The Toxic Effects of Ppz1 Overexpression Involve Nha1-Mediated Deregulation of K+ and H+ Homeostasis
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Strains and Culture Media
2.2. Generation of Nha1 Variants
2.3. Media Acidification Tests
2.4. Preparation of Protein Extracts and Inmmunoblot
2.5. Intracellular pH Measurements
2.6. Potassium Content and Efflux Measurements
2.7. Fluorescence Microscopy
2.8. Statistics
3. Results
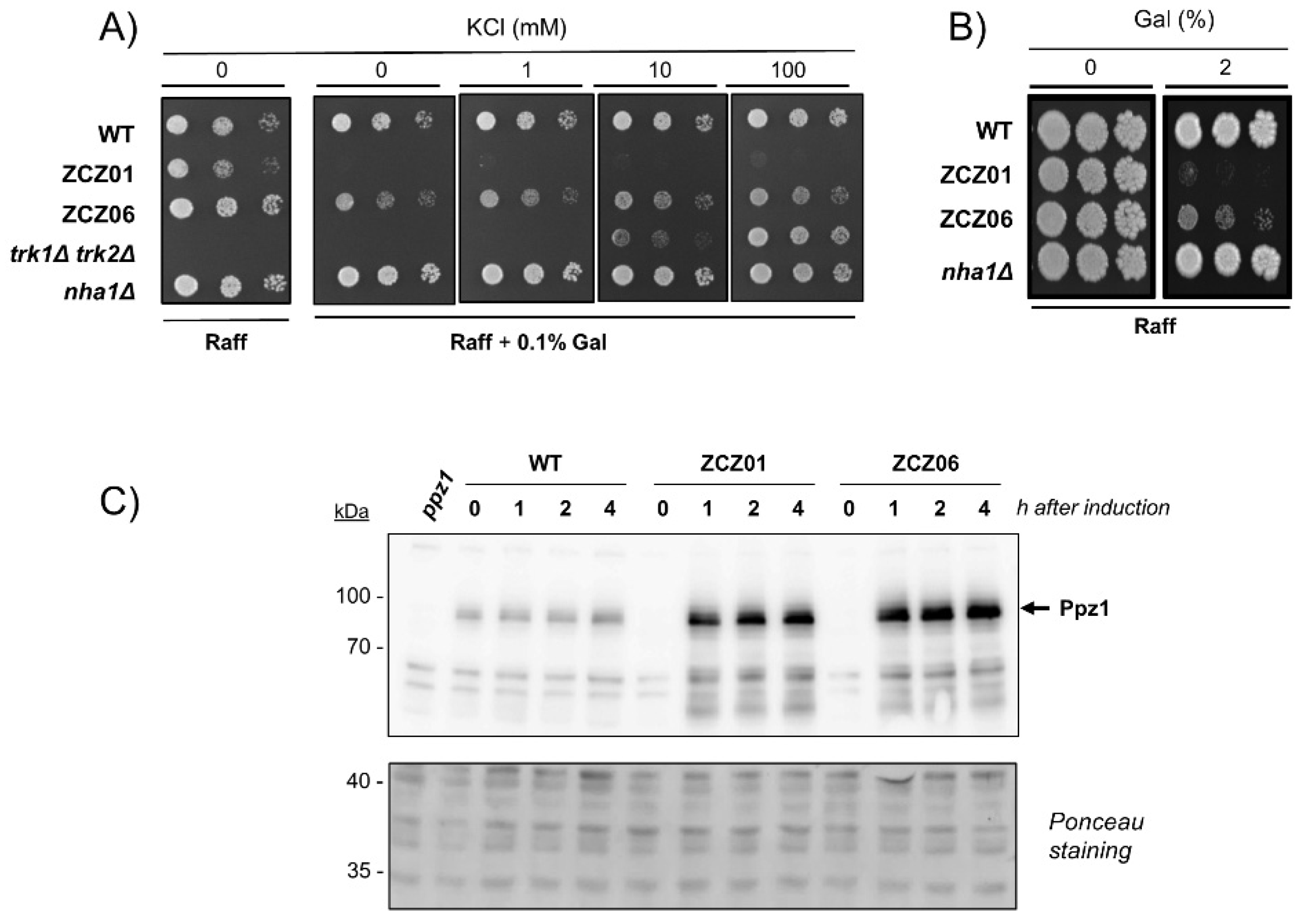
3.1. The Toxic Effects of Ppz1 Overexpression Are Supressed by Deletion of the NHA1 Antiporter Gene but Not by Potassium Supplementation
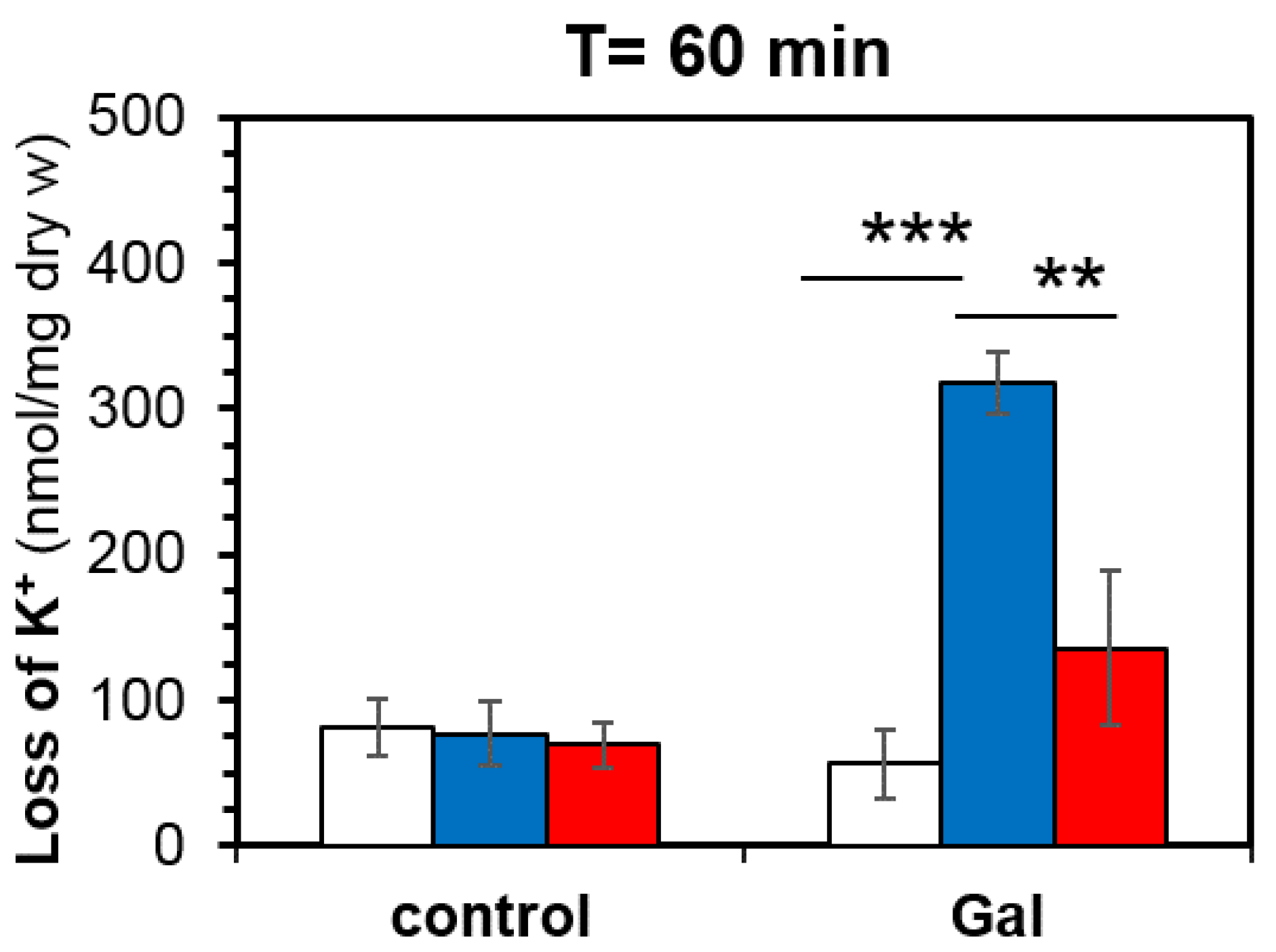
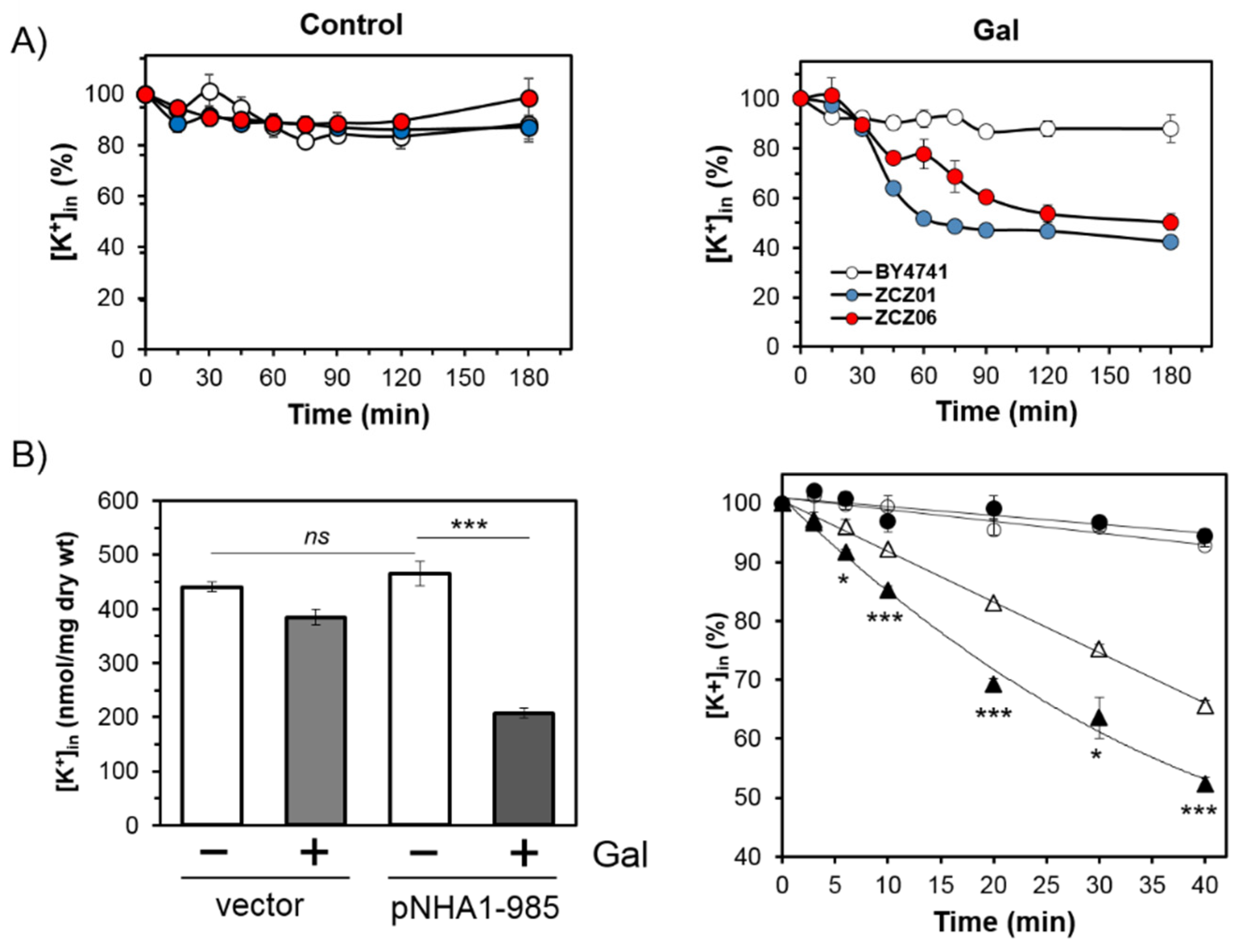
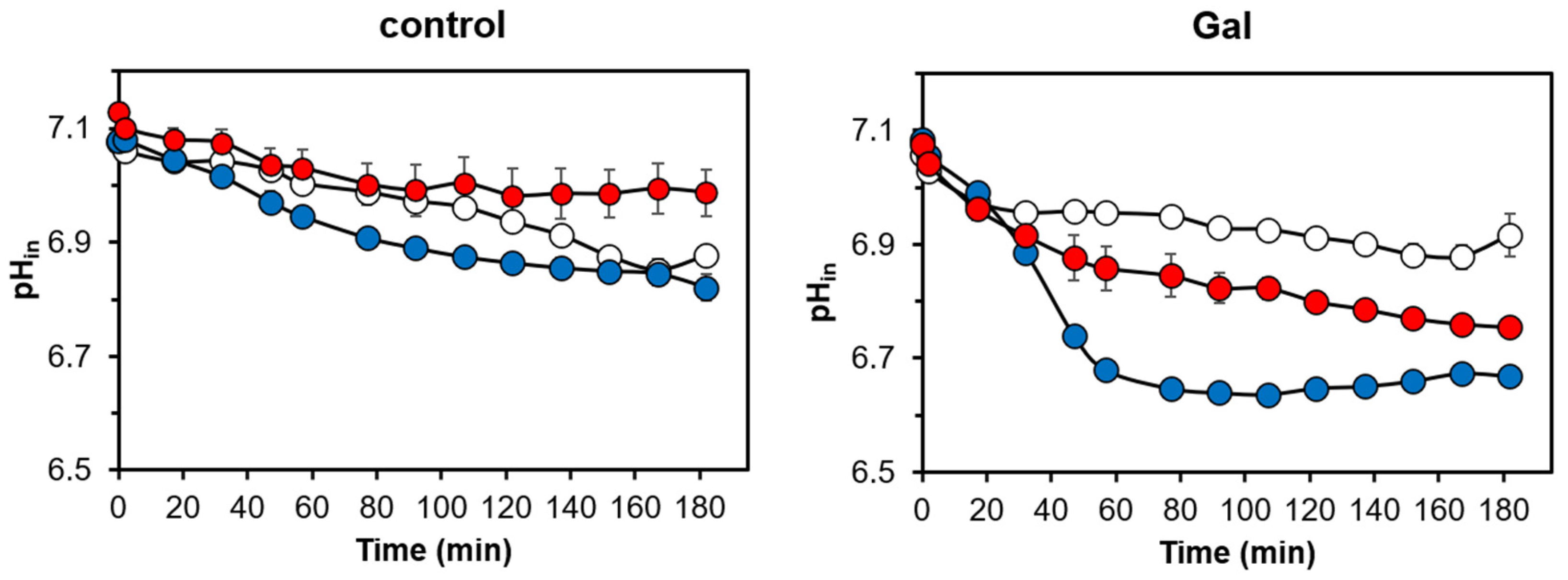
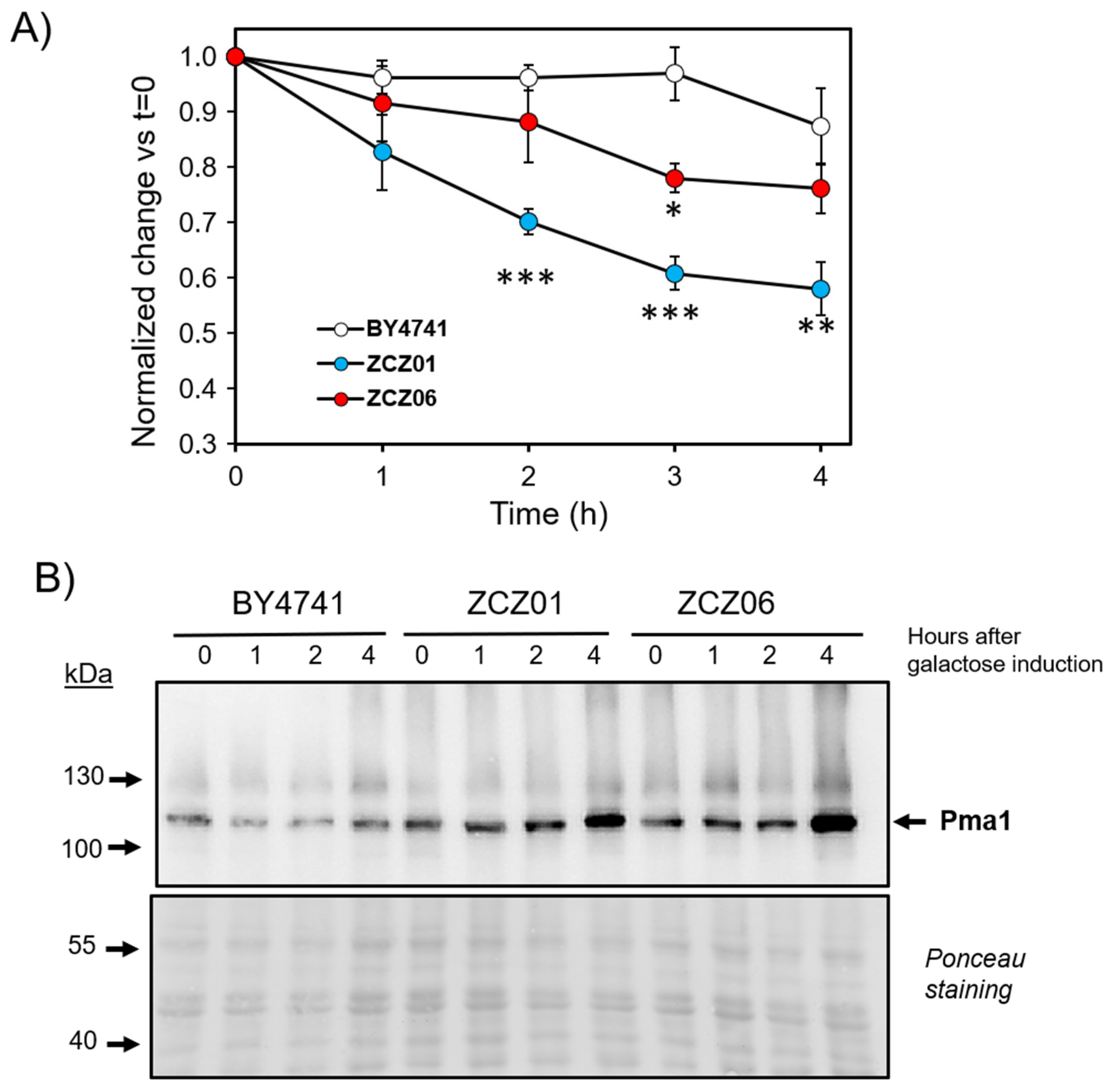
3.2. Cells Overexpressing Ppz1 Show Noticiable Loss of Intracellular Potassium Levels and Increased Intracellular Acidification
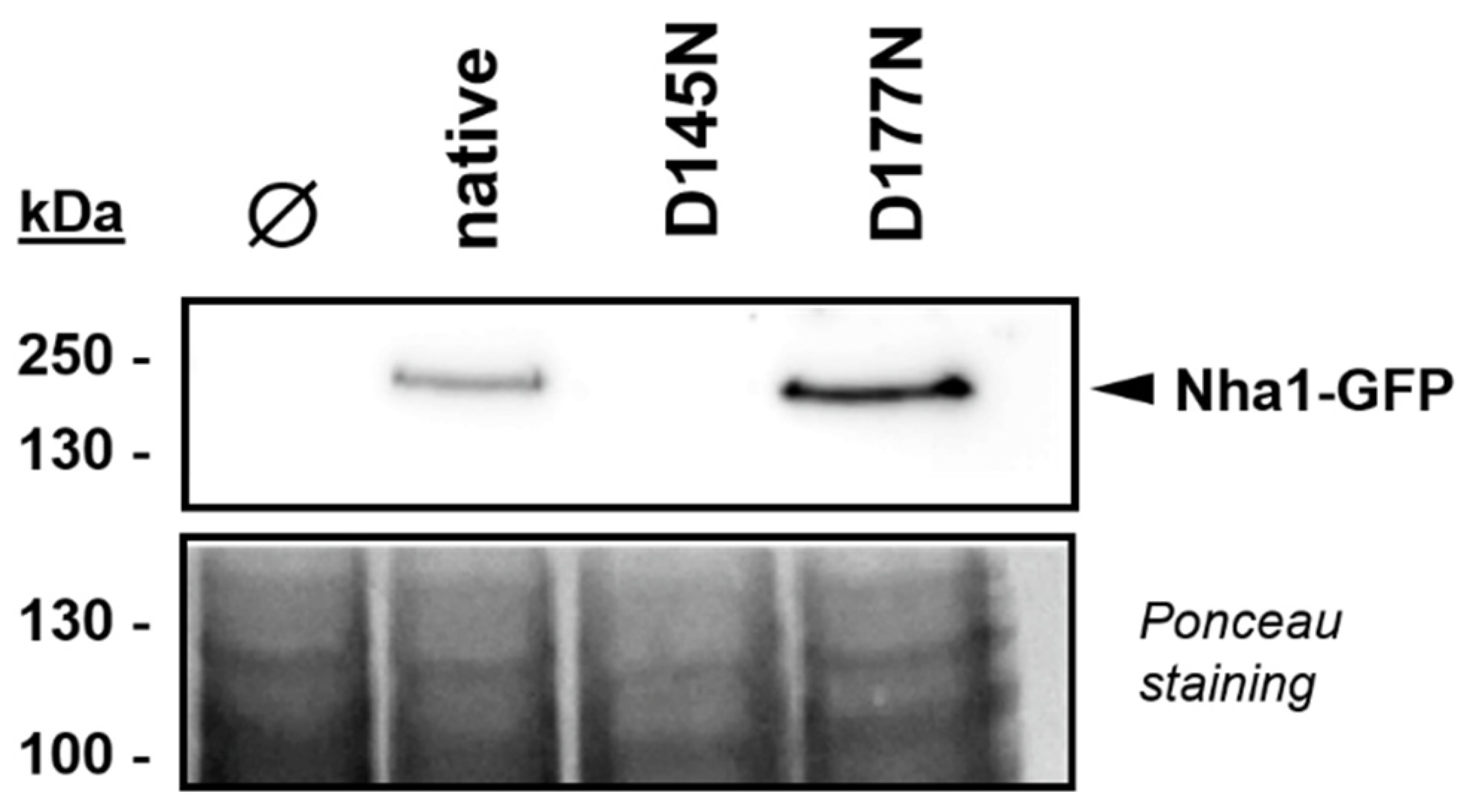
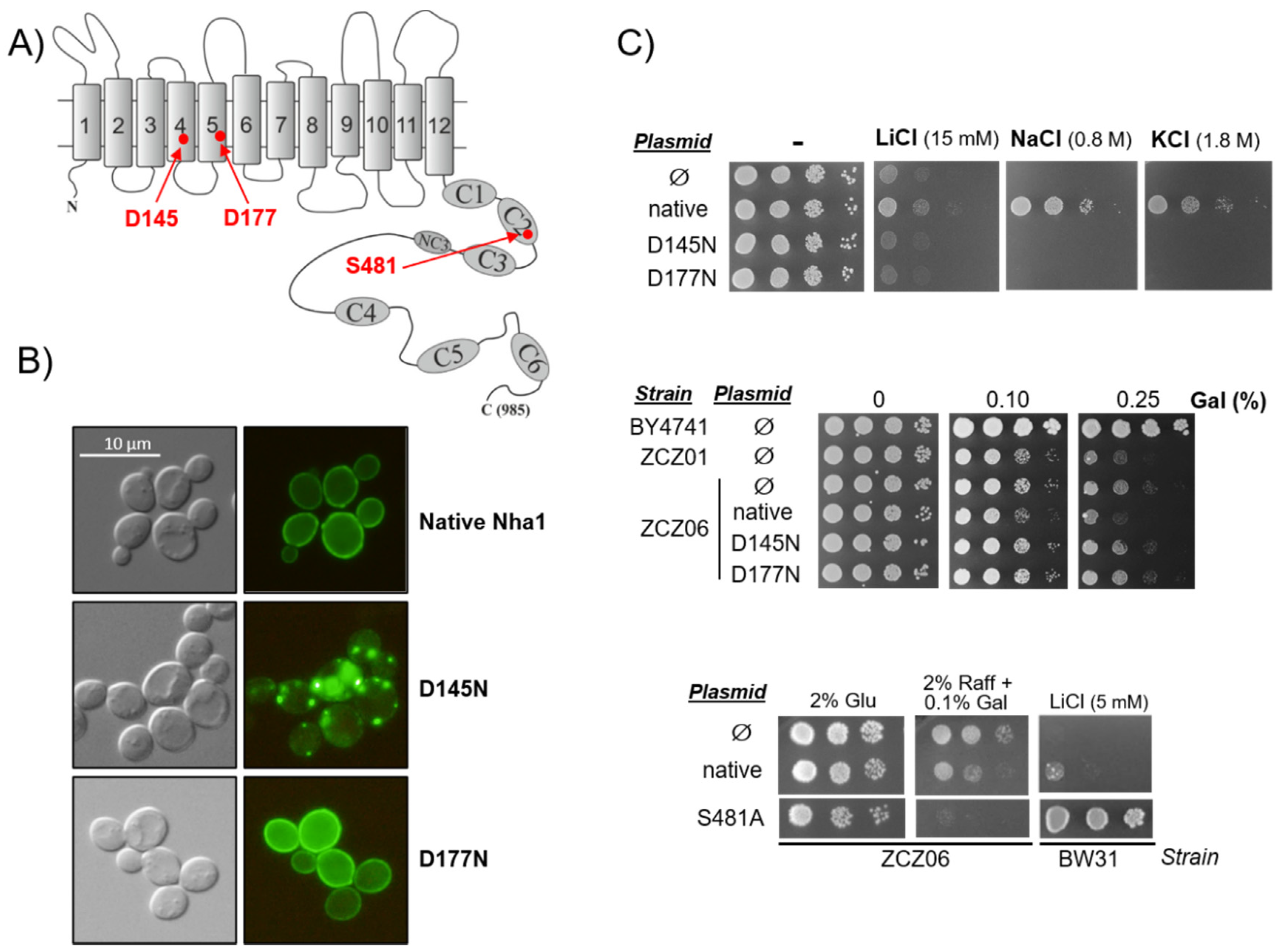
3.3. Functionally Impaired Versions of Nha1 Do Not Counteract the Effect of Nha1 Loss in Ppz1-Overexpressing Cells
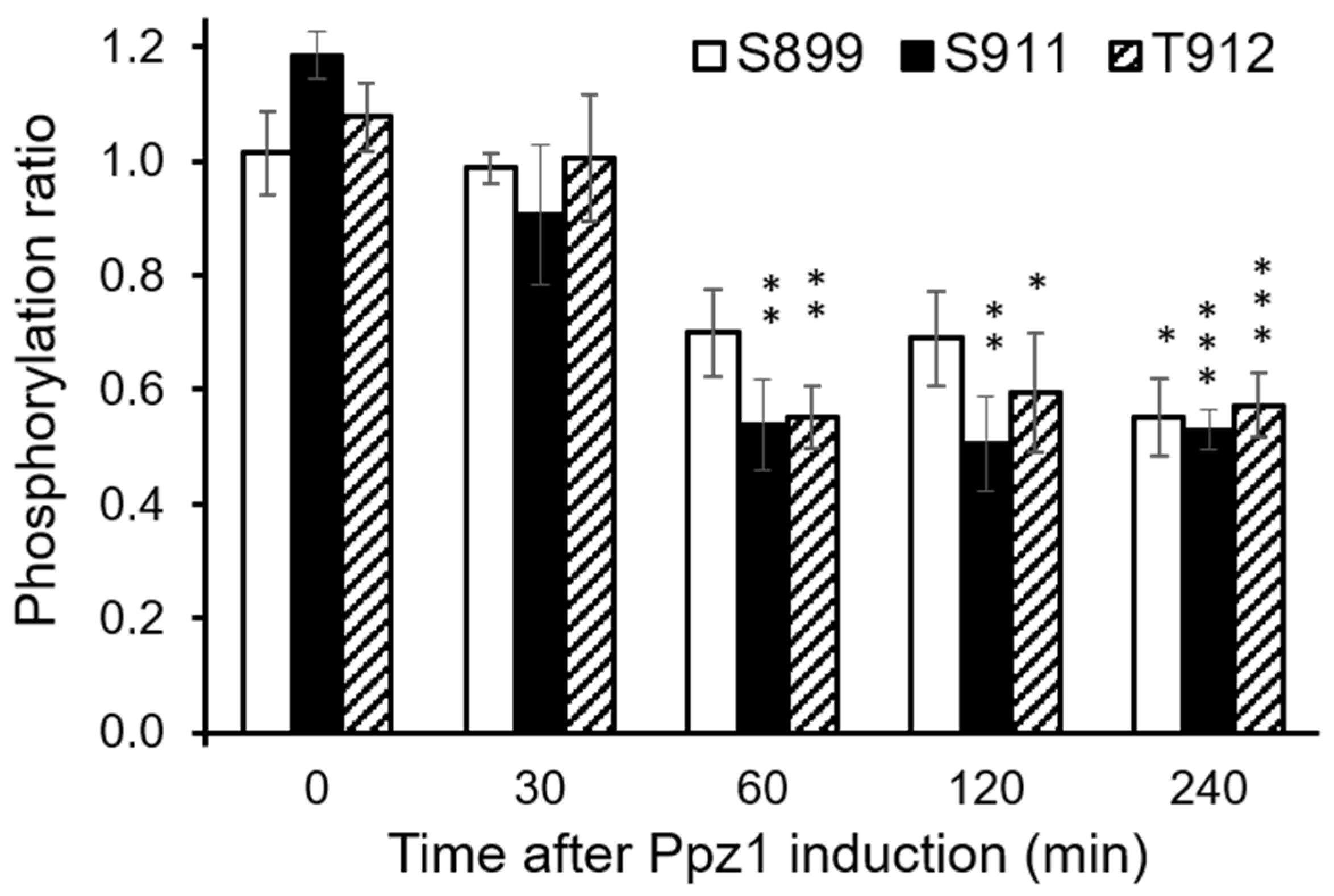
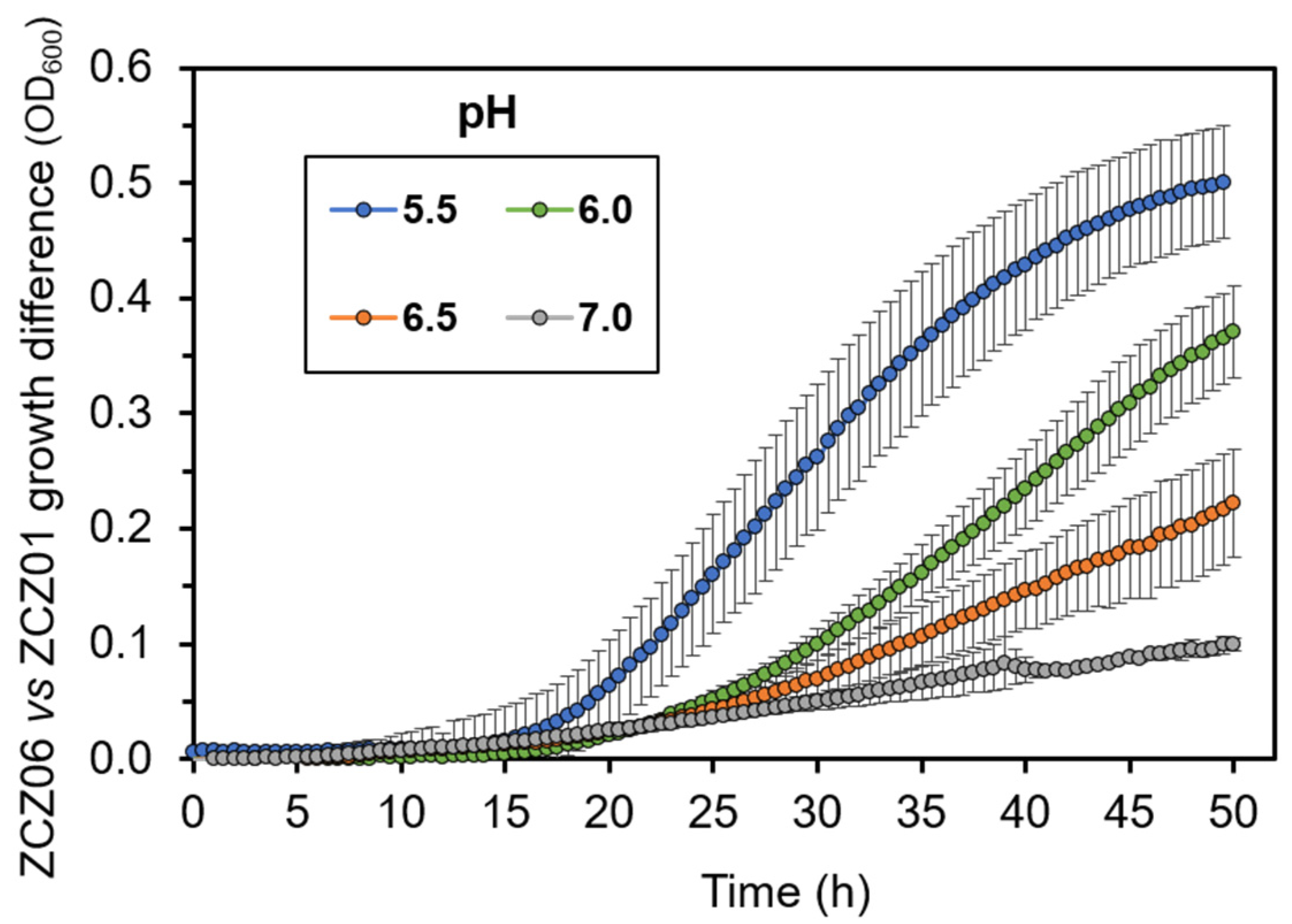
3.4. Overexpression of Ppz1 Limits the Capacity for Acidification of the Medium
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Name | Sequence * |
|---|---|
| NHA1_D145N_Fwd | 5′-CTGCGTGCATTACCGCAACAAATCCTATTCTGGCGCAGTCGG-3′ |
| NHA1_D145N_Rew | 5′-CCGACTGCGCCAGAATAGGATTTGTTGCGGTAATGCACGCAG-3′ |
| Nha1D177N-F | 5‘-TCAGGCTGCAATAATGGTATGGCCTTTC-3‘ |
| Nha1D177N-R | 5‘-GAAAGGCCATACCATTATTGCAGCCTGA-3‘ |
References
- Posas, F.; Casamayor, A.; Morral, N.; Ariño, J. Molecular cloning and analysis of a yeast protein phosphatase with an unusual amino-terminal region. J. Biol. Chem. 1992, 267, 11734–11740. [Google Scholar] [CrossRef]
- Lee, K.S.; Hines, L.K.; Levin, D.E. A pair of functionally redundant yeast genes (PPZ1 and PPZ2) encoding type 1-related protein phosphatases function within the PKC1-mediated pathway. Mol. Cell Biol. 1993, 13, 5843–5853. [Google Scholar] [PubMed]
- Ariño, J.; Velázquez, D.; Casamayor, A. Ser/thr protein phosphatases in fungi: Structure, regulation and function. Microb. Cell 2019, 6. [Google Scholar] [CrossRef]
- Offley, S.R.; Schmidt, M.C. Protein phosphatases of Saccharomyces cerevisiae. Curr. Genet. 2019, 65, 41–55. [Google Scholar] [CrossRef]
- De Nadal, E.; Clotet, J.; Posas, F.; Serrano, R.; Gomez, N.; Ariño, J. The yeast halotolerance determinant Hal3p is an inhibitory subunit of the Ppz1p Ser/Thr protein phosphatase. Proc. Natl. Acad. Sci. USA 1998, 95, 7357–7362. [Google Scholar] [CrossRef]
- Muñoz, I.; Simón, E.; Casals, N.; Clotet, J.; Ariño, J. Identification of multicopy suppressors of cell cycle arrest at the G1-S transition in Saccharomyces cerevisiae. Yeast 2003, 20, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, A.; Muñoz, I.; Serrano, R.; Gonzalez, A.; Simon, E.; Arino, J. Functional characterization of the Saccharomyces cerevisiae VHS3 gene: A regulatory subunit of the Ppz1 protein phosphatase with novel, phosphatase-unrelated functions. J. Biol. Chem. 2004, 279, 34421–34430. [Google Scholar] [CrossRef]
- Ruiz, A.; Gonzalez, A.; Munoz, I.; Serrano, R.; Abrie, J.A.; Strauss, E.; Arino, J. Moonlighting proteins Hal3 and Vhs3 form a heteromeric PPCDC with Ykl088w in yeast CoA biosynthesis. Nat. Chem. Biol. 2009, 5, 920–928. [Google Scholar] [CrossRef]
- Abrie, J.A.A.; González, A.; Strauss, E.; Ariño, J. Functional mapping of the disparate activities of the yeast moonlighting protein Hal3. Biochem. J. 2012, 442, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Clotet, J.; Posas, F.; De Nadal, E.; Arino, J. The NH2-terminal extension of protein phosphatase PPZ1 has an essential functional role. J. Biol. Chem. 1996, 271, 26349–26355. [Google Scholar] [CrossRef]
- Makanae, K.; Kintaka, R.; Makino, T.; Kitano, H.; Moriya, H. Identification of dosage-sensitive genes in Saccharomyces cerevisiae using the genetic tug-of-war method. Genome Res. 2013, 23, 300–311. [Google Scholar] [CrossRef] [PubMed]
- Clotet, J.; Garí, E.; Aldea, M.; Ariño, J. The yeast Ser/Thr phosphatases Sit4 and Ppz1 play opposite roles in regulation of the cell cycle. Mol. Cell. Biol. 1999, 19, 2408–2415. [Google Scholar] [CrossRef]
- Calafí, C.; López-Malo, M.; Velázquez, D.; Zhang, C.; Fernández-Fernández, J.; Rodríguez-Galán, O.; de la Cruz, J.; Ariño, J.; Casamayor, A. Overexpression of budding yeast protein phosphatase Ppz1 impairs translation. Biochim. Biophys. Acta. Mol. Cell Res. 2020, 1867, 118727. [Google Scholar] [CrossRef]
- Velázquez, D.; Albacar, M.; Zhang, C.; Calafí, C.; López-Malo, M.; Torres-Torronteras, J.; Martí, R.; Kovalchuk, S.I.; Pinson, B.; Jensen, O.N.; et al. Yeast Ppz1 protein phosphatase toxicity involves the alteration of multiple cellular targets. Sci. Rep. 2020, 10, 15613. [Google Scholar] [CrossRef] [PubMed]
- Ádám, C.; Erdei, É.; Casado, C.; Kovács, L.; González, A.; Majoros, L.; Petrényi, K.; Bagossi, P.; Farkas, I.; Molnar, M.; et al. Protein phosphatase CaPpz1 is involved in cation homeostasis, cell wall integrity and virulence of Candida albicans. Microbiology 2012, 158, 1258–1267. [Google Scholar] [CrossRef] [PubMed]
- Manfiolli, A.O.; de Castro, P.A.; Dos Reis, T.F.; Dolan, S.; Doyle, S.; Jones, G.; Riaño Pachón, D.M.; Ulaş, M.; Noble, L.M.; Mattern, D.J.; et al. Aspergillus fumigatus protein phosphatase PpzA is involved in iron assimilation, secondary metabolite production, and virulence. Cell. Microbiol. 2017, 19, e12770. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-Z.; Li, B.; Pan, Y.-T.; Fang, Y.-L.; Li, D.-W.; Huang, L. Protein Phosphatase CgPpz1 Regulates Potassium Uptake, Stress Responses and Plant Infection in Colletotrichum gloeosporioides. Phytopathology 2021, in press. [Google Scholar] [CrossRef]
- Posas, F.; Camps, M.; Ariño, J. The PPZ protein phosphatases are important determinants of salt tolerance in yeast cells. J. Biol. Chem. 1995, 270, 13036–13041. [Google Scholar] [CrossRef]
- Ruiz, A.; Yenush, L.; Arino, J. Regulation of ENA1 Na(+)-ATPase gene expression by the Ppz1 protein phosphatase is mediated by the calcineurin pathway. Eukaryot. Cell 2003, 2, 937–948. [Google Scholar] [CrossRef]
- Yenush, L.; Merchan, S.; Holmes, J.; Serrano, R. pH-Responsive, Posttranslational Regulation of the Trk1 Potassium Transporter by the Type 1-Related Ppz1 Phosphatase. Mol. Cell Biol. 2005, 25, 8683–8692. [Google Scholar] [CrossRef] [PubMed]
- Yenush, L.; Mulet, J.M.; Ariño, J.; Serrano, R. The Ppz protein phosphatases are key regulators of K+ and pH homeostasis: Implications for salt tolerance, cell wall integrity and cell cycle progression. EMBO J. 2002, 21, 920–929. [Google Scholar] [CrossRef]
- Merchan, S.; Bernal, D.; Serrano, R.; Yenush, L. Response of the Saccharomyces cerevisiae Mpk1 mitogen-activated protein kinase pathway to increases in internal turgor pressure caused by loss of Ppz protein phosphatases. Eukaryot. Cell 2004, 3, 100–107. [Google Scholar] [CrossRef]
- Kinclova, O.; Ramos, J.; Potier, S.; Sychrova, H. Functional study of the Saccharomyces cerevisiae Nha1p C-terminus. Mol. Microbiol. 2001, 40, 656–668. [Google Scholar] [CrossRef]
- Bañuelos, M.A.; Sychrova, H.; Bleykasten-Grosshans, C.; Souciet, J.L.; Potier, S. The Nha1 antiporter of Saccharomyces cerevisiae mediates sodium and potassium efflux. Microbiology 1998, 144, 2749–2758. [Google Scholar]
- Kinclova-Zimmermannova, O.; Gaskova, D.; Sychrova, H. The Na+, K+/H+ -antiporter Nha1 influences the plasma membrane potential of Saccharomyces cerevisiae. FEMS Yeast Res. 2006, 6, 792–800. [Google Scholar] [CrossRef][Green Version]
- Proft, M.; Struhl, K. MAP kinase-mediated stress relief that precedes and regulates the timing of transcriptional induction. Cell 2004, 118, 351–361. [Google Scholar] [CrossRef]
- Sychrova, H.; Ramirez, J.; Pena, A. Involvement of Nha1 antiporter in regulation of intracellular pH in Saccharomyces cerevisiae. FEMS Microbiol. Lett. 1999, 171, 167–172. [Google Scholar] [CrossRef][Green Version]
- Simon, E.; Clotet, J.; Calero, F.; Ramos, J.; Arino, J. A screening for high copy suppressors of the sit4 hal3 synthetically lethal phenotype reveals a role for the yeast Nha1 antiporter in cell cycle regulation. J. Biol. Chem. 2001, 276, 29740–29747. [Google Scholar] [CrossRef]
- Papouskova, K.; Moravcova, M.; Masrati, G.; Ben-Tal, N.; Sychrova, H.; Zimmermannova, O. C5 conserved region of hydrophilic C-terminal part of Saccharomyces cerevisiae Nha1 antiporter determines its requirement of Erv14 COPII cargo receptor for plasma-membrane targeting. Mol. Microbiol. 2021, 115, 41–57. [Google Scholar] [CrossRef] [PubMed]
- Kinclova-Zimmermannova, O.; Sychrova, H. Functional study of the Nha1p C-terminus: Involvement in cell response to changes in external osmolarity. Curr. Genet. 2006, 49, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Smidova, A.; Stankova, K.; Petrvalska, O.; Lazar, J.; Sychrova, H.; Obsil, T.; Zimmermannova, O.; Obsilova, V. The activity of Saccharomyces cerevisiae Na+, K+/H+ antiporter Nha1 is negatively regulated by 14-3-3 protein binding at serine 481. Biochim. Biophys. Acta-Mol. Cell Res. 2019, 1866. [Google Scholar] [CrossRef] [PubMed]
- Adams, A.; Gottschling, D.E.; Kaiser, C.A.; Stearns, T. Methods in Yeast Genetics: A Cold Spring Harbor Laboratory Course Manual; Cold Spring Harbor Laboratory Press: Suffolk County, NY, USA, 1998; ISBN 0879695080. [Google Scholar]
- Navarrete, C.; Petrezsélyová, S.; Barreto, L.; Martínez, J.L.; Zahrádka, J.; Ariño, J.; Sychrová, H.; Ramos, J. Lack of main K+ uptake systems in Saccharomyces cerevisiae cells affects yeast performance in both potassium-sufficient and potassium-limiting conditions. FEMS Yeast Res. 2010, 10, 508–517. [Google Scholar] [CrossRef]
- Barreto, L.; Canadell, D.; Petrezsélyová, S.; Navarrete, C.; Maresová, L.; Peréz-Valle, J.; Herrera, R.; Olier, I.; Giraldo, J.; Sychrová, H.; et al. A genomewide screen for tolerance to cationic drugs reveals genes important for potassium homeostasis in Saccharomyces cerevisiae. Eukaryot. Cell 2011, 10, 1241–1250. [Google Scholar] [CrossRef]
- Hanscho, M.; Ruckerbauer, D.E.; Chauhan, N.; Hofbauer, H.F.; Krahulec, S.; Nidetzky, B.; Kohlwein, S.D.; Zanghellini, J.; Natter, K. Nutritional requirements of the BY series of Saccharomyces cerevisiae strains for optimum growth. FEMS Yeast Res. 2012, 12, 796–808. [Google Scholar] [CrossRef] [PubMed]
- Maresova, L.; Hoskova, B.; Urbankova, E.; Chaloupka, R.; Sychrova, H. New applications of pHluorin--measuring intracellular pH of prototrophic yeasts and determining changes in the buffering capacity of strains with affected potassium homeostasis. Yeast 2010, 27, 317–325. [Google Scholar] [CrossRef]
- Canadell, D.; González, A.; Casado, C.; Ariño, J. Functional interactions between potassium and phosphate homeostasis in Saccharomyces cerevisiae. Mol. Microbiol. 2015, 95, 555–572. [Google Scholar] [CrossRef] [PubMed]
- Kinclova-Zimmermannova, O.; Zavrel, M.; Sychrova, H. Identification of conserved prolyl residue important for transport activity and the substrate specificity range of yeast plasma membrane Na+/H+ antiporters. J Biol.Chem. 2005, 280, 30638–30647. [Google Scholar] [CrossRef]
- Mahmoud, S.; Planes, M.D.; Cabedo, M.; Trujillo, C.; Rienzo, A.; Caballero-Molada, M.; Sharma, S.C.; Montesinos, C.; Mulet, J.M.; Serrano, R. TOR complex 1 regulates the yeast plasma membrane proton pump and pH and potassium homeostasis. FEBS Lett. 2017, 591, 1993–2002. [Google Scholar] [CrossRef]
- Horak, J.; Wolf, D.H. Glucose-induced monoubiquitination of the Saccharomyces cerevisiae galactose transporter is sufficient to signal its internalization. J. Bacteriol. 2001, 183, 3083–3088. [Google Scholar] [CrossRef]
- Zimmermannova, O.; Salazar, A.; Sychrova, H.; Ramos, J. Zygosaccharomyces rouxii Trk1 is an efficient potassium transporter providing yeast cells with high lithium tolerance. FEMS Yeast Res. 2015, 15. [Google Scholar] [CrossRef]
- Ariño, J.; Ramos, J.; Sychrova, H. Monovalent cation transporters at the plasma membrane in yeasts. Yeast 2019, 36, 177–193. [Google Scholar] [CrossRef]
- Mitsui, K.; Yasui, H.; Nakamura, N.; Kanazawa, H. Oligomerization of the Saccharomyces cerevisiae Na+/H+ antiporter Nha1p: Implications for its antiporter activity. Biochim. Biophys. Acta 2005, 1720, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Kinclova-Zimmermannova, O.; Falson, P.; Cmunt, D.; Sychrova, H. A hydrophobic filter confers the cation selectivity of Zygosaccharomyces rouxii plasma-membrane Na+/H+ antiporter. J. Mol. Biol. 2015, 427, 1681–1694. [Google Scholar] [CrossRef] [PubMed]
- Banuelos, M.A.; Rodriguez-Navarro, A. P-type ATPases mediate sodium and potassium effluxes in Schwanniomyces occidentalis. J. Biol. Chem. 1998, 273, 1640–1646. [Google Scholar] [CrossRef]
- Zahradka, J.; van Heusden, G.P.; Sychrova, H. Yeast 14-3-3 proteins participate in the regulation of cell cation homeostasis via interaction with Nha1 alkali-metal-cation/proton antiporter. Biochim. Biophys. Acta 2012, 1820, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Tarassov, K.; Messier, V.; Landry, C.R.; Radinovic, S.; Serna Molina, M.M.; Shames, I.; Malitskaya, Y.; Vogel, J.; Bussey, H.; Michnick, S.W. An in vivo map of the yeast protein interactome. Science 2008, 320, 1465–1470. [Google Scholar] [CrossRef]
- Kane, P.M. Proton Transport and pH Control in Fungi. Adv. Exp. Med. Biol 2016, 892, 33–68. [Google Scholar] [PubMed]
- Eraso, P.; Portillo, F. Molecular mechanism of regulation of yeast plasma membrane H(+)-ATPase by glucose. Interaction between domains and identification of new regulatory sites. J. Biol. Chem. 1994, 269, 10393–10399. [Google Scholar] [CrossRef]
- Lecchi, S.; Nelson, C.J.; Allen, K.E.; Swaney, D.L.; Thompson, K.L.; Coon, J.J.; Sussman, M.R.; Slayman, C.W. Tandem phosphorylation of Ser-911 and Thr-912 at the C terminus of yeast plasma membrane H+-ATPase leads to glucose-dependent activation. J. Biol. Chem. 2007, 282, 35471–35481. [Google Scholar] [CrossRef] [PubMed]
- Burgstaller, W. Transport of small ions and molecules through the plasma membrane of filamentous fungi. Crit. Rev. Microbiol. 1997, 23, 1–46. [Google Scholar] [CrossRef]
- Kjellerup, L.; Gordon, S.; Cohrt, K.O.; Brown, W.D.; Fuglsang, A.T.; Winther, A.M.L. Identification of antifungal H+-ATPase inhibitors with effect on plasma membrane potential. Antimicrob. Agents Chemother. 2017, 61, e00032-17. [Google Scholar] [CrossRef] [PubMed]
- Olsen, L.F.; Andersen, A.Z.; Lunding, A.; Brasen, J.C.; Poulsen, A.K. Regulation of glycolytic oscillations by mitochondrial and plasma membrane H+-ATPases. Biophys. J. 2009, 96, 3850–3861. [Google Scholar] [CrossRef] [PubMed]
- Stevens, H.C.; Nichols, J.W. The proton electrochemical gradient across the plasma membrane of yeast is necessary for phospholipid flip. J. Biol. Chem. 2007, 282, 17563–17567. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albacar, M.; Sacka, L.; Calafí, C.; Velázquez, D.; Casamayor, A.; Ariño, J.; Zimmermannova, O. The Toxic Effects of Ppz1 Overexpression Involve Nha1-Mediated Deregulation of K+ and H+ Homeostasis. J. Fungi 2021, 7, 1010. https://doi.org/10.3390/jof7121010
Albacar M, Sacka L, Calafí C, Velázquez D, Casamayor A, Ariño J, Zimmermannova O. The Toxic Effects of Ppz1 Overexpression Involve Nha1-Mediated Deregulation of K+ and H+ Homeostasis. Journal of Fungi. 2021; 7(12):1010. https://doi.org/10.3390/jof7121010
Chicago/Turabian StyleAlbacar, Marcel, Lenka Sacka, Carlos Calafí, Diego Velázquez, Antonio Casamayor, Joaquín Ariño, and Olga Zimmermannova. 2021. "The Toxic Effects of Ppz1 Overexpression Involve Nha1-Mediated Deregulation of K+ and H+ Homeostasis" Journal of Fungi 7, no. 12: 1010. https://doi.org/10.3390/jof7121010
APA StyleAlbacar, M., Sacka, L., Calafí, C., Velázquez, D., Casamayor, A., Ariño, J., & Zimmermannova, O. (2021). The Toxic Effects of Ppz1 Overexpression Involve Nha1-Mediated Deregulation of K+ and H+ Homeostasis. Journal of Fungi, 7(12), 1010. https://doi.org/10.3390/jof7121010









