Selective Inhibition of Human Monoamine Oxidase B by 5-hydroxy-2-methyl-chroman-4-one Isolated from an Endogenous Lichen Fungus Daldinia fissa
Abstract
1. Introduction
2. Materials and Methods
2.1. Extracts of Endogenous Lichen Fungi and Evaluations of Inhibitory Activities
2.2. Monoamine Oxidase (MAO) Activity Assay
2.3. Cholinesterase Activity Assay
2.4. β-Site Amyloid Precursor Protein Cleaving Enzyme 1 (BACE-1) Activity Assay
2.5. Antioxidant Activity Assay
2.6. Culture and Extraction of the Selected Endogenous Lichen Fungi
2.7. Thin-Layer Chromatography
2.8. Structure Analysis through Nuclear Magnetic Resonance (NMR), Liquid Chromatography–Mass Spectrometry (LC-MS)
2.9. Inhibitory Activities of the Compound and Enzyme Kinetics
2.10. Inhibitor Reversibility Analysis
2.11. Cytotoxicity Analysis of the Compound
2.12. Docking Simulations and Molecular Dynamics of the Compounds with Monoamine Oxidase-A (MAO-A) and MAO-B
2.13. Pharmacokinetic Analysis of the Compound Using the In Silico Method
3. Results
3.1. Inhibitory Activities against the Enzymes and Antioxidant Activities of the Extracts
3.2. Isolation of Compounds from Endogenous Lichen Fungi 13 (ELF13) Using Prep Thin-Layer Chromatography (TLC)
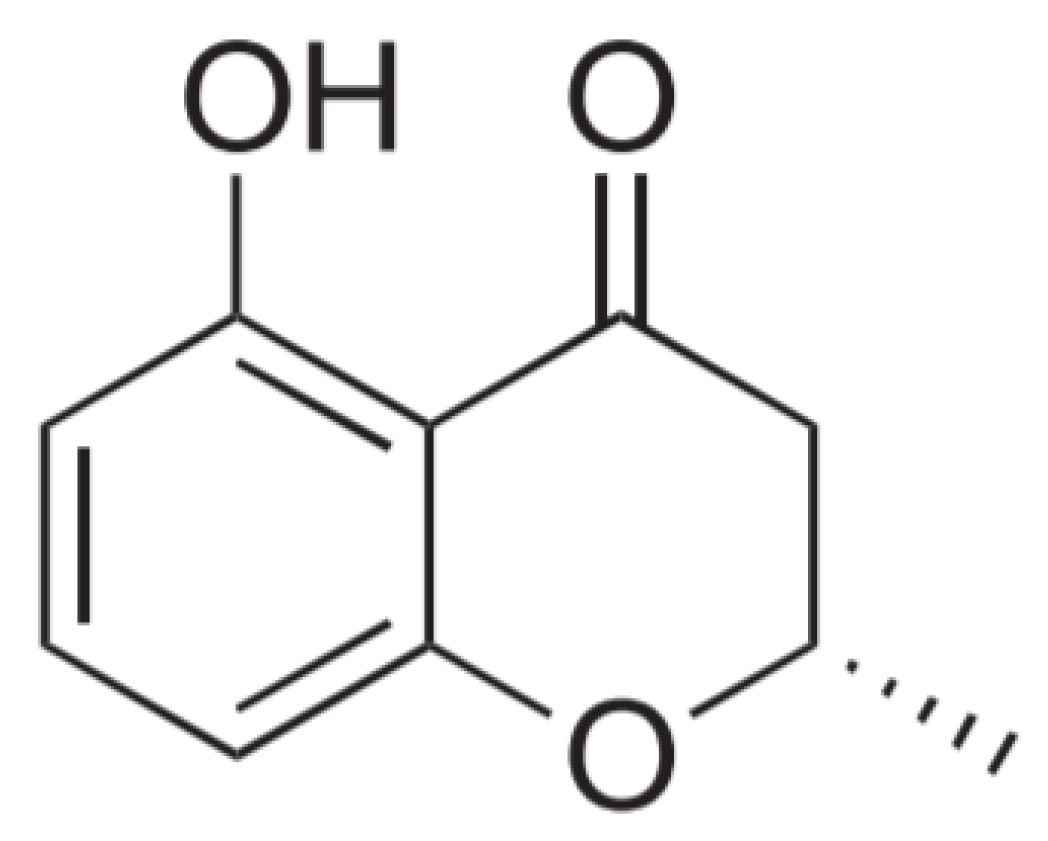
3.3. Molecular Structure Analysis of C2
3.4. Inhibitory Activities of HMC (5-hydroxy-2-methyl-chroman-4-one) against the Enzymes
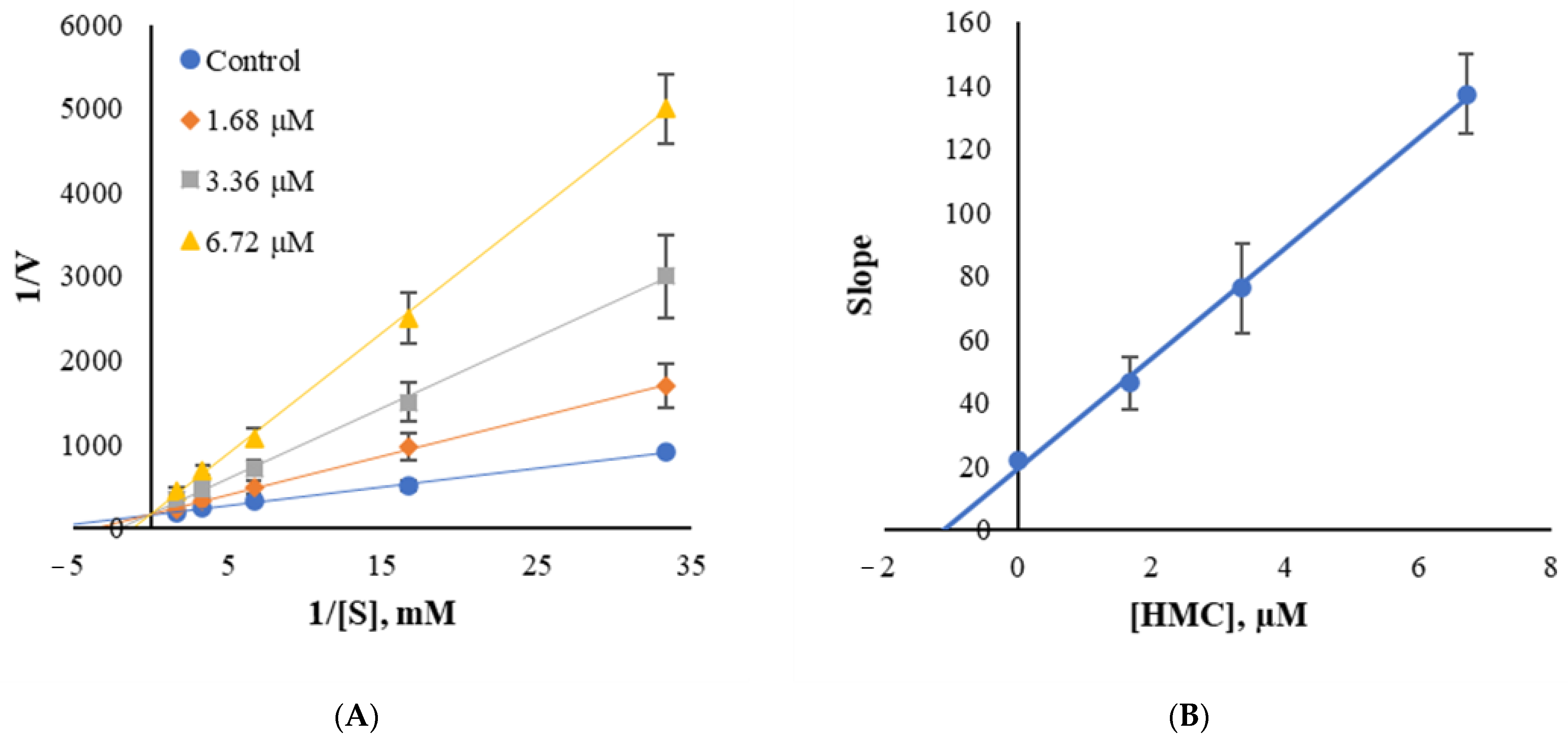
3.5. Inhibition Patterns of HMC
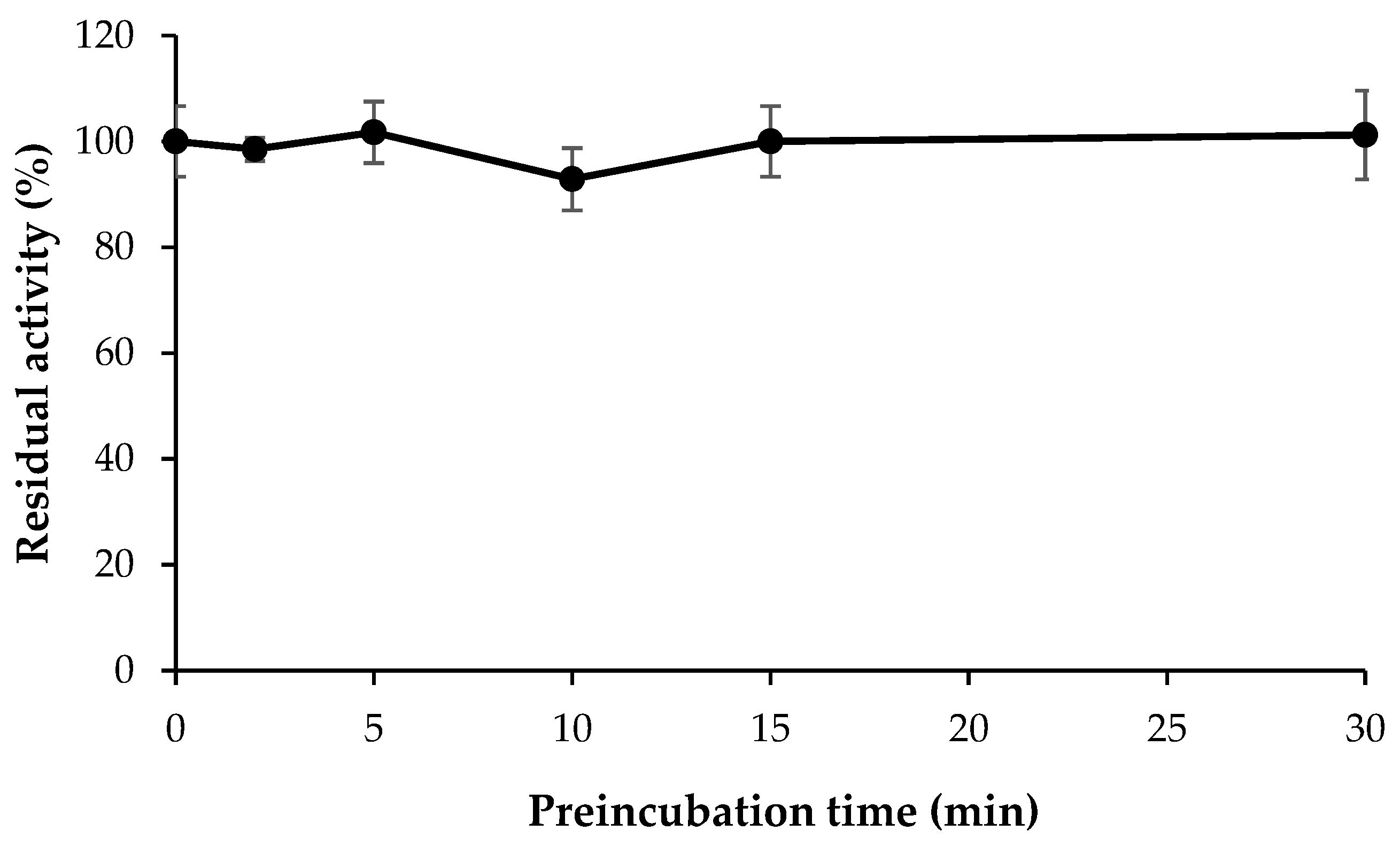
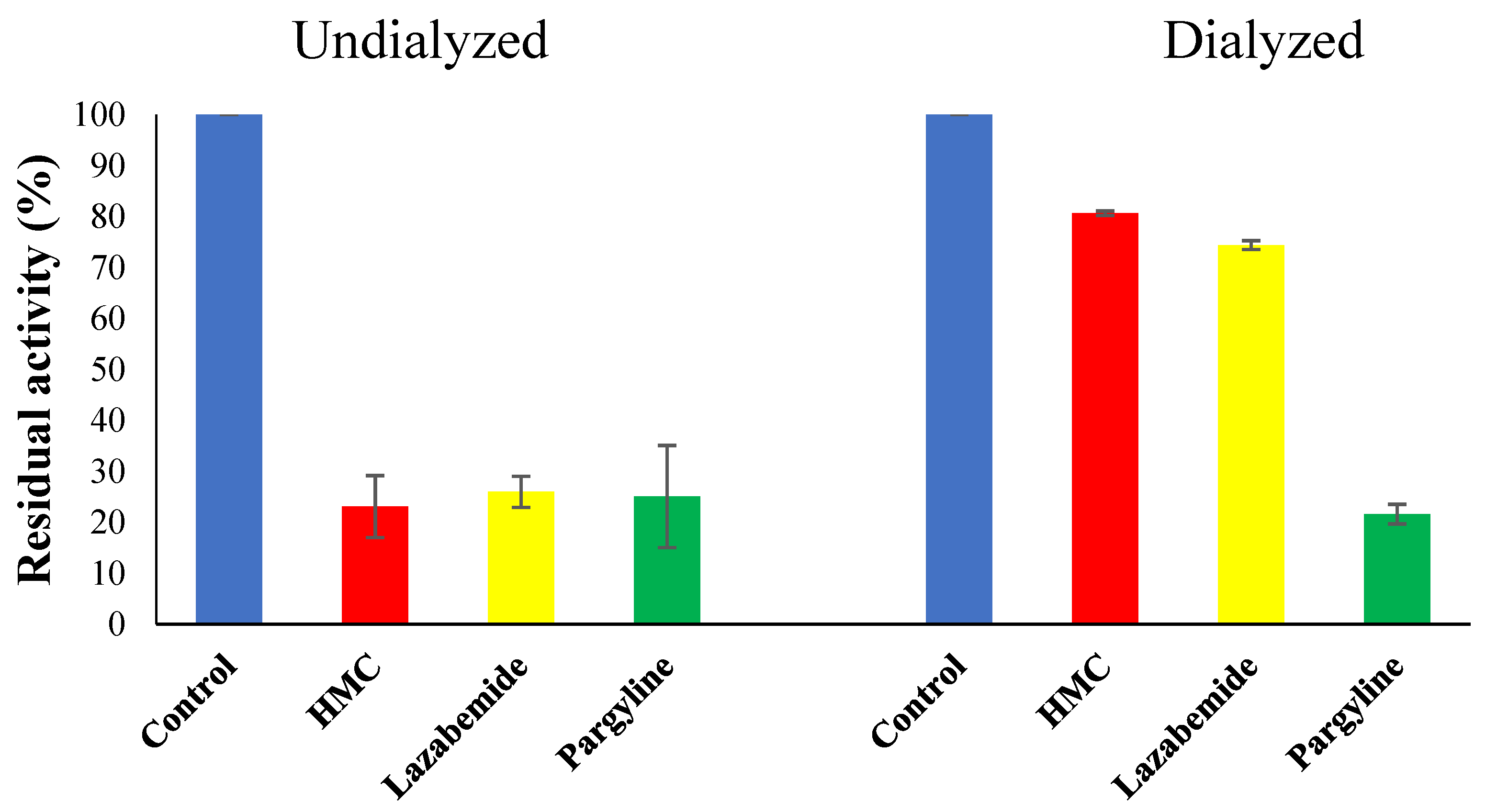
3.6. Inhibitor Reversibility of HMC
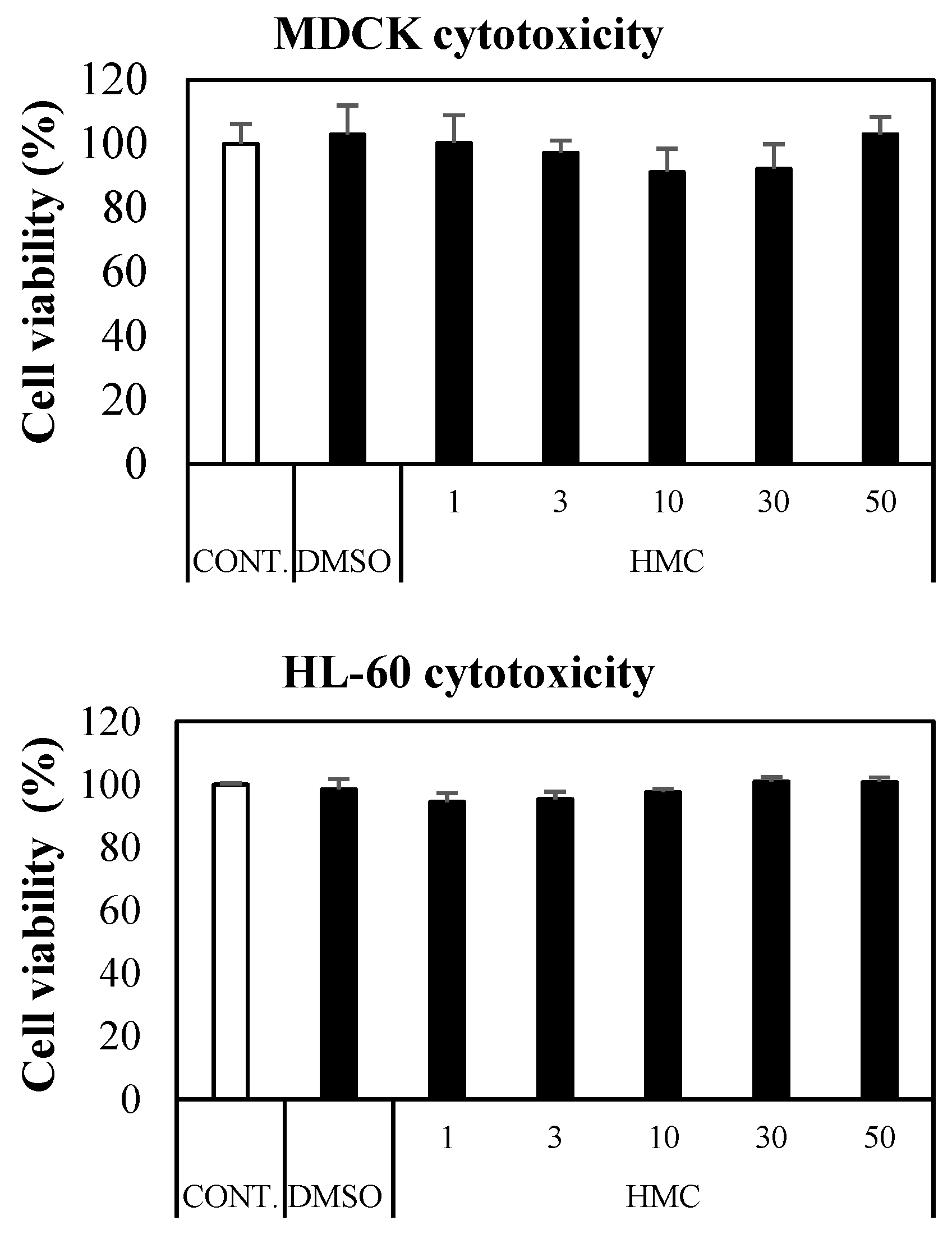
3.7. Cytotoxicity of HMC
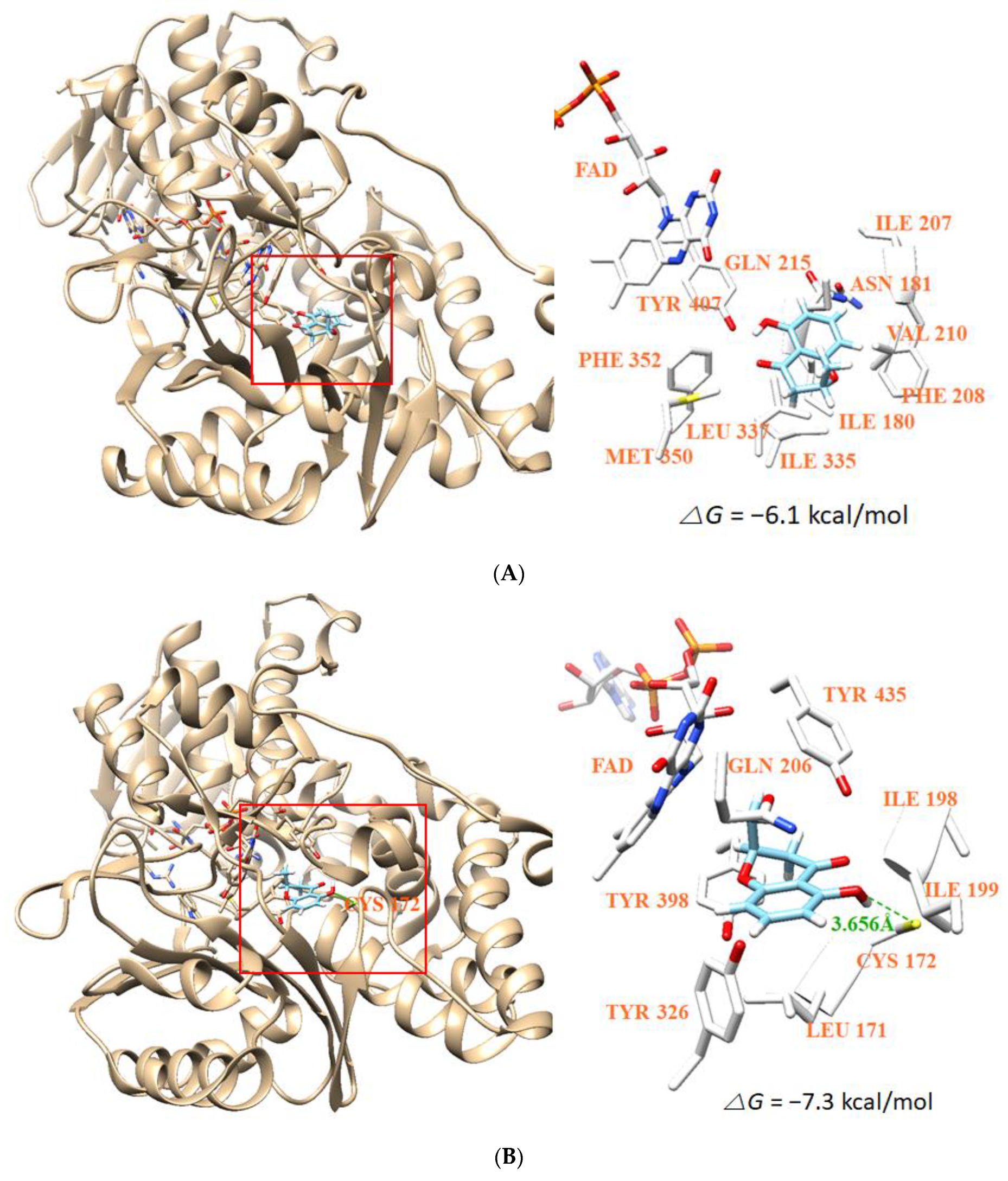
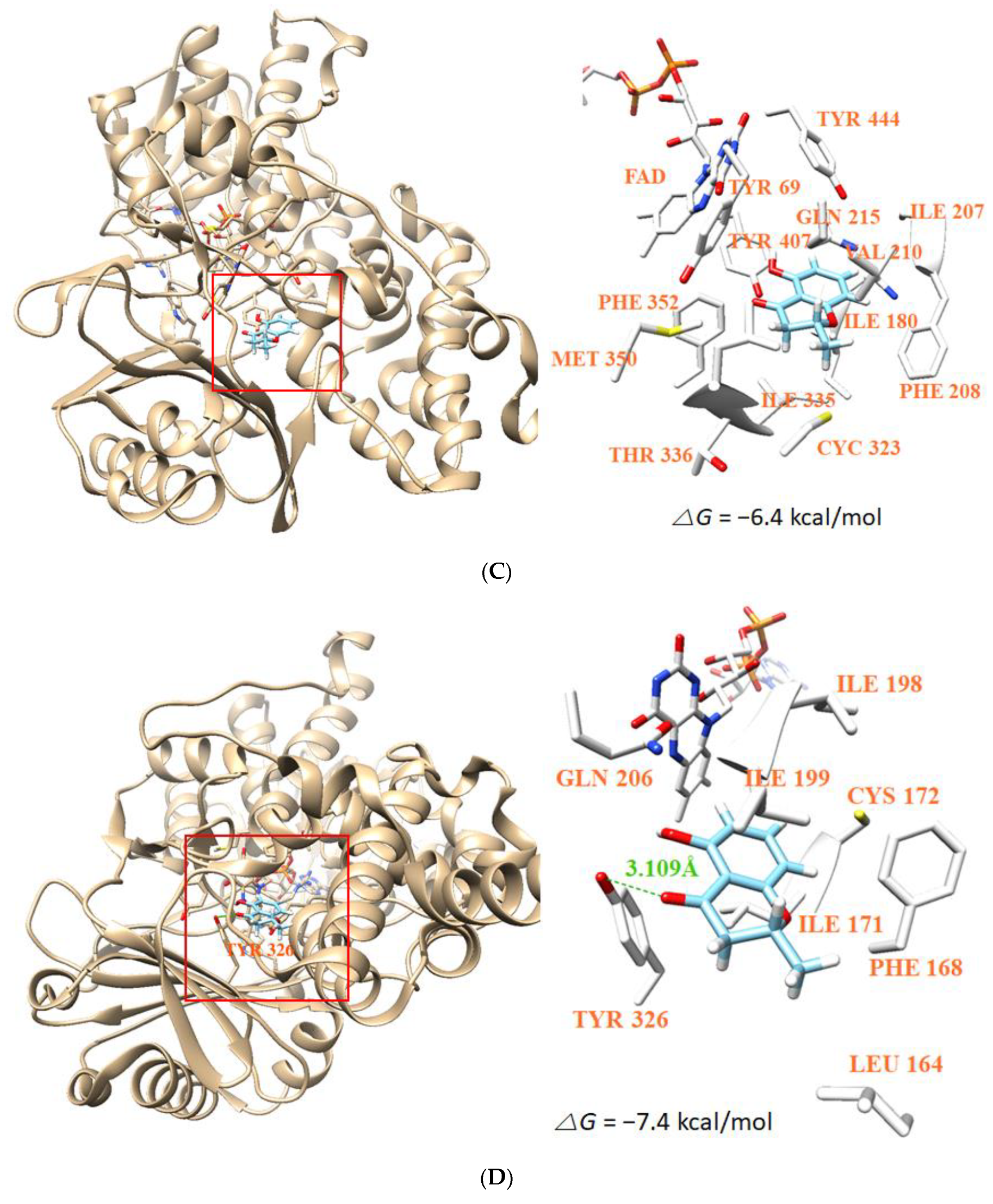
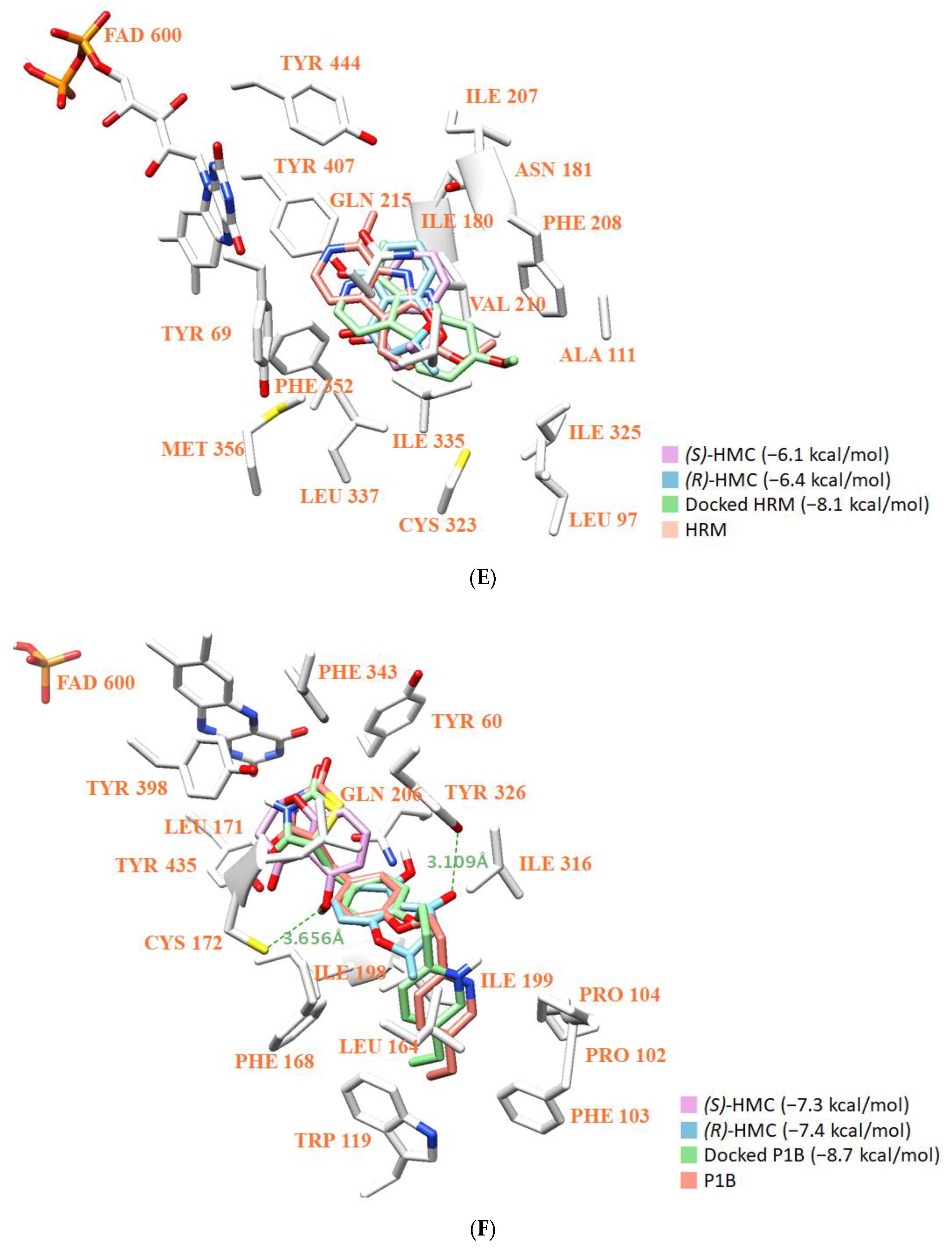
3.8. Molecular Docking Simulation and Molecular Dynamics
3.9. In Silico Pharmacokinetics of HMC
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Reece, J.B.; Taylor, M.R.; Simson, E.J.; Dickey, J.L. Campbell Biology: Concepts and Connections, 7th ed.; Pearson: London, UK, 2012. [Google Scholar]
- Berdy, J. Bioactive microbial metabolites. J. Antibiot. 2005, 58, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.M.; Kim, Y.J.; Gang, H.S.; Han, J.; Ha, H.H.; Kim, H. Antimicrobial activity of divaricatic acid isolated from the lichen Evernia mesomorpha against methicillin-resistant Staphylococcus aureus. Molecules 2018, 23, 3068. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.W. American arctic lichens: The macrolichens; Columbia University Press: New York, NY, USA, 1984. [Google Scholar]
- Olafsdottir, E.S.; Ingolfsdottir, K. Polysaccharides from lichens: Structural characteristics and biological activity. Planta Med. 2001, 67, 199–208. [Google Scholar] [CrossRef]
- Burns, A.; Iliffe, S. Alzheimer’s disease. BMJ 2009, 338, b158. [Google Scholar] [CrossRef] [PubMed]
- Förstl, H.; Kurz, A. Clinical features of Alzheimer’s disease. Eur. Arch. Psych. Clin. Neurosci. 1999, 249, 288–290. [Google Scholar] [CrossRef]
- Hardy, J.; Allsop, D. Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends Pharmacol. Sci. 1991, 12, 383–388. [Google Scholar] [CrossRef]
- Francis, P.T.; Palmer, A.M.; Snape, M.; Wilcock, G.K. The cholinergic hypothesis of Alzheimer’s disease: A review of progress. J. Neurol. Neurosurg. Psychiatry 1999, 66, 137–147. [Google Scholar] [CrossRef]
- de Zwart, P.L.; Jeronimus, B.F.; de Jonge, P. Empirical evidence for definitions of episode, remission, recovery, relapse and recurrence in depression: A systematic review. Epidemiol. Psychiatr. Sci. 2019, 28, 544–562. [Google Scholar] [CrossRef]
- Orhan, I.E. Potential of natural products of herbal origin as monoamine oxidase inhibitors. Curr. Pharm. Des. 2016, 22, 268–276. [Google Scholar] [CrossRef]
- Ramsay, R.R. Monoamine oxidases: The biochemistry of the proteins as targets in medicinal chemistry and drug discovery. Curr. Top. Med. Chem. 2012, 12, 2189–2209. [Google Scholar] [CrossRef]
- Youdim, M.B.; Edmondson, D.; Tipton, K.F. The therapeutic potential of monoamine oxidase inhibitors. Nat. Rev. Neurosci. 2006, 7, 295–309. [Google Scholar] [CrossRef] [PubMed]
- Bierer, L.M.; Haroutunian, V.; Gabriel, S.; Knott, P.J.; Carlin, L.S.; Purohit, D.P.; Perl, D.P.; Schmeidler, J.; Kanof, P.; Davis, K.L. Neurochemical correlates of dementia severity in Alzheimer’s disease: Relative importance of the cholinergic deficits. J. Neurochem. 1995, 64, 749–760. [Google Scholar] [CrossRef]
- Kumar, A.; Pintus, F.; Di Petrillo, A.; Medda, R.; Caria, P.; Matos, M.J.; Vina, D.; Pieroni, E.P.; Delogu, F.; Era, B.; et al. Novel 2-phenylbenzofuran derivatives as selective butyrylcholinesterase inhibitors for Alzheimer’s disease. Sci. Rep. 2018, 8, 4424. [Google Scholar] [CrossRef] [PubMed]
- Korábečný, J.; Nepovimová, E.; Cikánková, T.; Špilovská, K.; Vašková, L.; Mezeiová, E.; Kuča, K.; Hroudová, J. Newly developed drugs for Alzheimer’s disease in relation to energy metabolism, cholinergic and monoaminergic neurotransmission. Neuroscience 2018, 370, 191–206. [Google Scholar]
- Citron, M. Beta-secretase as a target for the treatment of Alzheimer’s disease. J. Neurosci. Res. 2002, 70, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.; Turner, R.T., 3rd; Koelsch, G.; Ghosh, A.K.; Tang, J. Memapsin 2 (beta secretase) as a therapeutic target. Biochem. Soc. Trans. 2002, 30, 530–534. [Google Scholar] [CrossRef][Green Version]
- Lee, H.W.; Ryu, H.W.; Kang, M.G.; Park, D.; Oh, S.R.; Kim, H. Potent selective monoamine oxidase B inhibition by maackiain, a pterocarpan from the roots of Sophora flavescens. Bioorg. Med. Chem. Lett. 2016, 26, 4714–4719. [Google Scholar] [CrossRef]
- Baek, S.C.; Choi, B.; Nam, S.J.; Kim, H. Inhibition of monoamine oxidase A and B by demethoxycurcumin and bisdemethoxycurcumin. J. Appl. Biol. Chem. 2018, 61, 187–190. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, J.V.; Feather-Stone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Lee, J.P.; Kang, M.G.; Lee, J.Y.; Oh, J.M.; Baek, S.C.; Leem, H.H.; Park, D.; Cho, M.L.; Kim, H. Potent inhibition of acetylcholinesterase by sargachromanol I from Sargassum siliquastrum and by selected natural compounds. Bioorg. Chem. 2019, 89, 103043. [Google Scholar] [CrossRef]
- Baek, S.C.; Park, M.H.; Ryu, H.W.; Lee, J.P.; Kang, M.G.; Park, D.; Park, C.M.; Oh, S.R.; Kim, H. Rhamnocitrin isolated from Prunus padus var. seoulensis: A potent and selective reversible inhibitor of human monoamine oxidase A. Bioorg. Chem. 2019, 83, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Asad, M.H.H.B.; Maity, S.; Zada, W.; Rizvanov, A.A.; Iqbal, J.; Babak, B.; Hussain, I. Fluoro-benzimidazole derivatives to cure Alzheimer’s disease: In-silico studies, synthesis, structure-activity relationship and in vivo evaluation for β secretase enzyme inhibition. Bioorg Chem. 2019, 88, 102936. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Food Sci. Technol. Lebensm Wiss Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Oh, J.M.; Kang, M.G.; Hong, A.; Park, J.E.; Kim, S.H.; Lee, J.P.; Baek, S.C.; Park, D.; Nam, S.J.; Cho, M.L.; et al. Potent and selective inhibition of human monoamine oxidase-B by 4-dimethylaminochalcone and selected chalcone derivatives. Int. J. Biol. Macromol. 2019, 137, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Gan, Z.-S.; Wang, Q.-Q.; Li, J.-H.; Wang, X.-L.; Wang, Y.-Z.; Du, H.-H. Iron reduces M1 macrophage polarization in RAW264.7 macrophages associated with inhibition of STAT1. Mediat. Inflamm. 2017, 2017, 8570818. [Google Scholar] [CrossRef] [PubMed]
- Trott, A.J. Olson, AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Son, S.Y.; Ma, J.; Kondou, Y.; Yoshimura, M.; Yamashita, E.; Tsukihara, T. Structure of human monoamine oxidase A at 2.2- Å resolution: The control of opening the entry for substrates/inhibitors. Proc. Natl. Acad. Sci. USA 2008, 105, 5739–5744. [Google Scholar] [CrossRef]
- Binda, C.; Aldeco, M.; Geldenhuys, W.J.; Tortorici, M.; Mattevi, A.; Edmondson, D.E. Molecular insights into human monoamine oxidase B inhibition by the glitazone anti-diabetes drugs. ACS Med. Chem. Lett. 2011, 3, 39–42. [Google Scholar] [CrossRef]
- Mills, J.E.; Dean, P.M. Three-dimensional hydrogen-bond geometry and probability information from a crystal survey. J. Comput. Aided Mol. Des. 1996, 10, 607–622. [Google Scholar] [CrossRef]
- Phillips, J.C.; Hardy, D.J.; Maia, J.D.C.; Stone, J.E.; Ribeiro, J.V.; Bernardi, R.C.; Buch, R.; Fiorin, G.; Hénin, J.; Jiang, W.; et al. Scalable molecular dynamics on CPU and GPU architectures with NAMD. J. Chem. Phys. 2020, 153, 044130. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Vanommeslaeghe, K.; Hatcher, E.; Acharya, C.; Kundu, S.; Zhong, S.; Shim, J.; Darian, E.; Guvench, O.; Lopes, P.; Vorobyov, I.; et al. CHARMM general force field: A force field for drug-like molecules compatible with the CHARMM all-atom additive biological force fields. J. Comput. Chem. 2010, 31, 671–690. [Google Scholar] [CrossRef] [PubMed]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [PubMed]
- Gray, L.E.; Gardner, H.W.; Weisleder, D.; Leib, M. Production and toxicity of 2,3-dihydro-5-hydroxy-2-methyl-4H-1-benzopyran-4-one by Phialophora gregata. Phytochemistry 1999, 50, 1337–1340. [Google Scholar] [CrossRef]
- Nadeau, A.K.; Sorensen, J.L. Polyketides produced by Daldinia loculata cultured from Northern Manitoba. Tetrahedron Lett. 2011, 52, 1697–1699. [Google Scholar] [CrossRef]
- Kekuda, T.R.P.; Lavanya, D.; Pooja, R. Lichens as promising resources of enzyme inhibitors: A review. J. Drug Deliv. Ther. 2019, 9, 665–676. [Google Scholar]
- Okuyama, E.; Hossain, C.F.; Yamazaki, M. Monoamine oxidase inhibitors from a lichen, Solorina crocea (L.) ACH. Jpn. J. Pharmacogn. 1991, 45, 159–162. [Google Scholar]
- Kinoshita, K.; Saito, D.; Koyama, K.; Takahashi, K.; Sato, Y.; Okuyama, E.; Fujimoto, H.; Yamazaki, M. Monoamine oxidase inhibitory effects of some lichen compounds and their synthetic analogues. J. Hattori. Bot. Lab. 2002, 92, 277–284. [Google Scholar]
- Dai, J.; Krohn, K.; Flörke, U.; Draeger, S.; Schulz, B.; Kiss-Szikszai, A.; Antus, S.; Kurtán, T.; van Ree, T. Metabolites from the endophytic fungus Nodulisporium sp. from Juniperus cedre. Eur. J. Org. Chem. 2006, 15, 3498. [Google Scholar] [CrossRef]
- Wei, B.; Yang, Z.-D.; Chen, X.-W.; Zhou, S.-Y.; Yu, H.-T.; Sun, J.-Y.; Yao, X.-J.; Wang, Y.-G.; Xue, H.Y. Colletotrilactam A-D, novel lactams from Colletotrichum gloeosporioides GT-7, a fungal endophyte of Uncaria rhynchophylla. Fitoterapia 2016, 113, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Carté, B.K.; Sidebottom, P.J.; Sek Yew, A.L.; Ng, S.; Huang, Y.; Butler, M.S. Circumdatin G, a new alkaloid from the fungus Aspergillus ochraceus. J. Nat. Prod. 2001, 64, 125–126. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Sun, H.; Wang, X. Recent advances in natural products from plants for treatment of liver diseases. Eur. J. Med. Chem. 2013, 63, 570–577. [Google Scholar] [CrossRef] [PubMed]
- Kamisuki, S.; Ishimaru, C.; Onoda, K.; Kuriyama, I.; Ida, N.; Sugawara, F.; Yoshida, H.; Mizushina, Y. Nodulisporol and nodulisporone, novel specific inhibitors of human DNA polymerase lambda from a fungus, Nodulisporium sp. Bioorg. Med. Chem. 2007, 15, 3109–3114. [Google Scholar] [CrossRef] [PubMed]
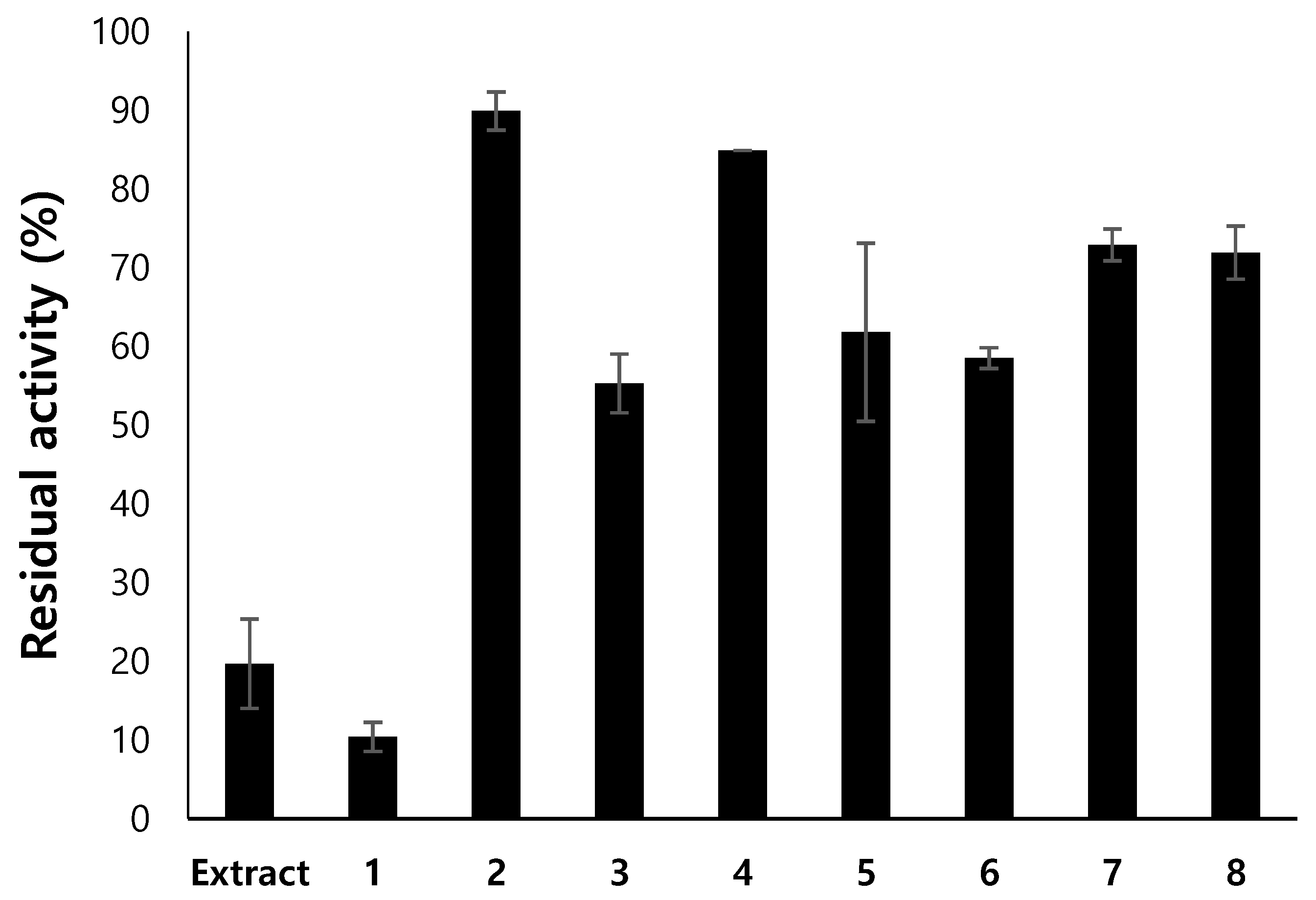
| ELF No. | Residual Activity at 20 µg/mL (%) | |||
|---|---|---|---|---|
| MAO-A | MAO-B | AChE | BChE | |
| 13 | - | 19.7 ± 5.67 | - | - |
| 22 | - | 22.2 ± 6.87 | - | - |
| 26 | - | 26.2 ± 1.40 | - | 26.3 ± 0.59 |
| 68 | 26.2 ± 5.76 | - | - | - |
| 71 | 43.6 ± 2.47 | - | - | - |
| 73 | - | 28.0 ± 1.32 | - | - |
| 74 | - | - | 41.0 ± 3.83 | - |
| 110 | - | 27.0 ± 1.16 | - | - |
| 172 | - | - | 40.8 ± 1.35 | - |
| ELF No. | Inhibition at 100 µg/mL (%) |
|---|---|
| DPPH | |
| 8 | 58.5 ± 1.94 |
| 84 | 39.0 ± 0.33 |
| 87 | 84.8 ± 0.50 |
| ELF13 | Residual Activity at 2 µg/mL (%) | % Inhibition b | ||||
|---|---|---|---|---|---|---|
| MAO-A | MAO-B | AChE | BChE | BACE-1 a | DPPH | |
| C1 | 57.1 ± 2.72 | 89.3 ± 3.68 | 81.0 ± 2.01 | 80.6 ± 1.31 | 93.4 ± 1.02 | 9.58 ± 0.29 |
| C2 | 55.8 ± 0.91 | 13.2 ± 0.48 | 73.0 ± 1.13 | 83.8 ± 5.89 | 98.6 ± 2.04 | 25.11 ± 2.12 |
| Compounds | IC50 (µM) | |||
|---|---|---|---|---|
| MAO-A | MAO-B | AChE | BChE | |
| HMC | 13.97 ± 2.07 | 3.23 ± 0.3 | >40 | >40 |
| Lazabemide | - | 0.063 ± 0.015 | - | - |
| Pargyline | - | 0.028 ± 0.0043 | - | - |
| Toloxatone | 1.08 ± 0.025 | - | - | - |
| Clorgyline | 0.0035 ± 0.00067 | - | - | - |
| Donepezil | - | - | 0.009 ± 0.0019 | 0.18 ± 0.0038 |
| Compound | GI Absorption | BBB Permeant | P-gp Substrate | CYP1A2 Inhibitor | CYP2C19 Inhibitor | CYP2C9 Inhibitor | CYP2D6 Inhibitor | CYP3A4 Inhibitor | Log Kp (Skin Permeation) |
|---|---|---|---|---|---|---|---|---|---|
| HMC | High | Yes | No | YES | No | No | No | No | −5.97 cm/s |
| Compound | Mw(g/mol) | cLog P | HBD | HBA | TPSA (Ų) | RB | Lipinski Violations |
|---|---|---|---|---|---|---|---|
| HMC | 178.18 | 1.70 | 1 | 3 | 46.53 | No | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeong, G.-S.; Kang, M.-G.; Han, S.-A.; Noh, J.-I.; Park, J.-E.; Nam, S.-J.; Park, D.; Yee, S.-T.; Kim, H. Selective Inhibition of Human Monoamine Oxidase B by 5-hydroxy-2-methyl-chroman-4-one Isolated from an Endogenous Lichen Fungus Daldinia fissa. J. Fungi 2021, 7, 84. https://doi.org/10.3390/jof7020084
Jeong G-S, Kang M-G, Han S-A, Noh J-I, Park J-E, Nam S-J, Park D, Yee S-T, Kim H. Selective Inhibition of Human Monoamine Oxidase B by 5-hydroxy-2-methyl-chroman-4-one Isolated from an Endogenous Lichen Fungus Daldinia fissa. Journal of Fungi. 2021; 7(2):84. https://doi.org/10.3390/jof7020084
Chicago/Turabian StyleJeong, Geum-Seok, Myung-Gyun Kang, Sang-Ah Han, Ji-In Noh, Jong-Eun Park, Sang-Jip Nam, Daeui Park, Sung-Tae Yee, and Hoon Kim. 2021. "Selective Inhibition of Human Monoamine Oxidase B by 5-hydroxy-2-methyl-chroman-4-one Isolated from an Endogenous Lichen Fungus Daldinia fissa" Journal of Fungi 7, no. 2: 84. https://doi.org/10.3390/jof7020084
APA StyleJeong, G.-S., Kang, M.-G., Han, S.-A., Noh, J.-I., Park, J.-E., Nam, S.-J., Park, D., Yee, S.-T., & Kim, H. (2021). Selective Inhibition of Human Monoamine Oxidase B by 5-hydroxy-2-methyl-chroman-4-one Isolated from an Endogenous Lichen Fungus Daldinia fissa. Journal of Fungi, 7(2), 84. https://doi.org/10.3390/jof7020084









