suggest population change of Phytophthora infestans in the Heilongjiang Province of China. Potato Res. 2012, 55, 217–224. [Google Scholar] [CrossRef]
- De Vrieze, M.; Gloor, R.; Massana Codina, J.; Torriani, S.; Gindro, K.; L’Haridon, F.; Bailly, A.; Weisskopf, L. Biocontrol activity of three Pseudomonas in a newly assembled collection of Phytophthora infestans isolates. Phytopathology 2019, 109, 1555–1565. [Google Scholar] [CrossRef] [PubMed]
- Hung, P.M.; Wattanachai, P.; Kasem, S.; Poaim, S. Biological Control of Phytophthora palmivora Causing Root Rot of Pomelo Using Chaetomium spp. Mycobiology 2015, 43, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.-F.; He, M.-H.; Chen, F.-P.; Zhu, W.; Yang, L.-N.; Wu, E.-J.; Guo, Z.-L.; Shang, L.-P.; Zhan, J. Comparative analyses of fungicide sensitivity and SSR marker variations indicate a low risk of developing azoxystrobin resistance in Phytophthora infestans. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef]
- Lamichhane, J.R.; Dachbrodt-Saaydeh, S.; Kudsk, P.; Messéan, A. Toward a reduced reliance on conventional pesticides in European agriculture. Plant Disease 2016, 100, 10–24. [Google Scholar] [CrossRef]
- Stephan, D.; Schmitt, A.; Carvalho, S.M.; Seddon, B.; Koch, E. Evaluation of biocontrol preparations and plant extracts for the control of Phytophthora infestans on potato leaves. Eur. J. Plant Pathol. 2005, 112, 235–246. [Google Scholar] [CrossRef]
- Najdabbasi, N.; Mirmajlessi, S.M.; Dewitte, K.; Landschoot, S.; Mänd, M.; Audenaert, K.; Ameye, M.; Haesaert, G. Biocidal activity of plant-derived compounds against Phytophthora infestans: An alternative approach to late blight management. Crop Prot. 2020, 138, 105315. [Google Scholar] [CrossRef]
- Dudareva, N.; Negre, F.; Nagegowda, D.A.; Orlova, I. Plant volatiles: Recent advances and future perspectives. Crit. Rev. Plant Sci. 2006, 25, 417–440. [Google Scholar] [CrossRef]
- Scala, A.; Allmann, S.; Mirabella, R.; Haring, M.A.; Schuurink, R.C. Green leaf volatiles: A plant’s multifunctional weapon against herbivores and pathogens. Int. J. Mol. Sci. 2013, 14, 17781–17811. [Google Scholar] [CrossRef] [PubMed]
- Paré, P.W.; Tumlinson, J.H. De novo biosynthesis of volatiles induced by insect herbivory in cotton plants. Plant Physiol. 1997, 114, 1161–1167. [Google Scholar] [CrossRef]
- Hatanaka, A. The biogeneration of green odour by green leaves. Phytochemistry 1993, 34, 1201–1218. [Google Scholar] [CrossRef]
- Brilli, F.; Loreto, F.; Baccelli, I. Exploiting plant volatile organic compounds (VOCs) in agriculture to improve sustainable defense strategies and productivity of crops. Front. Plant Sci. 2019, 10, 264. [Google Scholar] [CrossRef] [PubMed]
- Fall, R.; Karl, T.; Hansel, A.; Jordan, A.; Lindinger, W. Volatile organic compounds emitted after leaf wounding: On-line analysis by proton-transfer-reaction mass spectrometry. J. Geophys. Res. Atmos. 1999, 104, 15963–15974. [Google Scholar] [CrossRef]
- Piesik, D.; Pańka, D.; Delaney, K.J.; Skoczek, A.; Lamparski, R.; Weaver, D.K. Cereal crop volatile organic compound induction after mechanical injury, beetle herbivory (Oulema spp.), or fungal infection (Fusarium spp.). J. Plant Physiol. 2011, 168, 878–886. [Google Scholar] [CrossRef] [PubMed]
- Croft, K.P.; Juttner, F.; Slusarenko, A.J. Volatile products of the lipoxygenase pathway evolved from Phaseolus vulgaris (L.) leaves inoculated with Pseudomonas syringae pv phaseolicola. Plant Physiol. 1993, 101, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Shiojiri, K.; Kishimoto, K.; Ozawa, R.; Kugimiya, S.; Urashimo, S.; Arimura, G.; Horiuchi, J.; Nishioka, T.; Matsui, K.; Takabayashi, J. Changing green leaf volatile biosynthesis in plants: An approach for improving plant resistance against both herbivores and pathogens. Proc. Natl. Acad. Sci. USA 2006, 103, 16672–16676. [Google Scholar] [CrossRef] [PubMed]
- Pańka, D.; Piesik, D.; Jeske, M.; Baturo-Cieśniewska, A. Production of phenolics and the emission of volatile organic compounds by perennial ryegrass (Lolium perenne L.)/Neotyphodium lolii association as a response to infection by Fusarium poae. J. Plant Physiol. 2013, 170, 1010–1019. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, K.; Matsui, K.; Ozawa, R.; Takabayashi, J. Volatile C6-aldehydes and allo-ocimene activate defense genes and induce resistance against Botrytis cinerea in Arabidopsis thaliana. Plant Cell Physiol. 2005, 46, 1093–1102. [Google Scholar] [CrossRef]
- Ameye, M.; Allmann, S.; Verwaeren, J.; Smagghe, G.; Haesaert, G.; Schuurink, R.C.; Audenaert, K. Green leaf volatile production by plants: A meta-analysis. New Phytol. 2018, 220, 666–683. [Google Scholar] [CrossRef] [PubMed]
- Kuźnicki, D.; Meller, B.; Arasimowicz-Jelonek, M.; Braszewska-Zalewska, A.; Drozda, A.; Floryszak-Wieczorek, J. BABA-induced DNA methylome adjustment to intergenerational defense priming in potato to Phytophthora infestans. Front. Plant Sci. 2019, 10, 650. [Google Scholar] [CrossRef]
- Muthamilarasan, M.; Prasad, M. Plant innate immunity: An updated insight into defense mechanism. J. Biosci. 2013, 38, 433–449. [Google Scholar] [CrossRef]
- Kishimoto, K.; Matsui, K.; Ozawa, R.; Takabayashi, J. Components of C6-aldehyde-induced resistance in Arabidopsis thaliana against a necrotrophic fungal pathogen, Botrytis cinerea. Plant Sci. 2006, 170, 715–723. [Google Scholar] [CrossRef]
- Kishimoto, K.; Matsui, K.; Ozawa, R.; Takabayashi, J. Direct fungicidal activities of C6-aldehydes are important constituents for defense responses in Arabidopsis against Botrytis cinerea. Phytochemistry 2008, 69, 2127–2132. [Google Scholar] [CrossRef]
- Vicedo, B.; Flors, V.; de la O. Leyva, M.; Finiti, I.; Kravchuk, Z.; Real, M.D.; García-Agustín, P.; González-Bosch, C. Hexanoic acid-induced resistance against Botrytis cinerea in tomato plants. Mol. Plant Microbe Interact. 2009, 22, 1455–1465. [Google Scholar] [CrossRef]
- Kravchuk, Z.; Vicedo, B.; Flors, V.; Camañes, G.; González-Bosch, C.; García-Agustín, P. Priming for JA-dependent defenses using hexanoic acid is an effective mechanism to protect Arabidopsis against B. cinerea. J. Plant Physiol. 2011, 168, 359–366. [Google Scholar] [CrossRef]
- Finiti, I.; de la O. Leyva, M.; Vicedo, B.; Gómez-Pastor, R.; López-Cruz, J.; García-Agustín, P.; Real, M.D.; González-Bosch, C. Hexanoic acid protects tomato plants against Botrytis cinerea by priming defence responses and reducing oxidative stress. Mol. Plant Pathol. 2014, 15, 550–562. [Google Scholar] [CrossRef]
- Xin, Z.; Zhang, L.; Zhang, Z.; Chen, Z.; Sun, X. A tea hydroperoxide lyase gene, CsiHPL1, regulates tomato defense response against Prodenia Litura (Fabricius) and Alternaria Alternata f. sp. Lycopersici by modulating green leaf volatiles (GLVs) release and jasmonic acid (JA) gene expression. Plant Mol. Biol. Report. 2014, 32, 62–69. [Google Scholar] [CrossRef]
- Hamilton-Kemp, T.; McCracken, C.; Loughrin, J.; Andersen, R.; Hildebrand, D. Effects of some natural volatile compounds on the pathogenic fungi Alternaria alternata and Botrytis cinerea. J. Chem. Ecol. 1992, 18, 1083–1091. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, S.; Spencer, G.; Shasha, B. Volatile compounds from raspberry and strawberry fruit inhibit postharvest decay fungi. J. Food Sci. 1993, 58, 793–796. [Google Scholar] [CrossRef]
- Nakamura, S.; Hatanaka, A. Green-leaf-derived C6-aroma compounds with potent antibacterial action that act on both gram-negative and gram-positive bacteria. J. Agric. Food Chem. 2002, 50, 7639–7644. [Google Scholar] [CrossRef] [PubMed]
- Hubert, J.; Münzbergová, Z.; Santino, A. Plant volatile aldehydes as natural insecticides against stored-product beetles. Pest Manag. Sci. 2008, 64, 57–64. [Google Scholar] [CrossRef]
- Neri, F.; Mari, M.; Brigati, S.; Bertolini, P. Fungicidal activity of plant volatile compounds for controlling Monilinia laxa in stone fruit. Plant Dis. 2007, 91, 30–35. [Google Scholar] [CrossRef]
- De Lucca, A.; Carter-Wientjes, C.; Boue, S.; Bhatnagar, D. Volatile trans-2-hexenal, a soybean aldehyde, inhibits Aspergillus flavus growth and aflatoxin production in corn. J. Food Sci. 2011, 76, 381–386. [Google Scholar] [CrossRef]
- Ameye, M.; Audenaert, K.; De Zutter, N.; Steppe, K.; Van Meulebroek, L.; Vanhaecke, L.; De Vleesschauwer, D.; Haesaert, G.; Smagghe, G. Priming of wheat with the green leaf volatile Z-3-hexenyl acetate enhances defense against Fusarium graminearum but boosts deoxynivalenol production. Plant Physiol. 2015, 167, 1671–1684. [Google Scholar] [CrossRef]
- López-Gresa, M.P.; Payá, C.; Ozáez, M.; Rodrigo, I.; Conejero, V.; Klee, H.; Bellés, J.M.; Lisón, P. A new role for green leaf volatile esters in tomato stomatal defense against Pseudomonas syringe pv. tomato. Front. Plant Sci. 2018, 9, 1855. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Wang, S.; Hu, T.; Zhang, F.; Forrer, H.; Cao, K. Screening for plant extracts to control potato late blight. Front. Agric. China 2007, 1, 43–46. [Google Scholar] [CrossRef]
- Wang, C.-F.; Huang, L.-L.; Buchenauer, H.; Han, Q.-M.; Zhang, H.-C.; Kang, Z.-S. Histochemical studies on the accumulation of reactive oxygen species (O2− and H2O2) in the incompatible and compatible interaction of wheat—Puccinia striiformis f. sp. tritici. Physiol. Mol. Plant Pathol. 2007, 71, 230–239. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Huanga, Z.; Carter, N.; Lu, H.; Zhang, Z.; Wang-Pruski, G. Translocation of phosphite encourages the protection against Phy-tophthora infestans in potato: The efficiency and efficacy. Pestic. Biochem. Physiol. 2018, 152, 122–130. [Google Scholar] [CrossRef]
- Arseneault, T.; Pieterse, C.M.J.; Gérin-Ouellet, M.; Goyer, C.; Filion, M. Long-term induction of defense gene expression in potato by Pseudomonas sp. LBUM223 and Streptomyces scabies. Phytopathology 2014, 104, 926–932. [Google Scholar] [CrossRef]
- Saubeaua, G.; Perrinab, F.; Marnetc, N.; Andrivonaand, D.; Val, F. Hormone signalling pathways are differentially involved inquantitative resistance of potato to Phytophthora infestans. Plant Pathol. 2016, 65, 342–352. [Google Scholar] [CrossRef]
- Fielding, R.T.; Taylor, R.N. Architectural Styles and the Design of Network-Based Software Architectures; University of California: Irvine, CA, USA, 2000; Volume 7. [Google Scholar]
- Heil, M.; Kost, C. Priming of indirect defences. Ecol. Lett. 2006, 9, 813–817. [Google Scholar] [CrossRef]
- Frost, C.J.; Mescher, M.C.; Dervinis, C.; Davis, J.M.; Carlson, J.E.; De Moraes, C.M. Priming defense genes and metabolites in hybrid poplar by the green leaf volatile cis-3-hexenyl acetate. New Phytol. 2008, 180, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Quaglia, M.; Fabrizi, M.; Zazzerini, A.; Zadra, C. Role of pathogen-induced volatiles in the Nicotiana tabacum–Golovinomyces cichoracearum interaction. Plant Physiol. Biochem. 2012, 52, 9–20. [Google Scholar] [CrossRef]
- Engelberth, J.; Contreras, C.F.; Dalvi, C.; Li, T.; Engelberth, M. Early transcriptome analyses of Z-3-hexenol-treated Zea mays revealed distinct transcriptional networks and anti-herbivore defense potential of green leaf volatiles. PLoS ONE 2013, 8, e77465. [Google Scholar] [CrossRef] [PubMed]
- Gorman, Z.; Christensen, S.A.; Yan, Y.; He, Y.; Borrego, E.; Kolomiets, M.V. Green leaf volatiles and jasmonic acid enhance susceptibility to anthracnose diseases caused by Colletotrichum graminicola in maize. Mol. Plant Pathol. 2020, 21, 702–715. [Google Scholar] [CrossRef] [PubMed]
- Roy, B.; Krishnan, S.P.; Chandrasekaran, N.; Mukherjee, A. Toxic effects of engineered nanoparticles (metal/metal oxides) on plants using Allium cepa as a model system. In Comprehensive Analytical Chemistry; Elsevier: Amsterdam, The Netherlands, 2019; Volume 84, pp. 125–143. [Google Scholar]
- Dorey, S.; Kopp, M.; Geoffroy, P.; Fritig, B.; Kauffmann, S. Hydrogen peroxide from the oxidative burst is neither necessary nor sufficient for hypersensitive cell death induction, phenylalanine ammonia lyase stimulation, salicylic acid accumulation, or scopoletin consumption in cultured tobacco cells treated with elicitin. Plant Physiol. 1999, 121, 163–172. [Google Scholar] [PubMed]
- Balint-Kurti, P. The plant hypersensitive response: Concepts, control and consequences. Mol. Plant Pathol. 2019, 20, 1163–1178. [Google Scholar] [CrossRef]
- Spanu, P.D.; Panstruga, R. Biotrophic plant-microbe interactions. Front. Plant Sci. 2017, 8, 192. [Google Scholar] [CrossRef]
- Himanen, S.J.; Blande, J.D.; Klemola, T.; Pulkkinen, J.; Heijari, J.; Holopainen, J.K. Birch (Betula spp.) leaves adsorb and rerelease volatiles specific to neighbouring plants–a mechanism for associational herbivore resistance? New Phytol. 2010, 186, 722–732. [Google Scholar] [CrossRef] [PubMed]
- Quintana-Rodriguez, E.; Morales-Vargas, A.T.; Molina-Torres, J.; Ádame-Alvarez, R.M.; Acosta-Gallegos, J.A.; Heil, M. Plant volatiles cause direct, induced and associational resistance in common bean to the fungal pathogen Colletotrichum lindemuthianum. J. Ecol. 2015, 103, 250–260. [Google Scholar] [CrossRef]
- Cooke, L.; Schepers, H.; Hermansen, A.; Bain, R.; Bradshaw, N.; Ritchie, F.; Shaw, D.; Evenhuis, A.; Kessel, G.; Wander, J. Epidemiology and integrated control of potato late blight in Europe. Potato Res. 2011, 54, 183–222. [Google Scholar] [CrossRef]
- Mariette, N.; Mabon, R.; Corbière, R.; Boulard, F.; Glais, I.; Marquer, B.; Pasco, C.; Montarry, J.; Andrivon, D. Phenotypic and genotypic changes in French populations of Phytophthora infestans: Are invasive clones the most aggressive? Plant. Pathol. 2016, 65, 577–586. [Google Scholar] [CrossRef]
- Schepers, H.; Kessel, G.; Lucca, F.; Förch, M.; van Den Bosch, G.; Topper, C.; Evenhuis, A. Reduced efficacy of fluazinam against Phytophthora infestans in the Netherlands. Eur. J. Plant. Pathol. 2018, 151, 947–960. [Google Scholar] [CrossRef]
- Hansen, J.G. Results of the EuroBlight Potato Late Blight Monitoring In 2018. 2019. Available online: https://agro.au.dk (accessed on 5 June 2018).
- Njoroge, A.; Tusiime, G.; Forbes, G.; Yuen, J. Displacement of US-1 clonal lineage by a new lineage of Phytophthora infestans on potato in Kenya and Uganda. Plant. Pathol. 2016, 65, 587–592. [Google Scholar] [CrossRef]
- Kröner, A.; Mabon, R.; Corbière, R.; Montarry, J.; Andrivon, D. The coexistence of generalist and specialist clonal lineages in natural populations of the Irish Famine pathogen Phytophthora infestans explains local adaptation to potato and tomato. Mol. Ecol. 2017, 26, 1891–1901. [Google Scholar] [CrossRef]
- Dey, T.; Saville, A.; Myers, K.; Tewari, S.; Cooke, D.E.; Tripathy, S.; Fry, W.E.; Ristaino, J.B.; Roy, S.G. Large sub-clonal variation in Phytophthora infestans from recent severe late blight epidemics in India. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Michalska, A.M.; Sobkowiak, S.; Flis, B.; Zimnoch-Guzowska, E. Virulence and aggressiveness of Phytophthora infestans isolates collected in Poland from potato and tomato plants identified no strong specificity. Eur. J. Plant. Pathol. 2016, 144, 325–336. [Google Scholar] [CrossRef]
- Knapova, G.; Gisi, U. Phenotypic and genotypic structure of Phytophthora infestans populations on potato and tomato in France and Switzerland. Plant. Pathol. 2002, 51, 641–653. [Google Scholar] [CrossRef]
- Sagi, M.; Davydov, O.; Orazova, S.; Yesbergenova, Z.; Ophir, R.; Stratmann, J.W.; Fluhr, R. Plant respiratory burst oxidase homologs impinge on wound responsiveness and development in Lycopersicon esculentum. Plant. Cell 2004, 16, 616–628. [Google Scholar] [CrossRef]
- Torres, M.A.; Dangl, J.L. Functions of the respiratory burst oxidase in biotic interactions, abiotic stress and development. Curr. Opin. Plant. Biol. 2005, 8, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Ohura, I.; Kawakita, K.; Yokota, N.; Fujiwara, M.; Shimamoto, K.; Doke, N.; Yoshioka, H. Calcium-dependent protein kinases regulate the production of reactive oxygen species by potato NADPH oxidase. Plant. Cell 2007, 19, 1065–1080. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Yoshioka, M.; Asai, S.; Nomura, H.; Kuchimura, K.; Mori, H.; Doke, N.; Yoshioka, H. StCDPK5 confers resistance to late blight pathogen but increases susceptibility to early blight pathogen in potato via reactive oxygen species burst. New Phytol. 2012, 196, 223–237. [Google Scholar] [CrossRef] [PubMed]
- Boudsocq, M.; Sheen, J. CDPKs in immune and stress signaling. Trends Plant. Sci. 2013, 18, 30–40. [Google Scholar] [CrossRef]
- Ameye, M.; Van Meulebroek, L.; Meuninck, B.; Vanhaecke, L.; Smagghe, G.; Haesaert, G.; Audenaert, K. Metabolomics reveal induction of ROS production and glycosylation events in wheat upon exposure to the green leaf volatile Z-3-Hexenyl Acetate. Front. Plant. Sci. 2020, 11. [Google Scholar] [CrossRef]
- Bradley, D.J.; Kjellbom, P.; Lamb, C.J. Elicitor-and wound-induced oxidative cross-linking of a proline-rich plant cell wall protein: A novel, rapid defense response. Cell 1992, 70, 21–30. [Google Scholar] [CrossRef]
- Torres, M.A.; Jones, J.D.; Dangl, J.L. Reactive oxygen species signaling in response to pathogens. Plant. Physiol. 2006, 141, 373–378. [Google Scholar] [CrossRef]
- Legendre, L.; Rueter, S.; Heinstein, P.F.; Low, P.S. Characterization of the oligogalacturonide-induced oxidative burst in cultured soybean (Glycine max) cells. Plant. Physiol. 1993, 102, 233–240. [Google Scholar] [CrossRef]
- Levine, A.; Tenhaken, R.; Dixon, R.; Lamb, C. H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 1994, 79, 583–593. [Google Scholar] [CrossRef]
- Baker, C.J.; Orlandi, E.W. Active oxygen in plant pathogenesis. Annu. Rev. Phytopathol. 1995, 33, 299–321. [Google Scholar] [CrossRef]
- Bate, N.J.; Rothstein, S.J. C6-volatiles derived from the lipoxygenase pathway induce a subset of defense-related genes. Plant. J. 1998, 16, 561–569. [Google Scholar] [CrossRef]
- Bonasera, J.M.; Kim, J.F.; Beer, S.V. PR genes of apple: Identification and expression in response to elicitors and inoculation with Erwinia amylovora. BMC Plant. Biol. 2006, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Sarowar, S.; Zhao, Y.; Soria-Guerra, R.E.; Ali, S.; Zheng, D.; Wang, D.; Korban, S.S. Expression profiles of differentially regulated genes during the early stages of apple flower infection with Erwinia amylovora. J. Exp. Bot. 2011, 62, 4851–4861. [Google Scholar] [CrossRef] [PubMed]
- Cellini, A.; Buriani, G.; Rocchi, L.; Rondelli, E.; Savioli, S.; Rodriguez Estrada, M.T.; Cristescu, S.M.; Costa, G.; Spinelli, F. Biological relevance of volatile organic compounds emitted during the pathogenic interactions between apple plants and Erwinia amylovora. Mol. Plant. Pathol. 2018, 19, 158–168. [Google Scholar] [CrossRef]
- Schröder, M.; Hahlbrock, K.; Kombrink, E. Temporal and spatial patterns of 1, 3-β-glucanase and chitinase induction in potato leaves infected by Phytophthora infestans. Plant. J. 1992, 2, 161–172. [Google Scholar] [CrossRef]
- Hoegen, E.; Strömberg, A.; Pihlgren, U.; Kombrink, E. Primary structure and tissue-specific expression of the pathogenesis-related protein PR-1b in potato. Mol. Plant. Pathol. 2002, 3, 329–345. [Google Scholar] [CrossRef] [PubMed]
- van Loon, L.C.; Rep, M.; Pieterse, C.M. Significance of inducible defense-related proteins in infected plants. Annu. Rev. Phytopathol. 2006, 44, 135–162. [Google Scholar] [CrossRef]
- Lu, S.; Faris, J.D.; Sherwood, R.; Edwards, M.C. Dimerization and protease resistance: New insight into the function of PR-1. J. Plant. Physiol. 2013, 170, 105–110. [Google Scholar] [CrossRef]
- Asensi-Fabado, M.A.; Cela, J.; Müller, M.; Arrom, L.; Chang, C.; Munné-Bosch, S. Enhanced oxidative stress in the ethylene-insensitive (ein3-1) mutant of Arabidopsis thaliana exposed to salt stress. J. Plant. Physiol. 2012, 169, 360–368. [Google Scholar] [CrossRef]
- Shi, Y.; Tian, S.; Hou, L.; Huang, X.; Zhang, X.; Guo, H.; Yang, S. Ethylene signaling negatively regulates freezing tolerance by repressing expression of CBF and type-A ARR genes in Arabidopsis. Plant. Cell 2012, 24, 2578–2595. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Wang, X.; Fu, Y.; Tang, C.; Li, X.; Cheng, Y.; Feng, H.; Huang, L.; Kang, Z. TaEIL1, a wheat homologue of AtEIN3, acts as a negative regulator in the wheat–stripe rust fungus interaction. Mol. Plant. Pathol. 2013, 14, 728–739. [Google Scholar] [CrossRef] [PubMed]
- Bouchez, O.; Huard, C.; Lorrain, S.; Roby, D.; Balagué, C. Ethylene is one of the key elements for cell death and defense response control in the Arabidopsis lesion mimic mutant vad1. Plant. Physiol. 2007, 145, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Lin, J.; Chang, Y.; Jiang, C.-Z. Comparative transcriptomic analysis reveals that ethylene/H2O2-mediated hypersensitive response and programmed cell death determine the compatible interaction of sand pear and Alternaria alternata. Front. Plant. Sci. 2017, 8, 195. [Google Scholar] [CrossRef] [PubMed]
- Halim, V.A.; Altmann, S.; Ellinger, D.; Eschen-Lippold, L.; Miersch, O.; Scheel, D.; Rosahl, S. PAMP-induced defense responses in potato require both salicylic acid and jasmonic acid. Plant. J. 2009, 57, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Manosalva, P.M.; Park, S.-W.; Forouhar, F.; Tong, L.; Fry, W.E.; Klessig, D.F. Methyl esterase 1 (StMES1) is required for systemic acquired resistance in potato. Mol. Plant. Microbe Interact. 2010, 23, 1151–1163. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, S.M.; Odilbekov, F.; Burra, D.D.; Lenman, M.; Hedley, P.E.; Grenville-Briggs, L.; Alexandersson, E.; Liljeroth, E.; Andreasson, E. Intact salicylic acid signalling is required for potato defence against the necrotrophic fungus Alternaria solani. Plant. Mol. Biol. 2020, 104, 1–19. [Google Scholar] [CrossRef] [PubMed]
- McDowell, J.M.; Dangl, J.L. Signal transduction in the plant immune response. Trends Biochem. Sci. 2000, 25, 79–82. [Google Scholar] [CrossRef]
- Nawrath, C.; Heck, S.; Parinthawong, N.; Métraux, J.-P. EDS5, an essential component of salicylic acid–dependent signaling for disease resistance in Arabidopsis, is a member of the MATE transporter family. Plant. Cell 2002, 14, 275–286. [Google Scholar] [CrossRef]
- Vlot, A.C.; Dempsey, D.M.A.; Klessig, D.F. Salicylic acid, a multifaceted hormone to combat disease. Annu. Rev. Phytopathol. 2009, 47, 177–206. [Google Scholar] [CrossRef]
- Sanchez, L.; Courteaux, B.; Hubert, J.; Kauffmann, S.; Renault, J.-H.; Clément, C.; Baillieul, F.; Dorey, S. Rhamnolipids elicit defense responses and induce disease resistance against biotrophic, hemibiotrophic, and necrotrophic pathogens that require different signaling pathways in Arabidopsis and highlight a central role for salicylic acid. Plant. Physiol. 2012, 160, 1630–1641. [Google Scholar] [CrossRef] [PubMed]


| Gene | Primer Sequence (5′-3′): Forward (F), Reverse (R) | Function | Reference |
|---|---|---|---|
| stPR-1 | F: GGGAGAAGCCAAACTACAA R: TGAAATGAACCACCATCC | Marker gene in SA pathway | [51] |
| stLOX | F: CAGATCAGGCCCCGTTAATG R: CCTGTAAGTCCACCTTCACTTGTTG | Marker gene in JA synthesis | [52] |
| stWRKY1 | F: GGTTCTTGGGACTAATGG R: GTTTTTGACAGCCTTTTG | Transcriptional factor in SA pathway | [51] |
| EIN3 | F: CGACTCTGCTGCTACCGATGG R: GGTTCTTTCACTCTCAGGTTGCTC | Transcriptional factor in ET pathway | [53] |
| stRBOHb | F: GCTTCAATCAATCCAACAAGC R: GCGCCAATTTGGTCTAGC | Marker gene related to ROS production | This study |
| stCDPK4 | F: ACCATTCTGGGCTGAAACAC R: CCGGATGAGATCTTTTGCAC | Transcriptional factor in SA pathway; marker gene related to ROS production | This study |
| β-tubulin | F: ATGTTCAGGCGCAAGGCTT R: TCTGCAACCGGGTCATTCAT | Reference gene | [53] |
| actin | F: CATCCTGTCCTCCTAACTGAAGCC R: TCACCAGAGTCCAACACAATACCG | Reference gene | [53] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Najdabbasi, N.; Mirmajlessi, S.M.; Dewitte, K.; Ameye, M.; Mänd, M.; Audenaert, K.; Landschoot, S.; Haesaert, G. Green Leaf Volatile Confers Management of Late Blight Disease: A Green Vaccination in Potato. J. Fungi 2021, 7, 312. https://doi.org/10.3390/jof7040312
Najdabbasi N, Mirmajlessi SM, Dewitte K, Ameye M, Mänd M, Audenaert K, Landschoot S, Haesaert G. Green Leaf Volatile Confers Management of Late Blight Disease: A Green Vaccination in Potato. Journal of Fungi. 2021; 7(4):312. https://doi.org/10.3390/jof7040312
Chicago/Turabian StyleNajdabbasi, Neda, Seyed Mahyar Mirmajlessi, Kevin Dewitte, Maarten Ameye, Marika Mänd, Kris Audenaert, Sofie Landschoot, and Geert Haesaert. 2021. "Green Leaf Volatile Confers Management of Late Blight Disease: A Green Vaccination in Potato" Journal of Fungi 7, no. 4: 312. https://doi.org/10.3390/jof7040312
APA StyleNajdabbasi, N., Mirmajlessi, S. M., Dewitte, K., Ameye, M., Mänd, M., Audenaert, K., Landschoot, S., & Haesaert, G. (2021). Green Leaf Volatile Confers Management of Late Blight Disease: A Green Vaccination in Potato. Journal of Fungi, 7(4), 312. https://doi.org/10.3390/jof7040312







