Complex and Controversial Roles of Eicosanoids in Fungal Pathogenesis
Abstract
1. Introduction
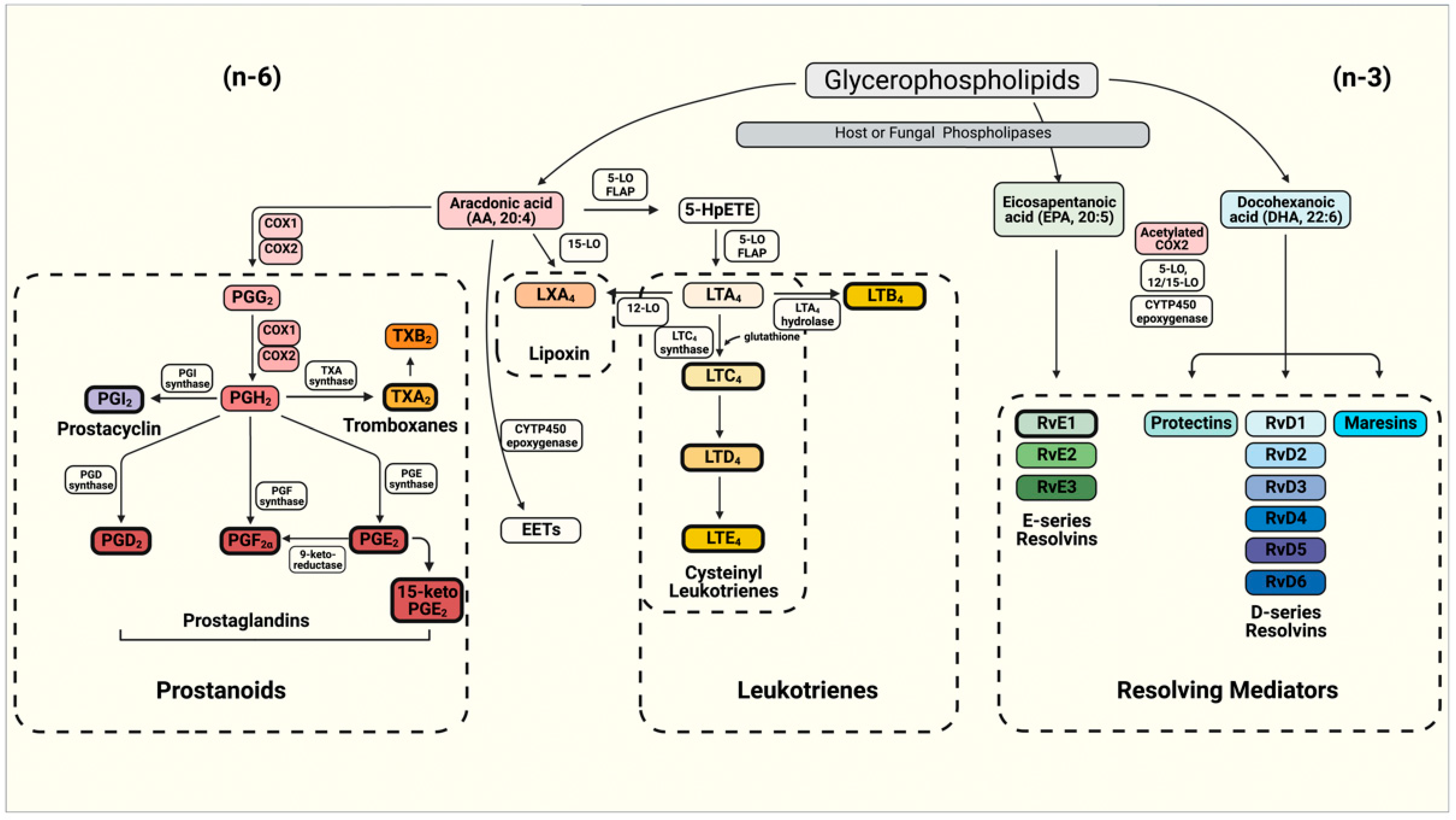
2. Molecular Basis of Eicosanoid Production in Fungi
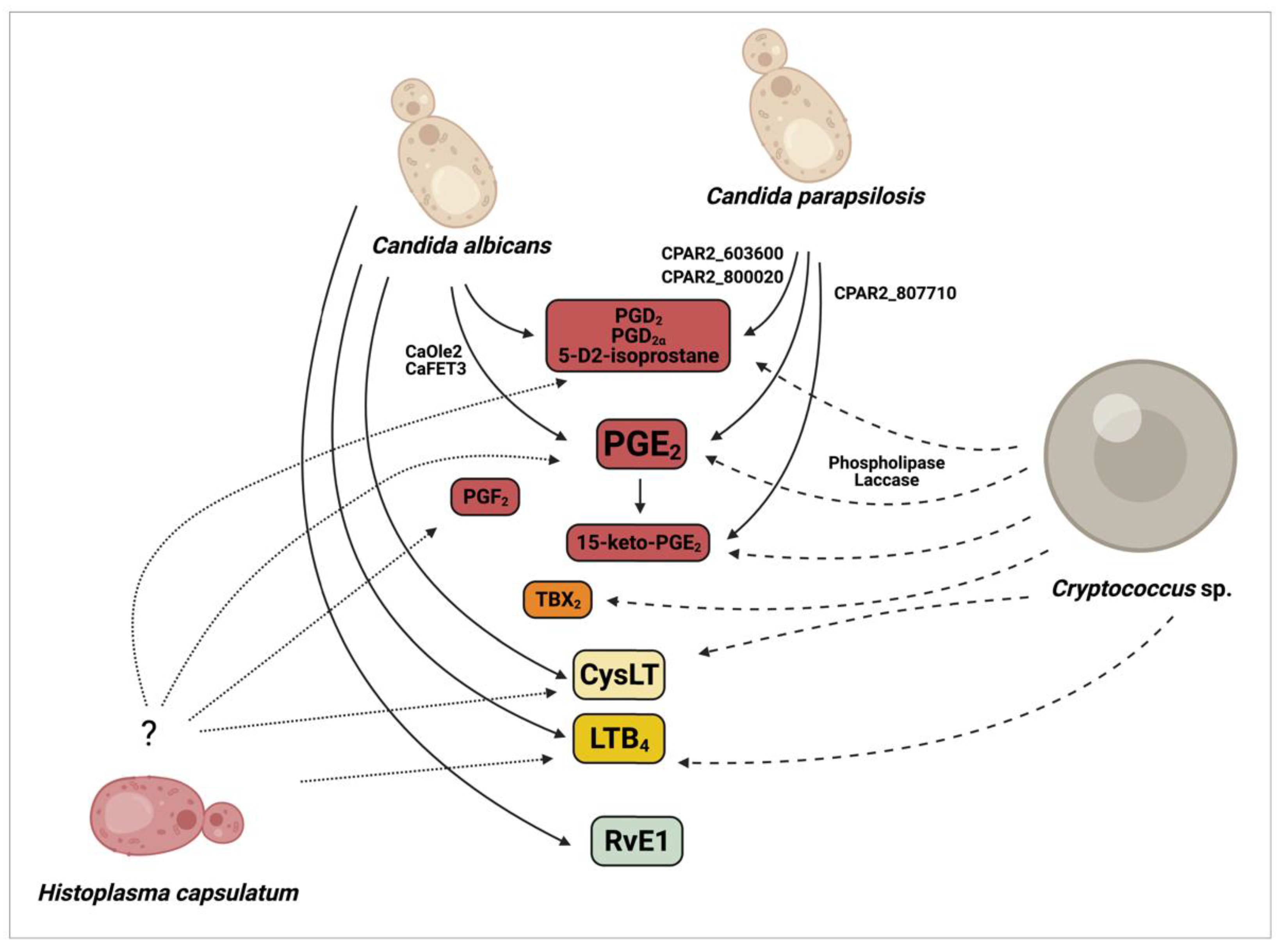
2.1. Production of Eicosanoids by Candida albicans and Non-Albicans Species
2.2. Production of Eicosanoids by Cryptococcus sp.
2.3. Production of Eicosanoids by Histoplasma Capsulatum
3. The Role of Eicosanoids during Fungal Infections
3.1. Eicosanoids in Candidiasis
3.2. Eicosanoids in Cryptococcosis
3.3. Eicosanoids in Histoplasmosis
4. Concluding Remarks
5. Future Trends
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviations
| 15-keto-PGE2 | Dehydrogenated form of Prostaglandin E2 |
| 3-HETE | 3-hydroxy-5,8,11,14- eicosatetraenoic acid |
| 3,18-di-HETE | 3,18-dihydroxy-5,8,11,14- eicosatetraenoic acid |
| 5- HpETE | 5-hydroperoxyeicosatetraenoic acid |
| 5-LO | Enzyme 5-lipoxygenase |
| AA | Arachidonic acid |
| AA (20:4, n-6) | Arachidonic acid (20-carbon, 4 insaturations, omega 6 family) |
| ABC | ATP-binding cassette transporter |
| APCs | Antigen presenting cells |
| BLT1 | Leukotriene B4 high-affinity receptor |
| BLT2 | Leukotriene B4 Low-affinity receptor |
| CaFET3 | Candida albicans multicopper ferroxidase |
| cAMP | Cyclic adenosine monophosphate |
| CaOLE2 | C. albicans fatty acid stearyl-coenzyme A desaturase |
| CNLAC1 | Cryptococcus neoformans laccase gene |
| CNS | Central nervous system |
| COX | Cyclooxygenase |
| CRTH2 | Chemoattractant receptor-homologous molecule expressed on Th2 cells; also known as DP2, PG DP2 receptor |
| CysLT | Cysteinyl leukotriene |
| CysLTR1 | Type 1 cysteinyl leukotriene receptor |
| CYTP450 | Cytochrome P450 oxidase |
| DCs | Dendritic cells |
| DHA | Docosahexaenoic acid |
| DP1 | Prostaglandin D2 receptor 1 |
| EP (1-4) | Rhodopsin-type receptors |
| EPA | Eicosapentaenoic acid |
| ERKs | Extracellular signal-regulate kinases |
| FAs | Fatty acids |
| FLA | 5-lipoxygenase activating protein |
| FP | Prostaglandin F receptor |
| Gal-1 | Galectin-1 |
| GPCRs | G-protein coupled receptors |
| IL | Interleukin |
| IP | Prostacyclin receptor |
| LOX | Lipoxygenase |
| LPL | Lysophospholipase |
| LPTA | Lysophospholipase transacylase |
| LT | Leukotriene |
| LTA4 | Leukotriene A4 |
| LTA4H | LTA4 hydrolase |
| LTB4 | Leukotriene B4 |
| LTD4 | Leukotriene D4 |
| LTE4 | Leukotriene E4 |
| LTF4 | Leukotriene F4 |
| LTs | Leukotrienes |
| n-3 | Omega-3 |
| n-6 | Omega-6 |
| PBMC- DM | Peripheral blood monocyte-derived macrophages |
| PGs | Prostaglandins |
| PGD2 | Prostaglandin D2 |
| PGE2 | Prostaglandin E2 |
| PGEx | Uncharacterized prostaglandin |
| PGF2 | Prostaglandin F2 |
| PGG2 | Prostaglandin G2 |
| PGH2 | Prostaglandin H2 |
| PGI2 | Prostacyclin |
| PI3K | Phosphoinositide 3-kinase |
| PKA | Protein kinase A |
| PLA | Phospholipase A |
| PLB | Phospholipase B |
| PMN | Polymorphonuclear neutrophil |
| PUFA | Polyunsaturated fatty acids |
| Rvs | Resolvins |
| RvD | D-series Resolvins (RvD1-6) |
| RvE1 | E-series Resolvins (RvE1-3) |
| RvT | 13-series resolvins |
| SPM | Specialized pro-resolving mediator |
| Th | T-helper |
| TNF-α | Tumor necrosis factor alpha |
| TP | Thromboxane receptor |
| TX | Thromboxane |
| TXA2 | Thromboxane A2 |
| TXS | Thromboxane synthase |
| Δlac1 | Laccase gene Cryptococcus neoformans mutant |
| Δplb1 | Phospholipase B1 Cryptococcus neoformans mutant |
References
- Kanamori, H.; Rutala, W.A.; Sickbert-Bennett, E.E.; Weber, D.J. Review of Fungal Outbreaks and Infection Prevention in Healthcare Settings during Construction and Renovation. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2015, 61, 433–444. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, M.; Robbins, N.; Wright, G.D. Combinatorial Strategies for Combating Invasive Fungal Infections. Virulence 2017, 8, 169–185. [Google Scholar] [CrossRef] [PubMed]
- Janbon, G.; Quintin, J.; Lanternier, F.; d’Enfert, C. Studying Fungal Pathogens of Humans and Fungal Infections: Fungal Diversity and Diversity of Approaches. Genes Immun. 2019, 20, 403–414. [Google Scholar] [CrossRef]
- Ells, R.; Kock, J.L.; Albertyn, J.; Pohl, C.H. Arachidonic Acid Metabolites in Pathogenic Yeasts. Lipids Health Dis. 2012, 11, 100. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.G.; Padilla, J.; Koumas, L.; Ray, D.; Phipps, R.P. Prostaglandins as Modulators of Immunity. Trends Immunol. 2002, 23, 144–150. [Google Scholar] [CrossRef]
- Ghannoum, M.A. Potential Role of Phospholipases in Virulence and Fungal Pathogenesis. Clin. Microbiol. Rev. 2000, 13, 122–143, table of contents. [Google Scholar] [CrossRef]
- Tilley, S.L.; Coffman, T.M.; Koller, B.H. Mixed Messages: Modulation of Inflammation and Immune Responses by Prostaglandins and Thromboxanes. J. Clin. Investig. 2001, 108, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Christie, W.W.; Harwood, J.L. Oxidation of Polyunsaturated Fatty Acids to Produce Lipid Mediators. Essays Biochem. 2020, 64, 401–421. [Google Scholar] [CrossRef]
- Ghannoum, M.A. Extracellular Phospholipases as Universal Virulence Factor in Pathogenic Fungi. Nihon Ishinkin Gakkai Zasshi Jpn. J. Med. Mycol. 1998, 39, 55–59. [Google Scholar] [CrossRef]
- Lehninger, A.L.; Nelson, D.L.; Cox, M.M. Lehninger Principles of Biochemistry, 4th ed.; W.H. Freeman: New York, NY, USA, 2005; ISBN 978-0-7167-4339-2. [Google Scholar]
- Serhan, C.N. Novel Eicosanoid and Docosanoid Mediators: Resolvins, Docosatrienes, and Neuroprotectins. Curr. Opin. Clin. Nutr. Metab. Care 2005, 8, 115–121. [Google Scholar] [CrossRef]
- Serhan, C.N.; Gotlinger, K.; Hong, S.; Arita, M. Resolvins, Docosatrienes, and Neuroprotectins, Novel Omega-3-Derived Mediators, and Their Aspirin-Triggered Endogenous Epimers: An Overview of Their Protective Roles in Catabasis. Prostagland. Other Lipid Mediat. 2004, 73, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Noverr, M.C.; Toews, G.B.; Huffnagle, G.B. Production of Prostaglandins and Leukotrienes by Pathogenic Fungi. Infect. Immun. 2002, 70, 400–402. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Colón, G.J.; Moore, B.B. Prostaglandin E2 as a Regulator of Immunity to Pathogens. Pharmacol. Ther. 2018, 185, 135–146. [Google Scholar] [CrossRef]
- Coleman, R.A.; Smith, W.L.; Narumiya, S. International Union of Pharmacology Classification of Prostanoid Receptors: Properties, Distribution, and Structure of the Receptors and Their Subtypes. Pharmacol. Rev. 1994, 46, 205–229. [Google Scholar]
- Pereira, P.A.T.; Assis, P.A.; Prado, M.K.B.; Ramos, S.G.; Aronoff, D.M.; de Paula-Silva, F.W.G.; Sorgi, C.A.; Faccioli, L.H. Prostaglandins D2 and E2 Have Opposite Effects on Alveolar Macrophages Infected with Histoplasma Capsulatum. J. Lipid Res. 2018, 59, 195–206. [Google Scholar] [CrossRef]
- Hardwick, J.P.; Eckman, K.; Lee, Y.K.; Abdelmegeed, M.A.; Esterle, A.; Chilian, W.M.; Chiang, J.Y.; Song, B.-J. Eicosanoids in Metabolic Syndrome. Adv. Pharmacol. 2013, 66, 157–266. [Google Scholar] [CrossRef]
- Jones, R.L. Prostanoid Receptors. In xPharm: The Comprehensive Pharmacology Reference; Elsevier: Amsterdam, The Netherlands, 2007; pp. 1–3. ISBN 978-0-08-055232-3. [Google Scholar]
- Sun, L.; Ye, R.D. Role of G Protein-Coupled Receptors in Inflammation. Acta Pharmacol. Sin. 2012, 33, 342–350. [Google Scholar] [CrossRef]
- Sugimoto, Y.; Narumiya, S. Prostaglandin E Receptors. J. Biol. Chem. 2007, 282, 11613–11617. [Google Scholar] [CrossRef]
- Narumiya, S.; Sugimoto, Y.; Ushikubi, F. Prostanoid Receptors: Structures, Properties, and Functions. Physiol. Rev. 1999, 79, 1193–1226. [Google Scholar] [CrossRef] [PubMed]
- Woodward, D.F.; Jones, R.L.; Narumiya, S. International Union of Basic and Clinical Pharmacology. LXXXIII: Classification of Prostanoid Receptors, Updating 15 Years of Progress. Pharmacol. Rev. 2011, 63, 471–538. [Google Scholar] [CrossRef] [PubMed]
- Tsuge, K.; Inazumi, T.; Shimamoto, A.; Sugimoto, Y. Molecular Mechanisms Underlying Prostaglandin E2-Exacerbated Inflammation and Immune Diseases. Int. Immunol. 2019, 31, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Ji, Z.; Tsalkova, T.; Mei, F. Epac and PKA: A Tale of Two Intracellular CAMP Receptors. Acta Biochim. Biophys. Sin. 2008, 40, 651–662. [Google Scholar] [CrossRef] [PubMed]
- Regan, J.W. EP2 and EP4 Prostanoid Receptor Signaling. Life Sci. 2003, 74, 143–153. [Google Scholar] [CrossRef]
- Breyer, R.M.; Bagdassarian, C.K.; Myers, S.A.; Breyer, M.D. Prostanoid Receptors: Subtypes and Signaling. Annu. Rev. Pharmacol. Toxicol. 2001, 41, 661–690. [Google Scholar] [CrossRef]
- Kobayashi, K.; Horikami, D.; Omori, K.; Nakamura, T.; Yamazaki, A.; Maeda, S.; Murata, T. Thromboxane A2 Exacerbates Acute Lung Injury via Promoting Edema Formation. Sci. Rep. 2016, 6, 32109. [Google Scholar] [CrossRef]
- Altieri, D.C.; Mannucci, P.M. Thromboxane Generation by Human Monocytes Enhances Platelet Function. J. Exp. Med. 1986, 164, 1815–1820. [Google Scholar] [CrossRef]
- Ashton, A.W.; Mukherjee, S.; Nagajyothi, F.N.U.; Huang, H.; Braunstein, V.L.; Desruisseaux, M.S.; Factor, S.M.; Lopez, L.; Berman, J.W.; Wittner, M.; et al. Thromboxane A2 Is a Key Regulator of Pathogenesis during Trypanosoma Cruzi Infection. J. Exp. Med. 2007, 204, 929–940. [Google Scholar] [CrossRef]
- Yang, C.-W.; Unanue, E.R. Neutrophils Control the Magnitude and Spread of the Immune Response in a Thromboxane A2-Mediated Process. J. Exp. Med. 2013, 210, 375–387. [Google Scholar] [CrossRef]
- Peters-Golden, M.; Canetti, C.; Mancuso, P.; Coffey, M.J. Leukotrienes: Underappreciated Mediators of Innate Immune Responses. J. Immunol. 2005, 174, 589–594. [Google Scholar] [CrossRef]
- Morato-Marques, M.; Campos, M.R.; Kane, S.; Rangel, A.P.; Lewis, C.; Ballinger, M.N.; Kim, S.-H.; Peters-Golden, M.; Jancar, S.; Serezani, C.H. Leukotrienes Target F-Actin/Cofilin-1 to Enhance Alveolar Macrophage Anti-Fungal Activity. J. Biol. Chem. 2011, 286, 28902–28913. [Google Scholar] [CrossRef] [PubMed]
- Tager, A.M.; Luster, A.D. BLT1 and BLT2: The Leukotriene B(4) Receptors. Prostagland. Leukot. Essent. Fatty Acids 2003, 69, 123–134. [Google Scholar] [CrossRef]
- Kanaoka, Y.; Boyce, J.A. Cysteinyl Leukotrienes and Their Receptors; Emerging Concepts. Allergy Asthma Immunol. Res. 2014, 6, 288–295. [Google Scholar] [CrossRef]
- Melo, C.F.O.R.; Bachur, L.F.; Delafiori, J.; Dabaja, M.Z.; de Oliveira, D.N.; Guerreiro, T.M.; Tararam, C.A.; Busso-Lopes, A.F.; Moretti, M.L.; Catharino, R.R. Does Leukotriene F4 Play a Major Role in the Infection Mechanism of Candida Sp.? Microb. Pathog. 2020, 149, 104394. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.H.P.; Canetti, C.; Ribeiro, R.A.; Cunha, F.Q. Neutrophil Migration Induced by IL-1beta Depends upon LTB4 Released by Macrophages and upon TNF-Alpha and IL-1beta Released by Mast Cells. Inflammation 2008, 31, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Olynych, T.J.; Jakeman, D.L.; Marshall, J.S. Fungal Zymosan Induces Leukotriene Production by Human Mast Cells through a Dectin-1-Dependent Mechanism. J. Allergy Clin. Immunol. 2006, 118, 837–843. [Google Scholar] [CrossRef]
- König, S.; Pace, S.; Pein, H.; Heinekamp, T.; Kramer, J.; Romp, E.; Straßburger, M.; Troisi, F.; Proschak, A.; Dworschak, J.; et al. Gliotoxin from Aspergillus Fumigatus Abrogates Leukotriene B4 Formation through Inhibition of Leukotriene A4 Hydrolase. Cell Chem. Biol. 2019, 26, 524–534.e5. [Google Scholar] [CrossRef]
- Lee, E.K.S.; Gillrie, M.R.; Li, L.; Arnason, J.W.; Kim, J.H.; Babes, L.; Lou, Y.; Sanati-Nezhad, A.; Kyei, S.K.; Kelly, M.M.; et al. Leukotriene B4-Mediated Neutrophil Recruitment Causes Pulmonary Capillaritis during Lethal Fungal Sepsis. Cell Host Microbe 2018, 23, 121–133.e4. [Google Scholar] [CrossRef]
- Caffrey-Carr, A.K.; Hilmer, K.M.; Kowalski, C.H.; Shepardson, K.M.; Temple, R.M.; Cramer, R.A.; Obar, J.J. Host-Derived Leukotriene B4 Is Critical for Resistance against Invasive Pulmonary Aspergillosis. Front. Immunol. 2017, 8, 1984. [Google Scholar] [CrossRef]
- Spite, M. Deciphering the Role of N-3 Polyunsaturated Fatty Acid-Derived Lipid Mediators in Health and Disease. Proc. Nutr. Soc. 2013, 72, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Haas-Stapleton, E.J.; Lu, Y.; Hong, S.; Arita, M.; Favoreto, S.; Nigam, S.; Serhan, C.N.; Agabian, N. Candida Albicans Modulates Host Defense by Biosynthesizing the Pro-Resolving Mediator Resolvin E1. PLoS ONE 2007, 2, e0001316. [Google Scholar] [CrossRef] [PubMed]
- Schwab, J.M.; Chiang, N.; Arita, M.; Serhan, C.N. Resolvin E1 and Protectin D1 Activate Inflammation-Resolution Programmes. Nature 2007, 447, 869–874. [Google Scholar] [CrossRef]
- Serhan, C.N. Resolution Phase of Inflammation: Novel Endogenous Anti-Inflammatory and Proresolving Lipid Mediators and Pathways. Annu. Rev. Immunol. 2007, 25, 101–137. [Google Scholar] [CrossRef]
- Sommer, C.; Birklein, F. Resolvins and Inflammatory Pain. F1000 Med. Rep. 2011, 3, 19. [Google Scholar] [CrossRef] [PubMed]
- Buczynski, M.W.; Dumlao, D.S.; Dennis, E.A. Thematic Review Series: Proteomics. An Integrated Omics Analysis of Eicosanoid Biology. J. Lipid Res. 2009, 50, 1015–1038. [Google Scholar] [CrossRef] [PubMed]
- van Dyk, M.S.; Kock, J.L.; Coetzee, D.J.; Augustyn, O.P.; Nigam, S. Isolation of a Novel Arachidonic Acid Metabolite 3-Hydroxy-5,8,11,14-Eicosatetraenoic Acid (3-HETE) from the Yeast Dipodascopsis Uninucleata UOFs-Y128. FEBS Lett. 1991, 283, 195–198. [Google Scholar] [CrossRef]
- Strauss, T.; Botha, A.; Kock, J.L.; Paul, I.; Smith, D.P.; Linke, D.; Schewe, T.; Nigam, S. Mapping the Distribution of 3-Hydroxylipins in the Mucorales Using Immunofluorescence Microscopy. Antonie Van Leeuwenhoek 2000, 78, 39–42. [Google Scholar] [CrossRef]
- Botha, A.; Kock, J.L.F.; Coetzee, D.J.; Van Dyk, M.S.; Van Der Berg, L.; Botes, P.J. Yeast Eicosanoids I. The Distribution and Taxonomic Value of Cellular Fatty Acids and Arachidonic Acid Metabolites in the Dipodascaceae and Related Taxa. Syst. Appl. Microbiol. 1992, 15, 148–154. [Google Scholar] [CrossRef]
- Noverr, M.C.; Phare, S.M.; Toews, G.B.; Coffey, M.J.; Huffnagle, G.B. Pathogenic Yeasts Cryptococcus Neoformans and Candida Albicans Produce Immunomodulatory Prostaglandins. Infect. Immun. 2001, 69, 2957–2963. [Google Scholar] [CrossRef]
- Erb-Downward, J.R.; Noggle, R.M.; Williamson, P.R.; Huffnagle, G.B. The Role of Laccase in Prostaglandin Production by Cryptococcus Neoformans. Mol. Microbiol. 2008, 68, 1428–1437. [Google Scholar] [CrossRef]
- Erb-Downward, J.R.; Noverr, M.C. Characterization of Prostaglandin E2 Production by Candida Albicans. Infect. Immun. 2007, 75, 3498–3505. [Google Scholar] [CrossRef]
- Deva, R.; Ciccoli, R.; Kock, L.; Nigam, S. Involvement of Aspirin-Sensitive Oxylipins in Vulvovaginal Candidiasis. FEMS Microbiol. Lett. 2001, 198, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Noverr, M.C.; Erb-Downward, J.R.; Huffnagle, G.B. Production of Eicosanoids and Other Oxylipins by Pathogenic Eukaryotic Microbes. Clin. Microbiol. Rev. 2003, 16, 517–533. [Google Scholar] [CrossRef]
- Alem, M.A.; Douglas, L.J. Prostaglandin Production during Growth of Candida Albicans Biofilms. J. Med. Microbiol. 2005, 54, 1001–1005. [Google Scholar] [CrossRef]
- Grózer, Z.; Tóth, A.; Tóth, R.; Kecskeméti, A.; Vágvölgyi, C.; Nosanchuk, J.D.; Szekeres, A.; Gácser, A. Candida Parapsilosis Produces Prostaglandins from Exogenous Arachidonic Acid and OLE2 Is Not Required for Their Synthesis. Virulence 2015, 6, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, T.; Thuer, E.; Heijink, M.; Tóth, R.; Bodai, L.; Vágvölgyi, C.; Giera, M.; Gabaldón, T.; Gácser, A. Eicosanoid Biosynthesis Influences the Virulence of Candida Parapsilosis. Virulence 2018, 9, 1019–1035. [Google Scholar] [CrossRef]
- Mishra, N.N.; Ali, S.; Shukla, P.K. Arachidonic Acid Affects Biofilm Formation and PGE2 Level in Candida Albicans and Non-Albicans Species in Presence of Subinhibitory Concentration of Fluconazole and Terbinafine. Braz. J. Infect. Dis. Off. Publ. Braz. Soc. Infect. Dis. 2014, 18, 287–293. [Google Scholar] [CrossRef]
- Arita, M.; Bianchini, F.; Aliberti, J.; Sher, A.; Chiang, N.; Hong, S.; Yang, R.; Petasis, N.A.; Serhan, C.N. Stereochemical Assignment, Antiinflammatory Properties, and Receptor for the Omega-3 Lipid Mediator Resolvin E1. J. Exp. Med. 2005, 201, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.J.; Pline, K.; Loynes, C.A.; Needs, S.; Aldrovandi, M.; Tiefenbach, J.; Bielska, E.; Rubino, R.E.; Nicol, C.J.; May, R.C.; et al. 15-Keto-Prostaglandin E2 Activates Host Peroxisome Proliferator-Activated Receptor Gamma (PPAR-γ) to Promote Cryptococcus Neoformans Growth during Infection. PLoS Pathog. 2019, 15, e1007597. [Google Scholar] [CrossRef] [PubMed]
- Ganendren, R.; Widmer, F.; Singhal, V.; Wilson, C.; Sorrell, T.; Wright, L. In Vitro Antifungal Activities of Inhibitors of Phospholipases from the Fungal Pathogen Cryptococcus Neoformans. Antimicrob. Agents Chemother. 2004, 48, 1561–1569. [Google Scholar] [CrossRef] [PubMed]
- Panepinto, J.C.; Williamson, P.R. Intersection of Fungal Fitness and Virulence in Cryptococcus Neoformans. FEMS Yeast Res. 2006, 6, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Williamson, P.R. Role of Laccase in the Biology and Virulence of Cryptococcus Neoformans. FEMS Yeast Res. 2004, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Valdez, P.A.; Vithayathil, P.J.; Janelsins, B.M.; Shaffer, A.L.; Williamson, P.R.; Datta, S.K. Prostaglandin E2 Suppresses Antifungal Immunity by Inhibiting Interferon Regulatory Factor 4 Function and Interleukin-17 Expression in T Cells. Immunity 2012, 36, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.G.; Lim, Y.S.; Tan, A.; Leong, R.; Pavelka, N. Fungal Symbionts Produce Prostaglandin E2 to Promote Their Intestinal Colonization. Front. Cell. Infect. Microbiol. 2019, 9, 359. [Google Scholar] [CrossRef] [PubMed]
- Filler, S.G.; Ibe, B.O.; Luckett, P.M.; Raj, J.U.; Edwards, J.E. Candida Albicans Stimulates Endothelial Cell Eicosanoid Production. J. Infect. Dis. 1991, 164, 928–935. [Google Scholar] [CrossRef]
- Ma, H.; Wan, S.; Xia, C.-Q. Immunosuppressive CD11b+Ly6Chi Monocytes in Pristane-Induced Lupus Mouse Model. J. Leukoc. Biol. 2016, 99, 1121–1129. [Google Scholar] [CrossRef]
- Yao, C.; Sakata, D.; Esaki, Y.; Li, Y.; Matsuoka, T.; Kuroiwa, K.; Sugimoto, Y.; Narumiya, S. Prostaglandin E2-EP4 Signaling Promotes Immune Inflammation through Th1 Cell Differentiation and Th17 Cell Expansion. Nat. Med. 2009, 15, 633–640. [Google Scholar] [CrossRef]
- Chizzolini, C.; Chicheportiche, R.; Alvarez, M.; de Rham, C.; Roux-Lombard, P.; Ferrari-Lacraz, S.; Dayer, J.-M. Prostaglandin E2 Synergistically with Interleukin-23 Favors Human Th17 Expansion. Blood 2008, 112, 3696–3703. [Google Scholar] [CrossRef]
- Castro, M.; Ralston, N.V.; Morgenthaler, T.I.; Rohrbach, M.S.; Limper, A.H. Candida Albicans Stimulates Arachidonic Acid Liberation from Alveolar Macrophages through Alpha-Mannan and Beta-Glucan Cell Wall Components. Infect. Immun. 1994, 62, 3138–3145. [Google Scholar] [CrossRef] [PubMed]
- Smeekens, S.P.; van de Veerdonk, F.L.; van der Meer, J.W.M.; Kullberg, B.J.; Joosten, L.A.B.; Netea, M.G. The Candida Th17 Response Is Dependent on Mannan- and -Glucan-Induced Prostaglandin E2. Int. Immunol. 2010, 22, 889–895. [Google Scholar] [CrossRef]
- Kalo-Klein, A.; Witkin, S.S. Prostaglandin E2 Enhances and Gamma Interferon Inhibits Germ Tube Formation in Candida Albicans. Infect. Immun. 1990, 58, 260–262. [Google Scholar] [CrossRef]
- Kim, Y.-G.; Udayanga, K.G.S.; Totsuka, N.; Weinberg, J.B.; Núñez, G.; Shibuya, A. Gut Dysbiosis Promotes M2 Macrophage Polarization and Allergic Airway Inflammation via Fungi-Induced PGE2. Cell Host Microbe 2014, 15, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Bernström, K.; Hammarström, S. A Novel Leukotriene Formed by Transpeptidation of Leukotriene E. Biochem. Biophys. Res. Commun. 1982, 109, 800–804. [Google Scholar] [CrossRef]
- Singh, R.K.; Gupta, S.; Dastidar, S.; Ray, A. Cysteinyl Leukotrienes and Their Receptors: Molecular and Functional Characteristics. Pharmacology 2010, 85, 336–349. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Shi, M. Neutrophil Swarming toward Cryptococcus Neoformans Is Mediated by Complement and Leukotriene B4. Biochem. Biophys. Res. Commun. 2016, 477, 945–951. [Google Scholar] [CrossRef]
- Zhu, L.; Maruvada, R.; Sapirstein, A.; Peters-Golden, M.; Kim, K.S. Cysteinyl Leukotrienes as Novel Host Factors Facilitating Cryptococcus Neoformans Penetration into the Brain. Cell. Microbiol. 2017, 19, e12661. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Liu, Y. Prostaglandin E2 Blockade Enhances the Pulmonary Anti-Cryptococcus Neoformans Immune Reaction via the Induction of TLR-4. Int. Immunopharmacol. 2015, 28, 376–381. [Google Scholar] [CrossRef]
- Wolf, J.E.; Massof, S.E.; Peters, S.P. Alterations in Murine Macrophage Arachidonic Acid Metabolism Following Ingestion of Nonviable Histoplasma Capsulatum. Infect. Immun. 1992, 60, 2559–2564. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.A.T.; Trindade, B.C.; Secatto, A.; Nicolete, R.; Peres-Buzalaf, C.; Ramos, S.G.; Sadikot, R.; Bitencourt, C.D.S.; Faccioli, L.H. Celecoxib Improves Host Defense through Prostaglandin Inhibition during Histoplasma Capsulatum Infection. Mediat. Inflamm. 2013, 2013, 950981. [Google Scholar] [CrossRef]
- Medeiros, A.I.; Sá-Nunes, A.; Soares, E.G.; Peres, C.M.; Silva, C.L.; Faccioli, L.H. Blockade of Endogenous Leukotrienes Exacerbates Pulmonary Histoplasmosis. Infect. Immun. 2004, 72, 1637–1644. [Google Scholar] [CrossRef]
- Secatto, A.; Rodrigues, L.C.; Serezani, C.H.; Ramos, S.G.; Dias-Baruffi, M.; Faccioli, L.H.; Medeiros, A.I. 5-Lipoxygenase Deficiency Impairs Innate and Adaptive Immune Responses during Fungal Infection. PLoS ONE 2012, 7, e31701. [Google Scholar] [CrossRef]
- Nicolete, R.; Secatto, A.; Pereira, P.A.T.; Soares, E.G.; Faccioli, L.H. Leukotriene B4-Loaded Microspheres as a New Approach to Enhance Antimicrobial Responses in Histoplasma Capsulatum-Infected Mice. Int. J. Antimicrob. Agents 2009, 34, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Metzemaekers, M.; Gouwy, M.; Proost, P. Neutrophil Chemoattractant Receptors in Health and Disease: Double-Edged Swords. Cell. Mol. Immunol. 2020, 17, 433–450. [Google Scholar] [CrossRef] [PubMed]
- Samuchiwal, S.K.; Boyce, J.A. Role of Lipid Mediators and Control of Lymphocyte Responses in Type 2 Immunopathology. J. Allergy Clin. Immunol. 2018, 141, 1182–1190. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, A.I.; Sá-Nunes, A.; Turato, W.M.; Secatto, A.; Frantz, F.G.; Sorgi, C.A.; Serezani, C.H.; Deepe, G.S.; Faccioli, L.H. Leukotrienes Are Potent Adjuvant during Fungal Infection: Effects on Memory T Cells. J. Immunol. 2008, 181, 8544–8551. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.C.; Secatto, A.; Sorgi, C.A.; Dejani, N.N.; Medeiros, A.I.; Prado, M.K.B.; Ramos, S.G.; Cummings, R.D.; Stowell, S.R.; Faccioli, L.H.; et al. Protective Effect of Galectin-1 during Histoplasma Capsulatum Infection Is Associated with Prostaglandin E2 and Nitric Oxide Modulation. Mediat. Inflamm. 2016, 2016, 5813794. [Google Scholar] [CrossRef]
- Bordon, A.P.; Dias-Melicio, L.A.; Acorci, M.J.; Calvi, S.A.; Serrão Peraçoli, M.T.; Victoriano de Campos Soares, A.M. Prostaglandin E2 Inhibits Paracoccidioides Brasiliensis Killing by Human Monocytes. Microbes Infect. 2007, 9, 744–747. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendoza, S.R.; Zamith-Miranda, D.; Takács, T.; Gacser, A.; Nosanchuk, J.D.; Guimarães, A.J. Complex and Controversial Roles of Eicosanoids in Fungal Pathogenesis. J. Fungi 2021, 7, 254. https://doi.org/10.3390/jof7040254
Mendoza SR, Zamith-Miranda D, Takács T, Gacser A, Nosanchuk JD, Guimarães AJ. Complex and Controversial Roles of Eicosanoids in Fungal Pathogenesis. Journal of Fungi. 2021; 7(4):254. https://doi.org/10.3390/jof7040254
Chicago/Turabian StyleMendoza, Susana Ruiz, Daniel Zamith-Miranda, Tamás Takács, Attila Gacser, Joshua D. Nosanchuk, and Allan J. Guimarães. 2021. "Complex and Controversial Roles of Eicosanoids in Fungal Pathogenesis" Journal of Fungi 7, no. 4: 254. https://doi.org/10.3390/jof7040254
APA StyleMendoza, S. R., Zamith-Miranda, D., Takács, T., Gacser, A., Nosanchuk, J. D., & Guimarães, A. J. (2021). Complex and Controversial Roles of Eicosanoids in Fungal Pathogenesis. Journal of Fungi, 7(4), 254. https://doi.org/10.3390/jof7040254







