Immunomodulatory Effects of Edible and Medicinal Mushrooms and Their Bioactive Immunoregulatory Products
Abstract
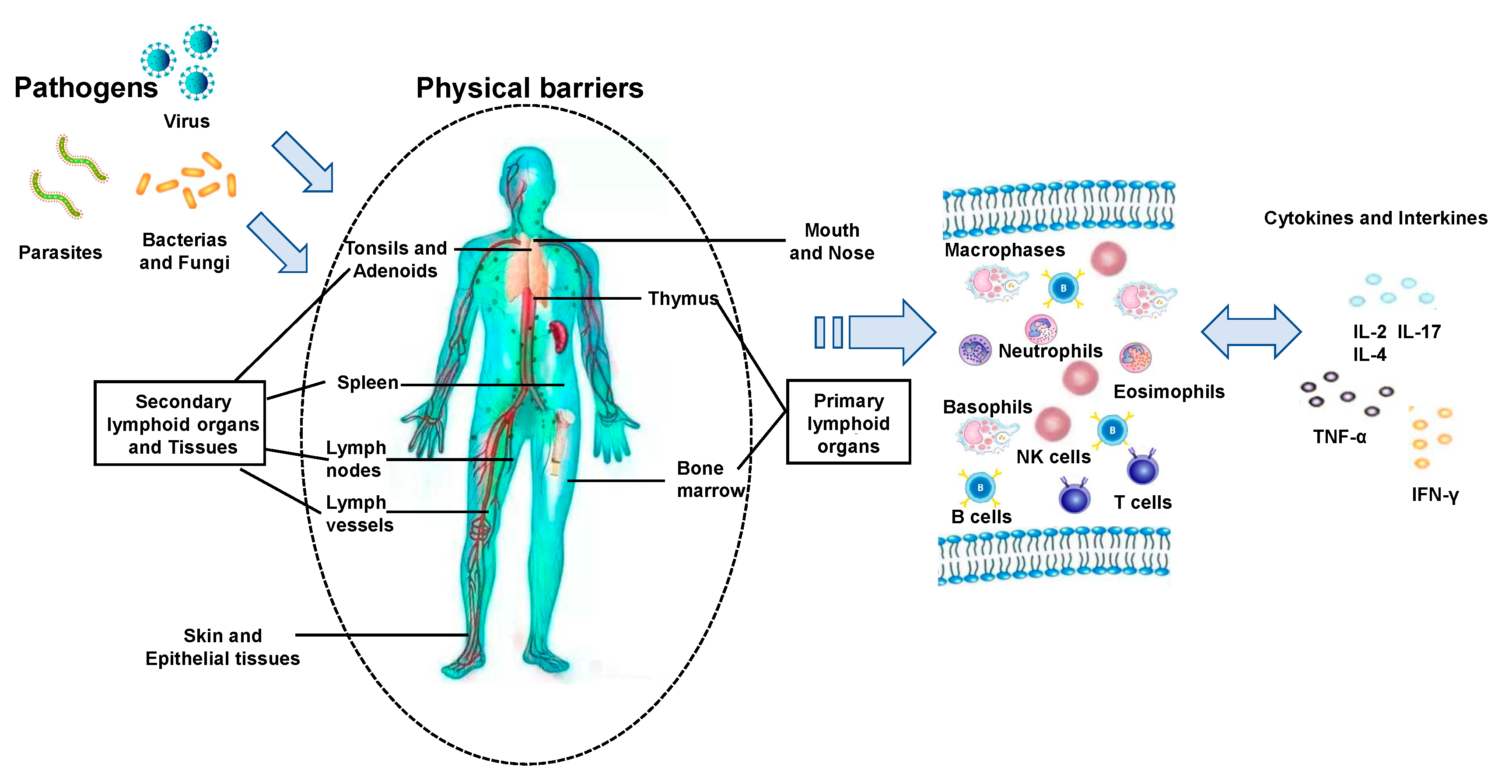
1. Introduction
2. Medicinal Mushrooms
3. Immunomodulatory Compounds and Commercial Products of Medicinal Mushrooms
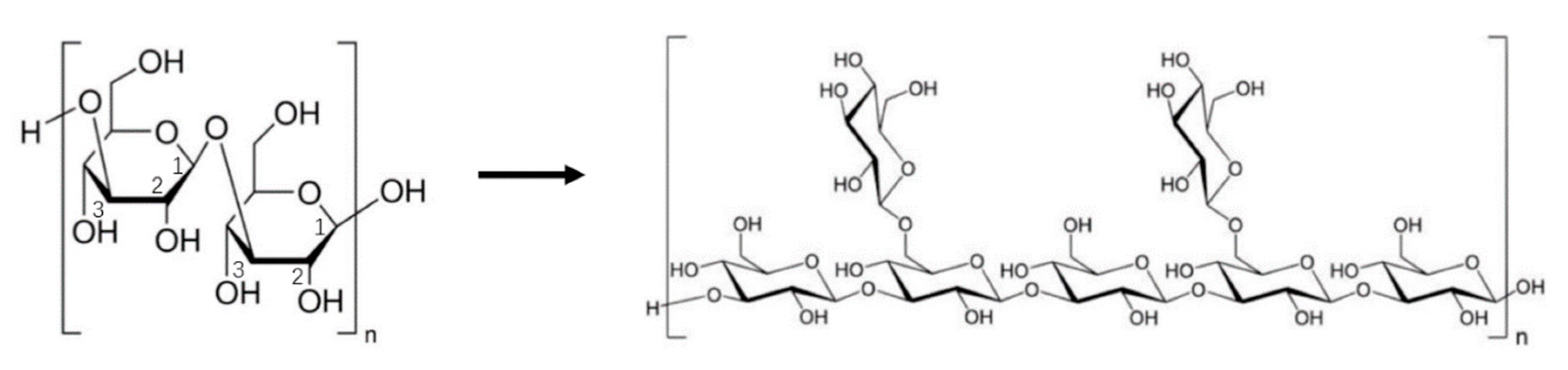
3.1. Polysaccharides
| Source | Active Compound | Immunomodulatory Effect | Refs |
|---|---|---|---|
| Agaricus blazei (syn. Agaricus brasiliensis) | Heteroglycan, Glycoprotein, Glucomannan-protein complex, β-1,3-d-glucan, with β-1,6-d-glucan branch | Stimulates Natural Killer (NK) cells, macrophages, dendritic cells, and granulocytes; induction of Tumor Necrosis Factor (TNF), Interferon (IFN)-γ, and Interleukin (IL)-8 production | [29] |
| Auricularia auricula-judae | AF1 β-1,3-d-glucan main chain with two β-1,6-d-glucosyl residues | Induces apoptosis of cancer cell | [30] |
| Gymnopus dryophilus (syn. Collybia dryophila) | β-d-glucan | Inhibits NO production in activated macrophages | [31] |
| Ophiocordyceps sinensis | β-d-glucan, heteroglycan, cordyglucan | Increase in IL-5 induction with decrease in IL-4 and IL-17 | [32] |
| Cryptoporus volvatus | β-1,3-d-Glucan | Decreases in TLR2 and activate NF-κB | [33] |
| Flammulina velutipes | Glycoprotein, Flammulina velutipes peptidoglycan (FVP), β-1,3-d-glucan | Increases NO, IL-1 production, and TNF-α secretion | [34] |
| Ganoderma lucidum | Ganoderan, Heteroglycan, mannoglucan, glycopeptide | Stimulates TNF-α, IL-1, IFN-γ production, activate NF-κB. | [35] |
| Grifola frondosa | Grifolan (1–6-monoglucosyl-branched β-1,3-d-glucan), proteoglycan, heteroglycan, galactomannan | Macrophage activation, induction of IL-1, IL-6, and TNF-α secretion | [36] |
| Hericium erinaceus | Heteroglycan, heteroglycan-peptide, β-1,3 branched-β-1,2-mannan | Induces NO production, increase expression of TNF-α, IL-1β, IL-12 | [15] |
| Inonotus obliquus | β-d-glucan | Enhance expression of IL-1β, IL-6, TNF-α, and inducible nitric oxide synthase (iNOS) in macrophages | [37] |
| Lentinula edodes (syn. Lentinus edodes) | Lentinan, glucan, mannoglucan, proteoglycan, β-(1-6)-d-glucan, α-(1-3)-d-glucan | Induces non-specific cytotoxicity in macrophage and enhance cytokine production Induces cytotoxic effect on a breast cancer cell line | [38,39,40] |
| Lentinus squarrosulus | Glucan | Activation of macrophages, splenocytes and thymocytes | [41] |
| Morchella esculenta | Galactomannan, β-1,3-d-glucan | Macrophage activation, activate NF-κB | [42] |
| Morchella conica | Galactomannan | Induces NO, IL-1β, IL-6 production | [43] |
| Naematelia aurantialba (syn. Tremella aurantialba) | Heteroglycan | Enhances mouse spleen lymphocyte proliferation | [44] |
| Pleurotus sp. ‘Florida’ (syn. Pleurotus florida) | α-1,6-glucan and α-1, 3-, β-1,6-d-glucan | Stimulates macrophages, splenocytes and thymocytes | [45,46] |
| Pleurotus ostreatus | Pleuran, heterogalactan, proteoglycan | Induces IL-4 and IFN-γ production | [47] |
| Poria cocos | β-pachyman, β-Glucan, β-1,3-d-glucan, α-1, 3-d-glucan | Promotes the immune reaction; increases the expression of cytokines | [48,49] |
| Sarcodon aspratus | Fucogalactan, 1,6-α-d-glucopyranosyl residue | Increases the release of TNF-α and NO in macrophage | [50] |
| Schizophyllum commune | Schizophyllan, 1,6-monoglucosyl branched β-1, 3-d-glucan | Activation of T cell, increases interleukin, and TNF-α production | [51] |
| Sparassis crispa | β-Glucan | Enhances IL-6 and INF-γ production | [52] |
| Taiwanofungus camphoratus (syn. Antrodia camphorate) | β-1,3-d-Gluco-pyranans with β-1,6-d-glucosyl branches, proteoglycan | Induction of INF-γ, TNF-α | [53] |
| Tropicoporus linteus (syn. Phellinus linteus) | Acidic polysaccharides | Activation of murine B cells, Induces IL-12 and IFN-γ production, Blocks NF-κB, TNF-α, IL-1α, IL-1β, and IL-4 production | [54] |
| Trametes versicolor | Polysaccharide peptide Krestin (PSK), β-1,3-glycosidic bond with β-1,6-glycosidic branches | Increases the expression of cytokines; stimulates the macrophage phagocytes | [1,55] |
| Tremella fuciformis | Acidic glucuronoxylomannan α-1,3-d-mannan backbone with β-linked D-glucuronic acid | Induces human monocytes to express interleukins | [56,57] |
| Macrocybe gigantea (syn. Tricholoma giganteum) | Polysaccharide-protein complex (PSPC) | Increases phagocytic function of macrophages by activating macrophages to release mediators such as NO and TNF-α and inhibits S180 and HL-60 cells | [13,27] |
| Xylaria nigripes | β-Glucan | Inhibits NO, IL-1β, IL-6, TNF-α, and IFN-γ production | [58] |
3.2. Mushroom Proteins and Protein–Conjugate Complexes
| Source | Lectin name | Immunomodulatory effect | Refs |
|---|---|---|---|
| Agaricus bisporus | Agaricus bisporus lectin (ABL) | Stimulate mice splenocytes mitogenicity and inhibit proliferation of L1210 and HT-29 cells | [70,71] |
| Agrocybe aegerita | Agrocybe aegerita lectin (AAL) | Inhibit proliferation of 4T1, HeLa, SW480 SGC7901, MGC803, BGC823, HL-60 and S180 cells | [72,73] |
| Amanita phalloides | - | Inhibit proliferation of L1210 cells | [74] |
| Floccularia luteovirens (syn. Armillaria luteovirens) | Armillaria luteovirens lectin (ALL) | Stimulate mice splenocytes mitogenicity and inhibit proliferation of L1210, Mouse myeloma MBL2 and HeLa cells | [75] |
| Boletus edulis | Boletus edulis lectin (BEL) | Stimulate mice splenocytes mitogenicity and inhibit proliferation of human hepatocyte carcinoma G2 (HepG2) and HT-29 cells | [76] |
| Boletus speciosus | Boletus speciosus hemagglutinin (BSH) | Inhibit proliferation of HepG2 and L1210 cells | [77] |
| Clitocybe nebularis | Clitocybe nebularis lectin (CNL) | Inhibit proliferation of human leukemic T cells | [78] |
| Flammulina velutipes | Flammulina velutipes agglutinin (FVA) | Stimulate mice splenocytes mitogenicity and inhibit proliferation of L1210 cells | [79] |
| Ganoderma capense | - | Stimulate mice splenocytes mitogenicity and inhibit proliferation of L1210, M1, HepG2 cells | [62] |
| Grifola frondosa | Grifola frondosa lectin (GFL) | Inhibit proliferation of HeLa | [80] |
| Hericium erinaceus (Syn. Hericium erinaceum) | Hericium erinaceus agglutinin (HEA) | Inhibit proliferation of HepG2 and human breast cancer MCF7 cells | [81] |
| Kurokawa leucomelas | Kurokawa leucomelas KL-15 | Inhibit proliferation of U937 cells | [82] |
| Lactarius flavidulus | Lactarius flavidulus lectin (LFL) | Inhibit proliferation of HepG2 and L1210 cells | [83] |
| Lignosus rhinocerotis | Lignosus rhinocerotis lectin (LRL) | Inhibit proliferation of HeLa, MCF7 and A549 cells | [84] |
| Marasmius oreades | Marasmius oreades agglutinin (MOA) | Inhibit proliferation of SW480, HepG2 and NIH-3T3 cells | [85] |
| Pholiota adiposa | Pholiota adiposa lectin (PAL) | Inhibit proliferation of HepG2 and MCF7 cells | [61] |
| Pleurotus citrinopileatus | - | Stimulate mice splenocytes mitogenicity and inhibit proliferation of S180 cells | [59] |
| Pleurotus eous | Pleurotus eous lectin (PEL) | Inhibit proliferation of MCF7, K562 and HepG2 | [86] |
| Cerioporus squamosus (syn. Polyporus squamosus) | Polyporus squamosus lectin 1a (PSL1a) | Inhibit proliferation of HeLa cells | [87] |
| Pseudosperma umbrinellum (syn. Inocybe umbrinella) | Inocybe umbrinella lectin (IUL) | Inhibit proliferation of HepG2 and MCF7 cells | [60] |
| Russula delica | - | Inhibit proliferation of HepG2 and MCF7 cells | [64] |
| Russula lepida | Russula lepida lectin (RLL) | Inhibit proliferation of HepG2 and MCF7 cells | [88] |
| Schizophyllum commune | Schizophyllum commune lectin (SCL) | Stimulate mice splenocytes mitogenicity and inhibit proliferation of KB, HepG2 and S180 cells | [63,89] |
| Stropharia rugosoannulata | Stropharia rugosoannulata lectin (SRL) | Inhibit proliferation of HepG2 and L1210 cells | [90] |
| Leucocalocybe mongolica (syn. Tricholoma mongolicum) | Tricholoma mongolicum lectin 1 (TML-1), Tricholoma mongolicum lectin 2 (TML-2) | Inhibit proliferation of S180 cells | [91] |
| Volvariella volvacea | Volvariella volvacea lectin (VVL) | Inhibit proliferation of S180 cells and enhance IL-2 and IFN-γ transcriptions | [92,93] |
| Xerocomellus chrysenteron (syn. Xerocomus chrysenteron) | Xerocomus chrysenteron lectin (XCL) | Inhibit proliferation of NIH-3T3 and HeLa cells | [94] |
| Xylaria hypoxylon | Xylaria hypoxylon lectin (XHL) | Inhibit proliferation of HepG2 cells | [95] |
| FIP Name | Source | Immunomodulatory Effect | Refs |
|---|---|---|---|
| FIP-aca | Taiwanofungus camphoratus (Syn. Antrodia camphorate) | Induce expression of different cytokines (IL-1b, IL-6, IL-12, TNF-α) and chemokines (CCL3, CCL4, CCL5, CCL10) | [96] |
| FIP-cru1 | Chroogomphis rutilus | Stimulate the proliferation of murine splenocytes and enhanced the secretion of IL-2 | [97] |
| FIP-dsq (FIP-dsq2) | Dichomitus squalens | Induce apoptosis and interrupt migration of A549 cells | [98] |
| FIP-fve | Flammulina velutipes | Stimulate mitogenesis in human peripheral lymphocytes, suppress systemic anaphylaxis reaction, enhance transcription of IL-3, INF-g | [99] |
| FIP-gja | Ganoderma japonicum | - | GenBank: AAX98241 |
| FIP-gat | Ganoderma atrum | - | [100] |
| FIP-glu1 (LZ-8) | Ganoderma lucidum | Enhance transcription of IL-2, IL-3, IL-4, IFN-g, TNF-α | [101] |
| FIP-gmi | Ganoderma microsporum | Down regulation of TNF-α | [102] |
| FIP-gsi | Ganoderma sinensis | Enhance production of IL-2, IL-3, IL-4, INF-g, TNF-a | [103] |
| FIP-gts | Ganoderma tsugae | Induce cytokine secretion, cellular proliferation of human peripheral mononuclear cells (HPBMCs), enhance IFN-g expression | [104] |
| FIP-glu2 (LZ-9) | Ganoderma lucidum | Activate THP-1 macrophages and induce pro-inflammatory cytokine transcription | [105] |
| FIP-SN15 | Intrageneric shuffled library | Induce U-251 MG cells apoptosis | [106] |
| FIP-Irh | Lignosus rhinocerotis | Inhibit the proliferation of MCF7, HeLa and A549 cancer cell lines | [84] |
| FIP-pcp | Poria cocos | Enhance production of IL-1b, IL-6, IL-18, TNF-a, NO | [107] |
| FIP-ppl | Postia placenta | Stimulate mouse splenocyte cell proliferation and enhance interleukin-2 (IL-2) release, inhibit proliferation and induce apoptotic effects on gastric tumor cells (MGC823) | [108] |
| FIP-tve2 (FIP-tvc) | Trametes versicolor | Increase human peripheral blood lymphocytes, enhanced production of TNF-a, NO | [109] |
| FIP-vvo | Volvariella volvacea | Enhance expression of IL-2, IL-4, IFN-g, TNF-a | [110] |
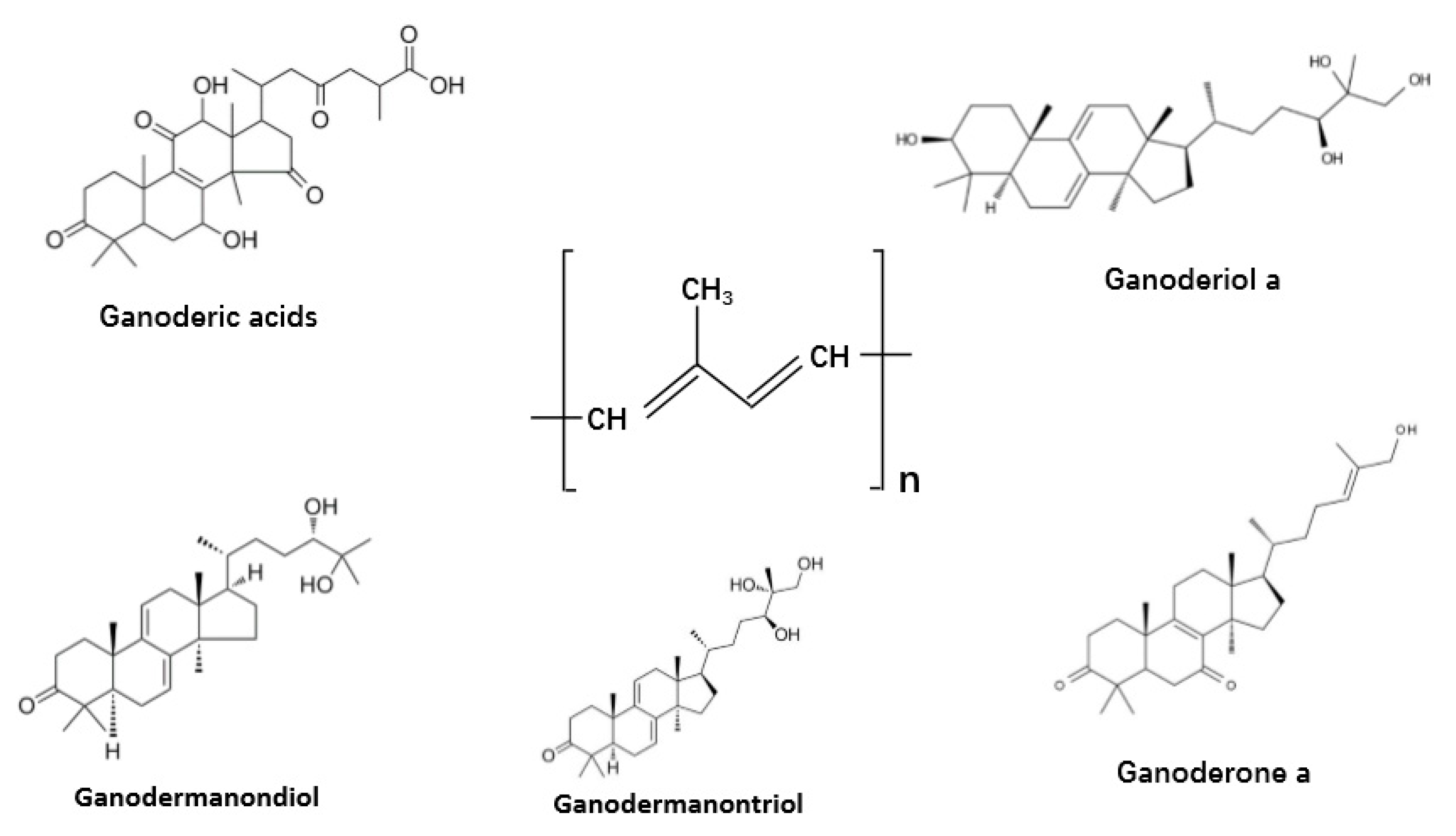
3.3. Terpenes and Terpenoids
| Type of Terpenes | Source | Compound | Immunomodulatory Effect | Refs |
|---|---|---|---|---|
| Monoterpenoids | Pleurotus cornucopiae | - | Inhibit the proliferation of HeLa and HepG2 cells | [114] |
| Sesquiterpenoids | Stereum hirsutum | - | Inhibit the proliferation of HepG2 and A549 cells | [111] |
| Inonotus rickii | 3α,6α-Hydroxycinnamolide | Inhibit the proliferation of SW480 cells | [112] | |
| Pleurotus cornucopiae | Pleurospiroketals A, B, C | Inhibit the proliferation of HeLa cells | [113] | |
| Anthracophyllum sp. BCC18695 | Anthracophyllone | Inhibit the proliferation of MCF7, NCI-H187, KB and Vero cells | [115] | |
| Flammulina velutipes | Enokipodins B, D, J 2,5-cuparadiene-1,4-dione Flammulinolides A, B, C, F | Inhibit the proliferation of HepG2, MCF7, SGC7901, KB, HeLa and A549 cells | [116,117] | |
| Neonothopanus nambi | Nambinones C | Inhibit the proliferation of NCI-H187 cells | [118] | |
| Diterpenoids | Cyathus africanus | Neosarcodonin O, 11-O-acetylcyathatriol, Cyathins H | Inhibit the proliferation of K562 and Hela cells | [119] |
| Pleurotus eryngii | Eryngiolide A | Inhibit the proliferation of Hela and HepG2 cells | [120] | |
| Sarcodon scabrosus | Sarcodonin G | Inhibit the proliferation of HOC-21, HEC-1, U251-SP, MM-1CB and HMV-1 cells | [121] | |
| Tricholoma sp. | Tricholomalide A, B, C | Inhibit the proliferation of HeLa cells | [122] | |
| Triterpenoids | Ganoderma boninense | Ganoboninketals A, B, C | Inhibit the proliferation of A549 and HeLa cells | [123] |
| Ganoderma orbiforme BCC 22324 | Ganoderic acid T and its C-3 epimer compound | Inhibit the proliferation of NCIH187, MCF7 and KB cells | [124] | |
| Ganoderma lucidum | lucialdehydes B, C, ganodermanondiol, ganodermanonol, ganoderic acid DM, ganoderic acid X | Inhibit the proliferation of T-47D, LLC, Meth-A, and Sarcoma 180 cells; Decrease the protein levels of CDK2, CDK6, p-Rb, cycle D1 and c-Myc in MCF7 cells; inhibit activity against topoisomerases I and II α and promote apoptosis | [125,126,127] | |
| Ganoderma concinna | 5α-lanosta-7,9(11),24-triene-3β-hydroxy-26-al, 5α-lanosta-7,9(11),24-triene-15α-26-dihydroxy-3-one, 8α,9α-epoxy-4,4,14α-trimethyl-3,7,11,15,20-pentaoxo-5α-pregrane | Induce apoptosis in promyelocyticleukemia HL-60 cells | [128] | |
| Ganoderma tsugae | Tsugaric acid A, 3β-hydroxy-5α-lanosta-8, 24-dien-21-oic acid | Inhibit the proliferation of HT-3, T-24, and CaSKi cells | [129] | |
| Hypholoma fasciculare (syn. Naematoloma fasciculare) | Fusciculol C, L, M, G | Inhibit the proliferation of HCT-15, SK-OV-3, SK-MEL-2 and A549 cells | [130] | |
| Astraeus odoratus | Astraodoric acids A, B, D | Inhibit the proliferation of KB, NCI-H187, and MCF7 cells | [131] | |
| Russula lepida Russula amarissima | Cucurbitane hydroxyl acid | Inhibit the proliferation of WISH, CAKI 1 and A549 cells | [132] | |
| Leucopaxillus gentianeus | Cucurbitacin B Leucopaxillone A | Inhibit the proliferation of MCF7, HepG2, kidney carcinoma CAKI-1 and A549 cells | [133] | |
| Hebeloma versipelle | 24(E)-3β-hydroxylanosta-8,24-dien-26-al-21-oic acid | Inhibit the proliferation of HL60, Bel-7402, SGC-7901 and A549 cells | [134] | |
| Tricholoma saponaceum | Saponaceol A | Inhibit the proliferation of HL-60 cells | [135] | |
| Elfvingia applanata | The methyl ester of elfvingic acid H | Inhibit the proliferation of Ehlrich and Kato III cells | [136] |
4. Immunomodulation and Other Human Health Effects of Medicinal Mushrooms
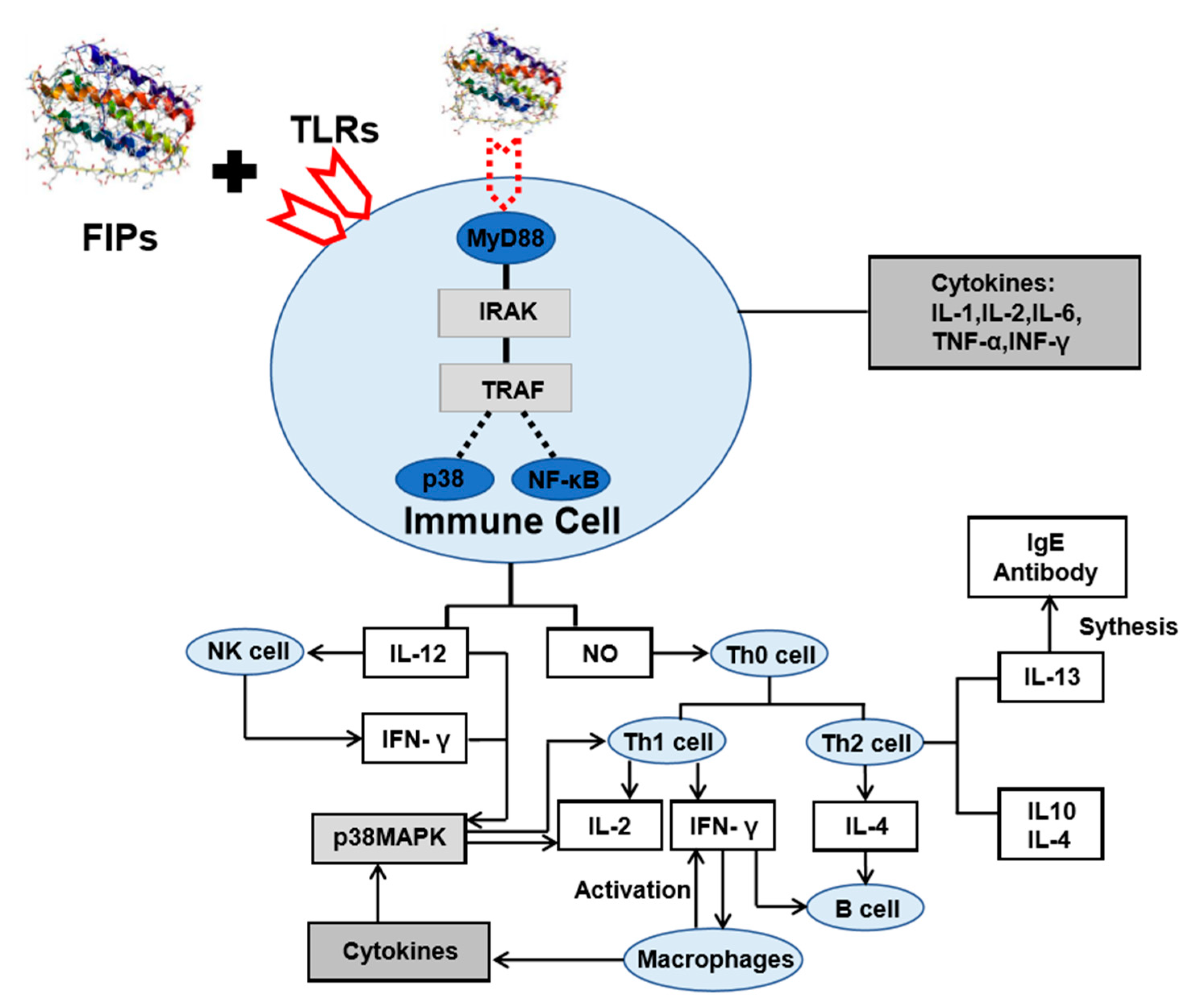
5. Mechanisms for the Immunomodulation Effects of Medicinal Mushroom Compounds
6. Relationship between Structure and Activity of Immunomodulatory Compounds from Medicinal Mushrooms
6.1. Polysaccharides
6.2. Lectins
6.3. FIPs
6.4. Terpenes and Terpenoids
7. Genomes and Molecular Techniques in the Study of Immunomodulatory Compounds in Medicinal Mushrooms
8. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- El Enshasy, H.A.; Hatti-Kaul, R. Mushroom Immunomodulators: Unique Molecules with Unlimited Applications. Trends Biotechnol. 2013, 31, 668–677. [Google Scholar] [CrossRef] [PubMed]
- Reis, F.S.; Martins, A.; Vasconcelos, M.H.; Morales, P.; Ferreira, I.C.F.R. Functional Foods Based on Extracts or Compounds Derived from Mushrooms. Trends. Food Sci. Technol. 2017, 66, 48–62. [Google Scholar] [CrossRef]
- Himanshi, R.; Shalinee, P.; Satyawati, S. Mushroom nutraceuticals for improved nutrition and better human health: A review. PharmaNutrition 2017, 5, 35–46. [Google Scholar] [CrossRef]
- Mannino, G.; Stefano, V.D.; Lauria, A.; Pitonzo, R.; Gentile, C. Vaccinium Macrocarpon (Cranberry)-Based Dietary Supplements: Variation in Mass Uniformity, Proanthocyanidin Dosage and Anthocyanin Profile Demonstrates Quality Control Standard Needed. Nutrients 2020, 12, 992. [Google Scholar] [CrossRef]
- Brya, P. Who Reads Food Labels? Selected Predictors of Consumer Interest in Front-of-Package and Back-of-Package Labels during and after the Purchase. Nutrients 2020, 12, 2605. [Google Scholar] [CrossRef]
- Shukla, S.; Bajpai, V.K.; Kim, M. Plants as Potential Sources of Natural Immunomodulators. Rev. Environ. Sci. Biol. 2014, 13, 17–33. [Google Scholar] [CrossRef]
- Wasser, S.P. Medicinal Mushroom Science: History, Current Status, Future Trends, and Unsolved Problems. Int. J. Med. Mushrooms 2010, 12, 1–16. [Google Scholar] [CrossRef]
- Davis, R.; Taylor, A.; Nally, R.; Benson, K.F.; Stamets, P.; Jensen, G.S. Differential Immune Activating, Anti-Inflammatory, and Regenerative Properties of the Aqueous, Ethanol, and Solid Fractions of a Medicinal Mushroom Blend. J. Inflamm. Res. 2020, 13, 117–131. [Google Scholar] [CrossRef]
- Chilton, J. A New Analytical Fingerprinting Method for Quality Control of Medicinal Mushroom Products. In Proceedings of the 2016 International Society for Mushroom Science, Amsterdam, The Netherlands, 30 May–2 June 2016; Sonnenberg, B., Ed.; International Society for Mushroom Science (ISMS): Amsterdam, The Netherlands, 2016; pp. 267–270. [Google Scholar]
- Dubey, S.K.; Chaturvedi, V.K.; Mishra, D.; Bajpeyee, A.; Tiwari, A.; Singh, M.P. Role of Edible Mushroom as A Potent Therapeutics for the Diabetes and Obesity. 3 Biotech 2019, 9. [Google Scholar] [CrossRef]
- Chaturvedi, V.K.; Agarwal, S.; Gupta, K.K.; Ramteke, P.W.; Singh, M.P. Medicinal Mushroom: Boon for Therapeutic Applications. 3 Biotech 2018, 8. [Google Scholar] [CrossRef]
- Wong, J.H.; Ng, T.B.; Chan, H.H.L.; Liu, Q.; Man, G.C.W.; Zhang, C.Z.; Guan, S.; Ng, C.C.W.; Fang, E.F.; Wang, H.; et al. Mushroom Extracts and Compounds with Suppressive Action on Breast Cancer: Evidence from Studies Using Cultured Cancer Cells, Tumor-Bearing Animals, and Clinical Trials. Appl. Microbiol. Biotechnol. 2020, 104, 4675–4703. [Google Scholar] [CrossRef]
- Moradali, M.F.; Mostafavi, H.; Ghods, S.; Hedjaroude, G.A. Immunomodulating and Anticancer Agents in the Realm of Macromycetes Fungi (Macrofungi). Int. Immunopharmacol. 2007, 7, 701–724. [Google Scholar] [CrossRef]
- Gao, Y.H.; Zhou, S.F.; Chen, G.L.; Dai, X.H.; Ye, J.X. A Phase I/II Study of a Ganoderma lucidum (Curt.: Fr.) P. Karst. Extract (Ganopofy) in Patients with Advanced Cancer. Int. J. Med. Mushrooms 2002, 4, 207–214. [Google Scholar] [CrossRef]
- Lee, J.S.; Cho, J.Y.; Hong, E.K. Study on Macrophage Activation and Structural Characteristics of Purified Polysaccharides from the Liquid Culture Broth of Hericium erinaceus. Carbohydr. Polym. 2009, 78, 162–168. [Google Scholar] [CrossRef]
- Riede, I. Tumor Therapy with Amanita phalloides (Death Cap): Stabilization of B-Cell Chronic Lymphatic Leukemia. J. Altern. Complement. Med. 2010, 16, 1129–1132. [Google Scholar] [CrossRef]
- Van, G.; Leo, J.L.D. Culinary-Medicinal Mushrooms: Must Action Be Taken? Int. J. Med. Mushrooms 2009, 11, 281–286. [Google Scholar] [CrossRef]
- Chakraborty, I.; Sen, I.K. Bioactive Polysaccharides from Natural Sources: A review on the Antitumor and Immunomodulating Activities. Biocatal. Agric. Biotechnol. 2019, 22, 101425. [Google Scholar] [CrossRef]
- Ina, K.; Kataoka, T.; Ando, T. The Use of Lentinan for Treating Gastric Cancer. Anti-Cancer Agent Med. Chem. 2013, 13, 681–688. [Google Scholar] [CrossRef]
- Ngwuluka, N.C.; Ochekpe, N.A.; Aruoma, O.I. Functions of Bioactive and Intelligent Natural Polymers in the Optimization of Drug Delivery. In Industrial Applications for Intelligent Polymers and Coatings; Springer International Publishing: New York, NY, USA, 2016; pp. 165–184. [Google Scholar] [CrossRef]
- Cui, J.; Chisti, Y. Polysaccharopeptides of Coriolus versicolor: Physiological Activity, Uses, and Production. J. Shanxi Med. Univ. 2003, 21, 109–122. [Google Scholar] [CrossRef]
- Oba, K.; Teramukai, S.; Kobayashi, M.; Matsui, T.; Kodera, Y.; Sakamoto, J. Efficacy of Adjuvant Immunochemotherapy with Polysaccharide K for Patients with Curative Resections of Gastric Cancer. Cancer Immunol. Immun. 2007, 56, 905–911. [Google Scholar] [CrossRef] [PubMed]
- Hattori, T.S.; Komatsu, N.; Shichijo, S.; Itoh, K. Protein-Bound Polysaccharide K Induced Apoptosis of the Human Burkitt Lymphoma Cell Line, Namalwa. Biomed. Pharm. 2004, 58, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Hirose, K.; Hakozaki, M.; Ohno, M.; Saito, Y.; Izutani, R.; Noguchi, J.; Hori, Y.; Okumoto, S.; Kuroda, D. Induction of Gene Expression for Immunomodulating Cytokines in Peripheral Blood Mononuclear Cells in Response to Orally Administered PSK, An Immunomodulating Protein-bound Polysaccharide. Cancer Immunol. Immun. 1995, 40, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Price, L.A.; Wenner, C.A.; Sloper, D.T.; Slaton, J.W.; Novack, J.P. Role for Toll-like Receptor 4 in TNF-alpha Secretion by Murine Macrophages in Response to Polysaccharide Krestin, a Trametes versicolor mushroom extract. Fitoterapia 2010, 81, 914–919. [Google Scholar] [CrossRef]
- Kidd, P.M. The Use of Mushroom Glucans and Proteoglycans in Cancer Treatment. Altern. Med. Rev. 2000, 5, 4–27. [Google Scholar]
- Ooi, V.E.C. Pharmacological Studies on Certain Mushrooms from China. Int. J. Med. Mushrooms 2001, 3, 1. [Google Scholar] [CrossRef]
- Biedron, R.; Tangen, J.M.; Maresz, K.; Hetland, G. Agaricus blazei Murill—Immunomodulatory Properties and Health Benefits. Funct. Foods Health Dis. 2012, 2, 428–447. [Google Scholar] [CrossRef]
- Firenzuoli, F.; Gori, L.; Lombardo, G. The Medicinal Mushroom Agaricus blazei Murrill: Review of Literature and Pharmaco-Toxicological Problems. Evid.-Based Complement. Altern. Med. 2008, 5, 3–15. [Google Scholar] [CrossRef]
- Ma, Z.; Wang, J.; Zhang, L.; Zhang, Y.; Ding, K. Evaluation of Water Soluble β-d-glucan from Auricularia auricular-judae as Potential Anti-tumor Agent. Carbohydr. Polym. 2010, 80, 977–983. [Google Scholar] [CrossRef]
- Pacheco-Sanchez, M.; Boutin, Y.; Angers, P.; Gosselin, A.; Tweddell, R.J. A Bioactive (1 -> 3)-, (1 -> 4)-beta-d-glucan from Collybia dryophila and Other Mushrooms. Mycologia 2006, 98, 180–185. [Google Scholar] [CrossRef]
- Chen, P.X. Properties of Cordyceps sinensis: A Review. J. Funct. Foods 2013, 5, 550–569. [Google Scholar] [CrossRef]
- Yao, H.Y.; Zhang, L.H.; Shen, J.; Shen, H.J.; Jia, Y.L.; Yan, X.F.; Xie, Q.M. Cyptoporus Polysaccharide Prevents Lipopolysaccharide-Induced Acute Lung Injury Associated with Down-regulating Toll-like Receptor 2 Expression. J. Ethnopharmacol. 2011, 137, 1267–1274. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Wang, Y.; Wang, Y.; Chen, T.; Tang, H.; Wang, M. Purification, Characterization and Immuno-Modulating Properties of Polysaccharides Isolated from Flammulina velutipes Mycelium. Am. J. Chin. Med. 2010, 38, 191–204. [Google Scholar] [CrossRef]
- Zhu, X.L.; Chen, A.F.; Lin, Z.B. Ganoderma lucidum Polysaccharides Enhance the Function of Immunological Effector Cells in Immunosuppressed Mice. J. Ethnopharmacol. 2007, 111, 219–226. [Google Scholar] [CrossRef]
- Matsui, K.; Kodama, N.; Nanba, H. Effects of Maitake (Grifola frondosa) D-Fraction on the Carcinoma Angiogenesis. Cancer Lett. 2001, 172, 193–198. [Google Scholar] [CrossRef]
- Dong, P.W.; Lee, J.S.; Kwon, D.S.; Lee, K.E.; Shin, W.C.; Hong, E.K. Immunostimulating Activity by Polysaccharides Isolated from Fruiting Body of Inonotus obliquus. Mol. Cells 2011, 31, 165–173. [Google Scholar] [CrossRef]
- Mizuno, T. The Extraction and Development of Antitumor-Active Polysaccharides from Medicinal Mushrooms in Japan (Review). Int. J. Med. Mushrooms 1999, 1, 9–29. [Google Scholar] [CrossRef]
- Bisen, P.S.; Baghel, R.K.; Sanodiya, B.S.; Thakur, G.S.; Prasad, G.B.K.S. Lentinus edodes: A Macrofungus with Pharmacological Activities. Curr. Med. Chem. 2010, 17, 2419–2430. [Google Scholar] [CrossRef]
- Morales, D.; Rutckeviski, R.; Villalva, M.; Abreu, H.; Smiderle, F.R. Isolation and Comparison of α- and β-d-glucans from Shiitake Mushrooms (Lentinula edodes) with Different Biological Activities. Carbohydr. Polym. 2019, 229, 115521. [Google Scholar] [CrossRef] [PubMed]
- Bhunia, S.K.; Dey, B.; Maity, K.K.; Patra, S.; Mandal, S.; Maiti, S.; Maiti, T.K.; Sikdar, S.R.; Islam, S.S. Isolation and Characterization of an Immunoenhancing Glucan from Alkaline Extract of an Edible mushroom, Lentinus squarrosulus (Mont.) Singer. Carbohyd. Res. 2011, 346, 2039–2044. [Google Scholar] [CrossRef]
- Cui, H.L.; Chen, Y.; Wang, S.S.; Kai, G.Q.; Fang, Y.M. Isolation, Partial Characterisation and Immunomodulatory Activities of Polysaccharide from Morchella esculenta. J. Sci. Food Agric. 2011, 91, 2180–2185. [Google Scholar] [CrossRef]
- Su, C.A.; Xu, X.Y.; Liu, D.Y.; Ming, W. Isolation and Characterization of Exopolysaccharide with Immunomodulatory Activity from Fermentation Broth of Morchella conica. DARU J. Pharm. Sci. 2013, 21, 5. [Google Scholar] [CrossRef]
- Du, X.; Zhang, J.; Yang, Y.; Tang, Q.; Jia, W.; Pan, Y. Purification, Chemical Modification and Immunostimulating Activity of Polysaccharides from Tremella aurantialba Fruit Bodies. J. Zhejiang Univ. Sci. B 2010. [Google Scholar] [CrossRef]
- Roy, S.K.; Das, D.; Mondal, S.; Maiti, D.; Bhunia, B.; Maiti, T.K.; Islam, S.S. Structural Studies of an Immunoenhancing Water-soluble Glucan Isolated from Hot Water Extract of an Edible Mushroom, Pleurotus florida, Cultivar Assam Florida. Carbohyd. Res. 2009, 344, 2596–2601. [Google Scholar] [CrossRef] [PubMed]
- Dey, B.; Bhunia, S.K.; Maity, K.K.; Patra, S.; Mandal, S.; Maiti, S.; Maiti, T.K.; Sikdar, S.R.; Islam, S.S. Glucans of Pleurotus florida Blue variant: Isolation, Purification, Characterization and Immunological Studies. Int. J. Biol. Macromol. 2012, 50, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Gern, R.M.M.; Wisbeck, E.; Rampinelli, J.R.; Ninow, J.L.; Furlan, S.A. Alternative Medium for Production of Pleurotus ostreatus Biomass and Potential Antitumor Polysaccharides. Bioresour. Technol. 2008, 99, 76–82. [Google Scholar] [CrossRef]
- Chen, X.; Xu, X.; Zhang, L.; Kennedy, J.F. Flexible Chain Conformation of (1→3)-β-d-glucan from Poria cocos sclerotium in NaOH/urea Aqueous Solution. Carbohyd. Polym. 2009, 75, 586–591. [Google Scholar] [CrossRef]
- Lin, Y.; Zhang, L.; Chen, L.; Jin, Y.; Zeng, F.; Jin, J.; Wan, B.; Cheung, P.C.K. Molecular Mass and Mntitumor Activities of Sulfated Derivatives of α-glucan from Poria cocos Mycelia. Int. J. Biol. Macromol. 2004, 34, 231–236. [Google Scholar] [CrossRef]
- Han, X.Q.; Wu, X.M.; Chai, X.Y.; Chen, D.; Dai, H.; Dong, H.L.; Ma, Z.Z.; Gao, X.M.; Tu, P.F. Isolation, Characterization and Immunological Activity of a Polysaccharide from the Fruit Bodies of an Edible Mushroom, Sarcodon aspratus (Berk.) S. Ito. Food Res. Int. 2011, 44, 489–493. [Google Scholar] [CrossRef]
- Hobbs, C. The Chemistry, Nutritional Value, Immunopharmacology, and Safety of the Traditional Food of Medicinal Split-Gill Fugus Schizophyllum commune Fr.:Fr. (Schizophyllaceae). A Literature Review. Int. J. Med. Mushrooms 2005, 7, 127–140. [Google Scholar] [CrossRef]
- Bimczok, D.; Wrenger, J.; Schirrmann, T.; Rothkotter, H.J.; Wray, V.; Rau, U. Short Chain Regioselectively Hydrolyzed Scleroglucans Induce Maturation of Porcine Dendritic Cells. Appl. Microbiol. Biotechnol. 2009, 82, 321–331. [Google Scholar] [CrossRef]
- Liu, J.J.; Huang, T.S.; Hsu, M.L.; Chen, C.C.; Lin, W.S.; Lu, F.J.; Chang, W.H. Antitumor Effects of the Partially Purified Polysaccharides from Antrodia camphorata and the Mechanism of Its Action. Toxicol. Appl. Pharm. 2004, 201, 186–193. [Google Scholar] [CrossRef]
- Wu, S.J.; Liaw, C.C.; Pan, S.Z.; Yang, H.C.; Ng, L.T. Phellinus linteus Polysaccharides and Their Immunomodulatory Properties in Human Monocytic Cells. J. Funct. Foods 2013, 5, 679–688. [Google Scholar] [CrossRef]
- Rau, U.; Kuenz, A.; Wray, V.; Nimtz, M.; Wrenger, J.; Cicek, H. Production and Structural Analysis of the Polysaccharide Secreted by Trametes (Coriolus) versicolor ATCC 200801. Appl. Microbiol. Biotechnol. 2009, 81, 827–837. [Google Scholar] [CrossRef]
- Wasser, S. Medicinal Mushrooms as a source of Antitumor and Immunomodulating Polysaccharides. Appl. Microbiol. Biotechnol. 2002, 60, 258–274. [Google Scholar] [CrossRef]
- Baets, S.D.; Vandamme, E.J. Extracellular Tremella polysaccharides: Structure, properties and applications. Biotechnol. Lett. 2001, 23, 1361–1366. [Google Scholar] [CrossRef]
- Ko, H.J.; Song, A.; Lai, M.N.; Ng, L.T. Immunomodulatory Properties of Xylaria nigripes in Peritoneal Macrophage Cells of Balb/c Mice. J. Ethnopharmacol. 2011, 138, 762–768. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.R.; Liu, Q.H.; Wang, H.X.; Ng, T.B. A Novel Lectin with Potent Antitumor, Mitogenic and HIV-1 Reverse Transcriptase Inhibitory Activities from the Edible Mushroom Pleurotus citrinopileatus. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2008, 1780, 51–57. [Google Scholar] [CrossRef]
- Zhao, J.K.; Wang, H.X.; Ng, T.B. Purification and Characterization of a Novel Lectin from the Toxic Wild Mushroom Inocybe umbrinella. Toxicon 2009, 53, 360–366. [Google Scholar] [CrossRef]
- Zhang, G.Q.; Sun, J.; Wang, H.X.; Ng, T.B. A Novel Lectin with Antiproliferative Activity from the Medicinal Mushroom Pholiota adiposa. Acta. Biochim. Pol. 2009, 56, 415–421. [Google Scholar] [CrossRef]
- Ngai, P.H.K.; Ng, T.B. A Mushroom (Ganoderma capense) Lectin with Spectacular Thermostability, Potent Mitogenic Activity on Splenocytes, and Antiproliferative Activity toward Tumor Cells. Biochem. Biophys. Res. Commun. 2004, 314, 988–993. [Google Scholar] [CrossRef]
- Han, C.H.; Liu, Q.H.; Ng, T.B.; Wang, H.X. A Novel Homodimeric Lactose-binding Lectin from the Edible Split Gill Medicinal Mushroom Schizophyllum commune. Biochem. Biophys. Res. Commun. 2005, 336, 252–257. [Google Scholar] [CrossRef]
- Zhao, S.; Zhao, Y.; Li, S.; Zhao, J.; Zhang, G.; Wang, H.; Ng, T.B. A Novel Lectin with Highly Potent Antiproliferative and HIV-1 Reverse Transcriptase Inhibitory Activities from the Edible Wild Mushroom Russula delica. Glycoconj. J. 2010, 27, 259–265. [Google Scholar] [CrossRef]
- Licastro, F.; Morini, M.C.; Kretz, O.; Dirheimer, G.; Creppy, E.E.; Stirpe, F. Mitogenic Activity and Immunological Properties of Bolesatine, A Lectin Isolated from the Mushroom Boletus satanas Lenz. Int. J. Biochem. 1993, 25, 789. [Google Scholar] [CrossRef]
- Ho, J.C.K.; Sze, S.C.W.; Shen, W.Z.; Liu, W.K. Mitogenic Activity of Edible Mushroom Lectins. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2004, 1671, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Silvana, A.; Paola, M.; Cerdeiras, M.; Fraguas, L.F. Screening for Lectins from Basidiomycetes and Isolation of Punctularia atropurpurascens Lectin. J. Basic Microbiol. 2014, 54, 89–96. [Google Scholar] [CrossRef]
- Amano, K.; Katayama, H.; Saito, A.; Ando, A.; Nagata, Y.; Yoshiho, N. Aleuria aurantia Lectin Exhibits Antifungal Activity Against Mucor racemosus. Biosci. Biotechnol. Biochem. 2012, 76, 967–970. [Google Scholar] [CrossRef]
- Rana, T.; Bera, A.K.; Das, S.; Bhattacharya, D.; Pan, D.; Bandyopadhyay, S.; Mondal, D.K.; Samanta, S.; Bandyopadhyay, S.; Das, S.K. Pleurotus florida Lectin Normalizes Duration Dependent Hepatic oxidative Stress Responses Caused by Arsenic in Rat. Exp. Toxicol. Pathol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.Q.; Chen, Q.J.; Hua, J.; Liu, Z.L.; Sun, Y.; Xu, X.; Han, P.; Wang, H.X. An Inulin-Specific Lectin with Anti-HIV-1 Reverse Transcriptase, Antiproliferative, and Mitogenic Activities from the Edible Mushroom Agaricus bitorquis. BioMed Res. Int. 2019, 2019, 1341370. [Google Scholar] [CrossRef]
- Yu, L.G.; Fernig, D.G.; Smith, J.A.; Milton, J.D.; Rhodes, J.M. Reversible Inhibition of Proliferation of Epithelial Cell Lines by Agaricus bisporus (Edible Mushroom) Lectin. Cancer Res. 1993, 53, 4627–4632. [Google Scholar] [CrossRef]
- Yang, Q.; Yin, Y.; Pan, Y.; Ye, X.; Xu, B.; Yu, W.; Zeng, H.; Sun, H. Anti-metastatic Activity of Agrocybe aegerita Galectin (AAL) in a Mouse Model of Breast Cancer Lung Metastasis. J. Func. Foods 2018, 41, 163–170. [Google Scholar] [CrossRef]
- Zhao, C.; Sun, H.; Tong, X.; Qi, Y. An Antitumour Lectin from the Edible Mushroom Agrocybe aegerita. Biochem. J. 2003, 374, 321–327. [Google Scholar] [CrossRef]
- Lutsik-Kordovsky, M.D.; Stasyk, T.V.; Stoika, R.S. Analysis of Cytotoxicity of Lectin and Non-lectin Proteins from Amanita Mushrooms. Exp. Oncol. 2001, 23, 43–45. [Google Scholar]
- Feng, K.; Liu, Q.H.; Ng, T.B.; Liu, H.Z.; Li, J.Q.; Chen, G.; Sheng, H.Y.; Xie, Z.L.; Wang, H.X. Isolation and Characterization of a Novel Lectin from the Mushroom Armillaria luteo-virens. Biochem. Biolphys. Res. Commun. 2006, 345, 1573–1578. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Li, C.; Ng, T.B.; Wang, H.X. A Lectin with Mitogenic Activity from the Edible wild Mushroom Boletus edulis. Process Biochem. 2007, 42, 1620–1624. [Google Scholar] [CrossRef]
- Sun, J.; Ng, T.B.; Wang, H.X.; Zhang, G.Q. A Novel Hemagglutinin with Antiproliferative Activity against Tumor Cells from the Hallucinogenic Mushroom Boletus speciosus. BioMed Res. Int. 2014, 2014, 340467. [Google Scholar] [CrossRef]
- Pohleven, J.; Obermajer, N.A.; Saboti, J.A.; Lovar, S.; Sep, I.K.; Kos, J.; Kralj, B.; Trukelj, B.; Brzin, J.E. Purification, Characterization and Cloning of a Ricin B-like Lectin from Mushroom Clitocybe nebularis with Antiproliferative Activity Against Human Leukemic T cells. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2009, 1790, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.B.; Ngai, P.H.K.; Xia, L. An Agglutinin with Mitogenic and Antiproliferative Activities from the Mushroom Flammulina velutipes. Mycologia 2006, 98, 167–171. [Google Scholar] [CrossRef]
- Kawagishi, H.; Nomura, A.; Mizuno, T.; Kimura, A.; Chiba, S. Isolation and Characterization of a Lectin from Grifola frondosa Fruiting Bodies. Biochim. Biophys. Acta (BBA)-Gen. Subj. 1990, 1034, 247–252. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, G.; Ng, T.B.; Wang, H. A Novel Lectin with Antiproliferative and HIV-1 Reverse Transcriptase Inhibitory Activities from Dried Fruiting Bodies of the Monkey Head Mushroom Hericium erinaceum. J. Biomed. Biotechnol. 2010, 2010, 716515. [Google Scholar] [CrossRef]
- Koyama, Y.; Katsuno, Y.; Miyoshi, N.; Hayakawa, S.; Mita, T.; Muto, H.; Isemura, S.; Aoyagi, Y.; Isemura, M. Apoptosis Induction by Lectin Isolated from the Mushroom Boletopsis leucomelas in U937 Cells. Biosci. Biotechnol. Biochem. 2002. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, H.; Ng, T.B. Purification and Characterization of a Lectin with Antiproliferative Activity toward Cancer Cells from the Dried Fruit Bodies of Lactarius flavidulus. Carbohyd. Res. 2011, 346, 2576–2581. [Google Scholar] [CrossRef]
- Pushparajah, V.; Fatima, A.; Chong, C.H.; Gambule, T.Z.; Chan, C.J.; Ng, S.T.; Tan, C.S.; Fung, S.Y.; Lee, S.S.; Tan, N.H. Characterisation of a New Fungal Immunomodulatory Protein from Tiger Milk mushroom, Lignosus rhinocerotis. Sci. Rep. 2016, 6, 30010. [Google Scholar] [CrossRef] [PubMed]
- Cordara, G.; Winter, H.C.; Goldstein, I.J.; Krengel, U.; Sandvig, K. The Fungal Chimerolectin MOA Inhibits Protein and DNA Synthesis in NIH/3T3 Cells and May Induce BAX-mediated Apoptosis. Biochem. Biophys. Res. Commun. 2014, 447, 586–589. [Google Scholar] [CrossRef]
- Mahajan, R.G.; Patil, S.I.; Mohan, D.R.; Shastry, P. Pleurotus eous Mushroom Lectin (PEL) with Mixed Carbohydrate Inhibition and Antiproliferative Activity on Tumor Cell Lines. J. Biochem. Mol. Biol. Biophys. 2002, 6, 341–345. [Google Scholar] [CrossRef]
- Manna, D.; Pust, S.; Torgersen, M.L.; Cordara, G.; Sandvig, K. Polyporus squamosus Lectin La (PSL1a) Exhibits Cytotoxicity in Mammalian Cells by Disruption of Focal Adhesions, Inhibition of Protein Synthesis and Induction of Apoptosis. PLoS ONE 2017, 12, e0170716. [Google Scholar] [CrossRef]
- Zhang, G.; Sun, J.; Wang, H.; Ng, T.B. First Isolation and Characterization of a Novel Lectin with Potent Antitumor Activity from a Russula Mushroom. Phytomedicine 2010, 17, 775–781. [Google Scholar] [CrossRef]
- Chumkhunthod, P.; Rodtong, S.; Lambert, S.J.; Fordham-Skelton, A.P.; Reynolds, C.D. Purification and Characterization of an N-acetyl-d-galactosamine-specific Lectin from the Edible Mushroom Schizophyllum commune. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2006, 1760, 326–332. [Google Scholar] [CrossRef]
- Zhang, W.; Tian, G.; Geng, X.; Zhao, Y.; Tzi, N.; Zhao, L.; Wang, H. Isolation and Characterization of a Novel Lectin from the Edible Mushroom Stropharia rugosoannulata. Molecules 2014, 19, 19880–19891. [Google Scholar] [CrossRef]
- Wang, H.X.; Ng, T.B.; Ooi, V.E.C.; Liu, W.K.; Chang, S.T. Actions of Lectins from the Mushroom Tricholoma mongolicum on Macrophages, Splenocytes and Life-span in Sarcoma-bearing Mice. Anticancer Res. 1997, 17, 419–424. [Google Scholar]
- Liua, W.K.; Ho, J.C.K.; Ng, T.B. Suppression of Cell Cycle Progression by a Fungal Lectin: Activation of Cyclin-dependent Kinase Inhibitors. Biochem. Pharmacol. 2001, 61, 33–37. [Google Scholar] [CrossRef]
- She, Q.B.; Ng, T.B.; Liu, W.K. A Novel Lectin with Potent Immunomodulatory Activity Isolated from Both Fruiting Bodies and Cultured Mycelia of the Edible Mushroom Volvariella volvacea. Biochem. Biophys. Res. Commun. 1998. [Google Scholar] [CrossRef]
- Marty-Detraves, C.; Francis, F.; Baricault, L.; Fournier, D.; Paquereau, L. Inhibitory Action of a New lectin from Xerocomus chrysenteron on Cell-substrate Adhesion. Mol. Cell. Biochem. 2004, 258, 49–55. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, H.; Ng, T.B. First Report of a Xylose-specific Lectin with Potent Hemagglutinating, Antiproliferative and Anti-mitogenic Activities from a Wild Ascomycete Mushroom. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2006, 1760, 1914–1919. [Google Scholar] [CrossRef]
- Sheu, F.; Chien, P.J.; Hsieh, K.Y.; Chin, K.L.; Huang, W.T.; Tsao, C.Y.; Chen, Y.F.; Cheng, H.C.; Chang, H.H. Purification, Cloning, and Functional Characterization of a Novel Immunomodulatory Protein from Antrodia camphorata (Bitter Mushroom) That Exhibits TLR2-Dependent NF-κB Activation and M1 Polarization within Murine Macrophages. J. Agric. Food Chem. 2009, 57, 4130–4141. [Google Scholar] [CrossRef]
- Lin, J.W.; Guan, S.Y.; Duan, Z.W.; Shen, Y.H.; Li, T.L. Gene Coning of a Novel Fungal Immunomodulatory Protein from Chroogomphis rutilus and Its Expression in Pichia Pastoris. J. Chem. Technol. Biotechnol. 2016, 91, 2761–2768. [Google Scholar] [CrossRef]
- Li, S.; Jiang, Z.; Sun, L.; Liu, X.; Huang, Y.; Wang, F.; Xin, F. Characterization of a new fungal immunomodulatory protein, FIP-dsq2 from Dichomitus squalens. J. Biol. 2017, 246, 45–51. [Google Scholar] [CrossRef]
- Wang, P.H.; Hsu, C.I.; Tang, S.C.; Huang, Y.L.; Lin, J.Y. Fungal Immunomodulatory Protein from Flammulina velutipes Induces Interferon-γ Production Through p38 Mitogen-Activated Protein Kinase Signaling Pathway. J. Agric. Food Chem. 2004. [Google Scholar] [CrossRef]
- Su, K.Q.; Wang, X.F.; Zhou, X.W. Cloning and Bioinformatics Analysis of Fugal Immunomodulatory Protein Gene from Ganoderma astum. J. Shanghai Jiaotong Univ. 2012, 30, 65–71. (In Chinese) [Google Scholar]
- Li, Q.Z.; Wang, X.F.; Bao, T.W.; Liang, R.; Lin, J.; Zhou, X. In Vitro Synthesis of a Recombinant Fungal Immunomodulatory Protein from Lingzhi or Reishi Medicinal Mushroom, Ganoderma lucidum (W.Curt.:Fr.) P.Karst. (Aphyllophoromycetideae) and Analysis of Its Immunomodulatory Activity. Int. J. Med. Mushrooms 2010, 12, 347–358. [Google Scholar] [CrossRef]
- Lin, C.H.; Hsiao, Y.M.; Ou, C.C.; Lin, Y.W.; Chiu, Y.L.; Lue, K.H.; Chang, J.G.; Ko, J.L. GMI, a Ganoderma Immunomodulatory Protein, Down-regulates Tumor Necrosis Factor α-Induced Expression of Matrix Metalloproteinase 9 via NF-κB Pathway in Human Alveolar Epithelial A549 Cells. J. Agric. Food Chem. 2010, 58, 12014–12021. [Google Scholar] [CrossRef]
- Li, Q.; Wang, X.; Chen, Y.; Lin, J.; Zhou, X. Cytokines Expression Induced by Ganoderma sinensis Fungal Immunomodulatory Proteins (FIP-gsi) in Mouse Spleen Cells. Appl. Biochem. Biotechnol. 2010, 162, 1403–1413. [Google Scholar] [CrossRef]
- Hsiao, Y.M.; Huang, Y.L.; Tang, S.C.; Shieh, G.J.; Lai, J.Y.; Wang, P.H.; Ying, T.H.; Ko, J.L. Effect of a Fungal Immunomodulatory Protein from Ganoderma tsugae on Cell Cycle and Interferon-gamma Production Through Phosphatidylinositol 3-kinase Signal Pathway. Process Biochem. 2008, 43, 423–430. [Google Scholar] [CrossRef]
- Bastiaan-Net, S.; Chanput, W.; Hertz, A.; Zwittink, R.D.; Mes, J.J.; Wichers, H.J. Biochemical and Functional Characterization of Recombinant Fungal Immunomodulatory Proteins (rFIPs). Int. Immunopharmacol. 2013, 15, 167–175. [Google Scholar] [CrossRef]
- Cong, W.R.; Xu, H.; Liu, Y.; Li, Q.Z.; Li, W.; Zhou, X.W. Production and Functional Characterization of a Novel Fungal Immunomodulatory Protein FIP-SN15 Shuffled from Two Genes of Ganoderma species. Appl. Microbiol. Biotechnol. 2014, 98, 5967–5975. [Google Scholar] [CrossRef]
- Chang, H.H.; Sheu, F. A Novel Fungal Immunomodulatory Protein (PCP) Isolated from Poria cocos Activates Mouse Peritoneal Macrophage Involved in Toll-like Receptor 4. FASEB J. 2007, 21, 702–715. [Google Scholar] [CrossRef]
- Li, S.Y.; Shi, L.J.; Ding, Y.; Nie, Y.; Tang, X.M. Identification and Functional Characterization of a Novel Fungal immunomodulatory Protein from Postia placenta. Food Chem. Toxicol. 2015, 78, 64–70. [Google Scholar] [CrossRef]
- Feng, L.; Wen, H.A.; Zhang, Y.J.; Min, A.; Liu, X.Z. Purification and Characterization of a Novel Immunomodulatory Protein from the Medicinal Mushroom Trametes versicolor. Sci. China Life Sci. 2011, 54, 91–97. [Google Scholar] [CrossRef]
- Hsu, H.Y.; Hua, K.F.; Wu, W.C.; Hsu, J.; Weng, S.T.; Lin, T.L.; Liu, C.Y.; Hseu, R.S.; Huang, C.T. Reishi Immuno-modulation Protein Induces Interleukin-2 Expression via Protein Kinase-dependent Signaling Pathways Within Human T Cells. J. Cell. Physiol. 2008, 215, 15–26. [Google Scholar] [CrossRef]
- Ma, K.; Bao, L.; Han, J.; Jin, T.; Yang, X.; Zhao, F.; Li, S.; Song, F.; Liu, M.; Liu, H. New Benzoate Derivatives and Hirsutane Type Sesquiterpenoids with Antimicrobial Activity and Cytotoxicity from the Solid-state Fermented Rice by the Medicinal Mushroom Stereum hirsutum. Food Chem. 2014, 143, 239–245. [Google Scholar] [CrossRef]
- Chen, H.P.; Dong, W.B.; Feng, T.; Yin, X.; Li, Z.H.; Dong, Z.J.; Li, Y.; Liu, J.K. Four New Sesquiterpenoids from Fruiting Bodies of the Fungus Inonotus rickii. J. Asian Nat. Prod. Res. 2014, 16, 1–6. [Google Scholar] [CrossRef]
- Wang, S.J.; Bao, L.; Han, J.J.; Wang, Q.X.; Yang, X.L.; Wen, H.A.; Guo, L.-D.; Li, S.J.; Zhao, F.; Liu, H.W. Pleurospiroketals A–E, Perhydrobenzannulated 5,5-Spiroketal Sesquiterpenes from the Edible Mushroom Pleurotus cornucopiae. J. Nat. Prod. 2013. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Bao, L.; Zhao, F.; Wang, Q.; Li, S.; Ren, J.; Li, L.; Wen, H.; Guo, L.; Liu, H. Isolation, Identification, and Bioactivity of Monoterpenoids and Sesquiterpenoids from the Mycelia of Edible Mushroom Pleurotus cornucopiae. J. Agric. Food Chem. 2013, 61, 5122–5129. [Google Scholar] [CrossRef]
- Intaraudom, C.; Boonyuen, N.; Supothina, S.; Tobwor, P.; Prabpai, S.; Kongsaeree, P.; Pittayakhajonwut, P. Novel Spiro-sesquiterpene from the Mushroom Anthracophyllum sp. BCC18695. Phytochem. Lett. 2013, 6, 345–349. [Google Scholar] [CrossRef]
- Li, L.; Yang, X.; Li, S.; Gao, H.; Yao, X.S.; Wen, H.; Liu, H.W. Bioactive Sesquiterpenoids from the Solid Culture of the Edible Mushroom Flammulina velutipes Growing on Cooked Rice. Food Chem. 2012. [Google Scholar] [CrossRef]
- Wang, Y.; Li, B.; Liu, D.; Yang, X.; Li, S.; Hao, G.; Yao, X.; Wen, H.; Liu, H. Two New Sesquiterpenes and Six Norsesquiterpenes from the Solid Culture of the Edible Mushroom Flammulina velutipes. Tetrahedron 2012, 68, 3012–3018. [Google Scholar] [CrossRef]
- Kanokmedhakul, S.; Lekphrom, R.; Kanokmedhakul, K.; Hahnvajanawong, C.; Bua-Art, S.; Saksirirat, W.; Prabpai, S.; Kongsaeree, P. Cytotoxic Sesquiterpenes from Luminescent Mushroom Neonothopanus nambi. Tetrahedron 2012, 68, 8261–8266. [Google Scholar] [CrossRef]
- Han, J.J.; Chen, Y.H.; Bao, L.; Yang, X.L.; Liu, D.; Li, S.J.; Zhao, F.; Liu, H. Anti-inflammatory and Cytotoxic Cyathane Diterpenoids from the Medicinal Fungus Cyathus africanus. Fitoterapia 2013, 84, 22–31. [Google Scholar] [CrossRef]
- Wang, S.J.; Li, Y.X.; Bao, L.; Han, J.J.; Yang, X.L.; Li, H.R.; Wang, Y.Q.; Li, S.J.; Liu, H.W. Eryngiolide A, a Cytotoxic Macrocyclic Diterpenoid with an Unusual Cyclododecane Core Skeleton Produced by the Edible Mushroom Pleurotus eryngii. Org. Lett. 2012, 14, 3672–3675. [Google Scholar] [CrossRef] [PubMed]
- Suzuki. Anti-proliferative and Apoptosis-inducible Activity of Sarcodonin G from Sarcodon scabrosus in HeLa Cells. Int. J. Oncol. 1992. [Google Scholar] [CrossRef]
- Tsukamoto, S.; Macabalang, A.D.; Nakatani, K.; Obara, Y.; Ohta, T. Tricholomalides A-C, New Neurotrophic Diterpenes from the Mushroom Tricholoma sp. J. Nat. Prod. 2004, 66, 1578–1581. [Google Scholar] [CrossRef]
- Ma, K.; Ren, J.; Han, J.; Bao, L.; Liu, H. Ganoboninketals A-C, Antiplasmodial 3,4-seco-27-Norlanostane Triterpenes from Ganoderma boninense Pat. J. Nat. Prod. 2014, 77, 1847–1852. [Google Scholar] [CrossRef] [PubMed]
- Isaka, M.; Chinthanom, P.; Kongthong, S.; Srichomthong, K.; Choeyklin, R. Lanostane Triterpenes from Cultures of the Basidiomycete Ganoderma orbiforme BCC 22324. Phytochemistry 2013. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.J.; Min, B.S.; Ahn, E.M.; Nakamura, N.; Lee, H.K.; Hattori, M. New Triterpene Aldehydes, Lucialdehydes A–C, from Ganoderma lucidum and Their Cytotoxicity against Murine and Human Tumor Cells. J. ChemInform. 2002. [Google Scholar] [CrossRef]
- Wu, G.S.; Lu, J.J.; Guo, J.J.; Li, Y.B.; Tan, W.; Dang, Y.Y.; Zhong, Z.F.; Xu, Z.T.; Chen, X.P.; Wang, Y.T. Ganoderic Acid DM, a Natural Triterpenoid, Induces DNA Damage, G1 Cell Cycle Arrest and Apoptosis in Human Breast Cancer Cells. Fitoterapia 2012, 83, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Li, C.H.; Chen, P.Y.; Chang, U.M.; Kan, L.S.; Fang, W.H.; Tsai, K.S.; Lin, S.B. Ganoderic Acid X, a lanostanoid Triterpene, Inhibits Topoisomerases and Induces Apoptosis of Cancer Cells. Life Sci. 2005, 77, 252–265. [Google Scholar] [CrossRef]
- Lez, A.G.G.; León, F.; Rivera, A.; Lez-Plata, J.G.; Padron, J.I. New Lanostanoids from the Fungus Ganoderma concinna. J. Nat. Prod. 2002, 65, 417–421. [Google Scholar] [CrossRef]
- Su, H.J.; Fann, Y.F.; Chung, M.I.; Won, S.J.; Lin, C.N. New Lanostanoids of Ganoderma tsugae. J. Nat. Prod. 2000, 63, 514–516. [Google Scholar] [CrossRef]
- Kim, K.H.; Moon, E.; Sang, U.C.; Sun, Y.K.; Kang, R.L. Lanostane Triterpenoids from the Mushroom Naematoloma fasciculare. J. Nat. Prod. 2013, 76, 845. [Google Scholar] [CrossRef]
- Arpha, K.; Phosri, C.; Suwannasai, N.; Mongkolthanaruk, W.; Sodngam, S. Astraodoric acids A–D: New Lanostane Triterpenes from Edible Mushroom Astraeus odoratus and Their Anti-Mycobacterium Tuberculosis H37Ra and Cytotoxic Activity. J. Agric. Food Chem. 2012, 60, 9834–9841. [Google Scholar] [CrossRef]
- Clericuzio, M.; Cassino, C.; Corana, F.; Vidari, G. Terpenoids from Russula lepida and R. amarissima (Basidiomycota, Russulaceae). Phytochemistry 2012, 84, 154–159. [Google Scholar] [CrossRef]
- Clericuzio, M.; Tabasso, S.; Bianco, M.A.; Pratesi, G.; Beretta, G.; Tinelli, S.; Zunino, F.; Vidari, G. Cucurbitane Triterpenes from the Fruiting Bodies and Cultivated Mycelia of Leucopaxillus gentianeus. J. Nat. Prod. 2006, 69, 1796–1799. [Google Scholar] [CrossRef]
- Shao, H.J.; Chen, Q.; Fei, W.; Zhang, Y.L.; Luo, D.Q.; Liu, J.K. A New Cytotoxic Lanostane Triterpenoid from the Basidiomycete Hebeloma versipelle. Cheminform 2010, 37, 828–831. [Google Scholar] [CrossRef]
- Yoshikawa, K.; Kuroboshi, M.; Ahagon, S.; Arihara, S. Three Novel Crustulinol Esters, Saponaceols A-C, from Tricholoma saponaceum. Chem. Pharm. Bull. 2004. [Google Scholar] [CrossRef]
- Yoshikawa, K.; Nishimura, N.; Bando, S.; Arihara, S.; Matsumura, E. New Lanostanoids, Elfvingic Acids A−H, from the Fruit Body of Elfvingia applanata. J. Nat. Prod. 2002. [Google Scholar] [CrossRef]
- Mallard, B.; Leach, D.N.; Wohlmuth, H.; Tiralongo, J. Synergistic Immuno-modulatory Activity in Human Macrophages of a Medicinal Mushroom Formulation Consisting of Reishi, Shiitake and Maitake. PLoS ONE 2019, 14. [Google Scholar] [CrossRef]
- Carbonero, E.R.; Gracher, A.H.P.; Komura, D.L.; Marcon, R.; Freitas, C.S.; Baggio, C.H.; Santos, A.R.S.; Torri, G.; Gorin, P.A.J.; Iacomini, M. Lentinus edodes heterogalactan: Antinociceptive and anti-inflammatory effects. Food Chem. 2008, 111, 531–537. [Google Scholar] [CrossRef]
- Finimundy, T.C.; Dillon, A.J.P.; Henriques, J.A.P.; Ely, M.R. A Review on General Nutritional Compounds and Pharmacological Properties of the Lentinula edodes. Mushroom. Int. J. Food Sci. Nutr. 2014, 5, 1095–1105. [Google Scholar] [CrossRef]
- Lee, H.H.; Lee, J.S.; Cho, J.Y.; Kim, Y.E.; Hong, E.K. Study on Immunostimulating Activity of Macrophage Treated with Purified Polysaccharides from Liquid Culture and Fruiting Body of Lentinus edodes. J. Microbiol. Biotechnol. 2009, 19, 566–572. [Google Scholar] [CrossRef]
- Chen, H.; Ju, Y.; Li, J.; Yu, M. Antioxidant activities of polysaccharides from Lentinus edodes and Their Significance for Disease Prevention. Int. J. Biol. Macromol. 2012, 50, 214–218. [Google Scholar] [CrossRef]
- Rincao, V.P.; Yamamoto, K.A.; Ricardo, N.M.P.S.; Soares, S.A.; Meirelles, L.D.P.; Nozawa, C.; Linhares, R.E.C. Polysaccharide and Extracts from Lentinula edodes: Structural Features and Antiviral activity. J. Virol. 2012, 9. [Google Scholar] [CrossRef]
- Ulziijargal, E.; Yang, J.H.; Lin, L.Y.; Chen, C.P.; Mau, J.L. Quality of Bread Supplemented with Mushroom Mycelia. Food Chem. 2013, 138, 70–76. [Google Scholar] [CrossRef]
- Kim, S.Y.; Kang, M.Y.; Kim, M.Y. Quality Characteristics of Noodle Added with Browned Oak Mushroom (Lentinus edodes). Korean J. Food Cook. Sci. 2008, 24, 665–671. (In Korean) [Google Scholar]
- Parab, D.N.; Dhalagade, J.R.; Sahoo, A.K.; Ranveer, R.C. Effect of Incorporation of Mushroom (Pleurotus sajor-caju) Powder on Quality Characteristics of Papad (Indian snack food). Int. J. Food Sci. Nutr. 2012, 63, 866–870. [Google Scholar] [CrossRef]
- Singla, R.; Ghosh, M.; Ganguli, A. Phenolics and Antioxidant Activity of a Ready-to-eat Snack Food Prepared from the Edible Mushroom (Agaricus bisporous). Food Sci. Nutr. 2009, 39, 227–234. [Google Scholar] [CrossRef]
- Margaret, B.; Emma, D.; Tiwari, B.K.; Brennan, C.S. Enrichment of Extruded Snack Products with Coproducts from Chestnut Mushroom (Agrocybe aegerita) Production: Interactions between Dietary Fiber, Physicochemical Characteristics, and Glycemic Load. J. Agric. Food Chem. 2012, 60, 4396–4401. [Google Scholar]
- Ribeiro, A.; Ruphuy, G.; Lopes, J.C.; Dias, M.M.; Barros, L.; Barreiro, F.; Ferreira, I.C.F.R. Spray-drying Microencapsulation of Synergistic Antioxidant Mushroom Extracts and Their use as Functional Food Ingredients. Food Chem. 2015, 188, 612–618. [Google Scholar] [CrossRef]
- Yuan, Q.; Zhang, X.; Ma, M.; Long, T.; Xiao, C.; Zhang, J.; Liu, J.; Zhao, L. Immunoenhancing Glucuronoxylomannan from Tremella Aurantialba Bandoni et Zang and Its Low-Molecular-Weight Fractions by Radical Depolymerization: Properties, Structures and Effects on Macrophages. Carbohydr. Polym. 2020, 238, 116–184. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Meng, M.; Zhao, J.; Wang, X.; Wang, C. Immunomodulatory Effects of the Polysaccharide from Craterellus cornucopioides Via Activating the TLR4-NFkappaB Signaling Pathway in Peritoneal Macrophages of BALB/c Mice. Int. J. Biol. Macromol. 2020, 160, 871–879. [Google Scholar] [CrossRef]
- Li, Y.; You, L.; Dong, F.; Yao, W.; Chen, J. Structural Characterization, Antiproliferative and Immunoregulatory Activities of a Polysaccharide from Boletus leccinum Rugosiceps. Int. J. Biol. Macromol. 2020, 157, 106–118. [Google Scholar] [CrossRef]
- Tan, X.; Chen, W.; Jiao, C.; Liang, H.; Yun, H.; He, C.; Chen, J.; Ma, X.; Xie, Y. Anti-tumor and Immunomodulatory Activity of the Aqueous Extract of Sarcodon imbricatus in Vitro and in Vivo. Food Funct. 2020, 11, 1110–1121. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.X.; Li, J.Z.; Wan, C.Y.; Hua, W.; Xiao, H.K.; Lv, Y.P.; Dong, M.H. Antitumor and Immunomodulatory Activity of Pleurotus Eryngii Extract. J. Food Biochem. 2014, 39, 19–27. [Google Scholar] [CrossRef]
- Borchers, A.T.; Krishnamurthy, A.; Keen, C.L.; Meyers, F.J.; Gershwin, M.E. The Immunobiology of Mushrooms. Exp. Biol. Med. 2008, 233, 259–276. [Google Scholar] [CrossRef] [PubMed]
- Xin, M.; He, B.L.; Li, X.L. Antitumor Polysaccharides from Mushrooms: A Review on the Structural Characteristics, Antitumor Mechanisms and Immunomodulating Activities. Carbohydr. Res. 2016, 30–41. [Google Scholar] [CrossRef]
- Kim, F.; Sakagami, H.; Tanuma, S.I.; Konno, K. Stimulation of Interferon-γ-induced Human Myelogenous Leukemic Cell Differentiation by High Molecular Weight PSK Subfraction. Anticancer Res. 1990, 10, 55–58. [Google Scholar] [CrossRef]
- Adachi, Y.; Ohno, N.; Ohsawa, M.; Oikawa, S.; Yadomae, T. Change of Biological Activities of (1.RAR.3)-.BETA.-d-glucan from Grifola frondosa upon Molecular Weight Reduction by Heat Treatment. Chem. Pharm. Bull. 1990, 38, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Chihara, G.; Hamuro, J.; Maeda, Y.; Arai, Y.; Fukuoka, F. Antitumour Polysaccharide Derived Chemically from Natural Glucan (Pachyman). Nature 1970, 225, 943–944. [Google Scholar] [CrossRef]
- Bae, I.Y.; Kim, H.W.; Yoo, H.J.; Kim, E.S.; Lee, S.; Dong, Y.P.; Lee, H.G. Correlation of Branching Structure of Mushroom β-glucan with its Physiological Activities. Food Res. Int. 2013, 51, 195–200. [Google Scholar] [CrossRef]
- Ren, L.; Perera, C.; Hemar, Y. Antitumor Activity of Mushroom Polysaccharides: A Review. Food Funct. 2012, 3, 1118–1130. [Google Scholar] [CrossRef]
- Palleschi, A.; Bocchinfuso, G.; Coviello, T.; Alhaique, F. Molecular Dynamics Investigations of the Polysaccharide Scleroglucan: First Study on the Triple Helix Structure. Carbohydr. Res. 2005, 340, 2154–2162. [Google Scholar] [CrossRef]
- Zhang, P.; Cheung, P.C.K. Evaluation of Sulfated Lentinus edodes α-(1 → 3)-d-glucan as a Potential Antitumor Agent. J. Agric. Chem. Soc. Jpn. 2002, 66, 1052–1056. [Google Scholar] [CrossRef]
- Singh, R.S.; Walia, A.K.; Kennedy, J.F. Mushroom Lectins in Biomedical Research and Development. Int. J. Boil. Macromol. 2019, 151, 1340–1350. [Google Scholar] [CrossRef]
- Švajger, U.; Pohleven, J.; Kos, J.; Štrukelj, B.; Jeras, M. CNL, a Ricin B-like Lectin from Mushroom Clitocybe nebularis, Induces Maturation and Activation of Dendritic Cells via the Toll-like Receptor 4 Pathway. Immunology 2011, 134, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Pohleven, J.; Renko, J.; Magister, S.; Smith, S.; Magister, D.F.; Kuenzler, M.; Strukelj, B.; Turk, D.; Kos, J.; Sabotic, J. Bivalent Carbohydrate Binding Is Required for Biological Activity of Clitocybe nebularis Lectin (CNL), the N,N′-Diacetyllactosediamine (GalNAcβ1-4GlcNAc, LacdiNAc)-Specific Lectin from Basidiomycete C. nebularis. J. Biol. Chem. 2012, 287, 10602–10612. [Google Scholar] [CrossRef]
- Zhang, L.Q.; Zhang, Z.Y.; Wei, Z.X. Fungal Immunomodulatory Proteins: Characteristic, Potential Antitumor Activities and Their Molecular Mechanisms. Drug Discov. Today 2018. [Google Scholar] [CrossRef]
- Wang, X.F.; Li, Q.Z.; Bao, T.W.; Cong, W.R.; Song, W.X.; Zhou, X.W. In Vitro Rapid Evolution of Fungal Immunomodulatory Proteins by DNA Family Shuffling. Appl. Microbiol. Biotechnol. 2013, 97, 2455–2465. [Google Scholar] [CrossRef]
- Jeong, Y.T.; Yang, B.K.; Jeong, S.C.; Kim, S.M.; Song, C.H. Ganoderma applanatum: A Promising Mushroom for Antitumor and Immunomodulating Activity. Phytother. Res. Ptr. 2008, 22, 614–619. [Google Scholar] [CrossRef]
- Tao, N.; Cui, X.H.; Cai, H.M.; Ma, Y.H.; Zhao, Y.C.; Chen, W.M. Establishment of Genetic Transformation System of Agrocybe aegerita Using PEG-Mediated Method. Mycosystema 2020, 39, 1100–1108. [Google Scholar] [CrossRef]
- Sonnenberg, A.S.M.; Gao, W.; Lavrijssen, B.; Hendrickx, P.; Visser, R.G.F. A Detailed Analysis of the Recombination Landscape of the Button Mushroom Agaricus bisporus Var. bisporus. Fungal Genet. Biol. 2016, 93, 35–45. [Google Scholar] [CrossRef]
- Morin, E.; Kohler, A.; Baker, A.R.; Foulongne-Oriol, M.; Lombard, V.; Nagy, L.G.; Ohm, R.A.; Patyshakuliyeva, A.; Brun, A.; Aerts, A.L.; et al. Genome Sequence of the Button Mushroom Agaricus bisporus Reveals Mechanisms Governing Adaptation to a Humic-rich Ecological Niche (vol 109, pg 17501, 2012). Proc. Natl. Acad. Sci. USA 2013, 110, 4146. [Google Scholar] [CrossRef]
- Chen, M.Y.; Liao, J.H.; Guo, Z.J.; Li, H.R.; Lu, Z.H.; Cai, D.F.; Wang, Z.S. The Expression Vector Construction and Transformation of Thermotolerance-related Gene of Agaricus bisporus. Mycosystema 2009, 28, 797–801. (In Chinese) [Google Scholar]
- Cheng, K.; Zhu, Z.; Wang, J.; Chen, J.; Bao, D.; Chen, M.; Zhang, J.; Tan, Q. Progress in the Genetic Transformation of Edible Fungi. Acta Edulis Fungi 2012, 19, 92–99. (In Chinese) [Google Scholar]
- Kurata, A.; Fukuta, Y.; Mori, M.; Kishimoto, N.; Shirasaka, N. Draft Genome Sequence of the Basidiomycetous Fungus Flammulina velutipes TR19. Genome Announc. 2016, 4, e00505–e00516. [Google Scholar] [CrossRef]
- Young-Jin, P.; Hun, B.K.; Seonwook, L.; Changhoon, K.; Hwanseok, R.; Hyungtae, K.; Jeong-Sun, S.; Hae-Ran, P.; Dae-Eun, Y.; Jae-Young, N.; et al. Whole Genome and Global Gene Expression Analyses of the Model Mushroom Flammulina velutipes Reveal a High Capacity for Lignocellulose Degradation. PLoS ONE 2014, 9, e93560. [Google Scholar] [CrossRef]
- Hyeokjun, Y.; You, Y.H.; Ju-Ri, W.; Young-Jin, P.; Kong, W.S.; Byoung-Moo, L.; Jong-Guk, K. The Mitochondrial Genome of the White-Rot Fungus Flammulina velutipes. J. Gen. Appl. Microbiol. 2012, 58, 331–337. [Google Scholar] [CrossRef][Green Version]
- Ko, J.L.; Lin, S.J.; Hsu, C.I.; Kao, C.L.; Lin, J.Y. Molecular Cloning and Expression of a Fungal Immunomodulatory Protein, FIP-fve, from Flammulina velutipes. J. Med. Assoc. 1997, 96, 517–524. [Google Scholar] [CrossRef]
- Xu, H.; Kong, Y.Y.; Chen, X.; Guo, M.Y.; Bai, X.H.; Lu, Y.J.; Li, W.; Zhou, X.W. Recombinant FIP-gat, a Fungal Immunomodulatory Protein from Ganoderma atrum, Induces Growth Inhibition and Cell Death in Breast Cancer Cells. J. Agric. Food Chem. 2016, 64, 2690–2698. [Google Scholar] [CrossRef]
- Li, Q.Z.; Chang, Y.Z.; He, Z.M.; Chen, L.; Zhou, X.W. Immunomodulatory Activity of Ganoderma lucidum Immunomodulatory Protein Via PI3K/Akt and MAPK Signaling Pathways in RAW264.7 Cells. J. Cell. Physiol. 2019, 234, 23337–23348. [Google Scholar] [CrossRef]
- Chen, S.L. Genome Sequence of the Model Medicinal Mushroom Ganoderma lucidum. Nat. Commun. 2012, 3. [Google Scholar] [CrossRef]
- Liu, D.; Jing, G.; Dai, W.; Kang, X.; Zhuo, H.; Zhang, H.M.; Wei, L.; Le, L.; Ma, J.; Xia, Z. The Genome of Ganderma lucidum Provide Insights into Triterpense Biosynthesis and Wood Degradation. PLoS ONE 2012. [Google Scholar] [CrossRef]
- Zhu, Y.; Xu, J.; Sun, C.; Zhou, S.; Xu, H.; Nelson, D.R.; Qian, J.; Song, J.; Luo, H.; Xiang, L. Chromosome-level Genome Map Provides Insights into Diverse Defense Mechanisms in the Medicinal Fungus Ganoderma sinense. Sci. Rep. 2015, 5, 11087. [Google Scholar] [CrossRef]
- Wang, P.H.; Yang, S.F.; Chen, G.D.; Han, C.P.; Chen, S.C.; Lin, L.Y.; Ko, J.L. Human Nonmetastatic Clone 23 Type 1 Gene Suppresses Migration of Cervical Cancer Cells and Enhances the Migration Inhibition of Fungal Immunomodulatory Protein from Ganoderma tsugae. Reprod. Sci. 2007, 14, 475–485. [Google Scholar] [CrossRef]
- Gong, W.; Wang, Y.; Xie, C.; Zhou, Y.; Peng, Y. Whole Genome Sequence of an Edible and Medicinal mushroom, Hericium erinaceus (Basidiomycota, Fungi). Genomics 2020, 112. [Google Scholar] [CrossRef]
- Liu, L.; Xiao, Z.; Guo, L.; Lin, J.; You, L.; Liao, J. Establishment of Genetic Transformation System of Hericium erinaceus Using PEG Mediated Method. Mycosystema 2014, 33, 121–128. (In Chinese) [Google Scholar]
- Gong, W.B.; Li, L.; Zhou, Y.; Bian, Y.B.; Kwan, H.S.; Cheung, M.K.; Xiao, Y. Genetic Dissection of Fruiting Body-related Traits Using Quantitative Trait loci Mapping in Lentinula edodes. Appl. Microbiol. Biotechnol. 2016, 100, 5437–5452. [Google Scholar] [CrossRef]
- Qu, J.; Zhao, M.; Tom, H.; Feng, X.; Zhang, J.; Huang, C. Identification and Characterization of Small Noncoding RNAs in Genome Sequences of the Edible Fungus Pleurotus ostreatus. BioMed Res. Int. 2016, 2016, 2503023. [Google Scholar] [CrossRef]
- Riley, R.; Salamov, A.A.; Brown, D.W.; Nagy, L.G.; Floudas, D.; Held, B.W.; Levasseur, A.; Lombard, V.; Morin, E.; Otillar, R. Extensive Sampling of Basidiomycete Genomes Demonstrates Inadequacy of the White-Rot/Brown-Rot Paradigm for Wood Decay Fungi. Proc. Natl. Acad. Sci. USA 2014, 111, 9923–9928. [Google Scholar] [CrossRef]
- Yong, W.; Zeng, F.; Chau, H.C.; Zhang, Y.; Ching, L.F.C. The Mitochondrial Genome of the Basidiomycete Fungus Pleurotus ostreatus (oyster mushroom). FEMS Microbiol. Lett. 2010. [Google Scholar] [CrossRef]
- Martinez, D.; Challacombe, J.; Morgenstern, I.; Hibbett, D.; Schmoll, M.; Kubicek, C.P.; Ferreira, P.; Ruiz-Duenas, F.J.; Martinez, A.T.; Kersten, P.; et al. Genome, Transcriptome, and Secretome Analysis of Wood Decay Fungus Postia placenta Supports Unique Mechanisms of Lignocellulose Conversion. Proc. Natl. Acad. Sci. USA 2009, 106, 1954–1959. [Google Scholar] [CrossRef]
- Li, F.; Wen, H.; Liu, X.; Zhou, F.; Chen, G. Gene Cloning and Recombinant Expression of a Novel Fungal Immunomodulatory Protein from Trametes versicolor. Protein Expr. Purif. 2012, 82, 339–344. [Google Scholar] [CrossRef]
- Floudas, D.; Binder, M.; Riley, R.; Barry, K.; Blanchette, R.A.; Henrissat, B.T.; Martinez, A. The Paleozoic Origin of Enzymatic Lignin Decomposition Reconstructed from 31 Fungal Genomes. Science 2012, 336, 1715–1719. [Google Scholar] [CrossRef]
- Sun, X.; Huang, W.; Xiao, S.; Liang, C.; Zhang, S.; Liu, Z.; Sun, F. Extracellular Expression and Efficient Purification of a Functional Recombinant Volvariella volvacea Immunomodulatory Protein (FIP-vvo) Using Pichia Pastoris System. Protein Expr. Purif. 2014, 94, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Bao, D.; Gong, M.; Zheng, H.; Chen, M.; Zhang, L.; Wang, H.; Jiang, J.; Wu, L.; Zhu, Y.; Zhu, G. Sequencing and Comparative Analysis of the Straw Mushroom (Volvariella volvacea) Genome. PLoS ONE 2013, 8, e58294. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R.A.A.; Boudrant, J.; Meyer, D.; Manno, N.; Demarchis, M.; Paoletti, M.G. Current views on fungal chitin/chitosan, human chitinases, food preservation, glucans, pectins and inulin: A tribute to Henri Braconnot, precursor of the carbohydrate polymers science, on the chitin bicentennial. Carbohydr. Polym. 2012, 87, 995–1012. [Google Scholar] [CrossRef]
- Yuan, X.; Zheng, J.; Jiao, S.; Cheng, G.; Feng, C.; Du, Y.; Liu, H. A review on the preparation of chitosan oligosaccharides and application to human health, animal husbandry and agricultural production. Carbohydr. Polym. 2019, 220, 60–70. [Google Scholar] [CrossRef]
- Grundemann, C.; Reinhardt, J.K.; Lindequist, U. European medicinal mushrooms: Do they have potential for modern medicine? An update. Phytomedicine 2020, 66, 153131. [Google Scholar] [CrossRef] [PubMed]
- Wanmuang, F.; Leopairut, J.; Kositchaiwat, C.; Wananukul, W.; Bunyaratvej, S. Fatal fulminant hepatitis associated with Ganoderma lucidum (Lingzhi) mushroom powder. J. Med. Assoc. Thail. 2007, 90, 179–181. [Google Scholar]
- Hisamochi, A.; Kage, M.; Arinaga, T.; Ide, T.; Miyajima, I.; Ogata, K.; Kuwahara, R.; Koga, Y.; Kumashiro, R.; Sata, M. Drug-induced liver injury associated with Agaricus blazei Murill which is very similar to autoimmune hepatitis. Clin. J. Gastroenterol. 2013, 6, 139–144. [Google Scholar] [CrossRef]


| MM Species | Common Name | Taxonomy | Geographic/Ecological Distribution |
|---|---|---|---|
| Agrocybe aegerita | Black Poplar mushroom | Basidiomycota Agaricomycetes Agaricales Bolbitiaceae | North temperate and subtropical zone |
| Agaricus bisporus | Button mushroom, Portobello mushroom, Common mushroom | Basidiomycota Agaricomycetes Agaricales Agaricaceae | USA, China, France, Netherlands, United Kingdom, Italy, Poland, Spain, Germany, Canada, Ireland, Belgium, Indonesia, Hungary and Mexico |
| Agaricus blazei (syn. Agaricus brasiliensis) | Royal Sun Agaricus, Almond Portobello | Basidiomycota Agaricomycetes Agaricales Agaricaceae | America, Brasil, Japan, China |
| Amanita phalloides | Death Cap | Basidiomycota Agaricomycetes Agaricales Amanitaceae | Europe, North American, Asia |
| Boletus edulis | Cep, Porcini, Penny Bun Bolete | Basidiomycota Agaricomycetes Boletales Boletaceae | China, Italy, France, Swiss, Germany |
| Boletus speciosus | Red-Capped Butter Bolete | Basidiomycota Agaricomycetes Boletales Boletaceae | Eastern North America, Southwest of China and Europe |
| Chroogomphus rutilus | Copper Spike | Basidiomycota Agaricomycetes Boletales Gomphidiaceae | China |
| Clitocybe nebularis | Clouded Funnel | Basidiomycota Agaricomycetes Agaricales Tricholomataceae | China, Japan, Taiwan, Europe, North America, North Africa |
| Cryptoporus volvatus | Veiled Polypore | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Trunks of pine, fir and spruce |
| Dichomitus squalens | Common White-Rot fungus | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Trunks of conifers such as pine and larch |
| Flammulina velutipes | Golden Needle mushroom | Basidiomycota Agaricomycetes Agaricales Physalacriaceae | Subtropical zone such as Japan, Russia, Australia and other countries as well as Europe, North America |
| Floccularia luteovirens (syn. Armillaria luteovirens) | Scaly Yellow mushroom | Basidiomycota Agaricomycetes Agaricales Tricholomataceae | Meadow at altitudes of 3000–4000 m above sea level |
| Ganoderma atrum | Black Ling-zhi | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Tropical regions |
| Ganoderma capense | Dark Ling-zhi | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Tropical regions |
| Ganoderma japonicum | Bloody Ling-zhi | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Majority in tropical and subtropical regions of Asia, Australia, Africa and America, minority in temperate zone |
| Ganoderma lucidum | Reitake, Ling-zhi, Spirit Plant Reishi | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Majority in tropical and subtropical regions of Asia, Australia, Africa and America, minority in temperate zone |
| Ganoderma microsporum | Small-Spored Ling-zhi | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Subtropics zone |
| Ganoderma lingzhi | Ling-zhi | Basidiomycota Agaricomycetes Polyporales Polyporaceae | China, North Korea, Japan |
| Ganoderma sinensis | Zi-zhi | Basidiomycota Agaricomycetes Polyporales Polyporaceae | China, North Korea, Japan |
| Ganoderma tsugae | Hemlock Varnish Shelf | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Northern and Montaine zone |
| Grifola frondosa | Maitake Hen of the Woods | Basidiomycota Agaricomycetes Polyporales Grifolaceae | Japan, China |
| Hericium erinaceus | Lion’s Mane mushroom, Bearded Tooth mushroom, Monkey-Head mushroom | Basidiomycota Agaricomycetes Russulales Hericiaceae | Broad-leaved forest or coniferous and broad-leaved mixed forest in northern temperate zone such as Western Europe, North America, China, Japan, Russia |
| Inonotus obliquus | Clinker Polypore, Birch Conk, Chaga | Basidiomycota Agaricomycetes Hymenochaetacles Hymenochaetaceae | Russia, China |
| Lentinula edodes | Shiitake, Black Forest mushroom, Golden Oak mushroom | Basidiomycota Agaricomycetes Agaricales Omphalotaceae | Distributed in an arc area on the west side of the Pacific Ocean, Japan, Papua New Guinea, Nepal, the Mediterranean coast and northern Africa |
| Lignosus rhinocerotis | Tiger Milk mushroom | Basidiomycota Agaricomycetes Polyporales Polyporaceae | China, Indonesia, Philippines, Sri Lanka, Australia, Thailand, Malaysia, Papua New Guinea and rainforests of East Africa |
| Leucocalocybe mongolica (syn. Tricholoma mongolicum) | Mongolia mushroom | Basidiomycota Basidiomycetes Agaricales Agaricales incertae sedis | Inner Mongolia in China |
| Marasmius oreades | Fairy Ring mushroom | Basidiomycota Agaricomycetes Agaricales Marasmiaceae | North America and Asia |
| Morchella esculenta | Common Morel, Yellow Morel, Sponge Morel | Ascomycota Pezizomycetes Pezizales Morohellaceae | Widely cultured over the world such as France, Germany, America, India, China, Russia, Sweden, Mexico, Spain, Czechoslovakia and Pakistan |
| Morchella conica | Black Morel, Sponge mushroom | Ascomycota Pezizomycetes Pezizales Morohellaceae | Distributed under broad-leaved forest, coniferous broad-leaved mixed forest, forest edge open space and weeds |
| Naematelia aurantialba (syn. Tremella aurantialba) | Golden Tremella | Basidiomycota Tremellomycetes Tremellales Naemateliaceae | Mountain forest of quercus, mutualism with Stereum spp. |
| Ophiocordyceps sinensis | Caterpillar fungus, Himalaya Viagra | Ascomycota Sordariomycetes Hypocreales Ophiocordycipitaceae | Southwest China, Nepal |
| Pholiota adiposa | Chestnut mushroom | Basidiomycota Agaricomycetes Agaricales Strophariaceae | Distributed on the dead willows in the forest in China |
| Pleurotus citrinopileatus | Golden Oyster mushroom, Tamogitake | Basidiomycota Agaricomycetes Agaricales Pleurotaceae | Widely cultured all over the world |
| Pleurotus ostreatus | Oyster mushroom | Basidiomycota Agaricomycetes Agaricales Pleurotaceae | Widely cultured all over the world |
| Cerioporus squamosus (syn. Polyporus squamosus) | Dryad’s Saddle, Pheasant’s Back mushroom | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Widely distributed in hardwood forest of North America, Australia, Asia and Europe |
| Poria cocos | Fuling, China Root | Basidiomycota Agaricomycetes Polyporales Laetiporaceae | Parasitic on the roots of Pinaceae plants, mainly distributed in China |
| Rhodonia placenta (syn. Postia placenta) | Rosy Crust | Basidiomycota Agaricomycetes Polyporales Dacryobolaceae | Widely distributed all over the world |
| Pseudosperma umbrinellum (syn. Inocybe umbrinella) | Fibrous Hat | Basidiomycota Agaricomycetes Agaricales Inocybaceae | France |
| Russula delica | Milk-White Brittlegill | Basidiomycota Agaricomycetes Russulales Russulaceae | Taiga forest and mixed forests |
| Russula lepida | Rosy Russula | Basidiomycota Agaricomycetes Russulales Russulaceae | Widely distributed all over the world |
| Sarcodon aspratus | Black Tiger Paw | Basidiomycota Agaricomycetes Thelephorales Thelephoraceae | Southwest of China |
| Schizophyllum commune | Split Gill | Basidiomycota Agaricomycetes Agaricales Schizophyllaceae | Widely distributed all over the world |
| Stropharia rugosoannulata | Wine Cap Stropharia, Garden Giant, Burgundy mushroom, King Stropharia | Basidiomycota Agaricomycetes Agaricales Strophariaceae | Europe, North America, Asia |
| Taiwanofungus camphoratus (syn. Antrodia camphorate) | Poroid Brown-rot fungus, Stout Camphor fungus | Basidiomycota Agaricomycetes Polyporales incertae sedis | Mountain forest in Taiwan with altitudes of 450–2000 m |
| Trametes versicolor (syn. Polystictus versicolor) | Turkey Tail fungus | Basidiomycota Agaricomycetes Polyporales Polyporaceae | Global distribution; Broad-leaf woods |
| Tropicoporus linteus (syn. Phelllinus linteus) | Mesima, Black Hoof fungus | Basidiomycota Agaricomycetes Hymenochaetales Hymenochaetaceae | Distributed on the dead trees and trunks in China |
| Xerocomellus chrysenteron (syn. Xerocomus Chrysenteron) | Red Cracking Bolete | Basidiomycota Agaricomycetes Agaricales Agaricales incertae sedis | China |
| Xylaria hypoxylon | Candlestick fungus, Candlesnuff fungus, Carbon Antlers, Stag’s Horn fungus | Ascomycota Sordariomycetes Xylariales Xylariaceae | Northern Europe |
| Xylaria nigripes | Dead Moll’s Fingers | Ascomycota Sordariomycetes Xylariales Xylariaceae | China, mutualism with white ant |
| Volvariella volvacea | Straw mushroom | Basidiomycota Basidiomycetes Agaricales Pluteaceae | China, East Asia, Southeast Asia |
| Medicinal Mushroom | Genome Size Mb | Number of Genes | GC Content (%) | Known Genes Related to Immunomodulatory Effects | Genetic Manipulations (Transformation Method) | Refs |
|---|---|---|---|---|---|---|
| Agrocybe aegerita | 44.7908 | 14110 | 49.2 | Polyethylene glycol–mediated transformation (PEG) | [169] | |
| Agaricus bisporus | 30.78 | 10863 | 46.5 | PEG, Electroporation, Particle bombardment, Agrobacterium tumefaciens-mediated transformation (ATMT) | [170,171,172,173] | |
| Flammulina velutipes | 35.64 | 49.76 | Fip-fve | PEG, Electroporation, Electro-injection, Restriction enzyme-mediated integration (REMI), ATMT | [173,174,175,176,177] | |
| Ganoderma atrum | fip-gat | [178] | ||||
| Ganoderma lucidum | 43.68 | 55.4 | fip-glu, Mevalonate (MVA) pathway genes: AACT (acetyl-CoA acetyltransferase); HMGS (3-hydroxy-3-methylglutaryl-CoA synthase), HMGR (3-hydroxy-3-methylglutaryl-CoA reductase), MVK (mevalonate kinase), MPK (phosphomevalonate kinase), MVD (pyrophosphomevalonate decarboxylase), IDI (isopentenyl-diphosphate isomerase), GPPs (geranyl diphosphate synthase), FPPs (farnesyl diphosphate synthase), SQS (squalene synthase), SE (squalene monooxygenase), OSC (2,3-oxidosqualene-lanosterol cyclase), P450 (cytochrome P450), UGTs (uridine diphosphate glycosyltransferases) | PEG, Electroporation, REMI | [173,179,180,181] | |
| Ganoderma sinensis | 48.96 | 15478 | 55.6 | fip-gsi | [103,182] | |
| Ganoderma tsugae | 45.5 | fip-gts | [183] | |||
| Hericium erinaceus (syn. Hericium erinaceum) | 41.21 | 52.43 | ATMT | [184,185] | ||
| Lentinula edodes | 39.92 | 12051 | 46 | PEG, Electro-injection, REMI, ATMT | [173,186] | |
| Pleurotus ostreatus | 34.36 | 12296 | 50.76 | PEG, Electroporation, REMI, Particle bombardment | [173,187,188,189] | |
| Postia placenta | 66.6724 | 12716 | 47.2 | fip-ppl | [108,190] | |
| Trametes versicolor | 44.794 | 14572 | 57.3 | fip-tvc | [191,192] | |
| Volvariella volvacea | 35.72 | 48.8 | fip-vvo | PEG, Particle bombardment, ATMT | [193,194] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, S.; Gao, Q.; Rong, C.; Wang, S.; Zhao, Z.; Liu, Y.; Xu, J. Immunomodulatory Effects of Edible and Medicinal Mushrooms and Their Bioactive Immunoregulatory Products. J. Fungi 2020, 6, 269. https://doi.org/10.3390/jof6040269
Zhao S, Gao Q, Rong C, Wang S, Zhao Z, Liu Y, Xu J. Immunomodulatory Effects of Edible and Medicinal Mushrooms and Their Bioactive Immunoregulatory Products. Journal of Fungi. 2020; 6(4):269. https://doi.org/10.3390/jof6040269
Chicago/Turabian StyleZhao, Shuang, Qi Gao, Chengbo Rong, Shouxian Wang, Zhekun Zhao, Yu Liu, and Jianping Xu. 2020. "Immunomodulatory Effects of Edible and Medicinal Mushrooms and Their Bioactive Immunoregulatory Products" Journal of Fungi 6, no. 4: 269. https://doi.org/10.3390/jof6040269
APA StyleZhao, S., Gao, Q., Rong, C., Wang, S., Zhao, Z., Liu, Y., & Xu, J. (2020). Immunomodulatory Effects of Edible and Medicinal Mushrooms and Their Bioactive Immunoregulatory Products. Journal of Fungi, 6(4), 269. https://doi.org/10.3390/jof6040269






