The Effect of Monochromatic LED Light Wavelengths and Photoperiods on Botrytis cinerea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungal Isolate
2.2. Light Treatments
2.3. B. cinerea Cultivation Parameters
2.4. Evaluation of the Morphological and Phenotypic Characteristics
2.5. Statistical Analysis
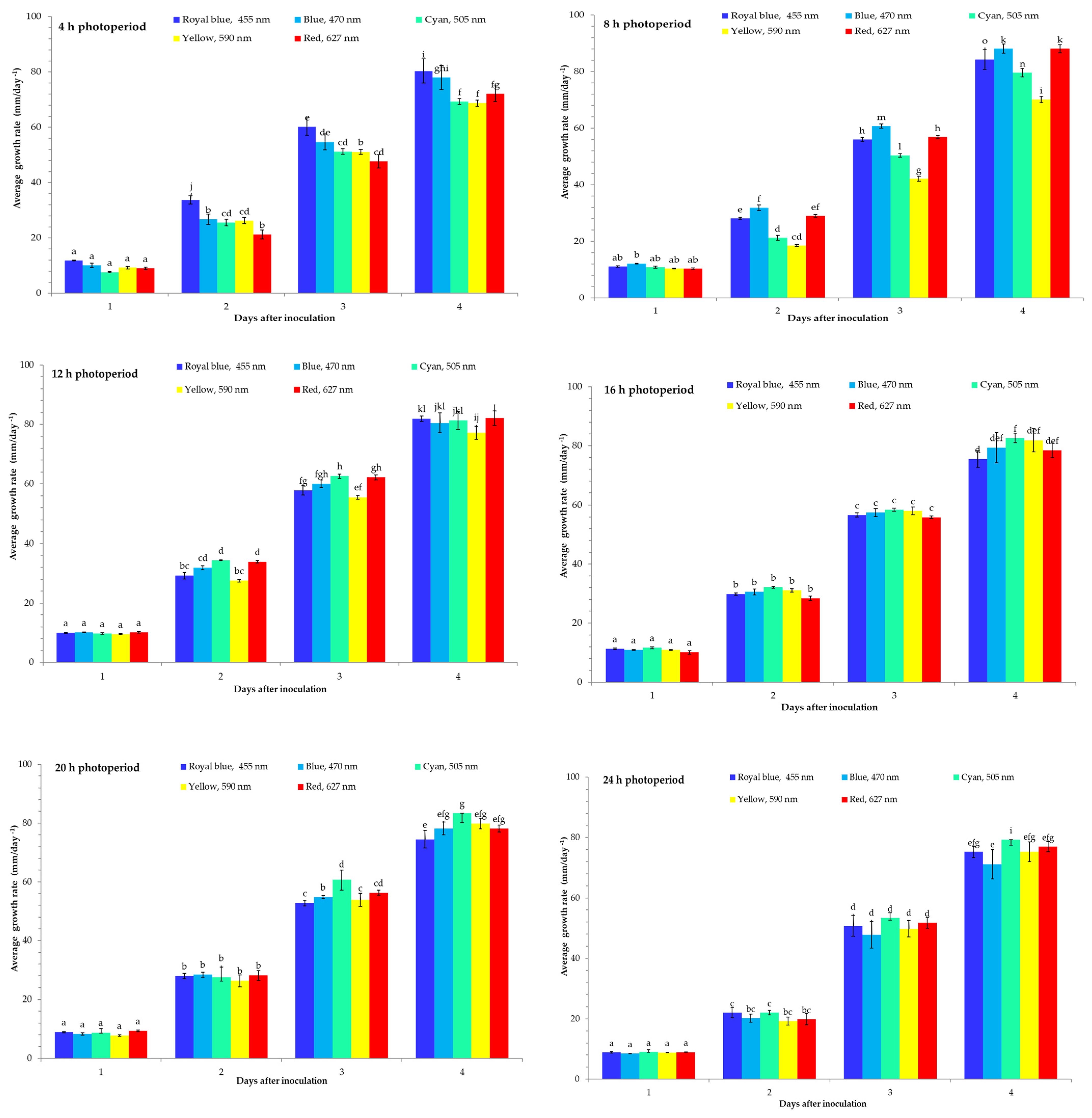
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Elad, Y.; Vivier, M.; Fillinger, S. Botrytis, the good, the bad and the ugly. In Botrytis–the Fungus, the Pathogen and Its Management in Agricultural Systems; Fillinger, S., Elad, Y., Eds.; Springer: New York, NY, USA; Dordrecht, The Netherlands; London, UK, 2016; pp. 1–15. [Google Scholar]
- Carisse, O. Epidemiology and Aerobiology of Botrytis spp. In Botrytis–the Fungus, the Pathogen and its Management in Agricultural Systems; Fillinger, S., Elad, Y., Eds.; Springer: New York, NY, USA; Dordrecht, The Netherlands; London, UK, 2016; pp. 127–148. [Google Scholar]
- Hua, L.; Yong, C.; Zhanquan, Z.; Boqiang, L.; Guozheng, Q.; Shipping, T. Pathogenic mechanisms and control strategies of Botrytis cinerea causing post-harvest decay in fruits and vegetables. Food Qual. Safe 2018, 2, 111–119. [Google Scholar] [CrossRef] [Green Version]
- Rasiukevičiūtė, N.; Rugienius, R.; Šikšnianienė, J.B. Genetic diversity of Botrytis cinerea from strawberry in Lithuania. Zemdirb.-Agric. 2018, 105, 265–270. [Google Scholar] [CrossRef] [Green Version]
- Shtienberg, D. Rational management of Botrytis-incited diseases: Integration of control measures and use of warning systems. In Botrytis: Biology, Pathology and Control; Elad, Y., Williamson, B., Tudzynski, P., Delen, N., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 335–347. [Google Scholar]
- Elad, Y. Cultural and integrated control of Botrytis. In Botrytis–the Fungus, the Pathogen and Its Management in Agricultural Systems; Fillinger, S., Elad, Y., Eds.; Springer: New York, NY, USA; Dordrecht, The Netherlands; London, UK, 2016; pp. 149–164. [Google Scholar]
- Fung, F.; Wang, H.S.; Menon, S. Food safety in the 21st century. Biomed. J. 2018, 41, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Rather, I.A.; Koh, W.Y.; Paek, W.K.; Lim, J. The Sources of chemical contaminants in food and their health implications. Front. Pharm. 2017, 8, 830. [Google Scholar] [CrossRef] [Green Version]
- Tiilikkala, K.; Lindqvist, I.; Hagner, M.; Setälä, H.; Perdikis, D. Use of botanical pesticides in modern plant protection. In Pesticides in the Modern World-Pesticides Use and Management; Stoytcheva, M., Ed.; IntechOpen Limited: UK, London, 2011; pp. 259–272. [Google Scholar]
- Stentiford, G.D.; Becnel, J.; Weiss, L.M.; Keeling, P.J.; Didier, E.S.; Williams, B.P.; Bjornson, S.; Kent, M.L.; Freeman, M.A.; Brown, M.J.F.; et al. Microsporidia-Emergent Pathogens in the Global Food Chain. Trends Parasitol 2016, 32, 336–348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carisse, O.; Morissette-Thomas, V.; Van der Heyden, H. Lagged association between powdery mildew leaf severity, airborne inoculum, weather, and crop losses in strawberry. Phytopathology 2013, 103, 811–821. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.; Kook, H.-S.; Jang, Y.-J.; Lee, W.-H.; Kamala-Kannan, S.; Chae, J.-C.; Lee, K.-J. The effect of blue-light emitting diodes on antioxidant properties and resistance to Botrytis cinerea in tomato. J. Plant Pathol. Microbiol. 2013, 4, 203. [Google Scholar] [CrossRef] [Green Version]
- Kook, H.S.; Park, S.H.; Jang, Y.J.; Lee, G.W.; Kim, J.S.; Kim, H.M.; Oh, B.T.; Chae, J.C.; Lee, K.J. Blue LED (light-emitting diodes)-mediated growth promotion and control of Botrytis disease in lettuce. Acta Agric. Scand. B Soil Plant Sci. 2013, 63, 271–277. [Google Scholar] [CrossRef]
- Hasan, M.M.; Bashir, T.; Ghosh, R.; Lee, S.K.; Bae, H. An Overview of LEDs’ effects on the production of bioactive compounds and crop quality. Molecules 2017, 22, 1420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luksiene, Z.; Rasiukeviciute, N.; Zudyte, B.; Uselis, N. Innovative approach to sunlight activated biofungicides for strawberry crop protection: ZnO nanoparticles. J. Photoch. Photobio. B 2019, 203, 111656. [Google Scholar] [CrossRef]
- Neugart, S.; Schreiner, M. UVB and UVA as eustressors in horticultural and agricultural crops. Sci. Hort. 2018, 234, 370–381. [Google Scholar] [CrossRef]
- Morkeliūnė, A.; Rasiukevičiūtė, N.; Valiuškaitė, A. Pathogenicity of Colletotrichum acutatum to different strawberry cultivars and anthracnose control with essential oils. Zemdirb.-Agric. 2021, 108, 173–180. [Google Scholar] [CrossRef]
- Šernaitė, L.; Valiuškaitė, A.; Rasiukevičiūtė, N.; Dambrauskienė, E.; Viškelis, P. Effectiveness of mixtures and individual plant extracts and essential oils for biocontrol of Botrytis cinerea. Zemdirb.-Agric. 2020, 107, 147–152. [Google Scholar] [CrossRef]
- D’Souza, C.; Yuk, H.; Khoo, H.G.; Zhou, W. Application of light-emitting diodes in food production, postharvest preservation, and microbiological food safety. Compr. Rev. Food Sci. Food Saf. 2015, 14, 719–740. [Google Scholar] [CrossRef]
- Bian, Z.H.; Yang, Q.C.; Liu, W.K. Effects of light quality on the accumulation of phytochemicals in vegetables produced in controlled environments: A review. J. Sci. Food Agric. 2015, 95, 869–877. [Google Scholar] [CrossRef]
- Vaštakaitė, V.; Viršilė, A.; Brazaitytė, A.; Samuolienė, G.; Jankauskienė, J.; Novičkovas, A.; Duchovskis, P. Pulsed Light-Emitting diodes for a higher phytochemical level in microgreens. J. Agric. Food Chem. 2017, 65, 6529–6534. [Google Scholar] [CrossRef] [PubMed]
- Brazaitytė, A.; Viršilė, A.; Samuolienė, G.; Vaštakaitė-Kairienė, V.; Jankauskienė, J.; Miliauskienė, J.; Novičkovas, A.; Duchovskis, P. Response of mustard microgreens to different wavelengths and durations of UV-A LEDs. Front. Plant Sci. 2019, 10, 1153. [Google Scholar] [CrossRef] [Green Version]
- Bantis, F.; Smirnakou, S.; Ouzounis, T.; Koukounaras, A.; Ntagkas, N.; Radoglou, K. Current status and recent achievements in the field of horticulture with the use of light-emitting diodes (LEDs). Sci. Hortic. 2018, 235, 437–451. [Google Scholar] [CrossRef]
- Yu, Z.; Fischer, R. Light sensing and responses in fungi. Nat. Rev. Microbiol. 2019, 17, 25–36. [Google Scholar] [CrossRef]
- Schumacher, J. How light affects the life of Botrytis. Fungal Genet. Biol. 2017, 106, 26–41. [Google Scholar] [CrossRef]
- Ballaré, C.L. Light Regulation of Plant Defense. Annu. Rev. Plant Biol. 2014, 65, 335–363. [Google Scholar] [CrossRef]
- Liao, H.-L.; Alferez, F.; Burns, J.K. Assessment of blue light treatments on citrus postharvest diseases. Postharvest Biol. Technol. 2013, 81, 81–88. [Google Scholar] [CrossRef]
- Wang, H.; Jiang, Y.P.; Yu, H.J.; Xia, X.J.; Shi, K.; Zhou, Y.H.; Yu, J.Q. Light quality affects incidence of powdery mildew, expression of defence-related genes and associated metabolism in cucumber plants. Eur. J. Plant Pathol. 2010, 127, 125–135. [Google Scholar] [CrossRef]
- Alferez, F.; Liao, H.-L.; Burns, J.K. Blue light alters infection by Penicillium digitatum in tangerines. Postharvest Biol. Technol. 2012, 63, 11–15. [Google Scholar] [CrossRef]
- Xu, H.; Fu, Y.; Li, T.; Wang, R. Effects of different LED light wavelengths on the resistance of tomato against Botrytis cinerea and the corresponding physiological mechanisms. J. Integr. Agr. 2017, 16, 106–114. [Google Scholar] [CrossRef] [Green Version]
- Dėnė, L.; Valiuškaitė, A. Sensitivity of Botrytis cinerea isolates complex to plant extracts. Molecules 2021, 26, 4595. [Google Scholar] [CrossRef]
- Cherkupally, R.; Kota, R.; Amballa, H.; Reddy, B.N. In vitro antifungal potential of plant extracts against Fusarium oxysporum, Rhizoctonia solani and Macrophomina phaseolina. Ann. Plant Sci. 2017, 6, 1676–1680. [Google Scholar] [CrossRef] [Green Version]
- Campbell, C.L.; Madden, L.V. Introduction to Plant Disease Epidemiology; John Wiley & Sons: New York, NY, USA, 1990; p. 532. [Google Scholar]
- Tanović, B.; Hrustić, J.; Mihajlović, M.; Grahovac, M.; Delibašić, G. Botrytis cinerea in raspberry in Serbia I: Morphological and molecular characterization. Pestic. Phytomed. (Belgrade) 2014, 29, 237–247. [Google Scholar] [CrossRef]
- Suthaparan, A.; Torre, S.; Stensvand, A.; Herrero, M.L.; Pettersen, R.I.; Gadoury, D.M.; Gislerød, H.R. Specific light-emitting diodes can suppress sporulation of Podosphaera pannosa on greenhouse roses. Plant Dis. 2010, 94, 1105–1110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alsanius, B.W.; Karlsson, M.; Rosberg, A.K.; Dorais, M.; Naznin, M.T.; Khalil, S.; Bergstrand, K.-J. Light and microbial lifestyle: The impact of light quality on plant–microbe interactions in horticultural production systems—A review. Horticulturae 2019, 5, 41. [Google Scholar] [CrossRef] [Green Version]
- Imada, K.; Tanaka, S.; Ibaraki, Y.; Yoshimura, K.; Ito, S. Antifungal effect of 405-nm light on Botrytis cinerea. Lett. Appl. Microbiol. 2014, 59, 670–676. [Google Scholar] [CrossRef]
- Yu, S.-M.; Ramkumar, G.; Lee, Y.H. Light quality influences the virulence and physiological responses of Colletotrichum acutatum causing anthracnose in pepper plants. J. Appl. Microbiol. 2014, 115, 509–516. [Google Scholar] [CrossRef]
- Canessa, P.; Schumacher, J.; Hevia, M.A.; Tudzynski, P.; Larrondo, L.F. Assessing the effects of light on differentiation and virulence of the plant pathogen Botrytis cinerea: Characterization of the white collar complex. PLoS ONE 2013, 8, e84223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, P.; Zhang, C.; Xiao, H.; Wang, Y.; Toyoda, H.; Xu, L. Exploitable regulatory effects of light on growth and development of Botrytis cinerea. J. Plant. Pathol. 2013, 95, 509–517. [Google Scholar]
| LED Light | Photoperiods | |||||
|---|---|---|---|---|---|---|
| 4 h | 8 h | 12 h | 16 h | 20 h | 24 h | |
| Royal Blue, 455 nm | 23.4 ± 1.3 | 22.6 ± 0.7 | 22.5 ± 0.6 | 21.8 ± 0.6 | 20.6 ± 0.8 | 19.7 ± 1.0 |
| Blue, 470 nm | 21.3 ± 1.4 | 24.2 ± 0.6 | 22.9 ± 0.8 | 22.4 ± 1.1 | 21.3 ± 0.6 | 18.6 ± 1.5 |
| Cyan, 505 nm | 19.3 ± 0.6 | 20.4 ± 0.5 | 23.6 ± 0.7 | 23.2 ± 0.5 | 22.7 ± 1.2 | 20.6 ± 0.5 |
| Yellow, 590 nm | 19.5 ± 0.6 | 17.8 ± 0.4 | 21.3 ± 0.6 | 22.9 ± 0.9 | 21.1 ± 0.9 | 19.3 ± 1.1 |
| Red, 627 nm | 18.9 ± 1.0 | 23.2 ± 0.5 | 23.7 ± 0.6 | 21.7 ± 0.7 | 21.6 ± 0.6 | 19.8 ± 0.8 |
| Conidia Size, μm | LED Light | ||||
|---|---|---|---|---|---|
| Royal Blue, | Blue, | Cyan, | Yellow, | Red, | |
| 455 nm | 470 nm | 505 nm | 590 nm | 627 nm | |
| 4 h photoperiod | |||||
| Width | 6.5 ± 0.3 | 6.6 ± 0.3 | 6.4 ± 0.3 | 6.6 ± 0.3 | 6.8 ± 0.3 |
| Length | 8.3 ± 0.2 | 8.9 ± 0.2 | 9.0 ± 0.6 | 8.5 ± 0.2 | 9.8 ± 0.3 |
| 8 h photoperiod | |||||
| Width | 0 ± 0 | 6.8 ± 0.2 | 6.5 ± 0.3 | 5.9 ± 0.5 | 6.6 ± 0.2 |
| Length | 0 ± 0 | 8.8 ± 0.1 | 8.9 ± 0.3 | 8.5 ± 0.4 | 8.8 ± 0.4 |
| 12 h photoperiod | |||||
| Width | 7.9 ± 0.2 | 0 ± 0 | 7.0 ± 0.0 | 7.1 ± 0.3 | 7.5 ± 0.4 |
| Length | 11.1 ± 0 | 0 ± 0 | 10.4 ± 0.5 | 9.9 ± 0.4 | 12.2 ± 0.6 |
| 16 h photoperiod | |||||
| Width | 0 ± 0 | 0 ± 0 | 6.2 ± 0.5 | 6.4 ± 0.2 | 6.2 ± 0.3 |
| Length | 0 ± 0 | 0 ± 0 | 8.9 ± 0.3 | 10.2 ± 0.1 | 9.5 ± 0.2 |
| 20 h photoperiod | |||||
| Width | 0 ± 0 | 5.7 ± 0.4 | 0 ± 0 | 6.6 ± 0.4 | 6.7 ± 0.3 |
| Length | 0 ± 0 | 8.6 ± 0.2 | 0 ± 0 | 10.4 ± 0.3 | 11.2 ± 0.2 |
| 24 h photoperiod | |||||
| Width | 0 ± 0 | 0 ± 0 | 0 ± 0 | 7.3 ± 0.2 | 6.3 ± 0.3 |
| Length | 0 ± 0 | 0 ± 0 | 0 ± 0 | 9.1 ± 0.4 | 9.4 ± 0.4 |
| LED Light | |||||
|---|---|---|---|---|---|
| Royal Blue, 455 nm | Blue, 470 nm | Cyan, 505 nm | Yellow, 590 nm | Red, 627 nm | |
| 4 h photoperiod | |||||
| Sclerotia | S1 | S3 | S0 | S3 | S2 |
| Mycelium | M2 | M2 | M2 | M2 | M2 |
| 8 h photoperiod | |||||
| Sclerotia | S2 | S1 | S3 | S3 | S1 |
| Mycelium | M1 | M2 | M2 | M2 | M2 |
| 12 h photoperiod | |||||
| Sclerotia | S3 | S0 | S3 | S4 | S4 |
| Mycelium | M2 | M1 | M2 | M2 | M2 |
| 16 h photoperiod | |||||
| Sclerotia | S1 | S0 | S3 | S3 | S3 |
| Mycelium | M1 | M1 | M2 | M2 | M2 |
| 20 h photoperiod | |||||
| Sclerotia | S0 | S0 | S0 | S0 | S0 |
| Mycelium | M1 | M2 | M1 | M2 | M2 |
| 24 h photoperiod | |||||
| Sclerotia | S2 | S0 | S1 | S1 | S0 |
| Mycelium | M1 | M1 | M1 | M2 | M2 |
| LED Light | ||||
|---|---|---|---|---|
| Royal Blue, | Blue, | Cyan, | Yellow, | Red, |
| 455 nm | 470 nm | 505 nm | 590 nm | 627 nm |
| 4 h photoperiod | ||||
| 59.8 ± 0.0 | 43.8 ± 0.3 | 59.0 ± 0.0 | 48.2 ± 3.3 | 47.1 ± 7.5 |
| 8 h photoperiod | ||||
| 55.0 ± 1.0 | 44.8 ± 0.2 | 44.5 ± 2.0 | 43.8 ± 1.3 | 41.5 ± 3.5 |
| 12 h photoperiod | ||||
| 51.5 ± 1.0 | 46.8 ± 2.8 | 50.0 ± 5.0 | 47.3 ± 1.8 | 46.8 ± 1.8 |
| 16 h photoperiod | ||||
| 66.5 ± 5.7 | 51.5 ± 1.8 | 45.0 ± 5.5 | 60.5 ± 5.6 | 60.0 ± 1.0 |
| 20 h photoperiod | ||||
| 71.0 ± 3.5 | 58.3 ± 4.7 | 70.5 ± 1.8 | 75.0 ± 8.5 | 64.0 ± 1.0 |
| 24 h photoperiod | ||||
| 72.8 ± 0.7 | 84.0 ± 0.2 | 67.5 ± 4.0 | 65.8 ± 1.8 | 66.0 ± 1.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rasiukevičiūtė, N.; Brazaitytė, A.; Vaštakaitė-Kairienė, V.; Kupčinskienė, A.; Duchovskis, P.; Samuolienė, G.; Valiuškaitė, A. The Effect of Monochromatic LED Light Wavelengths and Photoperiods on Botrytis cinerea. J. Fungi 2021, 7, 970. https://doi.org/10.3390/jof7110970
Rasiukevičiūtė N, Brazaitytė A, Vaštakaitė-Kairienė V, Kupčinskienė A, Duchovskis P, Samuolienė G, Valiuškaitė A. The Effect of Monochromatic LED Light Wavelengths and Photoperiods on Botrytis cinerea. Journal of Fungi. 2021; 7(11):970. https://doi.org/10.3390/jof7110970
Chicago/Turabian StyleRasiukevičiūtė, Neringa, Aušra Brazaitytė, Viktorija Vaštakaitė-Kairienė, Asta Kupčinskienė, Pavelas Duchovskis, Giedrė Samuolienė, and Alma Valiuškaitė. 2021. "The Effect of Monochromatic LED Light Wavelengths and Photoperiods on Botrytis cinerea" Journal of Fungi 7, no. 11: 970. https://doi.org/10.3390/jof7110970
APA StyleRasiukevičiūtė, N., Brazaitytė, A., Vaštakaitė-Kairienė, V., Kupčinskienė, A., Duchovskis, P., Samuolienė, G., & Valiuškaitė, A. (2021). The Effect of Monochromatic LED Light Wavelengths and Photoperiods on Botrytis cinerea. Journal of Fungi, 7(11), 970. https://doi.org/10.3390/jof7110970









