Effects of Medicinal Fungi-Derived β-Glucan on Tumor Progression
Abstract
1. Introduction
2. β-Glucans Isolated from Different Sources
2.1. Differences in β-Glucan Based on Its Sources
2.2. Mushroom β-Glucans
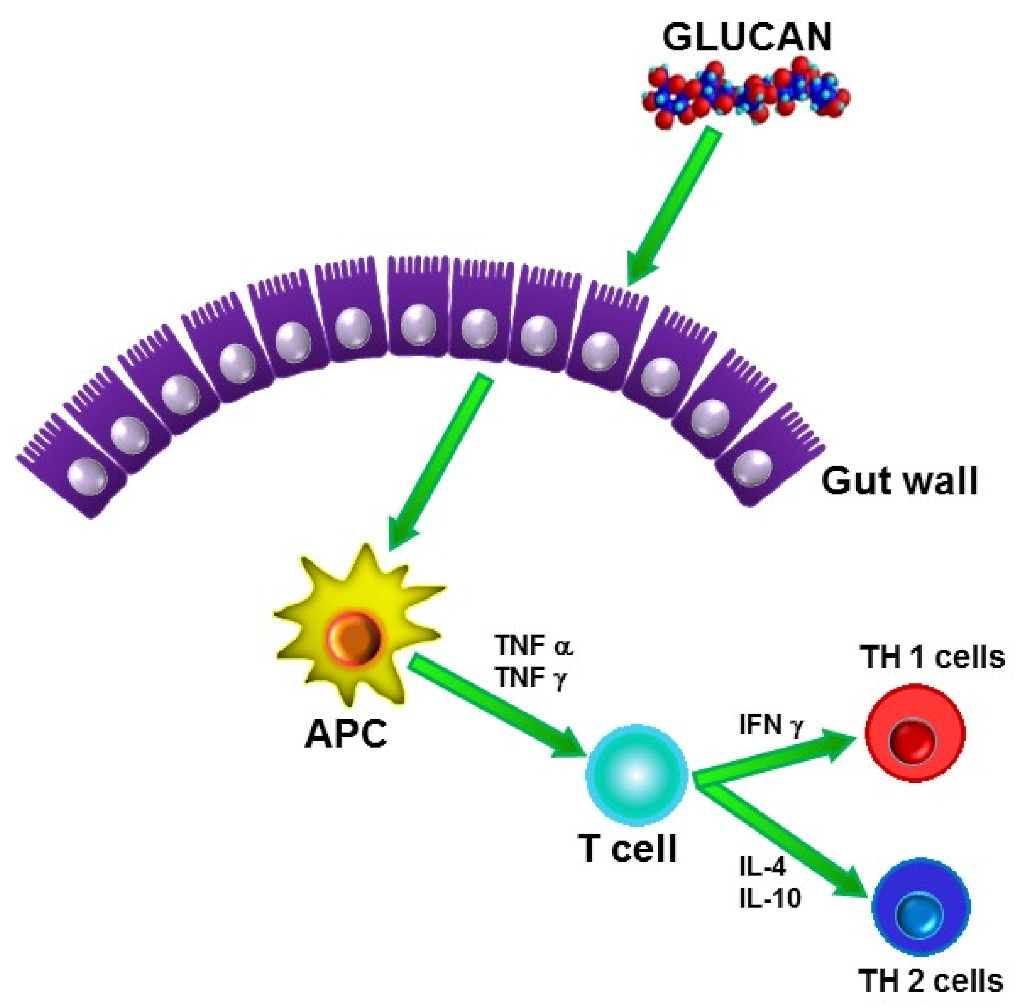
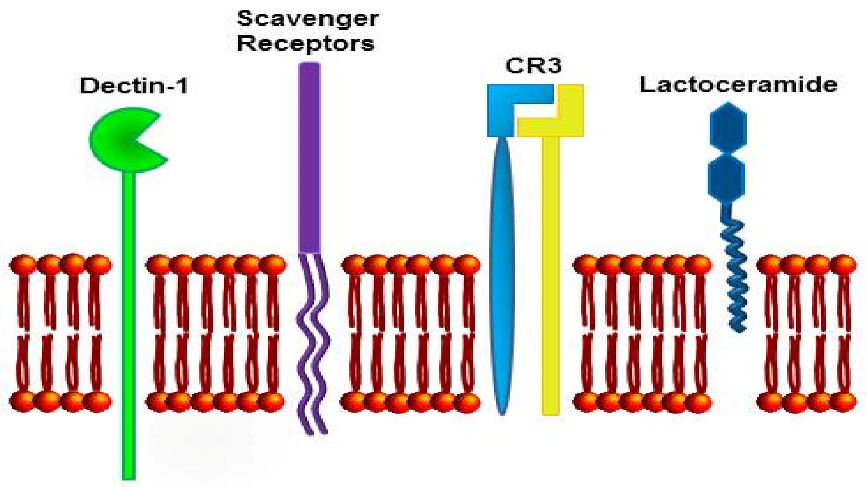
3. Mechanisms of Action
4. The Antitumor Effect of β-Glucan in Humans
5. Tumor-Associated Macrophages
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lind, J. A Treatise of the Scurvy: In Three Parts, Containing an Inquiry into the Nature, Causes, and Cure, of That Disease, Together with a Critical and Chronological View of What Has Been Published on the Subject; Sands, Murray, and Cochran: Edinburgh, UK, 1753. [Google Scholar]
- Jayachandran, M.; Chen, J.; Chung, S.S.M.; Xu, B. A critical review on the impacts of beta-glucans on gut microbiota and human health. J. Nutr. Biochem. 2018, 61, 101–110. [Google Scholar] [CrossRef]
- Bose, N.; Chan, A.S.; Guerrero, F.; Maristany, C.M.; Qiu, X.; Walsh, R.M.; Ertelt, K.E.; Jonas, A.B.; Gorden, K.B.; Dudney, C.M.; et al. Binding of soluble yeast beta-glucan to human neutrophils and monocytes is complement-dependent. Front. Immunol. 2013, 4, 230. [Google Scholar] [CrossRef] [PubMed]
- Bose, N.; Wurst, L.R.; Chan, A.S.; Dudney, C.M.; LeRoux, M.L.; Danielson, M.E.; Will, P.M.; Nodland, S.E.; Patchen, M.L.; Dalle Lucca, J.J.; et al. Differential regulation of oxidative burst by distinct beta-glucan-binding receptors and signaling pathways in human peripheral blood mononuclear cells. Glycobiology 2014, 24, 379–391. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vetvickova, J. B-glucan improves condiiton of chronic fatigue in mice by stimulation of immunity. Open Biochem. J. 2020, 14, 1–8. [Google Scholar] [CrossRef]
- Wang, W.J.; Wu, Y.S.; Chen, S.; Liu, C.F.; Chen, S.N. Mushroom beta-glucan may immunomodulate the tumor-associated macrophages in the Lewis lung carcinoma. BioMed Res. Int. 2015, 2015, 604385. [Google Scholar] [CrossRef]
- Yoon, T.J.; Koppula, S.; Lee, K.H. The effects of beta-glucans on cancer metastasis. Anticancer Agents Med. Chem. 2013, 13, 699–708. [Google Scholar] [CrossRef]
- Vetvicka, V. [Beta]-glucans as Natural Biological Response Modifiers; Nova Science Publishers, Inc.: New York, NY, USA, 2013. [Google Scholar]
- Bohn, J.A.; BeMiller, J.N. (1→3)-β-d-Glucans as biological response modifiers: A review of structure-functional activity relationships. Carbohydr. Polym. 1995, 28, 3–14. [Google Scholar] [CrossRef]
- Leung, P.H.; Zhang, Q.X.; Wu, J.Y. Mycelium cultivation, chemical composition and antitumour activity of a Tolypocladium sp. fungus isolated from wild Cordyceps sinensis. J. Appl. Microbiol. 2006, 101, 275–283. [Google Scholar] [CrossRef]
- Zhang, L.; Li, X.; Xu, X.; Zeng, F. Correlation between antitumor activity, molecular weight, and conformation of lentinan. Carbohydr. Res. 2005, 340, 1515–1521. [Google Scholar] [CrossRef]
- Yan, J.; Vetvicka, V.; Xia, Y.; Coxon, A.; Carroll, M.C.; Mayadas, T.N.; Ross, G.D. Beta-glucan, a “specific” biologic response modifier that uses antibodies to target tumors for cytotoxic recognition by leukocyte complement receptor type 3 (CD11b/CD18). J. Immunol. 1999, 163, 3045–3052. [Google Scholar]
- Novak, M.; Vetvicka, V. Glucans as biological response modifiers. Endocr. Metab. Immune Disord. Drug Targets 2009, 9, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Majtan, J.; Jesenak, M. beta-glucans: Multi-functional modulator of wound healing. Molecules 2018, 23, 806. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vetvickova, J. Combination therapy with glucan and coenzyme Q10 in murine experimental autoimmune disease and cancer. Anticancer Res. 2018, 38, 3291–3297. [Google Scholar] [CrossRef] [PubMed]
- Bashir, K.M.I.; Choi, J.S. Clinical and physiological perspectives of beta-glucans: The past, present, and future. Int. J. Mol. Sci. 2017, 18, 1906. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vetvickova, J. Glucans and cancer: Comparison of commercially available beta-glucans—Part IV. Anticancer Res. 2018, 38, 1327–1333. [Google Scholar] [CrossRef]
- Boulaka, A.; Christodoulou, P.; Vlassopoulou, M.; Koutrotsios, G.; Bekiaris, G.; Zervakis, G.I.; Mitsou, E.K.; Saxami, G.; Kyriacou, A.; Zervou, M.; et al. Genoprotective properties and metabolites of beta-glucan-rich edible mushrooms following their in vitro fermentation by human faecal microbiota. Molecules 2020, 25, 3554. [Google Scholar] [CrossRef]
- Muthusamy, G.; Joardar, S.N.; Samanta, I.; Isore, D.P.; Roy, B.; Maiti, T.K. Dietary administered purified β-glucan of edible mushroom (Pleurotus florida) provides immunostimulation and protection in broiler experimentally challenged with virulent Newcastle disease virus. J. Basic Appl. Zool. 2020, 81, 55. [Google Scholar] [CrossRef]
- Jesenak, M.; Banovcin, P.; Rennerova, Z.; Majtan, J. beta-Glucans in the treatment and prevention of allergic diseases. Allergol. Immunopathol. 2014, 42, 149–156. [Google Scholar] [CrossRef]
- Kirmaz, C.; Bayrak, P.; Yilmaz, O.; Yuksel, H. Effects of glucan treatment on the Th1/Th2 balance in patients with allergic rhinitis: A double-blind placebo-controlled study. Eur. Cytokine Netw. 2005, 16, 128–134. [Google Scholar]
- Vannucci, L.; Krizan, J.; Sima, P.; Stakheev, D.; Caja, F.; Rajsiglova, L.; Horak, V.; Saieh, M. Immunostimulatory properties and antitumor activities of glucans (Review). Int. J. Oncol. 2013, 43, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Beschin, A.; Bilej, M.; Hanssens, F.; Raymakers, J.; Van Dyck, E.; Revets, H.; Brys, L.; Gomez, J.; De Baetselier, P.; Timmermans, M. Identification and cloning of a glucan- and lipopolysaccharide-binding protein from Eisenia foetida earthworm involved in the activation of prophenoloxidase cascade. J. Biol. Chem. 1998, 273, 24948–24954. [Google Scholar] [CrossRef] [PubMed]
- Mazzei, M.; Fronte, B.; Sagona, S.; Carrozza, M.L.; Forzan, M.; Pizzurro, F.; Bibbiani, C.; Miragliotta, V.; Abramo, F.; Millanta, F.; et al. Effect of 1,3-1,6 beta-glucan on natural and experimental deformed wing virus infection in newly emerged honeybees (Apis mellifera ligustica). PLoS ONE 2016, 11, e0166297. [Google Scholar] [CrossRef] [PubMed]
- Duvic, B.; Soderhall, K. Purification and characterization of a beta-1,3-glucan binding protein from plasma of the crayfish Pacifastacus leniusculus. J. Biol. Chem. 1990, 265, 9327–9332. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vannucci, L.; Sima, P. The effects of beta-glucan on fish immunity. N. Am. J. Med. Sci. 2013, 5, 580–588. [Google Scholar] [CrossRef]
- Jacob, J.P.; Pescatore, A.J. Barley beta-glucan in poultry diets. Ann. Transl. Med. 2014, 2, 20. [Google Scholar] [CrossRef]
- Feletti, F.; De Bernardi di Valserra, M.; Contos, S.; Mattaboni, P.; Germogli, R. Chronic toxicity study on a new glucan extracted from Candida albicans in rats. Arzneimittelforschung 1992, 42, 1363–1367. [Google Scholar]
- Ferencik, M.; Kotulova, D.; Masler, L.; Bergendi, L.; Sandula, J.; Stefanovic, J. Modulatory effect of glucans on the functional and biochemical activities of guinea-pig macrophages. Methods Find. Exp. Clin. Pharmacol. 1986, 8, 163–166. [Google Scholar] [PubMed]
- Benkova, M.; Boroskova, Z.; Soltys, J. [Immunostimulatory effects of certain substances in experimental ascaridiasis in pigs]. Vet. Med. 1992, 36, 717–724. [Google Scholar]
- Buddle, B.M.; Pulford, H.D.; Ralston, M. Protective effect of glucan against experimentally induced staphylococcal mastitis in ewes. Vet. Microbiol. 1988, 16, 67–76. [Google Scholar] [CrossRef]
- Ayeka, P.A. Potential of mushroom compounds as immunomodulators in cancer immunotherapy: A review. Evid. Based Complement. Altern. Med. 2018, 2018, 7271509. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, M.; Hansen, R.; Ding, C.; Cramer, D.E.; Yan, J. Therapeutic potential of various beta-glucan sources in conjunction with anti-tumor monoclonal antibody in cancer therapy. Cancer Biol. Ther. 2009, 8, 218–225. [Google Scholar] [CrossRef]
- Novak, M.; Vetvicka, V. Beta-glucans, history, and the present: Immunomodulatory aspects and mechanisms of action. J. Immunotoxicol. 2008, 5, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vannucci, L.; Sima, P.; Richter, J. Beta glucan: Supplement or drug? From laboratory to clinical trials. Molecules 2019, 24, 1251. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vetvickova, J. Anti-infectious and anti-tumor activities of beta-glucans. Anticancer Res. 2020, 40, 3139–3145. [Google Scholar] [CrossRef] [PubMed]
- Blagodatski, A.; Yatsunskaya, M.; Mikhailova, V.; Tiasto, V.; Kagansky, A.; Katanaev, V.L. Medicinal mushrooms as an attractive new source of natural compounds for future cancer therapy. Oncotarget 2018, 9, 29259–29274. [Google Scholar] [CrossRef] [PubMed]
- Cypryk, W.; Ohman, T.; Eskelinen, E.L.; Matikainen, S.; Nyman, T.A. Quantitative proteomics of extracellular vesicles released from human monocyte-derived macrophages upon beta-glucan stimulation. J. Proteome Res. 2014, 13, 2468–2477. [Google Scholar] [CrossRef]
- Jozefowski, S.; Yang, Z.; Marcinkiewicz, J.; Kobzik, L. Scavenger receptors and beta-glucan receptors participate in the recognition of yeasts by murine macrophages. Inflamm. Res. 2012, 61, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Kogan, G.; Pajtinka, M.; Babincova, M.; Miadokova, E.; Rauko, P.; Slamenova, D.; Korolenko, T.A. Yeast cell wall polysaccharides as antioxidants and antimutagens: Can they fight cancer? Neoplasma 2008, 55, 387–393. [Google Scholar]
- Kogan, G.; Sandula, J.; Korolenko, T.A.; Falameeva, O.V.; Poteryaeva, O.N.; Zhanaeva, S.Y.; Levina, O.A.; Filatova, T.G.; Kaledin, V.I. Increased efficiency of Lewis lung carcinoma chemotherapy with a macrophage stimulator--yeast carboxymethyl glucan. Int. Immunopharmacol. 2002, 2, 775–781. [Google Scholar] [CrossRef]
- Legentil, L.; Paris, F.; Ballet, C.; Trouvelot, S.; Daire, X.; Vetvicka, V.; Ferrieres, V. Molecular interactions of beta-(1-->3)-glucans with their receptors. Molecules 2015, 20, 9745–9766. [Google Scholar] [CrossRef]
- Teplyakova, T.V.; Ilyicheva, T.N.; Andreeva, I.; Solovyanova, N. The activity of components of true tinder mushroom, Chaga Inonotus obliquus (Fr.) Pil. against viruses, bacteria and fungi (Abstract). In Proceedings of the 10th International Medicinal Mushroom Conference, Nantong, China, 19–22 September 2019; p. 117. [Google Scholar]
- Johnston, T.P.; Korolenko, T.A.; Pirro, M.; Sahebkar, A. Preventing cardiovascular heart disease: Promising nutraceutical and non-nutraceutical treatments for cholesterol management. Pharmacol. Res. 2017, 120, 219–225. [Google Scholar] [CrossRef] [PubMed]
- He, L.X.; Zhao, J.; Huang, Y.S.; Li, Y. The difference between oats and beta-glucan extract intake in the management of HbA1c, fasting glucose and insulin sensitivity: A meta-analysis of randomized controlled trials. Food Funct. 2016, 7, 1413–1428. [Google Scholar] [CrossRef] [PubMed]
- Baldassano, S.; Accardi, G.; Vasto, S. Beta-glucans and cancer: The influence of inflammation and gut peptide. Eur. J. Med. Chem. 2017, 142, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Shnyreva, A.V.; Shnyreva, A.A.; Espinoza, C.; Padron, J.M.; Trigos, A. Antiproliferative activity and cytotoxicity of some medicinal wood-destroying mushrooms from Russia. Int. J. Med. Mushrooms 2018, 20, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Teplyakova, T.V.; Ilyicheva, T.N.; Markovich, N.A. Prospects for the development of anti-influenza drugs based on medicinal mushrooms (review). Appl. Biochem. Microbiol. 2020, 56, 489–496. [Google Scholar] [CrossRef]
- Šandula, J.; Kogan, G.; Kačuráková, M.; Machová, E. Microbial (1→3)-β-d-glucans, their preparation, physico-chemical characterization and immunomodulatory activity. Carbohydr. Polym. 1999, 38, 247–253. [Google Scholar] [CrossRef]
- William, D.L.; Lowman, D.W.; Reale, M.; Ensley, H.E. Insights into the physicochemical characterization, chemistry, structure and synthesis of (1-3,1-6)- β-glucans. In Biology and Chemistry of Beta Glucan: Beta-Glucan, Structure, Chemistry and Specific Application; Vetvicka, V., Novak, M., Eds.; Bentham Science Publishers: Sharjah, United Arab Emirates, 2013; Volume 2. [Google Scholar]
- Theis, T.V.; Queiroz Santos, V.A.; Appelt, P.; Barbosa-Dekker, A.M.; Vetvicka, V.; Dekker, R.F.H.; Cunha, M.A.A. Fungal exocellular (1-6)-beta-d-glucan: Carboxymethylation, characterization, and antioxidant activity. Int. J. Mol. Sci. 2019, 20, 2337. [Google Scholar] [CrossRef] [PubMed]
- Pillemer, L.; Ecker, E.E. Anticomplementary factor in fresh yeast. J. Biol. Chem. 1941, 137, 139–142. [Google Scholar] [CrossRef]
- Di Luzio, N.R.; Riggi, S.J. The effects of laminarin, sulfated glucan and oligosaccharides of glucan on reticuloendothelial activity. J. Reticuloendothel. Soc. 1970, 8, 465–473. [Google Scholar]
- Chihara, G.; Maeda, Y.; Hamuro, J.; Sasaki, T.; Fukuoka, F. Inhibition of mouse sarcoma 180 by polysaccharides from Lentinus edodes (Berk.) Sing. Nature 1969, 222, 687–688. [Google Scholar] [CrossRef]
- Sari, M.; Prange, A.; Lelley, J.I.; Hambitzer, R. Screening of beta-glucan contents in commercially cultivated and wild growing mushrooms. Food Chem. 2017, 216, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.T.; Wasser, S.P. The role of culinary-medicinal mushrooms on human welfare with a pyramid model for human health. Int. J. Med. Mushrooms 2012, 14, 95–134. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.D.; Gordon, S. Immune recognition of fungal beta-glucans. Cell. Microbiol. 2005, 7, 471–479. [Google Scholar] [CrossRef]
- Kalac, P. A review of chemical composition and nutritional value of wild-growing and cultivated mushrooms. J. Sci. Food Agric. 2013, 93, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Wasser, S.P. Medicinal mushroom science: Current perspectives, advances, evidences, and challenges. Biomed. J. 2014, 37, 345–356. [Google Scholar] [CrossRef]
- Wasser, S.P. Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides. Appl. Microbiol. Biotechnol. 2002, 60, 258–274. [Google Scholar] [CrossRef]
- Grundemann, C.; Reinhardt, J.K.; Lindequist, U. European medicinal mushrooms: Do they have potential for modern medicine?—An update. Phytomedicine 2020, 66, 153131. [Google Scholar] [CrossRef] [PubMed]
- Shikov, A.N.; Pozharitskaya, O.N.; Makarov, V.G.; Wagner, H.; Verpoorte, R.; Heinrich, M. Medicinal plants of the Russian Pharmacopoeia; their history and applications. J. Ethnopharmacol. 2014, 154, 481–536. [Google Scholar] [CrossRef]
- Ren, L.; Perera, C.; Hemar, Y. Antitumor activity of mushroom polysaccharides: A review. Food Funct. 2012, 3, 1118–1130. [Google Scholar] [CrossRef]
- Wasser, S.P. Medicinal mushrooms in human clinical studies. Part I. Anticancer, oncoimmunological, and immunomodulatory activities: A review. Int. J. Med. Mushrooms 2017, 19, 279–317. [Google Scholar] [CrossRef]
- Byerrum, R.U.; Clarke, D.A.; Lucas, E.H.; Ringler, R.L.; Stevens, J.A.; Stock, C.C. Tumor inhibitors in Boletus edulis and other Holobasidiomycetes. Antibiot. Chemother. 1957, 7, 1–4. [Google Scholar]
- Meng, X.; Liang, H.; Luo, L. Antitumor polysaccharides from mushrooms: A review on the structural characteristics, antitumor mechanisms and immunomodulating activities. Carbohydr. Res. 2016, 424, 30–41. [Google Scholar] [CrossRef]
- Wasser, S.P.; Weis, A.L. Medicinal properties of substances occurring in higher Basidiomycetes mushrooms: Current perspectives (review). Int. J. Med. Mushrooms 1999, 1, 31–62. [Google Scholar] [CrossRef]
- Inomata, T.; Goodman, G.B.; Fryer, C.J.; Chaplin, D.J.; Palcic, B.; Lam, G.K.; Nishioka, A.; Ogawa, Y. Immune reaction induced by X-rays and pions and its stimulation by schizophyllan (SPG). Br. J. Cancer Suppl. 1996, 27, S122–S125. [Google Scholar]
- Shimizu, Y.; Hasumi, K.; Masubuchi, K. Augmenting effect of sizofiran on the immunofunction of regional lymph nodes in cervical cancer. Cancer 1992, 69, 1184–1194. [Google Scholar]
- Suzuki, I.; Itani, T.; Ohno, N.; Oikawa, S.; Sato, K.; Miyazaki, T.; Yadomae, T. Effect of a polysaccharide fraction from Grifola frondosa on immune response in mice. J. Pharmacobiodyn. 1985, 8, 217–226. [Google Scholar] [CrossRef]
- Hetland, G.; Tangen, J.M.; Mahmood, F.; Mirlashari, M.R.; Nissen-Meyer, L.S.H.; Nentwich, I.; Therkelsen, S.P.; Tjonnfjord, G.E.; Johnson, E. Antitumor, anti-inflammatory and antiallergic effects of Agaricus blazei mushroom extract and the related medicinal basidiomycetes mushrooms, Hericium erinaceus and Grifola frondosa: A review of preclinical and clinical studies. Nutrients 2020, 12, 1339. [Google Scholar] [CrossRef]
- Masuda, Y.; Matsumoto, A.; Toida, T.; Oikawa, T.; Ito, K.; Nanba, H. Characterization and antitumor effect of a novel polysaccharide from Grifola frondosa. J. Agric. Food Chem. 2009, 57, 10143–10149. [Google Scholar] [CrossRef]
- Akramiene, D.; Kondrotas, A.; Didziapetriene, J.; Kevelaitis, E. Effects of beta-glucans on the immune system. Medicina 2007, 43, 597–606. [Google Scholar] [CrossRef]
- Ito, K.; Masuda, Y.; Yamasaki, Y.; Yokota, Y.; Nanba, H. Maitake beta-glucan enhances granulopoiesis and mobilization of granulocytes by increasing G-CSF production and modulating CXCR4/SDF-1 expression. Int. Immunopharmacol. 2009, 9, 1189–1196. [Google Scholar] [CrossRef]
- Kobayashi, H.; Yoshida, R.; Kanada, Y.; Fukuda, Y.; Yagyu, T.; Inagaki, K.; Kondo, T.; Kurita, N.; Suzuki, M.; Kanayama, N.; et al. Suppressing effects of daily oral supplementation of beta-glucan extracted from Agaricus blazei Murill on spontaneous and peritoneal disseminated metastasis in mouse model. J. Cancer Res. Clin. Oncol. 2005, 131, 527–538. [Google Scholar] [CrossRef]
- Itoh, H.; Ito, H.; Amano, H.; Noda, H. Inhibitory action of a (1-->6)-beta-D-glucan-protein complex (F III-2-b) isolated from Agaricus blazei Murill (“himematsutake”) on Meth A fibrosarcoma-bearing mice and its antitumor mechanism. Jpn. J. Pharmacol. 1994, 66, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Tangen, J.M.; Holien, T.; Mirlashari, M.R.; Misund, K.; Hetland, G. Cytotoxic effect on human myeloma cells and leukemic cells by the Agaricus blazei Murill based mushroom extract, andosan. BioMed Res. Int. 2017, 2017, 2059825. [Google Scholar] [CrossRef] [PubMed]
- Tangen, J.M.; Tierens, A.; Caers, J.; Binsfeld, M.; Olstad, O.K.; Troseid, A.M.; Wang, J.; Tjonnfjord, G.E.; Hetland, G. Immunomodulatory effects of the Agaricus blazei Murrill-based mushroom extract AndoSan in patients with multiple myeloma undergoing high dose chemotherapy and autologous stem cell transplantation: A randomized, double blinded clinical study. BioMed Res. Int. 2015, 2015, 718539. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.N.; Chang, C.S.; Hung, M.H.; Chen, S.; Wang, W.; Tai, C.J.; Lu, C.L. The effect of mushroom beta-glucans from solid culture of ganoderma lucidum on inhibition of the primary tumor metastasis. Evid. Based Complement. Alternat. Med. 2014, 2014, 252171. [Google Scholar] [CrossRef]
- Rubel, R.; Santa, H.S.D.; Dos Santos, L.F.; Fernandes, L.C.; Figueiredo, B.C.; Soccol, C.R. Immunomodulatory and antitumoral properties of Ganoderma lucidum and Agaricus brasiliensis (agaricomycetes) medicinal mushrooms. Int. J. Med. Mushrooms 2018, 20, 393–403. [Google Scholar] [CrossRef]
- Ren, L.; Zhang, J.; Zhang, T. Immunomodulatory activities of polysaccharides from Ganoderma on immune effector cells. Food Chem. 2021, 340, 127933. [Google Scholar] [CrossRef] [PubMed]
- Dou, H.; Chang, Y.; Zhang, L. Coriolus versicolor polysaccharopeptide as an immunotherapeutic in China. Prog. Mol. Biol. Transl. Sci. 2019, 163, 361–381. [Google Scholar] [CrossRef]
- Chang, Y.; Zhang, M.; Jiang, Y.; Liu, Y.; Luo, H.; Hao, C.; Zeng, P.; Zhang, L. Preclinical and clinical studies of Coriolus versicolor polysaccharopeptide as an immunotherapeutic in China. Discov. Med. 2017, 23, 207–219. [Google Scholar] [PubMed]
- Kidd, P.M. The use of mushroom glucans and proteoglycans in cancer treatment. Altern. Med. Rev. 2000, 5, 4–27. [Google Scholar]
- Tsukagoshi, S.; Hashimoto, Y.; Fujii, G.; Kobayashi, H.; Nomoto, K.; Orita, K. Krestin (PSK). Cancer Treat. Rev. 1984, 11, 131–155. [Google Scholar] [CrossRef]
- Standish, L.J.; Wenner, C.A.; Sweet, E.S.; Bridge, C.; Nelson, A.; Martzen, M.; Novack, J.; Torkelson, C. Trametes versicolor mushroom immune therapy in breast cancer. J. Soc. Integr. Oncol. 2008, 6, 122–128. [Google Scholar]
- El-Deeb, N.M.; El-Adawi, H.I.; El-Wahab, A.E.A.; Haddad, A.M.; El Enshasy, H.A.; He, Y.W.; Davis, K.R. Modulation of NKG2D, KIR2DL and cytokine production by Pleurotus ostreatus glucan enhances natural killer cell cytotoxicity toward cancer cells. Front. Cell Dev. Biol. 2019, 7, 165. [Google Scholar] [CrossRef]
- Lavi, I.; Friesem, D.; Geresh, S.; Hadar, Y.; Schwartz, B. An aqueous polysaccharide extract from the edible mushroom Pleurotus ostreatus induces anti-proliferative and pro-apoptotic effects on HT-29 colon cancer cells. Cancer Lett. 2006, 244, 61–70. [Google Scholar] [CrossRef]
- Sasaki, T.; Takasuka, N. Further study of the structure of lentinan, an anti-tumor polysaccharide from Lentinus edodes. Carbohydr. Res. 1976, 47, 99–104. [Google Scholar] [CrossRef]
- Ladanyi, A.; Timar, J.; Lapis, K. Effect of lentinan on macrophage cytotoxicity against metastatic tumor cells. Cancer Immunol. Immunother. 1993, 36, 123–126. [Google Scholar] [CrossRef]
- Chihara, G.; Hamuro, J.; Maeda, Y.Y.; Shiio, T.; Suga, T.; Takasuka, N.; Sasaki, T. Antitumor and metastasis-inhibitory activities of lentinan as an immunomodulator: An overview. Cancer Detect. Prev. Suppl. 1987, 1, 423–443. [Google Scholar]
- Leibundgut-Landmann, S.; Osorio, F.; Brown, G.D.; Reis e Sousa, C. Stimulation of dendritic cells via the dectin-1/Syk pathway allows priming of cytotoxic T-cell responses. Blood 2008, 112, 4971–4980. [Google Scholar] [CrossRef]
- Zaidman, B.Z.; Yassin, M.; Mahajna, J.; Wasser, S.P. Medicinal mushroom modulators of molecular targets as cancer therapeutics. Appl. Microbiol. Biotechnol. 2005, 67, 453–468. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, Y.; Zhang, L.; Tian, Q. Mushroom polysaccharide lentinan for treating different types of cancers: A review of 12 years clinical studies in China. Prog. Mol. Biol. Transl. Sci. 2019, 163, 297–328. [Google Scholar] [CrossRef]
- Zhao, L.; Xiao, Y.; Xiao, N. Effect of lentinan combined with docetaxel and cisplatin on the proliferation and apoptosis of BGC823 cells. Tumor Biol. 2013, 34, 1531–1536. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, S.; Watanabe, S.; Imano, M.; Suga, T.; Nakazawa, S.; Hazama, S.; Oka, M. Improvement of QOL and prognosis by treatment of superfine dispersed lentinan in patients with advanced gastric cancer. Hepatogastroenterology 2010, 57, 172–177. [Google Scholar] [PubMed]
- Hazama, S.; Watanabe, S.; Ohashi, M.; Yagi, M.; Suzuki, M.; Matsuda, K.; Yamamoto, T.; Suga, Y.; Suga, T.; Nakazawa, S.; et al. Efficacy of orally administered superfine dispersed lentinan (beta-1,3-glucan) for the treatment of advanced colorectal cancer. Anticancer Res. 2009, 29, 2611–2617. [Google Scholar] [PubMed]
- Wang, W.; Dai, X.; Ouyang, X. [Efficacy of Lentinan combined with chemotherapy in advanced non-small cell lung cancer]. Zhongguo Fei Ai Za Zhi 2006, 9, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Vannucci, L.; Sima, P.; Vetvicka, V.; Krizan, J. Lentinan properties in anticancer therapy: A review on the last 12-year literature. Am. J. Immunol. 2017, 13, 50–61. [Google Scholar] [CrossRef]
- Teplyakova, T.V.; Ilyicheva, T.N.; Kosogova, T.A.; Wasser, S.P. Medicinal mushrooms against influenza viruses. Int. J. Med. Mushrooms 2021, 23, 1–11. [Google Scholar] [CrossRef]
- Yuan, H.; Lan, P.; He, Y.; Li, C.; Ma, X. Effect of the modifications on the physicochemical and biological properties of beta-glucan-a critical review. Molecules 2019, 25, 57. [Google Scholar] [CrossRef] [PubMed]
- Gantner, B.N.; Simmons, R.M.; Canavera, S.J.; Akira, S.; Underhill, D.M. Collaborative induction of inflammatory responses by dectin-1 and Toll-like receptor 2. J. Exp. Med. 2003, 197, 1107–1117. [Google Scholar] [CrossRef]
- Fang, J.; Wang, Y.; Lv, X.; Shen, X.; Ni, X.; Ding, K. Structure of a beta-glucan from Grifola frondosa and its antitumor effect by activating Dectin-1/Syk/NF-κB signaling. Glycoconj. J. 2012, 29, 365–377. [Google Scholar] [CrossRef]
- Hong, F.; Yan, J.; Baran, J.T.; Allendorf, D.J.; Hansen, R.D.; Ostroff, G.R.; Xing, P.X.; Cheung, N.K.; Ross, G.D. Mechanism by which orally administered beta-1,3-glucans enhance the tumoricidal activity of antitumor monoclonal antibodies in murine tumor models. J. Immunol. 2004, 173, 797–806. [Google Scholar] [CrossRef]
- Cheung, N.K.; Modak, S.; Vickers, A.; Knuckles, B. Orally administered beta-glucans enhance anti-tumor effects of monoclonal antibodies. Cancer Immunol. Immunother. 2002, 51, 557–564. [Google Scholar] [CrossRef]
- Salvador, C.; Li, B.; Hansen, R.; Cramer, D.E.; Kong, M.; Yan, J. Yeast-derived beta-glucan augments the therapeutic efficacy mediated by anti-vascular endothelial growth factor monoclonal antibody in human carcinoma xenograft models. Clin. Cancer Res. 2008, 14, 1239–1247. [Google Scholar] [CrossRef]
- Li, B.; Cai, Y.; Qi, C.; Hansen, R.; Ding, C.; Mitchell, T.C.; Yan, J. Orally administered particulate beta-glucan modulates tumor-capturing dendritic cells and improves antitumor T-cell responses in cancer. Clin. Cancer Res. 2010, 16, 5153–5164. [Google Scholar] [CrossRef] [PubMed]
- Albeituni, S.H.; Ding, C.; Liu, M.; Hu, X.; Luo, F.; Kloecker, G.; Bousamra, M., II; Zhang, H.G.; Yan, J. Yeast-derived particulate beta-glucan treatment subverts the suppression of myeloid-derived suppressor cells (MDSC) by inducing polymorphonuclear MDSC apoptosis and monocytic MDSC differentiation to APC in cancer. J. Immunol. 2016, 196, 2167–2180. [Google Scholar] [CrossRef]
- Geller, A.; Shrestha, R.; Yan, J. Yeast-derived beta-glucan in cancer: Novel uses of a traditional therapeutic. Int. J. Mol. Sci. 2019, 20, 3618. [Google Scholar] [CrossRef]
- Ishimoto, Y.; Ishibashi, K.I.; Yamanaka, D.; Adachi, Y.; Ito, H.; Igami, K.; Miyazaki, T.; Ohno, N. Protection against gut inflammation and sepsis in mice by the autodigested product of the Lingzhi medicinal mushroom, Ganoderma lingzhi (agaricomycetes). Int. J. Med. Mushrooms 2018, 20, 809–823. [Google Scholar] [CrossRef] [PubMed]
- Del Corno, M.; Gessani, S.; Conti, L. Shaping the innate immune response by dietary glucans: Any role in the control of cancer? Cancers 2020, 12, 155. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Zhu, X.; Huo, Z.; Liao, B.; Huang, J.; Wang, Z.; Song, C.; Hu, X.; Fang, J. A beta-glucan from Grifola frondosa effectively delivers therapeutic oligonucleotide into cells via dectin-1 receptor and attenuates TNFalpha gene expression. Int. J. Biol. Macromol. 2020, 149, 801–808. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vetvickova, J. Combination of glucan, resveratrol and vitamin C demonstrates strong anti-tumor potential. Anticancer Res. 2012, 32, 81–87. [Google Scholar]
- Hsiao, W.L.; Li, Y.Q.; Lee, T.L.; Li, N.; You, M.M.; Chang, S.T. Medicinal mushroom extracts inhibit ras-induced cell transformation and the inhibitory effect requires the presence of normal cells. Carcinogenesis 2004, 25, 1177–1183. [Google Scholar] [CrossRef]
- Zhang, X.; Li, T.; Liu, S.; Xu, Y.; Meng, M.; Li, X.; Lin, Z.; Wu, Q.; Xue, Y.; Pan, Y.; et al. beta-glucan from Lentinus edodes inhibits breast cancer progression via the Nur77/HIF-1alpha axis. Biosci. Rep. 2020, 40, BSR20201006. [Google Scholar] [CrossRef] [PubMed]
- Peyrin-Biroulet, L.; Lepage, C.; Jooste, V.; Gueant, J.L.; Faivre, J.; Bouvier, A.M. Colorectal cancer in inflammatory bowel diseases: A population-based study (1976–2008). Inflamm. Bowel Dis. 2012, 18, 2247–2251. [Google Scholar] [CrossRef] [PubMed]
- Triantafillidis, J.K.; Nasioulas, G.; Kosmidis, P.A. Colorectal cancer and inflammatory bowel disease: Epidemiology, risk factors, mechanisms of carcinogenesis and prevention strategies. Anticancer Res. 2009, 29, 2727–2737. [Google Scholar] [PubMed]
- Schwartz, B.; Hadar, Y. Possible mechanisms of action of mushroom-derived glucans on inflammatory bowel disease and associated cancer. Ann. Transl. Med. 2014, 2, 19. [Google Scholar] [CrossRef]
- Okamoto, T.; Kodoi, R.; Nonaka, Y.; Fukuda, I.; Hashimoto, T.; Kanazawa, K.; Mizuno, M.; Ashida, H. Lentinan from shiitake mushroom (Lentinus edodes) suppresses expression of cytochrome P450 1A subfamily in the mouse liver. Biofactors 2004, 21, 407–409. [Google Scholar] [CrossRef]
- Lavi, I.; Nimri, L.; Levinson, D.; Peri, I.; Hadar, Y.; Schwartz, B. Glucans from the edible mushroom Pleurotus pulmonarius inhibit colitis-associated colon carcinogenesis in mice. J. Gastroenterol. 2012, 47, 504–518. [Google Scholar] [CrossRef] [PubMed]
- Nowacka-Jechalke, N.; Nowak, R.; Lemieszek, M.K.; Rzeski, W.; Gawlik-Dziki, U.; Szpakowska, N.; Kaczynski, Z. Promising potential of crude polysaccharides from Sparassis crispa against colon cancer: An in vitro study. Nutrients 2021, 13, 161. [Google Scholar] [CrossRef]
- Hsu, W.H.; Qiu, W.L.; Tsao, S.M.; Tseng, A.J.; Lu, M.K.; Hua, W.J.; Cheng, H.C.; Hsu, H.Y.; Lin, T.Y. Effects of WSG, a polysaccharide from Ganoderma lucidum, on suppressing cell growth and mobility of lung cancer. Int. J. Biol. Macromol. 2020, 165, 1604–1613. [Google Scholar] [CrossRef]
- Maity, G.N.; Maity, P.; Choudhuri, I.; Bhattacharyya, N.; Acharya, K.; Dalai, S.; Mondal, S. Structural studies of a water insoluble beta-glucan from Pleurotus djamor and its cytotoxic effect against PA1, ovarian carcinoma cells. Carbohydr. Polym. 2019, 222, 114990. [Google Scholar] [CrossRef]
- Alonso, E.N.; Ferronato, M.J.; Gandini, N.A.; Fermento, M.E.; Obiol, D.J.; Lopez Romero, A.; Arevalo, J.; Villegas, M.E.; Facchinetti, M.M.; Curino, A.C. Antitumoral effects of D-Fraction from Grifola Frondosa (Maitake) mushroom in breast cancer. Nutr. Cancer 2017, 69, 29–43. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Saraswat-Ohri, S.; Vashishta, A.; Descroix, K.; Jamois, F.; Yvin, J.C.; Ferrieres, V. New 4-deoxy-(1-->3)-beta-D-glucan-based oligosaccharides and their immunostimulating potential. Carbohydr. Res. 2011, 346, 2213–2221. [Google Scholar] [CrossRef]
- Lin, T.Y.; Tseng, A.J.; Qiu, W.L.; Chao, C.H.; Lu, M.K. A sulfated glucan from Antrodia cinnamomea reduces Slug expression through regulation of TGFbeta/AKT/GSK3beta axis in lung cancer. Carbohydr. Polym. 2019, 210, 175–184. [Google Scholar] [CrossRef]
- Volman, J.J.; Helsper, J.P.; Wei, S.; Baars, J.J.; van Griensven, L.J.; Sonnenberg, A.S.; Mensink, R.P.; Plat, J. Effects of mushroom-derived beta-glucan-rich polysaccharide extracts on nitric oxide production by bone marrow-derived macrophages and nuclear factor-kappaB transactivation in Caco-2 reporter cells: Can effects be explained by structure? Mol. Nutr. Food Res. 2010, 54, 268–276. [Google Scholar] [CrossRef]
- Aleem, E. beta-Glucans and their applications in cancer therapy: Focus on human studies. Anticancer Agents Med. Chem. 2013, 13, 709–719. [Google Scholar] [CrossRef]
- Motta, F.; Gershwin, M.E.; Selmi, C. Mushrooms and immunity. J. Autoimmun. 2021, 117, 102576. [Google Scholar] [CrossRef]
- Steimbach, L.; Borgmann, A.V.; Gomar, G.G.; Hoffmann, L.V.; Rutckeviski, R.; de Andrade, D.P.; Smiderle, F.R. Fungal beta-glucans as adjuvants for treating cancer patients—A systematic review of clinical trials. Clin. Nutr. 2020, in press. [Google Scholar] [CrossRef]
- Chaichian, S.; Moazzami, B.; Sadoughi, F.; Haddad Kashani, H.; Zaroudi, M.; Asemi, Z. Functional activities of beta-glucans in the prevention or treatment of cervical cancer. J. Ovarian Res. 2020, 13, 24. [Google Scholar] [CrossRef] [PubMed]
- Rop, O.; Mlcek, J.; Jurikova, T. Beta-glucans in higher fungi and their health effects. Nutr. Rev. 2009, 67, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.-J. Influences of gut hormones on hepatocellular carcinoma. Endocrinol. Metab. Synd. 2015, 4, 1017–2161. [Google Scholar] [CrossRef]
- Mo, L.; Chen, Y.; Li, W.; Guo, S.; Wang, X.; An, H.; Zhan, Y. Anti-tumor effects of (1-->3)-beta-d-glucan from Saccharomyces cerevisiae in S180 tumor-bearing mice. Int. J. Biol. Macromol. 2017, 95, 385–392. [Google Scholar] [CrossRef]
- Li, X.; Luo, H.; Ye, Y.; Chen, X.; Zou, Y.; Duan, J.; Xiang, D. betaglucan, a dectin1 ligand, promotes macrophage M1 polarization via NFkappaB/autophagy pathway. Int. J. Oncol. 2019, 54, 271–282. [Google Scholar] [CrossRef]
- Morales, D.; Rutckeviski, R.; Villalva, M.; Abreu, H.; Soler-Rivas, C.; Santoyo, S.; Iacomini, M.; Smiderle, F.R. Isolation and comparison of alpha- and beta-D-glucans from shiitake mushrooms (Lentinula edodes) with different biological activities. Carbohydr. Polym. 2020, 229, 115521. [Google Scholar] [CrossRef]
- Wagener, J.; Striegler, K.; Wagener, N. alpha- and beta-1,3-Glucan synthesis and remodeling. Curr. Top. Microbiol. Immunol. 2020, 425, 53–82. [Google Scholar] [CrossRef]
- Jin, Y.; Li, P.; Wang, F. beta-glucans as potential immunoadjuvants: A review on the adjuvanticity, structure-activity relationship and receptor recognition properties. Vaccine 2018, 36, 5235–5244. [Google Scholar] [CrossRef]
- Mori, K.; Naganuma, M.; Mizuno, S.; Suzuki, H.; Kitazume, M.T.; Shimamura, K.; Chiba, S.; Sugita, A.; Matsuoka, K.; Hisamatsu, T.; et al. beta-(1,3)-Glucan derived from Candida albicans induces inflammatory cytokines from macrophages and lamina propria mononuclear cells derived from patients with Crohn’s disease. Intest. Res. 2018, 16, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jin, Z.; Zhang, W.; Xie, X.; Song, N.; Lv, T.; Wu, D.; Cao, Y. The preventable efficacy of beta-glucan against leptospirosis. PLoS Negl. Trop. Dis. 2019, 13, e0007789. [Google Scholar] [CrossRef]
- PDQ. Medicinal Mushrooms (PDQ®): Health Professional Version. In PDQ Cancer Information Summaries; Bethesda: Rockville, MD, USA, 2020; pp. 2002–2020. [Google Scholar]
- Murphy, E.J.; Rezoagli, E.; Major, I.; Rowan, N.J.; Laffey, J.G. beta-Glucan metabolic and immunomodulatory properties and potential for clinical application. J. Fungi 2020, 6, 356. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhang, X.D.; Jiang, Z. The application of fungal beta-glucans for the treatment of colon cancer. Anticancer Agents Med. Chem. 2013, 13, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hodge, J.; Liu, Q.; Wang, J.; Wang, Y.; Evans, T.D.; Altomare, D.; Yao, Y.; Murphy, E.A.; Razani, B.; et al. TFEB is a master regulator of tumor-associated macrophages in breast cancer. J. Immunother. Cancer 2020, 8, e000543. [Google Scholar] [CrossRef]
- Su, Y.; Chen, L.; Yang, F.; Cheung, P.C.K. Beta-d-glucan-based drug delivery system and its potential application in targeting tumor associated macrophages. Carbohydr. Polym. 2021, 253, 117258. [Google Scholar] [CrossRef] [PubMed]
- Salmaninejad, A.; Valilou, S.F.; Soltani, A.; Ahmadi, S.; Abarghan, Y.J.; Rosengren, R.J.; Sahebkar, A. Tumor-associated macrophages: Role in cancer development and therapeutic implications. Cell. Oncol. 2019, 42, 591–608. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Xu, J.; Lan, H. Tumor-associated macrophages in tumor metastasis: Biological roles and clinical therapeutic applications. J. Hematol. Oncol. 2019, 12, 76. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Marchesi, F.; Malesci, A.; Laghi, L.; Allavena, P. Tumour-associated macrophages as treatment targets in oncology. Nat. Rev. Clin. Oncol. 2017, 14, 399–416. [Google Scholar] [CrossRef]
- De Graaff, P.; Berrevoets, C.; Rsch, C.; Schols, H.A.; Verhoef, K.; Wichers, H.J.; Debets, R.; Govers, C. Curdlan, zymosan and a yeast-derived beta-glucan reshape tumor-associated macrophages into producers of inflammatory chemo-attractants. Cancer Immunol. Immunother. 2021, 70, 547–561. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; He, Y.; Zeng, P.; Liu, Y.; Zhang, M.; Hao, C.; Wang, H.; Lv, Z.; Zhang, L. Molecular basis for Poria cocos mushroom polysaccharide used as an antitumour drug in China. J. Cell. Mol. Med. 2019, 23, 4–20. [Google Scholar] [CrossRef]




| Name | Source | Type of Polymer |
|---|---|---|
| Lentinan | Lentinus edodes | Linear |
| Pleuran | Pleurotus ostreatus | Branched |
| Pachymaran | Poria cocos | Linear |
| Schizophyllan | Schizophyllum commune | Branched |
| Grifolan | Grifola frondosa | Branched |
| Maitake | Grifola frondosa | Branched |
| Pestolan | Pestolatia sp. | Linear |
| Coriolan | Coriolus versicolor | Linear |
| β-Glucan | Cryptoporus colvatus | Branched |
| Polycan | Aureobasidium pullans | Branched |
| β-Glucan | Ganoderma lucidum | Linear |
| β-Glucan | Agaricus blazei | Branched |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vetvicka, V.; Teplyakova, T.V.; Shintyapina, A.B.; Korolenko, T.A. Effects of Medicinal Fungi-Derived β-Glucan on Tumor Progression. J. Fungi 2021, 7, 250. https://doi.org/10.3390/jof7040250
Vetvicka V, Teplyakova TV, Shintyapina AB, Korolenko TA. Effects of Medicinal Fungi-Derived β-Glucan on Tumor Progression. Journal of Fungi. 2021; 7(4):250. https://doi.org/10.3390/jof7040250
Chicago/Turabian StyleVetvicka, Vaclav, Tamara V. Teplyakova, Alexandra B. Shintyapina, and Tatiana A. Korolenko. 2021. "Effects of Medicinal Fungi-Derived β-Glucan on Tumor Progression" Journal of Fungi 7, no. 4: 250. https://doi.org/10.3390/jof7040250
APA StyleVetvicka, V., Teplyakova, T. V., Shintyapina, A. B., & Korolenko, T. A. (2021). Effects of Medicinal Fungi-Derived β-Glucan on Tumor Progression. Journal of Fungi, 7(4), 250. https://doi.org/10.3390/jof7040250








